Abstract
Hepatitis B virus X protein (HBx) has been previously demonstrated to be associated with the regulation of cell proliferation; however, the exact mechanisms underlying this effect remain unclear. The present study aimed to investigate the regulatory mechanism of HBx on the cycle progression of primary renal tubular epithelial cells. Primary renal tubular epithelial cells of Sprague Dawley (SD) rats were separated and cultured. The morphology of cultured cells was characterized by immunohistochemical analysis and the results demonstrated that primary renal tubular epithelial cells with the expected morphology and distribution were successfully separated and cultured from SD rats. HBx gene pcDNA3.1/myc vector and empty vector were constructed and transfected into cells as HBx and empty groups, respectively. Following transfection, the mRNA and protein levels of HBx, cyclin A, cyclin D1 and cyclin E in cells were determined by reverse transcription-quantitative polymerase chain reaction and western blot analysis, respectively. The results demonstrated that following HBx gene transfection, the mRNA and protein levels of HBx, cyclin A, cyclin D1 and cyclin E in cells were significantly upregulated, compared with the empty control group (P<0.05). Furthermore, cell apoptosis and the cell cycle were evaluated by Annexin V-fluorescein isothiocyanate/propidium iodide staining and flow cytometry. HBx gene transfection significantly inhibited the cell apoptosis (P<0.05), promoted cell cycle progression from the G1 to S phase and arrested the cell cycle in the S phase. Therefore, the results of the present study indicated that HBx gene transfection may regulate the apoptosis and cell cycle of primary renal tubular epithelial cells by affecting the expression of cyclins. The results of the present study may improve the understanding of pathogenesis associated with HBV-associated glomerulonephritis, and may also provide insight and theoretical support for the future design and development of drugs for the treatment of hepatitis B virus.
Keywords: hepatitis B virus, hepatitis B virus X protein, nephropathy, renal tubular epithelial cells, cell cycle, cyclin
Introduction
The transmission of hepatitis B virus (HBV) is a major issue threatening public health worldwide, particularly in China. It is reported that in China, up to 10–20% of the population carry HBV (1). HBV infection leads to damage to multiple organs and in vivo lesions, with the kidney being among the susceptible vital organs. The morbidity of HBV-associated glomerulonephritis (HBV-GN) is a leading cause of secondary nephropathy in China (2). Nephron loss induced by HBV infection and the consequent imbalance of cell cycle progression are considered to be major factors in the pathogenesis of HBV-GN. Cell cycle progression is strictly regulated by genes and proteins that have been investigated extensively, including cyclin A, cyclin E, p16 and p21 proteins. When cell cycle progression is disturbed, the consequent cellular apoptosis or non-programmed cell death have an important role in renal injury.
The HBV gene is comprised of four open reading frames, which include the preS/S, P, C and X genes (3). The HBV X (HBx) gene guides the synthesis of the HBx protein, which is a unique non-structural protein of HBV. The HBx protein is a type of functional protein possessing various regulatory effects, such as transactivation, and it has an important regulatory role in virus replication, cell infection, cellular apoptosis induction and the triggering of inflammatory responses (4–10). Although HBx has been demonstrated to exert effects on the regulation of cell proliferation, the detailed regulatory mechanisms of the HBx protein are yet to be established.
Previous research conclusions concerning HBx have been based on transformed or immortalized cell lines, and the genetic and regulatory mechanisms of the cell cycle of these cell lines are often altered. This may misguide the understanding of the mechanisms underlying the effects of the HBx protein on the physiology of renal tubular epithelial cells and HBV replication. In order to exclude the variation from the transformed and immortalized cell lines and investigate the effects of HBx protein on the cell cycle progression of renal cells, primary renal tubular epithelial cells are more suitable model cells. However, to the best of our knowledge, no previous reports have employed primary renal tubular epithelial cells as model cells to investigate the effects of HBx in renal cells.
Therefore, the current study investigated the specific mechanisms underlying the effects of HBx protein in the regulation of the cell cycle progression of primary renal tubular epithelial cells by determining the expression levels of cell cycle-associated proteins following transfection of rat primary renal tubular epithelial cells with a HBx gene eukaryotic expression vector.
Materials and methods
Materials
Cellulose acetate membrane was purchased from EMD Millipore (Billerica, MA, USA). Collagenase type I, penicillin-streptomycin and epithelial cell growth factor were purchased from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany). Rabbit anti-Cyclin A monoclonal antibody (cat. no. ab181591; 1:2,000), rabbit anti-Cyclin D1 monoclonal antibody (cat. no. ab134175; 1:10,000), rabbit anti-Cyclin E polyclonal antibody (cat. no. ab71535; 1:2,000), rabbit anti-cytokeratin 18 monoclonal antibody (cat. no. ab32118; 1:400) and rabbit anti-HBx polyclonal antibody (cat. no. ab39716; 1:2,000) were all obtained from Abcam (Cambridge, MA, USA). Mouse anti-β actin (cat. no. TA-09; 1:2,000), horseradish peroxidase (HRP) conjugated goat anti-mouse IgG (cat. no. ZB-2305; 1:2,000), HRP goat anti-rabbit IgG (ZB-2301; 1:2,000) secondary antibodies and concentrated DAB kit (cat. no. ZLI-9017) were purchased from ZSGB-Bio, Inc. (Beijing, China). HRP conjugated polymer anti-rabbit IgG antibody (cat. no. SV0002; 1:100) was purchased from Boster Biological Technology (Pleasanton, CA, USA). Dulbecco's Modified Eagle's Medium (DMEM), fetal bovine serum (FBS) and trypsin were purchased from Gibco (Thermo Fisher Scientific, Inc., Waltham, MA, USA). KpnI and EcoRV endonucleases, T4 DNA ligase and TOP10 competent cells were from Beijing Transgen Biotech Co., Ltd. (Beijing, China). SuperSignal West Pico Chemiluminescent Substrate was purchased from Thermo Fisher Scientific, Inc.. Epithelial cell medium (EpiCM) was obtained from Nanjing KeyGen Biotech Co., Ltd. (Nanjing, Jiangsu, China). The HiFiScript cDNA Synthesis kit was purchased from CW Biotech Co., Ltd. (Beijing, China). TRNzol and Mini and Maxi Plasmid kits were from Tiangen Biotech Co., Ltd. (Beijing, China). TaKaRa Ex Taq was purchased Takara Biotechnology Co., Ltd. (Dalian, China). Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) apoptosis kit (cat. no. KGA105) was purchased from Nanjing KeyGen Biotech Co., Ltd. (Nanjing, China).
Animals
A total of 2 male Sprague Dawley (SD) rats (weight, 220 and 240 g; aged 8 weeks old) were purchased from the Experimental Animal Center of the Medical College of Zhejiang University (Hangzhou, China). The rats were raised with free access to water and food in a sterile environment of 25–28°C, 40–60% relative humidity and a 12/12 h light/dark cycle. Prior to the experiment, the rats were fasted for 12 h with free access to water. The study protocol was reviewed and approved by the Institutional Animal Care and Use Committee of Zhejiang University and was in accordance with the guidelines established by the Chinese Council of Animal Care (11).
Separation and culture of primary renal tubular epithelial cells
Rats were euthanized by cervical vertebra dislocation and immersed in 70% (v/v) ethanol for 2–3 min. Two kidneys were removed. Renal cortexes were cut into pieces (~1 mm) and ground on 80 mesh sieves. Following 2–3 washes with PBS, residue on the sieves was collected, pipetted repeatedly and centrifuged at 1,000 × g and room temperature for 5 min. The supernatant was discarded. The pellet was resuspended in 2 ml collagenase type-I (1 mg/ml) and incubated under vibration in a 37°C water bath for 30 min. The mixture was centrifuged at 1,000 × g at room temperature for 3 min. The supernatant was discarded again and the pellet was resuspended in 4 ml DMEM containing 10% (v/v) FBS and 1% (v/v) penicillin-streptomycin (10 mg/ml penicillin and 10 mg/ml streptomycin). Subsequently, primary cells were routinely cultured for 3 days at 37°C and 5% CO2, followed by washing with PBS three times. Trypsin buffer (2 ml, 0.25%), and a mixture of trypsin (0.25%) and EDTA (0.02%), were dropwise added to immerse the cells. Cells were subsequently trypsinized in a 37°C water bath for 1–2 min. 2X EpiCM containing 10% (v/v) FBS, 1% (v/v) epithelial cell growth factor (1 µg/l) and 1% (w/v) penicillin-streptomycin (10 mg/ml penicillin and 10 mg/ml streptomycin) was immediately added. Cells were subsequently subcultured at a ratio of 1:2.
Morphology characterization by immunohistochemical analysis
Cells (2.5×105) were seeded into 24-well plates and covered by coverslips. Following the culturing of cells for 36 h and 7 days time intervals, they were removed and washed with PBS three times. Cells were subsequently fixed in 4% (w/v) cold paraformaldehyde at 4°C for 30 min, permeabilized in 0.5% (w/v) Triton X-100 at 37°C for 5 min and incubated in deionized water containing 3% (w/v) H2O2 at room temperature for 10 min. Cells were subsequently blocked using 5% bovine serum albumin (Beyotime Institute of Biotechnology, Jiangsu, China) at 37°C for 30 min. After rinsing with PBS, cells were incubated in anti-cytokeratin 18 primary antibody (1:400) buffer at 4°C overnight. Cells were incubated in an equal volume of PBS in the control group. After rinsing with PBS, cells were incubated in HRP conjugated polymer anti-rabbit IgG antibody (1:100) buffer at 37°C for 1.5 h. Subsequently, development using a concentrated DAB kit, rinsing, counterstaining using 0.1% hematoxylin at room temperature for 3 min, dehydration using ethanol, transparentization using xylol and mounting were performed in sequence. The coverslips were observed under an optical microscope (XDZ-103; Shanghai Tiancheng Medical Flow Technology Co., Ltd., Shanghai, China) with Image Analysis 11.0 software (Beijing Changheng Rongchuang Technology Co., Ltd., Beijing, China).
Construction of HBx gene pcDNA3.1/myc vector
Primers were designed according to HBV genomic sequence of ayr subtype in GenBank (12) (https://www.ncbi.nlm.nih.gov/pubmed/6300776) and the HBx gene was amplified via polymerase chain reaction (PCR) using TaKaRa Ex Taq according to the manufacturer's protocol. The thermocycling conditions used were as follows: Pre-denaturation for 3 min at 96°C; followed by 23 cycles of denaturation for 20 sec at 95°C, annealing for 20 sec at 58°C, elongation for 30 sec at 72°C; and final extension for 1 min at 72°C. The sequences of primers were as follows: Forward, 5′-GGGGTACCGTGGCAGAGGTGAAAAAGTTGC-3′ and reverse, 5′-GCCGATATCCTAACATTGAGATTCCCGAG-3′. The forward and reverse primers contained KpnI and EcoRV endonuclease cutting sites, respectively. The pcDNA3.1/myc plasmid (Miaolingbio Bioscience & Technology Co., Ltd., Wuhan, China) and the amplified HBx segments were managed by double enzyme digestion with restriction endonucleases KpnI and EcoRV. Electrophoresis was performed and the enzyme-digested products were recovered and purified using an agarose gel DNA recovery kit (Tiangen Biotech Co., Ltd., Beijing, China), according to the manufacturer's protocol. Subsequently, ligation was performed with T4 DNA ligase. The ligated products were transformed into TOP10 competent cells and the cells were cultured based on ampicillin screening. Finally, DNA sequencing was performed by Sangon Bioetch Co., Ltd. (Shanghai, China) for identification.
Extraction of the HBx gene vector
Saved strains were inoculated into 200 ml LB media (Qingdao Hope Bio-Technology Co., Ltd., Qingdao, China) containing 100 mg/l ampicillin at 37°C for 14 h. Colonies containing plasmids on the LB plates were removed and added into 2 ml LB media containing ampicillin (100 µg/ml). When the optical density value of the bacterial solution reached ~2, the solution was centrifuged at 10,000 × g at room temperature for 1 min to collect the bacteria pellets. The pcDNA3.1/myc plasmids were extracted using Mini and Maxi Plasmid kits plasmid according to the manufacturer's protocol.
HBx gene transfection
When primary renal tubular epithelial cells, cultured in 4 ml DMEM containing 10% (v/v) FBS and 1% (v/v) penicillin-streptomycin (10 mg/ml penicillin and 10 mg/ml streptomycin), in 6-well plates reached 90% confluency (1.2×106), the culture medium was replaced by serum-free DMEM medium and cells were incubated again at 37°C for 48 h. The plasmid was gently mixed with Lipofectamine® 3000 (Invitrogen; Thermo Fisher Scientific, Inc.) and culture medium according to the manufacturer's protocol. The mixture was added into the 6-well plates (5 µg plasmid/well). Subsequently, cells were incubated at 37°C for 4 h followed by the replacement of the culture medium with medium containing serum at 37°C for another 48 h.
Determination of HBx and cell cycle-associated gene expression by reverse transcription-quantitative PCR (RT-qPCR)
Cells were divided into three groups: Control, empty and HBx. Cells transfected with empty plasmid pcDNA3.1(+) and recombinant plasmid pcDNA3.1(+)-HBx were designated as the empty and HBx groups, respectively. Cells without transfection served as the control group. Total RNA was extracted from cells (1.2×106) with TRNzol, according to the manufacturer's protocol. Primers sequences are presented in Table I. Reverse transcription was performed with a HiFiScript cDNA Synthesis kit, according to the manufacturer's protocol. qPCR was performed using UltraSYBR Mixture (cat. no. CW0957M; CWBIO, Inc., Beijing, China). Thermocycling parameters were set as follows: Pre-denaturation for 10 min at 95°C, followed by 40 cycles of denaturation for 10 sec at 95°C, annealing for 30 sec at 51.2°C and elongation for 30 sec at 72°C. β-actin served as internal control and the relative level to β-actin was calculated accordingly using classical 2-ΔΔCq method (13).
Table I.
Primers for reverse transcription-quantitative polymerase chain reaction.
| Gene | Primer sequence, 5′-3′ |
|---|---|
| Cyclin E | F: ATCTGGCGGTTGACTTT |
| R: GGCAGCGATGAAGAGTG | |
| Cyclin A | F: ACGAACATGTCGCTAGTGGT |
| R: GTGGGATTTTCTGGGTGTCCT | |
| Cyclin D1 | F: CAGAAGTGCGAAGAGGAA |
| R: GAAGCCAGGAACATACAAG | |
| HBx | F: CACTTCGCTTCACCTCTGC |
| R: TCGGTCGTTGACATTGCTG | |
| β-actin | F: ATCGTCCACCGTAAATGC |
| R: TGAAGTGGTAGTCGGGTG |
F, forward; R, reverse; HBx, hepatitis B virus X protein.
Determination of HBx and cell cycle-associated protein expression by western blot analysis
Cells were also divided into three groups for western blot analysis: Control, empty and HBx. Following transfection, the culture medium was discarded and cells were washed two times with ice-precooled PBS. Subsequently, cells (1.2×106) were scraped and centrifuged at 2,000 × g and 4°C for 5 min. Cell pellets were collected and resuspended in RIPA lysis buffer (cat. no. C1053; Applygen Technologies, Inc., Beijing, China) containing 10% (w/v) phenylmethanesulfonyl fluoride and 1% (w/v) phosphatase inhibitors for 20 min. This mixture was centrifuged at 10,000 × g at room temperature for 10 min and the supernatant was collected to obtain total cell protein. Gel loading buffer (5X) was added into total protein and the mixture was boiled for 5 min to denature proteins. Subsequently, proteins (2 µg per lane) were loaded to perform 10% SDS-PAGE electrophoresis. Proteins were transferred to a polyvinylidene difluoride membrane at 4°C. The membrane was incubated in blocking buffer that consisted of TBS with 0.1% Tween-20 (TBST) containing 5% (w/v) nonfat-dried milk on a horizontal shaker at room temperature for 1.5 h. Subsequently, the membrane was immersed in 3–5 ml with rabbit anti-Cyclin A antibody, rabbit anti-Cyclin D1, rabbit anti-Cyclin E, rabbit anti-HBx, and mouse anti-β actin primary antibodies at 4°C overnight. TBST was used to rinse the membrane three times for 10 min each time. The membrane was incubated with HRP conjugated goat anti-mouse IgG and goat anti-rabbit IgG secondary antibodies at room temperature for 1.5 h. The membrane was rinsed with TBST three times for 10 min each time. Following the addition of ECL solution, the membrane was exposed on a gel imaging system (ChemiDoc XRS; Bio-Rad Laboratories, Inc.). Gray values were determined using Quantity One software (v4.6.2; Bio-Rad Laboratories, Inc.).
Evaluation of cell apoptosis and the cell cycle by flow cytometry
Renal tubular epithelial cells in logarithmic phase were divided into three groups: Control, empty and HBx. Following transfection, cells were cultured for a further 24 h (1.2×106) and trypsinized for 5 min. Cells were centrifuged at 1,200 × g at room temperature for 5 min, washed and resuspended in 500 µl binding buffer (1X). Cells were gently mixed with Annexin V-FITC (5 µl) and incubated at 4°C in the dark for 30 min. After gentle mixing with PI (5 µl), the mixture was incubated at 4°C in the dark for another 5 min. Finally, the cell suspension was immediately evaluated on a flow cytometer (BD FACSCalibur; BD Biosciences, Franklin Lakes, NJ, USA). Data were analyzed using CellQuest software (v.17; BD Biosciences). In order to investigate the cell cycle, cells were cultured following transfection at 37°C for a further 24 h (1.2×106) and subsequently trypsinized for 5 min. Cell were fixed in pre-cooled 70% ethanol at 4°C for 3 h. Cells were then centrifuged at 1,200 × g at room temperature for 5 min, washed with PBS and then resuspended in 1 ml PI (50 µg/ml) containing 0.5% Triton X-100 at room temperature in the dark for 30 min. Finally, the cell suspension was evaluated using a flow cytometer (BD FACSCalibur; BD Biosciences), and data were subsequently analyzed using CellQuest software (v.17; BD Biosciences).
Statistical analysis
All experiments were independently repeated three times and data are presented as the mean + standard deviation. Statistical analysis was performed using SPSS 18.0 software (SPSS, Inc., Chicago, IL, USA). Statistical comparisons among groups were performed by one way analysis of variance followed by Tukey's post-hoc test. P<0.05 was considered to indicate a statistically significant difference.
Results
Morphology characterization of primary renal tubular epithelial cells
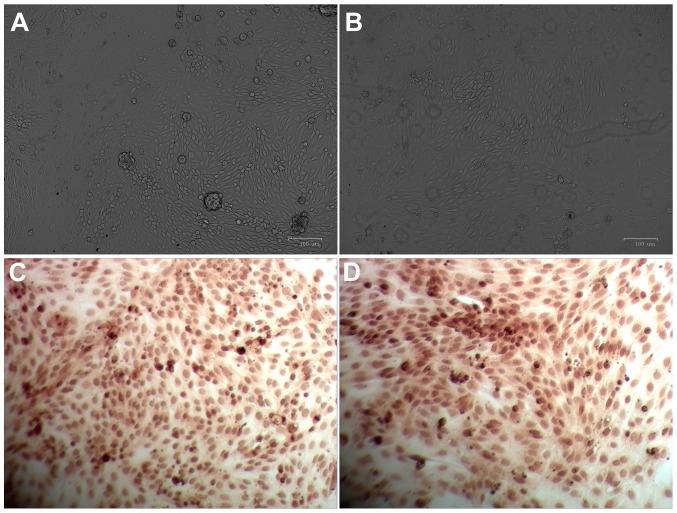
In the present study, cells that exhibited various intensities of non-uniform, scattered brown staining in the cytoplasm and perinuclear region, and with a shape resembling multilateral cobblestones with mutually tight arrangements, were considered to be verified as renal tubular epithelial cells. Images of primary renal tubular epithelial cells are presented in Fig. 1. When primary cells were cultured for 12 h, their anchorage growth was observed. After 24–36 h, epithelioid cells emerged from the periphery of the anchorage area and the whole cell population exhibited an ‘island-like’ distribution (Fig. 1A). A total of 6–7 days post-primary culture, cells grew all over the bottom of the culture bottle. At this time-point, cells were observed to resemble the shape of typical multilateral cobblestones with tight arrangements and high transparency (Fig. 1B). The present study employed immunohistochemical analysis to determine the levels of cytokeratin 18 specifically expressed in renal tubular epithelial cells and the results demonstrated the presence of brown granules with a non-uniform and scattered distribution in the cytoplasm and perinuclear region (Fig. 1C and D). These results indicated that these cells were verified to be renal tubular epithelial cells.
Figure 1.
Morphology characterization of primary renal tubular epithelial cells. Light microscopy images captured following culture for (A) 36 h and (B) 7 days. Immunohistochemical images captured following culture for (C) 36 h and (D) 7 days. At 36 h time intervals, the epithelioid cell population presented with an ‘island-like’ distribution; and at 7 day time intervals, cells were distributed as typical multilateral cobblestones with tight arrangements and high transparency. Brown granules exhibited non-uniform and scattered distributed in the cytoplasm and perinuclear region of cells. Magnification, ×20.
mRNA expression levels of HBx and cell cycle-associated genes
The mRNA levels of HBx and the cell cycle-associated genes cyclin A, cyclin D1 and cyclin E in renal tubular epithelial cells was determined by RT-qPCR. No significant differences were observed between the mRNA levels in the control and empty groups (P>0.05; Fig. 2). However, renal tubular epithelial cells in the HBx group exhibited significantly higher mRNA levels of HBx, cyclin A, cyclin D1 and cyclin E compared with the empty group (P<0.05; Fig. 2).
Figure 2.
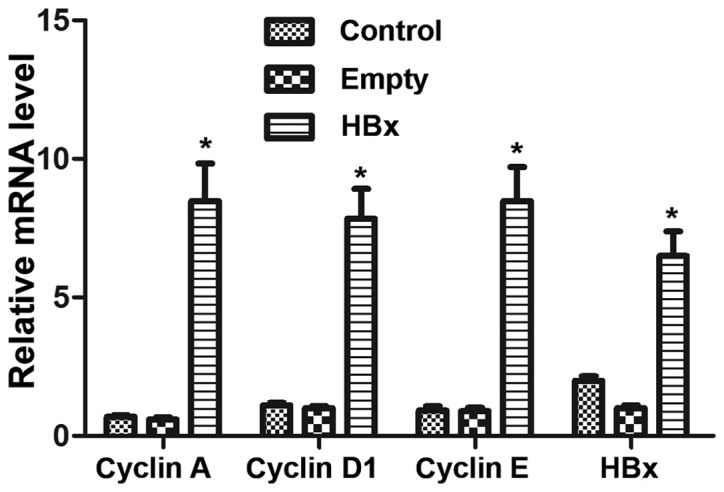
mRNA levels of HBx, cyclin A, cyclin D1 and cyclin E genes in renal tubular epithelial cells in the control, empty and HBx groups were determined by reverse transcription-quantitative polymerase chain reaction. Cells transfected with empty plasmid pcDNA3.1(+) and recombinant plasmid pcDNA3.1(+)-HBx were designated as the empty and HBx groups, respectively. Cells without transfection served as the control group. *P<0.05 vs. empty group. HBx, hepatitis B virus X protein.
HBx and cell cycle-associated protein levels
The protein levels of HBx and the cell cycle-associated proteins cyclin A, cyclin D1 and cyclin E in renal tubular epithelial cells were assessed by western blot analysis. As demonstrated in Fig. 3A, similar blots for β-actin were observed among the control, empty and HBx groups, indicating that the western blotting method was reliable. An obvious dark blot for HBx was observed in the HBx group, but no blot traces of HBx were observed in the control and empty groups. The quantified results demonstrated similar protein levels of cyclin A, cyclin D1 and cyclin E between the control and empty groups (P>0.05; Fig. 3B). Notably, in the HBx group, the protein expression of cyclin A, cyclin D1 and cyclin E were significantly upregulated compared with the empty group (P<0.05; Fig. 3B). Quantitatively, HBx protein was scarcely detected in the control and empty groups, while following HBx gene transfection, renal tubular epithelial cells exhibited significantly increased expression of HBx protein (Fig. 3B).
Figure 3.
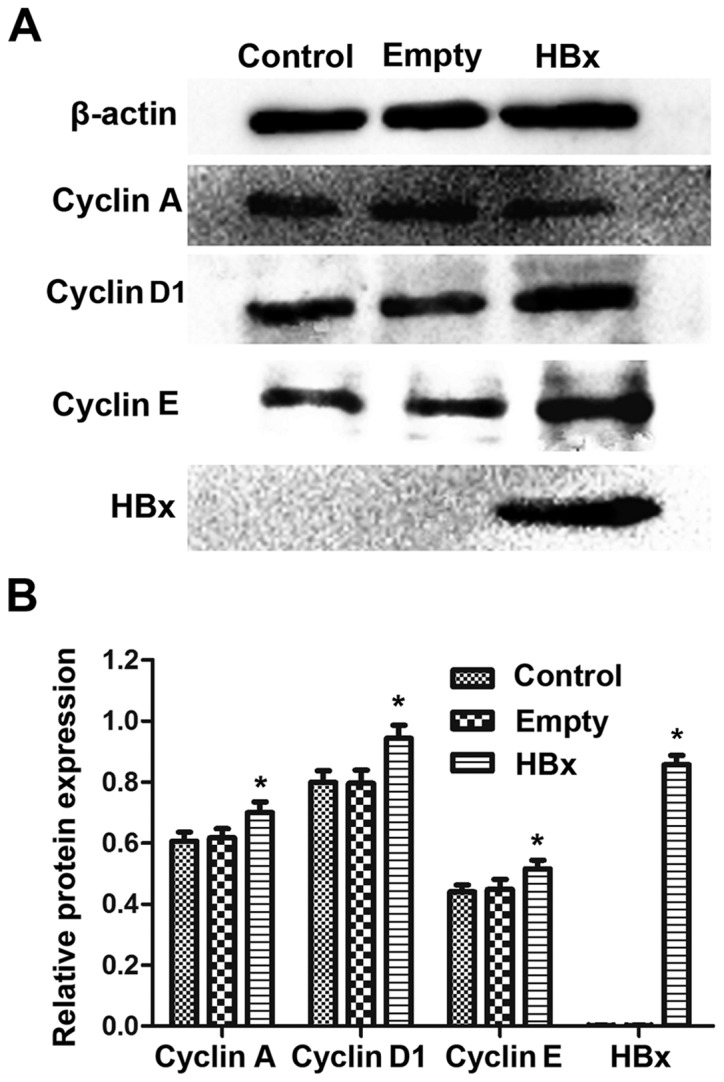
Protein levels of HBx, cyclin A, cyclin D1 and cyclin E in renal tubular epithelial cells in the control, empty and HBx groups were assessed western blot analysis. (A) Representative western blot bands for cyclin A, cyclin D1, cyclin E and HBx in control, empty and HBx groups are presented. (B) Densitometric analysis was performed to quantify the protein expression levels and perform statistical analysis. Cells transfected with empty plasmid pcDNA3.1(+) and recombinant plasmid pcDNA3.1(+)-HBx were designated as the empty and HBx groups, respectively. Cells without transfection served as the control group. *P<0.05 vs. empty group. HBx, hepatitis B virus X protein.
Cell apoptosis
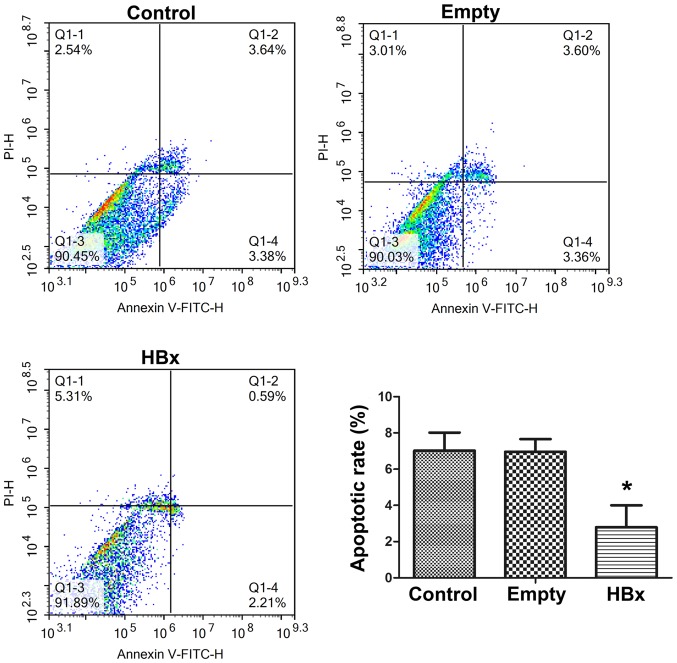
The cell apoptosis of renal tubular epithelial cells in the control, empty and HBx groups was evaluated by Annexin V-FITC/PI double staining and flow cytometry results are presented in Fig. 4. The apoptotic rates of cells in the control, empty and HBx groups were 7.02±1.02, 6.96±0.86 and 2.80±0.24%, respectively. Cells in the control and empty groups exhibited similar apoptotic rates, demonstrating that the transfection of the empty plasmid did not affect the apoptosis of renal tubular epithelial cells. Compared with the empty group, the apoptotic rate was markedly reduced by >60% following the transfection of the plasmid containing the HBx gene (P<0.05; Fig. 4), indicating that HBx gene transfection was able to effectively suppress the apoptosis of renal tubular epithelial cells.
Figure 4.
Cell apoptosis of renal tubular epithelial cells in the control, empty and HBx groups was evaluated by Annexin V-FITC/PI double staining and subsequent flow cytometry. Cells transfected with empty plasmid pcDNA3.1(+) and recombinant plasmid pcDNA3.1(+)-HBx were designated as the empty and HBx groups, respectively. Cells without transfection served as the control group. Q1-2 and Q1-4 quadrants represent cells undergoing apoptosis. *P<0.05 vs. empty group. HBx, hepatitis B virus X protein; FITC, fluorescein isothiocyanate; PI, propidium iodide.
Cell cycle
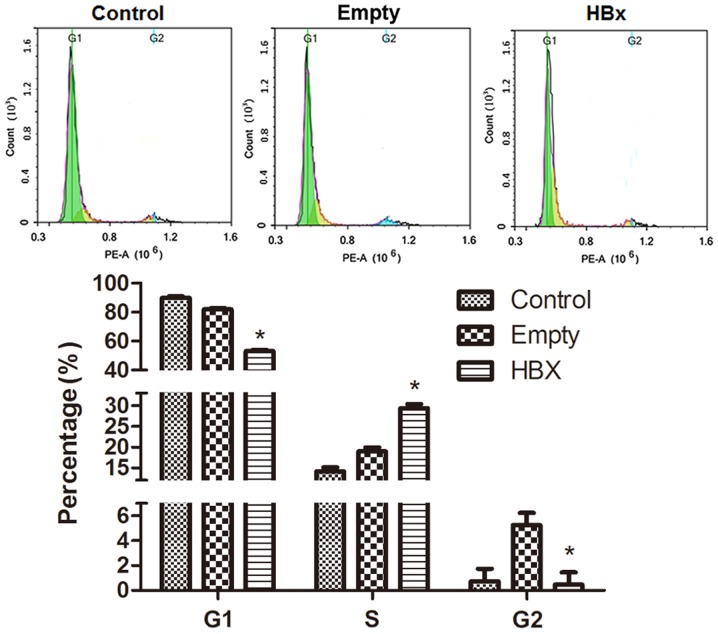
The cell cycle of renal tubular epithelial cells in the control, empty and HBx groups was evaluated by flow cytometry and results are presented in Fig. 5. The green peak area on the left, yellow peak area in the middle and blue peak area on the right represent cells in G1, S and G2 phases, respectively. The distribution of the cell cycle in the control group was comprised of G1 89.38% (Freq G1), S 14.15% (Freq S) and G2 0.74% (Freq G2); the majority of cells were in the presynthetic phase, a few cells were in the synthetic phase and a limited number of cells were in the postsynthetic phase. In the empty group, cells numbers in the S and G2 phases were marginally augmented. However, when cells were transfected with the HBx gene, G1 and S phase cell percentages were markedly decreased and increased, respectively, while the percentage in the G2 phase was similar to the control group. The results indicated that HBx gene transfection promoted the progression of renal tubular epithelial cells from the G1 to S phase.
Figure 5.
The cell cycle distribution of renal tubular epithelial cells in the control, empty and HBx groups was evaluated by flow cytometry. Cells transfected with empty plasmid pcDNA3.1(+) and recombinant plasmid pcDNA3.1(+)-HBx were designated as the empty and HBx groups, respectively. Cells without transfection served as the control group. *P<0.05 vs. empty group. HBx, hepatitis B virus X protein.
Discussion
Chronic hepatitis B in patients is usually associated with various types of extrahepatic damage, and HBV-GN is one of the most common forms of extrahepatic damage. However, an understanding of the risk factors and pathogenesis of HBV-GN remain unclear, and effective preventive and therapeutic strategies are also yet to be established (14). At present, direct infection of the kidney by HBV and subsequent immunological dysfunction induced by the infection are considered to be the major pathogenic causes of HBV-GN (15,16). In the nephridial tissues of patients with HBV-GN, obvious tubulointerstitial lesions were identified, which presented as inflammatory cell infiltration and interstitial fibrosis (17). The severity of interstitial lesions was reported to be positively associated with the infiltration degree of inflammatory cells and atrophy of renal tubular epithelial cells (HK-2 cells) was observed in the pathological tissues (17). The results of previous studies indicated that human renal tubular epithelial cells cultured in vitro were susceptible to HBV and HBV enhanced the secretion of transforming growth factor-β1 by human renal tubular epithelial cells (18,19). Other reports revealed that HBx protein was expressed in the renal tubular epithelial cells of patients with HBV-GN and was primarily distributed in the cytoplasm (20,21). Furthermore, its expression level in the renal tubular epithelial cells was demonstrated to be higher compared with in glomerular cells (20,21). Therefore, HBx may be principally replicated and transcribed in situ within renal tubular epithelial cells and the damage of renal tubular epithelial cells may be generated in this process.
In order to elucidate the damage mechanism of HBx on renal tubular epithelial cells, a renal tubular epithelial cell model with HBx transfection was established. It was previously reported that HBx transfection promoted the transdifferentiation of HK-2 cells, indicating that HBx may alter the microenvironment of HK-2 cells and facilitate epithelial-mesenchymal transition (22,23). Although the evolution from the damage of renal tubular epithelial cells to the final reduction of cell number was regulated by multiple mechanisms, the ultimate effect was altered cell cycle progression (24). The cell cycle is composed of a resting phase (G0), DNA presynthetic phase (G1), DNA synthetic phase (S), DNA postsynthetic phase (G2) and mitotic phase (M). Intervention in cell cycle progression influences the replication of HBV and, in addition, HBx protein was demonstrated to alter the cell apoptosis process of renal tubular epithelial cells during the pathological process of renal tubular interstitial fibrosis induced by HBV (25). Therefore, analysis of alterations in the expression of cell cycle-associated proteins and confirmation of the association with HBV replication level in vivo are required to investigate the mutual effects between HBx protein and the cell cycle.
Cyclins are a family of proteins with cell cycle-specific or temporal expression and catabolism. The cell cycle is regulated by various cyclins. Cyclin A regulates the transition from G1 to S phase, which is associated with DNA replication (26), while cyclin D1 and cyclin E are associated with G1 phase. Upregulation of cyclin A, cyclin D1 and cyclin E will facilitate the cell transition from G1 to S phase (27). The tumor suppressor genes p16 and p21 compete with multiples cyclins to bind to various cyclin-dependent kinase sites, inhibit the activation of cyclin and arrest the cell cycle in the G1 phase, which consequently regulates cells division and differentiation, and prevents the excessive proliferation of cells (28,29).
As a type of trans-acting factor with extensive biological functions, the HBx protein has been reported to activate intracellular signal transduction systems and regulate the cell proliferation through binding to cell transcription factors (30). In the past, it was widely considered that HBx may accelerate cell cycle progression and cell proliferation through shortening the G0/G1 phase. However, Clippinger et al (6) demonstrated that the effect of HBx on cell apoptosis was bidirectional in hepatocytes. Zhai et al (4) further revealed that the effect of HBx on the cell cycle was dose-dependent, but the specific mechanisms of this effect remain unclear (31).
Therefore, in the present study, HBx was transfected into primary renal tubular epithelial cells. Following HBx transfection, the mRNA expression of cyclin A, cyclin D1 and cyclin E were markedly increased, and the corresponding protein levels were also upregulated. These results indicated that HBx may influence the cell cycle progression of renal tubular epithelial cells by regulating the expression of cyclins and causing a rapid transition of cells from G0 to S phase via G1 phase. Thus, the present study also investigated the levels of cell apoptosis and cell cycle distribution, and the results demonstrated that HBx transfection markedly promoted the progression of renal tubular epithelial cells from the G1 to S phase and suppressed the cell apoptosis, which was consistent with the results of RT-qPCR and western blot analysis of cyclin expression. These results indicated that HBx protein may function in altering cell cycle progression.
Although the current study demonstrated that HBx protein may act on cyclin A, cyclin E and cyclin D1 to regulate the cell cycle of renal tubular epithelial cells, the further specific regulation mechanism requires future exploration. For example, the results of the present study do not indicate whether the HBx protein affects the expression of cyclin A, cyclin E and cyclin D1 by competitive binding or altering upstream signaling. However, dysfunction of the tumor suppressor gene p53 was reported to be associated with the overexpression of HBx (32,33). Our future studies will focus on the effects of p53, p21 and p16 genes on the HBx protein and HBV replication in primary renal tubular epithelial cells.
The present study revealed the effects of HBx on the cell cycle progression of primary renal tubular epithelial cells by transfection with HBx gene pcDNA3.1/myc vector. We also hypothesized that the HBx gene may integrate into the genomic DNA of hosts by recombination. As a multifunctional protein, HBx may function in the proliferation or apoptosis of host cells through stimulating signal transduction pathways in the cytoplasm and the activation of transcription factors in the nucleus (34). However, further studies are required to confirm whether the Hbx gene is able to integrate into the genomic DNA of host cells.
In conclusion, primary renal tubular epithelial cells of rats were separated and cultured to investigate the effect of HBx gene transfection on the expression of cyclins, cell apoptosis and the cell cycle. Consequently, HBx gene transfection was demonstrated to regulate the apoptosis and cell cycle of primary renal tubular epithelial cells by influencing the expression of cyclins. These results may improve the understanding of the pathogenesis of HBV-GN and may also provide insight and theoretical support for the design and development of future hepatitis B virus drugs.
Acknowledgements
Not applicable.
Funding
The present study was financially supported by the 2015 General Medical and Health Research Program of Zhejiang Province (Class A), Health and Family Planning Commission of Zhejiang Province, China (grant no. 2015KYA043).
Availability of data and materials
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.
Authors' contributions
WH and ML designed the study and wrote the manuscript. WH, ML, MH, YZhu, YZho, HD, GH, LL, QC and YL performed the experiments. MH, YZhu, YZho and HD collected and analyzed data.
Ethics approval and consent to participate
The study protocol was reviewed and approved by the Institutional Animal Care and Use Committee of Zhejiang University (Hangzhou, China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Zhang Q, Xue C. Research progress in pathogenesis of hepatitis B virus associated nephritis. Med Recap. 2013:2543–2545. [Google Scholar]
- 2.Yi Z, Jie YW, Nan Z. The efficacy of anti-viral therapy on hepatitis B virus-associated glomerulonephritis: A systematic review and meta-analysis. Ann Hepatol. 2011;10:165–173. [PubMed] [Google Scholar]
- 3.Seeger C, Mason WS. Hepatitis B virus biology. Microb Mol Biol Rev. 2000;64:51–68. doi: 10.1128/MMBR.64.1.51-68.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhai LL, Liu J, Xie YH. Dose-dependent modulation of cell apoptosis by hepatitis B virus X protein. J Microb Infect. 2011:72–77. [Google Scholar]
- 5.Xia LM, Huang WJ, Wu JG, Yang YB, Zhang Q, Zhou ZZ, Zhu HF, Lei P, Shen GX, Tian DA. HBx protein induces expression of MIG and increases migration of leukocytes through activation of NF-kappa B. Virology. 2009;385:335–342. doi: 10.1016/j.virol.2008.11.042. [DOI] [PubMed] [Google Scholar]
- 6.Clippinger AJ, Gearhart TL, Bouchard MJ. Hepatitis B virus X protein modulates apoptosis in primary rat hepatocytes by regulating both NF-kappa B and the mitochondrial permeability transition pore. J Virology. 2009;83:4718–4731. doi: 10.1128/JVI.02590-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tan C, Guo H, Zheng M, Chen Y, Huang W. Involvement of mitochondrial permeability transition in hepatitis B virus replication. Virus Res. 2009;145:307–311. doi: 10.1016/j.virusres.2009.08.001. [DOI] [PubMed] [Google Scholar]
- 8.He P, Shang H, Li D, Feng G. Effects of HBx gene on proliferation and apoptosis of human renal tubular epithelial cells. Chin J Nephrol. 2013;29:380–381. [Google Scholar]
- 9.Chen HY, Tang NH, Lin N, Chen ZX, Wang XZ. Hepatitis B virus X protein induces apoptosis and cell cycle deregulation through interfering with DNA repair and checkpoint responses. Hepatol Res. 2008;38:174–182. doi: 10.1111/j.1872-034X.2007.00213.x. [DOI] [PubMed] [Google Scholar]
- 10.Huang YQ, Wang LW, Yan SN, Gong ZJ. Effects of cell cycle on telomerase activity and on hepatitis B virus replication in HepG22.2.15 cells. Hepatobiliary Pancreat Dis Int. 2006;3:543–547. [PubMed] [Google Scholar]
- 11.General Administration of Quality Supervision, Inspection and Quarantine of China: Laboratory animal-Guideline of welfare ethical review (Draft for approval) 2017 Mar 18; [Google Scholar]
- 12.Ono Y, Onda H, Sasada R, Igarashi K, Sugino Y, Nishioka K. The complete nucleotide sequences of the cloned hepatitis B virus DNA; subtype adr and adw. Nucleic Acids Res. 1983;11:1747–1757. doi: 10.1093/nar/11.6.1747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C (T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 14.He P, Li H, Li D, Feng G. HBx gene promotes apoptosis in HK-2 cells through regulating apoptosis-relatedprotein ratio of Bax/Bcl-2. J Chin Med Univer. 2014;43:38–44. [Google Scholar]
- 15.An S, Zhang R, Yang W, Zhou H, Zhang Z, Yang Y, Guo X. Research advances in hepatitis B virus-associated glomerulonephritis. J Clin Hepatol. 2016;32:366–369. [Google Scholar]
- 16.Jiang W, Xu Y, Guan G, Ma R, Dong H. Renal amyloidosis and hepatitis B virus-associated glomerulonephritis in a patient with nephrotic syndrome. Chin Med J (Engl) 2014;127:1199. [PubMed] [Google Scholar]
- 17.Deng CL, Song XW, Liang HJ, Feng C, Sheng YJ, Wang MY. Chronic hepatitis B serum promotes apoptotic damage in human renal tubular cells. World J Gastroenterol. 2006;12:1752–1756. doi: 10.3748/wjg.v12.i11.1752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhu N, Zhou Y, Yuan WJ, Liu J, Shang MH, Wang L, Gu LJ. Toll-like receptor 4 deposition and its significance in hepatitis B virus associated nephropathy. Zhonghua Nei Ke Za Zhi. 2011;50:1008–1012. (In Chinese) [PubMed] [Google Scholar]
- 19.Han WL, Huang ZX. Experimental study of direct pathogenic effects of hepatitis B virus on human tubular kidney cell. J Wenzhou Med College. 2008;38:144–147. [Google Scholar]
- 20.Han WL, Huang ZX. Infection of hepatitis B virus on human tubular kidney cells in vitro. J Zhejiang Med Univer. 2010;32:1148–1150. [Google Scholar]
- 21.Hong L, Zhang J, Li Q, Min J, Lu J, Li F, Li H, Guo S. Role of NF-κB activiation by MHBst167/HBx in human renal tubular cells. Chin J Nephrol Dial Transplant. 2010;19:135–141. [Google Scholar]
- 22.Hong L, Zhang J, Min J, Lu J, Li F, Li H, Guo S, Li Q. A role for MHBst167/HBx in hepatitis B virus-induced renal tubular cell apoptosis. Nephrol Dial Transplant. 2010;25:2125–2133. doi: 10.1093/ndt/gfp737. [DOI] [PubMed] [Google Scholar]
- 23.Zhou Y, Wang X, Yuan W, Zhu N. Effect of hepatitis B virus X gene on transdifferentiation of human proximal tubular epithelial cells. Chin J Nephrol. 2012;28:956–960. [Google Scholar]
- 24.Zhou Y, Zhu N, Wang X, Wang L, Gu LJ, Yuan WJ. The role of the toll-like receptor TLR4 in hepatitis B virus-associated glomerulonephritis. Arch Virol. 2013;158:425–433. doi: 10.1007/s00705-012-1508-3. [DOI] [PubMed] [Google Scholar]
- 25.Marshall CB, Shankland SJ. Cell cycle regulatory proteins in podocyte health and disease. Nephron Exp Nephrol. 2007;106:e51–e59. doi: 10.1159/000101793. [DOI] [PubMed] [Google Scholar]
- 26.Madden CR, Slagle BL. Stimulation of cellular proliferation by hepatitis B virus X protein. Dis Markers. 2001;17:153–157. doi: 10.1155/2001/571254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhan SD, Huang JX. Progression of Cyclin A, PTEN, Survivin and p27 expression in esophageal cancer. J Modern Oncol. 2014:2245–2248. [Google Scholar]
- 28.Li YJ, Li YY. Advances in the research of cyclin E and p27 in gynecological. Foreign Med Sci (Obstet Gynecol Fascicle) 2007:177–180. [Google Scholar]
- 29.Lin T. Fujian Med Univer. Fuzhou: 2011. Expression and significance of Cell cycle regulatory proteins in the hepatitis B virus associated nephrits; p. 52. [Google Scholar]
- 30.Wu RS. Cell cycle regulation of p16, cdk4 and its role in the formation of middle ear cholesteatoma. J Basic Med Shandong Univer. 2004:53–55. [Google Scholar]
- 31.Lu HZ, Liu D, Zhou JH. Effects of HBV X protein on glomerular mesangial cell proliferation and proinflammation factor expression. Chongqing Med. 2011;40:2227–2230. [Google Scholar]
- 32.Matsuda Y, Ichida T. Impact of hepatitis B virus X protein on the DNA damage response during hepatocarcinogenesis. J Med Mol Morphol. 2009;42:138–142. doi: 10.1007/s00795-009-0457-8. [DOI] [PubMed] [Google Scholar]
- 33.Park SG, Min JY, Chung C, Hsieh A, Jung G. Tumor suppressor protein p53 induces degradation of the oncogenic protein HBx. Cancer Lett. 2009;282:229–237. doi: 10.1016/j.canlet.2009.03.019. [DOI] [PubMed] [Google Scholar]
- 34.Bouehard MJ, Sehneider RJ. The enigmatic X gene of hepatitis B virus. J Virol. 2004;78:12725–12734. doi: 10.1128/JVI.78.23.12725-12734.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.