To the Editor:
We read with interest the recent publication by Amsellem and colleagues in the Journal, titled “Roles for the CX3CL1/CX3CR1 and CCL2/CCR2 Chemokine Systems in Hypoxic Pulmonary Hypertension” (1). The authors demonstrated that CX3CR1 deficiency is protective against hypoxia-induced pulmonary hypertension (PH) by modulating monocyte recruitment, macrophage polarization, and pulmonary artery smooth muscle cell proliferation. The authors make a persuasive argument for CX3CR1 as a target for PH therapy. We completely concur and would like to add evidence that brain microglial cells and neuroinflammation are also involved. Neuroinflammation, the inflammatory response in the central nervous system, results from acute or chronic responses to injury by resident innate immune cells, and microglia are the principal component (2).
CX3CR1 is primarily expressed by microglial cells in the brain and is also present on resident monocytes in the periphery (2, 3). The CX3CL1/CX3CR1 axis has emerged as a key signaling pathway in the control of synaptic activity influencing short- and long-term neural plasticity (2). As a result, its importance in many neuroinflammatory diseases, including hypertension, is well appreciated (2–6).
Hypoxia is a known stimulus of sympathetic activity and is involved in microglia activation (7, 8). Furthermore, accumulating evidence links the activated sympathetic nervous system to PH. This includes increased sympathetic nerve trafficking, hyperoxia-mediated beneficial effects on muscle sympathetic nerve activity in patients with PH, and alleviation of right heart and PH pathophysiology by adrenergic antagonists in both human and animal models (7). In view of these observations and the study by Amsellem and colleagues (1), we explored the role of microglial cells in hypoxia-induced PH in wild-type (WT) and CX3CR1 knockout (CX3CR1GFP/GFP) mice. We hypothesized that microglial cells in autonomic brain regions, particularly in the paraventricular nucleus (PVN) of the hypothalamus, and enhanced sympathetic activity would be augmented in hypoxia-induced PH in WT mice. We further hypothesized that a lack of PH in response to hypoxia in CX3CR1GFP/GFP mice would be correlated with microglial numbers in the PVN and attenuation of sympathetic activity.
WT C57BL/6J and CX3CR1-deficient (CX3CR1GFP/GFP) mice were exposed to chronic hypoxia (10% O2) or normoxia for 3 weeks in a ventilated chamber (n = 5–8/group). A Millar pressure catheter (SPR-671; Millar Instruments) coupled to a signal transducer unit (PowerLab; ADInstruments) was used to measure cardiac and pulmonary hemodynamics. An ionized calcium-binding adaptor molecule 1 (Iba1)-specific antibody (019–19741; Wako Chemicals USA, Inc.) was used for immunohistochemical characterization and quantitation of microglial cells in 40 μm postfixed brain sections (4).
Chronic hypoxia exposure resulted in a significant increase (77%) in right ventricular (RV) systolic pressure (RVSP) in WT mice (Table 1). Increases in RV hypertrophy (RVH) and RV end diastolic pressure, and changes in contractility (+dP/dt, −dP/dt) were also observed in hypoxia-exposed WT mice (Table 1). In addition, a fivefold increase in the sympathetic/vagal balance (low frequency/high frequency ratio) was observed, suggesting an increase in sympathetic activity by hypoxia treatment in WT mice. In contrast, no significant changes in all of the above parameters were observed in CX3CR1GFP/GFP mice exposed to hypoxia. This lack of response to hypoxia by the RVSP and RVH was consistent with observations of Amsellem and colleagues (1).
Table 1.
Hemodynamic Parameters of Wild-Type (C57BL/6J) and CX3CR1-Deficient (CX3CR1GFP/GFP) Mice Exposed to Control Normoxia and Hypoxia
| WT-N | WT-H | CX3CR1GFP/GFP-N | CX3CR1GFP/GFP-H | P value | |
|---|---|---|---|---|---|
| RVSP, mm Hg | 19.5 ± 2 | 33.7 ± 2* | 23.6 ± 2 | 23.8 ± 3 | 0.0029 |
| RV/LV+S, g | 0.1390 ± 0.09 | 0.1923 ± 0.008† | 0.1292 ± 0.003 | 0.1615 ± 0.01 | 0.0019 |
| +dP/dt, mm Hg/s | 1,594 ± 120 | 2,254 ± 164* | 1,678 ± 93 | 1,823 ± 128 | 0.0082 |
| −dP/dt, mm Hg/s | −1,420 ± 40 | −2,184 ± 220* | −1,654 ± 65 | −1,350 ± 287 | 0.0106 |
| RVEDP, mm Hg | 1.91 ± 0.3 | 5.2 ± 0.8* | 2.4 ± 1 | 2.4 ± 0.3 | 0.0015 |
| LF/HF, % | 0.23 ± 0.08 | 1.19 ± 0.08* | 0.19 ± 0.03 | 0.29 ± 0.08 | <0.001 |
Definition of abbreviations: H = hypoxia; HF = high frequency; LF = low frequency; N = normoxia; RVEDP = right ventricular end diastolic pressure; RV/LV+S = right ventricular hypertrophy; RVSP = right ventricular systolic pressure; WT = wild-type.
Chronic hypoxia (10% O2) induced pulmonary hypertension in WT mice, but not in CX3CR1GFP/GFP mice, as demonstrated by RVSP, RV/LV+S, maximum (+dP/dt) and minimum (−dP/dt) dP/dt, and RVEDP measurements. Data are presented as mean ± SEM.
Significant differences were determined by one-way ANOVA followed by Newman-Keuls post hoc tests (n = 5–8/each group).
P < 0.05 versus WT-N; CX3CR1GFP/GFP -N and CX3CR1GFP/GFP-H.
P < 0.05 versus WT-N; CX3CR1GFP/GFP-N.
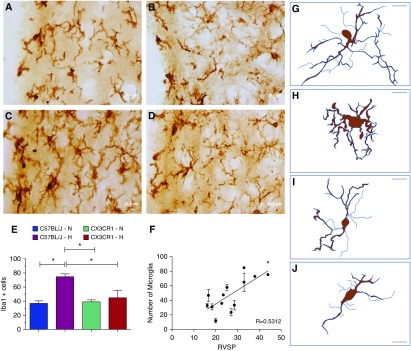
Next, we evaluated microglial cells in the PVN of control and hypoxia-treated WT and CX3CR1GFP/GFP mice. Only cells with a well-defined cell body were included in the analysis. The selection of the PVN as the representative autonomic brain region for this study was based on evidence that PVN microglia are involved in sympathetic activation, blood pressure control, and hypertension (9). Microglia cells are characterized as “resting” microglia when they exhibit a small cell body with thin and highly ramified branches extending in all directions. These ramified microglia can assume an amoeboid form and/or exhibit a larger cell body with well-defined and shorter branches, a process that is described as activation (10, 11). Representative images of Iba1 staining of microglia cells in WT mice exposed to hypoxia demonstrate a morphology consistent with activation (Figures 1C and 1H), in contrast to the “resting” microglia observed in WT-normoxia mice (Figures 1A and 1G). We also observed a twofold increase in the number of Iba1-positive cells in the PVN of WT-hypoxia mice as compared with WT controls (Figure 1E). A positive correlation between the number of microglia in the PVN and RVSP was observed in these animals (Figure 1F). A similar correlation with RVSP was also observed by Iba1 staining density in the PVN (data not shown). These data suggest that an increase in the PVN microglial cells was associated with an increase in the RVSP. In contrast, no significant changes in either numbers or intensity of staining were observed in CX3CR1GFP/GFP mice compared with controls (Figures 1C, 1D, 1H, and 1I). These data support the hypothesis that increases in microglial cells and sympathetic activity are associated with PH. Consistent with this hypothesis, data from CX3CR1GFP/GFP mice showed unchanged microglial cells and a lack of hypoxia-induced PH. In a separate preliminary experiment, we found an increase in microglial cells in the PVN of a monocrotaline rat model of PH, further supporting this contention (12). In addition, intraventricular infusion of monocrotaline-treated rats with minocycline attenuates RVSP and RVH, and significantly decreases PVN microglia (12). Minocycline is an antiinflammatory antibiotic that has commonly been used to inhibit microglial cells in vivo (13).
Figure 1.
Protection against hypoxia-induced pulmonary hypertension in CX3CR1-deficient mice correlates with decreased microglia activation. (A–D) Representative micrographs of immunohistochemical staining of microglia with an antibody against Iba1 using 3,3-diaminobenzidine detection in the paraventricular nucleus in wild-type (WT)-normoxia (A), CX3CR1-GFP/GFP- normoxia (B), WT-hypoxia (C), and CX3CR1-GFP/GFP-hypoxia (D) mice. Scale bars: 20 μm. (E) Chronic hypoxia induced an increase in the number of Iba1-positive cells (microglia) in WT mice, but not in CX3CR1GFP/GFP mice (*P < 0.05), and (F) a positive correlation between the number of microglia and the respective right ventricular systolic pressure (RVSP) was revealed (*P = 0.0057). Significant differences between groups were determined by one-way ANOVA followed by Newman-Keuls post hoc tests. Data are presented as mean ± SEM (n = 4/group). (G–J) Representative drawing of microglia captured in 100× images from WT-normoxia (G), CX3CR1-GFP/GFP-normoxia (H), WT-hypoxia (I), and CX3CR1-GFP/GFP-hypoxia (J) mice. Scale bars: 10 μm. H = hypoxia; Iba1 = ionized calcium-binding adaptor molecule 1; N = normoxia.
The CX3CL1/CX3CR1 axis constitutes a neuronal–microglial signaling system that is critical for neural plasticity, synaptic activity, and neuroimmune surveillance (2). Microglia in CX3CR1-deficient mice show an immature or reactive-like morphology and fail to modify in response to environmental effectors (14). This suggests that the absence of this chemokine system sets microglia in a “resting state.” It can also be observed in the representative drawings of microglia observed in 100× images from each group (Figures 1H and 1I). One could predict that a lack of their activation could alter microglia–neuron cross-talk that influences synaptic activity, leading to blunted sympathetic activity and an attenuated response to hypoxia-induced PH. This could be a selective effect on microglia–neuron cross-talk in the hypothalamus, as hypothalamic/pituitary axis function is normal in these mice (15).
Collectively, these observations represent the first correlative evidence of the involvement of microglial cells and possibly neuroinflammation in PH pathophysiology. Further investigations of the neuronal–microglial communication and transmission of these signals via autonomic regions to the cardiopulmonary system—for example, in microglia-depleted mice—are needed to delineate the precise role of microglia- and/or macrophage-expressed CX3CR1 in PH. Nonetheless, the observations presented herein highlight the need to consider the development of CX3CR1-targeted therapies for patients with PH.
Footnotes
Supported by National Institutes of Health grant HL102033.
Author disclosures are available with the text of this letter at www.atsjournals.org.
References
- 1.Amsellem V, Abid S, Poupel L, Parpaleix A, Rodero M, Gary-Bobo G, et al. Roles for the CX3CL1/CX3CR1 and CCL2/CCR2 chemokine systems in hypoxic pulmonary hypertension. Am J Respir Cell Mol Biol. 2017;56:597–608. doi: 10.1165/rcmb.2016-0201OC. [DOI] [PubMed] [Google Scholar]
- 2.Ransohoff RM, El Khoury J. Microglia in health and disease. Cold Spring Harb Perspect Biol. 2015;8:a020560. doi: 10.1101/cshperspect.a020560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Harrison JK, Jiang Y, Chen S, Xia Y, Maciejewski D, McNamara RK, et al. Role for neuronally derived fractalkine in mediating interactions between neurons and CX3CR1-expressing microglia. Proc Natl Acad Sci USA. 1998;95:10896–10901. doi: 10.1073/pnas.95.18.10896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shi P, Diez-Freire C, Jun JY, Qi Y, Katovich MJ, Li Q, et al. Brain microglial cytokines in neurogenic hypertension. Hypertension. 2010;56:297–303. doi: 10.1161/HYPERTENSIONAHA.110.150409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Haskó G. Receptor-mediated interaction between the sympathetic nervous system and immune system in inflammation. Neurochem Res. 2001;26:1039–1044. doi: 10.1023/a:1012305122327. [DOI] [PubMed] [Google Scholar]
- 6.Santisteban MM, Ahmari N, Carvajal JM, Zingler MB, Qi Y, Kim S, et al. Involvement of bone marrow cells and neuroinflammation in hypertension. Circ Res. 2015;117:178–191. doi: 10.1161/CIRCRESAHA.117.305853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ciarka A, Doan V, Velez-Roa S, Naeije R, van de Borne P. Prognostic significance of sympathetic nervous system activation in pulmonary arterial hypertension. Am J Respir Crit Care Med. 2010;181:1269–1275. doi: 10.1164/rccm.200912-1856OC. [DOI] [PubMed] [Google Scholar]
- 8.Kiernan EA, Smith SM, Mitchell GS, Watters JJ. Mechanisms of microglial activation in models of inflammation and hypoxia: Implications for chronic intermittent hypoxia. J Physiol. 2016;594:1563–1577. doi: 10.1113/JP271502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zubcevic J, Jun JY, Kim S, Perez PD, Afzal A, Shan Z, et al. Altered inflammatory response is associated with an impaired autonomic input to the bone marrow in the spontaneously hypertensive rat. Hypertension. 2014;63:542–550. doi: 10.1161/HYPERTENSIONAHA.113.02722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cerbai F, Lana D, Nosi D, Petkova-Kirova P, Zecchi S, Brothers HM, et al. The neuron-astrocyte-microglia triad in normal brain ageing and in a model of neuroinflammation in the rat hippocampus. PLoS One. 2012;7:e45250. doi: 10.1371/journal.pone.0045250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hilzendeger AM, Shenoy V, Raizada MK, Katovich MJ. Neuroinflammation in pulmonary hypertension: concept, facts, and relevance. Curr Hypertens Rep. 2014;16:469. doi: 10.1007/s11906-014-0469-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sharma RK, Oliveira AC, Kim S, Rigatto K, Zubcevic J, Rathinasabapathy A, et al. Involvement of neuroinflammation in the pathogenesis of monocrotaline-induced pulmonary hypertension. Hypertension. 2018;71:1156–1163. doi: 10.1161/HYPERTENSIONAHA.118.10934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Garrido-Mesa N, Zarzuelo A, Gálvez J. Minocycline: far beyond an antibiotic. Br J Pharmacol. 2013;169:337–352. doi: 10.1111/bph.12139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hellwig S, Brioschi S, Dieni S, Frings L, Masuch A, Blank T, et al. Altered microglia morphology and higher resilience to stress-induced depression-like behavior in CX3CR1-deficient mice. Brain Behav Immun. 2016;55:126–137. doi: 10.1016/j.bbi.2015.11.008. [DOI] [PubMed] [Google Scholar]
- 15.Milior G, Lecours C, Samson L, Bisht K, Poggini S, Pagani F, et al. Fractalkine receptor deficiency impairs microglial and neuronal responsiveness to chronic stress. Brain Behav Immun. 2016;55:114–125. doi: 10.1016/j.bbi.2015.07.024. [DOI] [PubMed] [Google Scholar]