Abstract
Objectives
Staphylococcus aureus (S. aureus) is the most commonly implicated organism in septic arthritis, a condition that may be highly destructive to articular cartilage. Previous studies investigating laboratory and clinical strains of S. aureus have demonstrated that potent toxins induced significant chondrocyte death, although the precise toxin or toxins that were involved was unknown. In this study, we used isogenic S. aureus mutants to assess the influence of alpha (Hla)-, beta (Hlb)-, and gamma (Hlg)-haemolysins, toxins considered important for the destruction of host tissue, on in situ bovine chondrocyte viability.
Methods
Bovine cartilage explants were cultured with isogenic S. aureus mutants and/or their culture supernatants. Chondrocyte viability was then assessed within defined regions of interest in the axial and coronal plane following live- and dead-cell imaging using the fluorescent probes 5-chloromethylfluorescein diacetate and propidium iodide, respectively, and confocal laser-scanning microscopy.
Results
Hla-producing mutants caused substantial chondrocyte death compared with the toxin-deficient control (Hla-Hlb-Hlg-), whilst mutants producing Hlb and Hlg in the absence of Hla induced minimal chondrocyte death. Coronal studies established that Hla-induced chondrocyte death started in the superficial zone of cartilage and spread to deeper layers, whereas Hlb and Hlg toxins were without significant effect.
Conclusion
This study identified Hla as a highly potent S. aureus toxin that caused rapid chondrocyte death in bovine cartilage, with other toxins or metabolic products produced by the bacteria playing a minor role. The identification of Hla in mediating chondrocyte death may assist in the development of therapeutic strategies aimed at reducing the extent of cartilage damage during and after an episode of septic arthritis.
Cite this article: I. D. M. Smith, K. M. Milto, C. J. Doherty, S. G. B. Amyes, A. H. R. W. Simpson, A. C. Hall. A potential key role for alpha-haemolysin of Staphylococcus aureus in mediating chondrocyte death in septic arthritis. Bone Joint Res 2018;7:457–467. DOI: 10.1302/2046-3758.77.BJR-2017-0165.R1.
Keywords: Septic arthritis, Articular cartilage, Chondrocyte death, Haemolysins, Confocal microscopy
Article focus
Septic arthritis can cause significant joint destruction, with Staphylococcus (S.) aureus being the causative organism in most cases.
A previous in vitro study demonstrated that unspecified S. aureus toxins induced significant in situ chondrocyte death.
Studies by others investigating a variety of S. aureus-related infections have identified alpha (Hla)-, beta (Hlb)-, and gamma (Hlg)-haemolysins as potentially key damage-promoting toxins.
Key messages
In situ chondrocyte viability was assessed by confocal laser-scanning microscopy in bovine cartilage explants exposed to isogenic S. aureus mutants and their isolated culture supernatants.
The results strongly suggest that Hla alone was the key chondrocyte death-inducing toxin, with Hlb and Hlg having no significant effect.
This provides insight to support the development of future therapeutic strategies aimed at reducing the extent of cartilage damage during and after an episode of septic arthritis.
Strengths and limitations
This study enabled the focused assessment of the influence of Hla, Hlb, and Hlg on in situ chondrocyte viability in the absence of the complexities of a host immune response.
A limitation of the study is the extrapolation of conclusions from a bovine cartilage explant model to the clinical setting, as the sensitivity of human chondrocytes to S. aureus haemolysins may be different.
Introduction
Septic arthritis may lead to permanent damage of articular cartilage.1,2 The current goal of treatment is to eradicate bacteria and associated toxins promptly, through a combination of joint lavage and intravenous antibiotics.3 However, despite these strategies, some cartilage damage, and thus joint injury, develops in up to 50% of cases.4-6 Whilst a range of bacterial species may cause septic arthritis,4 Staphylococcus (S.) aureus is most commonly implicated, accounting for 40% to 65% of cases.7,8 S. aureus is a highly virulent Gram-positive pathogen known to produce an array of virulence factors,9,10 which enable it to establish infection and contribute to the destruction of tissue.
Using a bovine cartilage explant model of S. aureus-induced septic arthritis, we previously identified that S. aureus toxins from both laboratory and clinical strains, isolated from patients with septic arthritis, induced significant chondrocyte death. However, the exact nature of these toxin(s) was not determined. Identification of the toxin(s) responsible for the chondrocyte death would represent an important step forward in the development of new therapeutic strategies aimed at reducing the extent of cartilage damage during and after an episode of septic arthritis.
Studies by others investigating a variety of S. aureus-related infections have implicated alpha (Hla)-, beta (Hlb)-, and gamma (Hlg)-haemolysins as key damage-promoting toxins,9,11-15 raising the possibility that these toxins may contribute to chondrocyte death. Hla forms cylindrical transmembrane heptameric pores,16,17 which permit the influx and efflux of ions and small molecules, culminating in disruption of the ionic equilibrium, osmotic swelling, and the death of cells.18 Similarly, Hlg is also a pore-forming toxin, although it has two functional bicomponent toxins (HlgAB and HlgCB) arising from three different subunits: HlgA, HlgB, and HlgC.19 In contrast, Hlb is a Mg2+-dependent sphingomyelinase C that degrades sphingomyelin in the outer phospholipid layer of the cell membrane.20
In the present work, we used the in vitro bovine cartilage explant model of S. aureus-induced septic arthritis to clarify the roles of Hla, Hlb, and Hlg on in situ chondrocyte viability.21 This model avoids the complexities of a host immune response, permitting a clearer assessment of their direct effects on cartilage. Appropriate isogenic S. aureus mutant strains, with respect to Hla, Hlb, and Hlg expression, all originating from the laboratory ‘wild-type’ strain 8325-4 through site-directed mutagenesis, were specifically chosen to test the roles of these toxins. S. aureus 8325-4 is a well-characterized prophage-cured derivative of strain NCTC8325 that produces large amounts of Hla, Hlb, and Hlg,15,22,23 and has chondrocyte-damaging potential of comparable potency to clinical strains of S. aureus.21 Following cartilage explant culture with isogenic mutants or their culture supernatants, in situ chondrocyte viability was visualized and quantified by confocal laser-scanning microscopy (CLSM). The hypothesis was that pore-forming Hla alone was the dominant destructive toxin to in situ chondrocyte viability.
Materials and Methods
Biochemicals and solutions
Biochemicals were obtained from Invitrogen Ltd. (Paisley, United Kingdom) unless otherwise stated. The culture medium was serum-free Dulbecco’s Modified Eagle’s Medium (DMEM; catalogue no: 41966029). The fluorescent dyes 5-chloromethylfluorescein diacetate (CMFDA) and propidium iodide (PI), were prepared as 1 mM stocks using dimethyl sulphoxide or water, respectively. Formaldehyde solution (4% volume/volume in normal saline) was obtained from Fisher Scientific (Loughborough, United Kingdom). The standard bacterial culture media were tryptone soya agar (TSA) and tryptone soya broth (TSB; Oxoid Ltd, Basingstoke, United Kingdom). Tetracycline (2 µg/ml) and erythromycin (10 µg/ml) were obtained from Sigma-Aldrich (Gillingham, United Kingdom) and added to TSA and TSB depending on the ‘mutation-coupled’ antibiotic resistance profile of the isogenic mutant investigated (Table I).14,22-24 Bacteria were stored at -80°C in 10% weight/volume skimmed milk (Oxoid Ltd).
Table I.
Staphylococcus aureus ‘haemolysin-knockout’ isogenic mutant strains, originating from wild-type 8325-4, used in the study
| Staphylococcus aureus strain | Genotype | Phenotype/given name | Source/reference |
|---|---|---|---|
| 8325-4 | Wild type | Hla+ Hlb+ Hlg+ | Novick22 |
| DU1090 | hla::Emr | Hla- Hlb+ Hlg+ | O'Reilly et al23 |
| DU5720 | hla::Emr hlb::φ42E | Hla- Hlb- Hlg+ | Patel et al24 |
| DU5938 | hlb::φ42E hla::Emr ΔhlgABC::Tcr | Hla- Hlb- Hlg- | Nilsson et al14 |
| DU5946 | hlb::φ42E ΔhlgABC::Tcr | Hla+ Hlb- Hlg- | Nilsson et al14 |
Tcr, Tetracycline resistance; Emr, Erythromycin resistance; Hla, alpha-haemolysin; Hlb, beta-haemolysin; Hlg, gamma-haemolysin
Bacterial strains
S. aureus strain 8325-4 and its associated isogenic mutants, containing combined mutations affecting Hla (hla::Emr), Hlb (hlb::φ42E), and Hlg (ΔhlgACB::Tcr) synthesis (Table I), were kindly provided by Professor Timothy J. Foster, Department of Microbiology, Trinity College, Dublin, Ireland. Mutants were established through site-directed mutagenesis of the Hla, Hlb, and Hlg genes, thereby rendering these genes inactive. All mutant strains were tested rigorously for phenotype and genotype at the time of creation by Southern hybridization and Western immunoblotting techniques.14,23-25
Erythromycin and tetracycline resistance associated with Hla and Hlg mutations, respectively, enabled the selective growth of specific mutants in antibiotic-loaded culture media. Nevertheless, prior to experimentation, the haemolysin production phenotype of the isogenic mutants was assessed by rabbit and sheep erythrocyte haemolysis assays. Fresh heparinized rabbit and sheep blood, obtained from the Scottish National Blood Transfusion Service (Penicuik, United Kingdom) and E&O Laboratories Ltd (Bonnybridge, United Kingdom), respectively, was used and red blood cell suspensions of approximately 5% haematocrit prepared in Hank’s balanced salt solution (HBSS; pH 7.4).
Bacterial strains were streaked onto TSA plates with or without tetracycline (2 µg/ml) and erythromycin (10 µg/ml), as appropriate. Following 24 hours of incubation (37°C), TSB (10 ml) was inoculated with several individual bacterial colonies from the 24 hours TSA plate of a given bacterial strain, and cultured in a shaking incubator (37°C; 24 hours). The 24-hour cultures were then centrifuged (3400 × g; ten minutes) and supernatants harvested and filter-sterilized (0.2 µm). Supernatants (250 µl) were then added to 5% haematocrit suspension (250 µl), to give a haematocrit of 2.5%, and a 50% supernatant dilution, mixed gently and incubated (37°C) for 12 hours. The addition of HBSS (250 µl) to 5% haematocrit suspension (250 µl) acted as the control. Following incubation, samples were centrifuged (8000 × g; one minute) and aliquots were taken for determination of haemoglobin at Abs540 on a Nanodrop spectrophotometer (Thermo Fisher Scientific, Waltham, Massachusetts). Haemolysis, as a percentage of complete lysis, was calculated using the formula: (Abs540 of sample/Abs540 of 100% haemolysis) × 100%. Complete (100%) lysis was achieved by freezing (-20°C; 12 hours) and thawing appropriate 2.5% erythrocyte suspensions.
Preparation of bacterial aspirates
When required, bacteria were thawed and streaked onto TSA plates with or without tetracycline (2 µg/ml) and erythromycin (10 µg/ml), as appropriate. Following 24 hours of incubation (37°C), TSB, containing antibiotics as required, was inoculated with several individual bacterial colonies from the 24-hour TSA plate of a given bacterial strain and cultured in a shaking incubator (37°C; 24 hours). Serial dilutions, to a maximum of 10-6, were performed on the 24-hour TSB culture in order to calculate the number of colony forming units (cfu) in 1 ml of 24-hour TSB. Thereafter, 100 µl of 10-4, 10-5, and 10-6 dilutions were spread onto TSA plates and incubated (37°C; 24 hours). Bacterial counts taken with a colony counter were performed on a number of cultures for each mutant strain that was studied and a count of approximately 1.0 × 109 cfu/ml was routinely obtained. Based on these results, a fresh 24-hour culture of each strain grown in 10 ml TSB was diluted in DMEM to produce a final bacterial concentration of approximately 1.0 × 105 cfu/ml DMEM.
Bovine osteochondral explants
Metacarpophalangeal joints of three-year-old cows were washed, skinned, de-hoofed, and opened under sterile conditions within six hours of slaughter. Only healthy joints, with no evidence of cartilage damage or degeneration, were used. Osteochondral explants with full depth cartilage and attached subchondral bone, were harvested from the convex weight-bearing articular surface (Fig. 1). As the thickness, cell density, physical characteristics, and biochemical characteristics of articular cartilage have been shown to vary according to mechanical load,26,27 explants were harvested from weight-bearing surfaces only in order to permit standardization.
Photographs showing osteochondral explant harvesting from the bovine metacarpophalangeal joint: a) a sterile number-24 scalpel blade was used to acquire explants from the proximal limb; b) due to the convex nature of the number-24 scalpel blade, a steady forward and backward ‘rocking’ movement of the blade yielded explants with approximately 0.5 mm subchondral bone attached; c) the explants were immediately transferred to a vessel containing sterile Dulbecco’s Modified Eagle’s Medium using the harvesting blade i.e. a ‘no touch’ technique; d) an example of a typical oval explant (scale bar = 1 cm).
Bacterial culture studies
Explants from each joint were placed into separate tissue culture flasks containing DMEM. Then, 25 µl (approximately 2.5 × 103 cfu) of a given isogenic mutant aspirate was injected into each flask. This concentration was chosen as control experiments identified that it produced a measurable degree of chondrocyte death within a reasonable time period that was neither overwhelming nor weak.21 Flasks were then incubated (37°C; 5% CO2) for 40 hours.
Bacterial culture-supernatant studies
The 40-hour cultures of specified isogenic mutant strains were pooled and centrifuged (3400 × g; ten minutes). The supernatant, containing the toxins, was then harvested and filter-sterilized. Osteochondral explants were subsequently incubated (37°C; 5% CO2) with the supernatants (3 ml of neat supernatant per experimental container) for six hours.
Fluorescent probes and fixation
At zero, 18, 24, and 40 hours (bacterial culture studies) or zero, two, four, and six hours (supernatant studies), explants were aseptically removed and trimmed so as to create two straight edges. Explants were then incubated (21°C; one hour) in penicillin (50 U/ml)-containing and streptomycin (50 µg/ml)-containing DMEM with CMFDA (10 µM) and PI (10 µM), to label living or dead chondrocytes green or red, respectively.28,29 Explants were subsequently fixed in 4% formaldehyde prior to storage at 4°C in phosphate buffered saline (PBS). For CLSM, explants were secured to the base of a Petri dish with Blu Tack (Bostik, Leicester, United Kingdom) and re-submerged in PBS.
CLSM
An upright Zeiss LSM510 Axioskop (Carl Zeiss Ltd, Welwyn Garden City, United Kingdom) CLSM, fitted with a ×10/0.3 dry objective, was used to acquire optical sections of CMFDA- and PI-labelled in situ chondrocytes in either the axial or coronal plane to a depth of 100 µm using established methods.21,28,29
Quantification of in situ chondrocyte death
For reconstructed 3D axial projections, the percentage cell death (number of dead cells/total number of living and dead cells) × 100) was calculated within a ‘field-of-view’ region of interest (ROI)21 measuring 921 µm × 461 µm × 100 µm (x-y-z axes, respectively) using Volocity 4 imaging software (Improvision, Coventry, United Kingdom), as previously described.21 For coronal projections, the percentage cell death was quantified at 100 µm intervals, within a ROI measuring 921 µm × 100 µm × 100 µm (x-y-z axis, respectively), to a depth of 400 µm from the surface of the cartilage.21 For each ROI, objects (individual cells) in both the green (living cells) and red (dead cells) channels were identified and counted using established methods.21,29
Statistical analysis
These were performed using SigmaPlot version 12 (Systat Software Inc, Chicago, Illinois); N referred to the number of feet obtained from independent animals (independent experiments) and n to the number of explants analyzed per experimental group at each timepoint. Replicates of explants for an experiment from the same animal were averaged to obtain a single observation for that animal. Parametric data were analyzed using one-way between-groups analysis of variance, with post hoc Dunnett’s test, and unpaired Student’s two-tailed t-tests. Wilcoxon signed-rank and Mann–Whitney U tests were used to analyze paired or non-paired non-parametric data, respectively. Data are presented as means and sd with the significance set at p < 0.05.
Results
Assessment of the toxin profiles of isogenic mutant strains
Hla, Hlb, and Hlg can be differentiated on the basis of the sensitivity of erythrocytes from different animal species to each toxin.30 Hla is strongly haemolytic to rabbit erythrocytes, Hlb is strongly haemolytic to sheep erythrocytes, and Hlg is haemolytic to rabbit erythrocytes, although it is less efficient than Hla.14,31,32 In order to determine the designated toxin production phenotypes of the isogenic mutant strains (Table I), rabbit and sheep erythrocyte haemolysis assays were conducted (Table II) and there was a significant difference between the supernatants for both the rabbit (p < 0.001) and sheep (p < 0.001) assays. The 8325-4 (Hla+Hlb+Hlg+) and Hla+Hlb-Hlg- supernatants induced significant rabbit erythrocyte haemolysis (p < 0.001 vs HBSS control) (Table II). The Hla-Hlb+Hlg+ and Hla-Hlb-Hlg+ supernatants also produced significant (p < 0.001) haemolysis but to a lesser extent. There was no evidence of rabbit erythrocyte haemolysis with the Hla-Hlb-Hlg- supernatant. The 8325-4 (Hla+Hlb+Hlg+) and Hla-Hlb+Hlg+ supernatants induced significant sheep erythrocyte haemolysis (p < 0.001). In contrast, no significant haemolysis was detected in the presence of the Hla+Hlb-Hlg-, Hla-Hlb-Hlg+, and Hla-Hlb-Hlg- culture supernatants. All isogenic mutant strains therefore produced the appropriate haemolysin phenotypes. Furthermore, the results indicated that the mutation inactivating the production of a particular toxin had no influence on the synthesis of other toxins. Thus, the production of Hla was not affected by the defective genes for Hlb and Hlb, and vice versa.
Table II.
The haemolytic activity of Staphylococcus aureus 8325-4 and isogenic mutant strain culture supernatants on rabbit and sheep erythrocytes; p-values represent supernatant versus Hank’s balanced salt solution (HBSS) control by post hoc Dunnett’s test (n = 3)
| Staphylococcus aureus strain | Phenotype | Mean rabbit erythrocyte haemolysis, % (sd) | Mean sheep erythrocyte haemolysis, % (sd) |
|---|---|---|---|
| 8325-4 | Hla+ Hlb+ Hlg+ | 49.9 (0.9), p < 0.001 | 53.2 (1.0), p < 0.001 |
| DU5946 | Hla+ Hlb- Hlg- | 53.8 (1.7), p < 0.001 | 4.1 (0.7), p = 0.35 |
| DU1090 | Hla- Hlb+ Hlg+ | 28 (0.9), p < 0.001 | 51.1 (1.4), p < 0.001 |
| DU5720 | Hla- Hlb- Hlg+ | 26.5 (1.4), p < 0.001 | 2.3 (0.9), p = 0.32 |
| DU5938 | Hla- Hlb- Hlg- | 5 (0.6), p = 0.06 | 1.8 (0.2), p = 0.08 |
| HBSS control | - | 3.9 (0.3) | 3.2 (1.1) |
Hla, alpha-haemolysin; Hlb, beta-haemolysin; Hlg, gamma-haemolysin
Toxins from wild-type Staphylococcus aureus 8325-4 caused rapid chondrocyte death
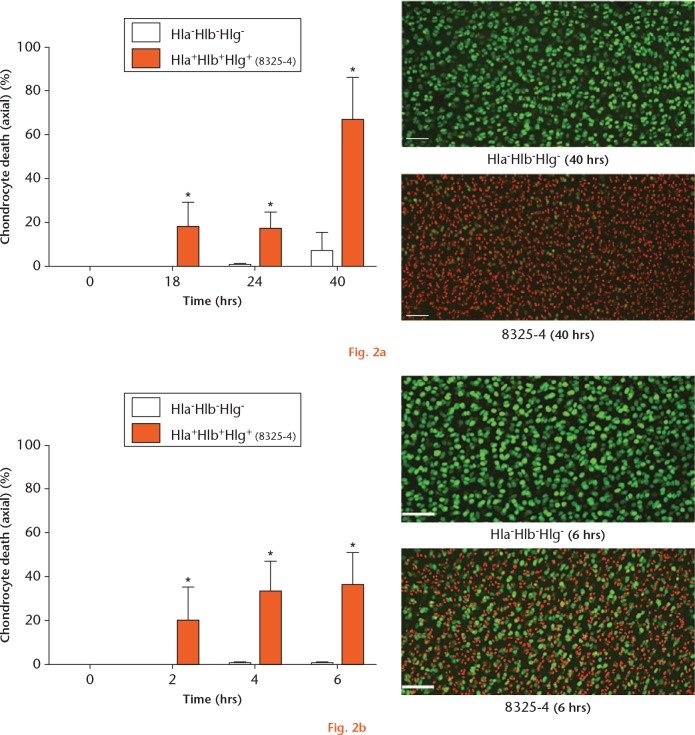
As a first step, in order to assess whether all three haemolysins may be involved in causing chondrocyte death, osteochondral explants were cultured in the presence of either the 8325-4 (Hla+Hlb+Hlg+) or Hla-Hlb-Hlg- strains. Chondrocyte death over 40 hours was then quantified in axial CLSM images. In explants cultured with S. aureus 8325-4 (Hla+Hlb+Hlg+), there was significant chondrocyte death at each timepoint (Fig. 2a). Between 24 and 40 hours, there was a substantial reduction in chondrocyte viability with 66.2% (sd 20.1%) (p = 0.029) cell death at 40 hours. In comparison, there was no significant chondrocyte death at 40 hours in those explants exposed to the Hla-Hlb-Hlg- strain (6.7% (sd 8.9%); p = 0.11; zero hours vs 40 hours). Thus, one, all, or a combination of these haemolysins were responsible for chondrocyte death.
Graphs showing the induction of chondrocyte death by Staphylococcus aureus haemolysins. a) Osteochondral explants cultured in the presence of the 8325-4 strain demonstrated significantly greater chondrocyte death at all timepoints. In comparison, chondrocyte death in the presence of the Hla-Hlb-Hlg- strain was minimal (N = 4 (n = 8); *p < 0.05, 8325-4 vs Hla-Hlb-Hlg- strain by Mann–Whitney U test). Confocal laser-scanning microscopy (CLSM) images displayed the marked difference between the two strains at 40 hours. b) Osteochondral explants exposed to the 8325-4 culture supernatant demonstrated rapid and significant chondrocyte death over six hours compared with explants exposed to the Hla-Hlb-Hlg- supernatant (N = 4 (n = 8); *p < 0.05, 8325-4 vs Hla-Hlb-Hlg- supernatant by Mann–Whitney U test). The CLSM images demonstrate the marked difference between the two supernatants at six hours (in this and subsequent figures, the scale bar = 100 µm).
In order to assess the chondrocyte death-inducing potential of the 40 hours 8325-4 (Hla+Hlb+Hlg+) culture toxins independent of bacteria, a toxin-rich supernatant was prepared. Fresh osteochondral explants were exposed to either 8325-4 (Hla+Hlb+Hlg+) or Hla-Hlb-Hlg- culture supernatants. In comparison with explants exposed to the Hla-Hlb-Hlg- supernatant, those exposed to the 8325-4 (Hla+Hlb+Hlg+) supernatant demonstrated significant chondrocyte death at each timepoint (Fig. 2b). There was no measurable chondrocyte death at any timepoint in explants exposed to the Hla-Hlb-Hlg- supernatant.
Hlb and Hlg induced minimal in situ chondrocyte death
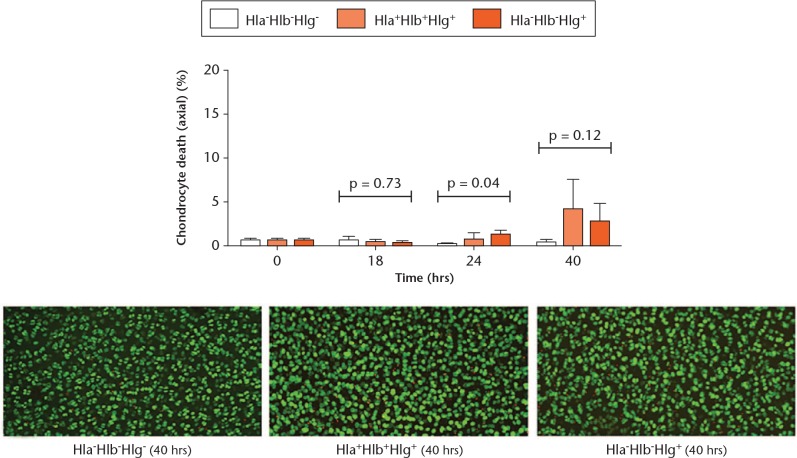
In order to evaluate the contributions of Hlb and Hlg, osteochondral explants were cultured with the Hla-Hlb+Hlg+ and Hla-Hlb-Hlg+ strains. The Hla-Hlb-Hlg- strain was used as a control as it does not cause significant chondrocyte death over 40 hours (Fig. 2a). Although slightly more chondrocyte death occurred in explants exposed to the Hla-Hlb-Hlg+ strain at 24 hours (p = 0.027), the level of chondrocyte death was very low (1.3% (sd 0.5%)) and at the detection limit (Fig. 3). By 40 hours, there was no difference between the strains (p = 0.12). Thus, Hlb and Hlg had no significant role in the induction of in situ chondrocyte death.
Fig. 3.
Graph showing that beta-haemolysin (Hlb) and gamma-haemolysin (Hlg) had a negligible effect on in situ chondrocyte viability. Osteochondral explants cultured in the presence of the Hla-Hlb-Hlg-, Hla-Hlb+Hlg+, and Hla-Hlb-Hlg+ strains exhibited minimal chondrocyte death at each timepoint (N = 4 (n = 8); p-values are shown from one-way analysis of variance; *p < 0.05 vs Hla-Hlb-Hlg- strain by post hoc Dunnett’s test). The confocal laser-scanning microscopy images show examples of the chondrocyte death induced by each strain at 40 hours.
Hla alone induced significant and rapid in situ chondrocyte death
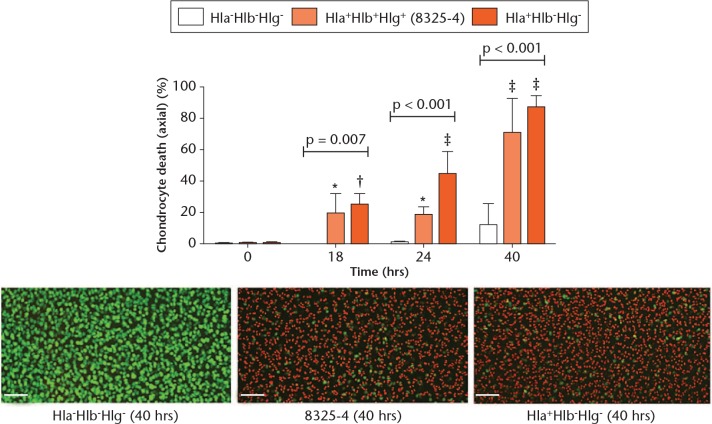
In order to test whether Hla alone was a key damaging toxin to in situ chondrocyte viability, explants were cultured in the presence of 8325-4 (Hla+Hlb+Hlg+), Hla+Hlb-Hlg-, or Hla-Hlb-Hlg- (control). Compared with the control, explants exposed to 8325-4 (Hla+Hlb+Hlg+) and Hla+Hlb-Hlg- exhibited significant chondrocyte death at each timepoint (Fig. 4). Although there was a significant difference between the 8325-4 (Hla+Hlb+Hlg+) and Hla+Hlb-Hlg- strains at 24 hours (p = 0.01), there was no difference at 40 hours (p = 0.2, unpaired Student’s two-tailed t-test).
Fig. 4.
Graph showing that the 8325-4 and Hla+Hlb-Hlg- strains had comparable potencies. In comparison with the Hla-Hlb-Hlg-strain control group, the 8325-4 and Hla+Hlb-Hlg- strains induced similar chondrocyte death over the experimental period (N = 4 (n = 8); p-values are shown from one-way analysis of variance; *p < 0.05; †p < 0.01; ‡p < 0.001 vs Hla-Hlb-Hlg- strain by post hoc Dunnett’s test). Confocal laser-scanning microscopy images represent the chondrocyte death induced by each strain at 40 hours.
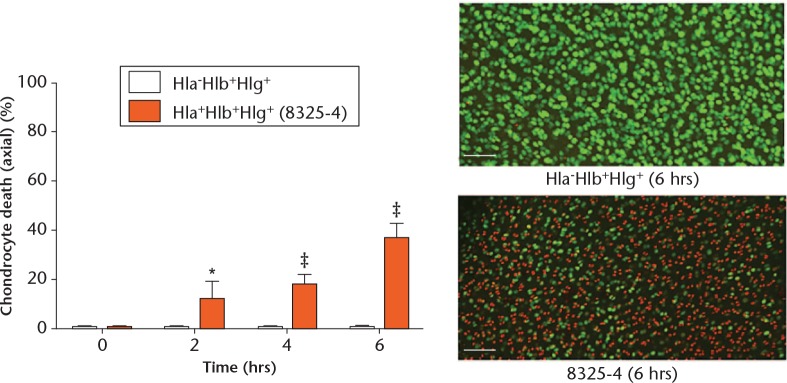
In order to test further whether Hla was the sole cause of chondrocyte death, explants were exposed to the 40 hours culture supernatants of the 8325-4 (Hla+Hlb+Hlg+) and Hla-Hlb+Hlg+ strains, where the only difference between the supernatants was the presence or absence of Hla, respectively. There was negligible chondrocyte death in the explants exposed to the Hla-Hlb+Hlg+ supernatant, while there was significant chondrocyte death at each timepoint in those exposed to the 8325-4 (Hla+Hlb+Hlg+) supernatant (Fig. 5). These results therefore confirmed that S. aureus Hla was the only toxin capable of inducing substantial and significant in situ chondrocyte death. Other bacterial products/toxins had a negligible effect on chondrocyte viability, as shown by the lack of effect when the explants were cultured in Hla-Hlb+Hlg+ or Hla-Hlb-Hlg- supernatants (Figs 2b and 5).
Fig. 5.
Graph showing that alpha-haemolysin (Hla) alone affected in situ chondrocyte viability. Osteochondral explants cultured with Hla-Hlb+Hlg+ supernatant were compared with those cultured with 8325-4 supernatant. The only difference between the two culture supernatants was the presence or absence of Hla. There was significant chondrocyte death at each timepoint in explants exposed to the 8325-4 supernatant in comparison with those exposed to the Hla-Hlb+Hlg+ supernatant (N = 4 (n = 8); *p < 0.05; ‡p < 0.001 by unpaired Student’s two-tailed t-test). The confocal laser-scanning microscopy images display the influence of Hla on in situ chondrocyte viability.
Hla-induced chondrocyte death commenced within the superficial zone of cartilage
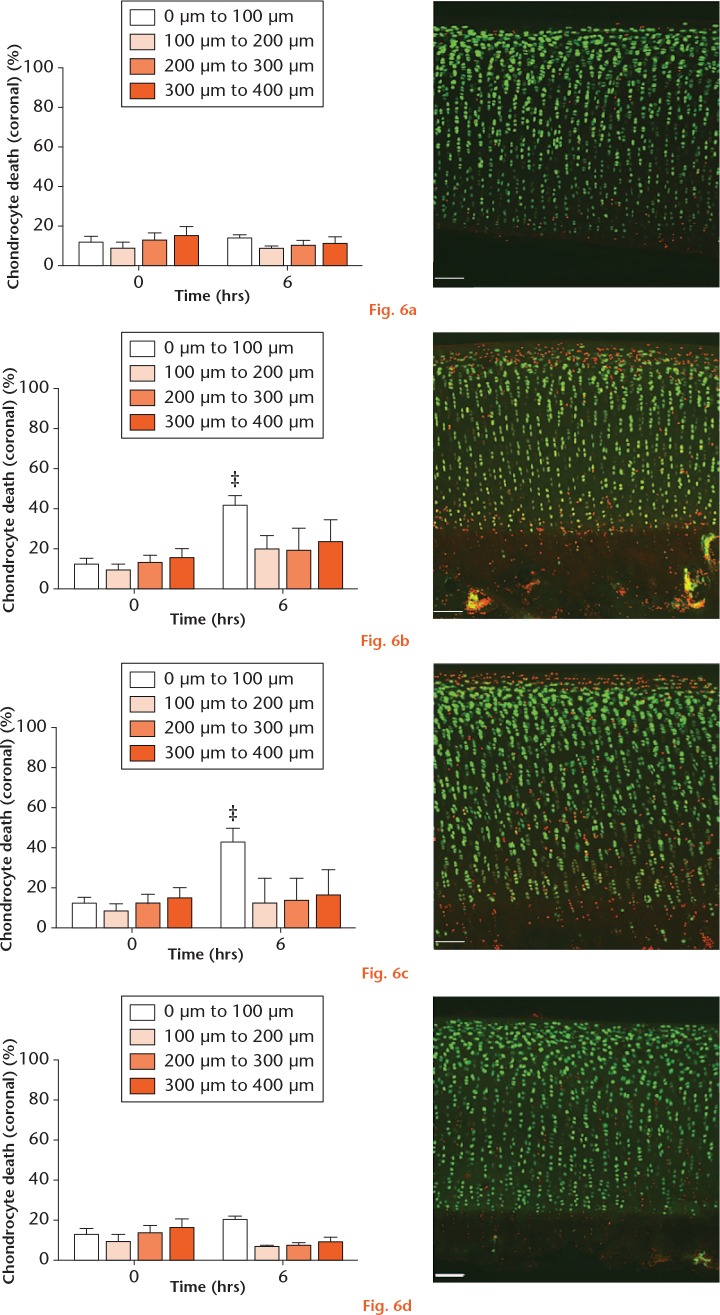
The axial studies described above involved imaging osteochondral explants to a depth of approximately 100 µm from the articular surface, thereby permitting quantification of chondrocyte death within the entirety of the superficial zone (SZ) and a portion of the middle zone (MZ). In order to establish whether Hla-induced chondrocyte death started in the SZ and to assess whether Hlb and Hlg did not selectively induce chondrocyte death within deeper layers of cartilage (i.e. cell death > 100 µm that would not be detected on axial imaging due to depth limitations), a coronal study was performed. Osteochondral explants were exposed to 8325-4 (Hla+Hlb+Hlg+), Hla+Hlb-Hlg-, Hla-Hlb+Hlg+, or Hla-Hlb-Hlg- strain culture supernatants and imaged coronally. As reported previously,29 a dead cell artefact was induced by the scalpel cut at zero hours (Fig. 6).
Graphs showing the pattern of alpha-haemolysin-induced chondrocyte death. Over 40 hours, compared with the a) Hla-Hlb-Hlg- control group, there was significantly more chondrocyte death within the first 100 µm of explants exposed to the b) Hla-containing 8325-4 and c) Hla+Hlb-Hlg- supernatants. d) There was no change in chondrocyte viability in explants exposed to the Hla-Hlb+Hlg+ strain supernatant (N = 4 (n = 8); ‡p < 0.001 vs Hla-Hlb-Hlg- strain supernatant by post hoc Dunnett’s test). a) to d) The confocal laser-scanning microscopy inserts show examples of the zonal pattern of chondrocyte death following explant exposure to the 40-hour culture supernatant of the strain indicated.
Explants exposed to the Hla-Hlb-Hlg- supernatant (control) exhibited no significant chondrocyte death above zero-hour values for each depth interval at six hours (p = 0.07) (Fig. 6a). Compared with the control group, explants exposed to the 8325-4 (Hla+Hlb+Hlg+) and Hla+Hlb-Hlg- supernatants exhibited significant chondrocyte death (p < 0.001) within the first depth interval (0 µm to 100 µm) (Figs 6a to 6c). In those explants exposed to the Hla-Hlb+Hlg+ supernatant, there was no change in chondrocyte viability within the first depth interval (p = 0.28) or throughout the analyzed depth of cartilage (Fig. 6d). This further confirmed that Hlb and Hlg had no significant role in the induction of chondrocyte death within the SZ, but also that Hlb and Hlg did not specifically induce chondrocyte death within deeper layers.
Discussion
Our main finding was that S. aureus Hla alone could induce rapid chondrocyte death, starting in the SZ of cartilage, whereas, in comparison, Hlb and Hlg induced negligible chondrocyte death. The lethal influence of Hla on chondrocyte viability is of potential importance in understanding the mechanism of cartilage destruction both during and after an episode of S. aureus-induced septic arthritis.
As chondrocytes in healthy cartilage do not normally divide once skeletal maturity is achieved,33 chondrocyte loss may ultimately have catastrophic consequences. Chondrocytes govern the turnover of matrix and thus cartilage devoid of chondrocytes will ultimately degrade, as maintenance of the matrix will be lost. This has been demonstrated in an in vivo study on rabbits by Simon et al,34 in which the long-term effect of localized cryotherapy-induced chondrocyte death was investigated. Six months after the cryotherapy, the cartilage was structurally intact despite the absence of viable chondrocytes. However, by 12 months there was extensive cartilage fibrillation and softening, changes considered to be amongst the first macroscopic changes associated with degenerative joint disease.35
Due to a lack of long-term prospective radiographic surveillance studies, how long it takes for cartilage destruction to become apparent following a supposedly treated episode of septic arthritis is currently not known. However, the potential rapid consequences of inadequately treated septic arthritis caused by S. aureus may be more evident. In a case report by Kim et al,36 the details of a case of ‘missed’ septic arthritis of the hip in a 61-year-old woman were presented. At the time of the onset of symptoms, plain radiographs were reported as normal and the diagnosis of ‘hip sprain’ was proposed. Two months later, with progressively worsening symptoms, aspiration of the hip confirmed S. aureus-induced sepsis. By this stage, however, repeat radiographs demonstrated complete loss of joint space and bone destruction. Whilst such an untreated case is rare, it highlights the cartilage-damaging potential of S. aureus-induced septic arthritis and the importance of early diagnosis and treatment. Given the lethal role of Hla observed in the current study, it is recommended that infected synovial fluid be removed promptly from the affected joint, with surgical washout probably offering the most comprehensive form of lavage.
The in vitro findings presented here differed from previous in vivo studies and it is important to consider possible explanations. Using murine models of septic arthritis caused by S. aureus, Gemmell et al15 and Nilsson et al14 concluded that Hla in combination with protein A and Hlg, respectively, were crucial for the development and progression of S. aureus-induced septic arthritis. Protein A is a cell wall surface protein that aids S. aureus survival in the presence of a host immune response by binding the Fc portion of IgG and the Fc receptors of polymorphonuclear leukocytes, thereby preventing bacterial opsonization and phagocytosis.9,37 However, the experimental model in the current study included no immune response. Furthermore, purified Hla has been shown to lyse eukaryotic cells, including erythrocytes38 and monocytic cells,39 thereby demonstrating lytic activity in the absence of protein A and other bacterial products. Thus, it seems probable that the Hla-induced chondrocyte death observed in the present study was independent of protein A. Whilst Hlg induced negligible chondrocyte death (Fig. 3), it is possible that it may play a destructive role in vivo as it may stimulate the release of inflammatory mediators.14,40,41
The establishment of Hla as a chondrocyte death-inducing toxin and the recent identification of A-disintegrin and metalloprotease 10 (ADAM10) as its receptor on eukaryotic cells,17,42-44 offers potential future therapeutic targets. By attempting to block the activity of Hla, for example by Hla-neutralizing antibodies or selective blockade of ADAM10, it is possible that chondrocyte death may be reduced during septic arthritis caused by S. aureus, thereby minimizing the extent of subsequent cartilage loss. Support for this comes from a murine study of pneumonia caused by S. aureus,45 which demonstrated protection in those animals immunized with a mutant form of Hla (HlaH35L) that was unable to form transmembrane pores and thus unable to lyse cells. The administration of Hla-specific antibodies to non-vaccinated animals also conferred protection. This is corroborated further in a recent study by Corrado et al46 in which the administration of a four-component vaccine, comprising five S. aureus antigens including a genetically detoxified derivative of Hla,10 to a murine model of S. aureus septic arthritis conferred protection to bacterial-mediated cellular toxicity.
A further potential therapeutic target is the Hla-induced cell death pathway following formation of the Hla pore through the plasma membrane. It is currently believed that the formation of pores triggers a cascade of events that ultimately culminates in cell death.18,47-49 It has been suggested that alterations in intracellular concentrations of Ca2+, Na+, and K+ may be key events in the Hla-induced cell death pathway after the formation of pores.50 However, there remains some uncertainty as to the exact roles of these ions. Some studies have suggested that the influx of Ca2+ through the membrane-spanning Hla pore is the main trigger of cellular death mechanisms,48 while others have suggested that Hla pores do not permit the passage of divalent cations and that the important primary trigger is the influx of Na+ coupled with the efflux of K+.49,51 Regardless, the influx of cations is accompanied by water, leading to swelling and lysis of cells. In addition to disturbance of the ionic equilibrium, activation of stress-signalling pathways, and the egress of vital molecules, such as adenosine triphosphate from the cell membrane are also thought to contribute to cell death.18,47-49 However, the mechanism of Hla-induced cell death may be concentration dependent as Jonas et al52 identified that when isolated peripheral blood human T lymphocytes were exposed to a high concentration of Hla, the plasma membrane became permeable to Ca2+. In contrast, when the cells were exposed to low concentrations of Hla, the plasma membrane was found to be impermeable to Ca2+, thereby suggesting that Hla pore size, and indeed the probable mechanism of cell death, may be dependent on the concentration of Hla. Clearly, a further understanding of this pathway is required before this avenue can be explored as a potential therapeutic target.
We used a bovine cartilage explant model of S. aureus-induced septic arthritis and live- and dead-cell CLSM imaging in this study to assess the influence of Hla, Hlb, and Hlg on in situ chondrocyte viability. The rationale behind such a model and its advantages have been discussed previously.21 A potential limitation is the extrapolation of conclusions from this model of septic arthritis to the clinical setting as the sensitivity of human chondrocytes to S. aureus haemolysins may be different. Whilst it is possible that human chondrocytes may be sensitive to Hlb and Hlg, Chubinskaya et al53 have demonstrated greater ADAM10 expression in human chondrocytes compared with bovine chondrocytes and thus the impact of Hla on human chondrocytes may be more important. They have also reported increased ADAM10 expression by SZ chondrocytes compared with chondrocytes from deeper layers in healthy human cartilage.53 This may offer an additional explanation, beyond that of the close proximity of SZ chondrocytes to the culture medium harbouring Hla, for the observed zonal pattern of chondrocyte death in both the current (Fig. 6) and previous studies.21 In any event, future work should be directed at assessing the sensitivity of in situ human chondrocytes to Hla, Hlb, and Hlg toxins.
Using an established in vitro model of S. aureus-induced septic arthritis, we have demonstrated that Hla alone has a rapid and lethal influence on in situ chondrocytes, with Hlb and Hlg having no significant effect on their viability. The identification of Hla as the key destructive toxin to chondrocytes may provide insight to support the development of therapeutic strategies aimed at reducing the extent of cartilage damage during and after an episode of septic arthritis caused by S. aureus.
Footnotes
Author Contributions: I. D. M. Smith: Study conception and design, Collecting and assembling the data, Analyzing and interpreting the data, Drafting, critically revising, and approving the manuscript.
K. M. Milto: Collecting and assembling the data, Analyzing and interpreting the data, Critically revising and approving the manuscript.
C. J. Doherty: Collecting and assembling the data, Approving the manuscript.
S. G. B. Amyes: Study conception and design, Critically revising and approving the manuscript.
A. H. R. W. Simpson: Study conception and design, Critically revising and approving the manuscript.
A. C. Hall: Study conception and design, Analyzing and interpreting the data, Drafting, critically revising, and approving the manuscript.
Conflict of Interest Statement: None declared
Follow us @BoneJointRes
Funding Statement
The support of Orthopaedic Research UK (Clinical Research Fellowship awarded to Innes D.M. Smith, ref no. 477) and The Royal College of Surgeons of Edinburgh (Small Project Grant awarded to Innes D.M. Smith, ref no. SRG/11/043) is gratefully acknowledged. The authors thank Professor Timothy J. Foster, Trinity College, Dublin, for supplying S. aureus strain 8325-4 and its associated mutants, Dr Trudi Gillespie, IMPACT facility, The University of Edinburgh, for CLSM guidance, and Scotbeef Ltd., for providing bovine feet.
References
- 1. Mathews CJ, Kingsley G, Field M, et al. Management of septic arthritis: a systematic review. Postgrad Med J 2008;84:265-270. [DOI] [PubMed] [Google Scholar]
- 2. Shirtliff ME, Mader JT. Acute septic arthritis. Clin Microbiol Rev 2002;15:527-544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Mathews CJ, Weston VC, Jones A, Field M, Coakley G. Bacterial septic arthritis in adults. Lancet 2010;375:846-855. [DOI] [PubMed] [Google Scholar]
- 4. Goldenberg DL. Septic arthritis. Lancet 1998;351:197-202. [DOI] [PubMed] [Google Scholar]
- 5. Kaandorp CJ, Krijnen P, Moens HJ, Habbema JD, van Schaardenburg D. The outcome of bacterial arthritis: a prospective community-based study. Arthritis Rheum 1997;40:884-892. [DOI] [PubMed] [Google Scholar]
- 6. Krieg AM. A possible cause of joint destruction in septic arthritis. Arthritis Res 1999;1:3-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Gupta MN, Sturrock RD, Field M. A prospective 2-year study of 75 patients with adult-onset septic arthritis. Rheumatology (Oxford) 2001;40:24-30. [DOI] [PubMed] [Google Scholar]
- 8. Kaandorp CJ, Dinant HJ, van de, Laar MA, et al. Incidence and sources of native and prosthetic joint infection: a community based prospective survey. Ann Rheum Dis 1997;56:470-475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Cunningham R, Cockayne A, Humphreys H. Clinical and molecular aspects of the pathogenesis of Staphylococcus aureus bone and joint infections. J Med Microbiol 1996;44:157-164. [DOI] [PubMed] [Google Scholar]
- 10. Torre A, Bacconi M, Sammicheli C, et al. Four-component Staphylococcus aureus vaccine 4C-staph enhances Fcγ receptor expression in neutrophils and monocytes and mitigates S. aureus infection in neutropenic mice. Infect Immun 2015;83:3157-3163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Dajcs JJ, Thibodeaux BA, Girgis DO, O’Callaghan RJ. Corneal virulence of Staphylococcus aureus in an experimental model of keratitis. DNA Cell Biol 2002;21:375-382. [DOI] [PubMed] [Google Scholar]
- 12. Hayashida A, Bartlett AH, Foster TJ, Park PW. Staphylococcus aureus beta-toxin induces lung injury through syndecan-1. Am J Pathol 2009;174:509-518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. O’Callaghan RJ, Callegan MC, Moreau JM, et al. Specific roles of alpha-toxin and beta-toxin during Staphylococcus aureus corneal infection. Infect Immun 1997;65:1571-1578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Nilsson I-M, Hartford O, Foster T, Tarkowski A. Alpha-toxin and gamma-toxin jointly promote Staphylococcus aureus virulence in murine septic arthritis. Infect Immun 1999;67:1045-1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Gemmell CG, Goutcher SC, Reid R, Sturrock RD. Role of certain virulence factors in a murine model of Staphylococcus aureus arthritis. J Med Microbiol 1997;46:208-213. [DOI] [PubMed] [Google Scholar]
- 16. Gouaux E. alpha-Hemolysin from Staphylococcus aureus: an archetype of beta-barrel, channel-forming toxins. J Struct Biol 1998;121:110-122. [DOI] [PubMed] [Google Scholar]
- 17. Ezekwe EAD, Jr, Weng C, Duncan JA. ADAM10 Cell surface expression but not activity is critical for Staphylococcus aureus α-hemolysin-mediated activation of the NLRP3 inflammasome in human monocytes. Toxins (Basel) 2016;8:95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Dinges MM, Orwin PM, Schlievert PM. Exotoxins of Staphylococcus aureus. Clin Microbiol Rev 2000;13:16-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Prévost G, Cribier B, Couppié P, et al. Panton-Valentine leucocidin and gamma-hemolysin from Staphylococcus aureus ATCC 49775 are encoded by distinct genetic loci and have different biological activities. Infect Immun 1995;63:4121-4129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Coleman DC, Arbuthnott JP, Pomeroy HM, Birkbeck TH. Cloning and expression in Escherichia coli and Staphylococcus aureus of the beta-lysin determinant from Staphylococcus aureus: evidence that bacteriophage conversion of beta-lysin activity is caused by insertional inactivation of the beta-lysin determinant. Microb Pathog 1986;1:549-564. [DOI] [PubMed] [Google Scholar]
- 21. Smith IDM, Winstanley JP, Milto KM, et al. Rapid in situ chondrocyte death induced by Staphylococcus aureus toxins in a bovine cartilage explant model of septic arthritis. Osteoarthritis Cartilage 2013;21:1755-1765. [DOI] [PubMed] [Google Scholar]
- 22. Novick R. Properties of a cryptic high-frequency transducing phage in Staphylococcus aureus. Virology 1967;33:155-166. [DOI] [PubMed] [Google Scholar]
- 23. O’Reilly M, de Azavedo JC, Kennedy S, Foster TJ. Inactivation of the alpha-haemolysin gene of Staphylococcus aureus 8325-4 by site-directed mutagenesis and studies on the expression of its haemolysins. Microb Pathog 1986;1:125-138. [DOI] [PubMed] [Google Scholar]
- 24. Patel AH, Nowlan P, Weavers ED, Foster T. Virulence of protein A-deficient and alpha-toxin-deficient mutants of Staphylococcus aureus isolated by allele replacement. Infect Immun 1987;55:3103-3110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Supersac G, Piémont Y, Kubina M, Prévost G, Foster TJ. Assessment of the role of gamma-toxin in experimental endophthalmitis using a hlg-deficient mutant of Staphylococcus aureus. Microb Pathog 1998;24:241-251. [DOI] [PubMed] [Google Scholar]
- 26. Castano Oreja MT, Quintáns Rodriguez M, Crespo Abelleira A, et al. Variation in articular cartilage in rabbits between weeks six and eight. Anat Rec 1995;241:34-38. [DOI] [PubMed] [Google Scholar]
- 27. Roberts S, Weightman B, Urban JPG, Chappell D. Mechanical and biochemical properties of human articular cartilage from the femoral head after subcapital fracture. J Bone Joint Surg [Br] 1986;68-B:418-422. [DOI] [PubMed] [Google Scholar]
- 28. Huntley JS, Simpson AH, Hall AC. Use of non-degenerate human osteochondral tissue and confocal laser scanning microscopy for the study of chondrocyte death at cartilage surgery. Eur Cell Mater 2005;9:13-22. [DOI] [PubMed] [Google Scholar]
- 29. Amin AK, Huntley JS, Bush PG, Simpson AH, Hall AC. Osmolarity influences chondrocyte death in wounded articular cartilage. J Bone Joint Surg [Am] 2008;90-A:1531-1542. [DOI] [PubMed] [Google Scholar]
- 30. Thelestam M, Möllby R, Wadström T. Effects of staphylococcal alpha-, beta-, delta-, and gamma-hemolysins on human diploid fibroblasts and HeLa cells: evaluation of a new quantitative assay for measuring cell damage. Infect Immun 1973;8:938-946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Traber KE, Lee E, Benson S, et al. agr function in clinical Staphylococcus aureus isolates. Microbiology 2008;154:2265-2274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Burnside K, Lembo A, de Los Reyes M, et al. Regulation of hemolysin expression and virulence of Staphylococcus aureus by a serine/threonine kinase and phosphatase. PLoS One 2010;5:e11071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Archer CW. Skeletal development and osteoarthritis. Ann Rheum Dis 1994;53:624-630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Simon WH, Richardson S, Herman W, Parsons JR, Lane J. Long-term effects of chondrocyte death on rabbit articular cartilage in vivo. J Bone Joint Surg [Am] 1976;58-A:517-526. [PubMed] [Google Scholar]
- 35. Pearle AD, Warren RF, Rodeo SA. Basic science of articular cartilage and osteoarthritis. Clin Sports Med 2005;24:1-12. [DOI] [PubMed] [Google Scholar]
- 36. Kim P, Mierau D, Loback D. Septic arthritis of the hip: a case report. J Can Chiropr Assoc 1993;37:151-158. [Google Scholar]
- 37. Widaa A, Claro T, Foster TJ, O’Brien FJ, Kerrigan SW. Staphylococcus aureus protein A plays a critical role in mediating bone destruction and bone loss in osteomyelitis. PLoS One 2012;7:e40586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cooper LZ, Madoff MA, Weinstein L. Hemolysis of rabbit erythrocytes by purified staphylococcal alpha-toxin. I. Kinetics of the lytic reaction. J Bacteriol 1964;87:127-135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Craven RR, Gao X, Allen IC, et al. Staphylococcus aureus alpha-hemolysin activates the NLRP3-inflammasome in human and mouse monocytic cells. PLoS One 2009;4:e7446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Suttorp N, Fuhrmann M, Tannert-Otto S, Grimminger F, Bhadki S. Pore-forming bacterial toxins potently induce release of nitric oxide in porcine endothelial cells. J Exp Med 1993;178:337-341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Siqueira JA, Speeg-Schatz C, Freitas FI, et al. Channel-forming leucotoxins from Staphylococcus aureus cause severe inflammatory reactions in a rabbit eye model. J Med Microbiol 1997;46:486-494. [DOI] [PubMed] [Google Scholar]
- 42. Wilke GA, Bubeck Wardenburg J. Role of a disintegrin and metalloprotease 10 in Staphylococcus aureus alpha-hemolysin-mediated cellular injury. Proc Natl Acad Sci USA 2010;107:13473-13478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Powers ME, Kim HK, Wang Y, Bubeck Wardenburg J. ADAM10 mediates vascular injury induced by Staphylococcus aureus α-hemolysin. J Infect Dis 2012;206:352-356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Inoshima I, Inoshima N, Wilke GA, et al. A Staphylococcus aureus pore-forming toxin subverts the activity of ADAM10 to cause lethal infection in mice. Nat Med 2011;17:1310-1314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Bubeck Wardenburg J, Schneewind O. Vaccine protection against Staphylococcus aureus pneumonia. J Exp Med 2008;205:287-294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Corrado A, Donato P, Maccari S, et al. Staphylococcus aureus-dependent septic arthritis in murine knee joints: local immune response and beneficial effects of vaccination. Sci Rep 2016;6:38043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Bhakdi S, Tranum-Jensen J. Alpha-toxin of Staphylococcus aureus. Microbiol Rev 1991;55:733-751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Suttorp N, Seeger W, Dewein E, Bhakdi S, Roka L. Staphylococcal alpha-toxin-induced PGI2 production in endothelial cells: role of calcium. Am J Physiol 1985;248:C127-C134. [DOI] [PubMed] [Google Scholar]
- 49. Walev I, Martin E, Jonas D, et al. Staphylococcal alpha-toxin kills human keratinocytes by permeabilizing the plasma membrane for monovalent ions. Infect Immun 1993;61:4972-4979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Berube BJ, Bubeck Wardenburg J. Staphylococcus aureus α-toxin: nearly a century of intrigue. Toxins (Basel) 2013;5:1140-1166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Valeva A, Walev I, Gerber A, et al. Staphylococcal alpha-toxin: repair of a calcium-impermeable pore in the target cell membrane. Mol Microbiol 2000;36:467-476. [DOI] [PubMed] [Google Scholar]
- 52. Jonas D, Walev I, Berger T, et al. Novel path to apoptosis: small transmembrane pores created by staphylococcal alpha-toxin in T lymphocytes evoke internucleosomal DNA degradation. Infect Immun 1994;62:1304-1312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Chubinskaya S, Mikhail R, Deutsch A, Tindal MH. ADAM-10 protein is present in human articular cartilage primarily in the membrane-bound form and is upregulated in osteoarthritis and in response to IL-1alpha in bovine nasal cartilage. J Histochem Cytochem 2001;49:1165-1176. [DOI] [PubMed] [Google Scholar]