Abstract
G-box-binding factors are plant transcription factors, involved in a wide range of biological processes including abiotic stress responses. In this study, we analyzed the expression of OsGBF1 during salt stress in two contrasting Oryza sativa spp. indica genotypes, Rasi and Tellahamsa. Two-day-old seedlings were exposed to NaCl stress under two different conditions. One set was exposed to 100 mM NaCl before transferring to 250 mM (induction stress), while another set was transferred directly to 250 mM (shock stress). During early induction stress, OsGBF1 was up-regulated in Rasi when compared to Tellahamsa. We cloned full-length OsGBF1 from these two genotypes, and analyzed the sequences. Our analysis indicated the presence of transcript variants, which are designated as OsGBF1a and 1b. OsGBF1b variant retained introns, which lead to the generation of premature termination codon. OsGBF1b transcript levels were not significantly different at 12-h of induction stress in Tellahamsa and Rasi. At 24-h of shock stress, OsGBF1b was up-regulated in both genotypes and the transcript was more in Rasi. Since, OsGBF1a and 1b are predicted to be splice variants, we examined expression pattern of OsSKIP, a splicing factor and component of the spliceosome. In induction stress, OsSKIP was up-regulated at 12- and 24-h in Rasi when compared to Tellahamsa. On the contrary, at 24-h shock stress, OsSKIP was down-regulated in Rasi when compared to Tellahamsa. It is possible that OsSKIP expression was increased in Rasi during induction stress for accurate splicing that could aid in tolerance. This is the first report on OsGBF1 splice variant and the variant could have specific functions linked to stress tolerance in rice.
Electronic supplementary material
The online version of this article (10.1007/s13205-018-1370-4) contains supplementary material, which is available to authorized users.
Keywords: Induced salt stress, Intron retention, Premature termination codon, OsSKIP
Introduction
Transcription factors (TFs) are proteins that bind to DNA-regulatory sequences in the promoter regions of target genes to modulate the process of transcription. The major transcription factors (TFs) belong to the family of basic leucine zipper (bZIP), AP2/ERF, MYB, NAC, WRKY, Zn finger proteins (Golldack et al. 2011) and one of the largest families of plant TFs that specifically interact with the G-box element 5′ CACGTG 3′ are classified as G-box-binding factors (GBFs) (De-Vetten and Ferl 1994). Most of the genes that are responsive to cold, salt, dehydration, hypoxia and abscisic acid (ABA) are known to have G-boxes in their promoters (Shinozaki and Yamaguchi-Shinozaki 1997; Menkens et al. 1995). The G-box-binding factors have the modular structures like leucine zipper at the C terminal, proline rich domain at the N terminal and basic regions (De-Vetten and Ferl 1994). The two families of TFs that are considered as GBFs are, bZIP and basic helix loop helix (bHLH) proteins (Siberil et al. 2001). In Arabidopsis bZIP proteins are divided into ten groups (A, B, C, D, E, F, G, H, I, and S) based on similar basic region and additional conserved motifs, among them group G comprises GBFs (Jakoby et al. 2002). In Arabidopsis, three cDNA clones encoding GBF proteins, namely, GBF1, GBF2 and GBF3 have been isolated. GBF1 and GBF2 are expressed in leaves (light and dark grown) and roots, whereas GBF3 is expressed in roots and dark grown leaves (Schindler et al. 1992). In wheat, GBF1 expression was up-regulated within 24-h of exogenous ABA treatment (Sun et al. 2015). Since, GBFs can be important players in stress response; we attempted to identify prospective TF linked to salinity stress response in rice. We are reporting the discovery of OsGBF1 splice variant with introns. We hypothesize that this variant could have specific functions linked to stress tolerance in rice.
Materials and methods
Salinity stress imposition, RNA isolation and cDNA synthesis
To study the relevance of OsGBFs, 2-day-old seedlings of Oryza sativa ssp. indica genotypes, Rasi and Tellahamsa were exposed to salinity stress under laboratory conditions (Jayaprakash et al. 1998). The seedlings were segregated into three sets; one set was exposed to 100 mM NaCl before transferring to 250 mM NaCl (induction stress), second set was transferred directly to 250 mM NaCl (shock stress) and third set was allowed to grow in water (control). After 12-h of induction stress, seedlings from 100 mM were transferred to 250 mM NaCl, whereas other two sets were continued in the same treatments (Supplementary Fig. 1a). Seedlings were harvested at 12- and 24-h post-stress imposition, frozen in liquid nitrogen and used for gene expression studies. Total RNA was isolated from 100 mg tissue by modified phenol–chloroform method (Sajeevan et al. 2014). All RNA samples were treated with 1 U of DNaseI enzyme (MBI Fermentas, Hanover, MD, USA) at 37 °C for 45 min and heat inactivated at 75 °C for 10 min. First-strand cDNA was synthesized from total RNA (3 µg) in a final reaction volume of 20 µl using 40 picomol of oligo (dT) primers, 1× reaction buffer with 10 mM dNTPs, and 200 U Moloney Murine Leukemia virus reverse transcriptase (MMLV-RT; MBI Fermentas, Hanover, MD, USA) at 42 °C.
Expression analysis
Expression of OsGBF1a, OsGBF1b and OsSKIP was studied by semi-quantitative reverse transcriptase (RT)-PCR using specific primers (Table 1). PCR was performed at an annealing temperature of 57 °C (30 cycles) for OsGBF1a and 56 °C (33 cycles) for OsGBF1b. OsActin1 was used as the loading control. The intensity of amplified products separated on agarose gel was quantified using ImageJ 1.45 s software (http://imagej.nih.gov/ij). A quantitative difference in expression was calculated as the ratio of band intensities of target gene to that of housekeeping gene. The relative expression ratio from three independent replicates was calculated and represented. Significant difference between the values was calculated using Student’s t test.
Table 1.
List of primers used and their application
| OsGBF1 F1 | ATGGGAAATGACGAAGCTGTAGTTACTC | Full-length amplification |
| OsGBF1 R1 | TTACCTTGCGGCTACAGCATCAGTC | Full-length amplification |
| OsGBF1a F2 | ATCCAGCCCTTAGTCCA | Expression of variant-a |
| OsGBF1a R2 | CCACCAGTTTCCACGCCTGATT | Expression of variant-a |
| OsGBF1b F3 | GCCCATTGAGCATGGAGCCAG | Expression of variant-b |
| OsGBF1b R3 | CTAAGCTGCAATCTCCACTGCATTG | Expression of variant-b |
| OsSKIP F4 | GGTGCTTCAGAGAGGTCTGG | Expression |
| OsSKIP R4 | CCTCCACCCTCGTAATCATCT | Expression |
| OsActin F | CCATAATGAAGTGTGATGT | Internal control |
| OsActin R | GGACCTGACTCGTCATACTC | Internal control |
Cloning of full-length OsGBF1
To analyze the features of OsGBF1, full-length gene was cloned using cDNA generated from salt stressed tissues of Rasi and Tellahamsa using gene-specific primers (Table 1). The gene sequence reported in NCBI (http://www.ncbi.nlm.gov/) database (AY606941.1) was used to design specific primers (Table 1). The PCR products were cloned using the InsTAclone PCR cloning kit (MBI Fermentas Hanover, MD, USA), and sequenced (ABI 3730Xl sequencer). The identity of the cloned gene was confirmed by BLASTn analysis in NCBI database.
Results and discussion
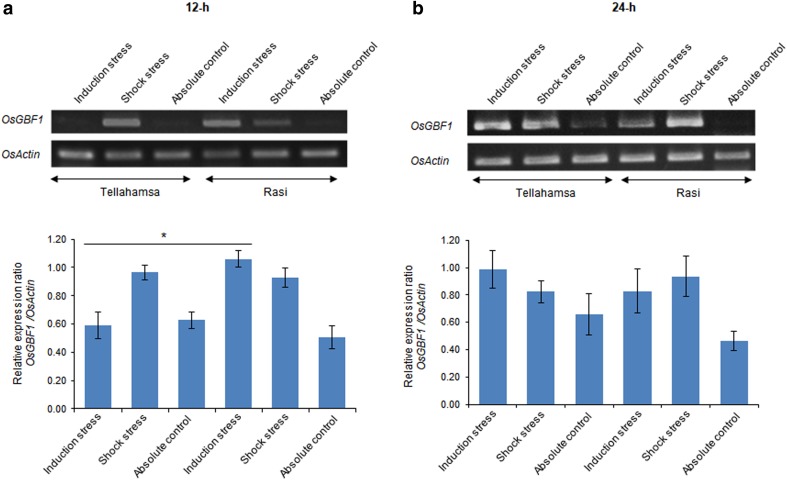
The rice genotypes, Rasi and Tellahamsa used in this study have been found to be tolerant and sensitive to salinity stress, respectively. There was significant difference in growth between Rasi and Tellahamsa under salinity stress. At the end of shock stress, 18% reduction in growth was observed in Tellahamsa, whereas only 5.9% reduction was noticed in Rasi, suggesting that Rasi is relatively tolerant to salinity stress (Supplementary Fig. 1b). OsGBF1 was up-regulated at 12-h of induction stress in Rasi compared to Tellahamsa. At 24-h post-induction stress, GBF1 expression was increased in Tellahamsa. However, in Rasi, GBF1 expression was increased in both induction and shock stresses when compared to control (Fig. 1a, b). Similar results were reported in wheat by Sun et al. (2015). Triticum aestivum GBF1 transcript levels were increased upon exposure to 200 mM NaCl, indicating the stress responsive nature of the gene. Sequence analysis of OsGBF1 showed identity with salinity stress inducible bZIP protein. In tolerant genotype Rasi, the expression level of GBF1 was more than Tellahamsa, suggesting that this regulon might have a role in stress response in rice.
Fig. 1.
Expression of OsGBF1 in Rasi and Tellahamsa under salinity stress. a OsGBF1a expression 12-h of stress. b OsGBF1a expression 24-h of stress. The bar diagram indicates relative expression of the gene at 12- and 24-h of stress. Asterisk (*) and line drawn above the bars indicate significant difference in similar stresses and control treatments (t test; P < 0.05)
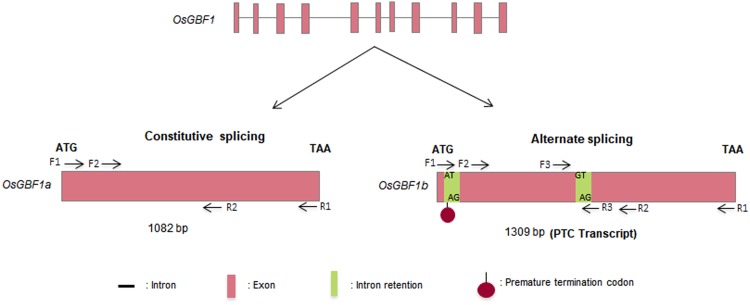
We cloned two transcript variants of GBFs, 1082 and 1309 bp from stressed cDNA of Rasi and Tellahamsa. BLASTn analysis of the 1082 and 1309 bp variants showed 99 and 100% sequence identity with a query coverage of 100 and 83%, respectively, with O. sativa (indica cultivar group) salt stress inducible bZIP transcript. Since the transcript identified was similar to GBF1, these variants have been designated as OsGBF1a and OsGBF1b. Genomic DNA sequence analysis of OsGBF1 revealed the presence of 11 exons and 10 introns. In OsGBF1a introns are spliced; however, in OsGBF1b, the introns between exons 1 and 2, 5 and 6 were retained (Fig. 2). The lengths of both introns are 112 bp, with the first and second intron at 60 and 520 nucleotides, respectively, from translation start site (TSS). OsGBF1b sequence has been deposited in GenBank under the accession number JZ903929. The common alternate splicing events reported include intron retention (IR), exon skipping, alternative 5′ or 3′ site selection, and mutually exclusive exons, of which IR is common in plants (Syed et al. 2012). The first intron which we are reporting starts with AT and ends with AG. However, some rare donor–acceptor sites such as AT-AC, GC-AG, GT-GG have also been reported (Dubrovina et al. 2013). The second intron starts with GT and ends with AG, as reported in plants (Dubrovina et al. 2013). OsGBF1b has no open reading frame due to premature termination codon (PTC) at 66 nucleotides from the start site (Fig. 2). In Arabidopsis, IR in mRNA results in generation of PTC (Feng et al. 2015). The alternative splicing of Circadian Clock Associated 1 (CCA1) and serine/arginine-rich (SR) pre-mRNA’s produced PTC containing transcripts due to IR (Filichkin et al. 2015; Reddy and Shad 2011).
Fig. 2.
Schematic representation of OsGBF1 splice variants, OsGBF1a and OsGBF1b. Arrows indicate position of the primers used in this study, F- and R-represents forward and reverse primer, respectively. For description of F1-R1, F2-R2, and F3-R3 refer Table 1. AT-AG and GT-AG correspond to donor–acceptor splice sites in intron 1 and 2, respectively
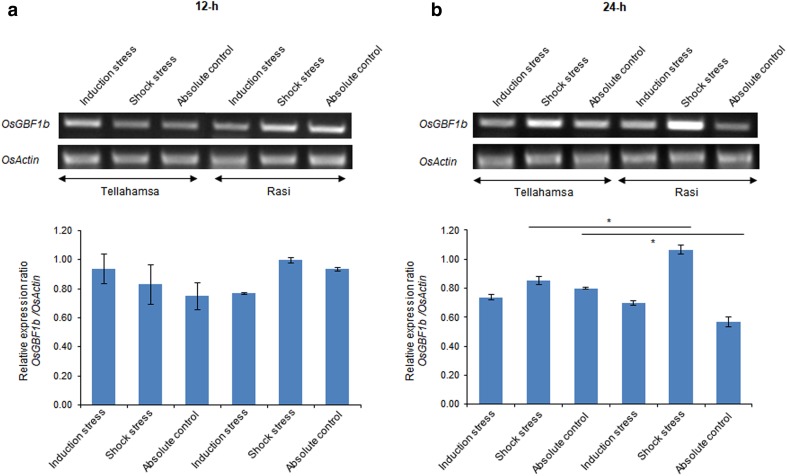
To analyze the expression of OsGBF1b under control and salt stress conditions in rice genotypes, we designed variant-specific primer (Table 1; Fig. 2). At 12-h post-induction stress, OsGBF1b expression was similar in Tellahamsa and Rasi. However, in Rasi the transcript was down-regulated in induction stress compared to control and shock stress (Fig. 3a). At 24-h, OsGBF1b expression was up-regulated in both the genotypes during shock stress, and the expression levels were high in Rasi as compared to Tellahamsa (Fig. 3b). It was documented that alternate splice forms are high in salt stress-induced Arabidopsis as compared to control, with IR being prevalent event under salt stress generating a PTC (Ding et al. 2014; Feng et al. 2015). The alternate spliced genes include those that have a role in abiotic stress response, hormone signaling, transcriptional regulation, and RNA processing (Ding et al. 2014). It is intriguing to speculate as to what could be the purpose of this transcript in stress conditions. Transcripts with PTC and/or long 3′ untranslated regions (UTRs), splice junction downstream of stop codon, PTC with downstream splice junction, and open reading frames in 5′ UTR are substrates for nonsense mediated decay (NMD) (Kalyna et al. 2012). In Arabidopsis, analysis of At5g37055 (SERRATED LEAVES AND EARLY FLOWERING—SEF) revealed three different IR transcripts (involving intron-1 and -2) containing PTC, but not targeted to NMD pathway, on the contrary transcript generated by alternative 3′ splice site (involving exon-3) containing PTC is targeted to NMD pathway (Kalyna et al. 2012). In our study OsGBF1b was up-regulated in shock stress at 24-h, which makes us to predict that this variant is probably not a substrate of NMD. At 24-h post-shock stress, both genotypes showed an increase in OsGBF1b expression. Our observation is in accordance with the documentation that spliced variants increase with the salt concentration (Feng et al. 2015). Analysis of differential alternate spliced genes under salt stress included those with role in abiotic stress, suggesting that alternative splicing is not a random process, but is responsive to stress (Ding et al. 2014). OsGBF1b noticed in our study seems to be generated not by aberrant splicing, and the increase in expression that we have observed indicates that this variant probably has a role in salinity stress tolerance.
Fig. 3.
Expression of OsGBF1b in Rasi and Tellahamsa under salinity stress. a OsGBF1b expression 12-h of stress. b OsGBF1b expression 24-h of stress. The bar diagram indicates relative expression of the gene at 12- and 24-h of stress. Asterisk (*) and line drawn above the bars indicate significant difference in similar stresses and control treatments (t test; P < 0.05)
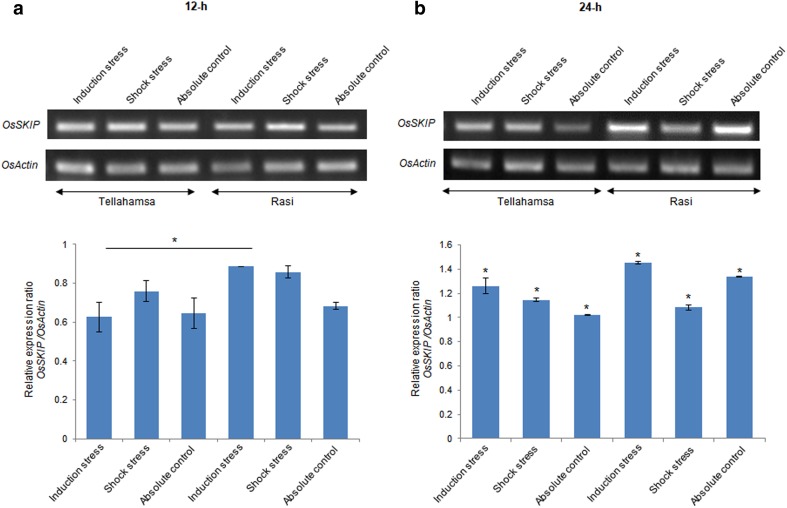
Accurate splicing of transcripts is essential for protein expression. Since, OsGBF1b variant showed differential expression, we studied the expression profile of OsSKIP under salt stress. OsSKIP (a homolog of pre-mRNA splicing factor 45 in yeast), part of spliceosome complex closely associated with SR45 (Wang et al. 2012). At 12- and 24-h of induction stress, OsSKIP expression was up-regulated in Rasi, when compared to Tellahamsa. However, at 24-h shock stress, OsSKIP expression was down-regulated in Rasi when compared to Tellahamsa (Fig. 4a, b). OsSKIP is constitutively expressed in leaf, root, stem, and is up-regulated during salinity stress in rice (Hou et al. 2009). It has been reported that SKIP under salt stress is required for splicing of pre-mRNA encoding salt responsive genes such as NHX1, RD29A and P5CS1 (Feng et al. 2015). Hence, OsSKIP expression was probably increased in Rasi during induction of salt stress for accurate splicing of stress responsive genes that could aid in tolerance. This is the first report on the discovery of OsGBF1 splice variant, which shows intron retention resulting in generation of premature termination codon. We hypothesize that this variant could have specific functions linked to stress tolerance.
Fig. 4.
Expression of OsSKIP under salinity stress. a 12-h stress. b 24-h stress. The bar diagram indicates relative expression of the gene at 12- and 24-h of stress. Asterisk (*) and line drawn above the bars indicates significant difference in similar stresses and control treatments (t test; P < 0.05)
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1 Salinity stress imposition in Rasi and Tellahamsa. (a) Schematic representation of stress imposition. (b) Percent reduction in growth over control of Rasi and Tellahamsa seedlings at the end of stress. Significant difference are indicated by small letters assessed by Duncan’s Multiple Range Test (DMRT) (P = 0.05). (TIF 16629 KB)
Acknowledgements
We thank Niche Area of Excellence-Indian Council for Agriculture Research (NAE-ICAR) Grant File. No. 10 (15)/2012 and DST-FIST Government of India, New Delhi, India, and Dow AgroSciences, Indianapolis, USA (No./S-8/C-3/5055/2011-12) for providing financial support.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
The research has been conducted with the consent of the authorities and that the research does not involve human participants and/or animals.
References
- De Vetten NC, Ferl RJ. Transcriptional regulation of environmentally inducible genes in plants by an evolutionary conserved family of G-box binding factors. Int J Biochem. 1994;26:1055–1068. doi: 10.1016/0020-711X(94)90128-7. [DOI] [PubMed] [Google Scholar]
- Ding F, Cui P, Wang Z, Zhang S, Ali S, Xiong L. Genome-wide analysis of alternative splicing of pre-mRNA under salt stress in Arabidopsis. BMC Genom. 2014;15:431–444. doi: 10.1186/1471-2164-15-431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dubrovina AS, Kiselev KV, Zhuravlev YN. The role of canonical and noncanonical pre-mRNA splicing in plant stress responses. BioMed Res Int. 2013;2013:Article ID 264314. doi: 10.1155/2013/264314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng J, Li J, Gao Z, Lu Y, Yu J, Zheng Q, Yan S, Zhang W, He H, Ma L, Zhu Z. SKIP confers osmotic tolerance during salt stress by controlling alternative gene splicing in Arabidopsis. Mol Plant. 2015;6:1038–1052. doi: 10.1016/j.molp.2015.01.011. [DOI] [PubMed] [Google Scholar]
- Filichkin SA, Cumbie JS, Dharmawardhana P, Jaiswal P, Chang JH, Palusa SG, Reddy ASN, Megraw M, Mockler TC. Environmental stresses modulate abundance and timing of alternatively spliced circadian transcripts in Arabidopsis. Mol Plant. 2015;8:207–227. doi: 10.1016/j.molp.2014.10.011. [DOI] [PubMed] [Google Scholar]
- Golldack D, Lüking I, Yang O. Plant tolerance to drought and salinity: stress regulating transcription factors and their functional significance in the cellular transcriptional network. Plant Cell Rep. 2011;30:1383–1391. doi: 10.1007/s00299-011-1068-0. [DOI] [PubMed] [Google Scholar]
- Hou X, Xie K, Yao J, Qi Z, Xiong L. A homolog of human ski-interacting protein in rice positively regulates cell viability and stress tolerance. Proc Natl Acad Sci USA. 2009;106:6410–6415. doi: 10.1073/pnas.0901940106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jakoby M, Weisshaar B, Dröge-Laser W, Vicente-Carbajosa J, Tiedemann J, Kroj T, Parcy F. bZIP transcription factors in Arabidopsis. Trends Plant Sci. 2002;7:106–111. doi: 10.1016/S1360-1385(01)02223-3. [DOI] [PubMed] [Google Scholar]
- Jayaprakash TL, Ramamohan G, Krishnaprasad BT, Prasad TG, Mathew MK, Udayakumar M. Genotypic variability in differential expression of lea2 and lea3 genes and proteins in response to salinity stress in finger millet (Eleusine coracana Gaertn) and Rice (Oryza sativa L.) seedlings. Ann Bot. 1998;82:513–522. doi: 10.1006/anbo.1998.0712. [DOI] [Google Scholar]
- Kalyna M, Simpson CG, Syed NH, Lewandowska D, Marquez Y, Kusenda B, Marshall J, Fuller J, Cardle L, McNicol J, Dinh HQ, Barta A, Brown JWS. Alternative splicing and nonsense-mediated decay modulate expression of important regulatory genes in Arabidopsis. Nucleic Acids Res. 2012;40:2454–2469. doi: 10.1093/nar/gkr932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menkens AE, Schindler U, Cashmore AR. The G-box: a ubiquitous regulatory DNA element in plants bound by the GBF family of bZIP proteins. Trends Biochem Sci. 1995;20:506–510. doi: 10.1016/S0968-0004(00)89118-5. [DOI] [PubMed] [Google Scholar]
- Reddy AS, Shad AG. Plant serine/arginine-rich proteins: roles in precursor messenger RNA splicing, plant development, and stress responses. WIREs RNA. 2011;2:875–889. doi: 10.1002/wrna.98. [DOI] [PubMed] [Google Scholar]
- Sajeevan RS, Shivanna MB, Nataraja KN. An efficient protocol for total RNA isolation from healthy and stressed tissues of mulberry (Morus sp.) and other species. Am J Plant Sci. 2014;5:2057–2065. doi: 10.4236/ajps.2014.513221. [DOI] [Google Scholar]
- Schindler U, Menkens AE, Beckmann H, Ecker JR, Cashmore AR. Heterodimerization between light-regulated and ubiquitously expressed Arabidopsis GBF bZIP proteins. EMBO J. 1992;11:1261–1273. doi: 10.1002/j.1460-2075.1992.tb05170.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinozaki K, Yamaguchi-Shinozaki K. Gene expression and signal transduction in water-stress response. Plant Physiol. 1997;115:327–334. doi: 10.1104/pp.115.2.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siberil Y, Doireau P, Gantet P. Plant bZIP G-box binding factors modular structure and activation mechanisms. Eur J Biochem. 2001;268:5655–5666. doi: 10.1046/j.0014-2956.2001.02552.x. [DOI] [PubMed] [Google Scholar]
- Sun Y, Xu W, Jia Y, Wang M, Xia G. The wheat TaGBF1 gene is involved in the blue-light response and salt tolerance. Plant J. 2015;84:1219–1230. doi: 10.1111/tpj.13082. [DOI] [PubMed] [Google Scholar]
- Syed NH, Kalyna M, Marquez Y, Barta A, Brown JWS. Alternative splicing in plants—coming of age. Trends Plant Sci. 2012;17:616–623. doi: 10.1016/j.tplants.2012.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Wu F, Xie Q, Wang H, Wang Y, Yue Y, Gahura O, Ma S, Liu L, Cao Y, Jiao Y, Puta F, McClung CR, Xu X, Ma L. SKIP is a component of the spliceosome linking alternative splicing and the circadian clock in Arabidopsis. Plant Cell. 2012;24:3278–3295. doi: 10.1105/tpc.112.100081. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Fig. 1 Salinity stress imposition in Rasi and Tellahamsa. (a) Schematic representation of stress imposition. (b) Percent reduction in growth over control of Rasi and Tellahamsa seedlings at the end of stress. Significant difference are indicated by small letters assessed by Duncan’s Multiple Range Test (DMRT) (P = 0.05). (TIF 16629 KB)