Abstract
Lung cancer is the leading cause of cancer-related mortality worldwide. Despite recent advances in treatment, lung cancer remains an incurable disease. Matrine, an active compound isolated from Sophora flavescens, has been demonstrated to inhibit proliferation and induce apoptosis of tumor cells. However, the protective effects and molecular mechanisms of matrine in lung cancer remain elusive. In the present study, the lung cancer cells H1299 and A549 were used to investigate how matrine affects the proliferation, migration and apoptosis of lung cancer cells in vitro. It was demonstrated that matrine is able to significantly suppress the proliferation and colony formation of lung cancer cells in vitro. Using cell apoptosis analysis, wound-healing and Transwell assays, it was demonstrated that matrine induced cellular apoptosis and inhibited the migration of lung cancer cells. Further experiments revealed that matrine significantly suppressed the phosphorylation of protein kinase B (Akt) and glycogen synthase kinase-3β (GSK-3β). The present results suggested that matrine inhibits lung cancer cell proliferation, and induces cell apoptosis by suppressing the Akt/GSK-3β signaling pathway, which demonstrated that matrine may have therapeutic potential for lung cancer.
Keywords: lung cancer, matrine, proliferation, migration, invasion, apoptosis, protein kinase B, glycogen synthase kinase-3β
Introduction
Lung cancer remains one of the major causes of cancer-related mortality worldwide (1). In the past decade, tyrosine-kinase inhibitors, immunotherapy and chemotherapy have been the primary treatments for lung cancer, resulting in a median progression-free survival time of ~6 months and a response rate of ~30%, which appears to have reached a plateau of effectiveness in improving survival (2). Although the survival of patients with lung cancer has been improved with the emergence of these treatments, novel issues continue to arise, such as drug resistance and tumor recurrence. Therefore, significant advances are eagerly awaited (3), and there remains an urgent requirement to develop novel targeted drugs in order to improve patient outcomes.
Matrine (C15H24N2O; Fig. 1A), a natural component from the traditional Chinese medical herb Sophora flavescens, exhibits multiple pharmacological properties, including anti-inflammatory, anti-tumor and anti-fibrotic effects (4–6). The mechanisms of its anti-tumor activity are complex, and include cell proliferation, migration and apoptosis (7). Previous studies have demonstrated that matrine is able to negatively regulate the phosphoinositide 3-kinase (PI3K)/protein kinase B (Akt), Wnt and mitogen activated protein kinase (MAPK) signaling pathways in a number of human tumor types, including hepatocellular carcinoma (8), pancreatic cancer (9), and nasopharyngeal carcinoma (10). The Akt/glycogen synthase kinase-3β (GSK-3β) signaling pathway has been demonstrated to regulate important genes associated with proliferation and invasion, and thus control the growth and invasion of lung cancer cells (11,12). However, the effect of matrine on lung cancer cell growth and invasion in vitro, and whether the anti-tumor mechanisms of matrine are associated with the Akt/GSK-3β signaling pathway remain unclear.
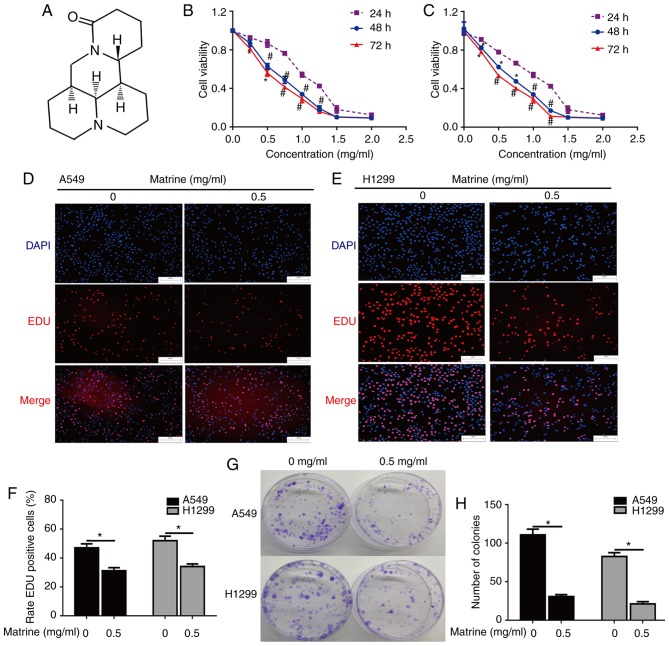
Figure 1.
The effect of matrine on A549 and H1299 cell proliferation and colony formation. (A) Chemical structure of matrine. Matrine inhibited (B) A549 and (C) H1299 cell proliferation, as determined by the Cell Counting Kit-8 assay. *P<0.05, #P<0.01 vs. 24 h. Matrine inhibited (D) A549 and (E) H1299 cell proliferation, as determined by the EdU assay. Scale bar=100 µm. (F) The ratio of EdU-positive A549 and H1299 cells suppressed by matrine was determined via the EdU assay. *P<0.05. (G and H) Matrine suppressed colony formation in human lung cancer cells. *P<0.05. Data are presented as the mean ± standard deviation (n=3). EdU, 5-ethynyl-2′-deoxyuridine.
In the present study, the effect of matrine on lung cancer was investigated in the A549 and H1299 cell lines, in order to clarify the underlying mechanism by which matrine inhibited the proliferation and induced the apoptotic ability of lung cancer.
Materials and methods
Reagents and antibodies
Matrine (>95% purity; Shanghai Yuanye Biotechnology Co., Ltd., Shanghai, China) was dissolved in PBS (10 mg/ml). Antibodies against phosphorylated (p)-Akt (Thr308) (sc-16646-R; Santa Cruz Biotechnology, Inc., Dallas, TX, USA), Akt (2620S; Cell Signaling Technology, Inc., Danvers, MA, USA), β-actin (66009–1-Ig; Proteintech Group, Inc., Chicago, IL, USA), GSK-3β (9832; Cell Signaling Technology, Inc.), p-GSK-3β (9323; Cell Signaling Technology, Inc.) were used in the present study. The secondary anti-rabbit-horseradish peroxidase (HRP) (ab6721) or anti-mouse-HRP antibodies (ab6728) were purchased from Abcam (Cambridge, UK).
Cell culture
A549 and H1299 human lung cancer cell lines were purchased from the cell bank of the Chinese Academy of Science (Shanghai, China). Lung cancer cells were cultured in Dulbecco's modified Eagle's medium (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.) and 1% penicillin/streptomycin, at 37°C in a humidified incubator containing 5% CO2.
Cell proliferation and colony formation assay
A549 or H1299 cells (2,000 cells/well) were seeded into 96-well plates and treated with matrine (0, 0.5, 1, 1.5 and 2 mg/ml) for 24, 48, and 72 h at 37°C. A Cell Counting Kit-8 (CCK-8; Dojindo Molecular Technologies, Inc., Kumamoto, Japan) assay was used to evaluate cell proliferation and viability according to the manufacturer's protocol. For the colony formation assay, 1,000 cells in DMEM containing 10% FBS and 0.5 mg/ml matrine were seeded into 35-mm plates at 37°C. After 14 days, the colonies were fixed in 100% methanol at 25°C for 15 min, and stained with crystal violet at 25°C for 10 min. All procedures were performed in triplicate.
Cell proliferation 5-ethynyl-2′-deoxyuridine (EdU) assay
Proliferation of A549 and H1299 were assessed using an EdU assay kit (Guangzhou RiboBio Co., Ltd., Guangzhou, China). Briefly, 2×104 cells in DMEM containing 10% FBS were treated with 0.5 mg/ml matrine or PBS in 24-well plates at 37°C for 24 h. Subsequently, the cells were incubated with EdU (50 µM) at 37°C. After 2 h, the cells were fixed in 4% formaldehyde at 25°C for 30 min and permeabilized with 0.5% Triton X-100 at 25°C for 1 min. Following washing with PBS for 5 min, Apollo® reaction solution from the EdU assay kit (100 µl) was added for 30 min at 25°C. Finally, the cells were incubated with Hoechst 33342 (100 µl) at 25°C for 30 min to stain the nucleus. The results of the cell proliferation assays were quantified with Image-Pro Plus software (6.0; Media Cybernetics, Inc., Rockville, MD, USA) and the ratio of Apollo-positive cells to Hoechst 33342-positive cells was recorded.
Apoptosis analysis
A549 or H1299 cells (5×104) in DMEM containing 10% FBS were treated with matrine (0.5 mg/ml) or PBS at 37°C for 24 h. Apoptosis was detected using a Hoechst 33258 fluorescence staining kit (Beyotime Institute of Biotechnology, Haimen, China) according to the manufacturer's protocol. The effect of matrine on cellular apoptosis was also evaluated using an Annexin-V Apoptosis Detection kit FITC (ebioscience; Thermo Fisher Scientific, Inc.) based on the manufacturer's protocol and apoptosis was determined using a flow cytometer. The results of cell apoptosis were analyzed with FlowJo software (10.0.7; FlowJo LLC, Ashland, OR, USA).
Wound-healing, migration and invasion assays
For the wound-healing assay, 2×105 cells (A549 and H1299) were plated in culture-insert wells (Ibidi GmbH, Martinsried, Germany) with DMEM containing 10% FBS at 37°C for 24 h. The culture-insert was subsequently removed and fresh DMEM containing 0.5 mg/ml matrine or an equal amount of PBS was added. The width of the healing monolayer wound was recorded after 24 h. For the migration assay, 3×104 cells in DMEM were seeded into the upper chambers of Transwell plates (Corning, Inc., Corning, NY, USA). Complete medium containing 10% FBS in the bottom chamber was used as a chemoattractant, and 0.5 mg/ml matrine was added to inhibit cell migration. For the invasion assay, 5×104 cells in DMEM were seeded in the upper chambers of Transwell plates with 10% Matrigel (Corning, Inc.) at 37°C for 6 h. Following, DMEM with 10% FBS was used in the bottom chamber as a chemoattractant and 0.5 mg/ml matrine was added to assess cell invasion. Following 24 h migration or invasion at 37°C, cells on the lower membrane of inserts were fixed in 100% methanol at 25°C for 15 min, stained with crystal violet at 25°C for 10 min and counted using light microscopy (magnification, ×100).
Western blotting
Western blotting was performed as previously described (5). Briefly, cell samples were lysed with RIPA buffer (Beyotime, Tianjin, China) and the concentration of total protein was determined using a BCA kit (Beyotime). A total of 30 µg protein from each sample was separated by 10% SDS-PAGE gels and transferred to a polyvinylidene difluoride membrane. Membranes were blocked in 5% milk in TBS-Tween-20 (TBS-T) at 25°C for 1 h and incubated overnight at 4°C with primary antibodies against Akt (1:1,000), p-Akt (Thr308) (1:1,000), GSK-3β (1:1,000), p-GSK-3β (1:1,000) and β-actin (1:2,000). Following washing in TBS-T, membranes were incubated with HRP-conjugated anti-mouse (1:2,000) or anti-rabbit (1:2,000) secondary antibodies at 25°C for 2 h prior to visualization with ECL detection system (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Densitometry analysis was performed using Image J software (1.48; National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
Data are presented as the mean ± standard deviation. Statistical analysis was performed using Tukey's post hoc test following one-way analysis of variance. Differences between two groups were evaluated for significance using Student's t-test. P<0.05 was considered to indicate a statistically significant difference.
Results
Matrine inhibits lung cancer cell proliferation and colony formation
To elucidate whether matrine inhibits the growth of lung cancer cells, a CCK-8 assay was used to analyze the proliferation of A549 and H1299 cells treated with different doses of matrine for 24, 48 and 72 h. The results demonstrated that matrine significantly inhibited cell growth, and that this inhibition was markedly increased with increasing matrine concentration (Fig. 1B and C). The EdU incorporation assay was also performed for more sensitive and specific evaluation of the effect of matrine on proliferation. As presented in Fig. 1D-F, the number of EdU-incorporating cells in A549 and H1299 groups exposed to 0.5 mg/ml matrine was significantly decreased compared with the corresponding control groups, suggesting that matrine treatment resulted in a significant suppression of cell proliferation. The effect of matrine on colony formation was further examined in lung cancer cells via a clonogenic assay. The results demonstrated that matrine significantly reduced the number of colonies-formed compared with the control groups. (Fig. 1G and H).
Matrine inhibits migration and invasion in lung cancer cells
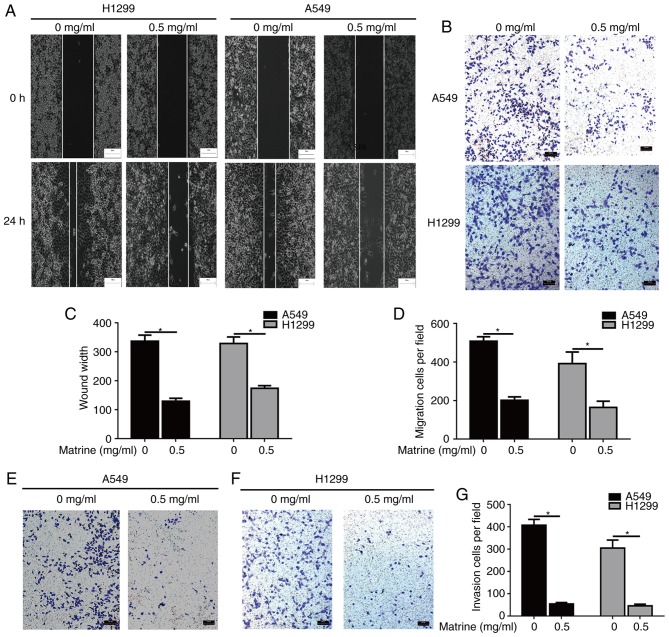
To evaluate whether matrine was able to reduce the mobility of lung cancer cells, wound-healing and Transwell assays were performed. The results of the wound-healing and Transwell assays demonstrated that matrine significantly inhibited the migration of A549 and H1299 cells (Fig. 2A-D). In addition, the effects of matrine on invasion were also evaluated, and it was observed that matrine treatment resulted in a significant decrease in the number of invasive lung cancer cells (Fig. 2E-G).
Figure 2.
Matrine suppresses migration and invasion in lung cancer cells. (A) Inhibition of cell migration by matrine was confirmed via wound-healing assay. Scale bar=100 µm. (B) Matrine reduced the number of migrated cells in the Transwell assay. Scale bar=100 µm. Quantified results of (C) wound-healing and (D) Transwell assays. (E-G) Matrine reduced the number of invasive cells in the Transwell assay. Scale bar=100 µm. Data are presented as the mean ± standard deviation (n=3). *P<0.05.
Matrine induces lung cancer cellular apoptosis
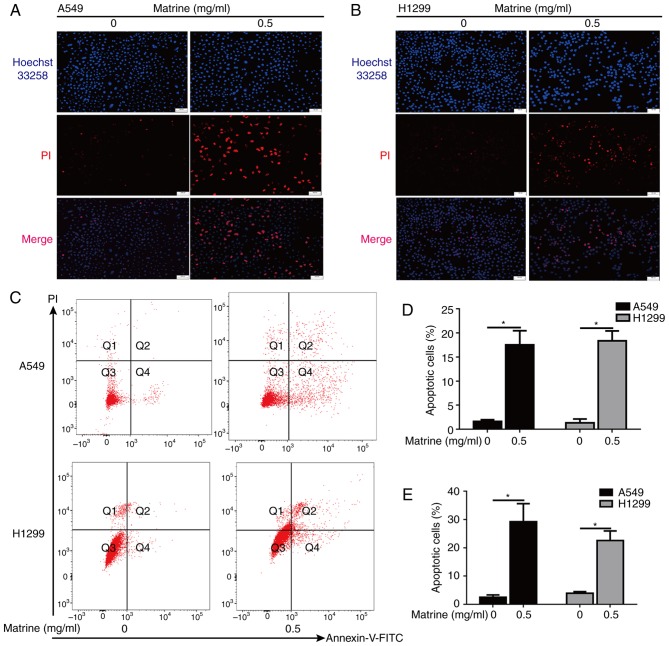
Hoechst 33342 staining of the matrine-treated A549 and H1299 cells revealed a condensed or fragmented chromatin staining pattern with brilliant blue fluorescent dots in the cell nuclei, as compared with a uniformly blue staining pattern in the control cells (Fig. 3A and B). In addition, the effects of matrine on apoptosis were further assessed by flow cytometry assays (Fig. 3C). Analysis of both experiments indicated that matrine induced a significant increase in the number of apoptotic cells (Fig. 3D and E).
Figure 3.
Matrine induces A549 and H1299 cellular apoptosis. (A) Representative images of Hoechst 33258 staining in apoptotic A549 cells following the administration of matrine. Scale bar=100 µm, n=4. (B) Representative images of Hoechst 33258 staining in apoptotic H1299 cells following the administration of matrine. Scale bar=50 µm, n=4. (C) Lung cancer cells were treated with matrine for 24 h and then labeled with Annexin-V and PI, followed by flow cytometric analysis. (D) Quantified results of apoptotic cells in different cell lines determined by Hoechst 33258 staining. (E) Quantified results of apoptotic cells in different cell lines determined by flow cytometric analysis. Data are presented as the mean ± standard deviation (n=3). *P<0.05. PI, propidium iodide.
Matrine inhibits the Akt GSK-3β signaling pathway in lung cancer cells
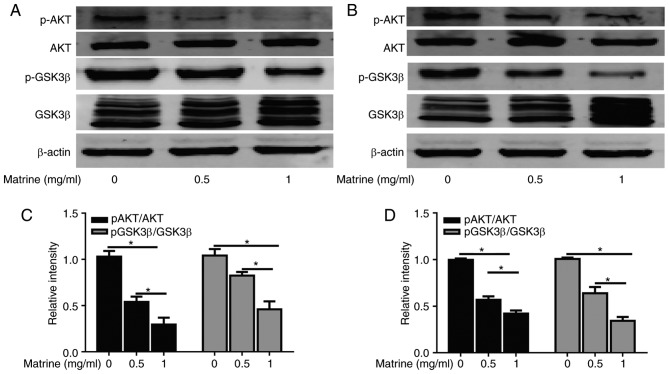
To elucidate the molecular mechanism underlying the suppression of proliferation and migration induced by matrine in lung cancer cells, western blotting was performed to analyze the classical signaling pathways associated with proliferation and migration. The degree of Akt phosphorylation at Thr308 was significantly inhibited in the matrine-treated cells compared with in the control groups, whereas no significant change was observed in the total Akt levels. Furthermore, matrine downregulated p-GSK-3β/GSK-3β protein expression in A549 and H1299 cells (Fig. 4). These results indicated that the anti-cancer effects of matrine may be associated with the Akt/GSK-3β pathway.
Figure 4.
Matrine regulates the Akt/GSK-3β pathway in A549 and H1299 cells. (A) Akt, p-Akt, GSK-3β, and p-GSK-3β (Ser9) protein expression levels in A549 cells following the administration of various doses of matrine, evaluated by immunoblotting with β-actin loading control. (B) Akt, p-Akt, GSK, and p-GSK-3β (Ser9) protein levels in H1299 cells following administration of various doses of matrine, evaluated by immunoblotting with β-actin loading control. Quantified results of the p-Akt/Akt and p-GSK-3β/GSK-3β ratios in (C) A549 and (D) H1299 cells following administration of matrine. Data are presented as the mean ± standard deviation (n=3). *P<0.05. Akt, protein kinase B; GSK-3β, glycogen synthase kinase-3β; p, phosphorylated.
Discussion
Matrine is an extract of Sophora flavescens, which has been shown to have inhibitory effects on numerous tumor types. However, the specific mechanisms underlying these inhibitory effects remains unclear, particularly in lung cancer. In the present study, the inhibitory effects of matrine on the human lung cancer cell lines A549 and H1299 were evaluated, and it was observed that matrine inhibited lung cancer cell proliferation, and induced apoptosis by blocking the Akt/GSK-3β pathway.
Previous studies have demonstrated that the combination of matrine and standard therapies can significantly improve the quality of life and prognosis of patients with cancer, suggesting its potential as an anti-cancer drug (13,14). Additionally, it is well documented that matrine can inhibit the proliferation of a variety of cancer cells (15,16). In the present study, it was demonstrated that matrine significantly inhibited the proliferation and colony formation of A549 and H1299 cells. Similar results were recently observed with the matrine derivative YF-18 in A549, H1975 and 95D cells (17). Migration and invasion are important indices for evaluating lung cancer malignancy, and are associated with the adhesion between cancer cells and the basement membrane, the depletion of the extracellular matrix and the formation of metastases (18–20). In the present study, the effect of matrine on the migration and invasion of lung cancer cells was evaluated using Transwell assays, and it was demonstrated that matrine could markedly inhibit cell mobility, as well as migration and invasion. The results were consistent with the lower mobility observed in prostate (4) and cervical cancer cells (21). However, the mechanisms underlying the inhibitory effect of matrine on tumor invasion and migration remain unclear, and may be associated with the following approaches: Inhibited degradation of proteolytic enzymes on the basement membrane and extracellular matrix (22); adhesion of lung cancer cells to the extracellular matrix (23); tumor angiogenesis; and the regulation-specific genes associated with cancer metastasis (24). It was also evaluated whether matrine was able to induce apoptosis in lung cancer cells via Hoechst 33342 staining and FACS analysis. The results demonstrated that the number of apoptotic A549 and H1299 cells following exposure to 0.5 mg/ml matrine, was significantly increased, suggesting that the anti-proliferative effect of matrine on lung cancer cells is associated with cellular apoptosis.
The Akt/GSK-3β pathway has a critical regulatory role in the signal transduction activity associated with cell proliferation, apoptosis, differentiation and survival (25). Deregulation of the Akt/GSK-3β signaling pathways has been implicated in the initiation and progression of various human malignancies. When the MAPK and PI3k/Akt pathways are blocked, the proliferation, invasion and metastasis of cancer cells has been demonstrated to be significantly inhibited (26,27). In addition, previous studies have demonstrated that matrine regulates the Akt signaling pathway, and inhibits hepatic stellate cell activation and dendritic cell maturation (28,29). Consistent with these studies, the present results demonstrated that matrine suppresses the Akt pathway by reducing the phosphorylation of Akt in lung cancer cells in a dose-dependent manner, which suggests that the inhibitory effect of matrine on these lung cancer cells was achieved via downregulation of the Akt pathway. In addition, a recent study demonstrated that matrine also modulates the Wnt/β-catenin self-renewal pathway, further resulting in the increased phosphorylation of β-catenin (Ser33/Ser37/Thr41), and that decreasing β-catenin levels, as well as its target gene Cyclin D1, ultimately suppressed cancer proliferation (30). In the present study, the effects of matrine on non-epidermal growth factor receptor (EGFR) mutant A549 and H1299 cell lines originated from epithelial and mesenchymal lung adenocarcinomas was investigated (31), and it was observed that matrine was able to inhibit the Akt pathway and subsequent GSK-3β activation, in order to ultimately inhibit proliferation and induce apoptosis in lung cancer cells. The phenomenon that epithelial cells transform to mesenchymal phenotype is closely associated with carcinogenicity, metastasis and poor prognosis in a number of tumor types, including non-small-cell lung carcinoma (NSCLC) (32,33). Further studies will investigate the effect of matrine on EGFR-mutant NSCLC cells and clarify the exact role of such downregulation in the inhibition of lung cancer cells by matrine.
In conclusion, the present findings suggest that matrine inhibits lung cancer cell proliferation, and induces cell apoptosis by suppressing the Akt/GSK-3β signaling pathway, offering possible mechanisms for its antitumor activity. The present study provides a foundation for further preclinical and clinical evaluations of matrine as a lung cancer therapy.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Natural Science Foundation of China (grant no. 81370174).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
ZG developed the idea for the study, XW, WX, JL, QL, JH and HL did the analyses and XW and HL wrote the paper. All authors have reviewed and approved the final version of the manuscript and have consented to its publication.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Hong X, Chen Q, Ding L, Liang Y, Zhou N, Fang W, Chen X, Wu H. Clinical benefit of continuing crizotinib therapy after initial disease progression in Chinese patients with advanced ALK-rearranged non-small-cell lung cancer. Oncotarget. 2017;8:41631–41640. doi: 10.18632/oncotarget.15892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pearce A, Bradley C, Hanly P, O'Neill C, Thomas AA, Molcho M, Sharp L. Projecting productivity losses for cancer-related mortality 2011–2030. Bmc Cancer. 2016;16:804. doi: 10.1186/s12885-016-2854-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Midha A, Dearden S, McCormack R. EGFR mutation incidence in non-small-cell lung cancer of adenocarcinoma histology: A systematic review and global map by ethnicity (mutMapII) Am J Cancer Res. 2015;5:2892–2911. [PMC free article] [PubMed] [Google Scholar]
- 4.Huang H, Du T, Xu G, Lai Y, Fan X, Chen X, Li W, Yue F, Li Q, Liu L, Li K. Matrine suppresses invasion of castration-resistant prostate cancer cells by downregulating MMP-2/9 via NF-κB signaling pathway. Int J Oncol. 2017;50:640–648. doi: 10.3892/ijo.2016.3805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Liu Z, Zhang Y, Tang Z, Xu J, Ma M, Pan S, Qiu C, Guan G, Wang J. Matrine attenuates cardiac fibrosis by affecting ATF6 signaling pathway in diabetic cardiomyopathy. Eur J Pharmacol. 2017;804:21–30. doi: 10.1016/j.ejphar.2017.03.061. [DOI] [PubMed] [Google Scholar]
- 6.Huang WC, Chan CC, Wu SJ, Chen LC, Shen JJ, Kuo ML, Chen MC, Liou CJ. Matrine attenuates allergic airway inflammation and eosinophil infiltration by suppressing eotaxin and Th2 cytokine production in asthmatic mice. J Ethnopharmacol. 2014;151:470–477. doi: 10.1016/j.jep.2013.10.065. [DOI] [PubMed] [Google Scholar]
- 7.Yong J, Wu X, Lu C. Anticancer advances of matrine and its derivatives. Curr Pharm Des. 2015;21:3673–3680. doi: 10.2174/1381612821666150122123748. [DOI] [PubMed] [Google Scholar]
- 8.Qian L, Liu Y, Xu Y, Ji W, Wu Q, Liu Y, Gao Q, Su C. Matrine derivative WM130 inhibits hepatocellular carcinoma by suppressing EGFR/ERK/MMP-2 and PTEN/AKT signaling pathways. Cancer Lett. 2015;368:126–134. doi: 10.1016/j.canlet.2015.07.035. [DOI] [PubMed] [Google Scholar]
- 9.Ma Y, Zou F, Xiong J, Wan W, Yin L, Li X, Bei Z, Yuan L, Meng S, Wang J, Song G. Effect of Matrine on HPAC cell migration by down-regulating the expression of MT1-MMP via Wnt signaling. Cancer Cell Int. 2015;15:59. doi: 10.1186/s12935-015-0210-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xie M, Yi X, Wang R, Wang L, He G, Zhu M, Qi C, Liu Y, Ye Y, Tan S, Tang A. 14-Thienyl methylene matrine (YYJ18), the derivative from matrine, induces apoptosis of human nasopharyngeal carcinoma cells by targeting MAPK and PI3K/Akt pathways in vitro. Cell Physiol Biochem. 2014;33:1475–1483. doi: 10.1159/000358712. [DOI] [PubMed] [Google Scholar]
- 11.Wei L, Yao Y, Zhao K, Huang Y, Zhou Y, Zhao L, Guo Q, Lu N. Oroxylin A inhibits invasion and migration through suppressing ERK/GSK-3β signaling in snail-expressing non-small-cell lung cancer cells. Mol Carcinog. 2016;55:2121–2134. doi: 10.1002/mc.22456. [DOI] [PubMed] [Google Scholar]
- 12.Ho MY, Liang SM, Hung SW, Liang CM. MIG-7 controls COX-2/PGE2-mediated lung cancer metastasis. Cancer Res. 2013;73:439–449. doi: 10.1158/0008-5472.CAN-12-2220. [DOI] [PubMed] [Google Scholar]
- 13.Chen J, Mei Q, Xu YC, Du J, Wei Y, Xu ZM. Effects of Matrine Injection on T-lymphocyte subsets of patients with malignant tumor after gamma knife radiosurgery. Zhong Xi Yi Jie He Xue Bao. 2006;4:78–79. doi: 10.3736/jcim20060121. (In Chinese) [DOI] [PubMed] [Google Scholar]
- 14.Yin H, Que R, Liu C, Ji W, Sun B, Lin X, Zhang Q, Zhao X, Peng Z, Zhang X, et al. Survivin-targeted drug screening platform identifies a matrine derivative WM-127 as a potential therapeutics against hepatocellular carcinoma. Cancer Lett. 2018;425:54–64. doi: 10.1016/j.canlet.2018.03.044. [DOI] [PubMed] [Google Scholar]
- 15.Wu L, Wang G, Liu S, Wei J, Zhang S, Li M, Zhou G, Wang L. Synthesis and biological evaluation of matrine derivatives containing benzo-α-pyrone structure as potent anti-lung cancer agents. Sci Rep. 2016;6:35918. doi: 10.1038/srep35918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhou N, Li J, Li T, Chen G, Zhang Z, Si Z. Matrine-induced apoptosis in Hep3B cells via the inhibition of MDM2. Mol Med Rep. 2017;15:442–450. doi: 10.3892/mmr.2016.5999. [DOI] [PubMed] [Google Scholar]
- 17.Wu L, Wang G, Wei J, Huang N, Zhang S, Yang F, Li M, Zhou G, Wang L. Matrine derivative YF-18 inhibits lung cancer cell proliferation and migration through down-regulating Skp2. Oncotarget. 2017;8:11729–11738. doi: 10.18632/oncotarget.14329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ali M, Wu Y, Ghosh D, Do BH, Chen K, Dawson MR, Fang N, Sulchek TA, El-Sayed MA. Nuclear membrane-targeted gold nanoparticles inhibit cancer cell migration and invasion. ACS Nano. 2017;11:3716–3726. doi: 10.1021/acsnano.6b08345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Labernadie A, Kato T, Brugués A, Serra-Picamal X, Derzsi S, Arwert E, Weston A, González-Tarragó V, Elosegui-Artola A, Albertazzi L, et al. A mechanically active heterotypic E-cadherin/N-cadherin adhesion enables fibroblasts to drive cancer cell invasion. Nat Cell Biol. 2017;19:224–237. doi: 10.1038/ncb3478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jacquemet G, Baghirov H, Georgiadou M, Sihto H, Peuhu E, Cettour-Janet P, He T, Perälä M, Kronqvist P, Joensuu H, Ivaska J. L-type calcium channels regulate filopodia stability and cancer cell invasion downstream of integrin signalling. Nat Commun. 2016;7:13297. doi: 10.1038/ncomms13297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu X, Zhou J, Cai D, Li M. Matrine inhibits the metastatic properties of human cervical cancer cells via downregulating the p38 signaling pathway. Oncol Rep. 2017;38:1312–1320. doi: 10.3892/or.2017.5787. [DOI] [PubMed] [Google Scholar]
- 22.Yang SF, Yang WE, Kuo WH, Chang HR, Chu SC, Hsieh YS. Antimetastatic potentials of flavones on oral cancer cell via an inhibition of matrix-degrading proteases. Arch Oral Biol. 2008;53:287–294. doi: 10.1016/j.archoralbio.2007.09.001. [DOI] [PubMed] [Google Scholar]
- 23.Zhou LE, Wang WJ, Bai JY, Cheng GF. Effects of ginkgolide B on arachidonic acid metabolizing enzymes and level of intracellular calcium in rat polymorphonuclear leukocytes. Yao Xue Xue Bao. 2001;36:92–95. (In Chinese) [PubMed] [Google Scholar]
- 24.Li H, Tan G, Jiang X, Qiao H, Pan S, Jiang H, Kanwar JR, Sun X. Therapeutic effects of matrine on primary and metastatic breast cancer. Am J Chin Med. 2010;38:1115–1130. doi: 10.1142/S0192415X10008512. [DOI] [PubMed] [Google Scholar]
- 25.Davis NM, Sokolosky M, Stadelman K, Abrams SL, Libra M, Candido S, Nicoletti F, Polesel J, Maestro R, D'Assoro A, et al. Deregulation of the EGFR/PI3K/PTEN/Akt/mTORC1 pathway in breast cancer: Possibilities for therapeutic intervention. Oncotarget. 2014;5:4603–4650. doi: 10.18632/oncotarget.2209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sagredo AI, Sagredo EA, Cappelli C, Báez P, Rodrigo AM, Blanco C, Tapia JC, Echeverria C, Cerda O, Stutzin A, et al. TRPM4 regulates Akt/GSK3-β activity and enhances β-catenin signaling and cell proliferation in prostate cancer cells. Mol Oncol. 2018;12:151–165. doi: 10.1002/1878-0261.12100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang PP, Wang PQ, Qiao CP, Zhang Q, Zhang JP, Chen F, Zhang X, Xie WF, Yuan ZL, Li ZS, Chen YX. Differentiation therapy of hepatocellular carcinoma by inhibiting the activity of AKT/GSK-3β/β-catenin axis and TGF-β induced EMT with sophocarpine. Cancer Lett. 2016;376:95–103. doi: 10.1016/j.canlet.2016.01.011. [DOI] [PubMed] [Google Scholar]
- 28.Wu J, Hu G, Dong Y, Ma R, Yu Z, Jiang S, Han Y, Yu K, Zhang S. Matrine induces Akt/mTOR signalling inhibition-mediated autophagy and apoptosis in acute myeloid leukaemia cells. J Cell Mol Med. 2017;21:1171–1181. doi: 10.1111/jcmm.13049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Niu H, Zhang Y, Wu B, Zhang Y, Jiang H, He P. Matrine induces the apoptosis of lung cancer cells through downregulation of inhibitor of apoptosis proteins and the Akt signaling pathway. Oncol Rep. 2014;32:1087–1093. doi: 10.3892/or.2014.3273. [DOI] [PubMed] [Google Scholar]
- 30.Hseu YC, Thiyagarajan V, Tsou HT, Lin KY, Chen HJ, Lin CM, Liao JW, Yang HL. In vitro and in vivo anti-tumor activity of CoQ0 against melanoma cells: inhibition of metastasis and induction of cell-cycle arrest and apoptosis through modulation of Wnt/β-catenin signaling pathways. Oncotarget. 2016;7:22409–22426. doi: 10.18632/oncotarget.7983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xu G, Yu H, Shi X, Sun L, Zhou Q, Zheng D, Shi H, Li N, Zhang X, Shao G. Cisplatin sensitivity is enhanced in non-small cell lung cancer cells by regulating epithelial-mesenchymal transition through inhibition of eukaryotic translation initiation factor 5A2. BMC Pulm Med. 2014;14:174. doi: 10.1186/1471-2466-14-174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chen L, Li YC, Wu L, Yu GT, Zhang WF, Huang CF, Sun ZJ. TRAF6 regulates tumour metastasis through EMT and CSC phenotypes in head and neck squamous cell carcinoma. J Cell Mol Med. 2018;22:1337–1349. doi: 10.1111/jcmm.13439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Liu L, Zhou XM, Yang FF, Miao Y, Yin Y, Hu XJ, Hou G, Wang QY, Kang J. TRIM22 confers poor prognosis and promotes epithelial-mesenchymal transition through regulation of AKT/GSK3β/β-catenin signaling in non-small cell lung cancer. Oncotarget. 2017;8:62069–62080. doi: 10.18632/oncotarget.18911. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.