Abstract
Reconstruction of large skeletal defects is challenging due to the requirement for large volumes of donor tissue and the often complex surgical procedures. Tissue engineering has the potential to serve as a new source of tissue for bone reconstruction, but current techniques are often limited in regards to the size and complexity of tissue that can be formed. Building tissue using an in vivo bioreactor approach may enable the production of appropriate amounts of specialized tissue, while reducing issues of donor site morbidity and infection. Large animals are required to screen and optimize new strategies for growing clinically appropriate volumes of tissues in vivo. In this article, we review both ovine and porcine models that serve as models of the technique proposed for clinical engineering of bone tissue in vivo. Recent findings are discussed with these systems, as well as description of next steps required for using these models, to develop clinically applicable tissue engineering applications.
Keywords: : bone regeneration, large animal models, periosteum, ovine model, porcine model
Introduction
There is a critical need for improved methods for reconstruction of complex skeletal defects that involve missing bone tissue. These defects can occur because of congenital absence of normal bone or acquired bone loss due to cancer treatment or trauma. They remain one of the most significant challenges for surgeons. Currently, the best replacement for lost tissue is uninjured autologous tissue-healthy bone obtained from an uninjured location on the patient, which is harvested and transferred into the defect, and current clinical methods are effective for transferring this type of tissue.1 However, these methods are limited by amounts of suitable bone tissue and resultant donor site morbidity. Tissue engineering has shown promise as a new source of tissues for reconstruction of these defects.2 Engineering living bone replacements in shapes and volumes relevant to clinical application could transform surgical practice.
The successful translation of tissue engineering therapies into clinical application requires overcoming a number of technical, biological, and surgical challenges. Many bioreactor systems have been developed for building new tissues in vitro. A recent study showed the potential of using bioreactors to generate a large volume of engineered bone.3 However, the overall complexity of tissue formed is limited due to the inability to sufficiently recreate the process of vascularized bone formation. Other techniques attempt to regenerate bone within the defect site.4,5 For many clinical circumstances this strategy is unsuitable because the local tissues are compromised as part of the underlying disease process and, therefore, have limited healing and regenerative capacity. This necessitates generating the tissue at a healthy site distant from the defect and later transferring the tissue. Utilizing the body's own regenerative capacity to serve as an in vivo bioreactor to grow complex tissues overcomes many of the challenges of in vitro tissue engineering.
Clinical Strategy
One strategy for bone tissue engineering is to exploit the natural osteogenic and angiogenic properties of the periosteum to serve as an in vivo bioreactor to enhance vascularized bone formation within implanted chambers of predetermined 3D shape.6,7 A presentation of the overall approach is provided in Figure 1.
FIG. 1.
A schematic summary of clinical strategy. The complex bone defect is first imaged and then the optimal structure determined to acquire custom design scaffold and tissue chamber shape. A custom-made tissue chamber is filled with biodegradable osteoconductive scaffolding, bioactive growth factors, and cells. The entire mold is then implanted into the patient adjacent to the periosteum of the inner table of the pelvis. After a period of time for tissue generation, the device is harvested to yield a custom fabricated bone flap, which is then surgically transferred to the defect location.
An image provides patient-specific information on the 3D structure of the defect to be reconstructed.
The optimal structure needed for reconstruction is determined using computational models.
Chambers are designed that specifically conform to the optimal structures determined.
The chambers are filled with a tissue engineering strategy (e.g., scaffolds, cells, and growth factors) and implanted against the periosteum in a secure and stable location away from the defect site.
After some optimal implantation time, the bone can be harvested and transferred along with the periosteum to the recipient location as a vascularized bone flap.
The success of this approach requires determination of the tissue engineering strategies that can control the shape and volume of bone formed in the implanted chambers. Translatable large animal models of the periosteum-based in vivo bioreactor allow screening and optimization of these strategies.
Clinical Cases of the In Vivo Bioreactor
The application of an in vivo bioreactor to generate vascularized bone has been explored clinically.7–11 The majority of these cases involved implantation of a preformed chamber into muscle or fascia.8,10,11 These studies have combined a variety of different scaffolds (e.g., autograft and hydroxyapatite) and, in some cases, osteogenic growth factors (e.g., BMP-7).9,10 While these studies provided support for the potential of in vivo bioreactors, it has been shown in large animal models that the periosteum generates greater bone volume than muscle.12 The periosteum-based bioreactor (Fig. 1) has been applied clinically for the prefabrication of a vascularized bone flap for transfer to a mandible defect.7 After excision of a buccal squamous cell carcinoma, reconstruction was first performed with a pectoralis major myocutaneous flap and a reconstruction plate. This failed and was followed by incomplete reconstruction with a combined free anterolateral thigh cutaneous flap, a radial forearm flap, and a free fibula flap. The limited remaining options for the patient resulted in the application of the in vivo bioreactor approach with morcellized bone graft (MBG) used as the biomaterial. Poly(methyl methacrylate) (PMMA) chambers were filled with MBG harvested from the iliac crest and the chamber implanted against the periosteum in the iliac crest. After 8 weeks, the bone was harvested and transferred to the defect location. The patient was followed out to 1 year, when the engineered bone enabled the placement of three dental implants. Histological samples revealed regenerated bone. While this study supports the clinical potential of the in vivo bioreactor, broad application requires the identification of alternative, nonautologous sources for the chamber contents.13–15
Large Animal Models
Small and large animal models have been developed to guide investigation into in vivo bone bioreactors. Small animal models result in relatively small tissue volumes.16,17 A critical limitation to clinical application is the inability to generate bone of sufficient volume to treat the defects encountered. In addition, the transport challenges presented do not sufficiently approximate the clinical situation. Therefore, large animal models are essential to test the feasibility of tissue engineering strategies for growing large complex volumes of bone.
Large animals, including sheep, minipigs, and nonhuman primates, have been investigated in the field for bone reconstruction using an in vivo bioreactor strategy. While nonhuman primates offer a number of advantages,18 the high cost and strict regulations limit broad application. A preclinical model of an in vivo bioreactor should: (1) mimic the clinical surgery techniques; (2) allow evaluation of vascularized bone formation of large volume and complex shape; (3) have an implantation site with high regenerative capacity and low infection risk; (4) be adaptable for different tissue engineering components; (5) allow quantitative evaluation of results; and (6) be available/adaptable in a wide range of clinical research centers. Below, we review two periosteum guided in vivo bioreactor models, ovine and porcine, that meet these requirements.
Surgical approach
Ovine model
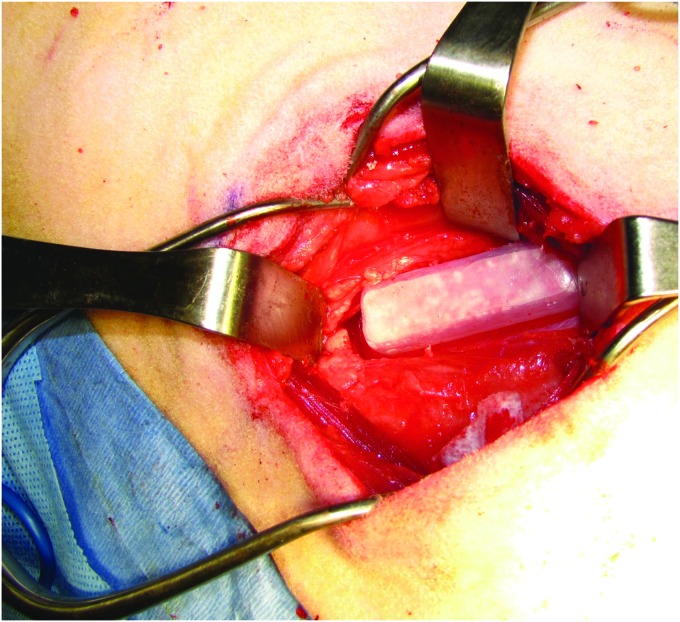
The ovine model is a well-described physiologically-relevant large animal platform in which multiple in vivo bioreactors can be implanted for the generation of mineralized bone tissue.6 Briefly, a section of bone along the ribs is resected to uncover the cambium layer of the periosteum. Bioreactors, generally fabricated from PMMA, are implanted with an open face against the periosteum (Fig. 2).
FIG. 2.
Surgical photograph of a bioreactor (1 × 1 × 4 cm) implanted against rib periosteum in a sheep. Bioreactors are implanted on alternating ribs with up to four implanted per animal.
Unlike bioreactors implanted within muscle, bioreactors implanted against the inner surface of the periosteum can generate viable ossified tissue in the ovine model without the addition of exogenous growth factors.12 Once implanted, tissues generated within bioreactors reach peak mineralization and then are harvested for either analysis or for use in reconstruction of a craniofacial defect.18 Tissues can be harvested with the accompanying pedicle (nerve and intercostal artery and vein) for transfer as a vascularized free bony flap (Fig. 3).
FIG. 3.
A harvested bioreactor (1 × 1 × 4 cm) after removal of generated tissues, a tissue flap with accompanying pedicle generated after 9 weeks of implantation, and a resected mandibular angle (1 × 4 cm) to create a large volume tissue defect for subsequent reconstruction with bioreactor-generated free tissue bone flaps.
In the sheep, these vessels are very small (∼1.5 and 0.5 mm in diameter for the artery and vein, respectively). They may be anastomosed to branches of the external carotid artery (transverse facial artery, internal maxillary artery, superficial temporal artery, or lingual artery) and accompanying vein using side-to-side and/or end-to-side technique with microsurgery (Fig. 4).
FIG. 4.
Completed anastomoses of a free tissue bone flap generated within an in vivo bioreactor used to reconstruct a large ovine mandibular angle defect. Note that the artery anastomosis was completed side to side, and the vein anastomosis was side to end. The lower edge of the tissue flap is located in the upper right corner (partially obscured by the retractor).
Porcine model
Minipigs have been used as a model for reconstruction of a mandibular defect with tissue using the latissimus dorsi as the implantation site for the in vivo bioreactor.19,20 In these studies, bovine hydroxyapatite (Bio-Oss) scaffold was combined with an osteogenic growth factor (BMP-7) in titanium cages to generate bone tissue. This approach has been applied clinically.10,21 However, the regenerative properties of the periosteum can provide significant advantages for bone regeneration compared to muscle fascia.12,22 The cambium of the periosteum includes osteogenic cells and growth factors that are not present in muscle, which enables bone formation even in the absence of exogenous cells or factors. Intramuscular implantation requires growth factors for bone formation.23
Recently, a periosteum guided porcine model analogous to the ovine model was developed. Briefly, the rib cage of skeletally mature Lanyu miniature pigs is exposed through one S-shape incision oriented parallel to the ribs. The periosteum covered on top of the ribs is incised sharply at three rib levels and peeled away from the bone for 8–10 cm using a periosteum elevator. A 4–6 cm bone segment is removed with a sharp bone-cutting instrument (Fig. 5A) allowing cambium layer of bottom part of periosteum exposed. Next, PMMA chambers (10 mm height, 8 mm inner diameter) are implanted (Fig. 5B).
FIG. 5.
Periosteum guided pig animal model. (A) Open periosteum area is between green dashed lines, inset shows removed rib segment. (B) PMMA chambers implanted against the periosteum, inset shows empty chamber before implantation. Bioreactors are implanted on alternating ribs with up to two chambers per rib and four ribs used per animal.
Chamber design
The structure of the chamber is critical for successful implementation in the animal model. The shape of the chamber must fit the prefabrication site and within the dimensions of the open periosteal surface. The chamber shape previously used in the majority of the sheep studies is not practical in minipigs.18,24 In a pilot pig study, the feasibility of a cylindrical shape PMMA chamber for the implantation against the periosteum was evaluated (Fig. 6A–C). While the total volume of these chambers is much less than the larger rectangular shapes implemented in ovine studies, the depth of bone formation into the chamber was the same (1 cm). This enables the critical evaluation of osteogenic penetration depth which is the primary variable serving as a tissue volume surrogate.6 In addition, the inner cylindrical shapes minimized the contribution from corners and edges.
FIG. 6.
(A) The exposed periosteal surface (green dashed line) after removal of a 6–8 cm rib segment. (B) Cylindrical shape PMMA molds implanted against periosteum. Examples are images of cylindrical inner PMMA molds with outer (C) cylindrical and (D) rectangular walls. (E) The PMMA mold with rectangular shape typically used in the ovine model. Transparent material around the molds is the “cuff” (ethyl-vinyl-acetate, EVA).
With cylindrical chambers, the fixation of the chamber was challenging. This problem did not occur in studies using chambers with identical inner cylindrical volumes but a rectangular outer shape (Fig. 6C). The new designs of the chambers that maintain the outer structure of rectangular and the inner design can be flexible based on the defect geometry (Fig. 6), allowing optimization of both geometry of the generated tissue, as well as subsequent removal.
The materials used for the fabrication of the chambers must be durable to maintain the defined shape. PMMA,18,25 dacron-polyurethane11 titanium cages,10 and resorbable poly-lactide mesh21 have all been used in previous studies. However, bioresorbable materials do not maintain shape.21 Furthermore, a fracture of a titanium cage in a human patient caused excessive necrotic tissue formation.10 These findings suggest that materials with nondegradable structure and low risk for inflammation are preferred for the chamber preparation.
Evaluation
Analysis of tissues generated within ovine and porcine in vivo bioreactors is typically performed using radiologic, histologic, and/or molecular assays.18,24,25 Microcomputed tomography (microCT) is a valuable technique as it is nondestructive; fixed tissues can be scanned, analyzed, and then used in other techniques. However, microCT only measures radiopaque material. It cannot differentiate viable from nonviable bone nor can it differentiate autologous graft from synthetic ceramic graft. Therefore, histology is a valuable tool for evaluating bioreactor-generated tissues. To examine bone tissue without demineralization, histology is best performed using methacrylate embedding techniques rather than paraffin.24 Image processing methods can be used to differentiate bone from synthetic graft for comparison. However, this method does not adequately separate viable bone from nonviable autologous graft. To assay the amount of living bone generated, the fractional depth of new bone has been quantified.24,25 In addition, histological scoring can be performed on sections.25 Finally, reverse transcriptase polymerase chain reaction of biopsies taken from tissues can be used to analyze gene expression.25
Vascularization is critical for successful bone formation. Immunohistochemical assays can be performed to analyze vascularization in large animals (e.g., endothelial cell markers: Willebrand factor and CD31; mural cell markers: alpha smooth muscle actin [αSMA]). Measure of vascular density (number of vessels/tissue area) and vessel maturity (number of αSMA positive vessels/number of CD31 positive vessels) can be quantified. In settings with large animal imaging systems, MRI or CT could potentially be used to evaluate vascularization.26,27
Recent Advances in Large Animal Models
Ovine model
In a previous review,6 we highlighted the extensive work characterizing the in vivo bioreactor in the ovine model, including optimization of implantation site,12 implantation time,18 and scaffold selection.18,28 However, since that time, significant advances in the in vivo bioreactor strategy for mandibular repair have occurred. These include the use of synthetic biphasic ceramic particles as scaffold material and successful reconstruction of large craniofacial defects.
Previously, morcellized autologous bone was demonstrated to be superior to devitalized bone, as well as biodegradable polymer-based scaffolds.18,28 In recent years, a variety of synthetic ceramic-based graft materials have become available for orthopedic and craniofacial reconstruction. While these scaffolds may lack some of the biological architecture and signals inherent to autologous bone, they offer other advantages such as tunable degradation properties and minimization of donor tissue.29 Therefore, to determine the efficacy of synthetic ceramic graft in producing bone in the ovine in vivo bioreactor, different ratios of autologous graft and clinically-available biphasic ceramic graft (85% beta-tricalcium phosphate, 15% hydroxyapatite) were evaluated.24 The ceramic scaffold was capable of supporting the growth of viable mineralized tissue the periosteum (Fig. 7).
FIG. 7.
Histological cross-section of a bioreactor initially filled with only synthetic biphasic ceramic scaffold (no autologous bone, no exogenous cells, no growth factors) after implantation against the sheep periosteum (bottom) for 9 weeks. Ceramic scaffold appears gray; bone appears dark pink. Greater bone formation occurs closer to the periosteum than the top of the bioreactor.
Incorporation of synthetic ceramic as a scaffold did not have a negative effect on the amount of mineralized tissue grown within the bioreactors. In a subsequent study, bioreactors were implanted with 100% morcellized autologous graft, 100% synthetic ceramic graft, or a 50%/50% (v/v) mixture.25 Regardless of the scaffold used, there were no significant differences between bioreactors in type of tissue generated, maturity of bone tissue, or presence of osteoclast-like cells indicating active remodeling.
Finally, the reconstruction of large craniofacial defects with mineralized tissues generated from in vivo bioreactors has successfully been performed in the ovine model.24,25 The transferred tissues have been generated from both bioreactors filled with autologous24 and synthetic graft.25 A similar approach to the in vivo bioreactor was recently explored in generating tissues appropriate for temporomandibular joint (TMJ) repair in a caprine model.30 Rather than implant scaffolds against the periosteum of the rib, scaffold was implanted against the transition region between the rib costal cartilage and bony periosteum. Thus, tissue generated had a cartilaginous end and bony end similar to the native TMJ. The scaffold was transferred as an avascular graft after 3 months to a TMJ defect 5 cm in length. By 6 months, the transferred tissue had properties indistinguishable from native condyle. Hypertrophic chondrocytes have been seen within bioreactors in the ovine bioreactor model25; these studies suggest that under the proper conditions, it may be possible to leverage the in vivo bioreactor to generate cartilaginous tissues.
Porcine model
Minipigs have been previously used as a model for reconstruction of a mandibular defect with tissue using the latissimus dorsi as the implantation site.19,20 In the development of a periosteum-guided model, empty chambers were tested as a surgical control with the new design of PMMA chambers (cylindrical inner and rectangular outer shape, Fig. 6). At harvest following 4 weeks of implantation the chambers were found still affixed to the periosteum and partially covered with soft tissue.
Chambers containing MBG maintained the cylindrical internal shape at 4 and 8 weeks (Fig. 8A). Qualitatively, microCT images demonstrated the presence of MBG throughout the chamber volume at 4 weeks (Fig. 8B, C). Histological staining revealed that osteocytes and small amounts of fibrovascular tissue formed within the chamber. Mineralization was observed locally around the MBG (Fig. 8D, E). The total bone volume determined from microCT decreased (30%) from 4 to 8 weeks. However, this technique cannot differentiate between MBG and new bone formation. New bone depth assessed from H&E stains did not show a significant difference between weeks 4 and 8.
FIG. 8.
(A) A gross picture of the harvested morcellized bone graft after chamber removal at 4 weeks. (B) A microCT image of the bone graft in A. (C) 3D reconstruction of the regenerated bone volume based on microCT images. (D) A corresponding H&E stained section of image B, scale bar is 1 mm. Purple and dark pink stained cell nuclei and bone, respectively. (E) A higher magnification image from the selected area, showing osteocytes inside the lacuna of the bone matrix. Scale bar is 100 μm.
Comparison Between the Ovine and Porcine Models
Both the sheep and porcine model mimic the surgical technique used in human patients in today's operating room quite well. In addition, the use of rib periosteum as the model implantation site provides a region of high regenerative capacity with low risk of infection. While similar to the ovine model, the porcine model is less expensive in both animal costs and the amount of material used. The size of the rib is smaller in the porcine model, and movement is more vigorous in pigs relative to sheep. The smaller volume potentially allows for screening a larger number of conditions in a single animal. However, the sheep model can be used to examine larger and more complex volumes consistent with clinical application. While the overall volume of tissue that can be formed is lower in the porcine model, it is still designed to assess the critical dimension related to bone invasion depth. The use of sheep may be limited in some institutions due to Q-fever, a zoonotic infection caused by Coxiella burnetii.
Recent findings for the porcine model suggest that MBGs maintained the chamber shape at 4 and 8 weeks, but the regenerated bone volume decreased 30% from week 4 to 8. The loss in bone volume resulted primarily from a decrease in the depth of new bone formation. In the ovine model bone depth reached a maximum at 9 weeks and decreased between 12 and 24 weeks.18 It is unclear why these differences occur, but the sheep model appeared to more closely mimic the absolute depth of bone formation observed in clinical application. Future studies should investigate the role of chamber volume on bone penetration. Both the sheep and porcine models result in incomplete bone formation within the chambers at ∼8 weeks, which is consistent with clinical observations when using MBG.
Discussion and Future Directions
These animal models enable investigators to screen tissue-engineering strategies for large volume tissue formation for an in vivo bioreactor application. The composition of the implants is critical for success of the in vivo bioreactor approach. Bone regression was seen for MBG in the porcine and ovine studies,18 but ceramic supplemented MBGs were successfully transferred to mandibular defect region.25 The development of biomaterial systems, including autologous stem cells and angiogenic and osteogenic factors, might overcome the issues regarding composition of the implants.27,31
A critical question remains determining the optimal time of implantation before transfer to the defect. The current evaluation methods rely on sacrificing the animals and harvesting samples for imaging and analysis. This is costly and time consuming and not possible clinically. The development and evaluation of imaging modalities that could be used to nondestructively monitor tissue development are critical in the field of tissue engineering.32 The location of the implanted chambers limits the application to primarily X-ray and MR-based methods. Techniques based on X-ray phase contrast have been shown to enable the simultaneous evaluation of soft tissue, mineralization, and scaffold structure in bone applications.33 However, these techniques are currently limited to ex vivo analysis. Combination of the in vivo bioreactor with X-ray and MR imaging methods should be evaluated as a means for noninvasive monitoring and assessment.
Topological optimization is a powerful computational technique that could also be utilized to provide better design of the structures required for reconstruction of defects, improving outcomes, and limiting the volume of materials needed.34,35 To find the most optimized macrostructure to replace missing bone that contains the minimum amount of tissue positioned appropriately and that can be supported by remaining uninjured portions of the facial skeleton is novel.35 Topology optimization can also be used to suggest scaffold microstructures that enhance formation.36,37 To improve the biomechanical environments, various material properties such as elasticity that matches bone, bulk modulus and diffusivity, and anisotropy of bones are crucial to design.34 Overall, topological optimization provides a powerful tool for improving the design of 3D tissues for defect reconstruction and should be coupled to the in vivo bioreactor approach.
The recent work in the ovine model showing the successful reconstruction of large craniofacial defects with mineralized tissues generated from in vivo bioreactors was a critical step in accelerating work toward clinical application.24,25 Future studies need to continue to push the evaluation of the viability of tissues engineered in the vivo following transfer. However, clinical impact requires identification of the optimal methods of surgical transfer and an understanding of how the tissues perform following transfer to the defect.
Acknowledgments
This work was supported, in part, by funding from the National Institutes of Health (grants 5R01EB020604), the National Science Foundation (CBET-1263994, IIS-1125412), the Department of Veterans Affairs (5 I01 BX000418-06), and Chang-Gung Memorial Hospital (CMRPG3C1063). The work of AMT and AGM was supported by the Army, Navy, NIH, Air Force, VA, and Health Affairs to support the AFIRM II effort, under Award No. W81XWH-14-2-0004. The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick MD 21702-5014 is the awarding and administering acquisition office. Opinions, interpretations, conclusions, and recommendations are those of the author and are not necessarily endorsed by the Department of Defense. The work of A.S. was supported by the National Science Foundation (1521801).
Disclosure Statement
No competing financial interests exist.
References
- 1.Foster R.D., Anthony J.P., Sharma A., and Pogrel M.A. Vascularized bone flaps versus nonvascularized bone grafts for mandibular reconstruction: an outcome analysis of primary bony union and endosseous implant success. Head Neck 21, 66, 1999 [DOI] [PubMed] [Google Scholar]
- 2.McCullen S.D., Chow A.G., and Stevens M.M. In vivo tissue engineering of musculoskeletal tissues. Curr Opin Biotechnol 22, 715, 2011 [DOI] [PubMed] [Google Scholar]
- 3.Nguyen B.-N.B., Ko H., Moriarty R.A., Etheridge J.M., and Fisher J.P. Dynamic bioreactor culture of high volume engineered bone tissue. Tissue Eng Part A 22, 263, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kretlow J.D., Young S., Klouda L., Wong M., and Mikos A.G. Injectable biomaterials for regenerating complex craniofacial tissues. Adv Mater 21, 3368, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yang Y., Hallgrimsson B., and Putnins E.E. Craniofacial defect regeneration using engineered bone marrow mesenchymal stromal cells. J Biomed Mater Res Part A 99A, 74, 2011 [DOI] [PubMed] [Google Scholar]
- 6.Tatara A.M, Wong M.E., and Mikos A.G. In vivo bioreactors for mandibular reconstruction. J Dent Res 93, 1196, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cheng M., Brey E.M., Ulusal B.G., and Wei F.-C. Mandible augmentation for osseointegrated implants using tissue engineering strategies. Plast Reconstr Surg 118, 1e, 2006 [DOI] [PubMed] [Google Scholar]
- 8.Kokemueller H., Spalthoff S., Nolff M., et al. Prefabrication of vascularized bioartificial bone grafts in vivo for segmental mandibular reconstruction: experimental pilot study in sheep and first clinical application. Int J Oral Maxillofac Surg 39, 379, 2010 [DOI] [PubMed] [Google Scholar]
- 9.Heliotis M., Lavery K.M., Ripamonti U., Tsiridis E., and di Silvio L. Transformation of a prefabricated hydroxyapatite/osteogenic protein-1 implant into a vascularised pedicled bone flap in the human chest. Int J Oral Maxillofac Surg 35, 265, 2006 [DOI] [PubMed] [Google Scholar]
- 10.Warnke P., Springer I., Wiltfang J., and Acil Y. Growth and transplantation of a custome vascularized bone graft on a man. Lancet 364, 766, 2004 [DOI] [PubMed] [Google Scholar]
- 11.Orringer J., Shaw W., Borud L., Freymiller E., Wang S., and Markowitz B. Total mandibular and lower lip reconstruction with a prefabricated osteocutaneous Free Flaps 1, 793, 1999 [DOI] [PubMed] [Google Scholar]
- 12.Brey E.M., Cheng M.-H., Allori A., et al. Comparison of guided bone formation from periosteum and muscle fascia. Plast Reconstr Surg 119, 1216, 2007 [DOI] [PubMed] [Google Scholar]
- 13.Somo S.I., Akar B., Bayrak E.S., et al. Pore interconnectivity influences growth factor-mediated vascularization in sphere-templated hydrogels. Tissue Eng Part C Methods 21, 773, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Akar B., Jiang B., Somo S.I., et al. Biomaterials with persistent growth factor gradients in vivo accelerate vascularized tissue formation. Biomaterials 72, 61, 2015 [DOI] [PubMed] [Google Scholar]
- 15.Jiang B., Akar B., Waller T.M., Larson J.C., Appel A.A., and Brey E.M. Design of a composite biomaterial system for tissue engineering applications. Acta Biomater 10, 1177, 2014 [DOI] [PubMed] [Google Scholar]
- 16.Celik M., Tuncer S., Emekli U., and Kesim S. Histologic analysis of prefabricated vascularized bone grafts: an experimental study in rabbits. J Oral Maxifollac Surg 58, 292, 2000 [DOI] [PubMed] [Google Scholar]
- 17.Holt G.E., Halpern J.L., Dovan T.T., Hamming D., and Schwartz H.S. Evolution of an in vivo bioreactor. J Orthop Res 23, 916, 2005 [DOI] [PubMed] [Google Scholar]
- 18.Cheng M., Brey E.M., Allori A., et al. Ovine model for engineering bone segments. Tissue Eng 11, 214, 2005 [DOI] [PubMed] [Google Scholar]
- 19.Terheyden H., Jepsen S., and Rueger D.R. Mandibular reconstruction in miniature pigs with prefabricated vascularized bone grafts using recombinant human osteogenic protein-1: a preliminary study. Int J Oral Maxillofac Surg 28, 461, 1999 [PubMed] [Google Scholar]
- 20.Terheyden H., Warnke P., Dunsche A., et al. Mandibular reconstruction with prefabricated vascularized bone grafts using recombinant human osteogenic protein-1: an experimental study in miniature pigs. Part II: transplantation. Int J Oral Maxillofac Surg 30, 469, 2001 [DOI] [PubMed] [Google Scholar]
- 21.Warnke P.H., Wiltfang J., Springer I., et al. Man as living bioreactor: fate of an exogenously prepared customized tissue-engineered mandible. Biomaterials 27, 3163, 2006 [DOI] [PubMed] [Google Scholar]
- 22.Lin Z., Fateh A., Salem D.M., and Intini G. Periosteum: biology and applications in craniofacial bone regeneration. J Dent Res 93, 109, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhou M., Peng X., Mao C., Xu F., Hu M., and Yu G.Y. Primate mandibular reconstruction with prefabricated, vascularized tissue-engineered bone flaps and recombinant human bone morphogenetic protein-2 implanted in situ. Biomaterials 31, 4935, 2010 [DOI] [PubMed] [Google Scholar]
- 24.Tatara A.M., Kretlow J.D., Spicer P.P., et al. Autologously generated tissue-engineered bone flaps for reconstruction of large mandibular defects in an ovine model. Tissue Eng Part A 21, 1520, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tatara A.M., Shah S.R., Demian N., et al. Reconstruction of large mandibular defects using autologous tissues generated from in vivo bioreactors. Acta Biomater 45, 124, 2016 [DOI] [PubMed] [Google Scholar]
- 26.Weigand A., Beier J.P., Hess A., et al. Acceleration of vascularized bone tissue-engineered constructs in a large animal model combining intrinsic and extrinsic vascularization. Tissue Eng Part A 21, 1680, 2015 [DOI] [PubMed] [Google Scholar]
- 27.Young S., Kretlow J.D., Nguyen C., et al. Microcomputed tomography characterization of neovascularization in bone tissue engineering applications. Tissue Eng Part B Rev 14, 295, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Thomson R.C., Mikos A.G., Beahm E., et al. Guided tissue fabrication from periosteum using preformed biodegradable polymer scaffolds. Biomaterials 20, 2007 [DOI] [PubMed] [Google Scholar]
- 29.Watson E., Tatara A.M., Kontoyiannis D.P., and Mikos A.G. Inherently antimicrobial biodegradable polymers in tissue engineering. ACS Biomater Sci Eng acsbiomaterials 6b, 100, 2016 [DOI] [PubMed] [Google Scholar]
- 30.Wei J., Herrler T., Han D., et al. Autologous temporomandibular joint reconstruction independent of exogenous additives: a proof-of-concept study for guided self-generation. Sci Rep 6, 37904, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Roux B.M., Cheng M.-H., and Brey E.M. Engineering clinically relevant volumes of vascularized bone. J Cell Mol Med 19, 903, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Appel A.A., Anastasio M.A., Larson J.C., and Brey E.M. Imaging challenges in biomaterials and tissue engineering. Biomaterials 34, 6615, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Appel A.A., Larson J.C., Somo S., Zhong Z., Mikos A.G., et al. Imaging of poly(α-hydroxy-ester) scaffolds with X-ray phase-contrast microcomputed tomography. Tissue Eng Part C Methods 18, 859, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Guedes J.M., Rodrigues H.C., and Bendsøe M.P. A material optimization model to approximate energy bounds for cellular materials under multiload conditions. Struct Multidiscip Optim 25, 446, 2003 [Google Scholar]
- 35.Sutradhar A., Park J., Carrau D., Nguyen T.H., Miller M.J., and Paulino G.H. Designing patient-specific 3D printed craniofacial implants using a novel topology optimization method. Med Biol Eng Comput 54, 1123, 2016 [DOI] [PubMed] [Google Scholar]
- 36.Kang H., Long J.P., Urbiel Goldner G.D., Goldstein S.A., and Hollister S.J. A paradigm for the development and evaluation of novel implant topologies for bone fixation: implant design and fabrication. J Biomech 45, 2241, 2012 [DOI] [PubMed] [Google Scholar]
- 37.Hollister S.J. Porous scaffold design for tissue engineering. Nat Mater 4, 518, 2005 [DOI] [PubMed] [Google Scholar]