Summary
Background
Effective systemic therapies for patients with progressive neuroendocrine tumours of lung or gastrointestinal tract are limited. We aimed to assess the efficacy and safety of everolimus in this patient population.
Methods
In RADIANT-4, a randomised, double-blind, placebo-controlled, phase 3 study, patients with advanced, progressive, well-differentiated, nonfunctional lung or gastrointestinal neuroendocrine tumours were enrolled from 97 centres in 25 countries worldwide. Eligible patients were randomised in a 2:1 ratio to everolimus 10 mg/day or placebo, both with supportive care. Patients were stratified by tumour origin, performance status, and prior somatostatin analogue treatment. The primary endpoint was progression-free survival assessed by central radiology review. Overall survival was a key secondary endpoint. This trial is registered with ClinicalTrials.gov, number NCT01524783.
Findings
From April 2012 to August 2013, a total of 302 patients were enrolled, of whom, 205 were allocated to everolimus 10 mg/day and 97 to placebo. Median progression-free survival was 11.0 months (95% confidence interval [CI], 9.2–13.3) in the everolimus arm and 3.9 months (95% CI, 3.6–7.4) in the placebo arm. Everolimus was associated with a 52% reduction in the estimated risk of progression or death (hazard ratio [HR], 0.48; 95% CI, 0.35–0.67; p<0.00001). Although statistically not significant, a trend towards improved survival was observed in the first pre-planned interim overall survival analysis (HR, 0.64; 95% CI: 0.40, 1.05; one-sided p=0.037; boundary for statistical significance, 0.0002). Grade 3 or 4 drug-related adverse events (everolimus vs placebo) were relatively infrequent and included stomatitis (9% vs 0), diarrhoea (7% vs 2%), infections (7% vs 0), anaemia (4% vs 1%), fatigue (4% vs 1%), and hyperglycaemia (4% vs 0).
Interpretation
Treatment with everolimus was associated with significant improvement in progression-free survival in patients with progressive lung or gastrointestinal neuroendocrine tumours. The safety findings were consistent with the known side effect profile of everolimus.
Introduction
Neuroendocrine tumours (NET) are a group of heterogeneous malignancies arising from neuroendocrine cells throughout the body.1 Data from population-based registries indicate that 51% of NET arise from the gastrointestinal (GI) tract, 27% from lung, and 6% from pancreas.1 Clinically, NET are considered functional, if associated with symptoms of hormonal hypersecretion, or nonfunctional. Although carcinoid syndrome is classically associated with metastatic, well-differentiated NET of the small intestine, an analysis of National Comprehensive Cancer Centre database showed that the majority of NET (74%) are nonfunctional.2 Prognosis of NET varies based on the primary site, presence of metastatic disease, tumour grade, and stage at diagnosis.3
Advanced NET are incurable in nearly all cases. The somatostatin analogue (SSA), octreotide, approved for control of hormonal syndrome, has been shown to delay disease progression in patients with treatment-naïve midgut NET.4 Recently, lanreotide demonstrated a delay in tumour growth among patients with mostly stable, advanced, enteropancreatic NET.5 Although targeted therapies such as everolimus and sunitinib are approved for advanced pancreatic NET, where both drugs have been associated with improved progression-free survival (PFS),6–8 these agents are not approved for advanced lung NET or progressive GI NET.
Everolimus (Afinitor, Novartis Pharmaceuticals Corporation), a potent, oral inhibitor of mammalian target of rapamycin (mTOR), was previously shown to be associated with antitumour activity in advanced non-pancreatic NET.9–11 The RADIANT-2 study evaluated everolimus or placebo, both with octreotide long-acting repeatable (LAR), in patients with NET and carcinoid syndrome. In RADIANT-2 study, treatment with everolimus plus octreotide LAR was associated with a 5.1-month improvement in median PFS for the everolimus arm over the placebo arm.12 The difference, however, did not achieve statistical significance, possibly a result of imbalance in baseline characteristics between treatment arms and informative censoring due to discordance between local and central radiology review.12 The RAD001 in Advanced Neuroendocrine Tumours, Fourth Trial (RADIANT-4) was conducted to determine whether oral everolimus at a daily dose of 10 mg compared with placebo prolongs PFS in patients with advanced, nonfunctional, progressive NET of lung or GI origin.
Methods
Patients
Adult patients (age, ≥18 years) with pathologically confirmed, advanced (unresectable or metastatic), nonfunctional, well-differentiated (grade 1 or 2 according to the 2010 World Health Organization [WHO] classification13,14) NET of lung or GI origin were eligible for participation within 6 months from documented radiologic disease progression. Additional key inclusion criteria included measurable disease according to modified Response Evaluation Criteria In Solid Tumors version 1.0 (RECIST; webappendix pp 2–3 [amended protocol pp 76–78]);15 a WHO performance status score of 0 or 1; and adequate bone marrow, liver, and kidney function. Patients previously treated with SSA, interferon, one prior line of chemotherapy, and/or peptide receptor radionuclide therapies (PRRT) were allowed if disease progression was documented during or after last treatment. Antineoplastic therapy must have been discontinued for at least 4 weeks (6 months, if PRRT) before randomisation. Patients were ineligible if they had a history of or presented with carcinoid syndrome, poorly differentiated histology, or pancreatic NET. Patients who received more than one line of chemotherapy; prior therapy with mTOR inhibitors (sirolimus, temsirolimus, or everolimus); hepatic intra-arterial embolization within 6 months, or cryoablation or radiofrequency ablation of hepatic metastases within 2 months of randomisation; or chronic treatment with corticosteroids or other immunosuppressive agents were excluded.
The study was conducted in accordance with Good Clinical Practice, ethical principles of the Declaration of Helsinki, and local regulations. Independent Ethics Committee or Institutional Review Board at each participating centre reviewed and approved the study and all the amendments. All patients provided written informed consent. An independent Data Monitoring Committee provided ongoing oversight of safety and study conduct.
Randomisation and masking
Randomisation was by interactive voice response systems (webappendix amended protocol pp 59–61]). Patients, investigators, and sponsor were blinded to treatment assignment. Randomisation was stratified by prior SSA treatment (defined as continuous SSA for ≥12 weeks), tumour origin (based on prognostic level, grouped into two strata: stratum A [better prognosis], appendix, cecum, jejunum, ileum, duodenum, or NET of unknown primary; stratum B [worse prognosis], lung, stomach, colon [other than cecum] or rectum), and WHO performance status (0 vs 1). Premature unblinding was only allowed in case of emergency. The identity of experimental treatments was concealed by use of everolimus and placebo that were identical in packaging, labelling, appearance, and administration schedule.
Procedures
In RADIANT-4, an international, multicentre, double-blind, phase 3 study, patients were randomly assigned in a 2:1 ratio to receive oral everolimus at a dose of 10 mg/day or placebo, both with best supportive care. Concomitant SSAs during study were allowed only for control of emergent carcinoid symptoms (eg, flushing, diarrhoea) not manageable by standard therapy such as loperamide.
All patients who underwent randomisation were assessed for efficacy by cross-sectional imaging with multiphasic computed tomography or magnetic resonance imaging every 8 weeks during the first 12 months and every 12 weeks thereafter.
Dose reductions and treatment interruption for a maximum of 28 days were permitted for patients who did not tolerate therapy or to manage adverse events (AEs) deemed to be related to study treatment. Two dose reductions were allowed: 5 mg/day and subsequently, 5 mg every other day. Treatment continued until documented radiologic disease progression, start of new cancer therapy, development of an intolerable AE, or withdrawal of consent. Crossover to open-label everolimus after progression was not allowed and patients and investigator remained blinded until primary analysis.
Outcomes
The primary endpoint was central radiology-assessed PFS, defined as the time from randomisation to death or progression as per modified RECIST v1.0.14 Central radiology review, blinded to treatment assignment and local assessment, was conducted in real time (webappendix pp 3). PFS per investigator assessment was a supportive analysis. Overall survival (OS) was the key secondary endpoint. Other secondary endpoints included objective response rate, disease control rate (webappendix pp 4), health-related quality of life, WHO performance status, pharmacokinetics, changes in chromogranin A and neuron-specific enolase levels, and safety.
All randomly assigned patients were included in full analysis set. Primary efficacy analyses were evaluated based on the data from this population on an intent-to-treat basis.
The safety set included all patients who received at least one dose of the study drug with at least one postbaseline safety evaluation. AEs were assessed as per National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) v4.03 (http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf).
Statistical analysis
Based on the historical data, we assumed the median PFS in the control arm to be approximately 5 months.4 Sample size was estimated based on the ability to detect a clinically meaningful improvement in PFS, defined as a 41% reduction in the risk of disease progression or death (hazard ratio [HR], 0.59), corresponding to a prolongation in median PFS from 5 months with placebo to 8.5 months with everolimus. With a 2:1 randomisation and one-sided type 1 error of 2.5%, a total of 176 PFS events are needed for 91.3% power. Adjusting for estimated dropout rate of 15%, approximately 285 patients were required to be randomised in a 2:1 ratio between the everolimus and placebo arms.
OS analyses were to be performed if the primary endpoint was statistically significant using a group sequential design with two interim analyses and final analysis at approximately 191 events (one-sided significance level, 2.5%; webappendix pp 4). Lan-DeMets method with O’Brien-Fleming type stopping boundary was used to control the cumulative type-I error rate.
PFS and OS were estimated using the Kaplan–Meier method; comparison between the treatment arms was performed using a one-sided log-rank test, stratified according to the tumour origin, WHO performance status, and prior SSA treatment. The HR was estimated by a stratified Cox proportional hazards model. The protocol, including the statistical analysis plan, is available in the webappendix). This study is registered at ClinicalTrials.gov, number NCT01524783.
Role of the funding source
The study was designed by academic investigators and representatives of the sponsor. Data were collected electronically using data management systems of a contract research organisation designated by sponsor and were analysed by the sponsor’s statistical team. All authors contributed to the interpretation of data and subsequent writing, reviewing, and amending of manuscript; the first draft of manuscript was prepared by the first author and a medical writer employed by sponsor. All authors vouch for the accuracy and completeness of the data and attest that the study conformed to protocol and statistical analysis plan.
Results
Between April 03, 2012 and August 23, 2013, a total of 302 patients with advanced, nonfunctional NET of lung or GI origin were enrolled from 97 centres in 25 countries worldwide (Austria, Belgium, Canada, China, Colombia, Czech Republic, Germany, Greece, Hungary, Italy, Japan, Lebanon, Netherlands, Poland, Republic of Korea, Russia, Saudi Arabia, Slovak Republic, South Africa, Spain, Taiwan, Thailand, Turkey, United Kingdom, and the USA). Patients were randomly assigned to everolimus 10 mg/day (205 patients) or placebo (97 patients; figure 1). The baseline characteristics of patients in both the arms were generally well balanced (table 1).
Figure 1. Trial profile.
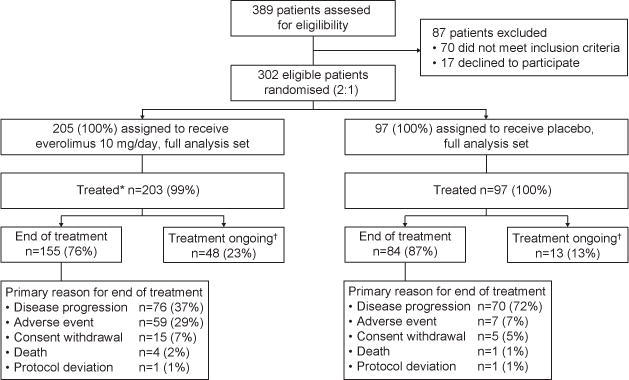
*The full analysis set contained all 302 randomized patients (205 patients in the everolimus arm and 97 in the placebo arm). Two patients randomized to everolimus were not treated and one patient randomized to everolimus received only placebo treatment, therefore the safety set contains 202 patients in the everolimus arm and 98 patients in the placebo arm.
†At the time of data cut-off (November 28, 2014).
Table 1.
Baseline Demographics and Disease Characteristics (Full Analysis Set).
| Characteristic | Everolimus N = 205 |
Placebo N = 97 |
|---|---|---|
| Median age, years (range) | 65 (22–86) | 60 (24–83) |
| Sex | ||
| Male | 89 (43%) | 53 (55%) |
| Female | 116 (57%) | 44 (45%) |
| WHO performance status* | ||
| 0 | 149 (73%) | 73 (75%) |
| 1 | 55 (27%) | 24 (25%) |
| Primary tumour site | ||
| Lung | 63 (31%) | 27 (28%) |
| Ileum | 47 (23%) | 24 (25%) |
| Rectum | 25 (12%) | 15 (16%) |
| NET of unknown primary site† | 23 (11%) | 13 (13%) |
| Jejunum | 16 (8%) | 6 (6%) |
| Stomach | 7 (3%) | 4 (4%) |
| Duodenum | 8 (4%) | 2 (2%) |
| Colon | 5 (2%) | 3 (3%) |
| Other‡ | 6 (3%) | 2 (2%) |
| Caecum | 4 (2%) | 1 (1%) |
| Appendix | 1 (1%) | 0 |
| Tumour grade¶ | ||
| Grade 1 | 129 (63%) | 65 (67%) |
| Grade 2 | 75 (37%) | 32 (33%) |
| Time from initial diagnosis to randomisation | ||
| ≤6 months | 26 (13%) | 12 (12%) |
| >6 months to ≤18 months | 51 (25%) | 25 (26%) |
| >18 months to ≤36 months | 41 (20%) | 22 (23%) |
| >36 months | 87 (42%) | 38 (39%) |
| Prior treatments§ | ||
| Surgery | 121 (59%) | 70 (72%) |
| Chemotherapy | 54 (26%) | 23 (24%) |
| Radiotherapy including PRRT | 44 (22%) | 19 (20%) |
| Locoregional and ablative therapies‖ | 23 (11%) | 10 (10%) |
| Somatostatin analogues | 109 (53%) | 54 (56%) |
| Disease sites** | ||
| Liver | 163 (80%) | 76 (78%) |
| Lymph node or lymphatic system | 85 (42%) | 45 (46%) |
| Lung | 45 (22%) | 20 (21%) |
| Bone | 42 (21%) | 15 (16%) |
| Peritoneum | 25 (12%) | 8 (8%) |
| Liver tumour burden | ||
| None | 34 (17%) | 14 (14%) |
| ≤10% | 119 (58%) | 61 (63%) |
| >10% to 25% | 29 (14%) | 8 (8%) |
| >25% | 21 (10%) | 14 (14%) |
| Unknown | 2 (1%) | 0 |
Data are n (%) unless otherwise stated.
One patient in the everolimus arm had WHO performance status of 2.
Patients with well-differentiated (grade 1 or 2) NET where primary tumour origin other than lung or GI tract were excluded by appropriate diagnostic procedures.
All patients, except one in the everolimus arm who had thymus as primary site, had primary tumour origin from GI.
Patients with WHO grade 1 or well-differentiated NET were classified as grade 1 and those with WHO grade 2 or moderately-differentiated NET as grade 2.13,14 Tumour grade was not available for one patient in the everolimus arm.
Few patients (≤5%) received other prior medications including immunotherapy, targeted therapy, and hormonal therapy other than SSA.
Included in this category are transarterial embolization, cryoablation, and radiofrequency ablation.
The sites as per target and nontarget lesion locations observed at baseline by central radiology review.
GI=gastrointestinal. NET=neuroendocrine tumours. PRRT=peptide receptor radionuclide therapy. SC=subcutaneous. SSA=somatostatin analogue. WHO=World Health Organization.
The most frequent sites of origin were the lung, ileum, and rectum. Median time from initial diagnosis to randomisation was 29.9 months (range, 0.7–258.4) in everolimus arm and 28.9 months (1.1–303.3) in the placebo arm. More than half of the population had a history of previous treatment with SSA therapy (mostly for tumour control). One-quarter of the patients had received chemotherapy. Both the arms were also comparable with respect to prior radiotherapy including PRRT and locoregional therapies, including transarterial embolization, cryoablation or radiofrequency ablation.
At the time of data analysis cutoff (November 28, 2014), 155 patients (76%) in the everolimus arm and 84 patients (87%) in the placebo arm had discontinued study treatment (figure 1). Common reasons for treatment discontinuation included disease progression, AEs, and withdrawal of consent.
Median PFS assessed by central review was 11.0 months (95% CI, 9.2–13.3) in the everolimus arm and 3.9 months (95% CI, 3.6–7.4) in the placebo arm. Everolimus was associated with a 52% reduction in the estimated risk of disease progression or death (HR, 0.48; 95% CI, 0.35–0.67; p<0.00001) (table 2 and figure 2A).
Table 2.
Progression-free Survival (Full Analysis Set).
| Variable | Everolimus N=205 | Placebo N=97 | Difference | Hazard ratio* for disease progression or death with everolimus (95% CI) | p-value† |
|---|---|---|---|---|---|
| Central radiology review | |||||
| PFS events‡ | 113 (55%) | 65 (67%) | |||
| Number censored | 92 (45%) | 32 (33%) | |||
| Median PFS, months | 11·0 | 3·9 | 7·1 | 0·48 (0·35–0·67) | <0·00001 |
| Local radiology review | |||||
| PFS events‡ | 98 (48%) | 70 (72%) | |||
| Number censored | 107 (52%) | 27 (28%) | |||
| Median PFS, months | 14·0 | 5·5 | 8·5 | 0·39 (0·28–0·54) | <0·00001 |
Hazard ratio was obtained from the stratified Cox model.
p-value was obtained from the one-sided stratified log-rank test.
Progression-free survival events include disease progression and death.
CI=confidence interval. PFS=progression-free survival.
Figure 2. Progression-free and overall survival.
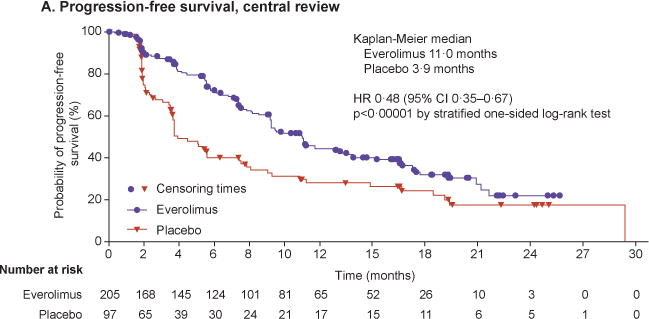
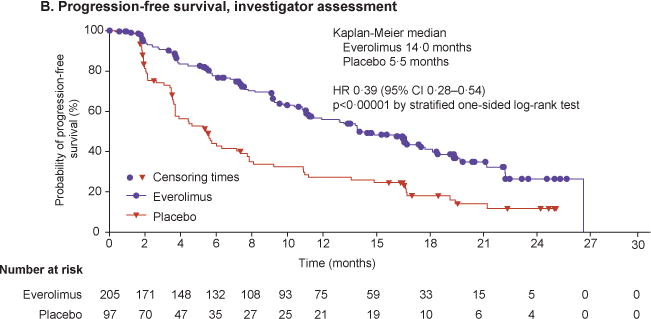
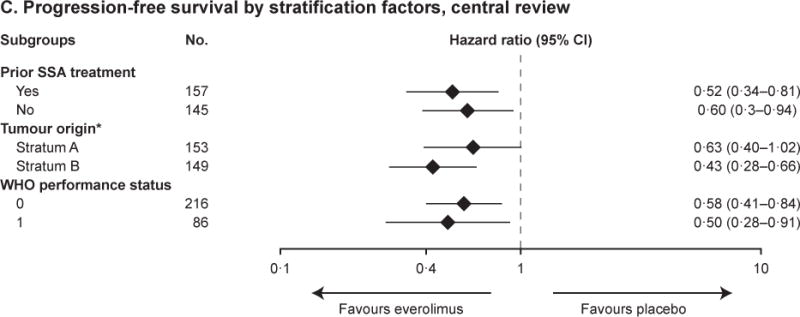
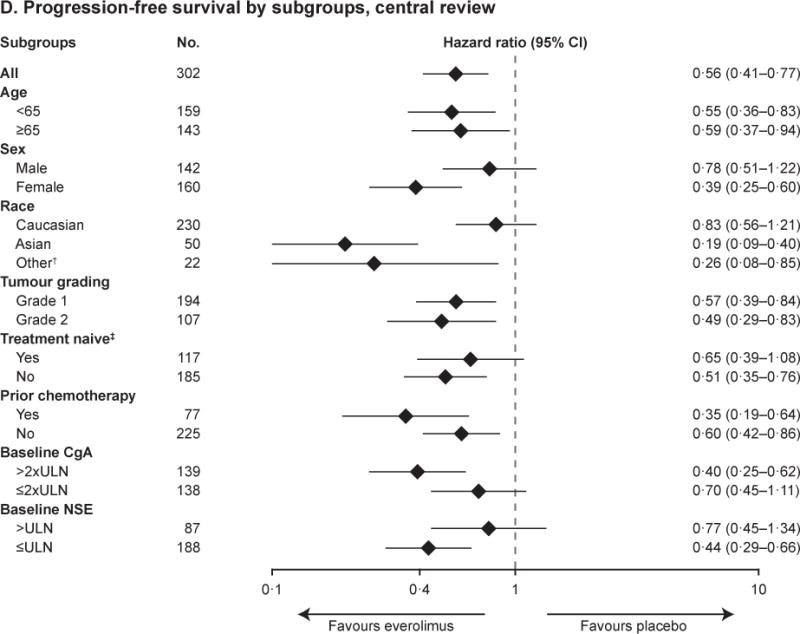
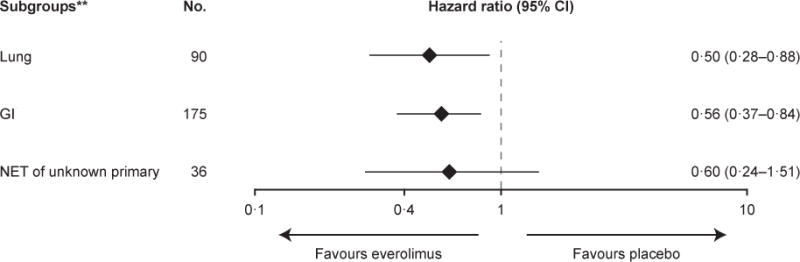
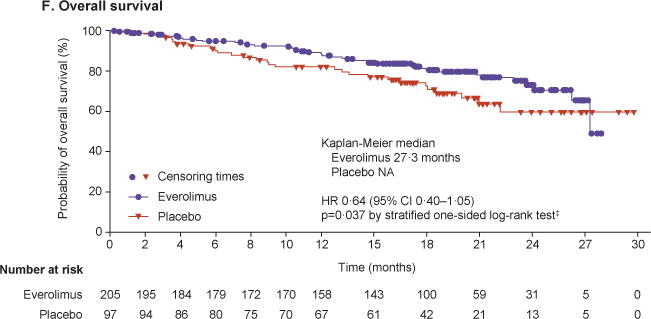
Kaplan–Meier curves are shown for progression-free survival as assessed by central radiology review (Panel A) and by local investigators (Panel B). Forest plots show the effect of study treatment on progression-free survival in predefined patient subgroups based on stratification factors (Panel C), major demographic and prognostic subgroups (Panel D) and a retrospective analysis in subgroups by primary tumour origin (Panel E). Subgroup results are reported based on central review. In the retrospective post hoc analysis, stomach, colon, rectum, appendix, caecum, ileum, duodenum, and jejunum are grouped under GI. The HRs in all subgroups are obtained from unstratified Cox model. Kaplan–Meier curves are also shown for overall survival (Panel F).
*Based on prognostic level, patients were divided into two strata. Stratum A (better prognosis) consisted of patients with tumour sites originating from appendix, cecum, jejunum, ileum, duodenum, and NET of unknown primary and stratum B (worse prognosis) comprised patients with primary tumours of lung, stomach, rectum, and colon (except the cecum). †Included Black. ‡Defined as no prior chemotherapy or no SSA therapy continuously for ≥12 weeks any time before study. **One patient with thymus as primary tumour origin was not included. ¶p-value boundary for significance at this interim analysis was 0.0002.
CI=confidence interval. CgA=chromogranin A. GI=gastrointestinal. NA=not available. NET=neuroendocrine tumours. NSE=neuron-specific enolase. SSA=somatostatin analogue. ULN=upper limit of normal. WHO=World Health Organization.
The estimated progression free survival rate at 12 months (per central review) was 44% in the everolimus arm and 28% in the placebo arm, indicating durable benefit with everolimus.
Findings by investigator assessment were consistent with central review. Median PFS was 14.0 months (95% CI, 11.2–17.7) with everolimus and 5.5 months (95% CI, 3.7–7.4) with placebo (HR, 0.39; 95% CI, 0.28–0.54; p<0.00001) (figure 2B). Consistent treatment benefits with everolimus were observed irrespective of the stratification factors (figure 2C).
Additional prespecified subgroup analyses of PFS per central radiologic assessment demonstrated a consistent positive treatment effect across major demographic and prognostic subgroups (figure 2D).
A retrospective analysis showed consistent beneficial effect across the subgroups based on primary tumour origin (lung, GI, or NET of unknown primary; figure 2E). A positive treatment effect regardless of the extent of liver metastasis was also seen (webappendix, figure S1, Supplementary Appendix).
A planned interim OS analysis was performed with a total of 70 deaths. The survival results from the first interim OS analysis favoured everolimus with 36% reduction in the estimated risk relative to placebo, although statistical significance was not attained (HR, 0.64; 95% CI, 0.40–1.05; p=0.037; Lan-DeMets O’brian Fleming boundary for significance at first interim analysis, 0.0002) (figure 2E). Data was not mature enough to provide accurate estimates of median OS. Kaplan–Meier estimates for overall survival at 25th percentile (25% of patients having survival events) were 23.7 months (95% CI, 17.6–27.3) in the everolimus arm and 16.5 months (95% CI, 9.0–21.0) in the placebo arm.
Confirmed objective responses (by central radiology review; all partial responses) were observed in four patients receiving everolimus (2%) and one patient receiving placebo (1%). Disease stabilisation was the best overall response in 165 patients (81%) in the everolimus arm vs 62 patients (64%) in the placebo arm. Thus, the prolongation in progression-free survival with everolimus was likely secondary to the stabilization of disease or minor tumour shrinkage and to fewer cases of progressive disease. Everolimus was associated with a higher disease control rate as compared with placebo (webappendix, table S1). Of patients that could be assessed for tumour shrinkage, 117 (64%) in the everolimus arm and 22 (26%) in the placebo arm experienced some degree of tumour shrinkage (figure 3).
Figure 3. Percentage change from baseline in size of target lesion, central review (full analysis set).
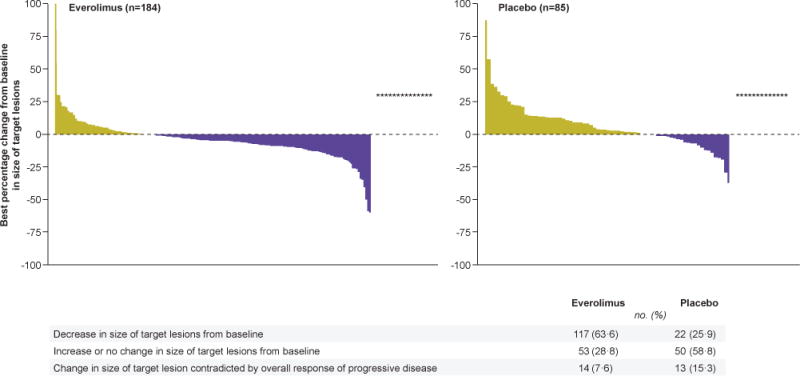
The plot shows the best percentage change from baseline in the size of the target lesion (ie, the best response in each patient) in the everolimus arm (left) and placebo arm (right). Fourteen patients (7.6%) in the everolimus arm and 13 patients (15.3%) in the placebo arm showed a change in the available target lesion that contradicted the overall response of progressive disease (marked as * in the graph). Patients for whom the best percentage change in target lesion was not available (21 patients receiving everolimus and 12 patients receiving placebo) or was available but contradicted by overall lesion response of unknown (none), were excluded from the analysis. Percentage above used N as denominator.
Findings from other secondary endpoints will be presented in future publications.
Safety
With a median follow-up period of 21 months, median duration of treatment was nearly double for the everolimus arm than the placebo arm (40.4 weeks [range, 0.7–120.4] vs 19.6 weeks [range, 4.0–130.3]). This difference in exposure should always be considered when comparing various rates of reported AEs that are not adjusted for treatment duration. The median relative dose intensity (the ratio of administered doses to planned doses) was 0.9 in the everolimus arm and 1.0 in the placebo arm. Without adjustment for duration of treatment, dose reductions or temporary treatment interruptions occurred in 135 patients (67%) receiving everolimus and 29 (30%) receiving placebo.
AEs were consistent with the known safety profile of everolimus and were mostly grade 1 or 2. Rates of on-treatment deaths (those occurring during the receipt of study medication or within 30 days of discontinuing therapy) were similar between the treatment arms (seven [3.5%] in the everolimus arm and three [3.1%] in the placebo arm). All except three deaths (1.5%) in the everolimus arm (one case each due to respiratory failure, septic shock, and cardiac failure) and two deaths (2.0%) in the placebo arm (one case each due to lung infection and dyspnoea) were attributed to disease progression.
Treatment-related AEs that occurred in at least 10% of patients are listed in table 3; the most common were stomatitis, diarrhoea, fatigue, infections, rash, and peripheral oedema. The most frequent grade 3 or 4 drug-related AEs included stomatitis, diarrhoea, infections, anaemia, and fatigue. Treatment discontinuation attributed to grade 3 or 4 AEs related to the study drug were reported in 24 patients (12%) receiving everolimus and three patients (3%) receiving placebo.
Table 3.
Treatment-Related Adverse Events Reported in at Least 10% of Patients (Safety Set).
| Adverse event | Everolimus N=202 |
Placebo N=98 |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| All grades | Grade 1 | Grade 2 | Grade 3 | Grade 4 | All grades | Grade 1 | Grade 2 | Grade 3 | Grade 4 | |
| Stomatitis* | 127 (63) | 72 (36) | 37 (18) | 18 (9) | 0 | 19 (19) | 17 (17) | 2 (2) | 0 | 0 |
| Diarrhoea | 63 (31) | 30 (15) | 18 (9) | 13 (6) | 2 (1) | 16 (16) | 10 (10) | 4 (4) | 2 (2) | 0 |
| Fatigue | 62 (31) | 35 (17) | 20 (10) | 5 (2) | 2 (1) | 24 (24) | 17 (17) | 6 (6) | 1 (1) | 0 |
| Infections† | 59 (29) | 12 (6) | 33 (16) | 10 (5) | 4 (2) | 4 (4) | 1 (1) | 3 (3) | 0 | 0 |
| Rash | 55 (27) | 42 (21) | 12 (6) | 1 (<1) | 0 | 8 (8) | 6 (6) | 2 (2) | 0 | 0 |
| Peripheral oedema | 52 (26) | 30 (15) | 18 (9) | 4 (2) | 0 | 4 (4) | 2 (2) | 1 (1) | 1 (1) | 0 |
| Nausea | 35 (17) | 26 (13) | 6 (3) | 2 (1) | 1 (<1) | 10 (10) | 7 (7) | 3 (3) | 0 | 0 |
| Asthenia | 33 (16) | 8 (4) | 22 (11) | 2 (1) | 1 (<1) | 5 (5) | 4 (4) | 1 (1) | 0 | 0 |
| Anaemia | 33 (16) | 5 (2) | 20 (10) | 8 (4) | 0 | 2 (2) | 0 | 1 (1) | 1 (1) | 0 |
| Decreased appetite | 32 (16) | 22 (11) | 9 (4) | 1 (<1) | 0 | 6 (6) | 2 (2) | 4 (4) | 0 | 0 |
| Noninfectious pneumonitis‡ | 32 (16) | 5 (2) | 24 (12) | 3 (1) | 0 | 1 (1) | 0 | 1 (1) | 0 | 0 |
| Dysgeusia | 30 (15) | 26 (13) | 3 (1) | 1 (<1) | 0 | 4 (4) | 4 (4) | 0 | 0 | 0 |
| Pruritus | 26 (13) | 19 (9) | 6 (3) | 1 (<1) | 0 | 4 (4) | 4 (4) | 0 | 0 | 0 |
| Cough | 26 (13) | 18 (9) | 8 (4) | 0 | 0 | 3 (3) | 3 (3) | 0 | 0 | 0 |
| Pyrexia | 22 (11) | 14 (7) | 4 (2) | 2 (1) | 2 (1) | 5 (5) | 4 (4) | 1 (1) | 0 | 0 |
| Hyperglycaemia | 21 (10) | 5 (2) | 9 (4) | 7 (3) | 0 | 2 (2) | 2 (2) | 0 | 0 | 0 |
| Dyspnoea | 21 (10) | 4 (2) | 15 (7) | 2 (1) | 0 | 4 (4) | 2 (2) | 1 (1) | 0 | 1 (1) |
Included in this category are stomatitis, aphthous stomatitis, mouth ulceration, and tongue ulceration.
All types of infections are included.
Included in this category are pneumonitis, interstitial lung disease, lung infiltration, and pulmonary fibrosis.
Noninfectious pneumonitis was observed for 32 patients (16%) in association with everolimus therapy. Most cases were of grade 1 or 2, grade 3 pneumonitis occurred in three patients (1%); no case of grade 4 was reported.
Discussion
In this randomised study of patients with advanced, progressive, nonfunctional NET of lung or GI origin, treatment with everolimus 10 mg/day significantly prolonged median PFS by 7.1 months compared with placebo (11.0 vs 3.9 months) by blinded central radiology review. This 2.8 fold improvement in median PFS corresponds with a reduction in risk of disease progression or death by 52% vs placebo (HR, 0.48; p<0.00001). This benefit was confirmed by the investigator-assessed PFS analysis. Subgroup analyses suggested a consistent treatment benefit across major subgroups.
The availability of targeted therapies has changed the treatment paradigm for patients with advanced NET. SSAs have now been established as a standard of care for effective treatment of carcinoid syndrome in functional NET. More recently SSAs have also been shown to control tumour growth in patients with advanced NET.4,5 Targeted therapies, such as everolimus and sunitinib, are approved in advanced, progressive, pancreatic NET.6–8 Effective antineoplastic therapy options for patients with advanced, progressive, nonfunctional NET of lung or GI tract, however, are limited.
The mTOR pathway is a central regulator of cellular proliferation, metabolism, protein synthesis, and autophagy. Although mTOR pathway mutations have been described in approximately 15% of pancreatic NET, somatic mutations in the mTOR pathway appear to be infrequent in NET of lung or GI origin.16–18 The activity of mTOR inhibitors in this setting is likely due to a combination of factors including inhibition of growth factor signalling, metabolic signalling, epigenetic regulation, and perhaps undefined genomic variations, which converge to activate mTOR pathway. Previously, using pair biopsy specimens, we demonstrated that everolimus therapy in patients with a variety of NET leads to significantly decreased S6 phosphorylation and consistent decrease in proliferation of tumour indicated by decreases in Ki-67 labeling.19,20 The findings from RADIANT-4 study validate the role of mTOR pathway in NET of lung or GI origin.
In the RADIANT-4 study, we evaluated everolimus as a monotherapy against placebo in nonfunctional NET including those arising from lung or GI tract, and demonstrated durable benefit in delaying tumour growth. Prospective stratification based on known prognostic factors in our study minimised confounding. Further, unlike RADIANT-2, crossover from placebo arm to everolimus arm after progression was not allowed during the blinded period in our study, which would avoid potential bias in the estimation of treatment effect on survival. Indeed, an interim OS analysis from RADIANT-4 showed a trend in survival benefit in favour of everolimus. Long-term OS results are awaited.
The safety and tolerability of everolimus in our study are consistent with the previous experience in advanced NET setting.6,12 The most frequent AEs observed with everolimus were of grade 1 or 2, and included stomatitis, diarrhoea, fatigue, infections, rash, and peripheral oedema; the frequencies were similar with those reported previously. Most everolimus-related AEs were manageable through dose modification or interruption without altering the duration of treatment.
In summary, everolimus, as compared with placebo, was associated with statistically significant and clinically meaningful prolongation of PFS in patients with advanced, progressive, nonfunctional lung or GI NET. The first interim OS analysis showed a trend towards improved survival in favour of everolimus, though it did not achieve threshold for statistical significance for this planned interim analysis. Up-to two additional OS analyses will be conducted according to the statistical plan as the survival follow-up matures. Everolimus was well tolerated and the safety findings were consistent with previous experience. Taken together with results from the prior RADIANT-3 study in pancreatic NET,6 everolimus has now been shown to have robust antitumour activity across a broad spectrum of NET including those arising from pancreas, lung, and GI tract.
Supplementary Material
Panel: Research in context.
Evidence before this study
We searched Medline for reports on clinical trials in advanced NET, with “mTOR” and “NET” as our primary search terms limiting the findings to include “nonfunctional”, “non-pancreatic” or “non-syndromic” NET. We did not limit our search by date. We identified no studies of mTOR inhibitors as monotherapy in the present population. We found that phase 3 RADIANT-2 study evaluated mTOR inhibitor everolimus in combination with octreotide LAR versus placebo plus octreotide LAR in patients with advanced NET and history of carcinoid symptoms (Pavel ME et al [2011]). Although the RADIANT-2 study did not meet the primary endpoint, the results provided an initial indication of potential antitumor effect of everolimus in the patients with nonpancreatic NET. Additionally, RAMSETE (RAD001 [everolimus] in Advanced and Metastatic Silent neuroEndocrine Tumours in Europe) a single-arm, multicenter, single-stage phase 2 trial showed that everolimus may be effective in nonsyndromic, nonpancreatic NET with diverse tumour origin sites (Pavel ME et al [2012]). We identified no phase 3 studies with mTOR inhibitors as monotherapy in the present study population.
Added value of this study
Advanced NET are incurable. Targeted therapies, such as everolimus and sunitinib, are approved in advanced, progressive, pancreatic NET. Effective antineoplastic therapy options for patients with advanced, progressive, nonfunctional NET of lung or GI tract are very limited. To our knowledge, RADIANT-4 is the first, large, randomised, placebo-controlled, phase 3 study that evaluated the efficacy and safety of mTOR inhibitor everolimus as monotherapy in this patient population. Everolimus was associated with a clinically meaningful 2.8 fold prolongation of progression-free survival versus placebo, indicating a statistically significant 52% risk reduction in favour of everolimus. The benefit of treatment with everolimus was maintained across majority of the prespecified subgroups. The adverse event findings were consistent with the known safety profile of everolimus.
Implications of all the available evidence
Taken together with results from the prior RADIANT-3 study in pancreatic NET, (Yao JC et al [2011]) the findings from RADIANT-4 study establishes robust, practice-changing evidence supporting the antitumor efficacy of everolimus across a broad spectrum of NET including those arising from pancreas, lung, or GI tract.
Acknowledgments
The study was sponsored by Novartis Pharmaceuticals Corporation. We thank the patients and investigators for their participation in the study; the worldwide network of research nurses, trial coordinators, and operations staff for their contributions. Additionally, we thank Du Lam, MD for assistance with protocol development and study conduct; Valentine Jehl, MSc and Hélène Cauwel, MSc for inputs on statistical aspects during protocol development; and Bhavik Shah, PhD and Rohit Kachhadiya, MPharm (Novartis Healthcare Pvt Ltd) for providing medical editorial assistance with this manuscript.
Funding Novartis Pharmaceuticals Corporation
Footnotes
Contributors
JCY, NF, SS, CS, JS, MHK, and MEP contributed to the study concept and design. JCY, NF, SS, RB, CC, EMW, JT, MR, HL, JWV, GDF, EVC, MT, YS, DYO, JS, MHK, and MEP recruited patients. NR performed the statistical analysis. JCY wrote the first draft of manuscript with the help of a medical writer. All authors contributed to the manuscript content, provided inputs for data interpretation, reviewed and critically revised the content, and approved the final version for submission.
Trial investigators
Austria: M Raderer, G Pall; Belgium: E Van Cutsem, I Borbath, K Geboes, M Peeters; Canada: T Asmis, W Kocha, D Rayson, J Ruether, S Singh, L Sideris, H Kennecke; China: J Wang, L Shen, J Xu, J Qian, L Jia; Colombia: LF Maya; Czech Republic: B Melichar, E Sedlackova, J Tomasek; Germany: M Pavel, J Bojunga, P Malfertheiner, H Lahner, A Vogel, M Weber, D Hörsch; Greece: G Kaltsas; Hungary: Z Papai, M Toth; Italy: C Carnaghi, G Luppi, N Fazio, P Tomassetti, G Delle Fave, G Cartenì, R. Buzzoni, C Barone, A Berruti, D Giuffrida, G Tortora, F Di Costanzo, S Tafuto; Japan: T Ito, N Okita, I Komoto; Lebanon: J Kattan, A Shamseddine; Netherlands: M. Tesselaar; Poland: B Jarzab, M Ruchala; Republic of Korea: TW Kim, YS Park, D-Y Oh, M-A Lee, HJ Choi; Russia: L Vladimirova; Saudi Arabia: H Raef; Slovakia (Slovak Republic): T Salek; South Africa: P Ruff; Spain: J Capdevila, R Salazar, JJR. Zoilo; Taiwan: J-S. Chen, C-C Wu, Y-Y Chen, Y Chao, K-H Yeh; Thailand: V Sriuranpong, S Thongprasert; Turkey: H Turna, A Sevinc; United Kingdom: J Valle, D Sarker, N Reed, J Cave, A Frilling, P Corrie; United States of America: P Fanta, J Yao, J Strosberg, U Verma, S Libutti, R Natale, R Pommier, S Lubner, A Starodub, M Kulke, D Sigal, B Polite, C Lieu, K Hande, D Reidy-Lagunes, A McCollum, and L Forero.
Conflicts of interest
JCY has received consulting or advisory fees from Ipsen, Lexicon, and Novartis and research funding from Novartis. NF has received honoraria from Ipsen, and Novartis, consulting or advisory fees from Ipsen, Lexicon, Novartis, and Italfarmaco, research funding from Novartis, and travel and accommodations expenses from Ipsen and Novartis. SS has received honoraria, consulting or advisory fees, travel and accommodations expenses, and research funding from Novartis. RB has received research funding from Italfarmaco, Novartis, and Otsuka and travel and accommodations expenses from Ipsen; Italfarmaco; and Novartis. EMW has received consulting or advisory fees from Celgene, Ipsen, and Novartis. JT has received honoraria, research funding, and travel and accommodations expenses from Novartis. MR has received honoraria from Celgene, Ipsen, Novartis, and Roche and consulting or advisory fees from Celgene, Ipsen, Novartis, and Roche. HL has received honoraria from Ipsen, Novartis, and Pfizer, consulting or advisory fees from Novartis and Pfizer, research funding from Novartis and travel and accommodations expenses from Ipsen, Novartis, and Pfizer. MV, LBP, NR, and CS are employees of and own shares in Novartis. JWV has received honoraria, consulting or advisory fees and research funding from Novartis. GDF has received consulting or advisory fees and research funding from Novartis. EVC has received research funding from Novartis. YS has received research funding Chugai Pharma, Lilly, Novartis, and Taiho Pharmaceutical. JS has received honoraria from Novartis, consulting or advisory fees from Ipsen, Lexicon, and Novartis, research funding from Novartis and Pfizer, and is on the speaker’s bureau for Bayer and Genentech. MHK has received consulting or advisory fees from Ipsen and Novartis. MEP has received honoraria from Ipsen, Lexicon, Novartis, and Pfizer, consulting or advisory fees from Ipsen, Lexicon, Novartis, and Pfizer, research funding from Novartis, and travel and accommodations expenses from Ipsen and Novartis. CC, MT, and DYO declare no conflicts of interest.
References
- 1.Yao JC, Hassan M, Phan A, et al. One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008;26:3063–72. doi: 10.1200/JCO.2007.15.4377. [DOI] [PubMed] [Google Scholar]
- 2.Choti MA, Bobiak S, Strosberg JR, et al. Prevalence of functional tumors in neuroendocrine carcinoma: An analysis from the NCCN NET database. J Clin Oncol. 2012;30:4126. (abstr). [Google Scholar]
- 3.Yao J, Phan A. Optimising therapeutic options for patients with advanced pancreatic neuroendocrine tumours. Eur Oncol Haematol. 2012;8:217–23. [Google Scholar]
- 4.Rinke A, Muller HH, Schade-Brittinger C, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009;27:4656–63. doi: 10.1200/JCO.2009.22.8510. [DOI] [PubMed] [Google Scholar]
- 5.Caplin ME, Pavel M, Ruszniewski P. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014;371:224–33. doi: 10.1056/NEJMoa1316158. [DOI] [PubMed] [Google Scholar]
- 6.Yao JC, Shah MH, Ito T, et al. Everolimus for advanced pancreatic neuroendocrine tumors. N Engl J Med. 2011;364:514–23. doi: 10.1056/NEJMoa1009290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Raymond E, Dahan L, Raoul JL, et al. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N Engl J Med. 2011;364:501–13. doi: 10.1056/NEJMoa1003825. [DOI] [PubMed] [Google Scholar]
- 8.Blumenthal GM, Cortazar P, Zhang JJ, et al. FDA approval summary: sunitinib for the treatment of progressive well-differentiated locally advanced or metastatic pancreatic neuroendocrine tumors. Oncologist. 2012;17:1108–13. doi: 10.1634/theoncologist.2012-0044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fazio N, Granberg D, Grossman A, et al. Everolimus plus octreotide long-acting repeatable in patients with advanced lung neuroendocrine tumors: analysis of the phase 3, randomized, placebo-controlled RADIANT-2 study. Chest. 2013;143:955–62. doi: 10.1378/chest.12-1108. [DOI] [PubMed] [Google Scholar]
- 10.Pavel ME, Wiedenmann B, Capdevila J, et al. RAMSETE: a single-arm, multicenter, single-stage phase II trial of RAD001 (everolimus) in advanced and metastatic silent neuro-endocrine tumours in Europe. J Clin Oncol. 2012;30:4122. (abstr). [Google Scholar]
- 11.Bajetta E, Catena L, Fazio N, et al. Everolimus in combination with octreotide long-acting repeatable in a first-line setting for patients with neuroendocrine tumors: an ITMO group study. Cancer. 2014;120:2457–63. doi: 10.1002/cncr.28726. [DOI] [PubMed] [Google Scholar]
- 12.Pavel ME, Hainsworth JD, Baudin E, et al. Everolimus plus octreotide long-acting repeatable for the treatment of advanced neuroendocrine tumours associated with carcinoid syndrome (RADIANT-2): a randomised, placebo-controlled, phase 3 study. Lancet. 2011;378:2005–12. doi: 10.1016/S0140-6736(11)61742-X. [DOI] [PubMed] [Google Scholar]
- 13.Rindi G, Klimstra DS, Arnold R, et al. Nomenclature and classification of neuroendocrine neoplasm of the digestive system. In: Bosman FT, Carniero F, Hruban RH, Theise ND, editors. WHO classification of tumours of the digestive system. 4th. Lyon: International Agency for Research on Cancer; 2010. pp. 13–14. [Google Scholar]
- 14.Klimstra DS, Modlin IR, Coppola D, Lloyd RV, Suster S. The pathologic classification of neuroendocrine tumors. Pancreas. 2010;39:707–12. doi: 10.1097/MPA.0b013e3181ec124e. [DOI] [PubMed] [Google Scholar]
- 15.Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors. J Natl Cancer Inst. 2000;92:205–16. doi: 10.1093/jnci/92.3.205. [DOI] [PubMed] [Google Scholar]
- 16.Fernandez-Cuesta L, Peifer M, Lu X, et al. Frequent mutations in chromatin-remodelling genes in pulmonary carcinoids. Nat Commun. 2014;5:3518. doi: 10.1038/ncomms4518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Francis JM, Kiezun A, Ramos AH, et al. Somatic mutation of CDKN1B in small intestine neuroendocrine tumors. Nat Genet. 2013;45:1483–86. doi: 10.1038/ng.2821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Banck MS, Kanwar R, Kulkarni AA, et al. The genomic landscape of small intestine neuroendocrine tumors. J Clin Invest. 2013;123:2502–08. doi: 10.1172/JCI67963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yao JC, Phan AT, Chang DZ, et al. Efficacy of RAD001 (everolimus) and octreotide LAR in advanced low- to intermediate-grade neuroendocrine tumors: results of a phase II study. J Clin Oncol. 2008;26:4311–18. doi: 10.1200/JCO.2008.16.7858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Meric-Bernstam F, Akcakanat A, Chen H, et al. PIK3CA/PTEN mutations and Akt activation as markers of sensitivity to allosteric mTOR inhibitors. Clin Cancer Res. 2012;18:1777–89. doi: 10.1158/1078-0432.CCR-11-2123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yao JC, Hainsworth JD, Wolin EM, et al. Multivariate analysis including biomarkers in the phase III RADIANT-2 study of octreotide LAR plus everolimus or placebo among patients with advanced neuroendocrine tumors. J Clin Oncol. 2012;30:4014. (abstr). [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


