Abstract
This paper reviews naturally occurring cell adhesion inhibitors derived from a plant, microbial and marine origin. Plant-derived inhibitors are classified according to a type of structure. Microbially and marine-derived inhibitors were described according to age. In addition, effects of inhibitors on cell proliferation and that of standards on cell adhesion are listed as much as possible.
Keywords: Cell adhesion, Inhibitor, Natural products, LFA-1, ICAM-1, HUVEC, PMA, LPS, TNF-α
Introduction
Cell adhesion molecules (CAMs) such as intercellular adhesion molecule 1 (ICAM-1, CD54 and immunoglobulin), vascular cell adhesion molecule 1 (VCAM-1) and E-selectin (CD62E) are critical in the regulation of immune response and inflammation. The extracellular interactions between specific CAMs expressing on the endothelium and leukocytes mediate leukocyte entry into tissues, T cell proliferation, and antigen presentation [1–4]. The key event in autoimmune disease is the migration of leukocytes to the disease site. Agents that inhibit leukocyte adhesion, transmigration and expression of related CAMs represent therapeutic potential as immunosuppressives and anti-inflammatory drugs. The major adhesive force for lymphocyte extravasation from the blood stream into tissue site is the protein–protein interaction of the adhesion molecules lymphocyte function-associated molecule 1 (LFA-1, CD11a/CD18 and β2 integrin) and its endothelial counter-receptor ICAM-1 [5, 6]. Monoclonal antibodies to ICAM-1 have been shown to inhibit lymphocyte transendothelial migration and have yielded very promising results in clinical trials for rheumatoid arthritis and organ transplantation [7, 8]. Therefore, the search for specific inhibitors of integrin-mediated cell adhesion with a small molecule in expectation of anti-inflammatory and anti-metastatic drugs started in the 1990s.
Generally, a cell adhesion inhibitor is categorized as target for cell–cell adhesion and for expression of cell adhesion molecules. Though certain small molecules such as flavonoids [9, 10] and others [11, 12] affecting expression of cell adhesion molecules are known, specific inhibitors for cell–cell contact are limited here in the review, as possible.
So far, some cell adhesion inhibitors based on synthetic methods and computer-aided drug design have been developed [13, 14]; however, natural products can still make unexpected structural discovery possible and they are believed to be a reservoir of resources for new types of drugs. This review introduces cell adhesion inhibitors focusing on those of naturally occurring plant, microbial and marine origin.
Cell adhesion inhibitors of plant origin
Terpenoid-sesquiterpene
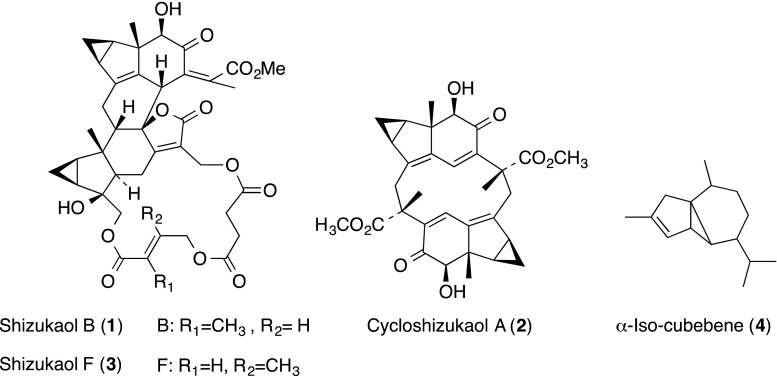
Chloranthus japonicas Sieb. (Chloranthaceae) is a perennial herb that grows in the southern part of Korea, Japan and China. It has been used for boils, dermatological disorder, and enteric fever in Korea as a folk remedy. Three active dimeric sesquiterpenoids of shizukaol B (1), cycloshizukaol A (2) and shizukaol F (3) were isolated from the MeOH extract of roots of C. japonicus [15]. These compounds inhibited phorbol 12-myristate-13-acetate (PMA)-induced homotypic aggregation of human promyelocytic leukemia (HL-60) cells without cytotoxicity with MIC values of 34.1 nM (1), 0.9 µM (2) and 27.3 nM (3), respectively. Although 1–3 did not affect the direct binding of LFA-1 to ICAM-1, these compounds markedly inhibited ICAM-1 expression in HL-60 cells in a dose-dependent manner. On the other hand, when human umbilical vein endothelial cells (HUVECs) were pretreated with 1–3 and stimulated with tumor necrosis factor α (TNF-α), adhesion of THP-1 cells to HUVECs decreased in a dose-dependent manner with IC50 values of 54.6 nM, 1.2 µM and 34.1 nM, respectively. In fact, 1 inhibited TNF-α-induced surface expression of the ICAM-1, VCAM-1 and E-selectin in HUVECs with IC50 values of 5.4 nM, 13.6 µM and 95.6 nM, respectively.
α-Iso-cubebene (4), a novel cubebene sesquiterpene from Schisandra chinensis (Schisandraceae), attenuated the activities of adhesion molecules in TNF-α-stimulated HUVECs [16]. α-Iso-cubebene (4) significantly suppressed the TNF-α-induced cell surface expression of VCAM-1 and E-selectin (43.8 and 29.6% inhibition, respectively) at 25 μg/mL, but not ICAM-1 expression. α-Iso-cubebene (4) attenuates TNF-α-stimulated endothelial adhesion to monocytes by inhibiting intracellular reactive oxygen species (ROS) production, the activation of redox-sensitive nuclear factor κB (NF-κB) transcription factor and expression of VCAM-1 and E-selectin.
Terpenoid-diterpene
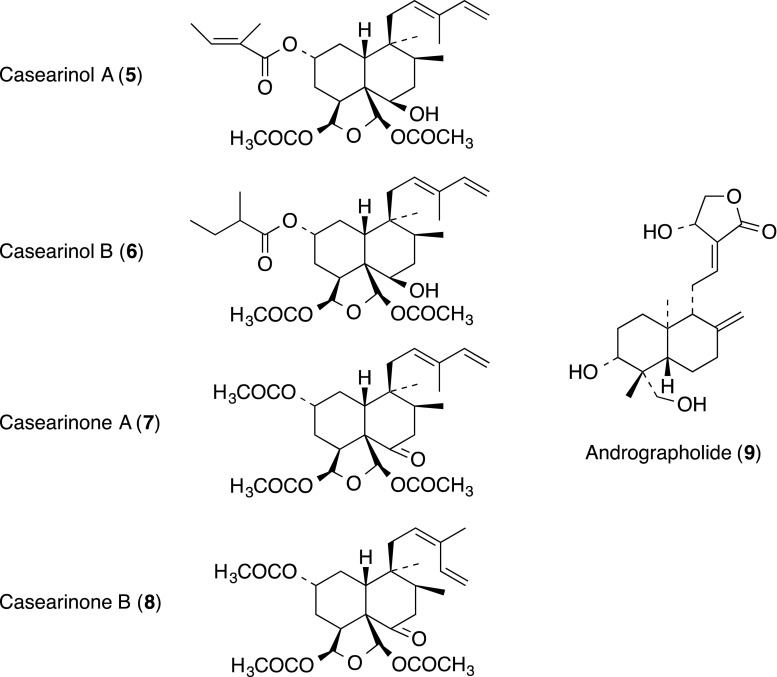
Four clerodane diterpenes, 18,19-diacetoxyclerodane 18,19-oxide acetals, casearinols A (5) and B (6), and casearinones A (7) and B (8), were isolated from the leaves of Casearia guianensis (Flacourtiaceae) [17]. Compounds 5–8 inhibited the binding of LFA-1 to ICAM-1. Quantitative data were obtained for casearinol A (5), which inhibited the binding of LFA-1 to ICAM-1 in a dose-responsive manner, yielding an IC50 of 50 µM. This is the first report of immunomodulatory activity for this class of diterpenes.
Andrographolide (9), an ent-labdane diterpenoid lactone isolated from the Chinese official herbal Andrographis paniculata (Acanthaceae), has been reported to have anticancer activity [18–20]. Jiang and co-workers reported that 9 inhibited the adhesion of gastric cancer cells with a highly expressing level of sialyl LewisX (SLeX) to the TNF-α-stimulated human endothelial cells by blocking E-selectin expression in a dose-dependent manner, in a concentration range of 1–10 µM [21].
Terpenoid-triterpene, steroid and related compound
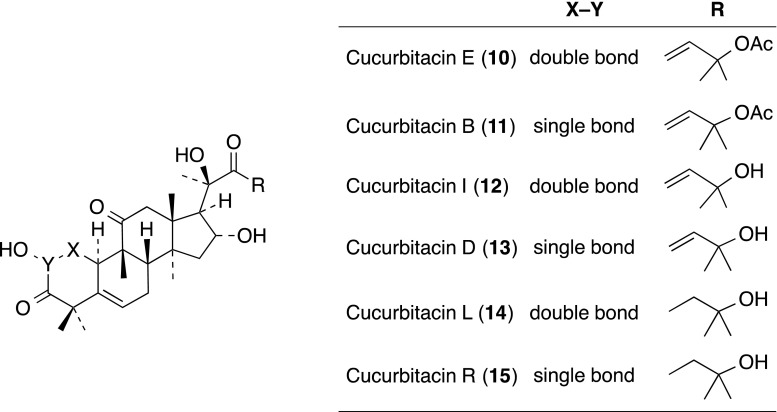
Cucurbitacin E (10) was isolated from CH2C12 extract of the stem and leaves of Conobea scoparioides Benth. (Scrophulariaceae) as an antagonist of CD18-mediated cell adhesion. Cucurbitacin E (10) is a tetracyclic triterpenoid with an unsaturated side chain present in various plant families such as the Cucurbitaceae, Scrophulariaceae, Euphorbiaceae, Liliaceae and Elaeocarpaceae. Cucurbitacin E (10) and five related analogues, cucurbitacins B (11), I (12), D (13), L (14) and R (15) obtained separately, were tested in the cell adhesion assay. Compounds 10–13 showed inhibition of JY/HeLa cell binding through LFA-1/ICAM-1-mediated adhesion, with IC50 values of 0.18, 0.30, 0.95 and 1.36 µM, respectively. Cucurbitacin E (10) was demonstrated to inhibit cell adhesion to HeLa cells by interfering with LFA-1 and not ICAM-1 [22].
Touihri-Barakati and co-workers reported that cucurbitacin B (10) from the leaves of Tunisian Ecballium elaterium (Cucurbitaceae) showed anti-integrin activity on human glioblastoma U87 cells, without being cytotoxic at concentrations up to 500 nM [23].
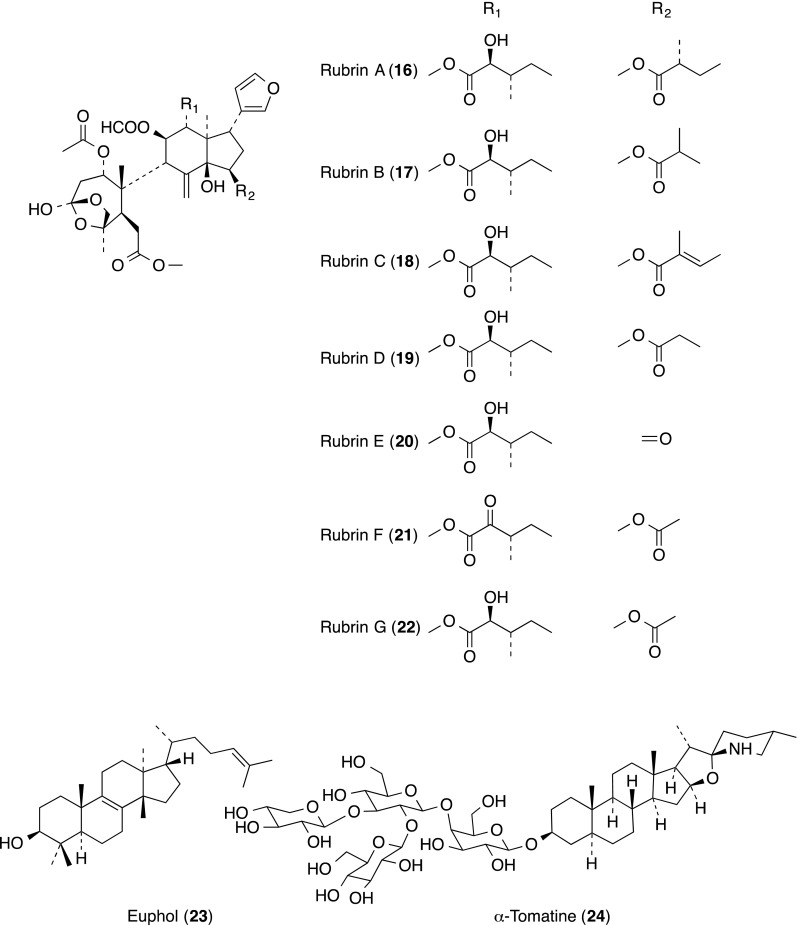
The extract from the root of Trichilia rubra (Meliaceae) was identified as having potent inhibitory activity in a bioassay for LFA-l/ICAM-I-mediated adhesion of JY and HeLa cells [24]. A series of seco-limonoids (16–22) with uncommon hemi ortho ester A-rings, was isolated. Compounds 16–22 exhibited potent inhibitory activity in the LFA-l/ICAM-1-mediated cell adhesion assay with IC50 values in the range of 10–25 nM. None of the compounds showed cytotoxicity at concentrations up to 20 µM.
The tetracyclic triterpene euphol (23) is the main constituent found in the sap of Euphorbia tirucalli (Euphorbiaceae), widely known in Brazilian traditional medicine for its use in the treatment of several kinds of cancer. The effect of euphol (23) on experimental models of colitis and the underlying mechanisms involved in its action has been reported [25]. The euphol (23) decreased lipopolysaccharide (LPS)-induced monocyte chemotactic protein 1 (MCP-1), TNF-α, interleukin 6 (IL-6) and interferon γ (IFN-γ), but increased IL-10 secretion from bone marrow-derived macrophages in vitro, and markedly inhibited both selectin (P- and E-selectin) and integrin (ICAM-1, VCAM-1 and LFA-1) expression in colonic tissue. Moreover, euphol (23) treatment markedly inhibited the activation of NF-κB in mouse colon tissue.
α-Tomatine (24), a glycoalkaloid isolated from Lycopersicon esculentum Linn, was reported to inhibit the PMA-induced abilities of adhesion, morphology/actin cytoskeleton arrangement, invasion and migration by cell–matrix adhesion assay, through blocking protein kinase Cα (PKC-α), extracellular signal-regulated kinase (ERK) and NF-κB activation. [26]
Lignane
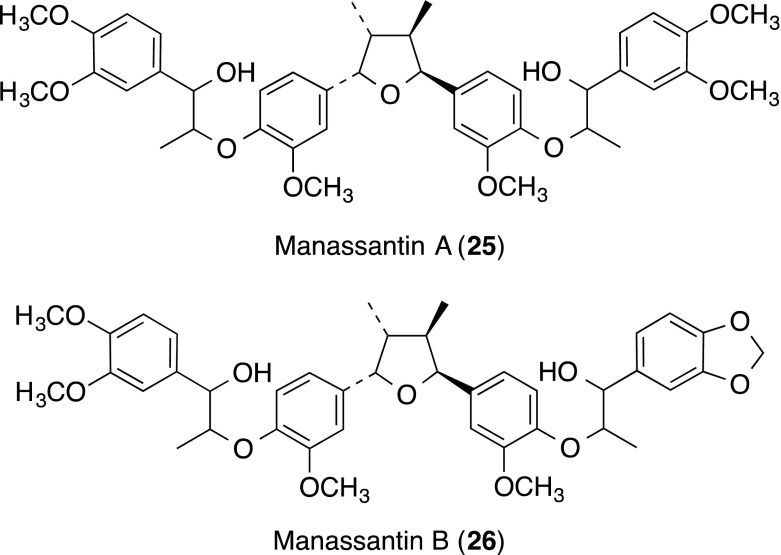
Manassantin A (25) and B (26), dineolignans isolated from Saururus chinensis, inhibited PMA-induced ICAM-1/LFA-1-mediated homotypic aggregation of HL-60 cells without cytotoxicity and with MIC values of 1.0 and 5.5 nM, respectively. After pretreating HUVECs with 25 and 26 followed by stimulation with TNF-α, adhesion of human acute monocytic leukemia cell line THP-1 to HUVECs decreased in a dose-dependent manner with IC50 values of 5 and 7 ng/mL, respectively, without cytotoxicity [27]. Both 25 and 26 also inhibited TNF-α-induced up-regulation of ICAM-1, VCAM-1 and E-selectin.
Flavonoide
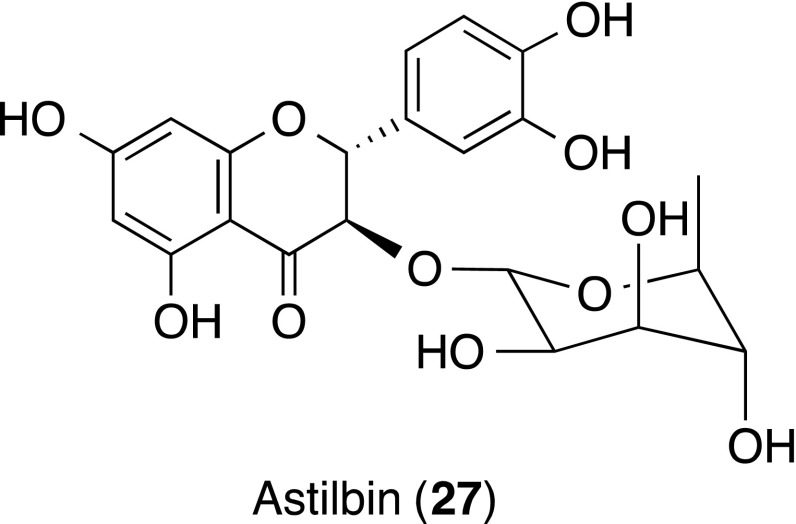
Astilbin [3,3′,4′,5,7-pentahydroxyflavanone 3-(6-deoxy-(l-mannopyranoside)] (27) from the rhizome of Smilax glabra (Liliaceae) was demonstrated to show a selective immunosuppressive feature [28]. The effect of 27 on concanavalin A (Con A)-induced liver injury by focusing on the TNF-α production and T lymphocyte adhesion was investigated. Inhibitory effect of 27 on the adhesion of Con A-activated human Jurkat T cells to endothelial cell line ECV-304 was reported.
Alkaloid
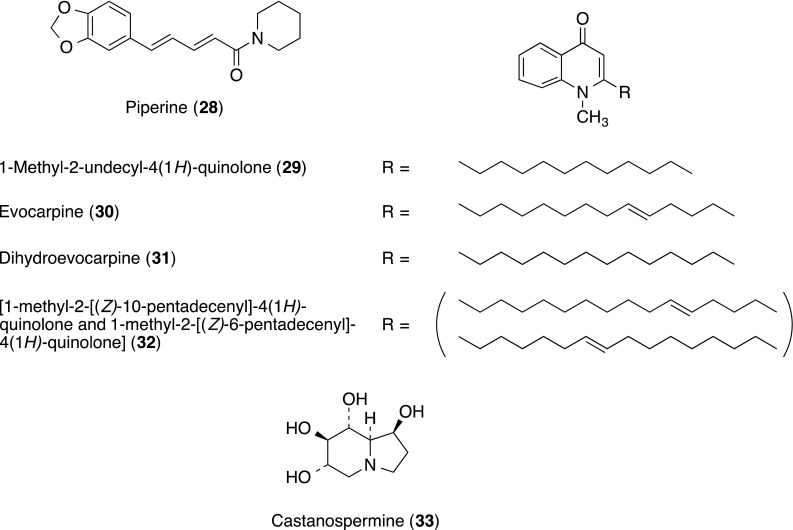
Piperine (28) and ethyl 3′,4′,5′-trimethoxycinnamate (34; the structure is shown in a section of other compounds described below) from the combined hexane and chloroform extracts of Piper longum (Piperaceae) were isolated as potent inhibitors of cell adhesion molecules on HUVECs [29]. Both 28 and 34 inhibited the TNF-α-induced expression of ICAM-1 at IC50 values of 45 and 25 µg/mL, respectively. In further study, 34 significantly blocked the adhesion of neutrophils to endothelium in a concentration-dependent manner. Compound 34 also significantly inhibited TNF-α-induced expression of VCAM-1 and E-selectin at 50 µg/mL. To elucidate its structure–activity relationship, effects of synthesized analogues of 34 and its thio, thiono analogues, and synthesized 7-hydroxy-4-methylcoumarin derivatives on cell adhesion molecules were studied [30, 31].
Lee and co-workers reported four quinolone alkaloids (29–32) isolated from the methanol extracts of Evodiae fructus, as the specific inhibitor on the binding of LFA-1 and ICAM-1 [32]. Evodiae fructus is natural medicine originated from Evodia rutaecarpa (Juss.) Benth. (Rutaceae), which has been used for treatment of gastrointestinal disorders and headaches, as an analgesic and antiemetic, and for amenorrhea in Korea.
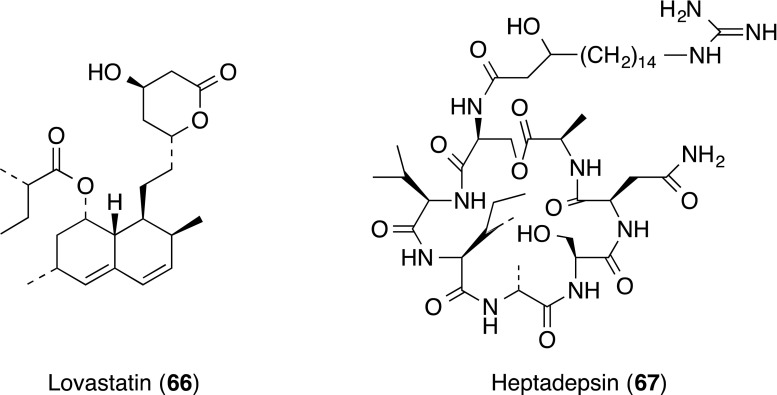
The four quinolone alkaloids inhibited the interaction of sICAM-1 and LFA-1 in THP-1 cells at IC50 values of > 150 (29), 109.8 (30), > 150 (31) and 40.9 μM (32), respectively [vs. lovastatin (66) as a positive control, IC50 33 µM]. On the other hand, they had no effect on direct binding assay using sVCAM-1 and E-selectin. They did not show cytotoxicity at the concentrations employed in the study (ca. 70–80% of THP-1 cell viability at 150 µM).
Among four quinolone alkaloids (29–32), cell adhesion inhibitory activity was suggested to be positively influenced by the presence of a double bond and an increase in aliphatic side chain length.
Castanospermine (33) is an indolizidine alkaloid originally isolated from the seeds of Castanospermum austral (the Australian Moreton Bay Chestnut, Fabaceae). Effects of 33 in a range of concentrations from 16,384 to 0.25 μM on mononuclear/endothelial cell binding and expression of their cell adhesion molecules were reported [33]. Upon HUVECs, 33 reduced expression of E-selectin, ICAM-1, ICAM-2 and platelet endothelial cell adhesion molecule (PECAM)-1, but increased it for P-selectin. Upon peripheral blood mononuclear cells, 33 reduced expression of L-selectin, LFA-1α, very-late antigen 4 (VLA-4; integrin α4β1), macrophage adhesion ligand 1 (Mac-1) and complement receptor 4 (CR-4; CD11c/CD18), but increased expression of P-selectin glycoprotein ligand 1 (PSGL-1) and PECAM-1. Similar changes of expression were found in the subset of lymphocytes and monocytes, but the reductions in LFA-1α and VLA-4 on lymphocytes and Mac-1 (CD11b/CD18) and CR-4 on monocytes were greater.
Other compounds
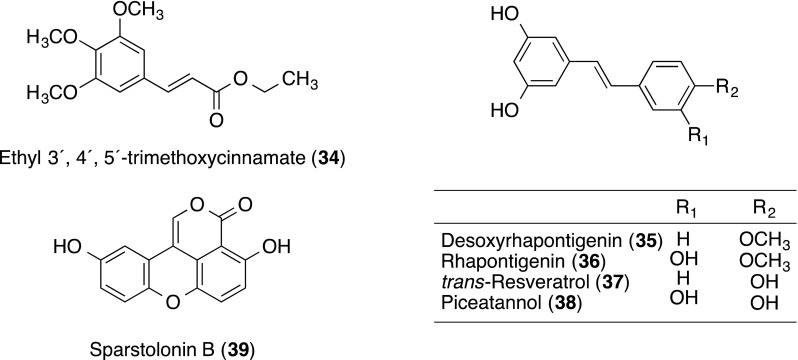
Lee and co-workers found an inhibitory effect of methanol extract of Rheum undulatum (Polygonaceae) rhizomes on cell adhesion in search for anti-inflammatory or anti-metastasis agents, and isolated six stilbenes from the by bioactivity-guided fractionation. Six stilbenes were identified as desoxyrhapontigenin (35), rhapontigenin (36), trans-resveratrol (37), piceatannol (38), piceatannol-3′-O-β-D-glucopyranoside and isorhapontin. Among them, 35–38 inhibited the direct binding between sICAM-1 and LFA-1 of the THP-1 cells in a dose-dependent manner with IC50 values of 50.1, 25.4, 33.4 and 45.9 μM, respectively (Table 1). Compounds 36, 37 and 38 also had an inhibitory effect on direct binding between sVCAM-1 and VLA-4 of THP-1 cells [34].
Table 1.
Inhibitory effect of 35–38 on cell adhesion-mediated LFA-1/sICAM-1 and E-selectin/VCAM-1
| LFA-1/sICAM-1 (µM) | E-selectin/sVCAM-1 (µM) | |
|---|---|---|
| 35 a | 50.1 | Inactive |
| 36 | 25.4 | > 100 |
| 37 | 33.4 | > 100 |
| 38 | 45.9 | 44.1 |
| Lovastatin (66) | 57.2 | – |
aCompound 35 showed cytotoxicity at 100 µM
In addition, compound 38 can interfere with the binding between integrin (LFA-1 and VLA-4) and immunoglobulins (ICAM-1 and VCAM-1). A lovastatin (66) was used as a positive control for the binding between sICAM-1 and LFA-1 of the THP-1 cells (IC50 57.2 µM; Table 1).
Sparstolonin B (39) is an isocoumarin compound isolated from the tubers of both Sparganium stoloniferum and Scirpus yagara. Sparstolonin B (39) inhibited LPS-induced expression of ICAM-1 and VCAM-1 in HUVECs at 10 and 100 µM, respectively [35].
Sparstolonin B (39) significantly suppressed the adhesion of THP-1 cells to LPS-activated HUVECs at a concentration of 100 µM. The inhibitory effect of 39 on LPS-induced phosphorylation of extracellular signal-regulated kinase (Erk1/2) and serine/threonine kinase (Akt, protein kinase B) was also reported.
Plant extract, etc.
Effects of crude plant extract, snake venom and other naturally occurring fatty acid derivatives on cell adhesion molecules are also reported. As they are not isolated as pure components, only references are shown [36–45].
Cell adhesion inhibitors of microbial origin
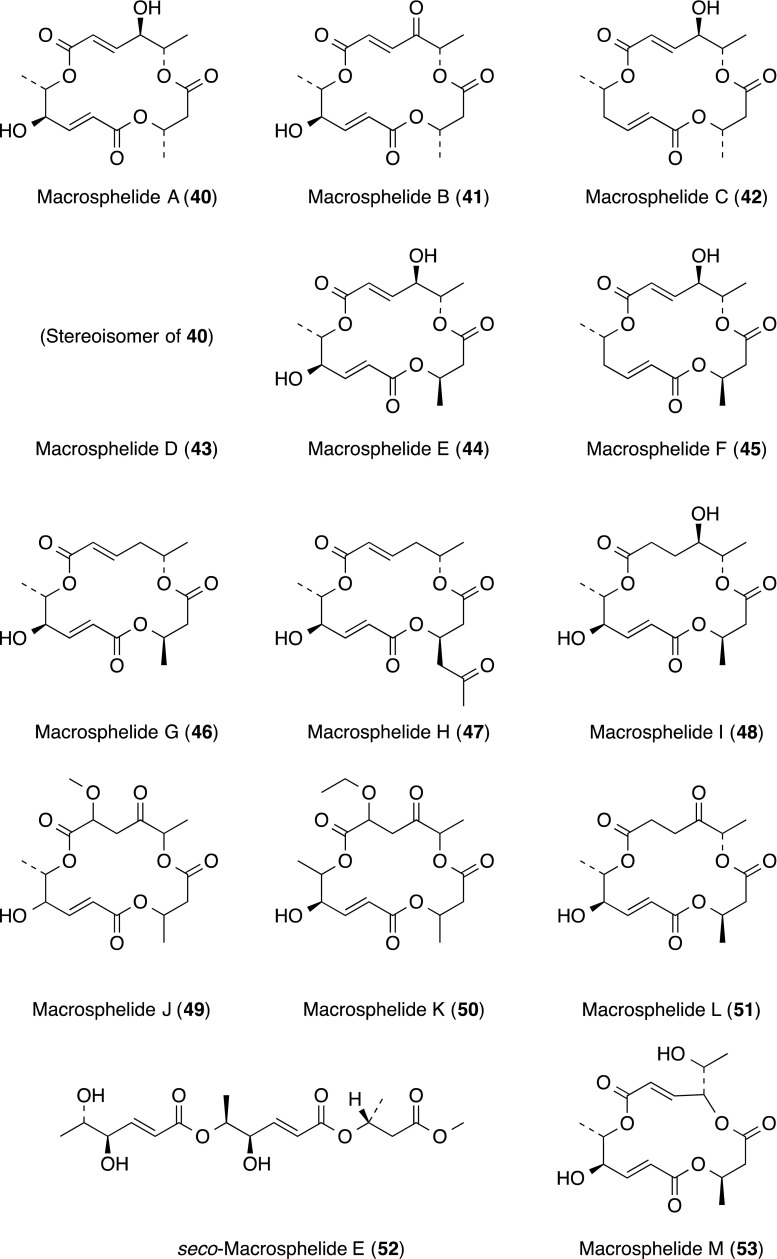
Macrosphelides (MSs) are 16-membered macrolides, embodying 3 ester linkages produced by several fungal strains. MSs A–D (40–43), J (49) and K (50) were originally isolated from the culture broth of Microsphaeropsis sp. FO-5050 as cell adhesion inhibitors [46–49]. At almost the same time, MSs A (40), C (42), E–I (44–48), L (51), seco-MS E (52) and MS-M (53) were isolated from a fungal strain of Periconia byssoides originally separated from the sea hare Aplysia kurodai [50–54].
MSs A–D (40–43) inhibited the adhesion of SLex-expressing HL-60 cells to endothelial cell leukocyte adhesion molecule 1 (ELAM-1)-expressing HUVECs in a dose-dependent fashion, with IC50 values of 3.5, 36, 67.5 and 25 µM, respectively. Among MSs, MS-A (40) showed the most potent inhibitory activity. On the one hand, MSs J (49) and K (50) were inactive (IC50 value: > 100 µg/mL).
It was suggested that they prevent the cell–cell adhesion by blocking the binding of SLex to ELAM-1. However, MSs showed no effect on the adhesion of sialyl Lewis A (SLea)-expressing HL-60 cells to HUVECs. Furthermore, pretreatment of HL-60 cells, not HUVECs, with MSs caused inhibition of the adhesion of HL-60 to HUVECs. These findings indicated that MSs specifically bound to SLex on HL-60 cells to block the cell–cell adhesion [55].
MSs proved to be effective in several in vivo models. In the mouse model of B16/BL6 melanoma lung metastasis, MS-B (41) caused a dose-dependent decrease in lung metastatic nodules without any toxic effect including body weight loss in the range of 5–20 mg/kg. Furthermore, its efficacy in combination therapy with anti-cancer drugs was demonstrated. Combined therapy of MS-B (41) and cisplatin (CDDP) induced remarkable lung metastasis inhibition without adverse effects of CDDP to the host [56, 57].
The total synthesis of MS-A (40) has been reported by several groups. A novel total synthesis have been accomplished by the group of the Kitasato Institute [58, 59]. The combinatorial synthesis of a 122-member MS library including MSs A (40), C (42), E (44) and F (45) has been achieved based on a unique strategy for a three-component coupling utilizing a palladium-catalyzed chemoselective carbonylation and an unprecedented macrolactonization on a polymer support [60]. Synthetic approaches to MS derivatives, based on medicinal chemistry, were reviewed [61]. At present, MS-A (40) is commercially available as a reagent.
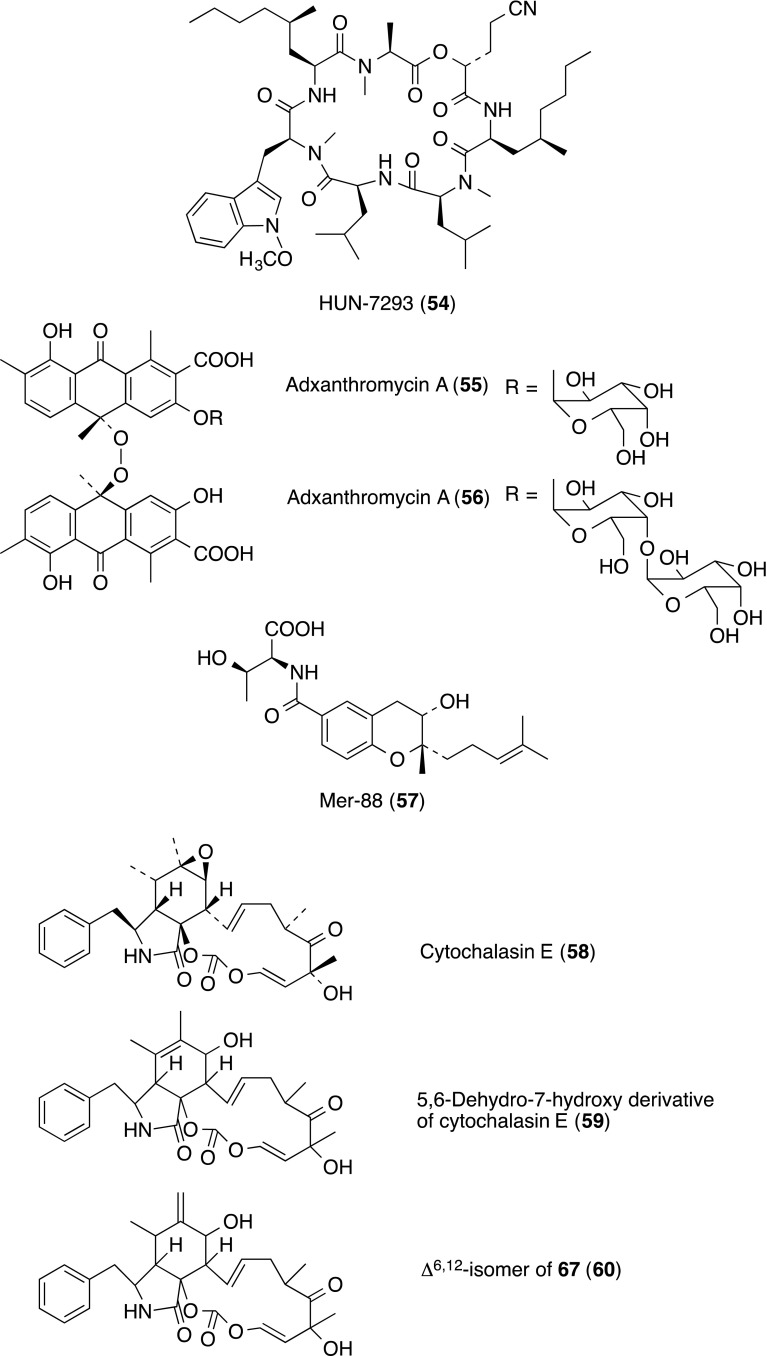
HUN-7293 (54) is a fungal cyclodepsipeptide that was first identified as an inhibitor of VCAM expression [62]. HUN-7293 (54) inhibited expression of VCAM-1 and ICAM-1 on TNF-α-stimulated human microvascular endothelial cells (HMEC-1), with IC50 values of 2 and 50 nM, respectively.
HUN-7293 (54) almost inhibited cell adhesion between the human Burkitt’s lymphoma B (BL 2) cell and TNF-α-stimulated HMEC-1 at a concentration of 20 nM. Total synthesis of 54 had done and evaluation of synthetic analogues as inhibitors of VCAM-1 expression was further reported [63, 64].
Nakagawa and co-workers found two inhibitors adxanthromycins A (55) and B (56) of ICAM-1/LFA-1-mediated cell adhesion molecule produced by Streptomyces sp. NA-148 [65–67]. The structure of 55 and 56 were characterized as dimeric anthrone peroxide skeleton containing an α-d-galactose for 55 and two α-d-galactose for 56.
Both 55 and 56 inhibited homotypic aggregation of Epstein–Barr virus (EBV)-immortalised B cell lymphoblastoid line (JY cell) from 1.5 µg/mL in a dose dependent manner. A complete inhibition was observed at 6.25 µg/mL.
The toxicity (IC50) of 55 and 56 against JY cell was 15.2 µg/mL. Compounds 55 and 56 also inhibited SKW-3 adhesion to soluble ICAM-1 in a dose-dependent manner with an IC50 of 18.8 and 25.0 µg/mL, respectively. The cell toxicity (IC50) of adxanthromycins against SKW-3 was 110.0 µg/mL. In the cell-free receptor binding assay, both 55 and 56 showed weak inhibition with an IC50 of 760 µg/mL. They were reported as the first example of inhibitors of ICAM-l/LFA-1-mediated adhesion molecule isolated from microbial sources.
The benzopyran derivative (57) was found in the culture of Streptomyces sp. Mer-88 as ICAM-1/LFA-1 binding inhibitors, ICAM-1 inhibitors and LFA-1 inhibitors [68]. Compound 57 inhibited binding between ICAM-1 and LFA-1 in the range of 31–2500 µg/mL dose dependently, without cytotoxicity up to a concentration of 1000 µg/mL. Then, Xu and co-workers reported isolation of the same compound, N-[[3,4-dihydro-3S-hydroxy-2S-methyl-2-(4′R-methyl-3′S-pentenyl)-2H-1-benzopyran-6-yl]carbonyl]-threonine (57), produced by Streptomyces xiamenensis, its structure, including the absolute configuration, and its anti-fibrotic properties [69].
Three derivatives of cytochalasin were isolated from the cultured broth of the fungal strain Mycotypha sp. UMF-006, as inhibitors of cell adhesion based on LFA-1/ICAM-1 [70]. These compounds were identified to be cytochalasin E (58), 5,6-dehydro-7-hydroxy derivative of cytochalasin E (59) and Δ6,12-isomer of 59 (60). All these components inhibited adhesion of HL-60 cells to CHO-ICAM-1 cells at IC50 values of 30 µg/mL for 58, 75 µg/mL for 59 and 90 µg/mL for 60.
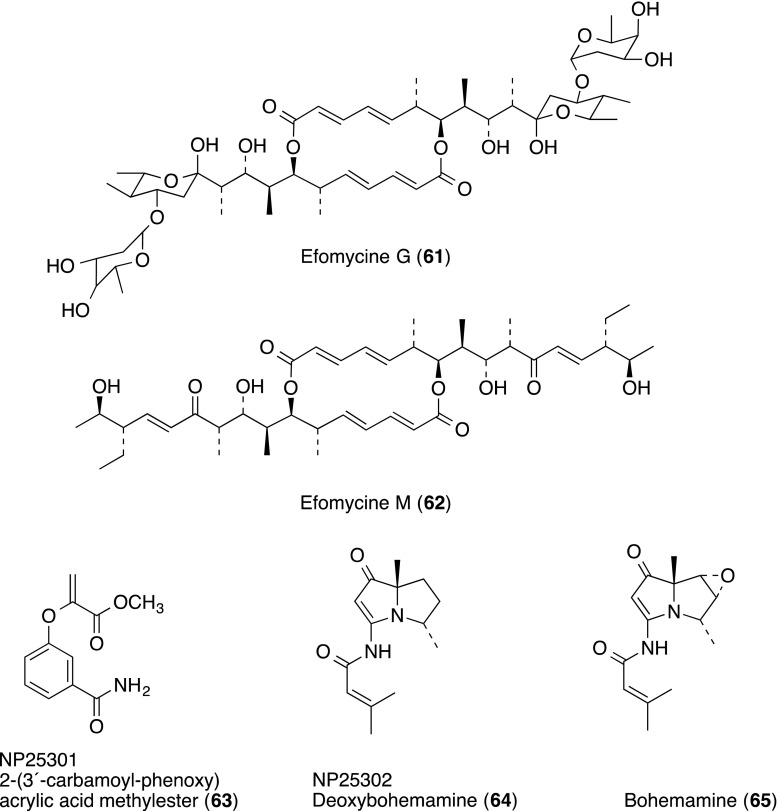
Members of the efomycine family from Streptomyces BS1261 were found to inhibit leukocyte adhesion, from a screening library of 20,000 natural compounds [71]. Finally, efomycines A B, E and G (61) were isolated as active substances [only structures of G (61) and M (62) were shown]. Members of the efomycine family inhibited the binding of human or porcine neutrophils by 50–60% at 10−5 M, whereas efomycine M (62) did not have a significant effect. Efomycine M (62) showed the most selective inhibitory effects on selectin-mediated leukocyte-endothelial adhesion in vitro, significantly diminishing rolling in mouse ear venules in vivo. In addition, efomycine M (70) alleviated cutaneous inflammation in two complementary mouse models of psoriasis, one of the most common chronic inflammatory skin disorders. Molecular modeling demonstrated a spatial conformation of efomycines mimicking naturally occurring selectin ligands.
Three compounds, NP25301 (63), NP25302 (deoxybohemamine 64) and bohemamine (65), inhibitors of cell adhesion based on LFA-1/ICAM-1, were isolated from the cultured broth of the strain Streptomyces sp. UMA-044 [72]. Compounds 63–65 inhibited adhesion of HL-60 cells to CHO-ICAM-1 cells at IC50 values of 29.5 µg/ml for 63, 24.3 µg/ml for 64 and 27.2 µg/ml for 65.
Random screening of chemical libraries identified the 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor lovastatin (66), a drug clinically used for lowering cholesterol levels, as an inhibitor of the LFA-1/ICAM-1 interaction [73]. Lovastatin (66) showed binding inhibition of recombinant ICAM-1 to purified LFA-1 with an IC50 value of 2.1 µM. Inhibitory effects of statin-derived compounds on the binding were also shown with a range of IC50 0.04–14 µM. The biological relevance of LFA-1 inhibition by statins with respect to the overall benefit of this drug class was reviewed [74].
Structurally related to known naturally occurring cyclic heptadepsipeptides, HUN-7293 (54), named heptadepsin (67), was isolated from the culture broth of Paenibacillus sp. [75]. Compound 67 inhibited LPS-stimulated adhesion between HUVECs and HL-60 cells with an IC50 value of 0.92 µg/mL, without showing any cytotoxicity up to 30 µg/mL. Compound 67 also inhibited cellular adhesion induced by lipid A, the active component of LPS, but it did not inhibit TNF-α- or IL-1β-induced cell adhesion. Heptadepsin (67) was shown to inactivate LPS by direct interaction with LPS and lipid A from the results of surface plasmon resonance analysis.
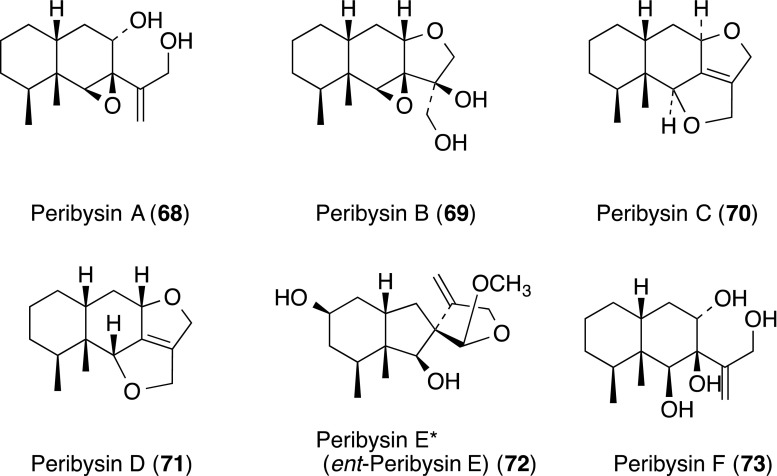
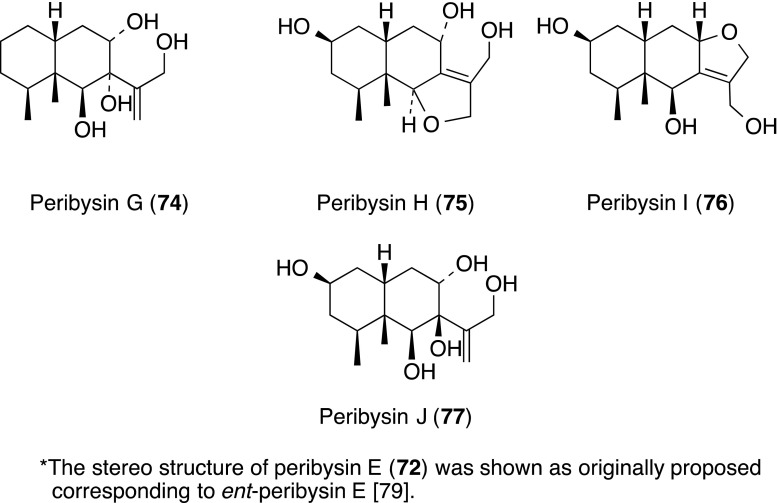
Peribysins A–J (68–77) including a furanofuran, were isolated from the culture of a strain Periconia byssoides OUPS-N133 separated from the sea hare Aplysia kurodai. They inhibited the adhesion of HL-60 cells to LPS-stimulated HUVECs with an IC50 range of 0.1–20 µM. Among them, compounds A (68) and D (71) showed the most potent cell adhesion inhibitory activity with IC50 values 0.3 and 0.1 µM, respectively, as compared to that of herbimycin A (standard, IC50 38 µM) [54, 76–78]. Interestingly, the producing strain OUPS-N133 of peribysins was the same as that of MSs A (40), C (42), E (44), F–I (45–48), L (51) and M (53). The total synthesis necessitated revision of the assignment of the absolute configuration of 72 [79].
Cell adhesion inhibitors derived from a marine organism
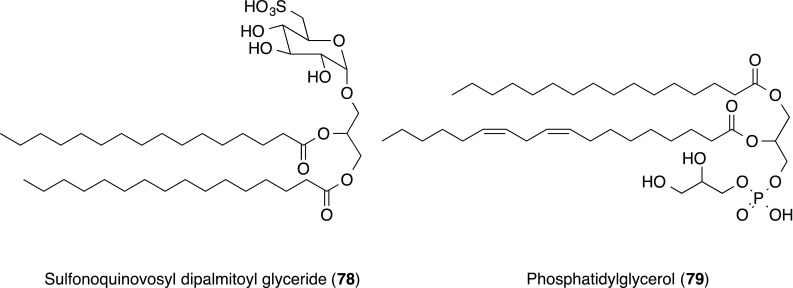
In the screening for P-selectin inhibitors, sulfonoquinovosyl dipalmitoyl glyceride (78) and phosphatidylglycerol (79) were isolated from the 85% EtOH extract of the marine alga Dictyochloris fragrans. Both 78 and 79 inhibited P-selectin binding to sulfatides in the P-selectin–IgG ELISA assay, with IC50 values of 5 and 1 µM, respectively [80].
The inhibitory effect of 78 on HL-60 cell adhesion to immobilized P-selectin receptor globulin (Rg) was only shown with an IC50 value of 40 µM. Compound 78 was further shown for its ability to inhibit (24%) the P-selectin-dependent binding of activated platelets to HL-60 cells.
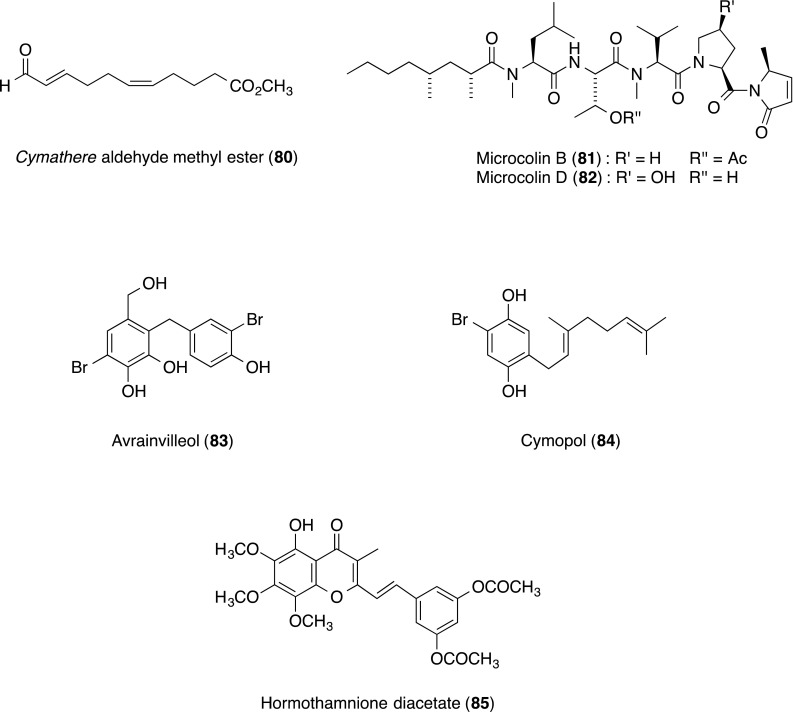
From a panel of 60 unusual marine natural products, 17 compounds inhibited LFA-1/ICAM-1-based cell aggregation without showing significant cytotoxicity in the primary assay. Six compounds inhibited the cell–cell adhesion of HL-60 cells to CHO-ICAM-1 cells. The unusual oxylipin Cymathere aldehyde methyl ester (80; IC50 3.5 µM), cyanobacterial lipopeptide microcolins B (81; IC50 0.15 µM) and D (82; IC50 0.9 µM), bromophenol avrainvilleol (83; IC50 2.2 µM), sesquiterpene cymopol (84; IC50 2.7 µM) and cryptophyte-derived compound styrylchromone hormothamnione diacetate (85; IC50 1.5 µM) significantly inhibited LFA-1/ICAM-1-mediated cell adhesion (Table 2). The pharmacological activity and structure–activity relationships of selected marine algal metabolites are described [81].
Table 2.
Effects of marine natural products (80–85) on cell adhesion of HL-60 cells to CHO-ICAM-1 cells, and on proliferation of CHO-ICAM-1 cells
| Compound | Sources | HL-60 | HL-60/CHO-ICAM-1 | CHO-ICAM-1 | |||
|---|---|---|---|---|---|---|---|
| (A) C. A.a | (B) C. P.b | S.I.c | (C) Cell adhesion | (D) C. P.b | |||
| MICd (µM) | IC50 (µM) | B/A | IC50 (µM) | IC50 (µM) | |||
| 80 | Cymathere aldehyde methyl ester | Cymathere triplicate | 5.9 | > 5.9 | > 1.0 | 3.5 ± 0.3 | 2.7 ± 0.5 |
| 81 | Microcolin B | Lyngbya majuscula | 1.7 | 1.3 ± 0.2 | 0.7 | 0.15 ± 0.09 | > 1.4 |
| 82 | Microcolin D | Lyngbya majuscula | 1.8 | > 1.8 | > 1.0 | 0.9 ± 0.1 | 1.1 ± 0.2 |
| 83 | Avrainvilleol | Avrainvillea longicaulis | 3.1 | > 3.1 | > 1.0 | 2.2 ± 0.2 | 0.8 ± 0.3 |
| 84 | Cymopol | Cymopolia barbata | 1.3 | > 3.8 | > 3.0 | 2.7 ± 0.2 | > 3.1 |
| 85 | Hormothamnione diacetate | Chrysophaeum taylori | 2.6 | > 2.6 | > 1.0 | 1.5 ± 0.2 | 0.6 ± 0.2 |
| Cytochalasin Be | 0.3 | > 2.6 | 8.9 | 1.2 ± 0.2 | 0.2 ± 0.01 | ||
Values are means of three independent determinations ± SE
aCell aggregation; bCell proliferation; cSpecific index; dMinimum inhibitory concentration; eUsed as a standard
Conclusions
As listed above, natural cell adhesion inhibitors are still proved essential to drug development due to their great variety of unexpected structures, such as plant-derived terpenoid (1–24), lignans (25–26), flavonoid (27) and alkaloid (28–33), and macrolide (40–53, 58–60), cyclodepsipeptide (54, 67), dimeric anthrone (55, 56), and furanofuran (68–77) of microbial origin, and marine-derived compounds (78–85).
Among those from different origin, it is interesting to note that casearinols (5, 6), casearinones (7, 8), andrographolide (9), lovastatin (66) and peribysins A–J (68–77) were structurally similar based on a decalin (decahydronaphthalene) skeleton with a side chain or condensed furanofuran.
Basically, all inhibitors covered this time were found using a cell-based assay for cell–cell adhesion or cell-soluble cell adhesion molecule. In a different way from the cell-based assay, the pharmacophore of sLex, recognized by a family of selectin, was used to search a three-dimensional database of chemical structures. As result of a search for a binding inhibitor between selectins and sLex, glycyrrhizin (structure was not shown), a saponin and sweet-tasting constituent of Glycyrrhiza glabra (liquorice, Fabaceae) root, was matched as a pharmacophore [82].
Commercially available cell adhesion inhibitors are almost all developed based on peptide [83, 84]. But naturally occurring inhibitors such as MSs A–D (40–43) were not only first isolated in the culture of Microsphaeropsis sp. FO-5050 as cell adhesion inhibitors, but also possess a novel macrocyclic skeleton with tri-ester groups. Furthermore, MSs (40–53), nontoxic against HL-60 cells and HUVECs, have recently become an active area of research for anticancer drugs [61]. It is of great interest that MSs (40–53) were found in the culture of Periconia byssoides OUPS-N133, a strain producing peribysins A–J (68–77) [54, 76–78]. Application of a diversity of microorganisms displays an infinite number of possibilities and unexploited production capacity. Therefore, natural products are still of particular interest as seeds for drug discovery.
Acknowledgements
I am indebted particularly to Prof. Kazuki Saito of the Graduate School of Pharmaceutical Sciences, Chiba University. I am deeply indebted to the late Prof. Kazuo Toriizuka of the School of Pharmacy, Showa University. I deeply grateful to Dr. Satoshi Ōmura, Distinguished Emeritus Professor of Kitasato University. I also sincerely thank Dr. Kanki Komiyama of Kitasato Research Center for Environmental Science, Prof. Yong-pil Kim and Prof. Masahiko Hayashi of the Department of Pharmacy, Iwaki Meisei University, for collaboration at the Kitasato Institute. This research was supported in part by a Grant-in-Aid for Scientific Research (C; no. 15K08002).
Footnotes
The original version of this article was revised due to a retrospective Open Access order.
This review is dedicated to the Japanese Society of Pharmacognosy (JSP) Award for Scientific Contributions 2015.
Change history
8/3/2018
The article Naturally occurring cell adhesion inhibitors, written by Satoshi Takamatsu, was originally published electronically on the publisher’s internet portal.
References
- 1.Picker LJ, Butcher EC. Physiological and molecular mechanisms of lymphocyte homing. Annu Rev Immunol. 1992;10:561–591. doi: 10.1146/annurev.iy.10.040192.003021. [DOI] [PubMed] [Google Scholar]
- 2.Osborn L. Leukocyte adhesion to endothelium in inflammation. Cell. 1990;62:3–6. doi: 10.1016/0092-8674(90)90230-C. [DOI] [PubMed] [Google Scholar]
- 3.Springer TA. Adhesion receptors of the immune system. Nature. 1990;346:425–434. doi: 10.1038/346425a0. [DOI] [PubMed] [Google Scholar]
- 4.Bradfield PF, Imhof BA. Adhesion mechanisms of endothelial cells. Handb Exp Pharmacol. 2004;165:405–436. doi: 10.1007/978-3-540-68170-0_13. [DOI] [PubMed] [Google Scholar]
- 5.Hynes RO. Integrins: versatility, modulation, and signaling in cell adhesion. Cell. 1992;69:11–25. doi: 10.1016/0092-8674(92)90115-S. [DOI] [PubMed] [Google Scholar]
- 6.Carlos TM, Harlan JM. Leukocyte-endothelial adhesion molecules. Blood. 1994;84:2068–2101. [PubMed] [Google Scholar]
- 7.Kavanaugh AF, Davis LS, Nichols LA, Norris SH, Rothlein R, Scharschmidt LA, Lipsky PE. Treatment of refractory rheumatoid arthritis with a monoclonal antibody to intercellular adhesion molecule 1. Arthritis Rheum. 1994;37:992–999. doi: 10.1002/art.1780370703. [DOI] [PubMed] [Google Scholar]
- 8.Haug CE, Colvin RB, Delmonico FL, Auchincloss H, Jr, Tolkoff-Rubin N, Preffer FI, Rothlein R, Norris S, Scharschmidt L, Cosimi AB. A phase I trial of immunosuppression with anti-ICAM-1 (CD54) mAb in renal allograft recipients. Transplantation. 1993;55:766–772. doi: 10.1097/00007890-199304000-00016. [DOI] [PubMed] [Google Scholar]
- 9.Kobuchi H, Roy S, Sen CK, Nguyen HG, Packer L. Quercetin inhibits inducible ICAM-1 expression in human endothelial cells through the JNK pathway. Am J Physiol. 1999;277:C403–C411. doi: 10.1152/ajpcell.1999.277.3.C403. [DOI] [PubMed] [Google Scholar]
- 10.Jia Z, Nallasamy P, Liu D, Shah H, Li JZ, Chitrakar R, Si H, McCormick J, Zhu H, Zhen W, Li Y. Luteolin protects against vascular inflammation in mice and TNF-α-induced monocyte adhesion to endothelial cells via suppressing IΚBα/NF-κB signaling pathway. J Nutr Biochem. 2015;26:293–302. doi: 10.1016/j.jnutbio.2014.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mun L, Jun MS, Kim YM, Lee YS, Kim HJ, Seo HG, Lee JH, Son KH, Lee DH, Kim YS, Park K, Chang KC. 7, 8-Didehydrocimigenol from Cimicifugae rhizoma inhibits TNF-α-induced VCAM-1 but not ICAM-1expression through up regulation of PPAR-γ in human endothelial cells. Food Chem Toxicol. 2011;49:166–172. doi: 10.1016/j.fct.2010.10.012. [DOI] [PubMed] [Google Scholar]
- 12.Jin M, Lee E, Yang JH, Lu Y, Kang S, Chang YC, Lee SH, Suh SJ, Kim CH, Chang HW. Deoxypodophyllotoxin inhibits the expression of intercellular adhesion molecule-1 induced by tumor necrosis factor-α in murine lung epithelial cells. Biol Pharm Bull. 2010;33:1–5. doi: 10.1248/bpb.33.1. [DOI] [PubMed] [Google Scholar]
- 13.Kelly TA, Jeanfavre DD, McNeil DW, Woska JR, Jr, Reilly PL, Mainolfi EA, Kishimoto KM, Nabozny GH, Zinter R, Bormann BJ, Rothlein R. Cutting edge: a small molecule antagonist of LFA-1-mediated cell adhesion. J Immunol. 1999;163:5173–5177. [PubMed] [Google Scholar]
- 14.Anderson ME, Siahaan TJ. Targeting ICAM-1/LFA-1 interaction for controlling autoimmune diseases: designing peptide and small molecule inhibitors. Peptides. 2003;24:487–501. doi: 10.1016/S0196-9781(03)00083-4. [DOI] [PubMed] [Google Scholar]
- 15.Kwon OE, Lee HS, Lee SW, Bae K, Kim K, Hayashi M, Rho MC, Kim YK. Dimeric sesquiterpenoids isolated from Chloranthus japonicus inhibited the expression of cell adhesion molecules. J Ethnopharmacol. 2006;104:270–277. doi: 10.1016/j.jep.2005.09.018. [DOI] [PubMed] [Google Scholar]
- 16.Choi YW, Kim HJ, Park SS, Chung JH, Lee HW, Oh SO, Kim BS, Kim JB, Chung HY, Yu BP, Kim CD, Yoon S. Inhibition of endothelial cell adhesion by the new anti-inflammatory agent α-iso-cubebene. Vasc Pharmacol. 2009;51:215–224. doi: 10.1016/j.vph.2009.05.008. [DOI] [PubMed] [Google Scholar]
- 17.Hunter MS, Corley DG, Carron CP, Rowold E, Kilpatrick BF, Durley RC. Four new clerodane diterpenes from the leaves of Casearia guianensis which inhibit the interaction of leukocyte function antigen 1 with intercellular adhesion molecule 1. J Nat Prod. 1997;60:894–899. doi: 10.1021/np970141n. [DOI] [PubMed] [Google Scholar]
- 18.Balmain A, Connolly JD. Minor diterpenoid constituents of Andrographis paniculata Nees. J Chem Soc Perkin. 1973;1:1247–1251. doi: 10.1039/p19730001247. [DOI] [Google Scholar]
- 19.Qizhen D, Jerz G, Winterhalter P. Separation of andrographolide and neoandrographolide from the leaves of Andrographis paniculata using high-speed counter-current chromatography. J Chromatogr A. 2003;984:147–151. doi: 10.1016/S0021-9673(02)01831-9. [DOI] [PubMed] [Google Scholar]
- 20.Chen L, Zhu H, Wang R, Zhou K, Jing Y, Qiu F. ent-Labdane diterpenoid lactone stereoisomers from Andrographis paniculata. J Nat Prod. 2008;71:852–855. doi: 10.1021/np0704452. [DOI] [PubMed] [Google Scholar]
- 21.Jiang CG, Li JB, Liu FR, Wu T, Yu M, Xu HM. Andrographolide inhibits the adhesion of gastric cancer cells to endothelial cells by blocking E-selectin expression. Anticancer Res. 2007;27:2439–2447. [PubMed] [Google Scholar]
- 22.Musza LL, Speight P, McElhiney S, Barrow CJ, Gillum AM, Cooper R, Killar LM. Cucurbitacins, cell adhesion inhibitors from Conobea scoparioides. J Nat Prod. 1994;57:1498–1502. doi: 10.1021/np50113a004. [DOI] [PubMed] [Google Scholar]
- 23.Touihri-Barakati I, Kallech-Ziri O, Ayadi W, Kovacic H, Hanchi B, Hosni K, Luis J. Cucurbitacin B purified from Ecballium elaterium (L.) A. Rich from Tunisia inhibits α5β1 integrin-mediated adhesion, migration, proliferation of human glioblastoma cell line and angiogenesis. Eur J Pharmacol. 2017;797:153–161. doi: 10.1016/j.ejphar.2017.01.006. [DOI] [PubMed] [Google Scholar]
- 24.Musza LL, Killar LM, Speight P, McElhiney S, Barrow C, Gillum AM, Copper R. Potent new cell adhesion inhibitory compounds from the root of Trichilia rubra. Tetrahedron. 1994;50:11369–11378. doi: 10.1016/S0040-4020(01)89279-6. [DOI] [Google Scholar]
- 25.Dutra RC, Claudino RF, Bento AF, Marcon R, Schmidt EC, Bouzon ZL, Pianowski LF, Calixto JB. Preventive and therapeutic euphol treatment attenuates experimental colitis in mice. PLoS One. 2011;6:e27122. doi: 10.1371/journal.pone.0027122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shi MD, Shih YW, Lee YS, Cheng YF, Tsai LY. Suppression of 12-O-tetradecanoylphorbol-13-acetate-induced MCF-7 breast adenocarcinoma cells invasion/migration by α-tomatine through activating PKCα/ERK/NF-κB-dependent MMP-2/MMP-9 expressions. Cell Biochem Biophys. 2013;66:161–174. doi: 10.1007/s12013-012-9465-8. [DOI] [PubMed] [Google Scholar]
- 27.Rho MC, Kwon OE, Kim K, Lee SW, Chung MY, Kim YH, Hayashi M, Lee HS, Kim YK. Inhibitory effects of manassantin A and B isolated from the roots of Saururus chinensis on PMA-induced ICAM-1 expression. Planta Med. 2003;69:1147–1149. doi: 10.1055/s-2003-818007. [DOI] [PubMed] [Google Scholar]
- 28.Wang J, Zhao Y, Xu Q. Astilbin prevents concanavalin A-induced liver injury by reducing TNF- α production and T lymphocytes adhesion. J Pharm Pharmacol. 2004;56:495–502. doi: 10.1211/0022357023033. [DOI] [PubMed] [Google Scholar]
- 29.Kumar S, Arya P, Mukherjee C, Singh BK, Singh N, Parmar VS, Prasad AK, Ghosh B. Novel aromatic ester from Piper longum and its analogues inhibit expression of cell adhesion molecules on endothelial cells. Biochemistry. 2005;44:15944–15952. doi: 10.1021/bi050941u. [DOI] [PubMed] [Google Scholar]
- 30.Kumar S, Singh BK, Arya P, Malhotra S, Thimmulappa R, Prasad AK, Van der Eycken E, Olsen CE, DePass AL, Biswal S, Parmar VS, Ghosh B. Novel natural product-based cinnamates and their thio and thiono analogs as potent inhibitors of cell adhesion molecules on human endothelial cells. Eur J Med Chem. 2011;46:5498–5511. doi: 10.1016/j.ejmech.2011.09.008. [DOI] [PubMed] [Google Scholar]
- 31.Kumar S, Singh BK, Kalra N, Kumar V, Kumar A, Prasad AK, Raj HG, Parmar VS, Ghosh B. Novel thiocoumarins as inhibitors of TNF-α induced ICAM-1 expression on human umbilical vein endothelial cells (HUVECs) and microsomal lipid peroxidation. Bioorg Med Chem. 2005;13:1605–1613. doi: 10.1016/j.bmc.2004.12.013. [DOI] [PubMed] [Google Scholar]
- 32.Lee SW, Chang JS, Lim JH, Kim MS, Park SJ, Jeong HJ, Kim MS, Lee WS, Rho MC. Quinolone alkaloids from Evodiae fructus inhibit LFA-1/ICAM-1-mediated cell adhesion. Bull Korean Chem Soc. 2010;31:64–68. doi: 10.5012/bkcs.2010.31.01.064. [DOI] [Google Scholar]
- 33.Hibberd AD, Trevillian PR, Clark DA, McElduff P, Cowden WB. The effects of Castanospermine, an oligosaccharide processing inhibitor, on mononuclear/endothelial cell binding and the expression of cell adhesion molecules. Transpl Immunol. 2012;27:39–47. doi: 10.1016/j.trim.2012.05.002. [DOI] [PubMed] [Google Scholar]
- 34.Lee SW, Hwang BS, Kim MH, Park CS, Lee WS, Oh HM, Rho MC. Inhibition of LFA-1/ICAM-1-mediated cell adhesion by stilbene derivatives from Rheum undulatum. Arch Pharm Res. 2012;35:1763–1770. doi: 10.1007/s12272-012-1008-8. [DOI] [PubMed] [Google Scholar]
- 35.Liang Q, Yu F, Cui X, Duan J, Wu Q, Nagarkatti P, Fan D. Sparstolonin B suppresses lipopolysaccharide-induced inflammation in human umbilical vein endothelial cells. Arch Pharm Res. 2013;36:890–896. doi: 10.1007/s12272-013-0120-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Minamiguchi K, Kumagai H, Masuda T, Kawada M, Ishizuka M, Takeuchi T. Thiolutin, an inhibitor of HUVEC adhesion to vitronectin, reduces paxillin in HUVECs and suppresses tumor cell-induced angiogenesis. Int J Cancer. 2001;93:307–316. doi: 10.1002/ijc.1321. [DOI] [PubMed] [Google Scholar]
- 37.Liu G, Bode A, Ma WY, Sang S, Ho CT, Dong Z. Two novel glycosides from the fruits of Morinda citrifolia (noni) inhibit AP-1 transactivation and cell transformation in the mouse epidermal JB6 cell line. Cancer Res. 2001;61:5749–5756. [PubMed] [Google Scholar]
- 38.Selistre-de-Araujo HS, Cominetti MR, Terruggi CH, Mariano-Oliveira A, De Freitas MS, Crepin M, Figueiredo CC, Morandi V. Alternagin-C, a disintegrin-like protein from the venom of Bothrops alternatus, modulates alpha2beta1 integrin-mediated cell adhesion, migration and proliferation. Braz J Med Biol Res. 2005;38:1505–1511. doi: 10.1590/S0100-879X2005001000007. [DOI] [PubMed] [Google Scholar]
- 39.Kim J, Lee H, Lee Y, Oh BG, Cho C, Kim Y, Shin M, Hong M, Jung SK, Bae H. Inhibition effects of Moutan Cortex Radicis on secretion of eotaxin in A549 human epithelial cells and eosinophil migration. J Ethnopharmacol. 2007;114:186–193. doi: 10.1016/j.jep.2007.07.038. [DOI] [PubMed] [Google Scholar]
- 40.Xu CX, Jin H, Chung YS, Shin JY, Woo MA, Lee KH, Palmos GN, Choi BD, Cho MH. Chondroitin sulfate extracted from the Styela clava tunic suppresses TNF-alpha-induced expression of inflammatory factors, VCAM-1 and iNOS by blocking Akt/NF-κB signal in JB6 cells. Cancer Lett. 2008;264:93–100. doi: 10.1016/j.canlet.2008.01.022. [DOI] [PubMed] [Google Scholar]
- 41.Lin CP, Chen YH, Chen JW, Leu HB, Liu TZ, Liu PL, Huang SL. Cholestin (Monascus purpureus rice) inhibits homocysteine-induced reactive oxygen species generation, nuclear factor-κB activation, and vascular cell adhesion molecule-1 expression in human aortic endothelial cells. J Biomed Sci. 2008;15:183–196. doi: 10.1007/s11373-007-9212-0. [DOI] [PubMed] [Google Scholar]
- 42.Sánchez EE, Rodríguez-Acosta A, Palomar R, Lucena SE, Bashir S, Soto JG, Pérez JC. Colombistatin: a disintegrin isolated from the venom of the South American snake (Bothrops colombiensis) that effectively inhibits platelet aggregation and SK-Mel-28 cell adhesion. Arch Toxicol. 2009;83:271–279. doi: 10.1007/s00204-008-0358-y. [DOI] [PubMed] [Google Scholar]
- 43.Zapolska-Downar D, Naruszewicz M. Propionate reduces the cytokine-induced VCAM-1 and ICAM-1 expression by inhibiting nuclear factor-κB (NF-κB) activation. J Physiol Pharmacol. 2009;60:123–131. [PubMed] [Google Scholar]
- 44.Chen C, Jin X, Meng X, Zheng C, Shen Y, Wang Y. Inhibition of TNFα-induced adhesion molecule expression by (Z)-(S)-9-octadecenamide, N-(2-hydroxyethyl,1-methyl) Eur J Pharmacol. 2011;660:305–309. doi: 10.1016/j.ejphar.2011.04.009. [DOI] [PubMed] [Google Scholar]
- 45.Tong H, Song J, Zhang Z, Mao D, Sun G, Jiang G. Inhibitory function of P-selectin-mediated leukocyte adhesion by the polysaccharides from Sanguisorba officinalis. Pharm Biol. 2015;53:345–349. doi: 10.3109/13880209.2014.919597. [DOI] [PubMed] [Google Scholar]
- 46.Hayashi M, Kim YP, Hiraoka H, Natori M, Takamatsu S, Kawakubo T, Masuma R, Komiyama K, Ōmura S. Macrosphelide, a novel inhibitor of cell-cell adhesion molecule. I. Taxonomy, fermentation, isolation and biological activities. J Antibiot. 1995;48:1435–1439. doi: 10.7164/antibiotics.48.1435. [DOI] [PubMed] [Google Scholar]
- 47.Takamatsu S, Kim YP, Hayashi M, Hiraoka H, Natori M, Komiyama K, Ōmura S. Macrosphelide, a novel inhibitor of cell–cell adhesion molecule. II. Physiochemical properties and structural elucidation. J Antibiot. 1996;49:95–98. doi: 10.7164/antibiotics.49.95. [DOI] [PubMed] [Google Scholar]
- 48.Takamatsu S, Hiraoka H, Kim YP, Hayashi M, Natori M, Komiyama K, Ōmura S. Macrosphelides C and D, novel inhibitors of cell adhesion. J Antibiot. 1997;50:878–880. doi: 10.7164/antibiotics.50.878. [DOI] [PubMed] [Google Scholar]
- 49.Fukami A, Taniguchi Y, Nakamura T, Rho MC, Kawaguchi K, Hayashi M, Komiyama K, Ōmura S. New members of the macrosphelides from Microsphaeropsis sp. FO-5050 IV. J Antibiot. 1999;52:501–504. doi: 10.7164/antibiotics.52.501. [DOI] [PubMed] [Google Scholar]
- 50.Numata A, Iritani M, Yamada T, Minoura K, Matsumura E, Yamori T, Tsuruo T. Novel antitumour metabolites produced by a fungal strain from a sea hare. Tetrahedron Lett. 1997;38:8215–8218. doi: 10.1016/S0040-4039(97)10198-8. [DOI] [Google Scholar]
- 51.Yamada T, Iritani M, Doi M, Minoura K, Ito T, Numata A. Absolute stereostructures of cell-adhesion inhibitors, macrosphelides C, E–G and I, produced by a Periconia species separated from an Aplysia sea hare. J Chem Soc Perkin. 2001;1:3046–3053. doi: 10.1039/b104337b. [DOI] [Google Scholar]
- 52.Yamada T, Iritani M, Minoura K, Numata A, Kobayashi Y, Wang YG. Absolute stereostructures of cell adhesion inhibitors, macrosphelides H and L, from Periconia byssoides OUPS-N133. J Antibiot. 2002;55:147–154. doi: 10.7164/antibiotics.55.147. [DOI] [PubMed] [Google Scholar]
- 53.Nakamura H, Ono M, Yamada T, Numata A, Akita H. Determination of the absolute stereostructure of seco-macrosphelide E produced by a fungal strain from a sea hare. Chem Pharm Bull. 2002;50:303–306. doi: 10.1248/cpb.50.303. [DOI] [PubMed] [Google Scholar]
- 54.Yamada T, Minoura K, Tanaka R, Numata A. Cell-adhesion inhibitors produced by a sea hare-derived Periconia sp. J Antibiot. 2007;60:370–375. doi: 10.1038/ja.2007.50. [DOI] [PubMed] [Google Scholar]
- 55.Hayashi M, Ōmura S. Actions of macrosphelide, a cell adhesion inhibitor. Jpn J Antibiot. 1998;51(Suppl A):68–71. [PubMed] [Google Scholar]
- 56.Fukami A, Iijima K, Hayashi M, Komiyama K, Ōmura S. Macrosphelide B suppressed metastasis through inhibition of adhesion of sLex/E-selectin molecules. Biochem Biophys Res Commun. 2002;291:1065–1070. doi: 10.1006/bbrc.2002.6572. [DOI] [PubMed] [Google Scholar]
- 57.Takamatsu S. Macrosphelides, cell–cell adhesive inhibitor of microbial origin. Showa Univ J Pharm Sci. 2013;4:37–46. [Google Scholar]
- 58.Sunazuka T, Hirose T, Harigaya Y, Takamatsu S, Hayashi M, Komiyama K, Ōmura S, Paul A, Sprengeler PA, Smith AB., III Relative and absolute stereochemistries and total synthesis of (+)-macrosphelides A and B, potent, orally bioavailable inhibitors of cell–cell adhesion. J Am Chem Soc. 1997;119:10247–10248. doi: 10.1021/ja971657w. [DOI] [Google Scholar]
- 59.Sunazuka T, Hirose T, Ōmura S. Efficient total synthesis of novel bioactive microbial metabolites. Acc Chem Res. 2008;41:302–314. doi: 10.1021/ar6000044. [DOI] [PubMed] [Google Scholar]
- 60.Takahashi T, Kusaka S, Doi T, Sunazuka T, Ōmura S. A combinatorial synthesis of a macrosphelide library utilizing a palladium-catalyzed carbonylation on a polymer support. Angew Chem Int Ed Engl. 2003;42:5230–52304. doi: 10.1002/anie.200352229. [DOI] [PubMed] [Google Scholar]
- 61.Paek SM. Development of advanced macrosphelides: potent anticancer agents. Molecules. 2015;20:4430–4449. doi: 10.3390/molecules20034430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Hommel U, Weber HP, Oberer L, Naegeli HU, Oberhauser B, Foster CA. The 3D-structure of a natural inhibitor of cell adhesion molecule expression. FEBS Lett. 1996;379:69–73. doi: 10.1016/0014-5793(95)01453-5. [DOI] [PubMed] [Google Scholar]
- 63.Boger DL, Keim H, Oberhauser B, Schreiner EP, Foster CA. Total synthesis of HUN-7293. J Am Chem Soc. 1999;121:6197–6205. doi: 10.1021/ja990918u. [DOI] [Google Scholar]
- 64.Schreiner EP, Kern M, Steck A, Foster CA. Synthesis of ether analogues derived from HUN-7293 and evaluation as inhibitors of VCAM-1 expression. Bioorg Med Chem Lett. 2004;14:5003–5006. doi: 10.1016/j.bmcl.2004.07.012. [DOI] [PubMed] [Google Scholar]
- 65.Koiwa T, Nakano T, Takahashi S, Koshino H, Yamazaki M, Takashi T, Nakagawa A. Adxanthromycin: a new inhibitor of ICAM-1/LFA-1 mediated cell adhesion from Streptomyces sp. NA-148. J Antibiot. 1999;52:198–200. doi: 10.7164/antibiotics.52.198. [DOI] [PubMed] [Google Scholar]
- 66.Nakano T, Koiwa T, Takahashi S, Nakagawa A. Adxanthromycins A and B, new inhibitors of ICAM-1/LFA-1 mediated cell adhesion molecule from Streptomyces sp. NA-148. I. Taxonomy, production, isolation and biological activities. J Antibiot. 2000;53:12–18. doi: 10.7164/antibiotics.53.12. [DOI] [PubMed] [Google Scholar]
- 67.Takahashi S, Nakano T, Koiwa T, Noshita T, Funayama S, Koshino H, Nakagawa A. Adxanthromycins A and B, new inhibitors of ICAM-1/LFA-1 mediated cell adhesion molecule from Streptomyces sp. NA-148. II. Physico-chemical properties and structure elucidation. J Antibiot. 2000;53:163–170. doi: 10.7164/antibiotics.53.163. [DOI] [PubMed] [Google Scholar]
- 68.Kawamura N, Tsuji E, Watanabe Y, Tsuchihashi K, Takako T (2000) Benzopyran derivatives, their manufacture with Streptomyces Mer-88 species, and their use for treatment of asthma and rheumatoid arthritis. Jpn. Kokai Tokkyo Koho: JP 2000072766 A 20000307
- 69.Xu MJ, Liu XJ, Zhao YL, Liu D, Xu ZH, Lang XM, Ao P, Lin WH, Yang SL, Zhang ZG, Xu J. Identification and characterization of an anti-fibrotic benzopyran compound isolated from mangrove-derived Streptomyces xiamenensis. Mar Drugs. 2012;10:639–654. doi: 10.3390/md10030639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Takamatsu S, Zhang Q, Schrader KK, elSohly HN, Walker LA. Characterization of Mycotypha metabolites found to be inhibitors of cell adhesion molecules. J Antibiot. 2002;55:585–592. doi: 10.7164/antibiotics.55.585. [DOI] [PubMed] [Google Scholar]
- 71.Schön MP, Krahn T, Schön M, Rodriguez ML, Antonicek H, Schultz JE, Ludwig RJ, Zollner TM, Bischoff E, Bremm KD, Schramm M, Henninger K, Kaufmann R, Gollnick HP, Parker CM, Boehncke WH. Efomycine M, a new specific inhibitor of selectin, impairs leukocyte adhesion and alleviates cutaneous inflammation. Nat Med. 2002;8:366–372. doi: 10.1038/nm0402-366. [DOI] [PubMed] [Google Scholar]
- 72.Zhang Q, Schrader KK, ElSohly HN, Takamatsu S. New cell–cell adhesion inhibitors from Streptomyces sp. UMA-044. J Antibiot. 2003;56:673–681. doi: 10.7164/antibiotics.56.673. [DOI] [PubMed] [Google Scholar]
- 73.Weitz-Schmidt G. Lymphocyte function-associated antigen-1 blockade by statins: molecular basis and biological relevance. Endothelium. 2003;10:43–47. doi: 10.1080/10623320303360. [DOI] [PubMed] [Google Scholar]
- 74.Kallen J, Welzenbach K, Ramage P, Geyl D, Kriwacki R, Legge G, Cottens S, Weitz-Schmidt G, Hommel U. Structural basis for LFA-1 inhibition upon lovastatin binding to the CD11a I-domain. J Mol Biol. 1999;292:1–9. doi: 10.1006/jmbi.1999.3047. [DOI] [PubMed] [Google Scholar]
- 75.Ohno O, Ikeda Y, Sawa R, Igarashi M, Kinoshita N, Suzuki Y, Miyake K, Umezawa K. Isolation of heptadepsin, a novel bacterial cyclic depsipeptide that inhibits lipopolysaccharide activity. Chem Biol. 2004;11:1059–1070. doi: 10.1016/j.chembiol.2004.05.016. [DOI] [PubMed] [Google Scholar]
- 76.Yamada T, Iritani M, Minoura K, Kawai K, Numata A. Peribysins A–D, potent cell-adhesion inhibitors from a sea hare-derived culture of Periconia species. Org Biomol Chem. 2004;2:2131–2135. doi: 10.1039/B404459B. [DOI] [PubMed] [Google Scholar]
- 77.Yamada T, Doi M, Miura A, Harada W, Hiramura M, Minoura K, Tanaka R, Numata A. Absolute stereostructures of cell-adhesion inhibitors, peribysins A, E, F and G, produced by a sea hare-derived Periconia sp. J Antibiot. 2005;58:185–191. doi: 10.1038/ja.2005.21. [DOI] [PubMed] [Google Scholar]
- 78.Yamada T, Minoura K, Tanaka R, Numata A. Cell-adhesion inhibitors produced by a sea hare-derived Periconia sp. II. Absolute stereostructures of peribysins H and I. J Antibiot. 2006;59:345–350. doi: 10.1038/ja.2006.48. [DOI] [PubMed] [Google Scholar]
- 79.Angeles AR, Dorn DC, Kou CA, Moore MA, Danishefsky SJ. Total synthesis of peribysin E necessitates revision of the assignment of its absolute configuration. Angew Chem Int Ed Eng. 2007;46:1451–1454. doi: 10.1002/anie.200604308. [DOI] [PubMed] [Google Scholar]
- 80.Golik J, Dickey JK, Todderud G, Lee D, Alford J, Huang S, Klohr S, Eustice D, Aruffo A, Agler ML. Isolation and structure determination of sulfonoquinovosyl dipalmitoyl glyceride, a P-selectin receptor inhibitor from the alga Dictyochloris fragrans. J Nat Prod. 1997;60:387–389. doi: 10.1021/np9606761. [DOI] [PubMed] [Google Scholar]
- 81.Takamatsu S, Nagle DG, Gerwick WH. Secondary metabolites from marine cyanobacteria and algae inhibit LFA-1/ICAM-1 mediated cell adhesion. Planta Med. 2004;70:127–131. doi: 10.1055/s-2004-815488. [DOI] [PubMed] [Google Scholar]
- 82.Rao BN, Anderson MB, Musser JH, Gilbert JH, Schaefer ME, Foxall C, Brandley BK. Sialyl Lewis X mimics derived from a pharmacophore search are selectin inhibitors with anti-inflammatory activity. J Biol Chem. 1994;269:19663–19666. [PubMed] [Google Scholar]
- 83.Zimmerman T, Blanco FJ. Inhibitors targeting the LFA-1/ICAM-1 cell-adhesion interaction: design and mechanism of action. Curr Pharm Des. 2008;14:2128–2139. doi: 10.2174/138161208785740225. [DOI] [PubMed] [Google Scholar]
- 84.Moral MEG, Siahaan TJ. Conjugates of cell adhesion peptides for therapeutics and diagnostics against cancer and autoimmune diseases. Curr Top Med Chem. 2018;18:11–19. doi: 10.2174/1568026618666180118154514. [DOI] [PMC free article] [PubMed] [Google Scholar]