Abstract
Lung cancer is the leading cause of global cancer-associated mortality, therefore it is important to reveal the molecular mechanisms of lung cancer progression and to develop novel therapeutic targets. The results of the present study identified that manganese-12 acetate (Mn12Ac) was able to significantly inhibit the migration and invasion of A549 cells. Western blotting demonstrated that treatment with Mn12Ac was able to upregulate E-cadherin, and downregulate N-cadherin and vimentin. It was also identified by a quantitative polymerase chain reaction analysis that Mn12Ac was able to reduce the mRNA expression levels of EMT-associated transcription factors Snail, Slug, Twist-related protein 1 and zinc finger E-box-binding homeobox 1. It was also demonstrated that Mn12Ac was able to reduce the expression levels of Wnt and β-catenin proteins, and suppress the phosphorylation of phosphoinositide 3-kinase (PI3K) and AKT in A549 cells. Notably, it was revealed that Mn12Ac was able to decrease the mRNA and protein expression levels of programmed death ligand-1. Taken together, the results suggested that Mn12Ac is able to inhibit cell migration, invasion and EMT in lung cancer cells by regulating the Wnt/β-catenin and PI3K/AKT signaling pathways.
Keywords: manganese-12 acetate, migration, epithelial-mesenchymal transition, Wnt, phosphoinositide 3-kinase
Introduction
Lung cancer is the leading cause of cancer mortality, and there were ~1,800,000 incident lung cancer cases in 2012 globally, representing ~13% of the total global cancer incidence (1). Lung cancer primarily consists of two histological types: Small cell lung cancer and non-small-cell lung cancer (NSCLC), which account for 15 and 85% of total cases of lung cancer, respectively (2). Lung adenocarcinoma (LAC), which is the most widespread histological type of NSCLC, causes >500,000 mortalities every year worldwide (3). Therefore, it is important to reveal the molecular mechanisms of LAC progression and development, and to develop novel therapeutic targets for patients with LAC.
Epithelial-mesenchymal transition (EMT) is an important process that is characterized by the loss of cell polarity and cell-cell contact, and is activated in tumor metastasis and resistance to chemoradiotherapy (4,5). Activation of EMT is associated with the altered expression of numerous genes, including the downregulation of epithelial markers such as occluden-1 and E-cadherin, and the upregulation of mesenchymal markers including N-cadherin and vimentin (6). A number of studies have demonstrated that EMT is a critical process in tumor invasion and metastasis in NSCLC (7–9).
Manganese-12 acetate (Mn12Ac) is a magnetically bistable molecule exhibiting a combination of strong magnetic anisotropy and high spin properties (10,11). However, there have been no studies concerning the association between Mn12Ac and disease therapy, particularly in cancer. In the present study, the biological function of Mn12Ac in lung cancer treatment was investigated.
Materials and methods
Cell culture
The human lung cancer A549 cell line (American Type Culture Collection, Manassas, VA, USA) was cultured in RPMI-1640 medium (Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA) with 10% fetal bovine serum (GE Healthcare, Chicago, IL, USA), 100 U/ml penicillin and 100 g/ml streptomycin at 37°C in a humidified incubator containing 5% CO2. Cells were treated with 100 nM Mn12Ac (dissolved in H2O, and H2O was used as control) and cultured at 37°C for 48 h, and subsequent experiments were performed.
Total RNA extraction and quantitative polymerase chain reaction (qPCR) assay
Total RNA was isolated from cancer cells using the RNeasy Mini kit according to the protocol of the manufacturer (Qiagen GmbH, Hilden, Germany) and used for the qPCR assay. Three independent experimental repeats of qPCR were performed.
qPCR was used to detect the mRNA expression of E-cadherin, N-cadherin, vimentin, zinc finger protein SNAI1 (Snail), zinc finger protein SNAI2 (Slug), twist-related protein 1 (Twist1), zinc finger E-box-binding homeobox 1 (ZEB1) and programmed death-ligand 1 (PD-L1). The PCR reactions were performed in a total volume of 20 µl, including 10 µl 2× Power SYBR® Green PCR Master Mix (Applied Biosystems; Thermo Fisher Scientific, Inc., Waltham, MA, USA), 2 µl cDNA (5 ng/µl) and 1 µl primer mix (10 µM each). PCR amplification and detection were performed in a LightCycler 480 II (Roche Applied Science, Penzberg, Germany) as follows: Initial denaturation at 95°C for 10 min, then 40 cycles of denaturation at 95°C for 15 sec and annealing and elongation at 60°C for 1 min. The relative gene expression was calculated using the comparative Cq method. The gene expression of the target gene was normalized to an endogenous reference gene (GAPDH), and data relative to the calibrator were calculated using the formula 2−ΔΔCq. ΔCq was calculated by subtracting the average GAPDH Cq value from the average Cq value of the gene of interest (12). The ratio defined the level of relative expression of the target gene to that of GAPDH. The primers were as follows: E-cadherin forward primer, 5′-GAACGCATTGCCACATACAC-3′; reverse primer, 5′-GAATTCGGGCTTGTTGTCAT-3′; N-cadherin forward primer, 5′-GTGCCATTAGCCAAGGGAATTCAGC-3′, reverse primer, 5′-GCGTTCCTGTTCCACTCATAGGAGG-3′; vimentin forward primer, 5′-TGAGTACCGGAGACAGGTCGAG-3′, reverse primer, 5′-TAGCAGCTTCAACGCAAAGTTC-3′; Snail forward primer, 5′-ACCACTATGCCGCGCTCTT-3′, reverse primer, 5′-GGTCGTAGGGCTGCTGGAA-3′; Slug forward primer, 5′-GCGCATGCTCCATTGTCTTAC-3′, reverse primer, 5′-AGGCACTTGGAAGGGGTATTG-3′; Twist1 forward primer, 5′-AGAAGTCTGCGGGCTGTGGCG-3′, reverse primer, 5′-GAGGGCAGCGTGGGGATGATC-3′; ZEB1 forward primer, 5′-CTACTCAACTACGGTCAGCCC-3′, reverse primer, 5′-TTGGGCGGTGTAGAATCAGAG-3′; programmed death ligand 1 (PD-L1) forward primer, 5′-GAACTACCTCTGGCACATCCT-3′, reverse primer, 5′-CACATCCATCATTCTCCCTTT-3′; GAPDH forward primer, 5′-AAATCCCATCACCATCTTCCAG-3′ and reverse primer, 5′-GAGTCCTTCCACGATACCAAAGTTG-3′.
Western blotting
The cells were lysed in the lysis buffer (20 mM Tris, 2 mM EDTA, 50 mM 2-mecaptoethanol, 10% glycerol, pH 7.4). The homogenates were placed on ice for 30 min and centrifuged at 12,000 × g for 15 min at 4°C. Subsequently, the protein concentration of the lysates was determined using a Protein Assay kit (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Equal amounts (15 µg) of total proteins were loaded onto a 10% PAGE, electrophoretically transferred to polyvinylidene difluoride membrane, and then blocked with 10% non-fat milk for 2 h at room temperature. The membranes were incubated with specific primary antibodies overnight at 4°C and probed with corresponding secondary antibodies (goat anti-rabbit IgG-HRP, sc-2004, 1:5,000 and goat anti-mouse IgG-HRP, sc-2005, 1:5,000, Santa Cruz Biotechnology, Inc., Dallas, TX, USA) for 1 h at room temperature. The protein bands were visualized using Enhanced Chemiluminescence Blotting Detection reagents (Applygen Technologies, Inc., Beijing, China). The primary antibodies used are as follows: E-cadherin (1:1,000; sc-71009; Santa Cruz Biotechnology, Inc.), N-cadherin (1:1,000; sc-59987; Santa Cruz Biotechnology, Inc.), vimentin (1:1,000; sc-80975; Santa Cruz Biotechnology, Inc.), Wnt1 (1:1,000; sc-6266; Santa Cruz Biotechnology, Inc.), β-catenin (1:1,000; sc-65480; Santa Cruz Biotechnology, Inc.), phosphorylated-phosphoinositide 3-kinase (p-PI3K; 1:1,000; 4228; Cell Signaling Technology, Inc., Danvers, MA, USA), PI3K (1:1,000; sc-376112; Santa Cruz Biotechnology, Inc.), p-AKT (1:1,000; sc-271964; Santa Cruz Biotechnology, Inc.), AKT (1:1,000; sc-1619; Santa Cruz Biotechnology, Inc.), PD-L1 (1:1,000; 13684; Cell Signaling Technology, Inc.) and β-actin (1:5,000; sc-517582; Santa Cruz Biotechnology, Inc.).
Transwell assay
For the Transwell invasion assay, 60 µl Matrigel® was diluted with precooled serum-free medium in a ratio of 1:4, and was added to the bottom of the Transwell chamber and incubated for 1 h at 37°C. A total of 2×105 cells (Mn12Ac treated cells and the H2O treated control cells) were seeded into the upper chambers (24-well insert; pore size 8 µm), while medium supplemented with 10% FBS (600 µl) was placed in the lower chamber. Following incubation at 37°C for 24 h, the cells on the upper chamber side of the inserts were removed gently with a cotton swab. The inserts were then fixed with 4% methanol for 15 min at room temperature and stained with 0.1% crystal violet for 15 min at room temperature. The mean number of migratory cells was calculated by counting five randomly selected fields each time under the light microscope at ×200 magnification. The experiments were repeated three times.
For the Transwell migration assay, the remaining protocol was the same as the Transwell invasion assay, except that the inserts were not pre-coated with Matrigel®.
Statistical analysis
All data were analyzed by unpaired Student's t-test or one-way analysis of variance with post hoc comparisons with Tukey's honest significant difference test for multiple group comparisons using SPSS version 20.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism (version 5.0; GraphPad, Inc., La Jolla, CA, USA). P<0.05 was considered to indicate a statistically significant difference.
Results
Mn12Ac is able to significantly inhibit the migration and invasion of A549 lung cancer cells
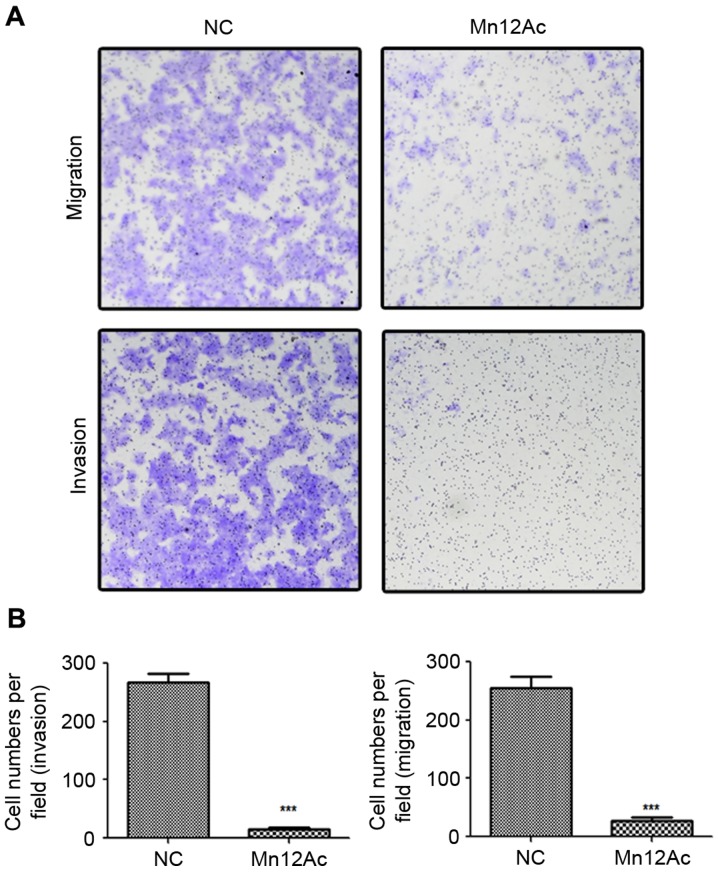
In order to investigate the anti-tumor activity of Mn12Ac, Transwell migration and invasion assays were performed to analyze the effects of Mn12Ac on lung cancer cell metastasis. The results demonstrated that Mn12Ac was able to significantly inhibit the migration and invasion of A549 cells compared with the negative control (Fig. 1A and B).
Figure 1.
Mn12Ac inhibits the migration and invasion of A549 lung cancer cells. (A) Transwell assay analysis of the migratory and invasive abilities of lung cancer cells with a light microscope at ×200 magnification. (B) Statistical analysis of the results from the Transwell migration and invasion assays. ***P<0.001 vs. negative group. Mn12Ac, manganese-12 acetate; NC, negative control.
Mn12Ac inhibits EMT in A549 lung cancer cells
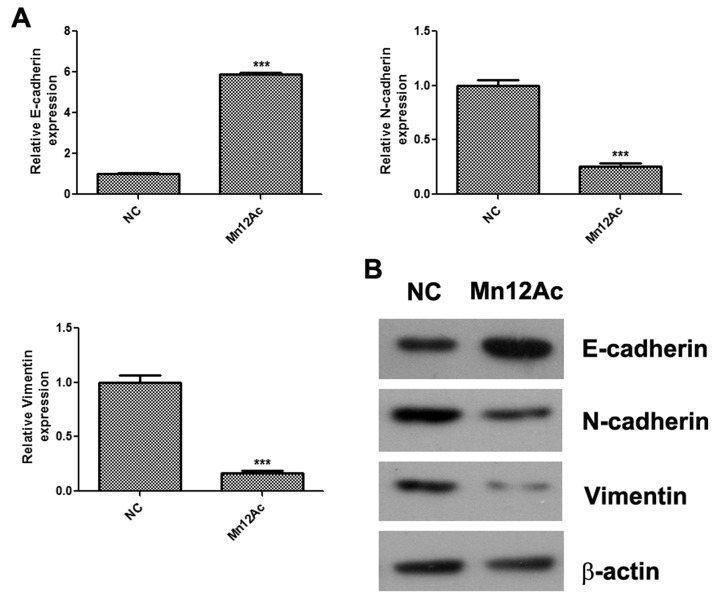
EMT is activated during tumor metastasis and serves an important role in the process (13); therefore whether Mn12Ac affected the EMT process in A549 lung cancer cells was investigated. The qPCR assay results indicated that Mn12Ac was able to significantly upregulate the epithelial marker E-cadherin, and downregulate the mesenchymal markers N-cadherin and vimentin in A549 cells (Fig. 2A). In addition, it was indicated by western blotting that Mn12Ac was also able to markedly upregulate the protein expression of E-cadherin and reduce the protein expression levels of N-cadherin and vimentin (Fig. 2B).
Figure 2.
Mn12Ac inhibits EMT in lung cancer cells. (A) mRNA expressions of EMT markers as detected using quantitative polymerase chain reaction analysis. (B) Protein expression of EMT markers as detected using western blotting assay. ***P<0.001 vs. negative control. Mn12Ac, manganese-12 acetate; NC, negative control; EMT, epithelial mesenchymal transition; E-cadherin, epithelial cadherin; N-cadherin, neural cadherin.
Mn12Ac significantly inhibits the expressions of EMT-associated transcription factors in A549 lung cancer cells
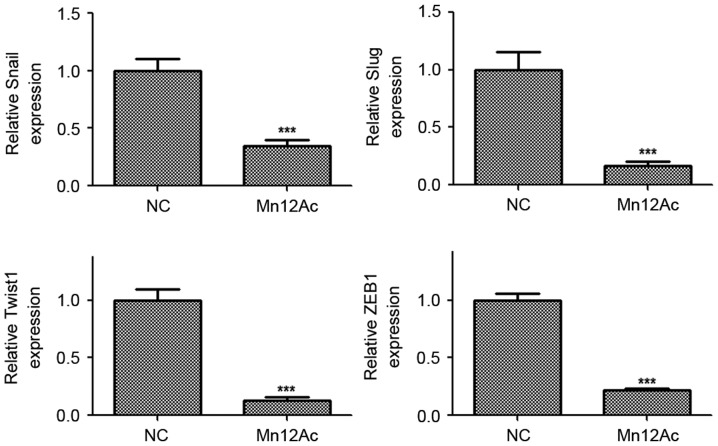
Snail, slug, twist1 and ZEB1 are EMT-associated transcription factors in cancer (14–16). Therefore, whether Mn12Ac regulated EMT via affecting these transcription factors was examined. Using qPCR, it was revealed that Mn12Ac was able to significantly decrease the mRNA expression levels of Snail, Slug, Twist1 and ZEB1 in A549 cancer cells (Fig. 3). These results suggested that Mn12Ac inhibited EMT in lung cancer cells by downregulating EMT-inducing transcription factors.
Figure 3.
Mn12Ac decreases the expression levels of EMT-associated transcription factors. ***P<0.001 vs. negative group. Mn12Ac, manganese-12 acetate; NC, negative control; EMT, epithelial mesenchymal transition; Twist1, twist-related protein 1; ZEB1, zinc finger E-box-binding homeobox 1.
Mn12Ac inhibits the Wnt/β-catenin and PI3K/AKT signaling pathways
The Wnt/β-catenin and PI3K/AKT signaling pathways serve important roles in the regulation of EMT (17–19), and it was hypothesized that Mn12Ac may inhibit EMT in lung cancer cells by regulating these two pathways. Using western blotting assay, it was identified that Mn12Ac was able to markedly reduce the expression levels of Wnt1 and β-catenin and inhibit the phosphorylation of PI3K and AKT in A549 lung cancer cells (Fig. 4).
Figure 4.
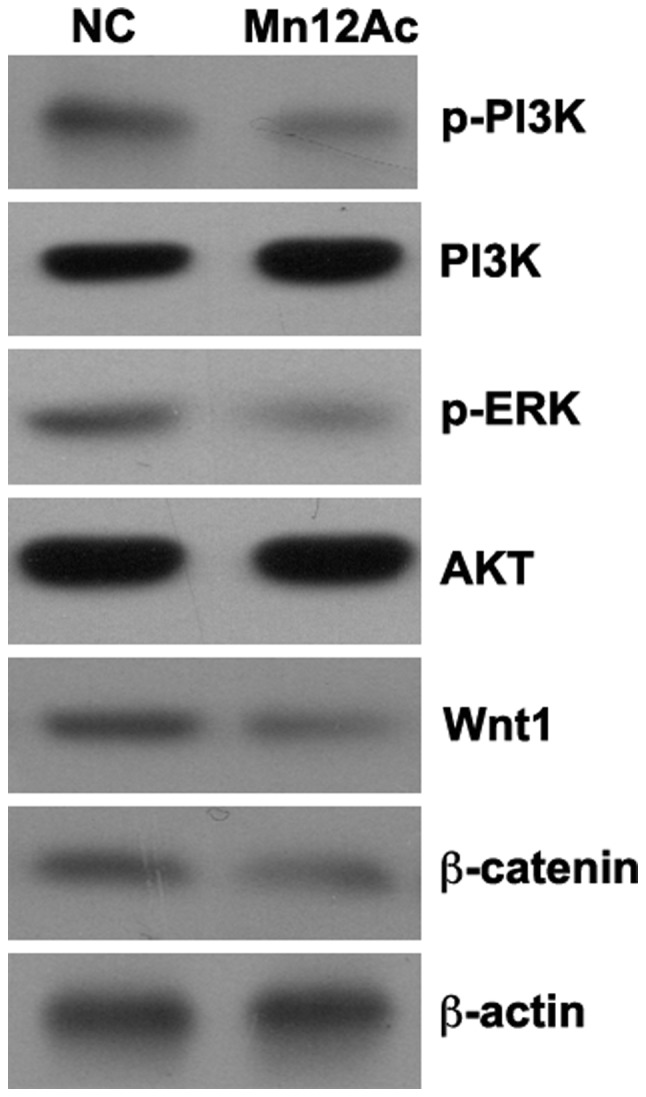
Mn12Ac inhibits the PI3K/AKT and Wnt/β-catenin signaling pathways. The expressions of Wnt1 and β-catenin and the phosphorylation of PI3K and AKT were assessed by western blot analysis. Mn12Ac, manganese-12 acetate; NC, negative control.
Mn12Ac decreases the mRNA and protein expression levels of PD-L1
PD-1/PD-L1-targeted immunotherapy has emerged as a promising therapeutic strategy for lung cancer, and PD-L1 expression was associated with EMT transition in lung cancer (20). Therefore, the present study investigated whether Mn12Ac affected PD-L1 in lung cancer. Notably, the results indicated that Mn12Ac was able to reduce the mRNA and protein expression levels of PD-L1 in A549 lung cancer cells by qPCR and western blotting assay, respectively (Fig. 5).
Figure 5.
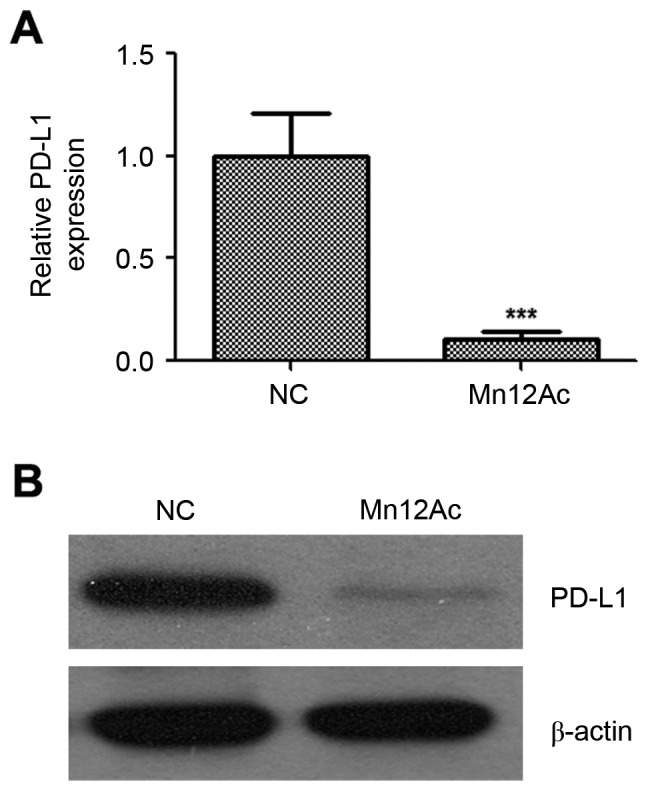
Mn12Ac decreases the PD-L1 expression in A549 lung cancer cells. (A) The mRNA expression of PD-L1 was assessed by quantitative polymerase chain reaction analysis. (B) Protein expression of PD-L1 was assessed by western blotting assay. ***P<0.001 vs. negative control. Mn12Ac, manganese-12 acetate; NC, negative control; PD-L1, programmed death-ligand 1.
Discussion
Human lung cancer is the most common cause of global cancer-associated mortality (21). Lung cancer develops through a multistep process with oncogenic mutations in lung epithelial cells (22). It is important to reveal the molecular mechanisms of LAC progression and development, and to develop novel therapeutic targets for patients with LAC.
Metastasis is the primary cause of cancer-associated mortality, and the epithelial EMT process is critical for epithelial cell cancer progression. The results of the present study demonstrated that Mn12Ac was able to significantly inhibit the migration and invasion of A549 lung cancer cells. These results suggested that Mn12Ac suppressed the EMT process in lung cancer cells. EMT is a morphological change in cells from an epithelial form to a fibroblast-like mesenchymal form. In addition to these morphological changes, key biomarkers that involved in the steps of the process include cell adhesion molecules such as E-cadherin, N-cadherin, and transcription factors Snail, Slug, Twist1 and ZEB1 (5,23). The expression levels of these EMT molecules were demonstrated to be associated with drug resistance, and the inhibition of EMT may interfere with tumor progression and drug resistance (24). The results of the present study suggested that Mn12Ac may inhibit the EMT process by downregulating the transcription factors Snail, Slug, Twist1 and ZEB1. In addition, the results of the present study suggest that Mn12Ac may promote drug sensitivity in cancer therapy.
The PI3K/AKT signaling pathway serves an important role in the EMT process and tumor metastasis. Shao et al (25) suggested that irisin may inhibit EMT and the invasion of lung cancer cells by regulating the PI3K/AKT/Snail signaling pathway. Tumor protein D52 inhibited the growth and metastasis of renal cell carcinoma cells by downregulating the levels of p-PI3K and p-Akt (26). Curcumin was also reported to suppress EMT in renal tubular epithelial cells through the inhibition of the Akt/mTOR signaling pathway (19). The Wnt/β-catenin signaling pathway was also associated with the EMT process. CDGSH iron sulfur domain 2 may promote proliferation and EMT in pancreatic cancer cells by upregulating the Wnt/β-catenin signaling pathway (27). Wang et al (28) reported that calpain small subunit 1 promoted EMT in human melanoma cells through the activation of the Wnt/β-catenin signaling pathway (28). Zuo et al (29) observed that long non-coding RNA protein SPRY4 intronic transcript 1 was able to modulate trophoblast cell invasion, migration and EMT processes by regulating the Wnt/β-catenin signaling pathway (29). The results of the present study revealed that Mn12Ac may inhibit the Wnt/β-catenin and PI3K/AKT signaling pathways in lung cancer cells.
PD-L1 in tumor cells is known to promote immune escape of cancer cells by interacting with programmed cell death protein 1 in tumor-infiltrating immune cells (30). Immunotherapy targeting these molecules is emerging as a novel strategy for the treatment of different types of cancer. Notably, the results of the present study indicated that Mn12Ac was able to markedly reduce the expression of PD-L1 at the level of mRNA and protein in lung cancer cells.
Taken together, the results of the present study revealed that Mn12Ac may significantly inhibit migration, invasion and EMT in lung cancer cells by suppressing the Wnt/β-catenin and PI3K/AKT signaling pathways, and Mn12Ac may also reduce the expression of PD-L1. In the future, additional studies should focus on the potential clinical applications of Mn12Ac.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Key Project of Kunming Technology Program (grant no. 2015-1-S-00877, 2015-1-S-00576).
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author.
Author's contributions
CZ, WQ and JH designed the work that led to the submission. HJ and XX performed RT-qPCR analysis. LP, ZW, TZ, JX, LL and LD conducted western blot analysis. CZ and JH drafted the manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Herbst RS, Heymach JV, Lippman SM. Lung cancer. N Engl J Med. 2008;359:1367–1380. doi: 10.1056/NEJMra0802714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Imielinski M, Berger AH, Hammerman PS, Hernandez B, Pugh TJ, Hodis E, Cho J, Suh J, Capelletti M, Sivachenko A, et al. Mapping the hallmarks of lung adenocarcinoma with massively parallel sequencing. Cell. 2012;150:1107–1120. doi: 10.1016/j.cell.2012.08.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Smith BN, Bhowmick NA. Role of EMT in metastasis and therapy resistance. J Clin Med. 2016;5 doi: 10.3390/jcm5020017. pii: E17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15:178–196. doi: 10.1038/nrm3758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Seton-Rogers S. Epithelial-mesenchymal transition: Untangling EMT's functions. Nat Rev Cancer. 2016;16:1. doi: 10.1038/nrc.2015.6. [DOI] [PubMed] [Google Scholar]
- 7.Wang DX, Zou YJ, Zhuang XB, Chen SX, Lin Y, Li WL, Lin JJ, Lin ZQ. Sulforaphane suppresses EMT and metastasis in human lung cancer through miR-616-5p-mediated GSK3β/β-catenin signaling pathways. Acta Pharmacol Sin. 2017;38:241–251. doi: 10.1038/aps.2016.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ye Z, Yin S, Su Z, Bai M, Zhang H, Hei Z, Cai S. Downregulation of miR-101 contributes to epithelial-mesenchymal transition in cisplatin resistance of NSCLC cells by targeting ROCK2. Oncotarget. 2016;7:37524–37535. doi: 10.18632/oncotarget.6852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen S, Jiao S, Jia Y, Li Y. Effects of targeted silencing of FOXC1 gene on proliferation and in vitro migration of human non-small-cell lung carcinoma cells. Am J Transl Res. 2016;8:3309–3318. [PMC free article] [PubMed] [Google Scholar]
- 10.Rakvin B, Zilić D, North JM, Dalal NS. Probing magnetic fields on crystals of the nanomagnet Mn12-acetate by electron paramagnetic resonance. J Magn Reson. 2003;165:260–264. doi: 10.1016/j.jmr.2003.08.010. [DOI] [PubMed] [Google Scholar]
- 11.Morello A, Bakharev ON, Brom HB, Sessoli R, de Jongh LJ. Nuclear spin dynamics in the quantum regime of a single-molecule magnet. Phys Rev Lett. 2004;93:197202. doi: 10.1103/PhysRevLett.93.197202. [DOI] [PubMed] [Google Scholar]
- 12.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 13.Guo C, Zhao D, Zhang Q, Liu S, Sun MZ. miR-429 suppresses tumor migration and invasion by targeting CRKL in hepatocellular carcinoma via inhibiting Raf/MEK/ERK pathway and epithelial-mesenchymal transition. Sci Rep. 2018;8:2375. doi: 10.1038/s41598-018-20258-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Amoroso MR, Matassa DS, Agliarulo I, Avolio R, Lu H, Sisinni L, Lettini G, Gabra H, Landriscina M, Esposito F. TRAP1 downregulation in human ovarian cancer enhances invasion and epithelial-mesenchymal transition. Cell Death Dis. 2016;7:e2522. doi: 10.1038/cddis.2016.400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liang W, Lai Y, Zhu M, Huang S, Feng W, Gu X. Combretastatin A4 regulates proliferation, migration, invasion, and apoptosis of thyroid cancer cells via PI3K/Akt signaling pathway. Med Sci Monit. 2016;22:4911–4917. doi: 10.12659/MSM.898545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hammers H, Fu C, Gerber S, Berg SVD, Steenwinkel F, Moriarty W, Keizman D, Kachhap S, Carducci MA. Epithelial-mesenchymal transition: A mechanism of resistance to VEGF pathway inhibition in genitourinary cancers. J Clin Oncol. 2012;30(5_suppl):S390. doi: 10.1200/jco.2012.30.5_suppl.390. [DOI] [Google Scholar]
- 17.Liu MH, Fu WJ, Cui YH, Guo QN, Zhou Y. Downregulation of Semaphorin-3F is associated with poor prognostic significance in osteosarcoma patients. Am J Cancer Res. 2016;6:2252–2262. [PMC free article] [PubMed] [Google Scholar]
- 18.Liu B, Pan CF, He ZC, Wang J, Wang PL, Ma T, Xia Y, Chen YJ. Long noncoding RNA-LET suppresses tumor growth and EMT in lung adenocarcinoma. Biomed Res Int. 2016;2016:4693471. doi: 10.1155/2016/4693471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhu FQ, Chen MJ, Zhu M, Zhao RS, Qiu W, Xu X, Liu H, Zhao HW, Yu RJ, Wu XF, et al. Curcumin suppresses epithelial-mesenchymal transition of renal tubular epithelial cells through the inhibition of Akt/mTOR pathway. Biol Pharm Bull. 2017;40:17–24. doi: 10.1248/bpb.b16-00364. [DOI] [PubMed] [Google Scholar]
- 20.Kim S, Koh J, Kim MY, Kwon D, Go H, Kim YA, Jeon YK, Chung DH. PD-L1 expression is associated with epithelial-to-mesenchymal transition in adenocarcinoma of the lung. Hum Pathol. 2016;58:7–14. doi: 10.1016/j.humpath.2016.07.007. [DOI] [PubMed] [Google Scholar]
- 21.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62:10–29. doi: 10.3322/caac.20138. [DOI] [PubMed] [Google Scholar]
- 22.Larsen JE, Minna JD. Molecular biology of lung cancer: Clinical implications. Clin Chest Med. 2011;32:703–740. doi: 10.1016/j.ccm.2011.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Arai K, Eguchi T, Rahman MM, Sakamoto R, Masuda N, Nakatsura T, Calderwood SK, Kozaki K, Itoh M. A novel high-throughput 3D screening system for EMT inhibitors: A pilot screening discovered the EMT inhibitory activity of CDK2 inhibitor SU9516. PLoS One. 2016;11:e0162394. doi: 10.1371/journal.pone.0162394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dopeso H, Jiao HK, Cuesta AM, Henze AT, Jurida L, Kracht M, Acker-Palmer A, Garvalov BK, Acker T. PHD3 controls lung cancer metastasis and resistance to EGFR inhibitors through TGFα. Cancer Res. 2018;78:1805–1819. doi: 10.1158/0008-5472.CAN-17-1346. [DOI] [PubMed] [Google Scholar]
- 25.Shao L, Li H, Chen J, Song H, Zhang Y, Wu F, Wang W, Zhang W, Wang F, Li H, Tang D. Irisin suppresses the migration, proliferation, and invasion of lung cancer cells via inhibition of epithelial-to-mesenchymal transition. Biochem Biophys Res Commun. 2017;485:598–605. doi: 10.1016/j.bbrc.2016.12.084. [DOI] [PubMed] [Google Scholar]
- 26.Zhao Z, Liu H, Hou J, Li T, Du X, Zhao X, Xu W, Xu W, Chang J. Tumor protein D52 (TPD52) inhibits growth and metastasis in renal cell carcinoma cells through the PI3K/Akt signaling pathway. Oncol Res. 2017;25:773–779. doi: 10.3727/096504016X14774889687280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yang Y, Bai YS, Wang Q. CDGSH iron sulfur domain 2 activates proliferation and EMT of pancreatic cancer cells via Wnt/β-catenin pathway and has prognostic value in human pancreatic cancer. Oncol Res. 2017;25:605–615. doi: 10.3727/096504016X14767450526417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang E, Wang D, Li B, Ma H, Wang C, Guan L, Zhang H, Yi L, Li S. Capn4 promotes epithelial-mesenchymal transition in human melanoma cells through activation of the Wnt/β-catenin pathway. Oncol Rep. 2017;37:379–387. doi: 10.3892/or.2016.5247. [DOI] [PubMed] [Google Scholar]
- 29.Zuo Q, Huang S, Zou Y, Xu Y, Jiang Z, Zou S, Xu H, Sun L. The Lnc RNA SPRY4-IT1 modulates trophoblast cell invasion and migration by affecting the epithelial-mesenchymal transition. Sci Rep. 2016;6:37183. doi: 10.1038/srep37183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chatterjee J, Dai W, Aziz NHA, Teo PY, Wahba J, Phelps DL, Maine CJ, Whilding LM, Dina R, Trevisan G, et al. Clinical use of programmed cell death-1 and its ligand expression as discriminatory and predictive markers in ovarian cancer. Clin Cancer Res. 2017;23:3453–3460. doi: 10.1158/1078-0432.CCR-16-2366. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the present study are available from the corresponding author.