Abstract
Inflammatory bowel disease (IBD) is a chronic incurable condition characterized by relapsing inflammation of the gut. Intestinal smooth muscle cells (SMCs) are affected structurally and functionally during IBD due to excessive production of different inflammatory mediators. Metformin is a widely used antidiabetic agent known to exert several anti-inflammatory effects in different tissues independently from its hypoglycemic effect. The aim of the present study was to investigate the effect of metformin on expression and secretion of different cytokines and chemokines from mouse colon SMCs (CSMCs) following induction of inflammation with lipopolysaccharide (LPS) in vitro. CSMCs from male BALB/c mice were isolated and cultured in Dulbecco's modified Eagle's medium and treated with LPS (1 µg/ml) and 0, 5, 10 or 20 mM metformin for 24 h. Expression and secretion of tumor necrosis factor-α (TNF-α), interleukin-1α (IL-1α), macrophage colony stimulating factor (M-CSF), T cell activation gene-3 (TCA-3) and stromal cell-derived factor-1 (SDF-1) was evaluated by ELISA. LPS-treated CSMCs demonstrated significantly increased expression of TNF-α, IL-1α, M-CSF, TCA-3 and SDF-1 when compared with the control group (P<0.05). Co-treatment with metformin (5 and 10 mM) significantly reduced their expression by ~20-40% when compared with LPS treatment alone (P<0.05). Furthermore, secretion of TNF-α, IL-1α, M-CSF and TCA-3 into the conditioned media was significantly decreased by metformin (5 and 10 mM; P<0.05). In addition, metformin decreased levels of LPS-induced nuclear factor-κB phosphorylation. These data suggest that metformin may provide beneficial anti-inflammatory effects on CSMCs and it may be utilized as an adjunct therapy for patients suffering from IBD.
Keywords: metformin, inflammation, inflammatory bowel disease, smooth muscle
Introduction
Mounting evidence suggests that intestinal smooth muscle cells (SMCs) may be involved in different inflammatory diseases that affect the bowel, leading to altered morphology, contractility and augmented production of various inflammatory cytokines (1,2). Studies conducted using different animal models of gastrointestinal diseases have demonstrated that growth and contractile properties of SMCs are substantially altered during mucosal inflammation in the gastrointestinal tract due to increased expression of different cytokines (3,4). Patients who suffer from IBD experience symptoms associated with abnormal intestinal motility, resulting from abnormal proliferation and contractility of intestinal SMCs (3). Numerous studies have demonstrated that intestinal SMCs may produce different inflammatory mediators, including interleukin (IL)-6 and tumor necrosis factor-α (TNF-α) during various pathological conditions (2,5). Shi and Sarna (6) demonstrated that TNF-α binds to cognate receptors expressed on human CSMCs, resulting in activation of nuclear factor (NF)-κB and induction of expression of different cytokines and chemokines, including monocyte chemotactic protein (MCP)-1, IL-8 and intercellular adhesion molecule-1. Furthermore, exposing human colonic (C)SMCs to different inflammatory stimuli led to enhanced expression of IL-1α, IL-6, IL-8, cyclooxygenase-2 and regulated on activation, normal T cell expressed and secreted (RANTES) (7).
Ulcerative colitis and Crohn's disease are two distinct forms of IBD, affecting the colon and small intestine, respectively, and are characterized by chronic and relapsing intestinal inflammation (8). IBD may result from dysregulation of the mucosal immune response triggered by a combination of genetic, environmental and immunological factors, resulting in mucosal and submucosal inflammation (8). Furthermore, IBD is usually associated with other co-morbid diseases, including rheumatoid arthritis, multiple sclerosis, systemic lupus, psoriasis, hypothyroidism and diabetes mellitus (9). Diabetes mellitus is one of the major conditions associated with IBD, resulting in significant clinical and therapeutic consequences (10). The incidence and prevalence of IBD and associated comorbidities are increasing worldwide, resulting in significant healthcare costs and impaired quality of life for patients (10,11). Despite advances in understanding IBD pathophysiology and its biological therapies, IBD remains a non-curable condition highlighting the need to develop novel treatment approaches (11).
Metformin is a biguanide derivative used in type 2 diabetes treatments as a first-line therapy and is one of the major prescribed oral hypoglycemic drugs (12). Metformin increases peripheral uptake of glucose, decreases hepatic glucose production and increases insulin sensitivity in liver and skeletal muscle (12). Metformin may mediate its hypoglycemic effect through several molecular mechanisms, including activation of the adenosine 5′-monophosphate kinase (AMPK) signaling pathway, which regulates different physiological processes, including cellular growth and proliferation, mitochondrial function and biogenesis, insulin sensitivity, inflammation, oxidative stress and autophagy (12).
Notably, clinical and experimental research has demonstrated an array of potential benefits of metformin beyond its hypoglycemic effect in an AMPK-dependent and independent manner (13). Metformin may exert anti-inflammatory, anticancer, cardioprotective and antiatherosclerotic effects, and it may decrease macrovascular complications of diabetes (14–17). It has been demonstrated that metformin may inhibit NF-κB activation and inflammatory marker expression through the AMPK signaling pathway by reducing signal transduction and activator of transcription (STAT)3 activity and tumor suppressor phosphatidylinositol 3,4,5-trisphosphate 3-phosphatase and dual-specificity protein phosphatase through inhibition of NF-κB and downstream inflammatory genes in multiple cell types (15).
In an experimental IBD model, metformin reduced disease activity index scores, decreased colonic histopathological score, reduced expression of inflammatory mediators and preserved colon length (18). Additionally, treatment with metformin upregulated phosphorylated (p)-AMPK levels and simultaneously inhibited expression of IL-17, p-STAT3 and p-mammalian target of rapamycin (18). Furthermore, metformin decreased expression of inflammatory cytokines in a dose-dependent manner in inflamed human intestinal epithelial HT-29 cells (18). Metformin also decreased phosphorylation and activation of pro-inflammatory proteins, including protein kinase B, p38, extracellular signal regulated kinase and protein kinase C in vascular endothelial cells under hyperglycemic conditions (19).
Mouse CSMCs have previously been identified as being capable of expressing multiple cytokines and chemokines including TNF-α, IL-1α, macrophage colony stimulating factor (M-CSF), T cell activation gene-3 (TCA-3) and stromal cell-derived factor-1 (SDF-1), when exposed to inflammatory stimuli including lipopolysaccharides (LPSs) (20). Although multiple studies have demonstrated that metformin suppresses NF-κB activation and cytokine production in various cell types, little information is available on the effect of metformin on CSMC expression and secretion of pro-inflammatory cytokines and chemokines (14,15). Therefore, the current study hypothesized that metformin regulates NF-κB signaling in CSMCs, by influencing cytokine and chemokine expression, and may provide a novel adjunct therapy to treat IBD particularly in patients with diabetes.
Materials and methods
Solutions, drugs and chemicals
A smooth muscle buffer (SMB) was prepared in-house and contained the following: 120 mM NaCl, 4 mM KCl, 2.6 mM KH2PO4, 2 mM CaCl2, 25 mM HEPES (N-2-hydroxyethylpiperazine-N'-2-ethanesulfonic acid), 14 mM glucose and 2.1% essential amino acid mixture; the pH was adjusted to 7.4. Tissue digestion solution contained 0.5 mg collagenase and 0.33 mg soybean trypsin inhibitor per ml of SMB. All of these chemicals were purchased from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany). Cell culture media used during incubation processes was prepared by adding 10% fetal bovine serum (FBS; GE Healthcare Life Sciences, Little Chalfont, UK), 100 U/ml penicillin, 100 µg/ml streptomycin and 2.5 µg/ml amphotericin B to Dulbecco's modified Eagle medium (DMEM) with L-glutamine (Capricorn Scientific GmbH, Ebsdorfergrund, Germany). Remaining reagents were purchased from EuroClone S.p.A. (Pero, Italy). LPS was purchased from Sigma-Aldrich (Merck KGaA). Metformin was purchased from Merck KGaA. Specific ELISA kits for mouse TNF-α (cat. no. RAB0477), M-CSF (cat. no. RAB0099), IL-1α (cat. no. RAB0271), TCA-3 (cat. no. RAB0041) and SDF-1 (cat. no. RAB0125) were purchased from Sigma-Aldrich (Merck KGaA). Nuclear protein extraction kit (cat. no. ab113474) and an NF-κB p65 (pS536) ELISA kit (cat. no. ab176663) were purchased from Abcam (Cambridge, UK). A 500-µm Nitex mesh was purchased from Sigma-Aldrich (Merck KGaA).
Animals
A total of 20 young mature male BALB/c mice (~12 weeks of age, 26.5–30 g) were provided by the animal house of Jordan University of Science and Technology (Irbid, Jordan). Mice were housed in the animal facility at the Jordan University of Science and Technology under 12-h light/dark cycles in polyethylene cages at −22°C and 50% humidity. Mice were fed standard chow rodent diet and water available ad libitum. Mice (age, ~14 weeks) were euthanized by inhalation of CO2 and the colon was excised. The colon was cut into pieces (2–3 cm in length) and placed in cold SMB. All procedures were approved and performed according to the guidelines of the Animal Care and Use Committee at Jordan University of Science and Technology.
Preparation of dispersed CSMCs
CSMCs were isolated from the circular and longitudinal smooth muscle layers of the colons of BALB/c mice (sacrificed at 14 weeks of age) by enzymatic digestion of muscle strips, followed by filtration, and centrifugation as previously described (20). Mucosa was scraped off murine colon tissue with fine scissors; tissues were cut into thin slices (2 mm long; 2 mm thin) and incubated for 20 min in SMB containing 0.5 mg/ml collagenase and 0.33 mg/ml soybean trypsin inhibitor in an agitated water bath at 31°C. Tissues were continuously supplied with 100% oxygen during the isolation procedure. Partly digested tissue was washed twice with 50 ml collagenase-free SMB and muscle cells were allowed to disperse spontaneously for 10 min in collagenase-free medium. Cells were harvested by filtration through 500-µm Nitex mesh and centrifuged twice at 350 × g for 10 min at 4°C to eliminate broken cells and organelles. The process was repeated 4–5 times. Cells were counted in a hemocytometer and viability was assessed using a trypan blue exclusion assay. Cell suspensions (100 µl) were mixed with 100 µl of 0.4% trypan blue dye (Thermo Fisher Scientific, Inc., Waltham, MA, USA) and stained at room temperature (23°C) for 1 min. Cells were then immediately loaded into hemocytometer (Thermo Fisher Scientific, Inc.) and examined under an inverted microscope (Nikon Corporation, Tokyo, Japan; magnification, ×40). In this assay, trypan blue dye permeates unviable cells; while viable cells exclude this dye because they possess intact plasma membranes. Therefore blue-stained cells were counted and considered unviable cells. It was identified that >95% of cells were viable and therefore were suitable for further experimentation.
Identification and characterization of CSMCs
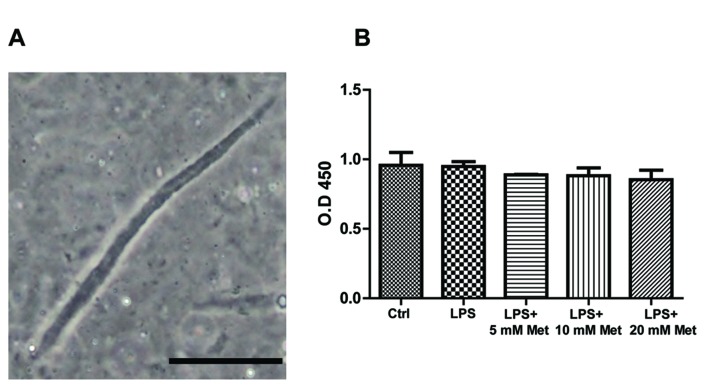
Isolated mouse CSMCs were viewed at a ×20 magnification using an inverted Nikon TMS-f microscope (Nikon Corporation). Fig. 1A presents CSMCs as spindle-shaped cells with an average length of ~140 µM.
Figure 1.
Identification and viability of mouse CSMCs. (A) A single, spindle-shaped CSMC under phase contrast microscopy; magnification, ×20. (B) Cell viability assay for cells treated with LPS (1 µg/ml) plus varying amounts of metformin (0, 5, 10 or 20 mM). Scale bar=50 µm. LPS, lipopolysaccharide; CSMC, colonic smooth muscle cell; Met, metformin; OD, optical density.
CSMC viability assay
Isolated CSMCs were cultured in 96-well plates at 1×104 cells/well (n=3 per group). Cells were serum-deprived for 24 h prior to treatment with LPS (1 µg/ml) and varying amounts of metformin (0, 5, 10 and 20 mM) at 37°C. Cell viability was assessed following 24 h using MTT assays (Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. CSMCs were incubated with MTT reagent for 4 h at 37°C. The MTT reagent was converted to an insoluble formazan. Formazan was then solubilized with a solubilizing reagent provided in the kit, and the concentration determined by optical density at 570 nm.
Quantification of protein expression by ELISA
A total of 10,000 dispersed mouse CSMCs (n=3 per group) were seeded per well in a 6-well plate containing DMEM with 10% FBS, 100 U/ml penicillin, 100 µg/ml streptomycin and 2.5 µg/ml amphotericin B. Cells were incubated in a humidified incubator with 95% air and 5% CO2 at 37°C for 24 h with LPS (1 µg/ml) and varying amounts of metformin (0, 5, 10 and 20 mM). Following the incubation period treated samples were centrifuged at 350 × g for 5 min at 4°C. Conditioned media was stored at −20°C for further analysis and cells pellets were lysed immediately. Cell lysates were prepared using BashingBeads Lysis tubes from Zymo Research Corp. (Irvine, CA, USA) and cell lysis buffer containing protease inhibitor cocktail provided the a whole cell extraction kit (Abcam; cat. no. ab113475), according to the manufacturer's protocol. A nuclear protein extraction kit (Abcam; cat. no. ab113474) was used to extract total nuclear proteins from another set of control and treated samples. Lysates were centrifuged for 10 min at 10,000 × g at 4°C and supernatants were collected for further analysis. Total protein concentration of supernatants was measured using the DC protein assay kit (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Protein concentration was adjusted to 100 µg/ml in all samples. Protein levels of specific cytokines were evaluated by ELISA assay. Specific ELISA kits for TNF-α, IL-1α, M-CSF, TCA-3, SDF-1 and nuclear NF-κB p65 (pS536) were used to measure cytokine levels in lysates and conditioned media for control and treated samples according to the manufacturer's protocols.
Statistical analysis
Statistical analyses were performed using GraphPad Prism 5.0 (GraphPad Software, Inc., La Jolla, CA, USA). One-way analysis of variance followed by Fisher's post-hoc analysis was used to examine significant differences between groups. All data are presented as mean ± standard error of the mean. Values presented are representative of three independent experiments performed in triplicate. P<0.05 was considered to indicate a statistically significant difference.
Results
CSMC viability assay
To ensure that neither LPS nor metformin affected CSMCs viability, an MTT assay was performed on the treated samples. The results indicated that cell viability was not significantly affected by LPS or metformin treatment following a 24 h period in all treatment groups, suggesting that growth of CSMCs remained unchanged during the treatment period (Fig. 1B).
Evaluation of cytokine and chemokine expression and secretion
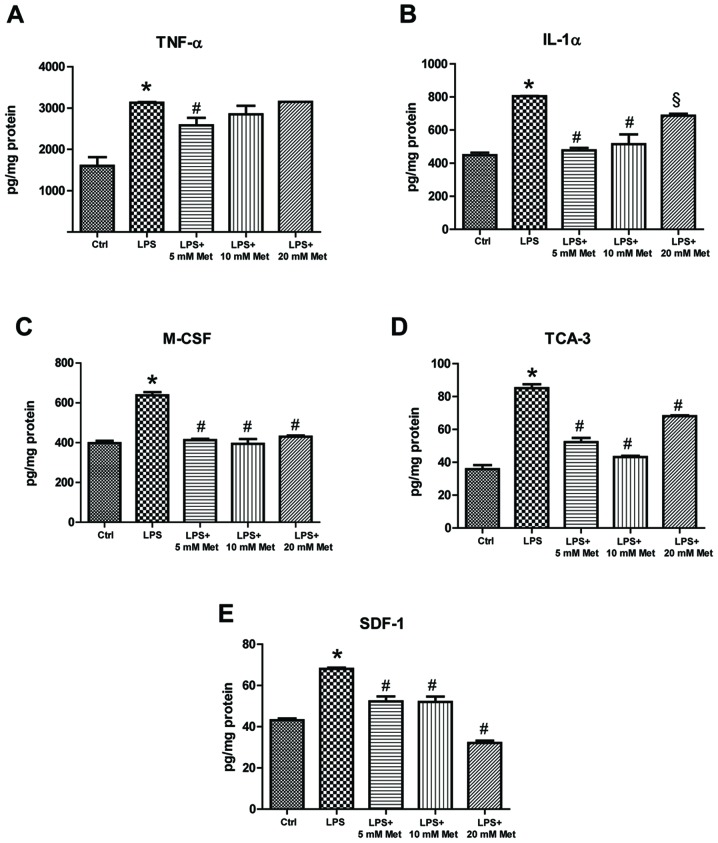
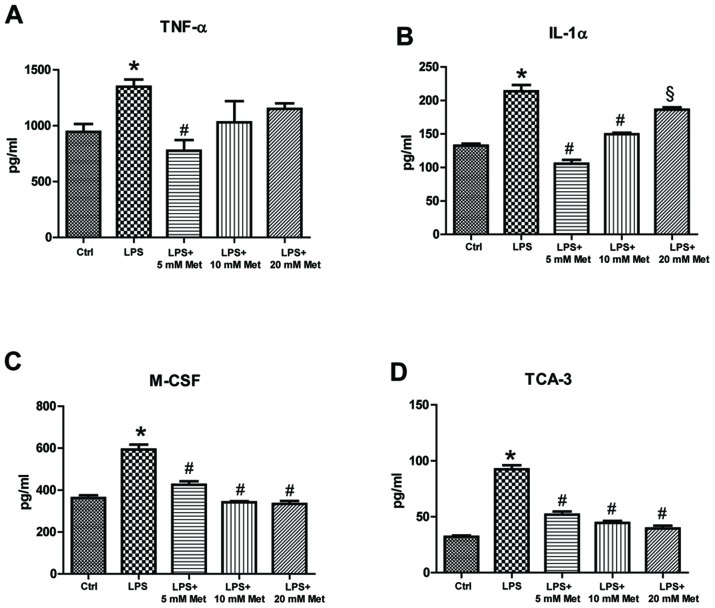
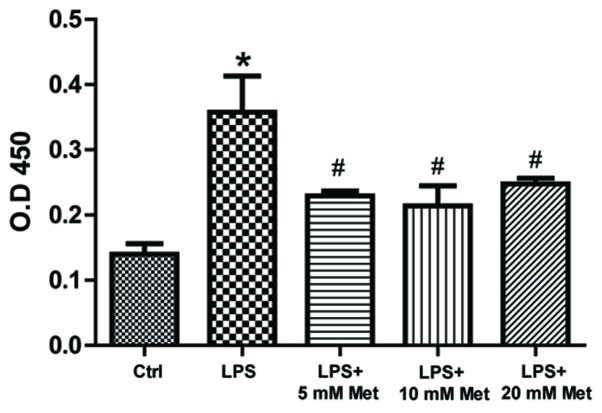
To measure cytokine and chemokine levels in the control and treated samples, specific ELISA assays were used. The data demonstrated that LPS treatment resulted in a significant increase (~1.5–2 fold) in TNF-α, IL-1α, M-CSF, TCA-3 and SDF-1 expression (P<0.05; Fig. 2). On the other hand, co-treatment with metformin (5 or 10 mM) significantly reduced expression by ~20-40% compared with LPS alone (P<0.05). Furthermore, evaluation of cytokine and chemokine secretion by CSMCs into the media was assessed by ELISA. The data demonstrated that TNF-α, IL-1α, M-CSF and TCA-3 levels were significantly elevated following LPS treatment, while co-treatment with metformin (5 and 10 mM) significantly reduced secretion into the media compared with LPS alone (P<0.05; Fig. 3). In addition, LPS treatment upregulated nuclear NF-κB p65 (pS536) protein levels, while co-treatment with metformin significantly reduced levels compared with LPS alone (P<0.05), suggesting that metformin may suppress inflammatory cytokine and chemokine expression and secretion by interfering with NF-κB signaling pathway activation (Fig. 4).
Figure 2.
Effect of metformin treatment on expression of inflammatory cytokines by mouse CSMCs, evaluated using ELISAs. Levels of (A) TNF-α, (B) IL-1α, (C) M-CSF, (D) TCA-3 and (E) SDF-1 in CSMCs treated with LPS (1 µg/ml) and varying amounts of metformin (0, 5, 10 and 20 mM). *P<0.01 vs. the Ctrl group; #P<0.01 vs. the LPS group; §P<0.05 vs. the LPS group. LPS, lipopolysaccharide; CSMC, colonic smooth muscle cell; Met, metformin; TNF-α, tumor necrosis factor-α; IL, interleukin; M-CSF, macrophage-colony stimulating factor; TCA-3, T cell activation gene-3; SDF-1, stromal cell-derived factor-1; Ctrl, control.
Figure 3.
Effect of metformin treatment on secretion of inflammatory cytokines by mouse CSMCs into the conditioned media, evaluated using ELISA. Levels of (A) TNF-α, (B) IL-1α, (C) M-CSF and (D) TCA-3 in CSMCs following treatment with LPS (1 µg/ml) and varying amounts of metformin (0, 5, 10 and 20 mM). *P<0.01 vs. the Ctrl group; #P<0.01 vs. the LPS group; §P<0.05 vs. the LPS group. LPS, lipopolysaccharide; CSMC, colonic smooth muscle cell; Met, metformin; TNF-α, tumor necrosis factor-α; IL, interleukin; M-CSF, macrophage-colony stimulating factor; TCA-3, T cell activation gene-3; Ctrl, control.
Figure 4.
Metformin reduces nuclear LPS-induced NF-κB phosphorylation. Total nuclear proteins were extracted and nuclear NF-κB p65 was assayed in the lysate in colonic smooth muscle cells following treatment with LPS (1 µg/ml) and varying amounts of metformin (0, 5, 10 and 20 mM) *P<0.01 vs. the Ctrl group; #P<0.01 vs. the LPS group. LPS, lipopolysaccharide; Met, metformin; NF-κB, nuclear factor-κB; OD, optical density; Ctrl, control.
Collectively, these results suggest that metformin may attenuate the expression and secretion of several cytokines and chemokines from mouse CSMCs in the presence of inflammatory stimulus. Fig. 5 represents an integrative hypothetical model for how metformin may exert its anti-inflammatory effect in CSMCs. It was hypothesized that metformin activates the AMPK pathway, thereby inhibiting downstream inflammatory gene expression in CSMCs.
Figure 5.
Proposed model for the mechanism by which metformin may suppress LPS-induced inflammatory gene expression in mouse colonic smooth muscle cells. Metformin may activate AMPK, which may interfere with LPS-induced NF-κB activation, phosphorylation and translocation to the nucleus, which in turn suppresses inflammatory cytokine and chemokine expression. Curved arrows indicate downstream activation and triangles indicate metformin. LPS, lipopolysaccharide; NF-κB, nuclear factor-κB; AMPK, adenosine 5′-monophsphate kinase; TLR4, Toll-like receptor 4.
Discussion
Most inflammatory conditions of the bowel result in activation and recruitment of different inflammatory cells that alter the surrounding environment, leading to activation of a complex integrated inflammatory cascade (21). These events result in major hallmarks of intestinal inflammation and loss of epithelial tight junctions (21). Furthermore, it has been reported that inflammatory conditions affecting the bowel may lead to significant functional and morphological changes in the intestinal SMCs. In the present study, it was demonstrated that metformin may exert significant anti-inflammatory effects on expression and secretion of different inflammatory mediators from mouse CSMCs under LPS-induced inflammation in vitro.
It was previously identified that mouse CSMCs are capable of expressing different cytokines and chemokines, including TNF-α, IL-1α, M-CSF, TCA-3 and SDF-1, when stimulated with LPS (20). Therefore, the anti-inflammatory effects of metformin on expression and secretion of these inflammatory mediators were investigated. The current study demonstrated that low doses of metformin (5 or 10 mM) rather than a high dose (20 mM) caused a significant reduction in expression and secretion of these inflammatory mediators in CSMCs. Additionally, metformin significantly reduced LPS-induced nuclear NF-kB p65 (S635) levels in CSMCs. These results indicated that metformin may be effective in suppressing expression and secretion of TNF-α, IL1-α, MCSF, TCA-3 and SDF-1 from CSMCs through inhibition of NF-kB p65 phosphorylation and nuclear translocation.
TNF-α is potent pro-inflammatory cytokine produced as a transmembrane protein that is released into medium through proteolytic cleavage via metalloproteinase TNF-converting enzyme (22). TNF-α is primarily secreted from activated macrophages, as well as a variety of other cell types (22). Abnormal production of TNF-α has been associated with different ailments and it has been reported that expression of both membrane-bound and soluble TNF-α by submucosal inflammatory cells is increased during active stages of IBD (22). TNF-α expression in IBD stimulates multiple pro-inflammatory actions, including initiation of systemic inflammatory response, increased angiogenesis, Paneth cell death, production of matrix metalloproteinases and damage of intestinal epithelial cells (23). Anti-TNF-α antibodies, including infliximab and adalimumab, are commonly used in the management of IBD with high success rates, demonstrating an essential role of TNF-α in the pathogenesis of IBD (24,25). In the current study, co-treatment with metformin attenuated LPS-induced expression and secretion of TNF-α, suggesting an important role of TNF-α derived from CSMCs in the development of colonic inflammation.
IL-1α is a key inflammatory mediator that serves an important role in early phases of IBD. Increased expression of IL-1α has been reported in experimental models of colitis and colon biopsies from IBD patients (26,27). It serves an important role in recruitment of neutrophils, stimulation of IL-6 production by macrophages and promotion of intestinal tumorigenesis (28). Cominelli and Pizarro (29) reported that blockage of IL-1α receptors inhibited inflammatory responses associated with immune complex-induced colitis, suggesting a central role for IL-1α in IBD pathogenesis. Expression of IL-1α is enhanced during different inflammatory conditions where it initiates a cascade of events to increase production of several inflammatory mediators. Enhanced production of both IL-1α and TNF-α together aggravates the inflammatory process by inducing a cascade of other pro-inflammatory events (23).
M-CSF is a cytokine produced by different cell types in the body, where it mediates expression of various inflammatory genes that control survival, proliferation, adhesion, migration and differentiation of macrophages (30,31). Effects of M-CSF are mediated through a tyrosine kinase receptor (M-CSFR) that is expressed primarily on mononuclear phagocytic cells and it has been demonstrated that M-CSF, M-CSFR and macrophages are increased in the gastrointestinal tract in the presence of IBD (32). It is postulated that M-CSF may serve an essential role in pathogenesis of IBD (31). Studies on animal models of experimental colitis demonstrated that blockage of M-CSF with a neutralizing antibody reduced symptoms of disease and cytokine expression by immune cells (31). The present study has demonstrated that metformin inhibited M-CSF expression and secretion by CSMCs in the presence of an inflammatory stimulus.
Chemotaxis is an important inflammatory process that serves an essential role in activation and recruitment of inflammatory cells during IBD. High levels of different chemokines, including IL-8 and RANTES, have been detected in colonic biopsies from patients with IBD during the active phase of colitis (33,34). Furthermore, mRNA levels of MCP-1 were demonstrated to increase in endothelial cells and intestinal SMCs during the active phase of the disease (35). In the present study, expression of TCA-3 and SDF-1 was increased following treatment with LPS, while co-treatment with metformin significantly reversed expression of these chemokines, suggesting that metformin may inhibit attraction and recruitment of inflammatory cells by reducing chemokine production from CSMCs.
Activated T lymphocytes are a major source of TCA-3, which plays an important role as a chemotactic factor for macrophages and leukocytes. TCA-3 is a β-chemokine that affects expression of other chemokines, including myoinhibitory-like protein-1α and MCP-1, thereby affecting leukocyte recruitment to inflammatory lesions (36). Chronically inflamed colons of IL-10 knockout mice displayed elevated levels of TCA-3 and other chemokines when compared with wild-type mice. Interestingly, remission of colitis in this animal model was associated with decreased expression of TCA-3 (37).
SDF-1 is a chemokine secreted by different inflammatory cells. SDF-1 expression is increased in intestinal epithelial cells in IBD, therefore it has been linked to IBD pathogenesis (33). In a mouse model of colitis, blockage of SDF-1 receptor reduced signs of colonic inflammation and reduced release of other pro-inflammatory mediators (38).
Despite the important role of CSMCs in IBD pathogenesis, their inflammatory and secretory functions are rarely investigated as the majority of studies focus on morphological alterations and abnormal contractility that occurs during gastrointestinal inflammation. Synthetic corticosteroids are commonly used drugs to treat IBD during different stages of the disease, to inhibit inflammatory cell function, cytokine production and to suppress disease activity (10). However, a large number of patients develop steroid resistance and become susceptible to relapse following treatment termination. Additionally, corticosteroid therapy is associated with serious side effects, including increased vulnerability to infection, fluid retention, hypertension, muscle wasting, hyperglycemia, insulin resistance and diabetes mellitus (10). Corticosteroid-induced diabetes is a major systemic manifestation in IBD and represents a challenge for treatment approaches (10).
Previous preclinical and clinical studies have demonstrated that oral hypoglycemic drugs like metformin may improve chronic inflammatory conditions in diabetic patients through reduction of hyperglycemia-induced oxidative stress, improvement of insulin sensitivity, dyslipidemia and its direct anti-inflammatory effects on different tissues (16,39). Several studies have demonstrated that metformin may downregulate the inflammatory process by inhibition of NF-κB activity through AMPK-dependent and independent signaling pathways, therefore it may be potentially used as an adjunct therapy of different inflammatory conditions, including atherosclerosis, diabetic nephropathy, nonalcoholic steatohepatitis, neurodegenerative disorders and asthma (15,39). Furthermore, metformin has been demonstrated to reduce cardiovascular events in diabetic patients through reductions in systemic inflammatory markers (40).
Metformin exerts its various functions through activation of AMPK, which is a serine/threonine protein kinase that functions as a cellular metabolic sensor and insulin sensitizer (12,13). It has been demonstrated that agents that activate AMPK may provide multiple anti-inflammatory effects through inhibition of other inflammatory cell functions and cytokine production (41,42). The mechanism by which metformin reduced expression of cytokines and chemokines in CSMCs was beyond the scope of the present study. It may be speculated that increased intracellular AMPK activity results in attenuation of NF-κB phosphorylation and translocation to the nucleus. NF-κB is an important transcription factor that controls different inflammatory functions and has been demonstrated to be suppressed by AMPK (41,43). In addition, several experimental studies have demonstrated inhibitory activities of metformin on NF-κB (13,15,19).
Cytokines and chemokines serve an important role in driving intestinal inflammation, local complications (including colon cancer) and systemic manifestations in patients with IBD, therefore agents that inhibit their production are considered extremely beneficial in treating IBD (23,44). However, therapies that target single cytokines, including infliximab, may only benefit certain subgroups of patients due to the nature of the cytokine network, where blockage of a specific cytokine may lead to development of a compensatory increase in other cytokines. Therefore, agents that inhibit multiple cytokines including metformin may provide great benefits for patients with IBD (23,45).
In conclusion, metformin, an AMPK activator, successfully attenuated expression and secretion of several cytokines and chemokines in mouse CSMCs following LPS challenge. Fig. 5 represents an integrative hypothetical model for how metformin may reduce inflammatory marker expression in CSMCs. Metformin may confer a great advantage in preventing chronic inflammatory diseases and colon cancer, beyond its ability to normalize blood glucose levels, and thus it may be possible to utilize it as an adjunct therapy to treat IBD. The present study has certain limitations since it was performed in vitro, therefore future work should evaluate anti-inflammatory effects of metformin on CSMCs in animal models of IBD in vivo, and potentially evaluate inflammatory gene expression in colonic tissues from IBD patients treated with metformin.
Acknowledgements
Not applicable.
Funding
The present study was funded by the Deanship of Research, Jordan University of Science and Technology, Irbid, Jordan (grant nos. 20160138 and 20150147).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
AA: Conception and design of study, acquisition of data, analysis and interpretation of data, drafting the manuscript and revising the manuscript critically for important intellectual content. MoA, OA and MaA: Analysis and interpretation of data and revising the manuscript critically for important intellectual content. DA: Acquisition of data, analysis and interpretation of data, drafting and revising the manuscript critically for important intellectual content.
Ethics approval and consent to participate
All procedures were approved and performed according to the guidelines of the Animal Care and Use Committee at Jordan University of Science and Technology, Irbid, Jordan.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Snape WJ, Jr, Williams R, Hyman PE. Defect in colonic smooth muscle contraction in patients with ulcerative colitis. Am J Physiol. 1991;261:G987–G991. doi: 10.1152/ajpgi.1991.261.6.G987. [DOI] [PubMed] [Google Scholar]
- 2.Severi C, Sferra R, Scirocco A, Vetuschi A, Pallotta N, Pronio A, Caronna R, Di Rocco G, Gaudio E, Corazziari E, Onori P. Contribution of intestinal smooth muscle to Crohn's disease fibrogenesis. Eur J Histochem. 2014;58:2457. doi: 10.4081/ejh.2014.2457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Vermillion DL, Huizinga JD, Riddell RH, Collins SM. Altered small intestinal smooth muscle function in Crohn's disease. Gastroenterology. 1993;104:1692–1699. doi: 10.1016/0016-5085(93)90647-U. [DOI] [PubMed] [Google Scholar]
- 4.Vrees MD, Pricolo VE, Potenti FM, Cao W. Abnormal motility in patients with ulcerative colitis: The role of inflammatory cytokines. Arch Surg. 2002;137:439–446. doi: 10.1001/archsurg.137.4.439. [DOI] [PubMed] [Google Scholar]
- 5.Shea-Donohue T, Notari L, Sun R, Zhao A. Mechanisms of smooth muscle responses to inflammation. Neurogastroenterol Motil. 2012;24:802–811. doi: 10.1111/j.1365-2982.2012.01986.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shi XZ, Sarna SK. Transcriptional regulation of inflammatory mediators secreted by human colonic circular smooth muscle cells. Am J Physiol Gastrointest Liver Physiol. 2005;289:G274–G284. doi: 10.1152/ajpgi.00512.2004. [DOI] [PubMed] [Google Scholar]
- 7.Salinthone S, Singer CA, Gerthoffer WT. Inflammatory gene expression by human colonic smooth muscle cells. Am J Physiol Gastrointest Liver Physiol. 2004;287:G627–G637. doi: 10.1152/ajpgi.00462.2003. [DOI] [PubMed] [Google Scholar]
- 8.Abraham C, Cho JH. Inflammatory bowel disease. N Engl J Med. 2009;361:2066–2078. doi: 10.1056/NEJMra0804647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bähler C, Schoepfer AM, Vavricka SR, Brüngger B, Reich O. Chronic comorbidities associated with inflammatory bowel disease: Prevalence and impact on healthcare costs in Switzerland. Eur J Gastroenterol Hepatol. 2017;29:916–925. doi: 10.1097/MEG.0000000000000891. [DOI] [PubMed] [Google Scholar]
- 10.Tamez-Pérez HE, Quintanilla-Flores DL, Rodríguez-Gutiérrez R, González-González JG, Tamez-Peña AL. Steroid hyperglycemia: Prevalence, early detection and therapeutic recommendations: A narrative review. World J Diabetes. 2015;6:1073–1081. doi: 10.4239/wjd.v6.i8.1073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fakhoury M, Negrulj R, Mooranian A, Al-Salami H. Inflammatory bowel disease: Clinical aspects and treatments. J Inflamm Res. 2014;7:113–120. doi: 10.2147/JIR.S65979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pernicova I, Korbonits M. Metformin-mode of action and clinical implications for diabetes and cancer. Nat Rev Endocrinol. 2014;10:143–156. doi: 10.1038/nrendo.2013.256. [DOI] [PubMed] [Google Scholar]
- 13.Cameron AR, Morrison VL, Levin D, Mohan M, Forteath C, Beall C, McNeilly AD, Balfour DJ, Savinko T, Wong AK, et al. Anti-inflammatory effects of metformin irrespective of diabetes status. Circ Res. 2016;119:652–665. doi: 10.1161/CIRCRESAHA.116.308445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hirsch HA, Iliopoulos D, Struhl K. Metformin inhibits the inflammatory response associated with cellular transformation and cancer stem cell growth. Proc Natl Acad Sci USA. 2013;110:972–977. doi: 10.1073/pnas.1221055110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim SA, Choi HC. Metformin inhibits inflammatory response via AMPK-PTEN pathway in vascular smooth muscle cells. Biochem Biophys Res Commun. 2012;425:866–872. doi: 10.1016/j.bbrc.2012.07.165. [DOI] [PubMed] [Google Scholar]
- 16.Kita Y, Takamura T, Misu H, Ota T, Kurita S, Takeshita Y, Uno M, Matsuzawa-Nagata N, Kato K, Ando H, et al. Metformin prevents and reverses inflammation in a non-diabetic mouse model of nonalcoholic steatohepatitis. PLoS One. 2012;7:e43056. doi: 10.1371/journal.pone.0043056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cao X, Li H, Tao H, Wu N, Yu L, Zhang D, Lu X, Zhu J, Lu Z, Zhu Q. Metformin inhibits vascular calcification in female rat aortic smooth muscle cells via the AMPK-eNOS-NO pathway. Endocrinology. 2013;154:3680–3689. doi: 10.1210/en.2013-1002. [DOI] [PubMed] [Google Scholar]
- 18.Lee SY, Lee SH, Yang EJ, Kim EK, Kim JK, Shin DY, Cho ML. Metformin ameliorates inflammatory bowel disease by suppression of the STAT3 signaling pathway and regulation of the between Th17/Treg balance. PLoS One. 2015;10:e0135858. doi: 10.1371/journal.pone.0135858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Isoda K, Young JL, Zirlik A, MacFarlane LA, Tsuboi N, Gerdes N, Schönbeck U, Libby P. Metformin inhibits proinflammatory responses and nuclear factor-kappaB in human vascular wall cells. Arterioscler Thromb Vasc Biol. 2006;26:611–617. doi: 10.1161/01.ATV.0000201938.78044.75. [DOI] [PubMed] [Google Scholar]
- 20.Al-Dwairi A, Alqudah TE, Al-Shboul O, Alqudah M, Mustafa AG, Alfaqih MA. Glucagon-like peptide-1 exerts anti-inflammatory effects on mouse colon smooth muscle cells through the cyclic adenosine monophosphate/nuclear factor-κB pathway in vitro. J Inflamm Res. 2018;11:95–109. doi: 10.2147/JIR.S152835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Berkes J, Viswanathan VK, Savkovic SD, Hecht G. Intestinal epithelial responses to enteric pathogens: Effects on the tight junction barrier, ion transport, and inflammation. Gut. 2003;52:439–451. doi: 10.1136/gut.52.3.439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Brynskov J, Foegh P, Pedersen G, Ellervik C, Kirkegaard T, Bingham A, Saermark T. Tumour necrosis factor alpha converting enzyme (TACE) activity in the colonic mucosa of patients with inflammatory bowel disease. Gut. 2002;51:37–43. doi: 10.1136/gut.51.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Neurath MF. Cytokines in inflammatory bowel disease. Nat Rev Immunol. 2014;14:329–342. doi: 10.1038/nri3661. [DOI] [PubMed] [Google Scholar]
- 24.Rizzo G, Pugliese D, Armuzzi A, Coco C. Anti-TNF alpha in the treatment of ulcerative colitis: A valid approach for organ-sparing or an expensive option to delay surgery? World J Gastroenterol. 2014;20:4839–4845. doi: 10.3748/wjg.v20.i17.4839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lv R, Qiao W, Wu Z, Wang Y, Dai S, Liu Q, Zheng X. Tumor necrosis factor alpha blocking agents as treatment for ulcerative colitis intolerant or refractory to conventional medical therapy: A meta-analysis. PLoS One. 2014;9:e86692. doi: 10.1371/journal.pone.0086692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Scarpa M, Kessler S, Sadler T, West G, Homer C, McDonald C, de la Motte C, Fiocchi C, Stylianou E. The epithelial danger signal IL-1α is a potent activator of fibroblasts and reactivator of intestinal inflammation. Am J Pathol. 2015;185:1624–1637. doi: 10.1016/j.ajpath.2015.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Papadakis KA, Targan SR. Role of cytokines in the pathogenesis of inflammatory bowel disease. Annu Rev Med. 2000;51:289–298. doi: 10.1146/annurev.med.51.1.289. [DOI] [PubMed] [Google Scholar]
- 28.Dionne S, D'Agata DD, Hiscott J, Vanounou T, Seidman EG. Colonic explant production of IL-1 and its receptor antagonist is imbalanced in inflammatory bowel disease (IBD) Clin Exp Immunol. 1998;112:435–442. doi: 10.1046/j.1365-2249.1998.00595.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cominelli F, Pizarro TT. Interleukin-1 and interleukin-1 receptor antagonist in inflammatory bowel disease. Aliment Pharmacol Ther. 1996;10:49–53. doi: 10.1046/j.1365-2036.1996.22164020.x. [DOI] [PubMed] [Google Scholar]
- 30.Ghia JE, Galeazzi F, Ford DC, Hogaboam CM, Vallance BA, Collins S. Role of M-CSF-dependent macrophages in colitis is driven by the nature of the inflammatory stimulus. Am J Physiol Gastrointest Liver Physiol. 2008;294:G770–G777. doi: 10.1152/ajpgi.00453.2007. [DOI] [PubMed] [Google Scholar]
- 31.Marshall D, Cameron J, Lightwood D, Lawson AD. Blockade of colony stimulating factor-1 (CSF-I) leads to inhibition of DSS-induced colitis. Inflamm Bowel Dis. 2007;13:219–224. doi: 10.1002/ibd.20055. [DOI] [PubMed] [Google Scholar]
- 32.Franzè E, Marafini I, de Simone V, Monteleone I, Caprioli F, Colantoni A, Ortenzi A, Crescenzi F, Izzo R, Sica G, et al. Interleukin-34 induces Cc-chemokine ligand 20 in gut epithelial cells. J Crohns Colitis. 2016;10:87–94. doi: 10.1093/ecco-jcc/jjv181. [DOI] [PubMed] [Google Scholar]
- 33.Werner L, Guzner-Gur H, Dotan I. Involvement of CXCR4/CXCR7/CXCL12 interactions in inflammatory bowel disease. Theranostics. 2013;3:40–46. doi: 10.7150/thno.5135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Danese S, Gasbarrini A. Chemokines in inflammatory bowel disease. J Clin Pathol. 2005;58:1025–1027. doi: 10.1136/jcp.2005.030916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mazzucchelli L, Hauser C, Zgraggen K, Zgraggen K, Wagner HE, Hess MW, Laissue JA, Mueller C. Differential in situ expression of the genes encoding the chemokines MCP-1 and RANTES in human inflammatory bowel disease. J Pathol. 1996;178:201–206. doi: 10.1002/(SICI)1096-9896(199602)178:2<201::AID-PATH440>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 36.Luo Y, Dorf ME. Beta-chemokine TCA3 binds to mesangial cells and induces adhesion, chemotaxis, and proliferation. J Immunol. 1996;156:742–748. [PubMed] [Google Scholar]
- 37.Scheerens H, Hessel E, de Waal-Malefyt R, Leach MW, Rennick D. Characterization of chemokines and chemokine receptors in two murine models of inflammatory bowel disease: IL-10-/-mice and Rag-2-/-mice reconstituted with CD4+ CD45RBhigh T cells. Eur J Immunol. 2001;31:1465–1474. doi: 10.1002/1521-4141(200105)31:5<1465::AID-IMMU1465>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 38.Xia XM, Wang FY, Zhou J, Hu KF, Li SW, Zou BB. CXCR4 antagonist AMD3100 modulates claudin expression and intestinal barrier function in experimental colitis. PLoS One. 2011;6:e27282. doi: 10.1371/journal.pone.0027282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Koh SJ, Kim JM, Kim IK, Ko SH, Kim JS. Anti-inflammatory mechanism of metformin and its effects in intestinal inflammation and colitis-associated colon cancer. J Gastroenterol Hepatol. 2014;29:502–510. doi: 10.1111/jgh.12435. [DOI] [PubMed] [Google Scholar]
- 40.Kothari V, Galdo JA, Mathews ST. Hypoglycemic agents and potential anti-inflammatory activity. J Inflamm Res. 2016;9:27–38. doi: 10.2147/JIR.S86917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Salminen A, Hyttinen JM, Kaarniranta K. AMP-activated protein kinase inhibits NF-κB signaling and inflammation: Impact on healthspan and lifespan. J Mol Med (Berl) 2011;89:667–676. doi: 10.1007/s00109-011-0748-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.He C, Li H, Viollet B, Zou MH, Xie Z. AMPK suppresses vascular inflammation in vivo by inhibiting signal transducer and activator of transcription-1. Diabetes. 2015;64:4285–4297. doi: 10.2337/db15-0107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Atreya I, Atreya R, Neurath MF. NF-kappaB in inflammatory bowel disease. J Intern Med. 2008;263:591–596. doi: 10.1111/j.1365-2796.2008.01953.x. [DOI] [PubMed] [Google Scholar]
- 44.Nair DG, Miller KG, Lourenssen SR, Blennerhassett MG. Inflammatory cytokines promote growth of intestinal smooth muscle cells by induced expression of PDGF-Rβ. J Cell Mol Med. 2014;18:444–454. doi: 10.1111/jcmm.12193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Guan Q, Zhang J. Recent advances: The imbalance of cytokines in the pathogenesis of inflammatory bowel disease. Mediators Inflamm. 2017;2017:4810258. doi: 10.1155/2017/4810258. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.