Abstract
Background:
The drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome is a life-threatening condition caused by different medications. The objective of this study was to analyze DRESS cases related to antiretroviral therapy in human immunodeficiency virus (HIV) patients.
Materials and Methods:
Systematic review of DRESS suspected cases in HIV patients associated to antiretrovirals published between January 1998 and April 2017. The registry of the severe cutaneous adverse reactions score was used to classify each report as a “definitive,” “probable,” “possible,” or “no” DRESS case. Clinical characteristics, management, and outcomes were evaluated.
Results:
Thirty-five case reports were analyzed involving 5 antiretrovirals: Abacavir in 10 (28.6%) cases, efavirenz in 6 (17.1%), nevirapine in 12 (34.3%), raltegravir in 6 (17.1%), and tenofovir in 1 (2.9%). Mean age of the patients was 40 ± 13 years, 65% of which were male. A total of 57.1% reports were classified as having a “definitive-probable” DRESS case. Management was based on withdrawal of the causal antiretroviral and corticosteroids in 68.6% of the cases. None of the patients died. Treatment with nevirapine or raltegravir, the longer onset of symptoms and the presence of lymphadenopathy, eosinophilia, liver involvement, and a longer time for clinical resolution were more frequent among “definitive-probable” DRESS cases.
Conclusions:
A DRESS syndrome has to be suspected in HIV patients with lymphadenopathy, eosinophilia, and liver involvement developing weeks after the initiation of nevirapine or raltegravir. Suspension of the causal antiretroviral and in most cases treatment with corticosteroids allowed adequate clinical control.
Key words: Antiretrovirals, drug hypersensitivity reactions, drug reaction with eosinophilia and systemic symptoms syndrome, highly active antiretroviral therapy, human immunodeficiency virus, management
INTRODUCTION
The introduction of highly active antiretroviral therapy (HAART) in the management of human immunodeficiency virus (HIV) infection has had a great impact in the natural evolution of the disease increasing life expectancy of infected patients.[1] The favorable impact of HAART has had some important downsides. These are mainly related to an increased incidence of adverse drug reactions, including drug hypersensitivity reactions (DHR), which are more frequent in HIV patients than in the general population.[2] While mild to moderate rash, the most common form of DHR, can be managed without discontinuation of the causal drug withdrawal is obligated in severe cases.[3]
The drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome is a potentially life-threatening delayed DHR characterized by extensive rash, fever, eosinophilia, and different degrees of organ involvement.[4] Factors implicated in the pathogenesis of DRESS syndrome include defects in drug metabolism, linked to deficiencies of detoxifying enzymes, leading to the accumulation of toxic reactive intermediates.[5] An association between certain human leukocyte antigen (HLA) and DRESS syndrome has also been described.[6] Additional factors include reactivation of herpes virus, mainly human herpes virus 6 (HHV-6).[7]
DRESS syndrome has an estimated incidence that ranges from 1 in 1000 to 1 in 10,000 new drug exposures.[8] In general, there is 2–8 weeks latency period between the initiation of the causal drug and the beginning of the clinical manifestations.[4] Aromatic anticonvulsants, allopurinol, and antimicrobial sulfonamides are the most commonly involved medications.[9,10] The DRESS syndrome has an associated mortality rate of 2% to 10%, mainly due to liver failure.[10,11] This relatively high mortality has been related to the prolonged latency period and the variable clinical expression that delays the suspension of the causal drug. In an attempt to facilitate the diagnosis of DRESS syndrome the European registry of severe cutaneous adverse reactions (RegiSCAR) has developed a scoring system, combining clinical and biological items.[12]
HIV patients frequently receive different medications for the prevention or the treatment of opportunistic infections, such as cotrimoxazole and tuberculostatic drugs, that have been involved in DRESS cases.[9,10,13] Although several DRESS reports in HIV patients associated to antiretrovirals have been published, to the best of our knowledge, a systematic review has not been undertaken.
The objective of this study was to analyze the clinical characteristics, management and prognosis of DRESS cases related to antiretroviral therapy in HIV patients.
MATERIALS AND METHODS
A search of DRESS syndrome suspected cases associated to HAART in HIV patients, published between January 1998 and April 2017, was carried out through EMBASE and MEDLINE databases. Search terms used included : “DRESS syndrome and HIV,” “drug-induced hypersensitivity syndrome and HIV,” “drug-induced hypersensitivity and antiretroviral,” and “DRESS syndrome and antiretroviral” Case reports in English, French, and Spanish languages were considered.
The following clinical and demographic variables were assessed: Age, gender, ethnicity, pregnancy, viral load, CD4 lymphocyte count, type of HAART, concomitant diagnosis of HHV-6 or other infections, and diagnostic test performed to rule out alternative diagnosis. Time of onset of symptoms, type of organ involvement, treatment administered, and outcomes were also analyzed. In addition, we recorded whether the RegiSCAR diagnostic score was provided by the report and when not given it was calculated based on the published criteria.[12] Thus, the following clinical and biological data were reviewed: The presence of fever (>38.5°C), enlarged lymph nodes (>2 sites, >1 cm), atypical lymphocytes, eosinophilia (eosinophil count >700/mm3 or >10% of the white blood cell count), skin rash (extent >50%, presence of edema, infiltration, purpura, scaling, or a skin biopsy suggesting DRESS), and internal organ involvement.
Hepatic involvement was considered when the level of alanine aminotransferase was greater than twice the normal values. Kidney involvement was defined as an elevation in serum creatinine levels 2 to –3 times above the baseline limits following the RIFLE criteria or if proteinuria was present.[14] Myalgias or rhabdomyolysis was considered a sign of muscular damage. Respiratory, gastrointestinal, or neurological symptoms without an alternative cause were also considered organ involvement. We also reviewed the time to resolution of the symptoms (<2 or >2 weeks) and whether at least 3 biological investigations were carried out to exclude alternative diagnosis. The score of each item ranged from-1 to 2. Patients with <2 points were classified as not having a DRESS syndrome while patients with 2–3 points, 4–5 and >5 were classified as having a “possible,” “probable,” or a “definite” DRESS syndrome, respectively. Cases with a “definitive” or a “probable” DRESS syndrome were pooled together and compared to those with a “possible” or “no” DRESS case.
Qualitative data are expressed as percentages and quantitative data as mean (±standard deviations). Continuous variables were compared using Mann–Whitney U-test and the categorical data using Chi-square or the Fisher's exact test as appropriate. Statistical significance was defined as a two-tailed value of P < 0.05. Logistic regression analysis was used to determine independent variables associated with “definitive-probable” DRESS cases including variables with a P < 0.1 in the univariate analysis. Statistical analysis was performed by the program SPSS (version 20.0; SPSS, Inc., Chicago, IL, USA).
RESULTS
Figure 1 depicts the flow chart followed during the selection process. A final sample of 35 cases was analyzed involving 5 antiretrovirals: Abacavir in 10 (28.6%) cases,[15,16,17,18,19,20,21,22,23,24] efavirenz in 6 (17.1%),[25,26,27,28,29,30] nevirapine in 12 (34.3%),[31,32,33,34,35,36,37,38,39,40] raltegravir in 6 (17.1%),[41,42,43,44,45,46] and tenofovir in 1 (2.9%) case.[47] Table 1 shows the characteristics of the included patients. Most cases involved middle-aged patients with a male gender preference. Among the females, 2 cases appeared during pregnancy or in the immediate postpartum. Although ethnicity was underreported, the Caucasian race was more frequent among “possible-no” DRESS cases, whereas 5 (83%) of the patients with a DRESS syndrome caused by raltegravir were of African origin.
Figure 1.
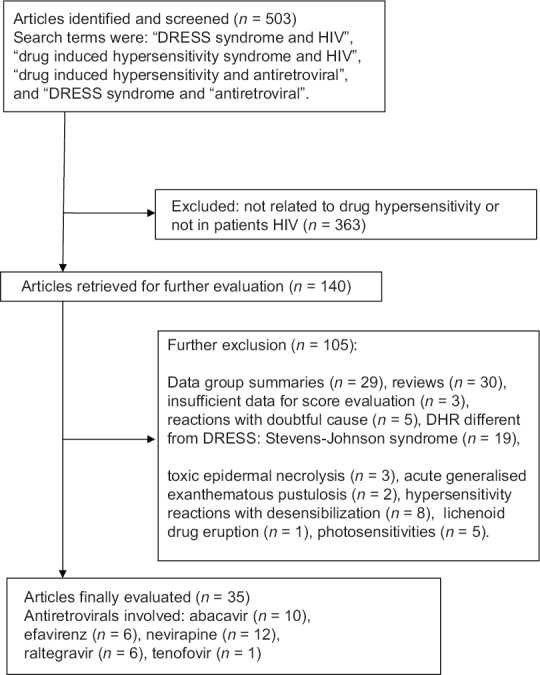
Flow diagram followed during the selection process. DRESS=Drug reaction with eosinophilia and systemic symptoms; HIV= Human immunodeficiency virus; DHR=Drug hypersensitivity reaction
Table 1.
Baseline findings of human immunodeficiency virus patients included in the study and univariate analysis of characteristics and outcomes of “definitive-probable” compared to “possible-no” drug reaction with eosinophilia and systemic symptoms cases
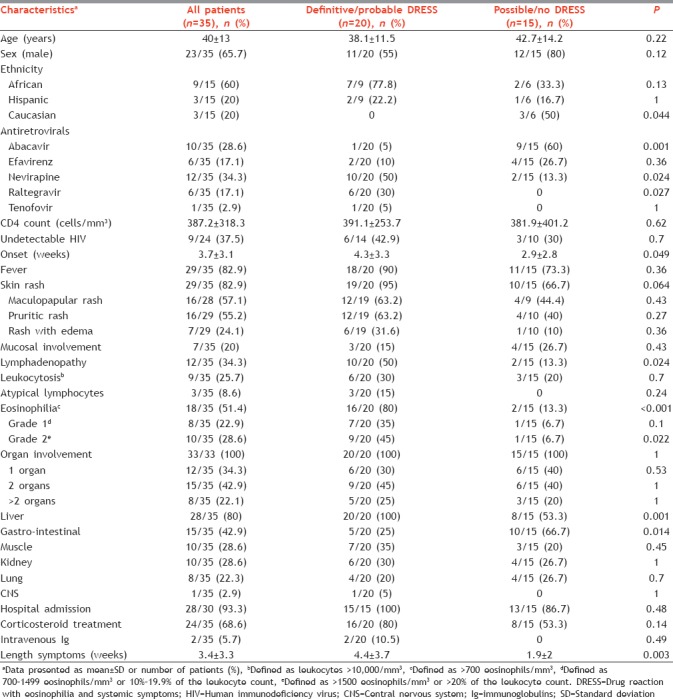
Fifteen out of 28 (53.5%) patients had more than 400 CD4/mm3. The mean CD4 count in patients receiving nevirapine or abacavir was 478.9 ± 184.1 cells/mm3 and 390.8 ± 454.3 cells/mm3, respectively (P = 0.31). Fever and skin rash, generally described as maculopapular, were the most common clinical manifestations. A skin biopsy was performed in 5 patients showing changes compatible with the diagnosis of DRESS syndrome in all of them. Eosinophilia was the most frequent hemotologic abnormality. A certain degree of organ involvement was present in all of the cases. The liver was the organ most frequently affected. A liver biopsy was performed in 2 patients demonstrating abnormalities that suggested a viral hepatitis in one. Gastrointestinal symptoms were present in 43% of the cases, mainly diarrhea. Although kidney dysfunction was common and generally reported as a mild elevation in serum creatinine levels, 2 cases receiving efavirenz developed an acute renal failure. One of them required hemodialysis with a renal biopsy showing nephritis with eosinophilic infiltrates. Less common was the central nervous system involvement appearing in one patient receiving nevirapine. Additional uncommon manifestations included a case of disseminated intravascular coagulation and another one of agranulocytosis in 2 patients taking abacavir.
There was information of at least 3 biological tests that were done to exclude alternative diagnosis in 68.5% of the analyzed case reports. The existence of an HHV-6 infection was evaluated in 17.1% of the cases with only one confirmed infection in a patient taking tenofovir. The existence of certain HLA alleles associated with DRESS syndrome predisposition was found in 3 out of the 6 patients in which the genetic test was performed. The HLA-B*5701 was found in one patient taking abacavir while another one with a nevirapine definitive DRESS case was homozygous for the HLA-DRB1*0701. The recently described HLA-B*53:01 was described in one patient with a raltegravir-induced DRESS syndrome.
Table 1 provides information about the management of the included patients. In 34 out of the 35 (97.1%) cases, the report clearly stated that the antiretroviral most likely involved was withdrawn from the treatment. The vast majority of the patients were hospitalized and 68.6% required systemic corticosteroids with different doses and duration. In addition, 2 patients receiving nevirapine were treated with intravenous immunoglobulins. After the suspension of the causal antiretroviral, normalization of the clinical and biological parameters was achieved after 2 weeks in 54.3% of the patients. None of the reviewed patients died.
The RegiSCAR score was only provided in 6 (17.1%) of the reviewed reports, and hence, it was calculated in the rest of the cases. Patients with a “definitive-probable” DRESS case had higher median RegiSCAR scores when compared to those with a “possible-no” DRESS case (5.1 ± 0.9 and 1.8 ± 1.1, respectively, P < 0.001). Table 2 summarizes the RegiSCAR classification for each of the 5 different antiretrovirals analyzed. Among the 20 (57.1%) patients with a “definitive-probable” DRESS case, 10 (50%) were taking nevirapine, and 6 (30%) raltegravir. Fifteen patients had a “possible-no” DRESS case, of which 9 (60%) were receiving abacavir and 4 (26.6%) efavirenz.
Table 2.
Classification of published drug reaction with eosinophilia and systemic symptoms cases according to the registry of severe cutaneous adverse reactions score

Table 1 also shows the clinical characteristics and outcomes of HIV patients with a “definitive-probable” DRESS syndrome compared to those with a “possible-no” DRESS case. In the univariate analysis, treatment with nevirapine or raltegravir, longer onset of symptoms since initiation of the involved drug and the presence of lymphadenopathy, eosinophilia, liver damage, and longer time for clinical and biological resolution after antiretroviral suspension were more frequent in patients with a “definitive-probable” DRESS case. On the other hand, treatment with abacavir and gastrointestinal symptoms were more prevalent among “possible-no” DRESS cases. None of the variables that were significant in the univariate analysis remained independently associated with a “definitive-probable” DRESS case in the multivariate analysis.
DISCUSSION
The actual guidelines for the use of antiretrovirals in naive HIV-infected adults include the use of two nucleoside reverse transcriptase inhibitors (NRTI) in combination with a third active agent from one of three drug classes: An integrase strand transfer inhibitor (INSTI), a non-NRTI (NNRTI) or a protease inhibitor (PI) with a pharmacokinetic enhancer.[48] In this review, we have analyzed 35 published reports of DHR associated to antiretrovirals in HIV patients with special focus on those catalogued as “definitive-probable” DRESS cases. Included reports involved 5 antiretrovirals: 2 NRTI (abacavir and tenofovir), 2 NNRTI (efavirenz and nevirapine) and 1 INSTI (raltegravir). No cases related to PI or to the newest antiretrovirals were found.
No age or gender differences were seen between “definitive-probable” and “possible-no” DRESS cases, as previously reported.[9] Caucasian ethnicity was more frequent among “possible-no” DRESS cases which were mostly related to abacavir. An association between HLA-B*57:01 and abacavir DHR has been described.[6] Abacavir induces loading of self-peptides into HLA-B*57:01, generating an array of neo-antigen peptides that drive polyclonal T-cell autoimmune responses and multiorgan systemic toxicity.[49] Routine HLA-B*57:01 screening and avoidance of abacavir in patients carrying the allele has reduced the incidence of DHR.[50] In this study, only one patient receiving abacavir carried the HLA-B*57:01 allele indicating that a negative test does not exclude the possibility of a DHR induced by the drug.[23] An association between nevirapine hypersensitivity in HIV patients and the HLA-DRB*101 allele has been described, which seems to be immune mediated and induced by a reactive metabolite.[51,52] Although none of the reviewed patients with a nevirapine-induced DRESS case had this allele, one had a different allele in the same genetic locus.[37] Interestingly, 5 (83%) of the raltegravir DRESS related cases were of African origin.[41,42,43,44,46] An association between raltegravir-induced DRESS cases and the HLA-B*53:01 allele in HIV patients of African origin has been recently described.[46] A pathogenic mechanism analogous to that causing abacavir DHR in B*57:01 positive HIV patients has been implicated in raltegravir-induced DRESS cases in HLA-B*53:01 positive patients.[46]
Regarding the CD4 count, DHR caused by nevirapine are more likely to occur at higher CD4 counts, for instance in healthy patients receiving the drug for postexposure prophylaxis.[53,54] In this study, although the mean CD4 count was higher in the group of patients receiving nevirapine compared to those taking abacavir, differences were not significant. A late-onset of symptoms after drug initiation is one of the main traits of DRESS syndrome.[5] Patients with a “definitive-probable” DRESS syndrome developed clinical symptoms later after antiretroviral initiation compared to those with a “possible-no” DRESS case, in consonance to what has been published.[9] Importantly, life-threating symptoms can appear in a matter of hours in patients who are re-exposed to abacavir after experiencing a previous DHR to the same drug.[24]
Fever and skin rash were the most common clinical manifestations in both groups. Mild to moderate delayed rash is the most frequent DHR induced by antiretroviral drugs in HIV patients. When is not accompanied by fever or other systemic signs or internal organ involvement it can be managed without drug withdrawal.[3] The cutaneous biopsy had a high diagnostic predictive value showing abnormalities compatible with a DRESS syndrome in all of the cases in which it was performed.[25,31,38,39,43] Mucosal involvement was scarcely reported, particularly in “definitive-probable” DRESS cases. Mucosal lesions in DRESS syndrome are usually milder and less hemorrhagic than in Steven-Johnson syndrome.[10] Lymphadenopathy and eosinophilia were more frequently described in “definitive-probable” DRESS cases. Interestingly, eosinophilia was not present in four patients with a “definitive-probable” DRESS case.[16,32,39,42] Therefore, the absence of eosinophilia does not rule out a DRESS syndrome.
Visceral damage usually determines the severity of DRESS syndrome. The liver was the organ most frequently affected in patients with a “definitive-probable” DRESS case, as shown previously.[9,10] A liver biopsy evidenced atypical lymphocytes and hepatocyte mitotic figures suggesting viral hepatitis in one patient.[38] A different pattern of organ dysfunction was described in “possible-no” DRESS cases with a higher frequency of gastrointestinal symptoms. This was due to the higher incidence of these symptoms in abacavir-induced DHR, which was the predominant antiretroviral in “possible-no” DRESS cases.[3]
The DRESS syndrome is difficult to diagnose as symptoms can mimic other diseases and can appear long after initial drug exposure. The diagnosis is mainly clinical and requires a high suspicion index. Identification of the culprit medication can be particularly challenging in HIV patients since many take multiple drugs. As supported by our review, the RegiSCAR score is a highly valuable diagnostic tool for the diagnosis of DRESS syndrome due to antiretrovirals in HIV patients. A slightly modified diagnostic criterion for DRESS syndrome has been proposed, which include data on HHV-6 reactivation.[55] There is an increasing evidence of the association between reactivation of HHV-6 and other herpes virus and DRESS syndrome. It has been demonstrated that tissue damage is caused by activated cytotoxic CD8+ T lymphocytes directed against viral antigens derived from the herpes virus.[7] It has been suggested that herpes virus reactivation could be induced by the capacity of certain drugs to interfere virus latency control. A minority of the evaluated patients were tested for the HHV-6 or for other herpes virus. Only one patient receiving efavirenz with a probable DRESS case had a positive PCR for HHV-6.[25] Although previous DRESS cases in HIV have shown evidence of herpes virus reactivation to our knowledge, the latter is the only one clearly associated with HAART.[56]
Management of DRESS syndrome requires immediate discontinuation of the offending drug and in severe cases hospital admission. The French Society of Dermatology has published therapeutic recommendations for DRESS syndrome including the use of corticosteroids in the case of relevant organ dysfunction.[57] In our review, patients with a “definitive-probable” DRESS syndrome more frequently required corticosteroid therapy while drug withdrawal was sufficient to control symptoms in most “possible-no” DRESS cases, despite having organ involvement. The use of intravenous immunoglobulins has shown to facilitate viral clearance and has been recommended in the presence of life-threating signs, always in combination with corticosteroids.[57,58] Two of the reviewed nevirapine-induced DRESS syndrome cases with persistent symptoms despite corticosteroids treatment responded to immunoglobulin therapy.[37,38] Treatment with antiretrovirals in combination with corticosteroids should be considered in severe cases with evidence of viral reactivation.[56,57]
Patients with a “definitive-probable” DRESS case required longer periods of time to achieve clinical and biological resolution after drug suspension. In a recent study variables associated with a prolonged evolution in patients with DRESS syndromes were a non-European ethnicity, severe hepatic cytolysis, and higher lymphocyte baseline count.[59] In addition, it has been suggested that patients with DRESS syndrome have an increased risk of developing autoimmune disorders.[60] Variables associated with a longer recovery or the incidence of autoimmune disorders could not be evaluated due to gaps in the provided information and the lack of follow-up data. Despite being recognized as a life threating condition, none of the reviewed patients died.
This study has important limitations mainly the small sample size and the fact that we have reviewed published case reports with a potential risk of publication bias. Second, the level of detail provided in each case report was not homogeneous and therefore subjected to a certain degree of data interpretation. Despite the limitations, we believe this review provides useful information.
CONCLUSION
In conclusion, the frequency of DRESS syndrome in HIV patients is declining with the use of the new antiretrovirals. A DRESS syndrome has to be suspected in HIV patients with lymphadenopathy, eosinophilia, and liver involvement that develop weeks after the initiation of nevirapine or raltegravir. The RegiSCAR score is a helpful diagnostic tool allowing prompt recognition of the condition. Withdrawal of the culprit drug and corticosteroids are the main therapeutic measures in patients with a suspected DRESS syndrome.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Crum NF, Riffenburgh RH, Wegner S, Agan BK, Tasker SA, Spooner KM, et al. Comparisons of causes of death and mortality rates among HIV-infected persons: Analysis of the pre-, early, and late HAART (highly active antiretroviral therapy) eras. J Acquir Immune Defic Syndr. 2006;41:194–200. doi: 10.1097/01.qai.0000179459.31562.16. [DOI] [PubMed] [Google Scholar]
- 2.Phillips E, Mallal S. Drug hypersensitivity in HIV. Curr Opin Allergy Clin Immunol. 2007;7:324–30. doi: 10.1097/ACI.0b013e32825ea68a. [DOI] [PubMed] [Google Scholar]
- 3.Borrás-Blasco J, Navarro-Ruiz A, Borrás C, Casterá E. Adverse cutaneous reactions associated with the newest antiretroviral drugs in patients with human immunodeficiency virus infection. J Antimicrob Chemother. 2008;62:879–88. doi: 10.1093/jac/dkn292. [DOI] [PubMed] [Google Scholar]
- 4.Bocquet H, Bagot M, Roujeau JC. Drug-induced pseudolymphoma and drug hypersensitivity syndrome (Drug Rash with Eosinophilia and Systemic Symptoms: DRESS) Semin Cutan Med Surg. 1996;15:250–7. doi: 10.1016/s1085-5629(96)80038-1. [DOI] [PubMed] [Google Scholar]
- 5.Choudhary S, McLeod M, Torchia D, Romanelli P. Drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome. J Clin Aesthet Dermatol. 2013;6:31–7. [PMC free article] [PubMed] [Google Scholar]
- 6.Mallal S, Nolan D, Witt C, Masel G, Martin AM, Moore C, et al. Association between presence of HLA-B*5701, HLA-DR7, and HLA-DQ3 and hypersensitivity to HIV-1 reverse-transcriptase inhibitor abacavir. Lancet. 2002;359:727–32. doi: 10.1016/s0140-6736(02)07873-x. [DOI] [PubMed] [Google Scholar]
- 7.Picard D, Janela B, Descamps V, D'Incan M, Courville P, Jacquot S, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): A multiorgan antiviral T cell response. Sci Transl Med. 2010;2:46ra62. doi: 10.1126/scitranslmed.3001116. [DOI] [PubMed] [Google Scholar]
- 8.Fiszenson-Albala F, Auzerie V, Mahe E, Farinotti R, Durand-Stocco C, Crickx B, et al. A 6-month prospective survey of cutaneous drug reactions in a hospital setting. Br J Dermatol. 2003;149:1018–22. doi: 10.1111/j.1365-2133.2003.05584.x. [DOI] [PubMed] [Google Scholar]
- 9.Cacoub P, Musette P, Descamps V, Meyer O, Speirs C, Finzi L, et al. The DRESS syndrome: A literature review. Am J Med. 2011;124:588–97. doi: 10.1016/j.amjmed.2011.01.017. [DOI] [PubMed] [Google Scholar]
- 10.Kardaun SH, Sekula P, Valeyrie-Allanore L, Liss Y, Chu CY, Creamer D, et al. Drug reaction with eosinophilia and systemic symptoms (DRESS): An original multisystem adverse drug reaction. Results from the prospective RegiSCAR study. Br J Dermatol. 2013;169:1071–80. doi: 10.1111/bjd.12501. [DOI] [PubMed] [Google Scholar]
- 11.Chen YC, Chiu HC, Chu CY. Drug reaction with eosinophilia and systemic symptoms: A retrospective study of 60 cases. Arch Dermatol. 2010;146:1373–9. doi: 10.1001/archdermatol.2010.198. [DOI] [PubMed] [Google Scholar]
- 12.Kardaun SH, Sidoroff A, Valeyrie-Allanore L, Halevy S, Davidovici BB, Mockenhaupt M, et al. Variability in the clinical pattern of cutaneous side-effects of drugs with systemic symptoms: Does a DRESS syndrome really exist? Br J Dermatol. 2007;156:609–11. doi: 10.1111/j.1365-2133.2006.07704.x. [DOI] [PubMed] [Google Scholar]
- 13.Lehloenya RJ, Dlamini S, Muloiwa R, Kakande B, Ngwanya MR, Todd G, et al. Therapeutic trial of rifabutin after rifampicin-associated DRESS syndrome in tuberculosis-human immunodeficiency virus coinfected patients. Open Forum Infect Dis. 2016;3:ofw130. doi: 10.1093/ofid/ofw130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P. Acute Dialysis Quality Initiative workgroup. Acute renal failure-definition, outcome measures, animal models, fluid therapy and information technology needs: The Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8:R204–12. doi: 10.1186/cc2872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sankatsing SU, Prins JM. Agranulocytosis and fever seven weeks after starting abacavir. AIDS. 2001;15:2464–5. doi: 10.1097/00002030-200112070-00021. [DOI] [PubMed] [Google Scholar]
- 16.Dargère S, Verdon R, Bouhier K, Bazin C. Disseminated intravascular coagulation as a manifestation of abacavir hypersensitivity reaction. AIDS. 2002;16:1696–7. doi: 10.1097/00002030-200208160-00022. [DOI] [PubMed] [Google Scholar]
- 17.Herring SJ, Krieger AC. Acute respiratory manifestations of the abacavir hypersensitivity. AIDS. 2006;20:301–2. doi: 10.1097/01.aids.0000199833.87606.95. [DOI] [PubMed] [Google Scholar]
- 18.Gervasoni C, Vigano O, Grinelli E, Ortu M, Galli M, Rusconi S. Abacavir hypersensitivity reaction after switching from the twice-daily to the once-daily formulation. AIDS Patient Care STDS. 2007;21:1–3. doi: 10.1089/apc.2006.0056. [DOI] [PubMed] [Google Scholar]
- 19.Fox J, Newton P, Daly R, Blume A, Marett B, Greene L, et al. An unusual abacavir reaction. AIDS. 2008;22:1520–2. doi: 10.1097/QAD.0b013e328305bd9e. [DOI] [PubMed] [Google Scholar]
- 20.Bonta PI, Vermeulen JN, Speelman P, Prins JM. Severe abacavir hypersensitivity reaction in a patient tested HLA-B*5701 negative. AIDS. 2008;31(22):1522–3. doi: 10.1097/QAD.0b013e3283065ba1. [DOI] [PubMed] [Google Scholar]
- 21.Calza L, Rosseti N, Biagetti C, Pocaterra D, Colangeli V, Manfredi R. Abacavir-induced reaction with fever and severe skin rash in a patient tested human leukocyte antigen-B*5701 negative. Int J STD AIDS. 2009;20:276–7. doi: 10.1258/ijsa.2008.008318. [DOI] [PubMed] [Google Scholar]
- 22.Janardhanan M, Amberkar VM, Vidyasagar S, Kumari KM, Holla SN. Hypersensitivity reaction associated with abacavir therapy in an Indian HIV patient – A case report. J Clin Diagn Res. 2014;8:HD01–2. doi: 10.7860/JCDR/2014/10063.4820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Haas C, Ziccardi MR, Borgman J. Abacavir-induced fulminant hepatic failure in a HIV/HCV co-infected patient. BMJ Case Rep 2015. 2015:pii: Bcr2015212566. doi: 10.1136/bcr-2015-212566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Shapiro M, Ward KM, Stern JJ. A near-fatal hypersensitivity reaction to abacavir: Case report and literature review. AIDS Read. 2001;11:222–6. [PubMed] [Google Scholar]
- 25.Bossi P, Colin D, Bricaire F, Caumes E. Hypersensitivity syndrome associated with efavirenz therapy. Clin Infect Dis. 2000;30:227–8. doi: 10.1086/313629. [DOI] [PubMed] [Google Scholar]
- 26.Behrens GM, Stoll M, Schmidt RE. Pulmonary hypersensitivity reaction induced by efavirenz. Lancet. 2001;357:1503–4. doi: 10.1016/S0140-6736(00)04663-8. [DOI] [PubMed] [Google Scholar]
- 27.Foti JL, Piatt JP. Hypersensitivity to efavirenz treated with corticosteroids in a 6-year-old child. AIDS Patient Care STDS. 2003;17:1–3. doi: 10.1089/108729103321042854. [DOI] [PubMed] [Google Scholar]
- 28.Angel-Moreno-Maroto A, Suárez-Castellano L, Hernández-Cabrera M, Pérez-Arellano JL. Severe efavirenz-induced hypersensitivity syndrome (not-DRESS) with acute renal failure. J Infect. 2006;52:e39–40. doi: 10.1016/j.jinf.2005.05.020. [DOI] [PubMed] [Google Scholar]
- 29.Leung JM, O'Brien JG, Wong HK, Winslow DL. Efavirenz-induced hypersensitivity reaction manifesting in rash and hepatitis in a Latino male. Ann Pharmacother. 2008;42:425–9. doi: 10.1345/aph.1K574. [DOI] [PubMed] [Google Scholar]
- 30.Curry E, Thomas M, Yehia M. Renal impairment and hypersensitivity reaction due to efavirenz. Nephrology (Carlton) 2008;13:541. doi: 10.1111/j.1440-1797.2008.00968.x. [DOI] [PubMed] [Google Scholar]
- 31.Bourezane Y, Salard D, Hoen B, Vandel S, Drobacheff C, Laurent R. DRESS (drug rash with eosinophilia and systemic symptoms) syndrome associated with nevirapine therapy. Clin Infect Dis. 1998;27:1321–2. [PubMed] [Google Scholar]
- 32.Sibaud V, Billon C, Barcat D, Bernard N, Geniaux M. Hypersensitivity syndrome to nevirapine. Therapie. 2000;55:320–2. [PubMed] [Google Scholar]
- 33.Sissoko D, Ajana F, de la Tribonnière X, Baclet V, Mouton Y. Cutaneous, hepatic and hematologic manifestations due to nevirapine: DRESS syndrome? Presse Med. 2000;29:1041–2. [PubMed] [Google Scholar]
- 34.Claudio GA, Martin AF, de Dios Perrino S, Velasco AA. DRESS syndrome associated with nevirapine therapy. Arch Intern Med. 2001;161:2501–2. doi: 10.1001/archinte.161.20.2501. [DOI] [PubMed] [Google Scholar]
- 35.Lanzafame M, Rovere P, De Checchi G, Trevenzoli M, Turazzini M, Parrinello A. Hypersensitivity syndrome (DRESS) and meningoencephalitis associated with nevirapine therapy. Scand J Infect Dis. 2001;33:475–6. doi: 10.1080/00365540152029990. [DOI] [PubMed] [Google Scholar]
- 36.Knudtson E, Para M, Boswell H, Fan-Havard P. Drug rash with eosinophilia and systemic symptoms syndrome and renal toxicity with a nevirapine-containing regimen in a pregnant patient with human immunodeficiency virus. Obstet Gynecol. 2003;101(5 Pt 2):1094–7. doi: 10.1016/s0029-7844(02)02620-0. [DOI] [PubMed] [Google Scholar]
- 37.Santos RP, Ramilo O, Barton T. Nevirapine-associated rash with eosinophilia and systemic symptoms in a child with human immunodeficiency virus infection. Pediatr Infect Dis J. 2007;26:1053–6. doi: 10.1097/INF.0b013e318125655d. [DOI] [PubMed] [Google Scholar]
- 38.Fields KS, Petersen MJ, Chiao E, Tristani-Firouzi P. Case reports: Treatment of nevirapine-associated dress syndrome with intravenous immune globulin (IVIG) J Drugs Dermatol. 2005;4:510–3. [PubMed] [Google Scholar]
- 39.Pott Junior H, Gosuen GC, Gales AC. DRESS syndrome due to nevirapine treated with methylprednisolone. Case Rep Med. 2013;2013:269501. doi: 10.1155/2013/269501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gill S, Sagar A, Shankar S, Nair V. Nevirapine-induced rash with eosinophilia and systemic symptoms (DRESS) Indian J Pharmacol. 2013;45:401–2. doi: 10.4103/0253-7613.114993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang KS, Modi GM, Hsu S. DRESS syndrome associated with raltegravir. Dermatol Online J. 2011;17:14. [PubMed] [Google Scholar]
- 42.Loulergue P, Mir O. Raltegravir-induced DRESS syndrome. Scand J Infect Dis. 2012;44:802–3. doi: 10.3109/00365548.2012.689850. [DOI] [PubMed] [Google Scholar]
- 43.Perry ME, Almaani N, Desai N, Larbalestier N, Fox J, Chilton D. Raltegravir-induced Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) syndrome-implications for clinical practice and patient safety. Int J STD AIDS. 2013;24:639–42. doi: 10.1177/0956462413481528. [DOI] [PubMed] [Google Scholar]
- 44.Ripamonti D, Benatti SV, Di Filippo E, Ravasio V, Rizzi M. Drug reaction with eosinophilia and systemic symptoms associated with raltegravir use: Case report and review of the literature. AIDS. 2014;28:1077–9. doi: 10.1097/QAD.0000000000000204. [DOI] [PubMed] [Google Scholar]
- 45.Yee BE, Nguyen NH, Lee D. Extensive pulmonary involvement with raltegravir-induced DRESS syndrome in a postpartum woman with HIV. BMJ Case Rep 2014. 2014:pii: Bcr2013201545. doi: 10.1136/bcr-2013-201545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Thomas M, Hopkins C, Duffy E, Lee D, Loulergue P, Ripamonti D, et al. Association of the HLA-B*53:01 allele with drug reaction With eosinophilia and systemic symptoms (DRESS) syndrome during treatment of HIV infection with raltegravir. Clin Infect Dis. 2017;64:1198–203. doi: 10.1093/cid/cix096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bayonne Kombo ES, Gathse A. Tenofovir hypersensitivity to tenofovir (DRESS) syndrome in a female patient infected by HIV. Med Mal Infect. 2013;43:134–5. doi: 10.1016/j.medmal.2013.01.008. [DOI] [PubMed] [Google Scholar]
- 48.Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents. Department of Health and Human Services. [Last accessed on 2016 Jul 20]. Available from: http://www.aidsinfo.nih.gov/contentfiles/lvguidelines/AdultandAdolescentGL.pdf .
- 49.Norcross MA, Luo S, Lu L, Boyne MT, Gomarteli M, Rennels AD, et al. Abacavir induces loading of novel self-peptides into HLA-B*57: 01: An autoimmune model for HLA-associated drug hypersensitivity. AIDS. 2012;26:F21–9. doi: 10.1097/QAD.0b013e328355fe8f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rauch A, Nolan D, Martin A, McKinnon E, Almeida C, Mallal S. Prospective genetic screening decreases the incidence of abacavir hypersensitivity reactions in the Western Australian HIV cohort study. Clin Infect Dis. 2006;43:99–102. doi: 10.1086/504874. [DOI] [PubMed] [Google Scholar]
- 51.Vitezica ZG, Milpied B, Lonjou C, Borot N, Ledger TN, Lefebvre A, et al. HLA-DRB1*01 associated with cutaneous hypersensitivity induced by nevirapine and efavirenz. AIDS. 2008;22:540–1. doi: 10.1097/QAD.0b013e3282f37812. [DOI] [PubMed] [Google Scholar]
- 52.Popovic M, Caswell JL, Mannargudi B, Shenton JM, Uetrecht JP. Study of the sequence of events involved in nevirapine-induced skin rash in Brown Norway rats. Chem Res Toxicol. 2006;19:1205–14. doi: 10.1021/tx0601152. [DOI] [PubMed] [Google Scholar]
- 53.Stern JO, Robinson PA, Love J, Lanes S, Imperiale MS, Mayers DL. A comprehensive hepatic safety analysis of nevirapine in different populations of HIV infected patients. J Acquir Immune Defic Syndr. 2003;34(Suppl 1):S21–33. doi: 10.1097/00126334-200309011-00005. [DOI] [PubMed] [Google Scholar]
- 54.Breining A, Guihot A, Warot D, Canestri A, Peytavin G, Bricaire F, et al. Persistent high nevirapine blood level with DRESS syndrome 12 days after interruption of antiretroviral therapy. BMJ Case Rep 2009. 2009:pii: Bcr0720080475. doi: 10.1136/bcr.07.2008.0475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Shiohara T, Iijima M, Ikezawa Z, Hashimoto K. The diagnosis of a DRESS syndrome has been sufficiently established on the basis of typical clinical features and viral reactivations. Br J Dermatol. 2007;156:1083–4. doi: 10.1111/j.1365-2133.2007.07807.x. [DOI] [PubMed] [Google Scholar]
- 56.Almudimeegh A, Rioux C, Ferrand H, Crickx B, Yazdanpanah Y, Descamps V. Drug reaction with eosinophilia and systemic symptoms, or virus reactivation with eosinophilia and systemic symptoms as a manifestation of immune reconstitution inflammatory syndrome in a patient with HIV? Br J Dermatol. 2014;171:895–8. doi: 10.1111/bjd.13079. [DOI] [PubMed] [Google Scholar]
- 57.Descamps V, Ben Saïd B, Sassolas B, Truchetet F, Avenel-Audran M, Girardin P, et al. Management of drug reaction with eosinophilia and systemic symptoms (DRESS) Ann Dermatol Venereol. 2010;137:703–8. doi: 10.1016/j.annder.2010.04.024. [DOI] [PubMed] [Google Scholar]
- 58.Galvao VR, Aun MV, Kalil J, Castells M, Giavina-Bianchi P. Clinical and laboratory improvement after intravenous immunoglobulin in drug reaction with eosinophilia and systemic symptoms. J Allergy Immunol Pract. 2014;2:107–10. doi: 10.1016/j.jaip.2013.11.008. [DOI] [PubMed] [Google Scholar]
- 59.Tetart F, Picard D, Janela B, Joly P, Musette P. Prolonged evolution of drug reaction with eosinophilia and systemic symptoms: Clinical, virologic, and biological features. JAMA Dermatol. 2014;150:206–7. doi: 10.1001/jamadermatol.2013.6698. [DOI] [PubMed] [Google Scholar]
- 60.Chen YC, Chang CY, Cho YT, Chiu HC, Chu CY. Long-term sequelae of drug reaction with eosinophilia and systemic symptoms: A retrospective cohort study from Taiwan. J Am Acad Dermatol. 2013;68:459–65. doi: 10.1016/j.jaad.2012.08.009. [DOI] [PubMed] [Google Scholar]


