Abstract
Potassium ions significantly contribute to the resting membrane potential of cells and, therefore, extracellular K+ concentration is a crucial regulator of cell excitability. Altered concentrations of extracellular K+ affect the resting membrane potential and cellular excitability by shifting the equilibria between closed, open and inactivated states for voltage-dependent ion channels that underlie action potential initiation and conduction. Hence, it is valuable to directly measure extracellular K+ dynamics in health and diseased states. Here, we describe how to make, calibrate and use monopolar K+-selective microelectrodes. We deployed them in adult hippocampal brain slices to measure electrically evoked K+ concentration dynamics. The judicious use of such electrodes is an important part of the tool-kit needed to evaluate cellular and biophysical mechanisms that control extracellular K+ concentrations in the nervous system.
Keywords: Neuroscience, Issue 135, Potassium, K+, ion-selective microelectrode, neuroscience, electrophysiology, brain slice, astrocyte, mouse, homeostasis, synapses
Introduction
Potassium ion concentrations are tightly regulated in the brain, and their fluctuations exert a powerful influence on the resting membrane potential of all cells. In light of these critical contributions, an important goal of biology is to determine the cellular and biophysical mechanisms that are used to tightly regulate the concentration of K+ in the extracellular space in different organs of the body1,2. An important requirement in these studies is the ability to measure K+ concentrations accurately. Although many components which contribute to potassium homeostasis in the brain in healthy and diseased states have been identified3,4,5, further progress has been slowed due to the specialized nature of preparing ion selective microelectrodes for potassium measurement. Microelectrode sensors represent the gold standard for measuring K+ concentrations in vitro, in tissue slices and in vivo.
Newer approaches for K+ monitoring are under development using optical sensors, however these do not detect a biologically relevant range of K+ concentrations or have not been fully vetted in biological systems, although initial results appear promising6,7,8. Compared to optical sensors, microelectrodes are fundamentally limited to a point source measurement of ions, although electrode arrays could improve the spatial resolution9. This article focuses on the single-barreled microelectrode sensors for monitoring K+ dynamics.
In this work, we report detailed stepwise procedures to make K+ selective microelectrodes, using a valinomycin-based potassium ionophore that permits highly selective (104 fold K+ to Na+ selectivity) K+ movement over membranes10. A naturally occurring polypeptide, valinomycin acts as a K+ permeable pore and facilitates the flow of K+ down it's electrochemical gradient. We also describe how to calibrate the electrodes, how to store and use them and finally how to deploy them to measure K+ concentration dynamics in acute hippocampal brain slices from adult mice. The use of such electrodes together with genetically modified mice that lack specific ion channels proposed to regulate extracellular K+ dynamics should reveal the cellular mechanisms used by the nervous system to control the ambient concentration of K+ in the extracellular milieu.
Protocol
All animal experiments were conducted in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals and were approved by the Chancellor's Animal Research Committee at the University of California, Los Angeles. All mice were housed with food and water available ad libitum in a 12 h light-dark environment. All animals were healthy with no obvious behavioral changes, were not involved in previous studies, and were sacrificed during the light cycle. Data for experiments were collected from adult mice (6-8 weeks old for all experiments).
1. Preparation of K+ selective microelectrodes
- Silanization of borosilicate glass
- Remove sufficient glass capillaries from packaging and place into a 50 mL conical tube. Fill conical tube to top with 1 M HCl. Wash electrodes with gentle agitation in HCl overnight or for a minimum of 6 hours.
- Briefly rinse capillaries with 70% ethanol and then dry completely at 100-120 °C for 6-8 hours. Store washed capillaries in containers with anhydrous calcium sulfate desiccant for up to 4 weeks before further use.
- Prior to silanization, pull capillaries to a fine tip using a microelectrode puller. The microelectrodes that we use are approximately 2-5 microns in diameter. Always handle washed capillaries with gloves, as oils from skin can interfere with silanization.
- Place microelectrodes into a glass container so that the electrodes are elevated from the bottom to prevent tip breakage. Fix the microelectrodes to the container using autoclavable tape or similar adhesive tape.
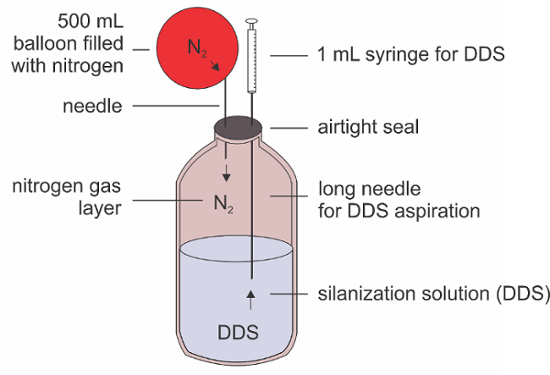
- Remove approximately 0.5 mL of 5% dichlorodimethylsilane (DDS) silanization solution from its container using the nitrogen replacement method (see Figure 2). Fill a balloon with nitrogen gas and attach a syringe or tube and needle to the balloon. Insert the needle into the DDS container while drawing up DDS into a separate syringe via a longer needle.
- Apply the silanization solution dropwise to the tips of the pipettes and immediately cover. Place the container holding the microelectrodes with silanization solution into a pre-warmed (170-180 °C) laboratory oven for 10-12 hours or at 200-220 °C for 30 minutes
- After the incubation, turn off the oven and then remove the plate from the oven. Be careful when removing the plate from the incubator, as it is extremely hot. Place the plate on a bench at room temperature for 10-15 minutes to allow the glassware to cool.
- Remove the microelectrodes from the plate (using a razor blade or scalpel blade to cut the tape) and place them into a desiccant filled airtight container. Silanized microelectrodes kept free of moisture can be used for up to 1 week following silanization.
- Priming the electrodes
- Prepare a stock solution of K+ ionophore cocktail: 5% w/v valinomycin, 93% v/v 1,2-dimethyl-3-nitrobenzene, 2% w/v potassium tetrakis(4-chlorophenyl)borate11. This solution is a faint yellow color. Keep in an airtight, opaque container at room temperature. If properly stored this solution can last many months.
- Prepare a stock solution of 10 mM HEPES buffered 300 mM NaCl at pH 7.4. Fix the electrode into a clamp and backfill with the buffered NaCl using a 28G microfil tip connected to a syringe. Observe that the saline solution has reached the end of the microelectrode tip. Confirm that the microelectrode is free of large bubbles that could interfere with the flow of current.
- Break the tip of the electrode to approximately 10-20 µm wide, using the blunt side of a scalpel or razor blade.
- Using a micropipette, apply a small droplet (~0.1 µl) of the K+ ionophore near the tip of the microelectrode. If the electrode has been properly silanized the droplet will be absorbed into the broken tip. Fill the electrode to 1-2 mm with the K+ ionophore, and remove excess using tissue paper.
2. Calibration of K+ Selective Microelectrodes
- Preparation of calibration solutions
- Prepare solutions of various concentrations of KCl in equal osmolarity artificial cerebrospinal fluid (ACSF) by replacing NaCl with KCl. We used 0.1, 1, 4.5, 10 and 100 mM K+ ACSF, for calibration of the electrodes. The recipes for these calibration solutions are listed in Table 1.
| Chemical | MW | final mM | 0.1 mM [K+] | 1 mM [K+] | 4.5 mM [K+] | 10 mM [K+] | 100 mM [K+] |
| (g / mol) | |||||||
| NaCl | 58.44 | varies | 1.51 g | 1.50 g | 1.44 g | 1.4 g | 0.345 g |
| KCl | 1 M stock | varies | 20 µl | 200 µl | 900 µl | 2 ml | 20 ml |
| CaCl2 | 1 M stock | 2 | 400 µl | ||||
| MgCl2 | 1 M stock | 1 | 200 µl | ||||
| NaH2PO4 | 119.98 | 1.2 | 0.29 g | ||||
| NaHCO3 | 84.01 | 26 | 0.437 g | ||||
| D-Glucose | 180.16 | 10 | 0.360 g | ||||
| Water | q.s. 200 ml |
Table 1. Potassium calibration solutions
- Microelectrode calibration
- Bubble all solutions with 95% O2/5% CO2 for at least 20 minutes before beginning the experiment. Begin perfusing the bath with 4.5 mM [K+] ACSF at a rate of 3 mL per minute. Place the K+ selective electrode into electrode holder attached to the electrode headstage on the manipulator. This headstage is connected to an appropriate amplifier. Insert the tip of the electrode into the bath perfusate.
- Ensure the Ag/AgCl ground electrode is bathed in the same solution and that the flow is steady. Apply calibration solutions in a stepwise fashion and record the potential changes in mV across the electrode tip. Wait for the potential at the electrode tip to reach a stable value before switching to the next solution
- Measure the steady state voltage change in response to the application of the calibration solutions to the electrode tip. Confirm that the slope of the electrode response is at least 52 and no greater than 58 mV per log change in [K+].
3. Preparation of Acute Hippocampal Brain Slices
- Preparation of slice solutions
- Prepare 500 mL sucrose cutting solution composed of: 194 mM sucrose, 30 mM NaCl, 4.5 mM KCl, 10 mM D-glucose, 1 mM MgCl2, 1.2 mM NaH2PO4, and 26 mM NaHCO3, 290-300 mOsm, saturated with 95% O2 and 5% CO2.
- Prepare 1-2 liters of recording solution (ACSF) composed of: 124 mM NaCl, 4.5 mM KCl, 1 mM MgCl2, 10 mM D-glucose, 2 mM CaCl2, 1.2 mM NaH2PO4, and 26 mM NaHCO3; pH 7.3 - 7.4 (after bubbling), 290 - 300 mOsm, saturated with 95% O2 and 5% CO2. Fill a beaker containing a brain slice holder chamber with recording solution and keep it at 32-34 °C. Fill the vibratome chamber with ice-water slush.
- Acute slice preparation
- Deeply anesthetize a mouse by placing it in a bell jar precharged with 2-3 mL isoflurane. Check for toe pinch reflex, and if non-responsive, rapidly decapitate it using a pair of sharp shears or guillotine as your animal protocol requires.
- Make a 2-3 cm incision using shears from the caudal portion of the skull to cut the scalp along the midline. While manually retracting the scalp, make two 1 cm horizontal incisions from the foramen magnum along the sides of the skull. Then, using fine shears, cut an incision the length of the skull, along the midline from the back of the skull to the nose.
- Using fine forceps inserted near the midline, retract the incised skull in two portions. Extract the mouse brain from the skull and use a blade to remove the cerebellum and olfactory bulbs, which are respectively located at the caudal and rostral portions of the brain. These can be identified by the large fissures, which separate them from the cortex.
- Mount the brain block onto the vibratome tray using super glue. Fill the vibratome tray with ice cold cutting solution.
- Cut tissue sections on the coronal plane at 300 µm thickness. Usually 6 coronal hippocampal slices can be collected.
- After each section is cut, immediately transfer the slices to the slice holding beaker warmed to 32-34°C. Keep the sections at this temperature for 20 min before removing the beaker containing the sections and place this at room temperature for at least 20-30 minutes prior to recording.
4. Measurement of Electrically Evoked K+ Dynamics
- Setting up the slice preparation
- Gently place the brain slice in the bath using a Pasteur pipette and gently hold it in place with a platinum harp with nylon strings.
- Ensure the tips of the bipolar stimulating electrode are parallel to one another and are level with the plane of the slice. Slowly, over the course of 6-7 seconds, insert the electrodes into CA3 stratum radiatum approximately 40-50 µm deep to stimulate Schaffer collaterals. In coronal sections, CA3 can be approximately identified as the portion of the hippocampus proper lateral to the granule cell layer at the hippocampal genu, with the stratum radiatum falling medial and ventral to the pyramidal cell layer.
- Carefully insert the K+-selective electrode into CA1 stratum radiatum approximately 50 µm deep, by slowly lowering the electrode over approximately 3-4 seconds. Allow the potential to stabilize across the electrode before applying stimulations to the slice: this usually takes 5 to 10 minutes. If the slice exhibits spontaneous changes in extracellular K+ then discard and repeat this process with a new slice.
- Measure evoked K+ release
- Apply trains of electrical stimulation (8 pulses) by manually depressing the trigger on the stimulator while digitally recording responses. Apply stimulation at 10 Hz and 1 ms pulse width, starting at 10 µA stimulus amplitude.
- Apply increasing stimulation amplitudes by a factor of 2 until a maximum K+ response amplitude is detected. If you do not see any response, move the position of the K+ electrode closer to the stimulation site in 100 µm in increments
- Determine the stimulus amplitude that produces the half maximal response. In our experience, this is between 40-160 µA, depending upon the preparation quality, age of the animal, and the distance between stimulation electrodes and K+ selective electrode.
- In the same slice, using a stimulus amplitude at one step lower than the amplitude that produces the half maximal response (e.g. if 80 µA produces a half-maximal response, use 40 µA) apply stimulus trains of increasing number of pulses. Initially, we have used trains of 1, 2, 4, 8, 16, 32, 64 and 128 pulses.
- To confirm that K+ signals are mediated by action potential firing of the electrically stimulated Schaffer collaterals bath, apply 0.5 µM TTX in ACSF for 10 minutes and repeat the stimulation protocol. No evoked responses should be observed.
- After finishing the slice experiment, confirm the electrode has maintained its responsivity by re-calibrating the electrode in the calibration solutions and ensuring the response has not deviated by more than 10% from the initial calibration
Representative Results
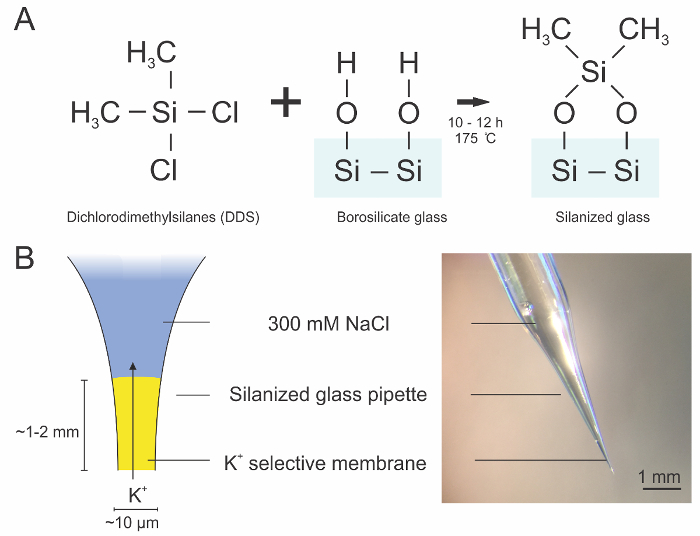
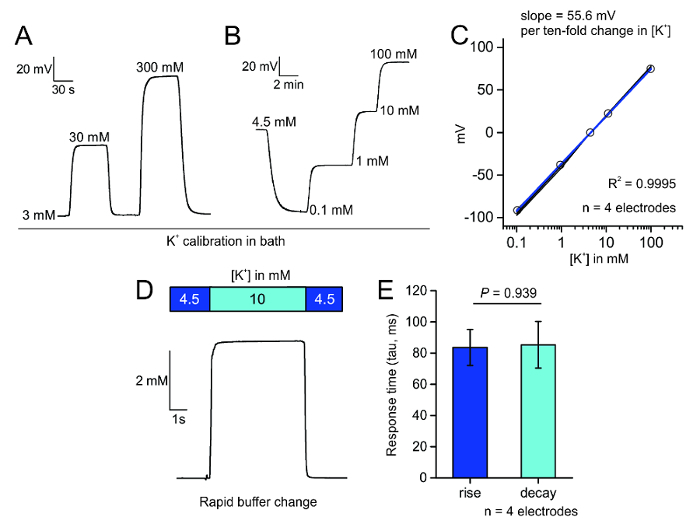
For selective measurement of extracellular K+, we prepared ion-selective microelectrodes coated with a hydrophobic layer through silanization of clean borosilicate glass pipettes (Figure 1A). This coating enables the K+ ionophore containing valinomycin to rest at the tip of the electrode and permit only K+ flux through a narrow opening at the electrode tip (Figure 1B). After priming the electrodes with the backfilled saline solution and the K+ ionophore, the electrodes can be tested for their rapid and linear response to stepwise changes in bath K+ concentrations (Figure 3A) and for their response to bath K+ changes over the calibration range (Figure 3B) in saline or ACSF in a manner predicted by the Nernst equation2. The change in the steady state potential can be plotted against the bath K+ concentration in order to determine the slope of the line, which should approximately be 58.2 mV per log [K+], according to the Nernst equation, and no less than 52 mV per log [K+] (Figure 3C). We additionally tested the responsiveness of the K+ selective electrodes and found that they responded to a 5.5 mM change in K+ with rise and decay time constants of approximately 85 ms (Figure 3D,E).
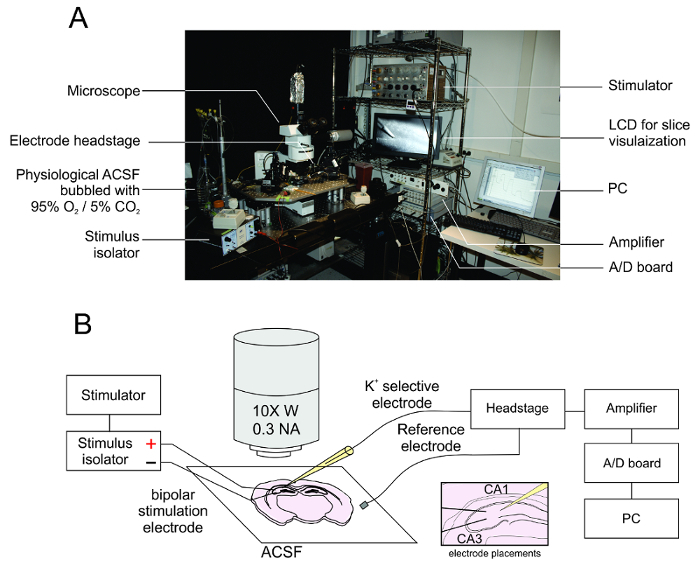
The electrophysiological recording rig consists of a standard upright microscope connected to an LCD display for identifying the placement of the stimulating and recording electrodes. No special optics are needed for visual placing of the K+ and stimulation electrodes; we use a 5x or 10x objective lens and white light from a halogen bulb, but a white LED could be used instead. The stimulating electrode is connected to the output of a stimulus isolator, which delivers depolarizing current via the timed delivery of pulses from a stimulator or other such timing device. In other words, the stimulator delivers trains of 2 V, 10-20 Hz timing pulses to the stimulus isolator. Upon receiving these pulses, the stimulus isolator then delivers the desired current to the stimulation electrodes. The recording electrode is connected to an electrode holder, connected to a headstage, amplifier and A/D board, which interface to a PC with electrophysiological recording software (Figure 4A). After the electrode has been successfully calibrated and the acute slices have been prepared, the slice can be placed in the ACSF perfusate. To stimulate the Schaffer collaterals, the K+-selective electrode is placed within the CA1 stratum radiatum and the field stimulation electrode is placed within the CA3 (Figure 4B).
Once the electrodes have been placed and the K+ recording has reached a stable baseline, then pulses of increasing current amplitude can be applied to the slice (Figure 5, top). The waveform of this activity appears as a rapid increase in K+ with an exponential decay rate, which is abolished with TTX application (Figure 5, bottom).
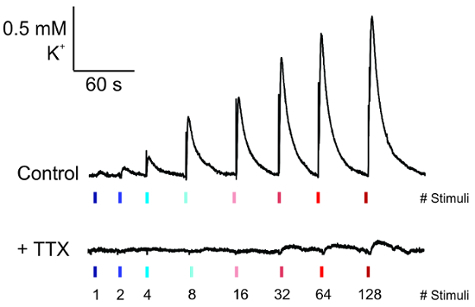
Figure 1:Diagram of silanization reaction and K+ selective microelectrode architecture. A. Schematic representation of silanization reaction that occurs between the exposed polar hydroxyl groups of the borosilicate glass and the silanization reagent dichlorodimethylsilane (DDS). This reaction renders the surface of the glass hydrophobic, which allows the K+ ionophore to form a thin membrane. B. Diagram of the K+ selective microelectrode. The electrode is backfilled with saline solution and the K+ selective solution is place in a 1-2 mm thick layer at the tip. Please click here to view a larger version of this figure.
Figure 2: Diagram of DDS extraction. Schematic representation of the nitrogen replacement procedure for DDS extraction from a container. DDS is volatile and flammable and can react violently with atmospheric gases when it is in high concentrations therefore it is necessary to replace the DDS removed with inert nitrogen gas. A balloon filled with nitrogen is connected to a needle via a syringe or appropriate tubing. This needle is inserted through the sealant on the container allowing nitrogen gas (N2) flow into the container. Separately, a long (3-10 cm) needle is connected to a 1 mL syringe and inserted into the container. This syringe is then used to extract DDS, while only nitrogen gas can enter the container. Please click here to view a larger version of this figure.
Figure 3: Calibration of microelectrodes. A. Bath perfusion application of different K+ concentrations in saline can rapidly and reversibly produce Nernstian changes in the potential across the electrode tip. B. Stepwise application of K+ in ACSF evokes a characteristic and stable change in the electrode tip potential. C. Plot of the mV change of the K+ selective electrode in response to increasing concentrations of K+ in four electrodes; the R2 is 0.9995 for these four electrodes. D. Fast perfusion system bath application of 10 mM K+ causes a step response in voltage across the K+ selective electrode tip. E. Plot of the measured response times (tau in ms); no difference between rise and decay time was detected (mean ± SEM, p = 0.939, two sample t-test). Please click here to view a larger version of this figure.
Figure 4: Apparatus for extracellular K+ measurement. A. The electrophysiology rig consists of a microscope fixed to an anti-vibration table with an attached camera and LCD display for slice visualization. The K+ electrode is fixed to a headstage, amplifier and analog to digital board which outputs the signal to an attached PC with electrophysiological recording software. The electrical stimulation electrodes are connected to a stimulus isolator which varies the stimulation amplitude and a stimulator for timing stimulus delivery. B. Diagram of the slice preparation and the location of the placement of the various electrodes at the slice. CA3 can be approximately identified as the portion of the hippocampus proper lateral to the granule cell layer at the hippocampal genu, with the stratum radiatum falling rostral to the pyramidal cell layer. Please click here to view a larger version of this figure.
Figure 5: Measurement of electrically evoked K+ release. Representative traces of K+ release from the recording electrode under basal conditions (top) and their loss upon the application of tetrodotoxin (0.25 µM) for 5 minutes prior to recording (bottom). Please click here to view a larger version of this figure.
Discussion
The method that we describe here has allowed us to assess K+ dynamics in response to electrical stimulation of Schaffer collaterals in acute hippocampal slices from adult mice. Our method of preparing K+ ion selective microelectrodes is similar to earlier described procedures12,13,14,15. However, this method has advantages over alternative electrode configurations in that it is rapid and uncomplicated to prepare K+ selective microelectrodes. After appropriate calibration, these electrodes were found to robustly measure K+ dynamics in slices during electrical stimulation, and such responses were blocked by TTX. In these experiments, stimulations of 80-160 uA at 10 Hz were used; however, optimization of stimulation conditions for a particular experiment and for a brain area of interest will be required. These values are listed as a guide.
The slope of the response of the electrodes should be 58.2 mV per log [K+]. Such a value is predicted from the Nernst and Nicolsky-Eisenman equations for a K+ selective semi-permeable membrane; the latter better accounts for interactions between ions16. If the electrode does not respond in the predicted manner, this could be for one of two primary reasons. First the silanization could be inadequate, causing the membrane to be lost or salt bridges to form. Confirm that the membrane is intact by observation through the microscope, there should be a clear interface between the pipette solution and the membrane. Another reason, could be the presence of bubbles in the pipette that impede the flow of current from the silver chloride wire. If bubbles are observed, then remove the pipette and flick vigorously to remove them. If these solutions fail, re-make another K+ selective electrode or repeat silanization for longer or at higher temperatures. However, it is important to repeat these key controls every time a new brain region is studied or a new microelectrode is tested.
Two specific amplifiers were used in these experiments, but other amplifiers could be used so long as the input impedance is greater than or equal to 500 MΩ. Having calibrated the electrodes with both 500 MΩ and 5 GΩ input impedances, we found there was no difference in the slope of the voltage response with either setting over a range of K+ concentrations (56.9 ± 0.7 and 56.5 ± 0.9 mV per ten-fold change in [K+] for 500 MΩ and 5 GΩ input impedances, respectively; P = 0.759, paired t-test, n = 4 electrodes).
We also used fast solution switches to estimate the response time of the electrodes to a known jump in [K+] from 4.5 to 10 mM (Figure 3D). The electrodes responded with rise and decay times (tau) of 85 ± 12 and 85 ± 15 ms, respectively. In relation to this, the solution exchange kinetics for our custom fast solution switcher was 85 ± 27 ms. Hence, the K+ selective electrodes respond with kinetics as fast as the solution switcher we employed and far faster than the dynamics of K+ in the extracellular milieu as a result of Schaffer collateral stimulation (Figure 5). These data suggest that K+ selective electrodes can be used to estimate the kinetics of K+ accumulation and clearance in brain tissue. However, in future work appropriate controls and calibrations will be needed on a case-by-case basis. We suggest that it is valuable to spend time understanding the solution exchange kinetics in your recording chamber.
We found three critically important factors that influence the quality and robustness of K+ measurements in slices. The first is the quality of the preparation, both the tissue health and age of the animals used appear to be relevant. For this study, we have used C57/Bl6N mice that are ~12 weeks old. On rare occasions, we found slices with spontaneous K+ fluctuations amounting to ~0.1 mM and lasting 5-10 seconds; these slices were discarded. The second is the quality of the recording microelectrode. The primary issue is the time and temperature that is used for silanization of the pulled glass capillary. We recommend >170 °C for at least 6 hours (up to overnight is acceptable) or at 200 °C for 30 minutes. Insufficient heating of the electrodes with the silanization reagent can lead to electrodes that do not maintain a steady state potential because of gradual loss of the K+ ionophore. Additionally, when preparing the electrodes, we recommend placing a thin layer (1-2 mm) of the K+ ionophore in the tip with a diameter of 10-20 µm i.e. at approximately the size of an average cell body (Figure 1B). Do not break the tip excessively wide, or the K+ selective membrane will lose integrity and the electrode will fail. This step may require some practice to achieve tips of the correct size. K+ selective electrodes with a too fine tip or too thick of a layer can have sluggish responses, compared to properly constructed electrodes. The third factor is the distance between the stimulation electrode and the K+ selective electrode. We have used an inter-electrode distance of approximately 500 µm, however the optimal distance can vary considerably with the individual brain area and will need to be carefully considered for the particular experiment at hand.
In addition to the single barrel configuration, there are currently several methods for making K+-selective microelectrodes in bipolar and concentric formats17. Compared to the published descriptions of these methods, single channel electrodes have two main disadvantages: 1) a slightly larger tip diameter (~10 vs 4 µm), which could cause greater disruption of the extracellular space comparted to bipolar and concentric electrodes, and 2) incompatibility with simultaneous measurement of multiple ion species as in the bipolar electrodes. However, the single channel electrodes offer several advantages. Specifically, these electrodes can be fabricated in less than five minutes and are therefore more disposable and can be made and calibrated quickly before experiments. Therefore, the risk of electrode breakage during the course experiments is less of a concern. Additionally, because the ground electrode and recording electrode are physically separated by the volume of the bath, there is no chance for the formation of salt bridges at the tip of the electrode, which can lead to electrode failure in concentric and bipolar electrodes. The response time of the single channel electrodes is faster than bipolar electrodes and likely comparable to concentric electrodes (~20 ms), although our fast perfusion system only permitted a measurement of response times on the order of 80 ms (Figure 3E). Furthermore, these electrodes offer lower noise compared to bipolar electrodes, which have greater tip resistance and require the use of amplifiers with higher input resistance. Lastly, these electrodes do not require the use of a specialized micromanipulator or headstage as is required for concentric electrodes. On balance, the advantages of the construction and ease of use of single channel electrodes outweighs the disadvantages.
The approach that we used here to measure K+ dynamics in slices can be used in many brain regions to study K+ regulation. Although this protocol demonstrates the usage of K+-selective electrodes for the measurements of the dynamics of electrically evoked potassium ions in brain tissues, this protocol can be broadly used in many different tissues where it is desirable to measure K+ dynamics. Such situations may include spontaneous dynamics and changes in response to pharmacological, optogenetic, or chemogenetic cellular activation. These microelectrodes can be made with sufficient quality and reliability as to permit rapid integration of this technique into any laboratory toolbox. The detailed analyses of K+ concentrations in health and diseased states will enable further detection and quantification of how various molecular and cellular components contribute to resting K+ concentrations in the brain3,18,19.
Disclosures
The authors have nothing to disclose.
Acknowledgments
The Khakh lab was supported by NIH MH104069. The Mody lab was supported by NIH NS030549. J.C.O. thanks the NIH T32 Neural Microcircuits Training Grant(NS058280).
References
- McDonough AA, Youn JH. Potassium homeostasis: The knowns, the unknowns, and the health benefits. Physiol Bethesda Md. 2017;32(2):100–111. doi: 10.1152/physiol.00022.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hille B. Ion channels of excitable membranes. Sunderland, MA: Sinauer; 2001. p. 507. [Google Scholar]
- Kofuji P, Ceelen P, Zahs KR, Surbeck LW, Lester HA, Newman EA. Genetic inactivation of an inwardly rectifying potassium channel (Kir4.1 subunit) in mice: Phenotypic impact in retina. J Neurosci. 2000;20(15):5733–5740. doi: 10.1523/JNEUROSCI.20-15-05733.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sibille J, Dao Duc K, Holcman D, Rouach N. The neuroglial potassium cycle during neurotransmission: role of Kir4.1 channels. PLoS Comput Biol. 2015;11(3):e1004137. doi: 10.1371/journal.pcbi.1004137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tong X, et al. Astrocyte Kir4.1 ion channel deficits contribute to neuronal dysfunction in Huntington's disease model mice. Nat Neurosci. 2014;17(5):694–703. doi: 10.1038/nn.3691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Datta D, Sarkar K, Mukherjee S, Meshik X, Stroscio MA, Dutta M. Graphene oxide and DNA aptamer based sub-nanomolar potassium detecting optical nanosensor. Nanotechnology. 2017;28(32):325502. doi: 10.1088/1361-6528/aa79e0. [DOI] [PubMed] [Google Scholar]
- Bandara HMD, et al. Palladium-Mediated Synthesis of a Near-Infrared Fluorescent K+ Sensor. J Org Chem. 2017;82(15):8199–8205. doi: 10.1021/acs.joc.7b00845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Depauw A, et al. A highly selective potassium sensor for the detection of potassium in living tissues. Chem Weinh Bergstr Ger. 2016;22(42):14902–14911. doi: 10.1002/chem.201602209. [DOI] [PubMed] [Google Scholar]
- Machado R, et al. Biofouling-Resistant Impedimetric Sensor for Array High-Resolution Extracellular Potassium Monitoring in the Brain. Biosensors. 2016;6(4) doi: 10.3390/bios6040053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose MC, Henkens RW. Stability of sodium and potassium complexes of valinomycin. Biochim Biophys Acta BBA - Gen Subj. 1974;372(2):426–435. [Google Scholar]
- Ammann D, Chao P, Simon W. Valinomycin-based K+ selective microelectrodes with low electrical membrane resistance. Neurosci Lett. 1987;74(2):221–226. doi: 10.1016/0304-3940(87)90153-4. [DOI] [PubMed] [Google Scholar]
- Amzica F, Steriade M. Neuronal and glial membrane potentials during sleep and paroxysmal oscillations in the neocortex. J Neurosci. 2000;20(17):6648–6665. doi: 10.1523/JNEUROSCI.20-17-06648.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amzica F, Steriade M. The functional significance of K-complexes. Sleep Med Rev. 2002;6(2):139–149. doi: 10.1053/smrv.2001.0181. [DOI] [PubMed] [Google Scholar]
- MacVicar BA, Feighan D, Brown A, Ransom B. Intrinsic optical signals in the rat optic nerve: role for K(+) uptake via NKCC1 and swelling of astrocytes. Glia. 2002;37(2):114–123. doi: 10.1002/glia.10023. [DOI] [PubMed] [Google Scholar]
- Chever O, Djukic B, McCarthy KD, Amzica F. Implication of Kir4.1 channel in excess potassium clearance: an in vivo study on anesthetized glial-conditional Kir4.1 knock-out mice. J Neurosci. 2010;30(47):15769–15777. doi: 10.1523/JNEUROSCI.2078-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall DG. Ion-selective membrane electrodes: A general limiting treatment of interference effects. J Phys Chem. 1996;100(17):7230–7236. [Google Scholar]
- Haack N, Durry S, Kafitz KW, Chesler M, Rose CR. Double-barreled and Concentric Microelectrodes for Measurement of Extracellular Ion Signals in Brain Tissue. J Vis Exp. 2015. p. e53058. [DOI] [PMC free article] [PubMed]
- Larsen BR, MacAulay N. Kir4.1-mediated spatial buffering of K(+): Experimental challenges in determination of its temporal and quantitative contribution to K(+) clearance in the brain. Channels Austin Tex. 2014;8(6):544–550. doi: 10.4161/19336950.2014.970448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mei L, et al. Long-term in vivo recording of circadian rhythms in brains of freely moving mice. Proceedings of the National Academy of Sciences. 2018;115:4276–4281. doi: 10.1073/pnas.1717735115. [DOI] [PMC free article] [PubMed] [Google Scholar]


