Introduction
Late gadolinium enhancement (LGE) and its application in cardiac magnetic resonance (CMR) regarding the ability to discern between various cardiomyopathies, magnetic resonance angiography, first pass perfusion, viability, and prognostic information in various pathologies makes it a valuable tool in advanced non-invasive imaging. It permits detection of abnormal myocardium based on the delivery and distribution of gadolinium (Gd) in the expanded extracellular space caused by fibrosis, protein infiltration, and/or myocardial disarray. Due to its high spatial resolution, LGE-CMR is able to distinguish scar patterns in different types of disease pathologies allowing the physician to expand on the differential diagnosis.1 It also adds value in identifying patients who are at increased risk for sudden cardiac death (SCD).2 Late gadolinium enhancement technique has been developed and validated for more than a decade.3 In this review, we will discuss the aspects of gadolinium-based imaging, and its application in CMR.
Safety of Gadolinium Based Contrast Agents
Gadolinium (an element with paramagnetic features) is able to be temporarily magnetized when placed in an external magnetic field. It shortens the T1 relaxation and provides enhancement of contrast in the images. Gadolinium is bound to a large molecule carrier. For this reason, when injected intravenously, Gd does not enter the intercellular spaces, and is quickly washed out of the blood pool. However, in areas with abnormal myocardium due to cell damage or increased interstitial space, there is an increased uptake and wash out is delayed. The distribution pattern of Gd enables us to distinguish between bright abnormal myocardium, and black normal myocardium.
Risk with Gadolinium Based Contrast Agents
Gadolinium is excreted by the kidneys, and have a half-life of two hours. Similar with the other radiocontrast agents, Gd can cause contrast-induced nephropathy. Nephrogenic systemic sclerosis (NSF), a condition that resembles scleroderma is a rare side effect of Gd contrast. The overall risk of NSF is low, and has declined since Gd was implicated as the cause. Certain factors such as chronic kidney disease, multiple exposures and/or exposure to certain Gd (non-ionic, linear) agents increased the risk of developing NSF after administration. Nephrogenic systemic sclerosis has not been reported in patients with normal renal function.4
Clinical Indications
A. Coronary Artery Disease (CAD)
a) Myocardial Viability
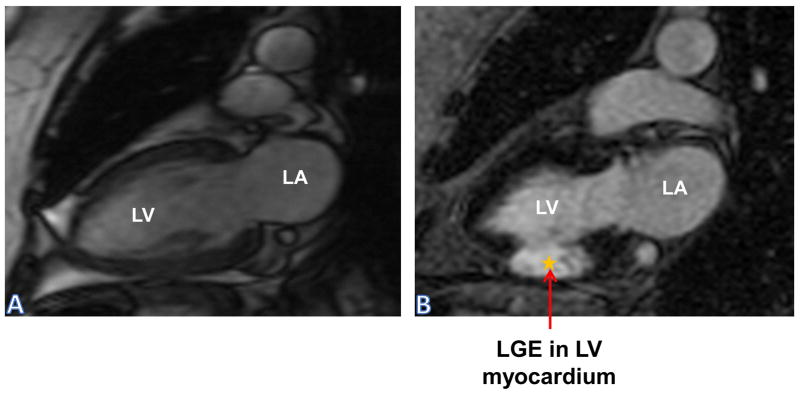
After numerous studies with validation, LGE-CMR imaging has become the gold standard imaging modality for assessing irreversibly injured myocardium, and its prognosis.5 Additionally, the CE-MARC trial has established the superior diagnostic accuracy of LGE-CMR over single-photon emission computed tomography (SPECT) in diagnosing CAD.6 Ischemic cardiomyopathy on LGE has a distribution pattern which typically involves subendocardium. The LGE-CMR has a much higher spatial resolution than SPECT imaging which allows for more accurate detection of subendocardial infarcts even without clinical myocardial necrosis.7 For this reason, CMR has a higher sensitivity in measurement of infarct size compared with SPECT in patients with acute myocardial infarction.8 Additionally, a significant percent of infarcts related to a right coronary or circumflex coronary artery distribution may be missed in SPECT imaging.9 Studies have revealed that LGE-CMR shows an inverse relationship with the extent of transmural enhancement and improvement in contractile function of the left ventricle (LV) which is valuable prognosticaly.10, 11 Several studies have revealed that LGE less than 25% of LV wall thickness predicts good functional recovery whereas LGE greater than 25% is associated with variable recovery of LV function following an intervention (PCI/CABG).7 The LGE-CMR is also a useful tool to detect microvascular obstruction (MVO). This can be seen after revascularization of epicardial coronary artery which has been occluded for a prolonged period. Microvascular obstruction is seen as subendocardial areas of low signal intensity which are surrounded by enhancement indicating fibrosis. The detection of MVO has prognostic implications as well, since it has demonstrated correlations with adverse events such as heart failure (HF), arrhythmia, death, and it is more accurate than catheterization.12
b) Stress Perfusion
An MR-IMPACT trial has shown perfusion CMR to be equivalent of SPECT for CAD, eliminating the need of radiation exposure.13 Stress perfusion CMR (using vasodilator stressor, regadenoson) is compared with resting perfusion CMR to identify areas of reduced myocardial perfusion. The perfusion defect extends from subendocardium towards the epicardium, and the severity is correlated to the extension of defect. Post stress LGE sequences are able to distinguish between viable and non-viable myocardium in the same setting. An abnormal myocardium will show hyper enhancement due to increase Gd concentration whereas viable myocardium will have no contrast uptake.13 (Figure 1)
Figure 1.
Coronary Artery Disease: Panel (A) SSFP sequence: Two chamber view showing left atrium and left ventricle shows normal thickness of inferior wall; Panel (B) LGE sequence showing areas of transmural hyper enhancement in mid-inferior wall suggesting infarction and non-viable myocardium.
LA: Left Atrium; LGE: Late Gadolinium Enhancement; LV: Left Ventricle; SSFP: Steady State Free Precision
c) Myocardial Wall Motion
One of significant advancement in stress CMR imaging is the acquisition of cine images which has allowed to assess LV volumes, function, and wall motion. To assess the wall motion on CMR, an identical pharmacological protocol similar to dobutamine stress echocardiography is used. Studies have shown that dobutamine stress CMR is safe with a higher diagnostic accuracy than dobutamine stress echocardiography.14, 15 Additionally, Jahnke et al has shown stress CMR predicts cardiovascular events in patients with known or suspected CAD.16 Supplementation of stress CMR with LGE-CMR allows us to assess the myocardial viability as well as perfusion of myocardium comprehensively.
B. Non-ischemic cardiomyopathies
a) Hypertrophic Cardiomyopathy (HCM)
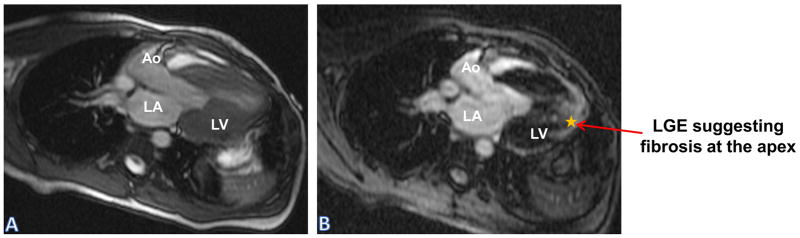
Hypertrophic cardiomyopathy represents the most common cause of SCD in the young, including trained athletes, and an important substrate for HF disability at any age. Various genetic mutations lead to a combination of myocyte hypertrophy, disarray and fibrosis.17 Late gadolinium enhancement, identified in up to 80% of HCM patients, represents areas of focal interstitial expansion due to myocardial fibrosis.17 In patients with HCM, typical patterns of LGE include patchy areas of hyper enhancement in the hypertrophied segments or at the right ventricle (RV) - LV insertion points. The hyper enhancement is predominantly subendocardial and extends towards midmyocardium.18 The importance of studying LGE in HCM is due to the fact that the disease has been associated with risk of malignant arrhythmias and sudden death.2 The amount of LGE (not just the presence) in a population of asymptomatic patients with HCM was an independent predictor of all-cause and cardiovascular mortality.18 Late gadolinium enhancement can help in identifying patients who are at risk for SCD without high risk factors, for implantation of Implantable Cardioverter Defibrillator (ICD). Chan et al have shown a continuous relationship between SCD and percentage of LGE.19 Chan et al concluded a greater than 15% LGE increases the risk of SCD by two-fold compared to patients with no LGE.19 (Figure 2)
Figure 2.
Hypertrophic Cardiomyopathy: Panel (A) SSFP sequence: left ventricle outflow tract image showing mid cavitary obliteration; Panel (B) LGE sequence showing hyper enhancement at the apex, suggestive of fibrosis in patient with hypertrophic cardiomyopathy.
Ao: Aorta; LA: Left Atrium; LGE: Late Gadolinium Enhancement; LV: Left Ventricle; SSFP: Steady State Free Precision
b) Idiopathic Dilated Cardiomyopathy (DCM)
In a significant proportion of patients with new onset HF and normal coronary arteries on cardiac catheterization, LGE-CMR may show an evidence of prior infarction by subendocardial or transmural enhancement in coronary artery distribution. This may be secondary to vasospasm, embolic phenomenon or spontaneous recanalization.20
A quarter of patients with DCM (but no CAD) may have some patchy or longitudinal mid myocardial hyper enhancement in non-coronary artery distribution which represents fibrosis, and may be a foci of arrhythmias.20 The recent study Venero et al has provided a cardinal proof on CMR’s role in predicting left ventricular assist device (LVAD) or heart transplantation requirement in patients with DCM.21 Venero et al concluded that in patients with DCM, the presence of mid myocardial stripe on LGE-CMR can help clinicians identify high risk patients who would need LVAD or heart transplant within 1 year of presentation.21
c) Sarcoidosis
Sarcoidosis is a multisystem granulomatous disease that can affect the myocardium in 20 to 30% of patients.22 Cardiac sarcoidosis presents with conduction abnormalities, congestive HF, and potentially fatal arrhythmias. Sudden death accounts for over half of the fatalities from cardiac sarcoidosis.23 Histological diagnosis of cardiac sarcoidosis by endomyocardial biopsy (EMB) is limited because of patchy cardiac infiltration which may result in false-negative result.24
Recent advances in imaging with CMR and positron emission tomography (PET) have increased diagnostic yield, and hence treatment opportunities in these patients. The CMR with contrast enhancement has high specificity for sarcoidosis.25
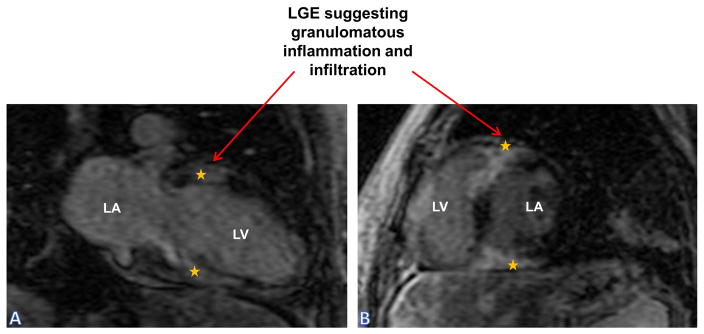
In a study of 155 patients with extra-cardiac sarcoidosis who underwent CMR, absence of LGE was associated with no adverse events even in patients with LV enlargement and dysfunction.25 The LGE-CMR images shows patchy, multifocal mid-myocardial or subepicardial enhancement, with a predilection for basal septum and lateral walls, in a non-coronary distribution.26 Enhancement of the RV side of basal septum is a characteristic finding. Occasionally, the enhancement scan can be transmural or in any distribution.26 Late gadolinium enhancement guided EMB may increase the diagnostic sensitivity for detecting cardiac involvement.24 In a recent meta-analysis presence of LGE in patients with cardiac sarcoidosis was found to be associated with increased arrhythmias and all-cause mortality.27 (Figure 3)
Figure 3.
Sarcoidosis: Panel (A) Two chamber (LA and LV) and Panel (B) Short axis view of LA and LV- both show areas of hyper enhancement secondary to granulomatous inflammation and infiltration in basal segments.
LA: Left Atrium; LGE: Late Gadolinium Enhancement; LV: Left Ventricle
d) Amyloidosis
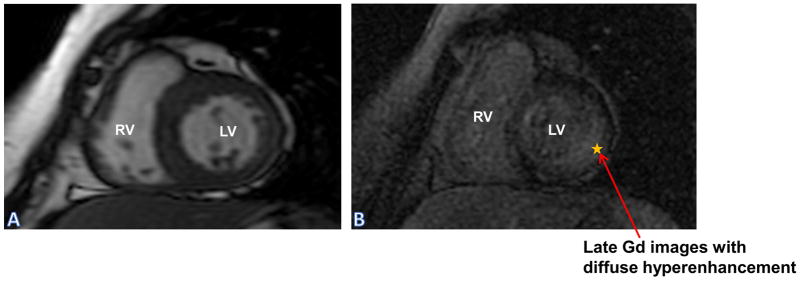
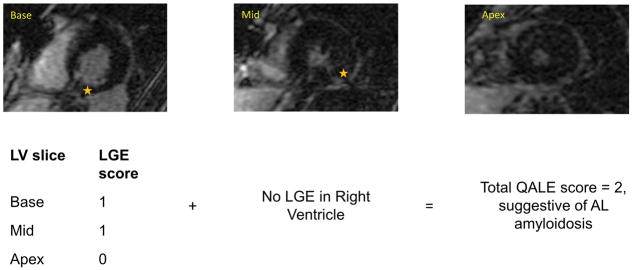
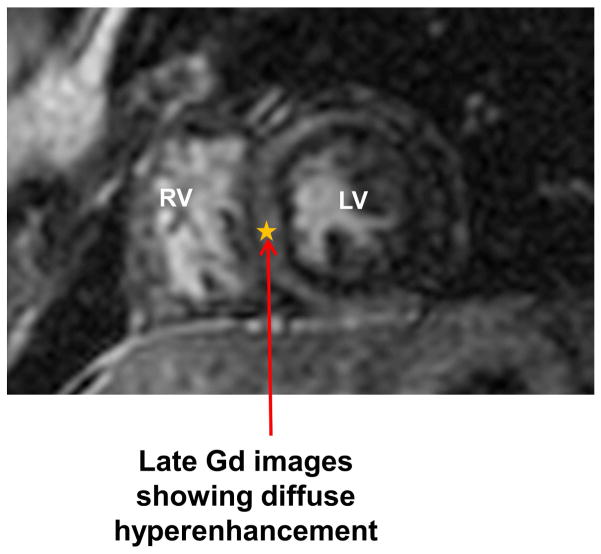
Cardiac amyloidosis is characterized by generalized deposition of amyloid fibrils in the interstitium which causes concentric thickening leading to restrictive cardiomyopathy. The typical finding of cardiac amyloidosis on LGE-CMR is the difficulty to optimally null the myocardium by choosing a T1 value. The LGE-CMR has a sensitivity and specificity of 86% for the identification of cardiac involvement in patients with systemic amyloidosis, detecting early abnormalities in patients with normal LV wall thickness, and appeared strongly related to HF severity.28 The pattern of LGE can be varied, most commonly described as a global subendocardial enhancement. However, it can involve both subendocardial and subepicardial regions, also with both patchy and diffuse distributions.29 In a recent meta-analysis, presence of diffuse gadolinium enhancement was associated with poor prognosis.30 Hence, late gadolinium enhancement imaging in patients with AL cardiac amyloidosis provides prognostic information over biomarkers. Additionally, the prognosis of cardiac amyloidosis depends upon the type of amyloid protein deposition.31–33 Query Amyloid Late Enhancement (QALE) score has been recently developed to improve the LGE-CMR’s diagnostic accuracy in differentiating transthyretin-related amyloidosis (ATTR) and AL types of cardiac amyloidosis.34 By differentiating different types of cardiac amyloidosis, the QALE score has created a new avenue for LGE-CMR imaging as a possibility to avoid biopsy in patients with cardiac amyloidosis. (Figure 4 and 5)
Figure 4.
Amyloidosis: Panel (A) SSFP sequence of short axis LV and RV shows moderate concentric hypertrophy; Panel (B) LGE sequence shows diffuse hyper enhancement in lateral wall.
LGE: Late Gadolinium Enhancement; LV: Left Ventricle; RV: Right Ventricle; SSFP: Steady State Free Precision
Figure 5.
Late Gadolinium Images of LV slices at Base, mid-LV and Apex. Non-circumferential patchy LGE pattern is seen at base and mid-LV. No LGE is seen at Apex.
LGE: Late Gadolinium Enhancement; LV: Left Ventricle; QALE: Query Amyloid Late Enhancement
e) Myocarditis
Myocarditis presents clinically as a flu-like illness with positive cardiac enzymes, along with wall motion abnormalities without CAD. An endomyocardial biopsy is currently gold standard for the diagnosis. Cardiac magnetic resonance’s accuracy of diagnosis of myocarditis has been increased by Lake Louise criteria as suggested by a large consensus group.5 Authors recommend the combined use of three tissue markers: 1) regional or global hyper signal intensity on T2-weighted images, 2) increased early gadolinium enhancement, and 3) at least one focal lesion on LGE with a non-ischemic distribution. When two or more of these tissue-based criteria were positive, myocarditis could be predicted with a diagnostic accuracy of 78% as compared with EMB.5 Studies have shown with the addition of CMR imaging to biopsy, the accuracy in diagnosis increases to 95%.7 The pattern of LGE in patients with myocarditis is most frequently subepicardial extending towards midmyocardium and sparing the subendocardium with a patchy distribution, frequently in inferior and lateral walls.35 With the resolution of myocarditis, the enhancement regress and there may be no LGE or mid-myocardial enhancement similar to DCM patients. In a recent study, LGE was found to be the best independent predictor of overall and cardiovascular mortality in a population with biopsy-proven myocarditis.36 (Figure 6)
Figure 6.
Myocarditis: LGE sequence shows hyper enhancement of entire septum, inferior and anterior wall consistent with myocarditis. Note, enhancement of subepicardium and sparing of subendocardium on inferior and anterior walls.
LGE: Late Gadolinium Enhancement; LV: Left Ventricle; RV: Right Ventricle
f) Arrhythmogenic right ventricular cardiomyopathy (ARVC)
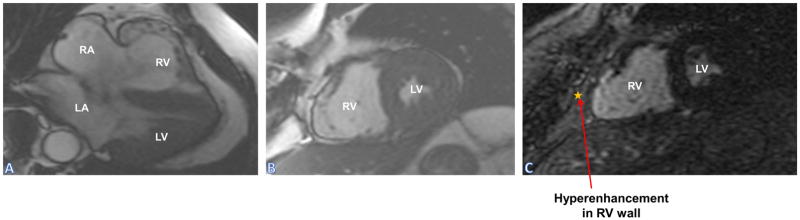
Diagnoses of ARVC is based on a set of major and minor criteria which include comprehensive assessment of electrical, anatomical and functional abnormalities.37 Right ventricle is difficult to image by transthoracic echo, but CMR provides good visualization and is considered gold standard. In a study by Tandri et al, LGE of RV myocardium in patients with ARVC correlated with inducible ventricular tachycardia and histopathology of fibro-fatty infiltration.38 (Figure 7)
Figure 7.
Arrhythmogenic right ventricular cardiomyopathy: Panels (A) and (B) SSFP sequence of four chamber and short axis showing RV dilatation; Panel (C) LGE sequence shows hyper enhancement in RV wall secondary to fibro fatty infiltration.
LA: Left Atrium; LGE: Late Gadolinium Enhancement; LV: Left Ventricle; RA: Right Atrium; RV: Right Ventricle; SSFP: Steady State Free Precision
g) Pulmonary artery hypertension (PAH)
The role of LGE-CMR in patients with PAH has been extensively studied over a past decade.39, 40 In general, majority of patients with PAH demonstrate LGE at RV insertion points. The presence of LGE at RV insertion point is a predictor of RV failure.41, 42 A recent study by Freed et al has suggested that the presence of LGE at RV insertion points is an independent predictor of adverse outcomes in patients with PAH.41
C. Pericardial Disease
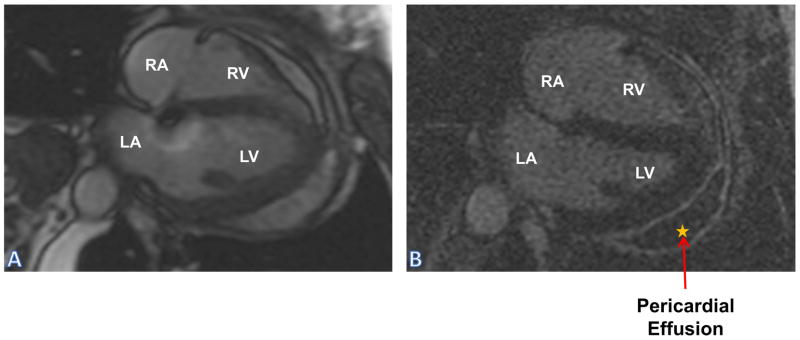
Constrictive pericarditis (CP) is characterized by thick pericardial fibrosis and frequent calcification that progressively leads to debilitating HF with constrictive physiology. Normal pericardium is thin (<2mm) and a poorly vascularized structure which has no significant LGE after contrast administration. Presence of pericardial LGE was more frequently associated with histological findings of pericardial inflammation, including increased neovascularization, fibroblast proliferation, and granulation tissue.43 In a small study by Feng et al, a trial of anti-inflammatory therapy based on presence of LGE in pericardium was associated with resolution of constrictive physiology and clinical improvement.43 (Figure 8)
Figure 8.
Effusive/Constrictive pericarditis: Panel (A) SSFP sequence of four chamber showing pericardial effusion with thickened pericardium; Panel (B) LGE sequence shows enhanced pericardium surrounding loculated effusion.
LA: Left Atrium; LGE: Late Gadolinium Enhancement; LV: Left Ventricle; RA: Right Atrium; RV: Right Ventricle
D. Cardiac Masses
Cardiac magnetic resonance plays an important role in the evaluation of cardiac masses of non-neoplastic and neoplastic origin, adding “precision” to diagnosis using a noninvasive imaging modality. Cardiac masses are usually evaluated in context with adjacent structures, regional wall motion, vascularity, and surface demarcation. Early gadolinium enhancement with high signal intensity is seen in vascular tumors such as hemangioma or angiosarcoma, whereas a thrombus or pericardial cyst has no contrast uptake, and therefore appears black. However, chronic and well organized thrombus may contain peripheral fibrous tissue which shows Gd enhancement.44
The presence of LGE on CMR is due to direct tumor invasion or necrosis which allows for intracellular expansion of Gd. Myxomas (most common primary cardiac tumor) are generally well defined, smooth, pedunculated mass located in the atria. As myxomas are made up of a mixture of tissue, necrosis, fibrosis, and foci of hemorrhage, it typically shows patchy LGE.45 On contrary to myxomas, benign conditions like fibroelastoma usually display a uniform pattern of LGE. Additionally, rare benign condition such as rhabdomyoma shows minimal or no enhancement on CMR.45 Malignant primary (i.e. angiosarcomas) and secondary (i.e. metastasis) cardiac tumors show heterogeneous enhancement on LGE-CMR due to peripheral fibrosis and central necrosis.46 (Figures 9 and 10)
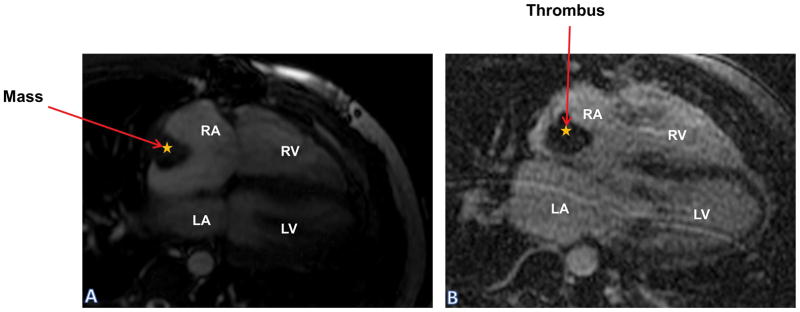
Figure 9.
Mass/Thrombus: Panel (A) SSFP sequence of four chamber showing filling defect in RA concerning for a myxoma; Panel (B) LGE sequence shows no enhancement of that mass suggesting against tumor and more likely a thrombus.
LA: Left Atrium; LGE: Late Gadolinium Enhancement; LV: Left Ventricle; RA: Right Atrium; RV: Right Ventricle; SSFP: Steady State Free Precision
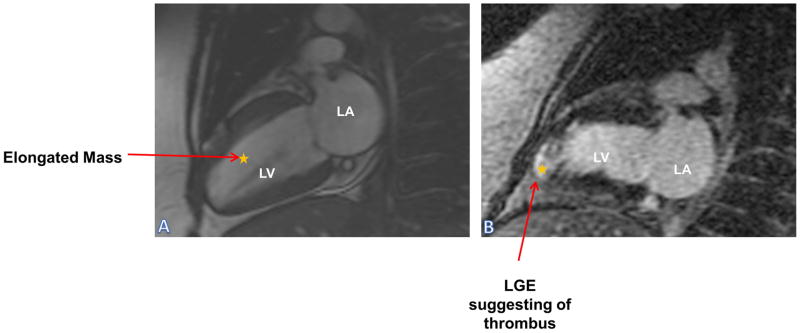
Figure 10.
Thrombus: Panel (A) SSFP sequence of two chamber showing an elongated mass in the LV; Panel (B) LGE sequence shows enhancement of the apex, suggesting infarction and subsequent long serpiginous thrombus formation.
LA: Left Atrium; LGE: Late Gadolinium Enhancement; LV: Left Ventricle; SSFP: Steady State Free Precision
E. Electrophysiological Procedures
Fluoroscopy plays a vital role during Electrophysiological (EP) procedures.47, 48 The increased radiation exposure to physicians and patients is harmful, and hence non-fluoroscopic techniques should be used when possible. Cardiac magnetic resonance offers less radiation exposure to both patients and providers during EP procedures.49 Additionally, the excellent spatial and temporal resolution of CMR provide an enhanced visualization of cardiac structures as well as better segregation between damaged and normal tissue. A technique using 3-D CMR angiography has been used to identify pulmonary vein anatomic variants [probable foci triggering atrial fibrillation (AF)] and assist in planning of AF ablation.50 An identification of abnormal foci producing AF before the procedure allows the electrophysiologist in selection of catheter, type of procedure, and minimize the procedure related complications. Monomorphic ventricular tachycardia (MVT) (a life threatening rhythm disorder) usually originates from scar. As described, LGE-CMR is the most reliable method to characterize regions of the scar tissue in any type of cardiomyopathy. The border-zone area of the scar tissue can give rise to MVT.51, 52 The LGE-CMR helps to correlate the scar morphology to possible MVT ablation sites, allowing higher successful rates of MVT ablation procedure.
F. Specific Population
Traditional evaluation of CAD with SPECT imaging has lower sensitivity in women compared with men.6 The use of stress CMR allows us to get higher diagnostic accuracy in specific populations such as women. Women with signs and symptoms of ischemia and non-obstructive CAD are more likely to present with coronary microvascular dysfunction (CMD). The assessment of myocardial perfusion with CMR allows us to detect CMD in this specific population.53
Cardiac magnetic resonance is considered to be a gold standard imaging technique in measurement of LV function and volumes.54, 55 Reduced LV ejection fraction (LVEF) (i.e. ≤35%) is considered as a marker for increased risk of SCD in patients with ischemic and non-ischemic cardiomyopathies. Use of implantable cardioverter defibrillator (ICD) is recommended for primary prevention in these patients who have a greater risk of SCD.56
However, the absolute number of SCD is much higher in patients with preserved LVEF given that this is a much larger subgroup.57 MUSTT study has highlighted that LVEF alone has lower sensitivity and specificity in predicting SCD.58 Evidence suggest that the focal or diffuse myocardial fibrosis help stratify the risk of SCD.59 The extent and regions of myocardial fibrosis provide a substrate for ventricular arrhythmias leading to SCD.60 The focal myocardial fibrosis can be readily detected by LGE-CMR. However, to identify the diffuse myocardial fibrosis T1 mapping techniques with quantification of extracellular volume can be used.61 The presence of myocardial fibrosis can help clinicians to identify the possible candidates for ICD placement to prevent SCD.55
Conclusion
The LGE-CMR has distinct advantages over other imaging modalities but the use is still limited by the availability of CMR scanners at different hospitals in US. With improvement in CMR techniques and sequences, the future to integrate LGE-CMR into routine clinical decision making looks promising. Clinical education of general cardiologists to use LGE-CMR for diagnosis and prognosis of cardiovascular diseases is an area of special emphasis in order to move the field forward.
Acknowledgments
Sources of Funding
Dr. Nirav Patel is supported by National Institutes of Health (NIH) grant 1T32HL129948-01A1.
Footnotes
Authors Contributions
Concept/design: PK, GA
Data analysis/interpretation: None
Drafting article: PK, NP, GA
Critical revision of article: PK, NP, GA
Approval of article: PK, NP, GA
Statistics: Not Applicable
Funding secured by: None
Data collection: None
References
- 1.Karamitsos TD, Francis JM, Myerson S, et al. The role of cardiovascular magnetic resonance imaging in heart failure. J Am Coll Cardiol. 2009;54:1407–1424. doi: 10.1016/j.jacc.2009.04.094. [DOI] [PubMed] [Google Scholar]
- 2.Kwon DH, Smedira NG, Rodriguez ER, et al. Cardiac magnetic resonance detection of myocardial scarring in hypertrophic cardiomyopathy: correlation with histopathology and prevalence of ventricular tachycardia. J Am Coll Cardiol. 2009;54:242–249. doi: 10.1016/j.jacc.2009.04.026. [DOI] [PubMed] [Google Scholar]
- 3.Soriano CJ, Ridocci F, Estornell J, et al. Noninvasive diagnosis of coronary artery disease in patients with heart failure and systolic dysfunction of uncertain etiology, using late gadolinium-enhanced cardiovascular magnetic resonance. J Am Coll Cardiol. 2005;45:743–748. doi: 10.1016/j.jacc.2004.11.037. [DOI] [PubMed] [Google Scholar]
- 4.Reiter T, Ritter O, Prince MR, et al. Minimizing risk of nephrogenic systemic fibrosis in cardiovascular magnetic resonance. J Cardiovasc Magn Reson. 2012;14:31. doi: 10.1186/1532-429X-14-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Friedrich MG, Sechtem U, Schulz-Menger J, et al. Cardiovascular magnetic resonance in myocarditis: A JACC White Paper. J Am Coll Cardiol. 2009;53:1475–1487. doi: 10.1016/j.jacc.2009.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Greenwood JP, Maredia N, Younger JF, et al. Cardiovascular magnetic resonance and single-photon emission computed tomography for diagnosis of coronary heart disease (CE-MARC): a prospective trial. Lancet. 2012;379:453–460. doi: 10.1016/S0140-6736(11)61335-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Doltra A, Amundsen BH, Gebker R, et al. Emerging concepts for myocardial late gadolinium enhancement MRI. Curr Cardiol Rev. 2013;9:185–190. doi: 10.2174/1573403X113099990030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lund GK, Stork A, Saeed M, et al. Acute myocardial infarction: evaluation with first-pass enhancement and delayed enhancement MR imaging compared with 201Tl SPECT imaging. Radiology. 2004;232:49–57. doi: 10.1148/radiol.2321031127. [DOI] [PubMed] [Google Scholar]
- 9.Ibrahim T, Bulow HP, Hackl T, et al. Diagnostic value of contrast-enhanced magnetic resonance imaging and single-photon emission computed tomography for detection of myocardial necrosis early after acute myocardial infarction. J Am Coll Cardiol. 2007;49:208–216. doi: 10.1016/j.jacc.2006.08.047. [DOI] [PubMed] [Google Scholar]
- 10.Beek AM, Kuhl HP, Bondarenko O, et al. Delayed contrast-enhanced magnetic resonance imaging for the prediction of regional functional improvement after acute myocardial infarction. J Am Coll Cardiol. 2003;42:895–901. doi: 10.1016/s0735-1097(03)00835-0. [DOI] [PubMed] [Google Scholar]
- 11.White JA, Yee R, Yuan X, et al. Delayed enhancement magnetic resonance imaging predicts response to cardiac resynchronization therapy in patients with intraventricular dyssynchrony. J Am Coll Cardiol. 2006;48:1953–1960. doi: 10.1016/j.jacc.2006.07.046. [DOI] [PubMed] [Google Scholar]
- 12.Wu KC. CMR of microvascular obstruction and hemorrhage in myocardial infarction. J Cardiovasc Magn Reson. 2012;14:68. doi: 10.1186/1532-429X-14-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schwitter J, Wacker CM, van Rossum AC, et al. MR-IMPACT: comparison of perfusion-cardiac magnetic resonance with single-photon emission computed tomography for the detection of coronary artery disease in a multicentre, multivendor, randomized trial. Eur Heart J. 2008;29:480–489. doi: 10.1093/eurheartj/ehm617. [DOI] [PubMed] [Google Scholar]
- 14.Wahl A, Paetsch I, Gollesch A, et al. Safety and feasibility of high-dose dobutamine-atropine stress cardiovascular magnetic resonance for diagnosis of myocardial ischaemia: experience in 1000 consecutive cases. Eur Heart J. 2004;25:1230–1236. doi: 10.1016/j.ehj.2003.11.018. [DOI] [PubMed] [Google Scholar]
- 15.Nagel E, Lehmkuhl HB, Bocksch W, et al. Noninvasive diagnosis of ischemia-induced wall motion abnormalities with the use of high-dose dobutamine stress MRI: comparison with dobutamine stress echocardiography. Circulation. 1999;99:763–770. doi: 10.1161/01.cir.99.6.763. [DOI] [PubMed] [Google Scholar]
- 16.Jahnke C, Nagel E, Gebker R, et al. Prognostic value of cardiac magnetic resonance stress tests: adenosine stress perfusion and dobutamine stress wall motion imaging. Circulation. 2007;115:1769–1776. doi: 10.1161/CIRCULATIONAHA.106.652016. [DOI] [PubMed] [Google Scholar]
- 17.Moon JC, Reed E, Sheppard MN, et al. The histologic basis of late gadolinium enhancement cardiovascular magnetic resonance in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2004;43:2260–2264. doi: 10.1016/j.jacc.2004.03.035. [DOI] [PubMed] [Google Scholar]
- 18.O’Hanlon R, Grasso A, Roughton M, et al. Prognostic significance of myocardial fibrosis in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2010;56:867–874. doi: 10.1016/j.jacc.2010.05.010. [DOI] [PubMed] [Google Scholar]
- 19.Chan RH, Maron BJ, Olivotto I, et al. Prognostic value of quantitative contrast-enhanced cardiovascular magnetic resonance for the evaluation of sudden death risk in patients with hypertrophic cardiomyopathy. Circulation. 2014;130:484–495. doi: 10.1161/CIRCULATIONAHA.113.007094. [DOI] [PubMed] [Google Scholar]
- 20.McCrohon JA, Moon JC, Prasad SK, et al. Differentiation of heart failure related to dilated cardiomyopathy and coronary artery disease using gadolinium-enhanced cardiovascular magnetic resonance. Circulation. 2003;108:54–59. doi: 10.1161/01.CIR.0000078641.19365.4C. [DOI] [PubMed] [Google Scholar]
- 21.Venero JV, Doyle M, Shah M, et al. Mid wall fibrosis on CMR with late gadolinium enhancement may predict prognosis for LVAD and transplantation risk in patients with newly diagnosed dilated cardiomyopathy—preliminary observations from a high-volume transplant centre. Esc Heart Failure. 2015;2:150–159. doi: 10.1002/ehf2.12041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Iwai K, Takemura T, Kitaichi M, et al. Pathological studies on sarcoidosis autopsy. II. Early change, mode of progression and death pattern. Acta Pathol Jpn. 1993;43:377–385. doi: 10.1111/j.1440-1827.1993.tb01149.x. [DOI] [PubMed] [Google Scholar]
- 23.Nery PB, Beanlands RS, Nair GM, et al. Atrioventricular block as the initial manifestation of cardiac sarcoidosis in middle-aged adults. J Cardiovasc Electrophysiol. 2014;25:875–881. doi: 10.1111/jce.12401. [DOI] [PubMed] [Google Scholar]
- 24.Aljaroudi WA, Flamm SD, Saliba W, et al. Role of CMR imaging in risk stratification for sudden cardiac death. JACC Cardiovasc Imaging. 2013;6:392–406. doi: 10.1016/j.jcmg.2012.11.011. [DOI] [PubMed] [Google Scholar]
- 25.Greulich S, Deluigi CC, Gloekler S, et al. CMR imaging predicts death and other adverse events in suspected cardiac sarcoidosis. JACC Cardiovasc Imaging. 2013;6:501–511. doi: 10.1016/j.jcmg.2012.10.021. [DOI] [PubMed] [Google Scholar]
- 26.Smedema JP, Snoep G, van Kroonenburgh MP, et al. Evaluation of the accuracy of gadolinium-enhanced cardiovascular magnetic resonance in the diagnosis of cardiac sarcoidosis. J Am Coll Cardiol. 2005;45:1683–1690. doi: 10.1016/j.jacc.2005.01.047. [DOI] [PubMed] [Google Scholar]
- 27.Coleman GC, Shaw PW, Balfour PC, Jr, et al. Prognostic Value of Myocardial Scarring on CMR in Patients With Cardiac Sarcoidosis. JACC Cardiovasc Imaging. 2017;10:411–420. doi: 10.1016/j.jcmg.2016.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Syed IS, Glockner JF, Feng D, et al. Role of cardiac magnetic resonance imaging in the detection of cardiac amyloidosis. JACC Cardiovasc Imaging. 2010;3:155–164. doi: 10.1016/j.jcmg.2009.09.023. [DOI] [PubMed] [Google Scholar]
- 29.Austin BA, Tang WH, Rodriguez ER, et al. Delayed hyper-enhancement magnetic resonance imaging provides incremental diagnostic and prognostic utility in suspected cardiac amyloidosis. JACC Cardiovasc Imaging. 2009;2:1369–1377. doi: 10.1016/j.jcmg.2009.08.008. [DOI] [PubMed] [Google Scholar]
- 30.Boynton SJ, Geske JB, Dispenzieri A, et al. LGE Provides Incremental Prognostic Information Over Serum Biomarkers in AL Cardiac Amyloidosis. JACC Cardiovasc Imaging. 2016;9:680–686. doi: 10.1016/j.jcmg.2015.10.027. [DOI] [PubMed] [Google Scholar]
- 31.Dubrey SW, Cha K, Anderson J, et al. The clinical features of immunoglobulin light-chain (AL) amyloidosis with heart involvement. QJM: An International Journal of Medicine. 1998;91:141–157. doi: 10.1093/qjmed/91.2.141. [DOI] [PubMed] [Google Scholar]
- 32.Lebovic D, Hoffman J, Levine BM, et al. Predictors of survival in patients with systemic light-chain amyloidosis and cardiac involvement initially ineligible for stem cell transplantation and treated with oral melphalan and dexamethasone. Br J Haematol. 2008;143:369–373. doi: 10.1111/j.1365-2141.2008.07327.x. [DOI] [PubMed] [Google Scholar]
- 33.Dungu JN, Anderson LJ, Whelan CJ, et al. Cardiac transthyretin amyloidosis. Heart. 2012;98:1546–1554. doi: 10.1136/heartjnl-2012-301924. [DOI] [PubMed] [Google Scholar]
- 34.Dungu JN, Valencia O, Pinney JH, et al. CMR-Based Differentiation of AL and ATTR Cardiac Amyloidosis. JACC: Cardiovascular Imaging. 2014;7:133–142. doi: 10.1016/j.jcmg.2013.08.015. [DOI] [PubMed] [Google Scholar]
- 35.Mahrholdt H, Wagner A, Deluigi CC, et al. Presentation, patterns of myocardial damage, and clinical course of viral myocarditis. Circulation. 2006;114:1581–1590. doi: 10.1161/CIRCULATIONAHA.105.606509. [DOI] [PubMed] [Google Scholar]
- 36.Parsai C, O’Hanlon R, Prasad SK, et al. Diagnostic and prognostic value of cardiovascular magnetic resonance in non-ischaemic cardiomyopathies. J Cardiovasc Magn Reson. 2012;14:54. doi: 10.1186/1532-429X-14-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Marcus FI, McKenna WJ, Sherrill D, et al. Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: proposed modification of the Task Force Criteria. Eur Heart J. 2010;31:806–814. doi: 10.1093/eurheartj/ehq025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tandri H, Saranathan M, Rodriguez ER, et al. Noninvasive detection of myocardial fibrosis in arrhythmogenic right ventricular cardiomyopathy using delayed-enhancement magnetic resonance imaging. J Am Coll Cardiol. 2005;45:98–103. doi: 10.1016/j.jacc.2004.09.053. [DOI] [PubMed] [Google Scholar]
- 39.Blyth KG, Groenning BA, Martin TN, et al. Contrast enhanced-cardiovascular magnetic resonance imaging in patients with pulmonary hypertension. Eur Heart J. 2005;26:1993–1999. doi: 10.1093/eurheartj/ehi328. [DOI] [PubMed] [Google Scholar]
- 40.Swift AJ, Rajaram S, Capener D, et al. LGE patterns in pulmonary hypertension do not impact overall mortality. JACC Cardiovasc Imaging. 2014;7:1209–1217. doi: 10.1016/j.jcmg.2014.08.014. [DOI] [PubMed] [Google Scholar]
- 41.Freed BH, Gomberg-Maitland M, Chandra S, et al. Late gadolinium enhancement cardiovascular magnetic resonance predicts clinical worsening in patients with pulmonary hypertension. J Cardiovasc Magn Reson. 2012;14:11. doi: 10.1186/1532-429X-14-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.McCann GP, Beek AM, Vonk-Noordegraaf A, et al. Delayed contrast-enhanced magnetic resonance imaging in pulmonary arterial hypertension. Circulation. 2005;112:e268. doi: 10.1161/CIRCULATIONAHA.104.512848. [DOI] [PubMed] [Google Scholar]
- 43.Feng D, Glockner J, Kim K, et al. Cardiac magnetic resonance imaging pericardial late gadolinium enhancement and elevated inflammatory markers can predict the reversibility of constrictive pericarditis after antiinflammatory medical therapy: a pilot study. Circulation. 2011;124:1830–1837. doi: 10.1161/CIRCULATIONAHA.111.026070. [DOI] [PubMed] [Google Scholar]
- 44.Paydarfar D, Krieger D, Dib N, et al. In vivo magnetic resonance imaging and surgical histopathology of intracardiac masses: distinct features of subacute thrombi. Cardiology. 2001;95:40–47. doi: 10.1159/000047342. [DOI] [PubMed] [Google Scholar]
- 45.Motwani M, Kidambi A, Herzog BA, et al. MR Imaging of Cardiac Tumors and Masses: A Review of Methods and Clinical Applications. Radiology. 2013;268:26–43. doi: 10.1148/radiol.13121239. [DOI] [PubMed] [Google Scholar]
- 46.Bruna J, Lockwood M. Primary heart angiosarcoma detected by computed tomography and magnetic resonance imaging. Eur Radiol. 1998;8:66–68. doi: 10.1007/s003300050341. [DOI] [PubMed] [Google Scholar]
- 47.Dickfeld T, Kato R, Zviman M, et al. Characterization of acute and subacute radiofrequency ablation lesions with nonenhanced magnetic resonance imaging. Heart Rhythm. 2007;4:208–214. doi: 10.1016/j.hrthm.2006.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Estner HL, Grazia Bongiorni M, Chen J, et al. Use of fluoroscopy in clinical electrophysiology in Europe: results of the European Heart Rhythm Association Survey. EP Europace. 2015;17:1149–1152. doi: 10.1093/europace/euv223. [DOI] [PubMed] [Google Scholar]
- 49.Nair GM, Nery PB, Redpath CJ, et al. Radiation safety and ergonomics in the electrophysiology laboratory: update on recent advances. Curr Opin Cardiol. 2016;31:11–22. doi: 10.1097/HCO.0000000000000246. [DOI] [PubMed] [Google Scholar]
- 50.Kato R, Lickfett L, Meininger G, et al. Pulmonary vein anatomy in patients undergoing catheter ablation of atrial fibrillation: lessons learned by use of magnetic resonance imaging. Circulation. 2003;107:2004–2010. doi: 10.1161/01.CIR.0000061951.81767.4E. [DOI] [PubMed] [Google Scholar]
- 51.Bello D, Fieno DS, Kim RJ, et al. Infarct morphology identifies patients with substrate for sustained ventricular tachycardia. J Am Coll Cardiol. 2005;45:1104–1108. doi: 10.1016/j.jacc.2004.12.057. [DOI] [PubMed] [Google Scholar]
- 52.Nazarian S, Bluemke DA, Lardo AC, et al. Magnetic resonance assessment of the substrate for inducible ventricular tachycardia in nonischemic cardiomyopathy. Circulation. 2005;112:2821–2825. doi: 10.1161/CIRCULATIONAHA.105.549659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Thomson LEJ, Wei J, Agarwal M, et al. Cardiac Magnetic Resonance Myocardial Perfusion Reserve Index Is Reduced in Women With Coronary Microvascular Dysfunction: A National Heart, Lung and Blood Institute-Sponsored Study From the Women’s Ischemia Syndrome Evaluation (WISE) Circulation. Cardiovascular imaging. 2015;8:e002481. doi: 10.1161/CIRCIMAGING.1114.002481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Pennell DJ. Cardiovascular magnetic resonance: twenty-first century solutions in cardiology. Clin Med (Lond) 2003;3:273–278. doi: 10.7861/clinmedicine.3-3-273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Su MY, Lin LY, Tseng YH, et al. CMR-verified diffuse myocardial fibrosis is associated with diastolic dysfunction in HFpEF. JACC Cardiovasc Imaging. 2014;7:991–997. doi: 10.1016/j.jcmg.2014.04.022. [DOI] [PubMed] [Google Scholar]
- 56.Kusumoto FM, Calkins H, Boehmer J, et al. HRS/ACC/AHA Expert Consensus Statement on the Use of Implantable Cardioverter-Defibrillator Therapy in Patients Who Are Not Included or Not Well Represented in Clinical Trials. Circulation. 2014;130:94–125. doi: 10.1161/CIR.0000000000000056. [DOI] [PubMed] [Google Scholar]
- 57.Goldberger JJ, Cain ME, Hohnloser SH, et al. American Heart Association/American College of Cardiology Foundation/Heart Rhythm Society Scientific Statement on Noninvasive Risk Stratification Techniques for Identifying Patients at Risk for Sudden Cardiac Death. A Scientific Statement From the American Heart Association Council on Clinical Cardiology Committee on Electrocardiography and Arrhythmias and Council on Epidemiology and Prevention. 2008;118:1497–1518. [PubMed] [Google Scholar]
- 58.Buxton AE, Lee KL, Hafley GE, et al. Limitations of ejection fraction for prediction of sudden death risk in patients with coronary artery disease: lessons from the MUSTT study. J Am Coll Cardiol. 2007;50:1150–1157. doi: 10.1016/j.jacc.2007.04.095. [DOI] [PubMed] [Google Scholar]
- 59.AlJaroudi WA, Flamm SD, Saliba W, et al. Role of CMR Imaging in Risk Stratification for Sudden Cardiac Death. JACC: Cardiovascular Imaging. 2013;6:392–406. doi: 10.1016/j.jcmg.2012.11.011. [DOI] [PubMed] [Google Scholar]
- 60.Wu TJ, Ong JJ, Hwang C, et al. Characteristics of wave fronts during ventricular fibrillation in human hearts with dilated cardiomyopathy: role of increased fibrosis in the generation of reentry. J Am Coll Cardiol. 1998;32:187–196. doi: 10.1016/s0735-1097(98)00184-3. [DOI] [PubMed] [Google Scholar]
- 61.Salerno M, Kramer CM. Advances in parametric mapping with CMR imaging. JACC Cardiovasc Imaging. 2013;6:806–822. doi: 10.1016/j.jcmg.2013.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]