Abstract
Background: Rheumatoid arthritis (RA) is a inflammatory disease that characterized with the destruction of synovial joint, which could induce disability. Inflammatory response mediated the RA. It has been reported that MiR-128-3p is significantly increased in RA, while the potential role was still unclear.
Methods: T cells in peripheral blood mononuclear cell (PBMC) were isolated from the peripheral blood from people of RA and normal person were used. Real-time PCR was performed to detect the expression of MiR-128-3p, while the protein expression of tumor necrosis factor-α-induced protein 3 (TNFAIP3) was determined using Western blot. The levels of IL-6 and IL-17 were measured using enzyme-linked immunosorbent assay (ELISA). The expression of CD69 and CD25 was detected using flow cytometry. The RA mouse model was constructed for verification of the role of MiR-128-3p.
Results: The expression of MiR-128-3p was significantly increased, while TNFAIP3 was decreased, the levels of IL-6 and IL-17 were also increased in the T cells of RA patients. Down-regulated MiR-128-3p significantly suppressed the expression of p-IkBα and CD69, and CD25in T cells. MiR-128-3p targets TNFAIP3 to regulate its expression. MiR-128-3p knockdown significantly suppressed the activity of nuclear factor κB (NF-κB) and T cells by up-regulating TNFAIP3, while cells co-transfected with si-TNFAIP3 abolished the effects of MiR-128-3p knockdown. The in vivo experiments verified the potential role of MiR-128-3p on RA.
Conclusion: Down-regulated MiR-128-3p significantly suppressed the inflammation response of RA through suppressing the activity of NF-κB pathway, which was mediated by TNFAIP3.
Keywords: inflammation, MiR-128-3p, NF-κB, Rheumatoid arthritis, TNFAIP3
Introduction
Rheumatoid arthritis (RA) is a inflammatory disease that characterized with the destruction of synovial joints, which severely threatens human joint. It has reported that the progressed RA could further lead to disability. Even though great progression has been made on effective medications for RA, the disability rate that induced by RA was still high. Genetic studies have reported that the rheumatoid factors and several inflammatory cytokines played an important role in the mechanism of RA, while the etiology of RA remains unclear.
Immunity role of T cells plays a critical role in RA. The abnormally T cells often evoke the dysregulated expression of several inflammatory mediators, such as tumor necrosis factor-α (TNF-α), interleukin-1(IL-1), IL-6, and IL-17. The nuclear factor κB (NF-κB), a transcription factor, is involved in the inflammatory process of RA, and the NF-κB signaling pathway has been reported to conduct the inflammatory mediators of RA [1,2]. To interference, the activity of NF-κB signaling pathway could effectively protect body against the aggression of inflammation [3]. Therefore, to explore the potential role of NF-κB signaling pathway is important for further RA study.
Tumor necrosis factor-α-induced protein 3 (TNFAIP3), also known as A20, is a cytoplasmic zinc finger protein that was originally identified as a dual inhibitor of NF-κB activation. Mounting studies have reported that the TNFAIP3 played an important role in the inflammation [4–6]. A recent study demonstrated that the expression of TNFAIP3 was significantly decreased in RA, and regulated the expression of several key cytokines of inflammatory process via negatively regulating the activity of NF-κB signaling pathway [7,8]. While the potential role of TNFAIP3 on RA was still undocumented.
MicroRNA (miRNA) is a class of noncoding RNA (ncRNA) with the length of ~22 nucleotides and regulates gene expression at the post-transcriptional level by binding the 3′-UTR of targeted genes. Recently, much miRNAs have been reported to mediate the cell apoptosis, signaling transduction, body metabolism, and immune response. Plenty miRNAs, such as miR-146a [9], miR-155 [10], miR-16 [11], miR-23b [12], miR-203 [13], miR-124a [14], miR-346 [15], and miR-223 [16], have been reported to mediated the synovial fibroblasts, peripheral blood mononuclear cells and T cells in RA, indicating the potential role of miRNA in RA. MiR-128-3p has been widely studied in various diseases, such as esophageal squamous-cell cancer [17], lung cancer [18], and kidney inflammation [19]. Mounting studies have reported that MiR-128-3p served as a tumor suppressor gene [18,20]. While the expression of miR-128 was significantly higher in RA patients than that in control [21]. However, whether miR-128 mediated the regulation mechanism of RA was still unclear.
In the present study, we first examined the inflammation-related cytokines; then we conducted in vitro experiments to identify the potential mechanism of RA; finally, the in vivo experiments were performed to verify our results. Our aims were to explore the potential role of MiR-128-3p on RA, and the potential mechanism of inflammatory response on RA. The present study will provide great help for further clinical therapy of RA.
Materials and methods
Patients
Twenty patients, who are diagnosed with RA in our hospital, were recruited in the present study. Another 20 healthy volunteers were acted as control. The study were approved by the ethic committee of Clinical Medical College, Yangzhou University. All patients were assigned the informed consent. Blood was collected after 12 h at the last medicine taking to minimize the effects of the drugs.
Isolation of T cells
To isolate T cells, the peripheral blood mononuclear cells (PBMCs) were separated from the blood samples. The blood was mixed with 2% dextran solution (Sigma, Japan) and cultured for 30 min in the room temperature. The supernatants were collected and layered using a density gradient centrifuge to separate the mononuclear cells. T cells were purified using antihuman CD3-coated magnetic beads. The concentration of T cells was cultured in RPMI-1640 medium containing 10% fetal bovine serum (FBS) at 37°C in a humidified atmosphere.
The activation of T cells and flow cytometry
T cells were cultured as described above and stimulated for 18 h with anti-CD3 and anti-CD28 antibodies or PMA/ionomycin to check respectively CD69 and CD25 expression. CD69-PE (BD Biosciences; 585/42BP) and CD25-PE (ImmunoStep, Salamanca, Spain; 585/42BP) expression were measured by fluorescence activated cell storing (FACS). The details were described in the article of Dong et al. [22].
Real-time PCR
The total RNA was isolated from T cells using the RNA Isolation Kit (Sigma-Aldrich), according to the manufacturer’s instruction. RNA quality was determined using the agarose gel electorphesis, and the concentration was detected using a Nanodrop Spectrophotometer. The cDNA was reverse transcripted using a QuantiTect Reverse Transcription Kit (Qiagen), according to the instruction. The real-time PCR was performed using an ABI Prism 7500 Fast Real-Time PCR system (Applied Biosystems, U.S.A.). Each experiment was repeated for three independent times, and U6 served as internal control.
Western blot
Cells were lysed using the RIPA Lysis buffer. The protein concentration was determined using micro bicinchoninic acid (BCA) method. The proteins were separated using 10% SDS-PAGE, and the equal amount of protein was transferred to PVDF membrane and incubated with the primary antibodies at 4°C for 24 h. Then the PVDF membrane was incubated with the second primary at room temperature for 1 h. β-Actin served as internal control. The ECL chemiluminescence was used to visualize the protein bands.
Luciferase reporter assay
The miRNA target interactions were based on the principle of the references [23,24]. The online TargetScan (v7.0; targetscan.org) was used to predict the potential binding site between MiR-128-3p and TNFAIP3 as described in the previous study [25]. To further identify the potential relationship, luciferase reporter assay was conducted. TNFAIP3 containing a wild-type or mutant 3′-UTR was constructed. HEK 293T cells were planted in 24-well plates, and the wild-type or mutant of TNFAIP3 and MiR-128-3p was co-transfected using Lipofectamine 2000. The activity was measured after 24 h using a Dual-Luciferase® Reporter Assay System (Promega Corporation, U.S.A.).
Cell transfection
Cells were planted in a 24-well plate for 24 h, the inhibitor, mimic, and their negative control (synthesized in Invitrogen, Shanghai, China) were transfected to the cells using Lipofectamine 2000 (Invitrogen), and the concentration of MiR-128-3p inhibitor and mimic was 100 nmol/l.
Enzyme-linked immunosorbent assay (ELISA)
The levels of IL-6 and IL-17 in the serum were detected using ELISA kit (USCN Life Science Inc., Wuhan, China). The samples were incubated with the enzyme-labeled antibody for 30 min at 37°C. The sulfuric acid of 2 M (0.05 ml) was used to suspend the reaction. The ultraviolet spectrophotometer (Eppendorf BioSpectrometer) was used to visualize the values with the OD = 450 nm.
Animals
The C57BL/6 mice were purchased from the Experimental Animals center of Shanghai. The mice were aged at 4–6 weeks and weighted 20–30 g. The mice were cultured in a cozy environment and supplied with plenty of food and water. A 12 h day–night circle was also provided. The animal experiments were approved by Clinical Medical College, Yangzhou University.
Establishment of RA mouse model
To establish the RA mice model, the mice were injected intradermally using 0.1 ml of an emulsion containing 100 μg of bovine type II collagen and complete Freund’s adjuvant (Arthrogen-CIA; Chondrex, Seattle, WA) into the base of the mice tail. After 2 weeks, 100 μg of type II collagen, dissolved and emulsified 1:1 with incomplete Freund’s adjuvant (Difco, Detroit, MI), was administered to the hind leg. To further identify the role of MiR-128-3p on RA in vivo, the RA mode was divided into two groups (n=6 in each group): negative control and miR-128 inhibitor group. MiR-128 inhibitor (60 nmol/l) was intravenous injection twice a week for 4 weeks.
The arthritis score calculation
The RA score of hind feet of the rats was calculated to assess the inflammation level of arthritis. The details were as follows: 0 score, no arthritis symptom in hind feet; 1 score, red with soft tissue swelling was observed in one or two hind feet; 2 scores, red with soft tissue swelling was observed in three to four joints in hind feet; 3 scores, red with soft tissue swelling was observed in five or even more joints in hind feet; 4 scores, hind feet showed severe arthritis symptoms.
Statistical analysis
All data were presented as means ± SD. Statistical differences were analyzed using student’s t-test for paired samples and ANOVA for multigroup samples. P<0.05 considered as statistical analysis.
Results
The expression pattern of MiR-128-3p and TNFAIP3 in clinical tissues
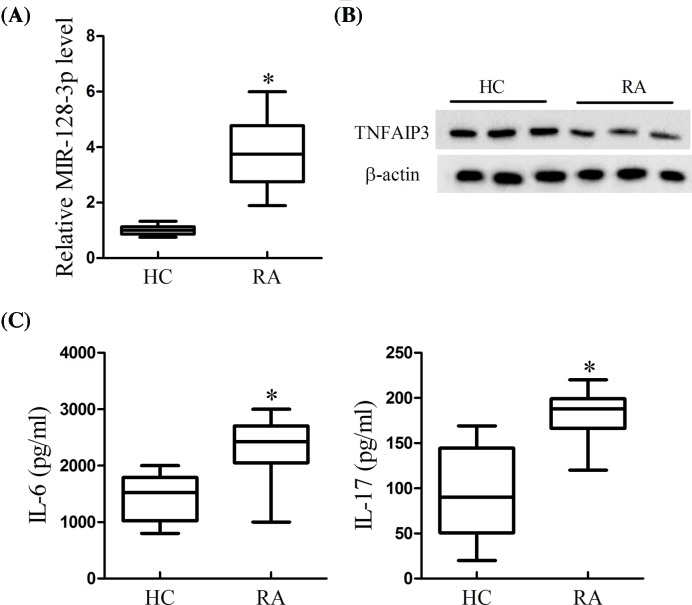
To learn the expression of MiR-128-3p and TNFAIP3 and the level of inflammatory cytokines in RA, the PBMCs of clinical RA patients were collected and the T cells were isolated. As is presented in Figure 1, the expression of MiR-128-3p was significantly increased (Figure 1A), while TNFAIP3 was decreased in T cells of RA patients compared with control (Figure 1B). In addition, the cytokines of IL-6 and IL-17 were markedly higher in RA patients rather than in control (Figure 1C). The results indicated that MiR-128-3p was higher in RA patients along with the inflammatory response. In addition, we analyzed the correlation between miR-128-3p expression and the clinical characteristics of RA patients. As presented in Table 1, the miR-128 expression was correlated with rheumatoid factor (RF), erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), and mean disease activity score in 28 joints (DAS28), but did not correlated with age, sex, and disease duration.
Figure 1. The expression pattern of MiR-128-3p and TNFAIP3 in clinical T cells of RA.
(A) The expression of MiR-128-3p was determined using real-time PCR. (B) The expression of TNFAIP3 was measured using Western blot. (C) The levels of IL-6 and IL-17 were detected using ELISA; *P<0.05 vs. HC.
Table 1. Characteristics of RA samples.
| Demographics | Active RA (n=20) | Healthy (n=20) |
|---|---|---|
| Age (years), mean (range) | 53 (26–77) | 49.1 (21–78) |
| Sex, female/male | 18/2 | 17/3 |
| Disease duration (years), mean (range) | 11 (2–23) | – |
| RF, positive/negative | 15/5* | – |
| ESR (mm/h), mean (range) | 65.23 (28–112)* | 7.5 (6–17) |
| CRP (mg/l), mean (range) | 69.62 (31–148)* | 2.8 (1–8) |
| Mean DAS28 | 5.91 (4.3–7.1)* | – |
Abbreviations: RF, rheumatoid factor; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; DAS28, disease activity score in 28 joints;
P<0.05 represented the relationship between miR-128 expression and the clinical characters.
MiR-128-3p knockdown significantly suppressed the signaling of NF-κB and decreased the activation of T cells
To explore the effects of MiR-128-3p on the pathway of NF-κB and the activation of T cells, the T cells were first activated and then transfected with MiR-128-3p inhibitor. The results presented that the expression of MiR-128-3p was significantly decreased (Figure 2A). MiR-128-3p knockdown significantly decreased the expression of p-IkBα, but did not affect the expression of IkBα (Figure 2B). Additionally, the MiR-128-3p inhibitor also suppressed the ratio of CD69 and CD25 (Figure 2C). Taken together, down-regulated MiR-128-3p suppressed the inflammatory response as well as the activity of NF-κB.
Figure 2. Effects of MiR-128-3p on the activity of NF-kB and T cells.
The activated T cells were transfected with MiR-128-3p inhibitor. (A) Real-time PCR was performed to determine the expression of MiR-128-3p. (B) Western blot was carried out to determine the protein expression of p-IkBα and IkBα. (C) The expression of CD69 and CD25 was measured using flow cytometry; *P<0.05 vs. NC.
The relationship between MiR-128-3p and TNFAIP3
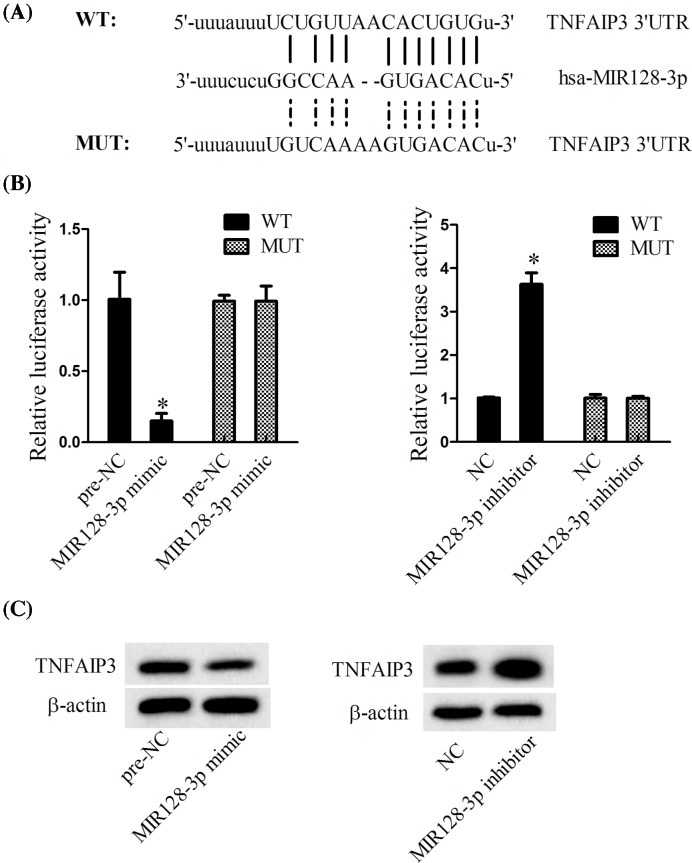
Online TargetScan predicted that MiR-128-3p could bind with the 3′-UTR of TNFAIP3 (weighted context++ score: −0.10; total context++ score: −0.11; aggregate PCT: <0.1). The luciferase reporter assay revealed that MiR-128-3p mimic significantly decreased the relative luciferase activity in WT-TNFAIP3 3′-UTR, while MiR-128-3p inhibitor dramatically promoted the relative luciferase activity in WT-TNFAIP3 3′-UTR (Figure 3B). Furthermore, MiR-128-3p mimic significantly decreased the expression of TNFAIP3, while MiR-128-3p inhibitor significantly promoted the expression of TNFAIP3, indicating that MiR-128-3p directly regulated the expression TNFAIP3.
Figure 3. The relationship between MiR-128-3p and TNFAIP3.
(A) Online TargetScan predicted that MiR-128-3p bound with the 3′-UTR of TNFAIP3. (B) Luciferase reporter assay was performed to determine the role of MiR-128-3p on the luciferase activity. (C) Western blot was performed to determine the role of MiR-128-3p on TNFAIP3; *P<0.05 vs. pre-NC or NC.
MiR-128-3p knockdown decreased the activation of NF-κB via up-regulating the expression of TNFAIP3
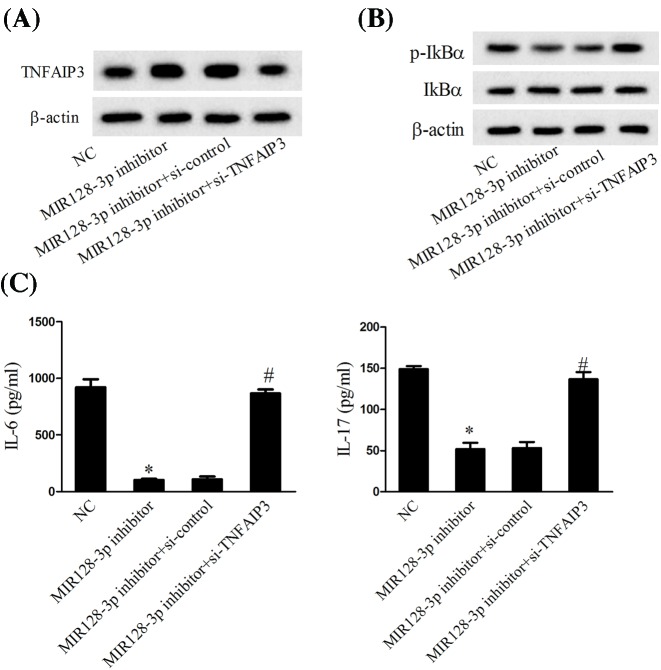
The activated T cells transfected with MiR-128-3p inhibitor significantly promoted the expression of TNFAIP3, while then co-transfected with si-TNFAIP3 abolished the effects of MiR-128-3p inhibitor (Figure 4A). T cells transfected with MiR-128-3p inhibitor decreased the expression of p-IkBα, while co-transfected with si-TNFAIP3 reversed the effects of MiR-128-3p inhibitor (Figure 4B). In addition, the MiR-128-3p inhibitor also decreased the cytokines of IL-6 and IL-17, while cells co-transfected with si-TNFAIP3 significantly reversed them (Figure 4C). Taken together, down-regulated MiR-128-3p promoted the expression of TNFAIP3, which further decreased the activation of NF-κB.
Figure 4. The role of MiR-128-3p knockdown on the activity of NF-κB.
(A) The expression of TNFAIP3 was determined using Western blot. (B) The expression of p-IkBα and IkBα was measured using Western blot. (C) The levels of IL-6 and IL-17 were determined using ELISA; *P<0.05 vs. NC; #P<0.05 vs. MiR-128-3p inhibitor +si-control.
Verification of the effects of MiR-128-3p in vivo
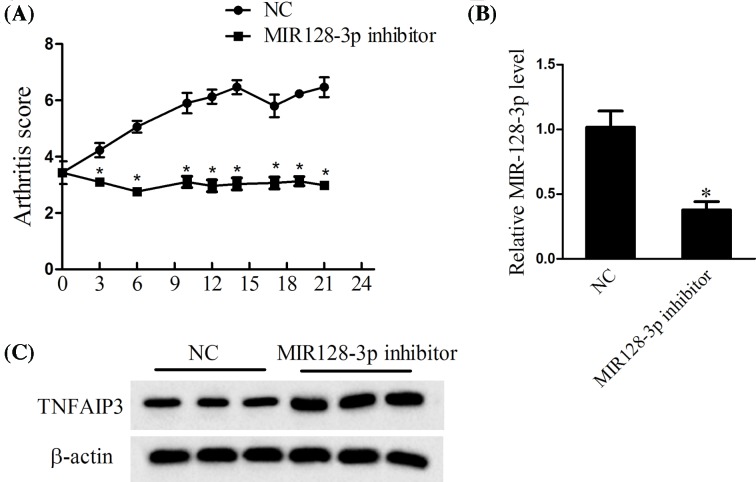
The rheumatoid arthritis mouse model was joint injection of MiR-128-3p inhibitor. The peripheral blood was used to detect the expression of TNFAIP3. The results revealed that the arthritis score was lower in MiR-128-3p inhibitor group than that in control (Figure 5A). The expression of MiR-128-3p was also decreased in MiR-128-3p inhibitor group (Figure 5B), while the expression of TNFAIP3 was increased in MiR-128-3p inhibitor group (Figure 5C). The results indicated that down-regulated MiR-128-3p protected joint against arthritis.
Figure 5. In vivo experiments to verify the expression of MiR-128-3p and TNFAIP3.
Arthritis mice were injected with MiR-128-3p inhibitor. (A) The arthritis scores were determined every 3 days. (B) The expression of MiR-128-3p was determined using real-time PCR. (C) The TNFAIP3 was determined using Western blot; *P<0.05 vs. NC.
Discussion
MiRNAs have been identified to mediate various process in body, the dysregulated miRNAs were often resulted in the diseases. For example, miR-486-5p served as a diagnostic marker in cervical cancer [26]; down-regulated miR-221-3p is associated with the poor prognosis of breast cancer [27]; miR-204-3p is a factor that induced familial mediterranean fever [28]; miR-146a and miR-155 showed dysregulated in oral lichen planus [29]. In addition, miRNA mediated inflammation in diseases, for example, miR-124 mediated the inflammatory pathogenesis of Parkinson’s disease [30]; miR-100 suppressed chronic vascular inflammation by stimulation of endothelial autophagy [31]. In addition, miR-548a-3p regulates the inflammatory response of RA by regulating the activity of TLR4/NF-κB signaling pathway [32]. MiR-10a-5p regulated joint inflammation in synoviocytes [33]. MiR-128-3p mediated the inflammation of rat kidney cells [19], and MiR-128-3p is a novel candidate oncogenic marker in T-cell acute lymphoblastic leukemia [34]. In the present study, MiR-128-3p was significantly increased in T cells of RA, indicating the potential role of MiR-128-3p in RA.
To further explore the potential role of MiR-128-3p on RA, the TNFAIP3 a negative regulator of NFκB activation was employed. TNFAIP3 was first discovered in human umbilical vein endothelial cells, and then researchers found that overexpressed TNFAIP3 dramatically suppressed the activation of NF-κB signaling pathway in various responses, such as the megakaryocytopoiesis [35], the differentiation of THP-1 cells [36], as well as the inflammation [37]. However, the potential targets between MiR-128-3p and TNFAIP3 were still did not documented. In the present study, we first found that TNFAIP3 was dominantly decreased in the T cells of RA. We further explored the potential role between MiR-128-3p and TNFAIP3. The online Targetscan predicted that MiR-128-3p bind with the TNFAIP 3′-UTR, the luciferase reporter assay further demonstrated that TNFAIP3 is a target gene of MiR-128-3p. The present study first identified the target relationship between TNFAIP3 and MiR-128-3p, indicating that MiR-128-3p mediated the mechanism of RA through regulating the expression of TNFAIP3.
The signaling pathway of NF-κB is widely participated in human body processes. Preliminary studies have identified that NF-κB is a key marker of RA in inflammation response [7,8]. Tumor necrosis factor-α (TNF-α) plays a critical role in TNFAIP3 suppressing the activity of NF-κB pathway [38]. The present study found that the knockdown of MiR-128-3p significantly decreased the p-IkBα, while due to that IkBα is a key marker in NF-κB pathway, thus we inferred that MiR-128-3p knockdown significantly decreased the activity of NF-κB pathway. Furthermore, knockdown of MiR-128-3p significantly promoted the expression of TNFAIP3, while cells co-transfected with MiR-128-3p inhibitor and si-TNFAIP3 altered the level of p-IkBα, indicating that MiR-128-3p regulated the NF-κB pathway is through regulating the expression of TNFAIP.
Inflammatory response is the main character of RA. From the present study, we found that the inflammatory cytokines IL-6 and IL-17 were significantly increased in T cells of RA, knockdown of MiR-128-3p significantly decreased the levels of IL-6 and IL-17, while cells co-transfected with MiR-128-3p inhibitor and si-TNFAIP3 abolished the effects of MiR-128-3p inhibitor, indicating that MiR-128-3p regulated the inflammatory response of RA is through regulating the expression of TNFAIP. The abnormally activity of T cells is also the main feature of RA [7]. The CD69 and CD25 is the main marker of T cells, thus, to examine the levels of CD69 and CD25 is important for measuring the activity of T cells. Here, we found that the knockdown of MiR-128-3p significantly decreased the ratio of CD69 and CD25, indicating that MiR-128-3p negatively regulated the activity of T cells.
In summary, the present study explored the potential mechanism of MiR-128-3p in RA, with the results of MiR-128-3p were significantly increased, the T cells and the NF-κB signaling pathway were activated, while down-regulating the expression of MiR-128-3p significantly suppressed the activation of T cells and the NF-κB signaling pathway, which was mediated by TNFAIP3, and further relieved the symptom of RA. The present study uncovered the mechanism of MiR-128-3p on RA, which provided great help for RA exploration. However, further study to verify the clinical use of MiR-128-3p for RA therapy is urgent.
Abbreviations
- CRP
C-reactive protein
- DAS28
mean disease activity score in 28 joints
- ELISA
enzyme-linked immunosorbent assay
- ESR
erythrocyte sedimentation rate
- IL
interleukin
- PBMC
peripheral blood mononuclear cell
- RA
rheumatoid arthritis
- RF
rheumatoid factor
- TNFAIP3
tumor necrosis factor-α-induced protein 3
Funding
The present study was supported by grants from National Natural Science Foundation of China [no. 81402936 and 81302576]; Jiangsu Provincial Natural Science Foundation of China [no. BK20131234]; Six talent peak research project in Jiangsu province [no. 2015-WSN-105]; the 333 project of Jiangsu Province, as well as by Jiangsu province science Qiang Wei young medical talents.
Author Contribution
Guoqing Li and Dan Liu designed the project; Zhongbin Xia performed the experiments; Zhongbin Xia and Fanru Meng wrote the paper; Ying Liu and Yuxuan Fang analyzed the statistics; Xia Wu and Chunwang Zhang conducted part of the experiments.
Competing Interests
The authors declare that there are no competing interests associated with the manuscript.
References
- 1.Campbell I.K., Gerondakis S., O’Donnell K. and Wicks I.P. (2000) Distinct roles for the NF-kappaB1 (p50) and c-Rel transcription factors in inflammatory arthritis. J. Clin. Invest. 105, 1799–1806 10.1172/JCI8298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Okamoto H. and Kobayashi A. (2011) Tyrosine kinases in rheumatoid arthritis. J. Inflamm. 8, 21 10.1186/1476-9255-8-21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rodriguez M.S., Egana I., Lopitz-Otsoa F., Aillet F., Lopez-Mato M.P., Dorronsoro A. et al. (2014) The RING ubiquitin E3 RNF114 interacts with A20 and modulates NF-kappaB activity and T-cell activation. Cell Death Dis. 5, e1399 10.1038/cddis.2014.366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mele A., Cervantes J.R., Chien V., Friedman D. and Ferran C. (2014) Single nucleotide polymorphisms at the TNFAIP3/A20 locus and susceptibility/resistance to inflammatory and autoimmune diseases. Adv. Exp. Med. Biol. 809, 163–183 10.1007/978-1-4939-0398-6_10 [DOI] [PubMed] [Google Scholar]
- 5.Yuk J.M., Kim T.S., Kim S.Y., Lee H.M., Han J., Dufour C.R. et al. (2015) Orphan nuclear receptor ERRalpha controls macrophage metabolic signaling and A20 expression to negatively regulate TLR-induced inflammation. Immunity 43, 80–91 10.1016/j.immuni.2015.07.003 [DOI] [PubMed] [Google Scholar]
- 6.Altonsy M.O., Sasse S.K., Phang T.L. and Gerber A.N. (2014) Context-dependent cooperation between nuclear factor kappaB (NF-kappaB) and the glucocorticoid receptor at a TNFAIP3 intronic enhancer: a mechanism to maintain negative feedback control of inflammation. J. Biol. Chem. 289, 8231–8239 10.1074/jbc.M113.545178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang X., Zhu L., Liao Z., Zhang F., Xu L., Xu Y. et al. (2014) Alternative expression pattern of MALT1-A20-NF-kappaB in patients with rheumatoid arthritis. J. Immunol. Res. 2014, 492872 10.1155/2014/492872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Elsby L.M., Orozco G., Denton J., Worthington J., Ray D.W. and Donn R.P. (2010) Functional evaluation of TNFAIP3 (A20) in rheumatoid arthritis. Clin. Exp. Rheumatol. 28, 708–714 [PMC free article] [PubMed] [Google Scholar]
- 9.Chen Z.Z., Zhang X.D., Chen Y. and Wu Y.B. (2017) The role of circulating miR-146a in patients with rheumatoid arthritis treated by Tripterygium wilfordii Hook F. Medicine 96, e6775 10.1097/MD.0000000000006775 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Migita K., Iwanaga N., Izumi Y., Kawahara C., Kumagai K., Nakamura T. et al. (2017) TNF-alpha-induced miR-155 regulates IL-6 signaling in rheumatoid synovial fibroblasts. BMC Res. Notes 10, 403 10.1186/s13104-017-2715-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Filkova M., Aradi B., Senolt L., Ospelt C., Vettori S., Mann H. et al. (2014) Association of circulating miR-223 and miR-16 with disease activity in patients with early rheumatoid arthritis. Ann. Rheum. Dis. 73, 1898–1904 10.1136/annrheumdis-2012-202815 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ham O., Lee C.Y., Song B.W., Lee S.Y., Kim R., Park J.H. et al. (2014) Up-regulation of miR-23b enhances the autologous therapeutic potential for degenerative arthritis by targeting PRKACB in synovial fluid-derived mesenchymal stem cells from patients. Mol. Cells 37, 449–456 10.14348/molcells.2014.0023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stanczyk J., Ospelt C., Karouzakis E., Filer A., Raza K., Kolling C. et al. (2011) Altered expression of microRNA-203 in rheumatoid arthritis synovial fibroblasts and its role in fibroblast activation. Arthritis Rheum. 63, 373–381 10.1002/art.30115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kawano S. and Nakamachi Y. (2011) miR-124a as a key regulator of proliferation and MCP-1 secretion in synoviocytes from patients with rheumatoid arthritis. Ann. Rheum. Dis. 70, i88–i91 10.1136/ard.2010.138669 [DOI] [PubMed] [Google Scholar]
- 15.Semaan N., Frenzel L., Alsaleh G., Suffert G., Gottenberg J.E., Sibilia J. et al. (2011) miR-346 controls release of TNF-alpha protein and stability of its mRNA in rheumatoid arthritis via tristetraprolin stabilization. PLoS One 6, e19827 10.1371/journal.pone.0019827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ogando J., Tardaguila M., Diaz-Alderete A., Usategui A., Miranda-Ramos V., Martinez-Herrera D.J. et al. (2016) Notch-regulated miR-223 targets the aryl hydrocarbon receptor pathway and increases cytokine production in macrophages from rheumatoid arthritis patients. Sci. Rep. 6, 20223 10.1038/srep20223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhao L., Li R., Xu S., Li Y., Zhao P., Dong W. et al. (2018) Tumor suppressor miR-128-3p inhibits metastasis and epithelial-mesenchymal transition by targeting ZEB1 in esophageal squamous-cell cancer. Acta Biochim. Biophys. Sin., 50, 171–180 10.1093/abbs/gmx132 [DOI] [PubMed] [Google Scholar]
- 18.Koh H., Park H., Chandimali N., Huynh D.L., Zhang J.J., Ghosh M. et al. (2017) MicroRNA-128 suppresses paclitaxel-resistant lung cancer by inhibiting MUC1-C and BMI-1 in cancer stem cells. Oncotarget 8, 110540–110551 10.18632/oncotarget.22818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shyamasundar S., Ong C., Yung L.L., Dheen S.T. and Bay B.H. (2017) MiR-128 regulates genes associated with inflammation and fibrosis of rat kidney cells in vitro. Anat. Rec., 301, 913–921, 10.1002/ar.23763 [DOI] [PubMed] [Google Scholar]
- 20.Zhao L., Li R., Xu S., Li Y., Zhao P., Dong W. et al. (2018) Tumor suppressor miR-128-3p inhibits metastasis and epithelial-mesenchymal transition by targeting ZEB1 in esophageal squamous-cell cancer. Acta Biochim. Biophys. Sin. 50, 171–180 10.1093/abbs/gmx132 [DOI] [PubMed] [Google Scholar]
- 21.Murata K., Furu M., Yoshitomi H., Ishikawa M., Shibuya H., Hashimoto M. et al. (2013) Comprehensive microRNA analysis identifies miR-24 and miR-125a-5p as plasma biomarkers for rheumatoid arthritis. PLoS One 8, e69118 10.1371/journal.pone.0069118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dong L., Wang X., Tan J., Li H., Qian W., Chen J. et al. (2014) Decreased expression of microRNA-21 correlates with the imbalance of Th17 and treg cells in patients with rheumatoid arthritis. J. Cell. Mol. Med. 18, 2213–2224 10.1111/jcmm.12353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Piletic K. and Kunej T. (2017) Minimal standards for reporting microRNA:target interactions. OMICS 21, 197–206 10.1089/omi.2017.0023 [DOI] [PubMed] [Google Scholar]
- 24.Desvignes T., Batzel P., Berezikov E., Eilbeck K., Eppig J.T., McAndrews M.S. et al. (2015) miRNA nomenclature: a view incorporating genetic origins, biosynthetic pathways, and sequence variants. Trends Genet. 31, 613–626 10.1016/j.tig.2015.09.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Agarwal V., Bell G.W., Nam J.W. and Bartel D.P. (2015) Predicting effective microRNA target sites in mammalian mRNAs. Elife 4, 10.7554/eLife.05005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li C., Zheng X., Li W., Bai F., Lyu J. and Meng Q.H. (2018) Serum miR-486-5p as a diagnostic marker in cervical cancer: with investigation of potential mechanisms. BMC Cancer 18, 61 10.1186/s12885-017-3753-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Deng L., Lei Q., Wang Y., Wang Z., Xie G., Zhong X. et al. (2017) Down-regulation of miR-221-3p and up-regulation of its target gene PARP1 are prognostic biomarkers for triple negative breast cancer patients and associated with poor prognosis. Oncotarget 8, 108712–108725 10.18632/oncotarget.21561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Koga T., Migita K., Sato T., Sato S., Umeda M., Nonaka F. et al. (2017) MicroRNA-204-3p inhibits lipopolysaccharide-induced cytokines in familial Mediterranean fever via the phosphoinositide 3-kinase gamma pathway. Rheumatology, 57, 718–726, 10.1093/rheumatology/kex451 [DOI] [PubMed] [Google Scholar]
- 29.Ahmadi-Motamayel F., Bayat Z., Hajilooi M., Shahryar-Hesami S., Mahdavinezhad A., Samie L. et al. (2017) Evaluation of the miRNA-146a and miRNA-155 expression levels in patients with oral lichen planus. Iran J. Immunol. 14, 316–324 [PubMed] [Google Scholar]
- 30.Yao L., Ye Y., Mao H., Lu F., He X., Lu G. et al. (2018) MicroRNA-124 regulates the expression of MEKK3 in the inflammatory pathogenesis of Parkinson’s disease. J. Neuroinflammation 15, 13 10.1186/s12974-018-1053-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pankratz F., Hohnloser C., Bemtgen X., Jaenich C., Kreuzaler S., Hoefer I.E. et al. (2017) MicroRNA-100 suppresses chronic vascular inflammation by stimulation of endothelial autophagy. Circ. Res., 122, 417–432, 10.1161/CIRCRESAHA.117.311428 [DOI] [PubMed] [Google Scholar]
- 32.Wang Y., Xu D., Yan S., Zhang L., Wang L., Cai X. et al. (2018) MiR-548a-3p regulates inflammatory response via TLR4/NF-kappaB signaling pathway in rheumatoid arthritis. J. Cell. Biochem. 10.1002/jcb.26659 [DOI] [PubMed] [Google Scholar]
- 33.Hussain N., Zhu W., Jiang C., Xu J., Wu X., Geng M. et al. (2018) Down-regulation of miR-10a-5p in synoviocytes contributes to TBX5-controlled joint inflammation. J. Cell. Mol. Med. 22, 241–250 10.1111/jcmm.13312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mets E., Van Peer G., Van der Meulen J., Boice M., Taghon T., Goossens S. et al. (2014) MicroRNA-128-3p is a novel oncomiR targeting PHF6 in T-cell acute lymphoblastic leukemia. Haematologica 99, 1326–1333 10.3324/haematol.2013.099515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.He Y., Xu L.L., Feng F.E., Wang Q.M., Zhu X.L., Wang C.C. et al. (2018) Mesenchymal stem cell deficiency influences megakaryocytopoiesis through the TNFAIP3/NF-kappaB/SMAD pathway in patients with immune thrombocytopenia. Br. J. Haematol., 180, 395–411 10.1111/bjh.15034 [DOI] [PubMed] [Google Scholar]
- 36.Osako M., Itsumi M., Yamaguchi H., Takeuchi H. and Yamaoka S. (2018) A20 restores phorbol ester-induced differentiation of THP-1 cells in the absence of nuclear factor-kappaB activation. J. Cell. Biochem. 119, 1475–1487 10.1002/jcb.26308 [DOI] [PubMed] [Google Scholar]
- 37.Aksentijevich I. and Zhou Q. (2017) NF-kappaB pathway in autoinflammatory diseases: dysregulation of protein modifications by ubiquitin defines a new category of autoinflammatory diseases. Front. Immunol. 8, 399 10.3389/fimmu.2017.00399 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lee H.R., Choi J., Lee S.H., Cho M.L. and Jue D.M. (2018) Intracelluar delivery of A20 protein inhibits TNFalpha-induced NF-kappaB activation. Protein Expr. Purif. 143, 14–19 10.1016/j.pep.2017.10.005 [DOI] [PubMed] [Google Scholar]