Abstract
The 8p11 myeloproliferative syndrome (EMS), a rare myeloproliferative disease, generally progresses rapidly and is characterized by chromosomal translocations of the fibroblast growth factor receptor 1 (FGFR1) gene. The FGFR1 gene is located at chromosome 8p11 and may fuse with distinct partner genes. The breakpoint cluster region gene located at chromosome 22 is one of these partner genes. The patients' clinical phenotype is primarily dependant on the partner gene that translocates with FGFR1. Of all the available examinations, determination of the chromosome karyotype is most essential for the diagnosis of EMS. In addition, regarding treatment, allogeneic hematopoietic stem cell transplantation is currently the optimal method. The present study presented a case of 8p11 myeloproliferative syndrome with t(8;22)(p11;q11). This represents a total of 8 and 11 chromosomal translocations, which form a BCR/FGFR1 fusion gene in the patient to produce the abnormal karyotype: 46,XY,t(8;22)(p11;q11). The difference between the current case and other EMS incidences is that the patient progressed slowly and the clinical manifestation was similar to chronic myeloid leukemia (CML).
Keywords: 8p11 myeloidproliferative syndrome, fibroblast growth factor receptor 1, chronic myeloid leukemia
Introduction
The 8p11 myeloproliferative syndrome (EMS) is an infrequent, aggressive hematological disease. At the molecular level, EMS is defined as chromosomal rearrangements of the fibroblast growth factor receptor 1 (FGFR1) gene. Among these karyotypes, there are distinct fusion partners, including the breakpoint cluster region (BCR) gene on chromosome 22. The FGFR1 fusion pattern is correlated with the phenotype and prognosis of the disease (1–3).
EMS presenting with t(8;22)(p11;q11) is infrequent in clinical practice, with only 14 cases reported to date, as presented in Table I. This type of disease may rapidly progress to acute myeloid leukemia (AML) or, less commonly, to acute lymphoblastic leukemia (ALL) (4–6).
Table I.
Previous case studies reporting on t(8;22)/breakpoint cluster region-fibroblast growth factor receptor 1 rearrangements.
| Author, year | Age (years), sex | Clinical manifestation prior to EMS diagnosis | Chromosomal abnormalities | Treatment | Outcome | (Refs.) |
|---|---|---|---|---|---|---|
| Fioretos, 2001 | 75, male | AML | 46,XY,t(8;22)(p11;q11) t(9;21)(q34;q22) | Not reported | Not reported | (3) |
| Demiroglou, 2001 | 65, female | CML-CP | Not reported | Hydroxyurea, IFNα | Alive on publication | (10) |
| Demiroglou, 2001 | 51, female | CML-CP | Not reported | Hydroxyurea, IFNα | Alive on publication | (10) |
| Pini, 2002 | 74, female | CML-CP | Not reported | Not reported | Not reported | (12) |
| Murati, 2005 | Not available | CML-CP and B-cell proliferation | Not available | Not available | Not reported | (13) |
| Agerstam, 2007 | 58, female | AML | 46,XY,t(8;22)(p11;q11) | Not reported | Not reported | (14) |
| Richebourg, 2008 | 56, female | CML, evolved to blast crisis | 47,XX,t(3;21)(q26;q22) t(8;22)(p11;q11), +der (22) | Hydroxyurea+IFNα, arsenic trioxide, cytarabine, daunorubicin+cytarabine | Death 24 months after presentation | (15) |
| Baldazzi, 2010 | 70, female | pre-ALL | 45,XX,del (3)(p11p21), del (7)(p12p15), t(8,22) (p11;q11), add (8)(p23), −9 | Induction combined chemotherapy | Death 24 months after presentation | (6) |
| Wakim, 2011 | 43, male | B-ALL | 45,XY,t6;11)(q11;p13), −7, t(8;22)(p11.2;q11.2), del (9) (p13p22) | Hyper-CVAD | Death | (5) |
| Haslam, 2012 | 21, male | B-ALL | 46,XY,t(8;22)(p12;q11)/45, idem, der (3;9)(q10;q10), dic (7;11)(p11;q13), +r (cp3) | FLAG, HSCT | Alive on publication | (9) |
| Morishige, 2012 | 50, male | Trilineage acute leukemia lymphoma | Not available | Cord blood | Alive on publication transplantation | (8) |
| Matikas, 2013 | 74, female | AML | 46,XX, del (5) q33q35, t(8;22)(p11;q11) | Mitoxantrone+etoposide, interferon, hydroxyurea, flutalazine | Alive on publication | (4) |
| Dolan, 2012 | 8, male | Juvenile myelomonocytic leukemia | 46,XY, inv (4)(p15.2q13), t(8;22)(p11;q11;q24) | Allogenic HSCT | Alive on publication | (7) |
| Qin, 2016 | 26, female | CML | 46,XX,t(8;22)(p11;q11) | Hydroxyurea | Alive on publication | (16) |
CML-CP, chronic myeloid leukemia in chronic phase; AML, acute myeloid leukemia; B-ALL, B-cell acute lymphoblastic leukemia; IFN, interferon; HSCT, hematopoietic stem cell transplantation; del, deletion; hyper-CVAD, hyperfractionated administration of cyclophosphamide, vincristine, doxorubicin and dexamethasone; idem, chromosomal abnormalities; der, translocation derived; dic, dimeric; FLAG, initial chemotherapy for acute monocytic leukemia; dic, dimeric centromeric; inv, inverted.
The present study reports on a case EMS with t(8;22)(p11;q11), presenting as chronic myeloid leukemia (CML), as distinguished by its slow progression, which differs from that of other cases presenting as AML, ALL and lymphoma.
Case report
Patient information
A 41-year-old male who had been presenting with progressive leukocytosis and thrombocytosis for 1 year was admitted to Tongji Hospital (Wuhan, China) in October 2016. The patient was formerly physically healthy and exhibited no obvious symptoms. Physical examination indicated no lymph node enlargement or hepatosplenomegaly, which was confirmed by ultrasonography and abdominal computed tomography. The initial complete blood analysis revealed hemoglobin levels of 139 g/l (normal range, 115-150 g/l), a white blood cell (WBC) count of 23×109/l (normal range, 3.5-9.5×109/l) and a platelet count of 492×109/l (normal range, 125-350×109/l). For the past year, the patient's WBC and platelet counts had been steadily increasing. Bone marrow (BM) cytology smears and BM biopsy indicated myelosis and granulocyte hyperplasia. BM cell differential analysis revealed 92% granulocytes (with a myeloid/erythroid cell ratio of 26:1). Polymerase chain reaction analysis indicated negativity for the BCR-Abelson murine leukemia fusion gene and the Janus kinase 2/Val617Phe mutation. Cytogenetic analysis of the BM was performed with 10 metaphase cells, all of which carried the 46,XY,t(8;22)(p11;q11) mutation. Fluorescence in situ hybridization (FISH) further validated this chromosome translocation. These results provided crucial evidence for the diagnosis of EMS, although unlike those of other EMS cases, the patient's clinical manifestations resembled those of CML.
EMS usually progresses rapidly and is associated with a poor prognosis. However, one more year after the initial diagnosis, the patient remained asymptomatic without any treatment, while his leucocytosis and thrombocytosis did not mitigate. According to previous studies, hematopoietic cell transplantation remains the only effective measure to control EMS (7–11). However, the patient refused to receive hematopoietic cell transplantation and was only followed up and monitored WBC count once a month for 2 years.
Examination results
BM cytology smears and biopsy
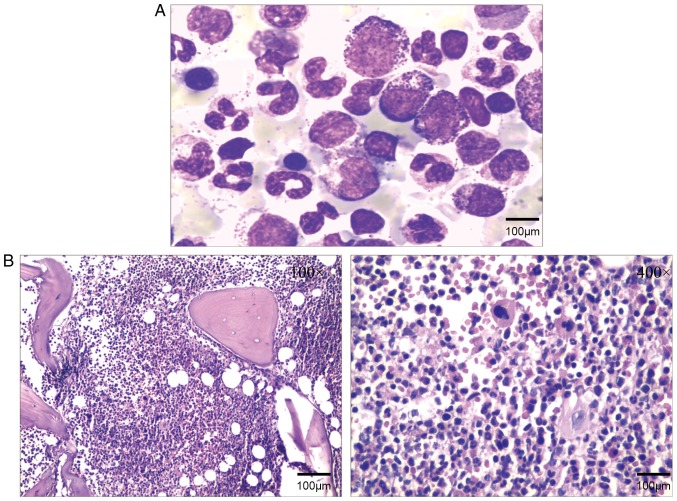
To analyze the myeloproliferative status, BM cytology smears and BM biopsy were performed at a hematology laboratory. The cytology and biopsy results indicated that the BM was hypercellular with granulocytic hyperplasia, as presented in Fig. 1A and B.
Figure 1.
(A) Bone marrow cytology stained with Swiss-stain, revealed active bone marrow hyperplasia, and proliferation of mostly granulocytes, while erythroid and megakaryocytic proliferation was inhibited. (B) Bone marrow biopsy indicated primarily granulocyte hyperplasia and active bone marrow hyperplasia.
Cytogenetics
Sufficient sample material obtained via BM aspiration was available to examine the BCR gene, which is located at 22q11. The initial analysis indicated that in the present case, FGFR1 crosses the breakpoint located on chromosome 22q11. Following initial diagnosis, 10 BM cells in metaphase were analyzed, and all of them were clonally abnormal karyotypes, while the abnormalities were 8 and 11 chromosomal translocations, forming a BCR/FGFR1 fusion gene in the patient. The abnormal karyotype 46,XY,t(8;22)(p11;q11) was observed in all of the cells, with no other abnormal karyotypes. The BCR-FGFR1 fusion gene is the characteristic chromosome karyotype in 8p11 myeloproliferative syndrome. At 2 months after the initial diagnosis, the karyotype remained identical, as indicated in Fig. 2. Although karyotype analysis is a simple and basic technology, it is of great significance for the diagnosis of EMS.
Figure 2.
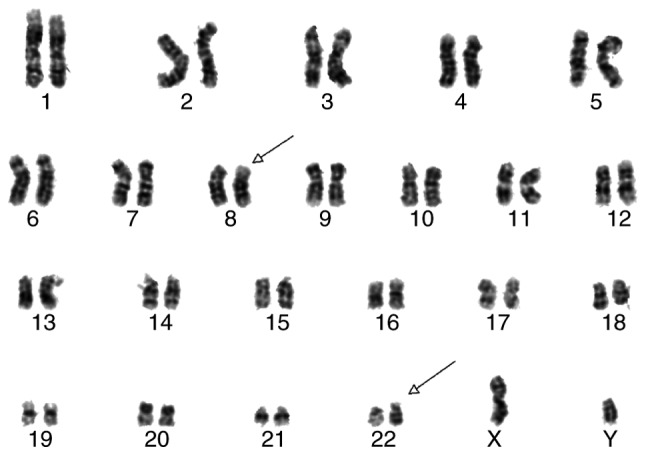
A total of 10 bone marrow cells in metaphase were subjected to karyotype analysis. All of them were clonal abnormal karyotypes with 8 and 11 chromosomal translocations, forming a breakpoint cluster region-fibroblast growth factor receptor 1 fusion gene in the patient (arrows).
FISH
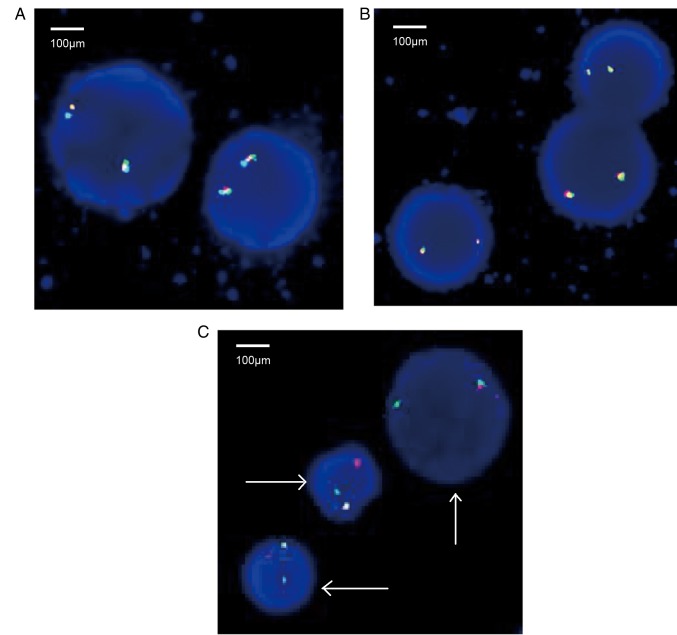
Once the gene has been cloned and characterized, FISH analysis may be performed. In the present study, the rearrangement of the FGFR1 gene was verified by FISH. A total of 200 interphase nuclei were examined for each probe, and no abnormal signals regarding the platelet-derived growth factor receptor (PDGFR)A (4q12) gene and PDGFRB (5q32) rearrangements were detected (Fig. 3A and B, respectively). However, ~79% of the nuclei had signals indicative of the FGFR1 (8p11) gene rearrangement (Fig. 3C). The results further confirmed the formation of the BCR-FGFR1 fusion gene.
Figure 3.
Rearrangement of the FGFR1 gene was also demonstrated by fluorescence in situ hybridization. A total of 200 cells in interphase were counted using three sets of probes. (A) Two fusion genes exhibiting red, green and blue staining. No abnormal signals of PDGFRA (4q12) gene rearrangement were detected; (B) two fusion genes exhibiting red, green and blue staining. No abnormal signals of PDGFRB (5q32) gene rearrangement were detected; (C) cells with red, green and blue fusion gene signals, indicating the presence of the FGFR1 (8p11) gene rearrangement using FISH analysis. The derivative chromosomes are indicated by arrows. FGFR1, fibroblast growth factor receptor 1; PDGFR, platelet-derived growth factor receptor.
Discussion
EMS is characterized by the following: i) Myeloproliferative neoplasm associated with eosinophilia; ii) Lymphadenopathy; iii) High tendency toward converting to AML; and iv) Reciprocal translocations involving 8p11 (2).
To the best of the author's knowledge, the present study reports on the 15th published case of t(8;22)/BCR-FGFR1 rearrangement. All other cases are presented in Table I. The FGFR1 gene encodes a receptor tyrosine kinase transmembrane protein. It consists of the following three parts: The extracellular domain, the transmembrane domain and the intracellular domain. Under normal circumstances, FGFR1 is in the form of oligomers, where binding of FGFR1 to its ligands, e g BCR, results in FGFR1 homodimerization and autophosphorylation, thus activating multiple effectors, including RAS/mitogen-activated protein kinase/phosphoinositide-3 kinase/phosphoinositide phospholipase C-γ, and providing proliferative and survival signals. BCR is one of the 15 fusion partners of FGFR1. The 15 rearrangement karyotypes are as follows: Myosin XVIIIA (MYO18A; 17q23), tripartite motif containing 24 (TIF1; 7q34), FGFR1 oncogene partner 2 (FGFR1OP2; 12p11), human endogenous retrovirus group K member (HERV-K; 19q13), BCR (22q11), centriolin (CEP110; 9q33), FGFR1 oncogene partner (FOP/FGFR10P; 6q27), zinc finger protein 198 (ZNF198; 13q11-12), cleavage and polyadenylation specific factor 6 (CPSF6; 12q15), tripartite motif containing 24 (TRIM24; 7q34), nuceloporin 98 (NUP98; 11p15), cut like homeobox 1 (CUX1; 7q22), translocated promoter region, nuclear basket protein (TPR; 1q25), RAN binding protein 2 (RANBP2; 2q12) and LPR binding FLII interacting protein 1 (LRRFIPI; 2q37). Among these translocations, t(8;13) is the most common (1–3).
Unlike other cases of FGFR1 rearrangement, which frequently manifest as eosinophilia and lymphadenopathy, the clinical presentation of the case of the present study was as CML. This particular clinical manifestation suggests a specific role of BCR during the development of the disease. FGFR1 has a critical role in the oncogenesis of EMS, and each FGFR1 fusion partner exerts a different influence on the malignant phenotype (2,4).
In other studies reporting on cases of EMS, which appear as leukemia or lymphoblastic lymphoma, accompanying chromosomal abnormalities frequently exist; however, in the present case, the only abnormal karyotype was t(8;22)(p11;q11). The unique clinical features of this patient may be attributed to the absence of any additional chromosomal abnormalities.
As for the treatment of EMS, numerous studies have reported that patients benefited from allogeneic hematopoietic stem cell transplantation (7–9). The application of FGFR1 inhibitors has also been reported in certain cases, but the clinical outcome was not improved (5). Therefore, allogeneic hematopoietic stem cell transplantation remains the primary treatment. In addition, chemotherapy is not ideal for patients who present with acute leukemia or lymphoblastic lymphoma. EMS has a rapidly progressing clinical course with a median survival time of <1 year (4,5).
The present study emphasizes the importance of accurate molecular diagnosis in FGFR1 rearrangement cases. In cases of non-classical clinical manifestations, accurate molecular diagnosis may avoid misdiagnoses. Conceivably, early diagnosis provides patients with the opportunity to adopt allogeneic hematopoietic stem cell transplantation in an early phase of the disease and may thus improve the clinical outcome of the patients.
Of note, EMS is a type of disease that may be misdiagnosed. In the present case report, a 41-year-old patient with a misdiagnosis of CML due to an increase in the number of white blood cells was subsequently diagnosed with EMS after undergoing a karyotype test. The present study may contribute to the improvement of the diagnosis of EMS in patients, which may first appear to have CML. EMS should be considered in patients with symptoms similar to those of the case of the present study.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
JJL analysed and interpreted the patient data. LM analyzed the hematological data. JJL and LM drafted and revised the manuscript. All authors read and approved the final manuscript.
Ethical approval and consent to participate
Not applicable.
Patient consent for publication
The current case report was published with informed consent of the patient, whose anonymity was preserved.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Kim WS, Park SG, Park G, Jang SJ, Moon DS, Kang SH. 8p11 myeloproliferative syndrome with t(1;8)(q25;p11.2): A case report and review of the literature. Acta Haematol. 2015;133:101–105. doi: 10.1159/000363441. [DOI] [PubMed] [Google Scholar]
- 2.Jackson CC, Medeiros LJ, Miranda RN. 8p11 myeloproliferative syndrome: A review. Hum Pathol. 2010;41:461–476. doi: 10.1016/j.humpath.2009.11.003. [DOI] [PubMed] [Google Scholar]
- 3.Fioretos T, Panagopoulos I, Lassen C, Swedin A, Billström R, Isaksson M, Strömbeck B, Olofsson T, Mitelman F, Johansson B. Fusion of the BCR and the Fibroblast Growth Factor Receptor-1 (FGFR1) genes as a result of t(8;22)(p11;q11) in a myeloproliferative disorder: The first fusion gene involving BCR but not ABL. Genes Chromosomes Cancer. 2001;32:302–310. doi: 10.1002/gcc.1195. [DOI] [PubMed] [Google Scholar]
- 4.Matikas A, Tzannou I, Oikonomopoulou D, Bakiri M. A case of acute myelogenous leukaemia characterised by the BCR-FGFR1 translocation. BMJ Case Rep. 2013;20 doi: 10.1136/bcr-2013-008834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wakim JJ, Tirado CA, Chen W, Collins R. t(8;22)/BCR-FGFR1 myeloproliferative disorder presenting as B acute lymphoblastic leukemia: Report of a case treated with sorafenib and review of the literature. Leuk Res. 2011;35:e151–153. doi: 10.1016/j.leukres.2011.05.013. [DOI] [PubMed] [Google Scholar]
- 6.Baldazzi C, Iacobucci I, Luatti S, Ottaviani E, Marzocchi G, Paolini S, Stacchini M, Papayannidis C, Gamberini C, Martinelli G, et al. B-cell acute lymphoblastic leukemia as evolution of a 8p11 myeloproliferative syndrome with t(8;22)(p11;q11) and BCRFGFR1 fusion gene. Leuk Res. 2010;34:e282–285. doi: 10.1016/j.leukres.2010.05.009. [DOI] [PubMed] [Google Scholar]
- 7.Dolan M, Cioc A, Cross NC, Neglia JP, Tolar J. Favorable outcome of allogeneic hematopoietic cell transplantation for 8p11 myeloproliferative syndrome associated with BCR-FGFR1 gene fusion. Pediatr Blood Cancer. 2012;59:194–196. doi: 10.1002/pbc.23404. [DOI] [PubMed] [Google Scholar]
- 8.Morishige S, Oku E, Takata Y, Kimura Y, Arakawa F, Seki R, Imamura R, Osaki K, Hashiguchi M, Yakushiji K, et al. A case of 8p11 myeloproliferative syndrome with BCR-FGFR1 gene fusion presenting with trilineage acute leukemia/lymphoma, successfully treated by cord blood transplantation. Acta Haematol. 2013;129:83–89. doi: 10.1159/000341289. [DOI] [PubMed] [Google Scholar]
- 9.Haslam K, Langabeer SE, Kelly J, Coen N, O'Connell NM, Conneally E. Allogeneic hematopoietic stem cell transplantation for a BCR-FGFR1 myeloproliferative neoplasm presenting as acute lymphoblastic leukemia. Case Report Hematol. 2012;2012:620967. doi: 10.1155/2012/620967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Demiroglu A, Steer EJ, Heath C, Taylor K, Bentley M, Allen SL, Koduru P, Brody JP, Hawson G, Rodwell R, et al. The t(8;22) in chronic myeloid leukemia fuses BCR to FGFR1: Transforming activity and specific inhibition of FGFR1 fusion proteins. Blood. 2001;98:3778–3783. doi: 10.1182/blood.V98.13.3778. [DOI] [PubMed] [Google Scholar]
- 11.Chase A, Grand FH, Cross NC. Activity of TKI258 against primary cells and cell lines with FGFR1 fusion genes associated with the 8p11 myeloproliferative syndrome. Blood. 2007;110:3729–3734. doi: 10.1182/blood-2007-02-074286. [DOI] [PubMed] [Google Scholar]
- 12.Pini M, Gottardi E, Scaravaglio P, Giugliano E, Libener R, Baraldi A, Muzio A, Cornaglia E, Saglio G, Levis A. A fourth case of BCR-FGFR1 positive CML-like disease with t(8;22) translocation showing an extensive deletion on the derivative chromosome 8p. Hematol J. 2002;3:315–316. doi: 10.1038/sj.thj.6200201. [DOI] [PubMed] [Google Scholar]
- 13.Murati A, Arnoulet C, Lafage-Pochitaloff M, Adélaide J, Derré M, Slama B, Delaval B, Popovici C, Vey N, Xerri L, et al. Dual lympho-myeloproliferative disorder in a patient with t(8;22) with BCR-FGFR1 gene fusion. Int J Oncol. 2005;26:1485–1492. [PubMed] [Google Scholar]
- 14.Agerstam H, Lilljebjörn H, Lassen C, Swedin A, Richter J, Vandenberghe P, Johansson B, Fioretos T. Fusion gene-mediated truncation of RUNX1 as a potential mechanism underlying disease progression in the 8p11 myeloproliferative syndrome. Genes Chromosomes Cancer. 2007;46:635–643. doi: 10.1002/gcc.20442. [DOI] [PubMed] [Google Scholar]
- 15.Richebourg S, Theisen O, Plantier I, Parry A, Soenen-Cornu V, Lepelley P, Preudhomme C, Renneville A, Laï JL, Roche-Lestienne C. Chronic myeloproliferative disorder with t(8;22)(p11;q11) can mime clonal cytogenetic evolution of authentic chronic myelogeneous leukemia. Genes Chromosomes Cancer. 2008;47:915–918. doi: 10.1002/gcc.20588. [DOI] [PubMed] [Google Scholar]
- 16.Qin YW, Yang YN, Bai P, Wang C. Chronic myelogenous leukemia-like hematological malignancy with t(8;22) in a 26-year-old pregnant woman: A case report. Oncol Lett. 2016;11:4131–4133. doi: 10.3892/ol.2016.4505. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.