Abstract
Background
Primary adenosquamous carcinoma (ASC) of the lung is a rare and aggressive disease. The accurate diagnosis of ASC based on small biopsies is challenging because of the mixed components within the tumor, and this may lead to suboptimal treatment. Furthermore, information about the efficacy of epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) in lung ASC is limited.
Patients and methods
Data on a cohort of patients with lung ASC who underwent surgery between October 2008 and December 2016 at a single institution were retrospectively reviewed.
Results
This study analyzed 148 patients. Differences between the pre- and post-resection diagnosis were observed. Based on the results of preoperative biopsy, patients were diagnosed as having squamous cell carcinoma (n=26), adenocarcinoma (n=20), poorly differentiated carcinoma (n=20), and large cell carcinoma (n=1), and finally diagnosed as having ASC based on histopathological examination of the surgical specimens. Thirty patients (20.3%) with EGFR-sensitizing mutations (TKI group) were treated with EGFR-TKIs after surgery, whereas the remaining patients (79.7%) with unknown EGFR-mutation status received chemotherapy or chemoradiotherapy alone (non-TKI group). TKI treatment was associated with better median overall survival (OS) (HR=0.619; p=0.034). Multivariate analysis identified the presence of EGFR-TKI treatment as an independent prognostic factor for OS (HR=0.471; p=0.003).
Conclusion
Discrepancies between the pre- and post-operative diagnosis reflect the inadequacy of non-resection approaches to the diagnosis of ASC. ASC patients harboring EGFR-sensitizing mutations who were treated with EGFR-TKIs showed a significantly better prognosis than those receiving chemotherapy or chemoradiotherapy alone.
Keywords: adenosquamous carcinoma, lung, EGFR, surgery, recurrence
Introduction
Adenosquamous carcinoma (ASC) is a relatively rare subtype of non-small-cell lung cancer (NSCLC), accounting for 0.4%–4% of all lung cancers.1 The overall prognosis is extremely poor, and most ASC patients with stage III disease cannot survive for more than 5 years.2 The World Health Organization (WHO) Classification of Lung Tumors defines ASC as “a carcinoma showing components of both squamous cell carcinoma (SCC) and adenocarcinoma (AC), with each comprising at least 10% of the tumor”.3 Although ASC is characterized by morphologically mixed tumors that contain adenomatous or squamous components, studies show that ASC has a more aggressive biological behavior and a poorer prognosis than pure AC or SCC,1,2,4,5 suggesting that ASC is not simply a mix of AC and SCC.6,7
The presence of mixed histological components within tumors potentially may lead to inaccurate tumor classification based on small biopsies, which are limited to a partial sampling of the lesion.8–10 This may affect the subsequent design of optimal treatment regimens. Accurate diagnosis is particularly important in patients with lung ASC because AC is a general indication for epidermal growth factor receptor (EGFR) mutation tests, and the survival advantage of pemetrexed treatment can only be achieved in patients with non-squamous histology.11,12 Patients with advanced-stage ASC who are misdiagnosed as SCC may miss out on the benefits of optimal treatment, highlighting the importance of an accurate diagnosis in lung cancer. In addition, excluding the presence of SCC histology is important because bevacizumab, which targets vascular endothelial growth factor, is associated with a high risk of bleeding in SCC and is approved only for the treatment of patients with non-squamous NSCLC.13 As a result, ASC patients in whom only the AC component is identified may not be indicated for bevacizumab treatment.
The best treatment options for ASC patients have not been specifically studied. EGFR tyrosine kinase inhibitors (EGFR-TKIs) are currently recommended as the first-line treatment option for advanced AC with EGFR-sensitizing mutations; however, the efficacy of EGFR-TKIs in patients with ASC remains to be evaluated in detail. Studies show that the incidence of EGFR mutations in East Asian patients with ASC is similar to that in patients with AC;14,15 however, the effect of EGFR-TKIs on the prognosis of ASC patients with EGFR mutation remains unclear because of the rarity of the disease and the potential impact of mixed components.
The objectives of the present study were to investigate whether the mixed structure of ASC affects the accuracy of diagnosis in all patients who have undergone a surgical resection and whether EGFR-TKI treatment can benefit patients with EGFR-positive mutations.
Patients and methods
This retrospective study was approved by the Institutional Ethics Committee of Shanghai Chest Hospital (KS (Y) 1802). Written informed consent was obtained from all patients.
Patients
Post-operatively pathologically confirmed ASC patients who underwent complete tumor resection and lymphadenectomy between January 2008 and December 2015 in the Department of Thoracic Surgery of Shanghai Chest Hospital (Shanghai, China) were included in the study.
These resected samples were reassessed based on the diagnostic criteria proposed by the 2015 edition of the WHO classification system.2
Subgroups
Each surgically resected tumor was systematically sampled according to standard procedures. Paraffin-embedded tumor specimens, which included the widest cross-sections, were reassessed by two senior clinical pathologists. The classification of adenomatous and squamous components within ASCs was performed by immunohistochemical staining of surgically resected tumors. Patients with ASC were subdivided into two groups according to post-surgical EGFR-TKI treatment as follows: the EGFR-TKI group, which included patients who received the TKI treatment in either an adjuvant or a recurrent setting, and the non-TKI group, which included patients who received only chemotherapy (CTx) or chemoradiotherapy (CRTx) over the entire treatment course. Specifically, in the EGFR-TKIs group, most patients received gefitinib (Iressa®, AstraZeneca; 250 mg once daily; n=27), whereas two patients received erlotinib (Tarceva®, Roche; 150 mg once daily; n=3). In the non-TKI group, CTx (cisplatin-based doublet) or CRTx (cisplatin-based doublet plus 45–70 Gy radiotherapy) was given.
Mutational analyses and clinical assessments
The mutational status of EGFR (exons 18–21) was determined using polymerase chain reaction-based direct sequencing and verified by DNA sequencing analysis. All mutations were verified by analysis of an independent polymerase chain reaction.
Objective tumor responses regarding complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) using the Response Evaluation Criteria in Solid Tumors (RECIST 1.1) were retrieved from the medical records.16
Follow-up
Follow-up information was acquired by review of the electronic medical records and by calls. The primary endpoints of the study were overall survival (OS), which was defined as the interval between the day of recurrence and the date of death by any cause or the last follow-up date, and post-operative OS, which was defined as the interval between the day of surgery and the date of death by any cause or the last follow-up date. Recurrence was defined as the proven detection of locoregional recurrence or distant metastasis.
Statistical analyses
Normally distributed continuous variables are presented as the mean ± SD or as the median and interquartile range, while categorical variables are presented as numbers and percentages. Categorical variables were compared using contingency table analysis and chi-squared tests. Survival curves were estimated using the Kaplan–Meier method and compared using the log-rank test. The variables found to be significant on univariate analysis (defined as a p-value of less than 0.15) were included in the multivariate analysis using the Cox proportional hazards model after backward stepwise Wald elimination. Other clinically relevant factors based on clinical knowledge and previous reports, such as age, gender, and smoking history, are also included in the Cox proportional hazards model. All tests were two sided and a p-value <0.05 was considered to indicate statistical significance. Statistical analyses were performed using SPSS software (version 19.0; IBM-SPSS, Armonk, NY, USA).
Results
The study included 148 patients with ASC. The clinical characteristics of the patients are listed in Table 1. The mean age was 59.9±10.4 years. Most of the patients were men (70.7%) and had a smoking history (57.4%). Women (53.3% vs 25.4%, p=0.003), never-smokers (70.0% vs 35.6%, p=0.001), a central tumor (56.7% vs 28.8%, p=0.008), and extended lobectomy (46.7% vs 22.0%, p=0.007) were significantly more frequent in the EGFR-TKI treatment group than in the CTx/CRTx alone group.
Table 1.
Clinical characteristics of patients with primary ASCs of the lung
| Characteristic | ASC
|
Subgroups
|
||
|---|---|---|---|---|
| Total, n (%) | EGFR-TKIs | CTx/CRTx | p-value | |
| Gender | 148 (100) | 30 | 118 | 0.003* |
| Male | 102 (70.7) | 14 | 88 | |
| Female | 46 (29.3) | 16 | 30 | |
| Age (years) | 0.325 | |||
| ≤60 | 72 (48.6) | 17 | 55 | |
| >60 | 76 (51.4) | 13 | 63 | |
| Smoking | 0.001* | |||
| No | 63 (42.6) | 21 | 42 | |
| Yes | 85 (57.4) | 9 | 76 | |
| Tumor location | 0.008* | |||
| Central | 51 (34.5) | 17 | 34 | |
| Peripheral | 97 (65.5) | 13 | 84 | |
| Type of resection | 0.007* | |||
| Lobectomy | 108 (73.0) | 16 | 92 | |
| Extended resection | 40 (27.0) | 14 | 26 | |
| Sleeve resection | 17 (11.5) | 5 | 12 | |
| Bilobectomy | 13 (8.8) | 6 | 7 | |
| Pneumonectomy | 10 (6.8) | 3 | 7 | |
| TNM stage | ||||
| I | 33 (22.3) | 5 | 28 | 0.299 |
| II | 40 (27.0) | 6 | 34 | |
| IIIa | 75 (50.7) | 19 | 56 | |
Note:
Statistically significant at p<0.05.
Abbreviations: ASC, adenosquamous carcinoma; EGFR, epidermal growth factor receptor; TKI, tyrosine kinase inhibitor; CTx, chemotherapy; CRTx, chemoradiotherapy.
Differential diagnosis between pre- and post-resection
A total of 67 patients (45.3%) were diagnosed pre-operatively with SCC (n=26), AC (n=20), poorly differentiated carcinoma (n=20), and large cell carcinoma (n=1), according to cytological and/or histological analysis, and finally diagnosed as ASC according to histopathological examination of the surgical specimens, which indicates a considerable inconsistency between pre- and post-operative diagnosis.
Efficacy of EGFR-TKI treatment
Thirty patients harboring sensitizing EGFR mutations, including DEL in exon 19 (n=16) and point mutation at codon 858 (L858R) in exon 21 (n=14), received EGFR-TKIs. Of those, 26 patients (86.7%) received EGFR-TKI treatment after recurrence, and four (13.3%) received TKIs as a postoperative adjuvant therapy. Of these 30 patients, 11 achieved PR and 12 had SD following EGFR-TKI treatment, accounting for a disease control rate of 76.7% (23/30). In the non-TKI group (n=118), only 15 patients had had a definitely prospective EGFR-mutation test, including 12 with a wild-type of EGFR and three with an EGFR-sensitizing mutation, and 89 patients never had the mutation detections. For the remaining 14 cases, the information on EGFR-mutation detection was missing.
In the non-TKI group, 21 patients (75.0%) with stage I, and all patients with stage II and IIIa disease received four to six cycles of CTx after surgery.
Survival analysis
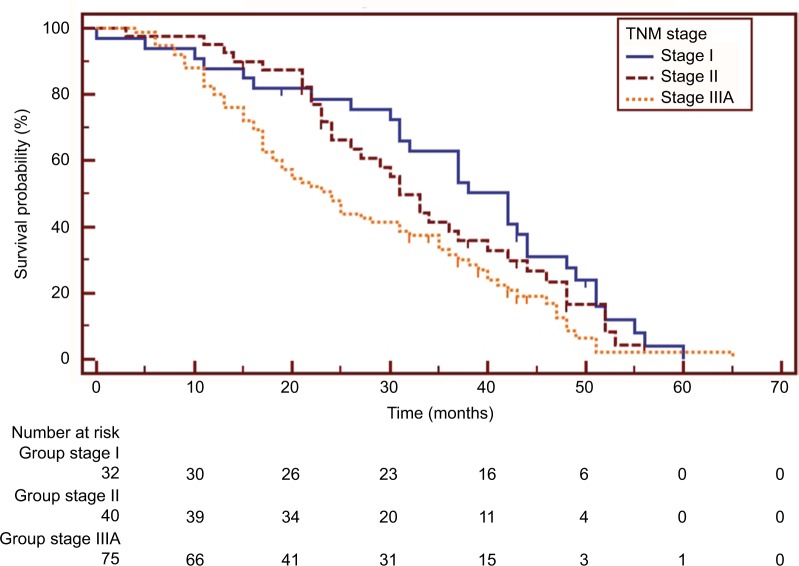
The survival data of one patient in the non-TKI group were missing. Overall, 131 patients (88.5%) had died at the last follow-up visit. The post-operative OS at 5 years and median OS were 1.6% and 31.0 (25.9–36.1) months, respectively. The median OS of patients with stage I, II, and IIIA disease was 42.0 (34.3–49.7), 31.0 (26.6–35.4), and 24.0 (18.9–29.1) months, respectively, and the difference was statistically significant (stage I vs IIIa: HR=0.579, 95% CI 0.385–0.872, p=0.011; stage II vs IIIa: HR=0.686, 95% CI 0.452–1.043, p=0.053) (Figure 1).
Figure 1.
Kaplan–Meier survival curves of overall survival analysis in patients with different disease stages (I vs IIIa: HR=0.579, 95% CI 0.385–0.872, p=0.011; II vs IIIa: HR=0.686, 95% CI 0.452–1.043, p=0.053).
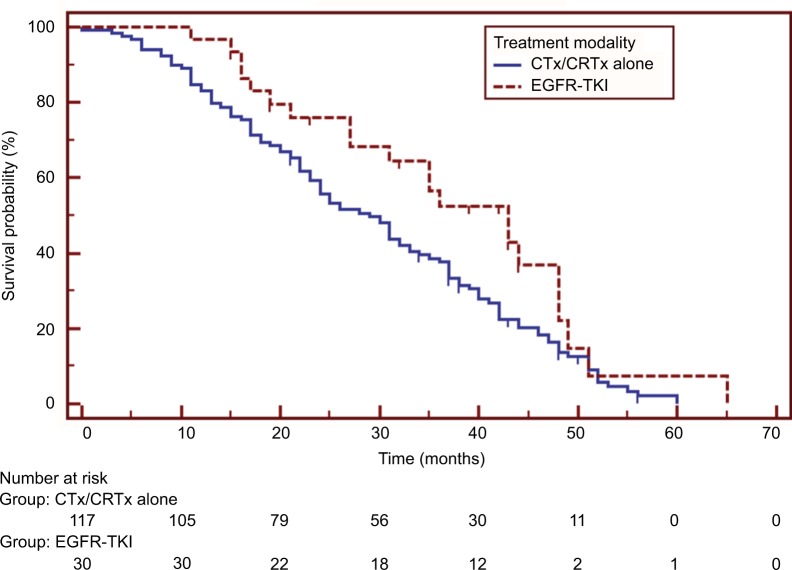
Patients in the EGFR-TKI treatment group had a significantly better median OS than those in the non-TKI group (HR=0.619, 95% CI 0.414–0.927, p=0.034), as shown in Figure 2. Univariate analysis demonstrated that pathological stages and treatment modalities were significant prognostic factors for post-operative OS (Table 2). Further multivariate analysis using the Cox proportional hazards model identified pathological stage (stage I vs IIIa: HR=0.463, 95% CI 0.293–0.731, p=0.001; stage II vs IIIa: HR=0.565, 95% CI 0.366–0.871, p=0.010) and the presence of EGFR-TKI treatment (HR=0.471, 95% CI 0.286–0.775, p=0.003) as independent prognostic factors of post-operative OS (Table 3).
Figure 2.
Survival comparisons between patients treated with epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKI group) and those with chemotherapy or chemoradiotherapy (CTx/CRTx alone group). Patients receiving EGFR-TKIs had a significantly longer survival (HR=0.619, 95% CI 0.414–0.927, p=0.034).
Table 2.
Impact of individual variables on the survival of adenosquamous carcinoma patients estimated by univariate analysis
| Variable | Post-operative OS | |
|---|---|---|
|
| ||
| Median (95% CI) (months) | p-value | |
| Gender | 0.185 | |
| Male | 31.0 (26.6–35.4) | |
| Female | 31.0 (19.4–42.6) | |
| Age (years) | 0.663 | |
| ≤60 | 28.0 (21.6–34.4) | |
| >60 | 32.0 (25.6–38.4) | |
| Smoking | 0.728 | |
| No | 31.0 (22.5–39.5) | |
| Yes | 31.0 (24.2–37.8) | |
| Tumor location | 0.798 | |
| Central | 33.0 (22.9–43.1) | |
| Peripheral | 31.0 (25.6–36.5) | |
| Type of resection | 0.873 | |
| Lobectomy | 31.0 (14.0–48.0) | |
| Extended resection | 31.0 (26.5–35.5) | |
| TNM stage | 0.017* | |
| I | 42.0 (34.3–49.7) | |
| II | 31.0 (26.6–35.4) | |
| IIIa | 24.0 (18.9–29.1) | |
| Treatment modality | 0.034* | |
| EGFR-TKIs | 43.0 (31.4–54.6) | |
| CTx/CRTx alone | 29.0 (23.7–34.3) | |
Note:
Statistically significant at p<0.05.
Abbreviations: CTx, chemotherapy; CRTx, chemoradiotherapy; EGFR, epidermal growth factor receptor; OS, overall survival; TKI, tyrosine kinase inhibitor.
Table 3.
Multivariate analysis
| Variable | Post-operative OS
|
||
|---|---|---|---|
| HR | 95% CI | p-value | |
| TNM stage | |||
| I | 0.463 | 0.293–0.731 | 0.001* |
| II | 0.565 | 0.366–0.871 | 0.010* |
| IIIa | Ref. | ||
| Treatment after surgery | |||
| EGFR-TKIs | 0.471 | 0.286–0.775 | 0.003* |
| CTx/CRTx alone | Ref. | ||
Note:
Statistically significant at p<0.05.
Abbreviations: CTx, chemotherapy; CRTx, chemoradiotherapy; EGFR, epidermal growth factor receptor; OS, overall survival; TKI, tyrosine kinase inhibitor; Ref., Reference.
Discussion
Lung ASC is a highly aggressive disease. The role of CRx/CRTx is limited because of the aggressive nature of the disease and the mixed components of ASCs, and patients with lung ASC usually experience a very short disease-free time after surgery. However, information about lung ASC is limited and little progress has been made regarding the optimal management of ASC patients. This is partly attributed to the fact that there are very few patients with ASC, which, based on our study, may not be the case.
The ASC patients included in the present study underwent complete resections, which provided a sufficient number of specimens for diagnosis. By contrast, cytological analysis or small biopsy provides limited diagnostic information and thus may not be truly representative of lung cancer with a mixed histology.8–10,17 Sixty-seven patients with post-operatively pathologically proven ASCs were pre-operatively misdiagnosed as having SCC (n=26), poorly differentiated SCC (n=20), AC (n=20), or large cell carcinoma (n=1) through cytology (diagnostic fine-needle aspiration or bronchial brush and lavage) or bronchial biopsy. This indicates a discrepancy in the diagnostic outcomes of ASC between pre-operative evaluations based on cytology or bronchial biopsy and post-operative diagnosis by immunohistochemistry, suggesting a diagnostic dilemma for ASC cases treated by non-resection approaches. This is an important issue in potential cases of ASC that are not surgical candidates, as patients with advanced-stage ASC who are misdiagnosed as SCC may miss out on the benefits of EGFR-mutation tests and TKI treatment. In addition, AC is one of the most frequent pre-operative diagnoses in our current series of ASC cases. Misdiagnosis can lead to severe complications, as SCC histology is associated with a high risk of pulmonary hemorrhage when treated with bevacizumab.13 Therefore, the identification of specific biomarkers of ASC is essential. In addition, our study may provide a possible interpretation of the histological differentiation between pre- and post-treatment in lung cancer cases showing “transdifferentiation” between SCC and AC caused by drug treatment stress.18,19 Some of these cases are associated with the high heterogeneity of the ASC and the inadequate sampling, rather than real transdifferentiation.
The incidence of EGFR mutations and the efficacy of EGFR-TKIs have been mainly evaluated in lung AC. However, in patients with non-AC NSCLCs, the value of detecting EGFR mutations remains unclear.20 In patients with ASC, genetic mutations are rarely evaluated, and research on the efficacy of EGFR-TKIs in ASC patients with EGFR mutations is limited. This is partly due to its rarity and the unclear value of assessing oncogenic mutations in ASCs. As a result, few relevant papers have been published, and most of these are retrospective evaluations of the frequency of EGFR mutations in ASCs.21–25 The present study compared EGFR-TKIs to conventional CTx in patients with lung ASCs. Survival comparisons between ASC patients with EGFR-sensitizing mutations who were treated with EGFR-TKIs and unselected ASC patients treated with CRx/CRTx alone after surgery indicated that ASC patients with EGFR-sensitizing mutations could benefit significantly from EGFR-TKI treatment.
The paucity of studies on the efficacy of EGFR-TKIs in lung ASC patients can be attributed to several factors, such as the relative rarity of ASC patients and the lack of awareness of the clinical significance of mutation tests in non-AC lung cancers. However, the insufficient diagnostic information provided by non-resected biopsies may be another contributory factor.
One of the major limitations of this study was the small sample size of ASC patients treated with TKIs. In addition, the frequency of EGFR mutations in the present cohort was not investigated. Therefore, both patients with mutant and wild-type EGFR may have been included in the non-TKI group, which may compromise the comparison of patient survival between the TKI and non-TKI groups. However, studies have shown no significant survival difference between adenosquamous lung cancer patients with wild-type EGFR and those with a mutation.21,23 In addition, the specific component within ASCs harboring EGFR mutations could not be identified because microdissection was not performed, and the mixed components were assessed separately. Finally, the effect of the different proportions of structural components within ASCs on the treatment response to TKIs was not evaluated because of the small sample size. Therefore, further studies are warranted.
Conclusion
The accuracy of non-resection approaches for the histological classification of lung ASC was relatively low, and a considerable number of potential ASC patients may miss out on optimal treatment owing to incorrect diagnostic information from the non-resected biopsies. EGFR mutation-positive ASC patients could achieve good disease control after EGFR-TKI treatment, and experience a significantly improved prognosis from EGFR-TKI treatment in comparison with unselected patients receiving standard CTx alone.
Acknowledgments
The authors are indebted to Keke Yu and Jie Xing (Department of Pathology of Shanghai Chest Hospital, Shanghai Jiaotong University) for pathological re-assessment.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Cooke DT, Nguyen DV, Yang Y, et al. Survival comparison of adenosquamous, squamous cell, and adenocarcinoma of the lung after lobectomy. Ann Thorac Surg. 2010;90(3):943–948. doi: 10.1016/j.athoracsur.2010.05.025. [DOI] [PubMed] [Google Scholar]
- 2.Gawrychowski J, Bruliński K, Malinowski E, Papla B. Prognosis and survival after radical resection of primary adenosquamous lung carcinoma. Eur J Cardiothorac Surg. 2005;27(4):686–692. doi: 10.1016/j.ejcts.2004.12.030. [DOI] [PubMed] [Google Scholar]
- 3.Travis WD, Brambilla E, Burke AP, Marx A, Nicholson AG. WHO classification of tumours of the lung, pleura, thymus and heart. Lyon: International Agency for Research on Cancer; 2015. [DOI] [PubMed] [Google Scholar]
- 4.Maeda H, Matsumura A, Kawabata T, et al. Adenosquamous carcinoma of the lung: surgical results as compared with squamous cell and adenocarcinoma cases. Eur J Cardiothorac Surg. 2012;41(2):357–361. doi: 10.1016/j.ejcts.2011.05.050. [DOI] [PubMed] [Google Scholar]
- 5.Filosso PL, Ruffini E, Asioli S, et al. Adenosquamous lung carcinomas: a histologic subtype with poor prognosis. Lung Cancer. 2011;74(1):25–29. doi: 10.1016/j.lungcan.2011.01.030. [DOI] [PubMed] [Google Scholar]
- 6.Bastide K, Ugolin N, Levalois C, Bernaudin JF, Chevillard S. Are adenosquamous lung carcinomas a simple mix of adenocarcinomas and squamous cell carcinomas, or more complex at the molecular level? Lung Cancer. 2010;68(1):1–9. doi: 10.1016/j.lungcan.2009.11.001. [DOI] [PubMed] [Google Scholar]
- 7.Zhao H, Yang H, Yao F, et al. Improved survival associated with a balanced structure between adenomatous and squamous components in patients with adenosquamous carcinoma of the lung. Eur J Surg Oncol. 2016;42(11):1699–1706. doi: 10.1016/j.ejso.2016.05.009. [DOI] [PubMed] [Google Scholar]
- 8.Shelton DA, Rana DN, Holbrook M, Taylor P, Bailey S. Adenosquamous carcinoma of the lung diagnosed by cytology?: a diagnostic dilemma. Diagn Cytopathol. 2012;40(9):830–833. doi: 10.1002/dc.21664. [DOI] [PubMed] [Google Scholar]
- 9.Chen J, Gao YD, Cao Y, Yang J, Luo GW. Surgical specimen histology revealed inadequacy of conventional transbronchial needle aspiration sample in the diagnosis of adenosquamous lung carcinoma. J Thorac Dis. 2015;7(4):680–686. doi: 10.3978/j.issn.2072-1439.2015.04.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang C, Yang H, Zhao H, et al. Clinical outcomes of surgically resected combined small cell lung cancer: a two-institutional experience. J Thorac Dis. 2017;9(1):151–158. doi: 10.21037/jtd.2017.01.07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Scagliotti GV, Parikh P, von Pawel J, et al. Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol. 2008;26(21):3543–3551. doi: 10.1200/JCO.2007.15.0375. [DOI] [PubMed] [Google Scholar]
- 12.Scagliotti G, Hanna N, Fossella F, et al. The differential efficacy of pemetrexed according to NSCLC histology: a review of two Phase III studies. Oncologist. 2009;14(3):253–263. doi: 10.1634/theoncologist.2008-0232. [DOI] [PubMed] [Google Scholar]
- 13.Johnson DH, Fehrenbacher L, Novotny WF, et al. Randomized phase II trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J Clin Oncol. 2004;22(11):2184–2191. doi: 10.1200/JCO.2004.11.022. [DOI] [PubMed] [Google Scholar]
- 14.Jia XL, Chen G. EGFR and KRAS mutations in Chinese patients with adenosquamous carcinoma of the lung. Lung Cancer. 2011;74(3):396–400. doi: 10.1016/j.lungcan.2011.04.005. [DOI] [PubMed] [Google Scholar]
- 15.Song Z, Lin B, Shao L, Zhang Y. Therapeutic efficacy of gefitinib and erlotinib in patients with advanced lung adenosquamous carcinoma. J Chin Med Assoc. 2013;76(9):481–485. doi: 10.1016/j.jcma.2013.05.007. [DOI] [PubMed] [Google Scholar]
- 16.Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1) Eur J Cancer. 2009;45(2):228–247. doi: 10.1016/j.ejca.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 17.Dragnev KH, Gehr G, Memoli VA, Tafe LJ. ALK-rearranged adenosquamous lung cancer presenting as squamous cell carcinoma: a potential challenge to histologic type triaging of NSCLC biopsies for molecular studies. Clin Lung Cancer. 2014;15(3):e37–e40. doi: 10.1016/j.cllc.2014.01.003. [DOI] [PubMed] [Google Scholar]
- 18.Jukna A, Montanari G, Mengoli MC, et al. Squamous cell carcinoma “transformation” concurrent with secondary T790M mutation in resistant EGFR-mutated adenocarcinomas. J Thorac Oncol. 2016;11(4):e49–e51. doi: 10.1016/j.jtho.2015.12.096. [DOI] [PubMed] [Google Scholar]
- 19.Longo L, Mengoli MC, Bertolini F, et al. Synchronous occurrence of squamous-cell carcinoma “transformation” and EGFR exon 20 S768I mutation as a novel mechanism of resistance in EGFR-mutated lung adenocarcinoma. Lung Cancer. 2017;103:24–26. doi: 10.1016/j.lungcan.2016.11.012. [DOI] [PubMed] [Google Scholar]
- 20.Song X, Wang Z. Clinical efficacy evaluation of tyrosine kinase inhibitors for non-adenocarcinoma lung cancer patients harboring EGFR-sensitizing mutations. Onco Targets Ther. 2017;10:3119–3122. doi: 10.2147/OTT.S134523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wang R, Pan Y, Li C, et al. Analysis of major known driver mutations and prognosis in resected adenosquamous lung carcinomas. J Thorac Oncol. 2014;9(6):760–768. doi: 10.1097/JTO.0b013e3182a406d1. [DOI] [PubMed] [Google Scholar]
- 22.Powrózek T, Krawczyk P, Ramlau R, et al. EGFR gene mutations in patients with adenosquamous lung carcinoma. Asia Pac J Clin Oncol. 2014;10(4):340–345. doi: 10.1111/ajco.12177. [DOI] [PubMed] [Google Scholar]
- 23.Morodomi Y, Okamoto T, Takenoyama M, et al. Clinical Significance of Detecting Somatic Gene Mutations in Surgically Resected Adeno-squamous Cell Carcinoma of the Lung in Japanese Patients. Ann Surg Oncol. 2015;22(8):2593–2598. doi: 10.1245/s10434-014-4218-0. [DOI] [PubMed] [Google Scholar]
- 24.Shiozawa T, Ishii G, Goto K, et al. Clinicopathological characteristics of EGFR mutated adenosquamous carcinoma of the lung. Pathol Int. 2013;63(2):77–84. doi: 10.1111/pin.12033. [DOI] [PubMed] [Google Scholar]
- 25.Fan L, Yang H, Yao F, et al. Clinical outcomes of epidermal growth factor receptor tyrosine kinase inhibitors in recurrent adenosquamous carcinoma of the lung after resection. Onco Targets Ther. 2017;10:239–245. doi: 10.2147/OTT.S114451. [DOI] [PMC free article] [PubMed] [Google Scholar]