Abstract
Schisandra sphenanthera is widely used as a tonic and restorative in many countries to enhance the function of liver and other organs. Wuzhi tablet (WZ) is a preparation of an ethanol extract of Schisandra sphenanthera. Our previous study demonstrated that WZ exerted a protective effect toward acetaminophen (APAP)-induced hepatotoxicity. However, the molecular mechanisms of this protection remain unclear. This study aimed to determine what molecular pathways contributed to the hepatoprotective effects of WZ against APAP toxicity. Administration of WZ 3 days before APAP treatment significantly attenuated APAP hepatotoxicity in a dose-dependent manner and reduced APAP-induced JNK activation. Treatment with WZ resulted in potent inhibition of CYP2E1, CYP3A11, and CYP1A2 activities and then caused significant inhibition of the formation of the oxidized APAP metabolite N-acetyl-p-benzoquinone imine–reduced glutathione. The expression of NRF2 was increased after APAP and/or WZ treatment, whereas KEAP1 levels were decreased. The protein expression of NRF2 target genes including Gclc, Gclm, Ho-1, and Nqo1 was significantly increased by WZ treatment. Furthermore, APAP increased the levels of p53 and its downstream gene p21 to trigger cell cycle arrest and apoptosis, whereas WZ pretreatment could inhibit p53/p21 signaling to induce cell proliferation-associated proteins including cyclin D1, CDK4, PCNA, and ALR to promote hepatocyte proliferation. This study demonstrated that WZ prevented APAP-induced liver injury by inhibition of cytochrome P450–mediated APAP bioactivation, activation of the NRF2–antioxidant response element pathway to induce detoxification and antioxidation, and regulation of the p53, p21, cyclin D1, CDK4, PCNA, and ALR to facilitate liver regeneration after APAP-induced liver injury.
Introduction
Acetaminophen (APAP)-induced hepatotoxicity is a leading cause of drug-induced liver injury and a persistent important public health concern in many countries (Larson et al., 2005). Metabolic activation of APAP to N-acetyl-p-benzoquinone imine (NAPQI) by cytochrome P450 enzymes is the critical initiating event of hepatic toxicity. Excess NAPQI depletes glutathione (GSH) and subsequently results in covalent binding of NAPQI with cellular proteins, which triggers initiation of the injury process with mitochondrial oxidative stress, ultimately leading to apoptosis and hepatocellular necrosis (Saito et al., 2010).
NRF2 regulates the expression of a battery of cytoprotective genes encoding intracellular detoxifying enzymes and antioxidant proteins, which are responsible for GSH synthesis, antioxidant defense system, conjugation, transport, and excretion of the metabolites through the antioxidant response element (ARE). NRF2 plays a critical role in the multiple steps related to the alleviation of APAP toxicity and thus is identified as a protective target resistant to liver injury (Gum and Cho, 2013). Furthermore, liver repair is a critical determinant of final outcome of toxicant-induced injury (Diehl and Chute, 2013). p53 is a tumor suppressor that plays an important role in regulating cell growth, DNA repair, and apoptosis (Shams et al., 2013). Under severe DNA damage by APAP overdose, p53 is activated to inhibit cell proliferation or trigger cell apoptosis (Zhao et al., 2012). Thus, downregulation of p53 level may serve to promote hepatocyte proliferation and liver repair after APAP-induced liver injury. Taken together, inhibition of APAP metabolic activation, alleviation in oxidative stress, and promotion in liver regeneration are important mechanisms involved in treatment of APAP-induced liver injury (Reid et al., 2005). Any drug and protective/therapeutic interventions targeting these molecular pathways will substantially protect or even completely eliminate APAP toxicity.
Schisandra sphenanthera, the dried ripe fruit of Schisandra sphenanthera Rehd. et Wils, has been widely used as a tonic and restorative in Asia and is now indexed in the Pharmacopoeia of China. Schisandra sphenanthera is capable of improving the body’s resistance to disease, stress, and other debilitating processes. Its adaptogenic action may help stimulate immune defenses, increase physical working capacity, boost recovery after surgery, normalize blood sugar and blood pressure, and afford a stress-protective effect against radiation, motion sickness, and aseptic inflammation (Panossian and Wikman, 2008). Moreover, Schisandra sphenanthera was shown to possess numerous pharmacological activities such as hepatoprotective effects against viral, chemical hepatitis and various hepatotoxins and improvement of kidney and heart function (Zhu et al., 2000; Xie et al., 2010). Wuzhi tablet (WZ), containing 7.5 mg Schisantherin A per tablet, is a preparation of an ethanol extract of Schisandra sphenanthera and widely used in the clinical practice in China to protect liver function in chronic hepatitis and liver dysfunction patients. Chemical fingerprint of WZ was determined by high performance liquid chromatography–diode array detector analysis in our previous study and its major active lignans include schisandrin A, schisandrin B, schisandrin C, schisandrol A, schisandrol B, and schisantherin A (Qin et al., 2014). The relative amounts of the six active lignans in WZ extract were compared and Schisantherin A and Schisandrin A were the two highest bioactive lignans in WZ, and other lignans were much lower than the above contents. Recently, WZ was found to have protective effects against APAP hepatotoxicity (Bi et al., 2013). Although the role of WZ in maintenance of normal fatty acid metabolism was identified as contributing to its protective effects against APAP-induced liver injury, many aspects of the molecular mechanisms underlying the hepatoprotective effect of WZ remain largely unknown. Therefore, the aim of the present study was to determine whether WZ exhibited an inhibitory effect on cytochrome P450 (P450)–mediated metabolic activation of APAP and whether WZ could regulate the NRF2-ARE and p53/p21 signaling pathway to prevent oxidative stress and promote liver regeneration.
Materials and Methods
Reagents.
Wuzhi tablets were manufactured by Fanglue Pharmaceutical Company (Guangxi, China), which has been quantified to 7.5 mg Schisantherin A per tablet. NADPH tetrasodium salt was obtained from AppliChem (Darmstadt, Germany). Acetaminophen, phenacetin, chlorzoxazone, 6-hydroxychlorzoxazone, nifedipine, dehydronifedipine, 4-methylpyrazole, α-naphthoflavone, ketoconazole, and reduced l-glutathione were obtained from Sigma-Aldrich (St. Louis, MO). Total JNK, p-JNK, SIRT1, p-p38, and glyceraldehyde-3-phosphate dehydrogenase antibodies for Western blot analysis were from Cell Signaling Technology (Danvers, MA). Antibodies against GCLC, p53, UGT1A1, and GCLM were obtained from Abcam (Cambridge, UK). CYP3A11, NRF2, NQO1, HO-1, and p21 antibodies were from Santa Cruz Biotechnology (Santa Cruz, CA). CYP1A2 and CYP2E1 antibodies were purchased from Boster Biotechnology Co., Ltd. (Wuhang, China). All other antibodies including p38, KEAP1, PCNA, cyclin D1, CDK4, and ALR were provided by Shanghai Sangon Biotech Co., Ltd. (Shanghai, China). The secondary antibody was obtained from Cell Signaling Technology.
Animals.
Male C57BL/6 mice (6–8 weeks old) were obtained from the Laboratory Animal Center of Sun Yat-sen University (Guangzhou, China) and maintained under controlled conditions (22–24°C, 55–60% humidity and 12-hour light/dark cycle) with free access to standard food and water. All procedures were in accordance with the Regulations of Experimental Animal Administration issued by the Ministry of Science and Technology of the People’s Republic of China (http://www.most.gov.cn). All animal protocols were approved by the Ethics Committee on the Care and Use of Laboratory Animals of Sun Yat-sen University.
Mice were randomly divided into six groups: 1) untreated control group, 2) WZ (700 mg/kg)-treated group, 3) APAP-treated group, 4) WZ (175 mg/kg)/APAP-treated group, 5) WZ (350 mg/kg)/APAP-treated group, and 6) WZ (700 mg/kg)/APAP-treated group. WZ and APAP solutions were prepared as described previously (Bi et al., 2013). Mice were administered WZ (175, 350, or 700 mg/kg) or saline solution by gavage seven times with an interval of 12 hours for 3 consecutive days. All animals were fasted overnight before APAP administration. Fifteen minutes after the last dose of WZ, a single dose of 400 mg/kg APAP was given by intraperitoneal injection. All mice were killed at 6 hours after APAP treatment. Serum samples and liver tissues were harvested, a portion of the liver was immediately fixed in 10% buffered formalin for histology, and the remaining tissues were flash frozen in liquid nitrogen and stored at −80°C for further use.
Histologic and Biochemical Assessment.
Liver tissues fixed in neutral buffered formalin were embedded in paraffin, cut into 3-µm-thick sections and stained with H&E according to a standard protocol. H&E-stained liver sections were examined using a LEICA DM5000B microscope (Leica, Heidelberg, Germany) and used for necrosis scoring.
Serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were determined to evaluate APAP-induced liver injury with a commercial AST or ALT assay kit (Kefang Biotech, Guangzhou, China) on a Beckman Synchron CX5 Clinical System. Oxidative stress induced by APAP was assessed by measuring hydrogen peroxide (H2O2) and malondialdehyde (MDA) levels in liver using commercially-available kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China).
Measurement of Liver CYP450 Enzyme Activity.
The effect of WZ on the activities of CYP450 isoenzymes responsible for APAP oxidation including CYP2E1, CYP1A2, and CYP3A11 (mouse CYP3A11 is a homolog to human CYP3A4) was investigated in mouse liver microsomes using a liquid chromatography–tandem mass spectrometry (LC-MS/MS)-based substrate “cocktail” incubation approach as previously described (He et al., 2007). Various classic inhibitors for P450 enzyme including 4-methylpyrazole (at 5 μM for CYP2E1), α-naphthoflavone (at 0.2 μM for CYP1A2), and ketoconazole (at 1.1 mM for CYP3A11) were used for validation of the developed method. Various concentrations of WZ extract from 0.0625 to 1 mg/ml and the mixed P450 probe substrates (phenacetin for CYP1A2, chlorzoxazone for CYP2E1, and nifedipine for CYP3A11) were “cocktail” incubated with liver microsomes. The metabolites (acetaminophen/6-hydroxychlorzoxazone/dehydronifedipine) of these three CYPs probe substrates in all samples were determined using previously developed LC-MS/MS methods.
ultra performance liquid chromatography–MS/MS analysis of N-acetyl-p-benzoquinone imine–reduced glutathione.
The oxidized APAP intermediate NAPQI, formed in mice microsomal incubation system, was trapped by reduced glutathione (GSH) and determined by an ultra performance liquid chromatography–MS/MS method. The incubation mixture of 150 μl contained 0.5 mM APAP, 5 mM GSH, WZ extract (0.0625, 0.125, 0.25, 0.5, 1 mg/ml), mouse liver microsomes (containing 30 μg protein), 0.4 mM NADPH, and potassium phosphate buffer (50 mM, pH 7.4). The NADPH was added to the incubation system to initiate the reaction. After incubating for 10 minutes at 37°C, the reaction was quenched by adding 300 μl ice-cold acetonitrile. The reaction mixtures were centrifuged at 16,000g for 15 minutes, and the supernatant was used for analysis of NAPQI-GSH conjugate formation. The sample was analyzed using Ultimate 3000 ultra performance liquid chromatography system (Dionex Corporation, Sunnyvale, CA) coupled with triple quadrupole mass spectrometer (TSQ Quantum Access, Thermo Fisher Scientific, Waltham, MA). Chromatographic separations were performed on Hypurity C18 5 μm column (150 × 2.1 mm, Thermo Fisher Scientific). The samples were eluted with 2% acetonitrile (v/v in water) and 0.1% (v/v) formic acid at a flow rate of 0.5 ml/min. Electrospray positive ionization mode was used for NAPQI-GSH analysis. Selective reaction monitoring experiments using m/z 457.1→328.1 was performed to profile and quantitate NAPQI-GSH.
Western Blot Analysis.
Liver whole cell, nuclear, cytosolic, or mitochondrial extracts were prepared from frozen liver tissues, and protein concentration was determined using Bradford method. Equivalent amounts of protein extracts were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then blotted onto polyvinylidene fluoride membranes (Millipore, Bedford, MA). Phosphorylated proteins were blocked with 5% bovine serum albumin in Tris-buffered saline, and other proteins were blocked with 5% non-fat milk in Tris-buffered saline. Membranes were incubated overnight at 4°C with primary antibodies, followed by secondary antibodies conjugated with horseradish peroxidase for 1 hour at room temperature. Protein-antibody complexes were detected using an electrochemiluminescence kit (Engreen Biosystem, Beijing, China) and exposed to an X-ray film (GE Healthcare, Piscataway, NJ). The intensity of protein bands was analyzed using ImageJ software (National Institutes of Health, Bethesda, MD).
Statistical Analysis.
Data are expressed as the mean ± S.E.M. To determine statistically significant difference between groups, two-tailed unpaired Student’s t test was carried out using GraphPad Prism 5 (GraphPad Software Inc., San Diego, CA). P value less than 0.05 was considered statistically significant.
Results
Pretreatment with WZ Dose-Dependently Protects against APAP-Induced Hepatotoxicity in Mice.
As expected, 400 mg/kg APAP treatment for 6 hours resulted in massive hepatic toxicity in mice as indicated by histopathological analysis of H&E-stained liver sections (Fig. 1A), increased ALT and AST activities (Fig. 1B), and necrosis scoring (Fig. 1C). The livers of APAP-treated mice displayed large area of centrilobular hepatocellular necrosis that extended into the hepatic lobule, whereas mice pretreated with 350 or 700 mg/kg WZ exhibited only a small, localized injury and necrosis that did not extensively radiate throughout the hepatic lobule. The ALT and AST levels were increased in parallel to the area of hepatic necrosis. Pretreatment with WZ for 3 consecutive days significantly attenuated the increase of ALT and AST levels by APAP. However, low doses of WZ were unable to protect against APAP toxicity. Taken together, these data indicated that WZ had a marked protective effect against APAP-induced liver injury in a dose-dependent manner.
Fig. 1.
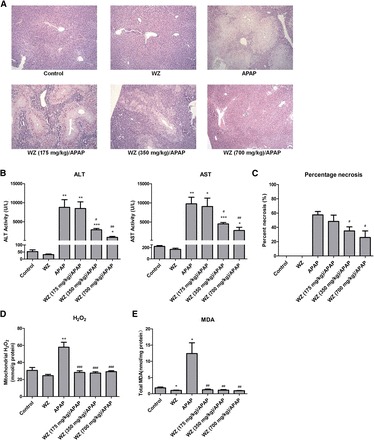
Pretreatment with WZ protects mice against APAP-induced hepatotoxicity. (A) H&E-stained liver sections, (B) serum ALT and AST activities, (C) percentage necrosis, (D) mitochondrial H2O2 levels, (E) total MDA levels from mice in each experimental group (n = 6). *P < 0.05, **P < 0.01, ***P < 0.001 versus control mice; #P < 0.05, ##P < 0.01, ###P < 0.001 versus APAP-treated mice.
Pretreatment with WZ Blocks APAP-Induced Oxidative Stress.
A rapid increase in mitochondrial H2O2 and hepatic MDA levels was observed after APAP challenge (Fig. 1, D and E) due to overproduction of reactive oxygen species and formation of lipid peroxidation. This aberrant increase was completely restored by WZ pretreatment, suggesting WZ could block APAP-induced oxidative stress. Compared with the control, WZ also exerted an effect on decreasing levels of both mitochondrial H2O2 and total MDA in uninjured livers.
Activation of JNK, a crucial signaling component of APAP-induced hepatotoxicity, was observed in APAP-treated mice, whereas WZ pretreatment suppressed APAP-induced phosphorylation of JNK. Compared with JNK1, JNK2 appeared to be markedly inactivated by WZ upon APAP exposure (Fig. 2). Furthermore, APAP treatment for 6 hours showed no effect on activation of p38 MAPK. However, the levels of phosphorylated p38 MAPK were significantly higher in liver of WZ-treated and WZ/APAP-treated mice compared with those of control and APAP-treated mice, indicating that WZ could activate p38 MAPK.
Fig. 2.
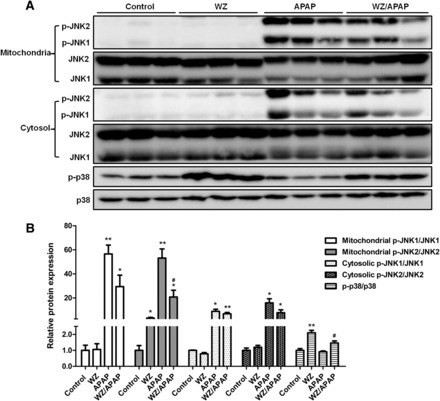
Pretreatment with WZ inactivates JNK, but activates p38 MAPK after APAP challenge. (A) Western blot analysis of p-JNK, JNK, p-p38, and p38 levels in livers from control, WZ (700 mg/kg)-treated, APAP-treated, WZ (700 mg/kg)/APAP-treated mice. (B) Densitometric analysis of Western blots (n = 3). *P < 0.05, **P < 0.01 versus control mice; #P < 0.05 versus APAP-treated mice.
WZ Inhibits the Activities of CYP450 Isoforms and NAPQI Formation.
The effects of WZ on the expression and enzymatic activities of CYP2E1, CYP1A2, and CYP3A11 in mice were next investigated. Expression of CYP2E1, CYP1A2 and CYP3A11 proteins was decreased after APAP challenge, but WZ pretreatment blocked this decline in P450 expression (Fig. 3, A and B). In addition, mice in the WZ-treated group exhibited a marked upregulation of CYP3A11 expression and slight downregulation of CYP1A2 levels, indicating that a component of this extract might be a pregnane X receptor (PXR) or constitutive androstane activator. WZ also exerted marked inhibitory effects on the activities of CYP2E1, CYP1A2, and CYP3A11 in a concentration-dependent manner, most notably CYP3A11 (Fig. 3C). Compared with controls, the activities of CYP2E1, CYP3A11, and CYP1A2 after treatment with 1 mg/ml WZ were reduced by 79, 86, and 40%, respectively. To further identify the effect of WZ on APAP bioactivation, the levels of NAPQI were determined. Pretreatment with WZ significantly inhibited the formation of the oxidized APAP metabolites, as evidenced by concentration-dependent decrease of NAPQI-GSH by WZ (Fig. 3D).
Fig. 3.
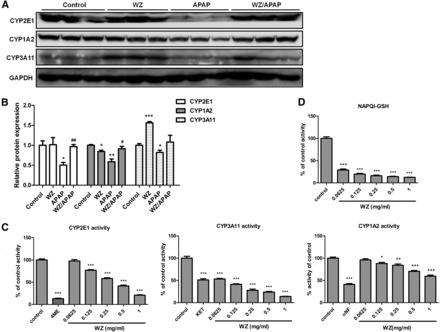
WZ possesses marked inhibitory effects on the activities of P450 enzymes and NAPQI-GSH formation. (A) Western blot analysis of CYP2E1, CYP1A2, CYP3A11, and glyceraldehyde-3-phosphate dehydrogenase levels in livers from control, WZ (700 mg/kg)-treated, APAP-treated, WZ (700 mg/kg)/APAP-treated mice. (B) Densitometric analysis of Western blots (n = 3). (C) Effect of WZ on the activities of CYP2E1, CYP1A2, CYP3A11 enzymes in mouse liver microsomes (n = 5). (D) Effect of WZ on NAPQI-GSH formation in mouse liver microsomes (n = 5). *P < 0.05, **P < 0.01, ***P < 0.001 versus control mice; #P < 0.05, ##P < 0.01 versus APAP-treated mice.
WZ Activates NRF2-ARE Signaling Pathway to Induce Detoxification and Antioxidant Effects.
The expression levels of proteins involved in NRF2-ARE pathway were measured to explore whether WZ activated this pathway to counteract APAP toxicity. The data indicated that KEAP1 expression was inhibited after WZ and/or APAP treatment compared with that in control mice (Fig. 4). NRF2 protein levels were increased after WZ pretreatment in uninjured and APAP-induced injured livers. APAP treatment resulted in decreased GCLC, GCLM, HO-1, NQO1, and UGT1A1 protein levels, which were significantly reversed by WZ. WZ alone could slightly upregulate GCLC, GCLM, HO-1, and UGT1A1 protein expression in control mice. These results revealed that activation of the NRF2-ARE signaling pathway by WZ might serve to limit APAP-induced liver damage.
Fig. 4.
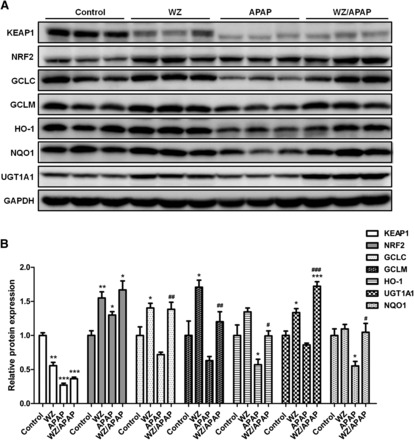
WZ activates NRF2-ARE signaling pathway in mice. (A) Western blot analysis of KEAP1, NRF2, GCLC, GCLM, HO-1, NQO1, UGT1A1, and glyceraldehyde-3-phosphate dehydrogenase proteins in livers from control, WZ (700 mg/kg)-treated, APAP-treated, WZ (700 mg/kg)/APAP-treated mice. (B) Densitometric analysis of Western blots (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001 versus control mice; #P < 0.05, ##P < 0.01, ###P < 0.001 versus APAP-treated mice.
WZ Suppresses p53/p21-Mediated Cell Cycle Arrest to Facilitate Liver Regeneration.
p53 and p21 levels were then investigated to explore whether WZ could block p53/p21 signaling to promote liver repair in mice after APAP-induced toxicity. p53 level was low or undetectable in control mice, and treatment with 400 mg/kg APAP significantly upregulated p53 protein levels, together with p21 (Fig. 5). However, the increase of p53 and p21 in APAP-treated mice was attenuated by WZ. Levels of cell cycle regulatory proteins cyclin D1 and CDK4 were decreased after APAP treatment, whereas cyclin D1 was markedly reversed by WZ pretreatment. Additionally, cyclin D1 was upregulated in WZ-treated mice compared with that of control mice. PCNA was strongly induced by WZ, and thus significantly increased after a decline caused by APAP in WZ/APAP-treated mice. Although WZ induced ALR expression in normal livers, decreased ALR caused by APAP was not restored to baseline levels in WZ/APAP-treated mice. Furthermore, WZ pretreatment before APAP challenge slightly increased SIRT1 protein levels to promote p53 degradation. These results suggest that WZ pretreatment prevented cell cycle arrest and cell apoptosis during APAP-induced liver injury through inhibiting the p53/p21 signaling to facilitate cell cycle progression and hepatocyte proliferation.
Fig. 5.
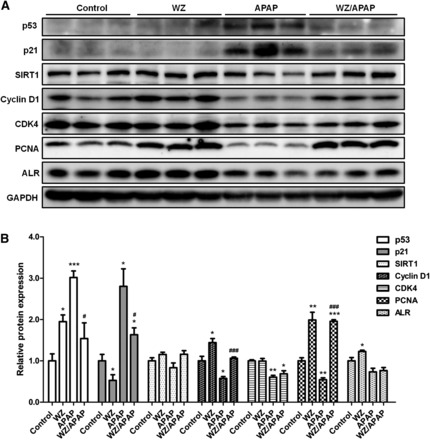
WZ suppresses p53/p21 signaling pathway in mice. (A) Western blot analysis of p53, p21, SIRT1, cyclin D1, CDK4, PCNA, ALR, and glyceraldehyde-3-phosphate dehydrogenase proteins in livers from control, WZ (700 mg/kg)-treated, APAP-treated, WZ (700 mg/kg)/APAP-treated mice. (B) Densitometric analysis of Western blots (n = 3). *P < 0.05, **P < 0.01, ***P < 0.001 versus control mice; #P < 0.05, ###P < 0.001 versus APAP-treated mice.
Discussion
The present study revealed a significant protective effect of WZ on APAP-induced liver injury and provided direct evidence for mechanisms underlying WZ’s protection against APAP toxicity. When moderate and high doses of WZ were administered before a toxic dose of APAP, mice were protected against APAP hepatotoxicity, as revealed by much less liver damage and lower serum AST and ALT levels. Furthermore, livers from mice pretreated with WZ before APAP challenge exhibited reduced oxidative stress with lower mitochondrial H2O2 and hepatic MDA levels compared with livers from mice treated for 6 hours with 400 mg/kg APAP. Our previous study indicated that WZ not only increased GSH levels in control mice but also partially reversed the decrease of hepatic GSH by APAP treatment with a slight increase (Bi et al., 2013). Evidence for decreased oxidative stress upon WZ pretreatment was also indicated by reduced activation of JNK. These results demonstrated that WZ possessed an antioxidant effect that likely contributed to its protective effect against APAP-induced liver injury.
APAP bioactivation is the crucial initiating step of liver injury. It is known that the reactive metabolite NAPQI derived from APAP is generated by P450-catalyzed oxidation mainly involving CYP2E1, CYP3A4, and CYP1A2 (Zaher et al., 1998; Wolf et al., 2007). Previous reports showed that Schisandra chinensis extract and its lignans activated PXR and induced the expression of CYP3A and CYP2C by reporter gene assays in primary hepatocyte cultures (Mu et al., 2006), which was consistent with our observations. Activation of PXR would enhance APAP-induced hepatic toxicity (Guo et al., 2004). In the current study, a marked inhibition by WZ of the CYP3A11, CYP2E1, and CYP1A2 enzymatic activities was observed, despite induction of their genes and increased protein expression by WZ administration. Several previous reports demonstrated that WZ significantly inhibited the CYP3A-mediated metabolism and the P-gp–mediated efflux contributing to increase the blood concentration of tacrolimus, paclitaxel, and cyclosporin A in rats (Qin et al., 2010; Jin et al., 2011; Xue et al., 2013). Schisandra lignan extract was found to exhibit a mechanism-based inhibition toward CYP3A in previous reports (Iwata et al., 2004; Lai et al., 2009), that is, Schisandra lignan extract caused irreversible inhibition of CYP3A until this enzyme was newly synthesized. Furthermore, the present observation is supported by similar studies of St. John’s wort, an extract from the flowers of the perennial plant Hypericum perforatum L, which indicates that St. John’s wort has inhibitory effects on the activity of enzymes/transporters but induces expression of drug-metabolizing enzymes and transporters (Xie and Kim, 2005). Enzymatic activity is regarded as an important determinant of final outcome of drug-interactions, either inhibition or induction of drug metabolism, compared with upstream gene and protein expression of metabolizing enzymes. The protective effect of WZ against APAP-induced liver injury may be attributed to the dominating inhibitory effects of CYP450 enzymes responsible for APAP metabolic activation over PXR activation and the induction effects on CYP450 enzymes, which is supported by the lower NAPQI formation after WZ pretreatment.
UGT1A1 is an important UDP-glucuronosyltransferase involved in the glucuronidation of APAP. Increased UGT1A1 expression by WZ may result in more APAP conjugated and eliminated. Additionally, mRNA expression of Ugt1a1, Ugt1a6, Ugt1a9, and Sult1a1 was significantly induced by WZ in uninjured livers (data not shown). Compared with APAP-treated mice, mice pretreated with WZ exhibited a dose-dependent increase in Ugt1a1, Ugt1a9, and Sult1a1 mRNA levels after APAP challenge. Furthermore, WZ not only significantly upregulated mRNA expression of Abcc2, Abcc3, and Abcc4 in uninjured livers but also reversed decline of Abcc2 and Abcc3 levels by APAP treatment (data not shown). Taken together, significant inhibition of P450s and upregulation of UGTs, sulfotransferases, and multidrug resistance–associated proteins by WZ are likely responsible for reducing NAPQI formation and thus contribute to the hepatic protection of WZ against APAP-induced hepatic toxicity.
Extensive studies have demonstrated that activation of the NRF2 signaling pathway serves to protect animals against liver injury produced by various hepatotoxicants including APAP (Enomoto et al., 2001). Glutamate cysteine ligase, composed of a catalytic (GCLC) and a modifier (GCLM) subunit, catalyzes the rate-limiting step in GSH biosynthesis, which is critical for the maintenance of cellular redox homeostasis and detoxification of electrophiles, including NAPQI. HO-1 is responsible for catabolism of heme released from drug metabolizing enzymes to protect the cell against oxidative injury, which is identified as a potential therapeutic target for hepatoprotection (Saberi et al., 2014). NQO1, an enzyme with antioxidant properties, catalyzes the reduction of cellular quinones. Recent studies have indicated that NQO1 possesses the capacity to enzymatically reduce NAPQI to amelioriate APAP toxicity (Moffit et al., 2007). Several studies revealed that some lignans isolated from fruits of Schisandra chinensis, including Schisandrin B, α-iso-cubebenol, and Tigloylgomisin H, exhibited anti-inflammatory activities and potential liver cancer prevention effects through activating NRF2 pathway (Lee et al., 2009; Park et al., 2011; Checker et al., 2012). However, the content of the above lignans is low in WZ tablets. Whether WZ induces NRF2-ARE signaling to counteract APAP toxicity has not been determined. Indeed, WZ pretreatment inhibited KEAP1 expression, and thus NRF2 is released from KEAP1 to translocate into the nucleus to activate downstream antioxidant target gene expression. APAP treatment decreased GCLC, GCLM, HO-1, and NQO1 expression, which was significantly reversed by WZ. Thus, coordinated regulation of detoxification enzymes and antioxidant genes by NRF2 during APAP hepatotoxicity is a mechanism by which hepatocytes may limit intracellular accumulation of APAP and oxidative damage. Mice pretreated with WZ are resistant to APAP hepatotoxicity by activating the NRF2-ARE pathway to induce detoxification and inhibit the deleterious effects of reactive oxygen species.
Additionally, previously published studies demonstrated that p38 MAPK triggered activation of NRF2 resulting in induction of the Ho-1 gene (Jang et al., 2012; Park et al., 2013), suggesting that WZ pretreatment seemed to activate NRF2-ARE pathway via p38 MAPK. ALR is identified to promote liver regeneration after partial hepatectomy and to augment hepatocyte proliferation (Polimeno et al., 2011). Recent studies have revealed that ALR is one of the ARE-regulated genes that can be induced by NRF2 activation (Dayoub et al., 2013). WZ upregulated the protein expression of ALR but was unable to prevent the decline of ALR protein levels by APAP treatment for 6 hour.
Another notable finding was that expression of p53 and p21 was significantly induced after APAP challenge, which may lead to inhibition of cell cycle activation as indicated by decreased CDK4, cyclin D1, PCNA, and ALR expression. WZ pretreatment inhibited p53 and p21 expression and thus induced cyclin D1 and PCNA levels. p53 is stabilized by multiple posttranslational modifications including acetylation and phosphorylation. p53, as a known target of SIRT1-mediated deacetylation, is regulated by SIRT1, which leads to mouse double minute 2 homolog–mediated p53 degradation (Langley et al., 2002). Very recently, SIRT1 was found to regulate the regenerative response in the liver through activation of the farnesoid X receptor and mammalian target of rapamycin signaling pathways (García-Rodríguez et al., 2014). This suggests that SIRT1 may be a potential target on cell cycle progression after APAP challenge. However, whether SIRT1 expression is affected by APAP and WZ is not yet reported. Decrease in SIRT1 levels followed by elevated p53 and p21 levels was observed upon APAP exposure, whereas SIRT1 levels were increased by WZ pretreatment. Moreover, JNK can activate p53 and then stabilize p53 via phosphorylation, leading to reduced ubiquitination and proteasomal degradation (Fuchs et al., 1998). Taken together, WZ pretreatment resulted in prevention of injury progression, paving the way for liver repair to take over and restore liver function.
Several studies have shown that acute liver injury induced by moderate doses of APAP is a dynamic process including initiation of injury, progression of injury, and compensatory liver repair (Mehendale, 2005; Bajt et al., 2008; Fan et al., 2014). In the present study, WZ suppresses NAPQI generation required for initiating cellular toxicity through inhibition of P450 enzyme activities and induction of UGT, sulfotransferase, and multidrug resistance–associated protein expression and prevents oxidative stress induced progression of liver injury by activating the NRF2-ARE pathway. Importantly, WZ can promote hepatocyte proliferation via regulating p53/p21 signaling, which will be beneficial to liver regeneration at later stage of APAP-induced liver injury. These results indicate the protective effects of WZ occur throughout the whole process of APAP-induced hepatotoxicity. Whether WZ could rescue APAP toxicity after ingestion will be further investigated.
Collectively, the current study clearly demonstrated that WZ prevented APAP-induced liver injury by inhibition of P450-mediated APAP metabolic activation, activation of the NRF2-ARE pathway to induce detoxification and antioxidation, and regulation of p53/p21-mediated cell cycle to facilitate liver regeneration after APAP-induced liver injury.
Abbreviations
- ALR
augmenter of liver regeneration
- ALT
alanine aminotransferase
- APAP
acetaminophen
- ARE
antioxidant response element
- AST
aspartate aminotransferase
- GCLC
glutamate-cysteine ligase catalytic subunit
- GCLM
glutamate-cysteine ligase modifier subunit
- GSH
reduced glutathione
- LC-MS/MS
liquid chromatography–tandem mass spectrometry
- MDA
malondialdehyde
- NAPQI
N-acetyl-p-benzoquinone imine
- P450
cytochrome P450
- PCNA
proliferating cell nuclear antigen
- PXR
pregnane X receptor
- WZ
Wuzhi tablet
Authorship Contributions
Participated in research design: Fan, Huang, Gonzalez, Bi.
Conducted experiments: Fan, Jiang, Yo. Wang, Tan, Zeng, Yi. Wang, Chen.
Performed data analysis: Fan, Jiang, Qu.
Wrote or contributed to the writing of the manuscript: Fan, Gonzalez, Bi.
Footnotes
This work was supported by the Natural Science Foundation of China [Grants 81373470, 81320108027], the Opening Project of Guangdong Provincial Key Laboratory of New Drug Design and Evaluation [Grant 2011A060901014], and the Fundamental Research Fund for the Central Universities [No. 13ykpy08].
References
- Bajt ML, Yan HM, Farhood A, Jaeschke H. (2008) Plasminogen activator inhibitor-1 limits liver injury and facilitates regeneration after acetaminophen overdose. Toxicol Sci 104:419–427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bi H, Li F, Krausz KW, Qu A, Johnson CH, Gonzalez FJ. (2013) Targeted Metabolomics of Serum Acylcarnitines Evaluates Hepatoprotective Effect of Wuzhi Tablet (Schisandra sphenanthera Extract) against Acute Acetaminophen Toxicity. Evid Based Complement Alternat Med 2013:985257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Checker R, Patwardhan RS, Sharma D, Menon J, Thoh M, Bhilwade HN, Konishi T, Sandur SK. (2012) Schisandrin B exhibits anti-inflammatory activity through modulation of the redox-sensitive transcription factors Nrf2 and NF-κB. Free Radic Biol Med 53:1421–1430. [DOI] [PubMed] [Google Scholar]
- Dayoub R, Vogel A, Schuett J, Lupke M, Spieker SM, Kettern N, Hildt E, Melter M, Weiss TS. (2013) Nrf2 activates augmenter of liver regeneration (ALR) via antioxidant response element and links oxidative stress to liver regeneration. Mol Med 19:237–244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diehl AM, Chute J. (2013) Underlying potential: cellular and molecular determinants of adult liver repair. J Clin Invest 123:1858–1860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enomoto A, Itoh K, Nagayoshi E, Haruta J, Kimura T, O’Connor T, Harada T, Yamamoto M. (2001) High sensitivity of Nrf2 knockout mice to acetaminophen hepatotoxicity associated with decreased expression of ARE-regulated drug metabolizing enzymes and antioxidant genes. Toxicol Sci 59:169–177. [DOI] [PubMed] [Google Scholar]
- Fan X, Chen P, Tan H, Zeng H, Jiang Y, Wang Y, Wang Y, Hou X, Bi H, Huang M. (2014) Dynamic and coordinated regulation of KEAP1-NRF2-ARE and p53/p21 signaling pathways is associated with acetaminophen injury responsive liver regeneration. Drug Metab Dispos 42:1532–1539. [DOI] [PubMed] [Google Scholar]
- Fuchs SY, Adler V, Pincus MR, Ronai Z. (1998) MEKK1/JNK signaling stabilizes and activates p53. Proc Natl Acad Sci USA 95:10541–10546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García-Rodríguez JL, Barbier-Torres L, Fernández-Álvarez S, Gutiérrez-de Juan V, Monte MJ, Halilbasic E, Herranz D, Álvarez L, Aspichueta P, Marín JJG, et al. (2014) SIRT1 controls liver regeneration by regulating bile acid metabolism through farnesoid X receptor and mammalian target of rapamycin signaling. Hepatology 59:1972–1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gum SI, Cho MK. (2013) Recent updates on acetaminophen hepatotoxicity: the role of nrf2 in hepatoprotection. Toxicol Res 29:165–172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo GL, Moffit JS, Nicol CJ, Ward JM, Aleksunes LA, Slitt AL, Kliewer SA, Manautou JE, Gonzalez FJ. (2004) Enhanced acetaminophen toxicity by activation of the pregnane X receptor. Toxicol Sci 82:374–380. [DOI] [PubMed] [Google Scholar]
- He F, Bi HC, Xie ZY, Zuo Z, Li JK, Li X, Zhao LZ, Chen X, Huang M. (2007) Rapid determination of six metabolites from multiple cytochrome P450 probe substrates in human liver microsome by liquid chromatography/mass spectrometry: application to high-throughput inhibition screening of terpenoids. Rapid Commun Mass Spectrom 21:635–643. [DOI] [PubMed] [Google Scholar]
- Iwata H, Tezuka Y, Kadota S, Hiratsuka A, Watabe T. (2004) Identification and characterization of potent CYP3A4 inhibitors in Schisandra fruit extract. Drug Metab Dispos 32:1351–1358. [DOI] [PubMed] [Google Scholar]
- Jang HJ, Kim YM, Tsoyi K, Park EJ, Lee YS, Kim HJ, Lee JH, Joe Y, Chung HT, Chang KC. (2012) Ethyl pyruvate induces heme oxygenase-1 through p38 mitogen-activated protein kinase activation by depletion of glutathione in RAW 264.7 cells and improves survival in septic animals. Antioxid Redox Signal 17:878–889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin J, Bi H, Hu J, Zeng H, Zhong G, Zhao L, Huang Z, Huang M. (2011) Effect of Wuzhi tablet (Schisandra sphenanthera extract) on the pharmacokinetics of paclitaxel in rats. Phytother Res 25:1250–1253. [DOI] [PubMed] [Google Scholar]
- Lai L, Hao H, Wang Q, Zheng C, Zhou F, Liu Y, Wang Y, Yu G, Kang A, Peng Y, et al. (2009) Effects of short-term and long-term pretreatment of Schisandra lignans on regulating hepatic and intestinal CYP3A in rats. Drug Metab Dispos 37:2399–2407. [DOI] [PubMed] [Google Scholar]
- Langley E, Pearson M, Faretta M, Bauer UM, Frye RA, Minucci S, Pelicci PG, Kouzarides T. (2002) Human SIR2 deacetylates p53 and antagonizes PML/p53-induced cellular senescence. EMBO J 21:2383–2396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larson AM, Polson J, Fontana RJ, Davern TJ, Lalani E, Hynan LS, Reisch JS, Schiødt FV, Ostapowicz G, Shakil AO, et al. Acute Liver Failure Study Group (2005) Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology 42:1364–1372. [DOI] [PubMed] [Google Scholar]
- Lee SB, Kim CY, Lee HJ, Yun JH, Nho CW. (2009) Induction of the phase II detoxification enzyme NQO1 in hepatocarcinoma cells by lignans from the fruit of Schisandra chinensis through nuclear accumulation of Nrf2. Planta Med 75:1314–1318. [DOI] [PubMed] [Google Scholar]
- Mehendale HM. (2005) Tissue repair: an important determinant of final outcome of toxicant-induced injury. Toxicol Pathol 33:41–51. [DOI] [PubMed] [Google Scholar]
- Moffit JS, Aleksunes LM, Kardas MJ, Slitt AL, Klaassen CD, Manautou JE. (2007) Role of NAD(P)H:quinone oxidoreductase 1 in clofibrate-mediated hepatoprotection from acetaminophen. Toxicology 230:197–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mu Y, Zhang J, Zhang S, Zhou HH, Toma D, Ren S, Huang L, Yaramus M, Baum A, Venkataramanan R, et al. (2006) Traditional Chinese medicines Wu Wei Zi (Schisandra chinensis Baill) and Gan Cao (Glycyrrhiza uralensis Fisch) activate pregnane X receptor and increase warfarin clearance in rats. J Pharmacol Exp Ther 316:1369–1377. [DOI] [PubMed] [Google Scholar]
- Panossian A, Wikman G. (2008) Pharmacology of Schisandra chinensis Bail.: an overview of Russian research and uses in medicine. J Ethnopharmacol 118:183–212. [DOI] [PubMed] [Google Scholar]
- Park EJ, Kim YM, Park SW, Kim HJ, Lee JH, Lee DU, Chang KC. (2013) Induction of HO-1 through p38 MAPK/Nrf2 signaling pathway by ethanol extract of Inula helenium L. reduces inflammation in LPS-activated RAW 264.7 cells and CLP-induced septic mice. Food Chem Toxicol 55:386–395. [DOI] [PubMed] [Google Scholar]
- Park SY, Park DJ, Kim YH, Kim Y, Choi YW, Lee SJ. (2011) Schisandra chinensis α-iso-cubebenol induces heme oxygenase-1 expression through PI3K/Akt and Nrf2 signaling and has anti-inflammatory activity in Porphyromonas gingivalis lipopolysaccharide-stimulated macrophages. Int Immunopharmacol 11:1907–1915. [DOI] [PubMed] [Google Scholar]
- Polimeno L, Pesetti B, Annoscia E, Giorgio F, Francavilla R, Lisowsky T, Gentile A, Rossi R, Bucci A, Francavilla A. (2011) Alrp, a survival factor that controls the apoptotic process of regenerating liver after partial hepatectomy in rats. Free Radic Res 45:534–549. [DOI] [PubMed] [Google Scholar]
- Qin XL, Bi HC, Wang CX, Li JL, Wang XD, Liu LS, Chen X, Huang M. (2010) Study of the effect of Wuzhi tablet (Schisandra sphenanthera extract) on tacrolimus tissue distribution in rat by liquid chromatography tandem mass spectrometry method. Biomed Chromatogr 24:399–405. [DOI] [PubMed] [Google Scholar]
- Qin XL, Chen X, Zhong GP, Fan XM, Wang Y, Xue XP, Wang Y, Huang M, Bi HC. (2014) Effect of Tacrolimus on the pharmacokinetics of bioactive lignans of Wuzhi tablet (Schisandra sphenanthera extract) and the potential roles of CYP3A and P-gp. Phytomedicine 21:766–772. [DOI] [PubMed] [Google Scholar]
- Reid AB, Kurten RC, McCullough SS, Brock RW, Hinson JA. (2005) Mechanisms of acetaminophen-induced hepatotoxicity: role of oxidative stress and mitochondrial permeability transition in freshly isolated mouse hepatocytes. J Pharmacol Exp Ther 312:509–516. [DOI] [PubMed] [Google Scholar]
- Saberi B, Ybanez MD, Johnson HS, Gaarde WA, Han D, Kaplowitz N. (2014) Protein kinase C (PKC) participates in acetaminophen hepatotoxicity through c-jun-N-terminal kinase (JNK)-dependent and -independent signaling pathways. Hepatology 59:1543–1554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito C, Lemasters JJ, Jaeschke H. (2010) c-Jun N-terminal kinase modulates oxidant stress and peroxynitrite formation independent of inducible nitric oxide synthase in acetaminophen hepatotoxicity. Toxicol Appl Pharmacol 246:8–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shams I, Malik A, Manov I, Joel A, Band M, Avivi A. (2013) Transcription pattern of p53-targeted DNA repair genes in the hypoxia-tolerant subterranean mole rat Spalax. J Mol Biol 425:1111–1118. [DOI] [PubMed] [Google Scholar]
- Wolf KK, Wood SG, Allard JL, Hunt JA, Gorman N, Walton-Strong BW, Szakacs JG, Duan SX, Hao Q, Court MH, et al. (2007) Role of CYP3A and CYP2E1 in alcohol-mediated increases in acetaminophen hepatotoxicity: comparison of wild-type and Cyp2e1(-/-) mice. Drug Metab Dispos 35:1223–1231. [DOI] [PubMed] [Google Scholar]
- Xie HG, Kim RB. (2005) St John’s wort-associated drug interactions: short-term inhibition and long-term induction? Clin Pharmacol Ther 78:19–24. [DOI] [PubMed] [Google Scholar]
- Xie Y, Hao H, Kang A, Liang Y, Xie T, Sun S, Dai C, Zheng X, Xie L, Li J, et al. (2010) Integral pharmacokinetics of multiple lignan components in normal, CCl4-induced hepatic injury and hepatoprotective agents pretreated rats and correlations with hepatic injury biomarkers. J Ethnopharmacol 131:290–299. [DOI] [PubMed] [Google Scholar]
- Xue XP, Qin XL, Xu C, Zhong GP, Wang Y, Huang M, Bi HC. (2013) Effect of Wuzhi tablet (Schisandra sphenanthera extract) on the pharmacokinetics of cyclosporin A in rats. Phytother Res 27:1255–1259. [DOI] [PubMed] [Google Scholar]
- Zaher H, Buters JT, Ward JM, Bruno MK, Lucas AM, Stern ST, Cohen SD, Gonzalez FJ. (1998) Protection against acetaminophen toxicity in CYP1A2 and CYP2E1 double-null mice. Toxicol Appl Pharmacol 152:193–199. [DOI] [PubMed] [Google Scholar]
- Zhao X, Cong X, Zheng L, Xu L, Yin L, Peng J. (2012) Dioscin, a natural steroid saponin, shows remarkable protective effect against acetaminophen-induced liver damage in vitro and in vivo. Toxicol Lett 214:69–80. [DOI] [PubMed] [Google Scholar]
- Zhu M, Yeung RY, Lin KF, Li RC. (2000) Improvement of phase I drug metabolism with Schisandra chinensis against CCl4 hepatotoxicity in a rat model. Planta Med 66:521–525. [DOI] [PubMed] [Google Scholar]


