Abstract
Since the development of CLARITY, a bioelectrochemical clearing technique that allows for three-dimensional phenotype mapping within transparent tissues, a multitude of novel clearing methodologies including CUBIC (clear, unobstructed brain imaging cocktails and computational analysis), SWITCH (system-wide control of interaction time and kinetics of chemicals), MAP (magnified analysis of the proteome), and PACT (passive clarity technique), have been established to further expand the existing toolkit for the microscopic analysis of biological tissues. The present study aims to improve upon and optimize the original PACT procedure for an array of intact rodent tissues, including the whole central nervous system (CNS), kidneys, spleen, and whole mouse embryos. Termed psPACT (process-separate PACT) and mPACT (modified PACT), these novel techniques provide highly efficacious means of mapping cell circuitry and visualizing subcellular structures in intact normal and pathological tissues. In the following protocol, we provide a detailed, step-by-step outline on how to achieve maximal tissue clearance with minimal invasion of their structural integrity via psPACT and mPACT.
Keywords: Neuroscience, Issue 135, Optical tissue clearing method, psPACT, mPACT, CLARITY, Passive tissue clearing technique, Central nervous system, Transparent CNS, Rodent, Mouse and rat
Introduction
A fundamental objective of scientific and clinical inquiry involves attaining a complete understanding of organ structure and function; however, the exceedingly complex nature of mammalian organs often serves as a barrier to fully achieving this aim1. CLARITY (Clear Lipid-exchanged Acrylamide-hybridized Rigid Imaging-compatible Tisssue-hYdrogel)2,3,4, which involves building an acrylamide-based hydrogel hybrid from intact tissues, achieves optical clearance of a variety of organs, including the brain, liver, and spleen, while preserving their structural integrity5. CLARITY has thus enabled not only visualization but also the opportunity to finely dissect complex cellular networks and tissue morphologies without the need for sectioning.
In order to achieve tissue clearance, CLARITY employs electrophoretic methods to remove the lipid content of the sample at hand. While CLARITY has been noted for producing physically stable tissue-hydrogel hybrids, studies have shown that its use of electrophoretic tissue clearing (ETC) methods yields variable results in terms of tissue quality, including browning, epitope damage, and protein loss5,6. To address these issues, modified protocols such as PACT (PAssive Clarity Technique), which replaces the ETC treatment with a passive, ionic-detergent based delipidation technique, have been developed7,8,9. Despite achieving a greater consistency in results, however, PACT requires more time to obtain maximal clearance. Furthermore, none of these techniques have yet been applied to the whole CNS form, or in larger rodent models such as rats and guinea pigs.
The present study seeks to address these limitations by proposing novel methodologies, psPACT (process-separate PACT) and mPACT (modified PACT), for facilitating the fast clearance of the whole CNS and internal organs in both mouse and rat models10. Specifically, psPACT processes tissues in 4% acrylamide and 0.25% VA-044 in two separate steps during hydrogel formation; mPACT essentially involves the same steps, but supplements the SDS-based clearing solution with 0.5% α-thioglycerol as a key reagent. Both techniques harness the endogenous systemic and cerebrospinal circulatory systems to significantly reduce the time needed to produce optical clearance. As a proof of principle, we demonstrate the use of confocal microscopy to analyze blood vessel patterns in the cleared tissues10.
Protocol
All procedures have been approved by the appropriate research ethics committee at Yonsei University College of Medicine. All experimental animals are sacrificed in accordance with the guidelines of the laboratory animal care committee at Yonsei University College of Medicine.
1. Preparation of Reagents
Caution: Paraformaldehyde (PFA), acrylamide and sodium dodecyl sulfate (SDS) are toxic irritants and thus should be handled in a fume hood with appropriate personal protective equipment (PPE; lab coat, gloves, protective eyewear).
4% acrylamide (A.A) solution (A4P0): Add 20 mL of 40% acrylamide solution to 180 mL of 0.1 M phosphate-buffered saline (PBS).
0.25% VA-044 solution: Add 0.5 g of 2,2´-Azobis[2-(2-imidazolin-2-yl)propane] dihydrochloride (VA-044) powder to 200 mL of 0.1 M phosphate-buffered saline (PBS). While the VA-044 powder can be stored at room temperature, it must be stored at 4 °C upon solubilization in PBS. For best results, prepare only the amount of solution needed for the experiment.
PACT cocktail solution: Add 10 g of acrylamide to 225 mL of 4% paraformaldehyde (PFA) solution and adjust the volume to 200 mL with 4% PFA. Prior to use, add 100 mg of VA-044 powder to 40 mL of mixture in a 50 mL conical tube.
PACT clearing solution buffer: Add 40 g of sodium dodecyl sulfate (SDS) to 350 mL of 0.1 M PBS and adjust the volume to 500 mL with 0.1 M PBS. For mPACT clearing solution, add 0.5% α-thioglycerol to the PACT clearing solution buffer.
The mice used in this study were 2-week-old BALB male mice; the rats used in this study were 2 week old male SD-rats.
2. Anesthesia and Perfusion Surgery
Caution: PFA and acrylamide are toxic irritants and thus should be handled in a fume hood with appropriate PPE.
Anesthetize the animal in a pathogen-free room with 30 mg/kg of zoletil using a 1 mL syringe with a 26-gauge needle. Monitor the animal for 5 -10 min.
Once the animal has reached a surgical plane of anesthesia, use the toe-pinch response method to determine the depth of anesthesia. Confirm unresponsiveness. NOTE: The following steps involving mouse and rat surgery follow a similar protocol used in a previous study11.
Make a 5-6 cm incision beneath the rib cage, through the integument and abdominal wall.
Use curved, blunt scissors to make an incision in the diaphragm, palpating the thoracic area to locate the site of incision beforehand.
Extend the incision across the entire length of the ribcage to expose the pleural cavity.
Carefully displace the lungs. Make a cut through the ribcage up to the collarbone. Using iris scissors, make a small incision to the posterior end of the left ventricle. Insert an 18-gauge blunt- or olive-tipped perfusion needle into the ascending aorta, passing through the cut ventricle.
Secure the needle and prevent leakage by clamping the heart with a hemostat. Alternatively, use a modified hemostat to clamp the aorta around the needle tip.
Make a large incision in the right atrium, taking care to ensure minimal damage to the descending aorta. The rat is now ready for perfusion.
3. Whole Perfusion and Dissection of Rat
Caution: PFA and acrylamide are toxic irritants and thus should be handled in a fume hood with appropriate PPE.
NOTE: The following whole perfusion steps are a similar to a protocol used in a previous study by Woo et al. (2016)10,11.
Attach the right atrium of heart to an 18-gauge needle, taking care to not introduce any air bubbles.
Using a 50 mL syringe, quickly and evenly pump 50 mL of cold 0.1 M PBS solution containing heparin (10 units/mL).
Connect the 18-gauge needle to the tube of the peristaltic pump.
Wash with 200 mL of cold 0.1 M PBS solution containing heparin (10 units/mL) at a circulation velocity of 10 mL/min.
Fix with 250 mL of cold 4% PFA solution at a circulation velocity of 10 mL/min. The rat should be stiff at this stage.
Collect and store the remaining PFA solution for disposal.
For PACT, perfuse tissues with a chilled PACT cocktail solution of 4% PFA, 4% acrylamide, and 0.25% VA-044 powder with an 18-gauge needle, and then remove the head and spine. Expose the skull by making a midline incision from the neck to the nose. This additional perfusion step is unnecessary for the psPACT and mPACT methods; after fixation, immediately isolate the head and spine and expose the skull.
Expose the base of the skull by removing any residual neck and spine muscle.
Use rongeurs and scissors to peel away the skull. Remove the brain and spinal cord.
Store the tissue in 4% PFA at 4 °C; tissues can be stored for up to 1 week.
4. Hydrogel Monomer Infusion and Polymerization of the Rat and Mouse CNS
Caution: Acrylamide, SDS, α-thioglycerol and PFA are irritants and thus should be handled in a fume hood and with appropriate PEE.
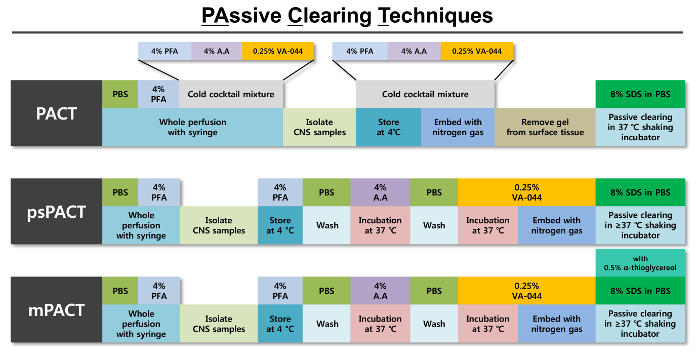
- PACT (Passive Clearing Technique)
- Isolate and culture the whole CNS (brain and spinal cord) from fixed mice and rats to a 50 mL tube containing a chilled PACT cocktail solution of 4% PFA, 4% acrylamide, and 0.25% VA-044 powder and store at 4 °C for 24 h. Ensure that the tissue is completely immersed in the solution.
- Embed the sample in nitrogen gas for 10 min using a tissue gel hybridization system connected to a nitrogen tank: Set the system to 37 °C. Transfer tissues to a 50 mL tube containing fresh cold (4 °C) PACT cocktail solution and connect with tube cap, then turn on the vacuum.
- To polymerize the hydrogel, place the tube containing the sample in a shaking incubator (150 rpm, 37 °C) for 3 h, or until polymerization is complete.
- Using blotting paper (see Table of Materials), remove the remaining polymerized hydrogel surrounding the tissues.
- Transfer the tissue to a 50 mL tube containing clearing solution (8% SDS in 0.1 M PBS, pH 8.0).
- Place the sample in a shaking incubator set to 37 °C and 150 rpm until the tissue has been cleared. On average, it takes about 20 days for the mouse CNS to achieve full clarity.
- psPACT (process separate Passive Clearing Technique)
- Isolate the whole CNS (brain and spinal cord) with bone cutter and scissors from PFA-fixed mice and rats on a clean bench.
- Transfer tissues to a 50 mL tube containing 4% PFA, and store at 4 °C for 24 h. Ensure that the tissue is completely immersed in the fixative.
- Wash the fixed tissue for 1 h in 0.1 M PBS, and then transfer to A4P0 solution (4% acrylamide in 0.1 M PBS). Store at 37 °C for 24 h.
- Wash the tissue for 5 min in 0.1 M PBS.
- Immerse the tissue in 0.25% VA-044 in 0.1 M PBS. Store at 37 °C for 6-24 h, and then transfer to fresh 0.25% VA-044/PBS solution.
- Embed the sample in nitrogen gas for 10 min using a tissue gel hybridization system (see Table of Materials) connected to a nitrogen tank: Transfer tissues to a 50 mL tube containing fresh cold (4 °C) 0.25% VA-044/PBS solution, and connect with tube cap. Turn on the vacuum.
- Transfer the tissue to clearing solution (8% SDS in 0.1 M PBS, pH 8.0).
- Incubate the sample in a shaking incubator set to 37 °C and 150 rpm until the tissue has been cleared. On average, it takes approximately 17 days for mouse CNS to achieve full clarity.
- mPACT (modified Passive Clearing Technique)
- Isolate the whole CNS (brain and spinal cord) with bone cutter and scissors from PFA-fixed mice and rats on a clean bench.
- Follow steps 4.2.2 - 4.2.8 of the psPACT protocol.
- Transfer the tissue to clearing solution. Note that unlike the PACT and psPACT protocols, mPACT requires a clearing solution consisting of 0.5% α-thioglycerol in addition to 8% SDS in 0.1 M PBS, pH 8.0. α-thioglycerol is an un-browning agent that helps clear tissue more rapidly and effectively. Ensure that the tissue is fully immersed in the solution.
- Place the sample in a shaking incubator set to 37 °C and 150 rpm until the tissue has been cleared. On average, it takes approximately 2 weeks for mouse CNS to achieve full clarity.
5. Refractive Index Matching and Immunostaining of Cleared CNS
NOTE: nRIMS (Nycodenz-based Refractive Index Matching Solution) consists of 0.8 g/mL Nycodenz powder dissolved in 30 mL base buffer (0.01% sodium azide and Tween-20 in 0.1 M PBS, pH 8.0). It is recommended that the solution is placed in a 37 °C shaking incubator to allow for proper solvation of the powder.
Incubate tissues in 0.1% Triton X-100 in 0.1 M PBS for 2 h, then block with 2% bovine serum albumin (BSA) in 0.1 M PBS for 6 h.
Wash sections three times in PBST (0.1% Tween-20 in 0.1 M PBS) for 2 h. Stain sections with primary antibodies (in this case, an anti-rabbit PECAM-CD31 antibody, which stains blood vessels) for 2 days.
Stain sections with secondary antibodies (in this case, a goat anti-rabbit IgG Cy3 fluorescent conjugate) in 2% BSA for 2 days.
Wash labeled tissues three times in PBST for 2 h, and store in 15 mL nRIMS for 2 - 10 days.
Before imaging, move labeled tissues to a small amount of nRIMS on 35- or 60-mm tissue culture dishes. Fix with silicone around the bottom edge of the dish.
Add 1.5 - 2 mL of fresh nRIMS.
6. Image processing
Acquire images of cleared tissues with tile scanning using a confocal laser-scanning microscopewith 10x magnification (see Table of Materials). For of Cy3-labeled tissue, use wavelengths of 550-600 nm.
Representative Results
Generation of a transparent model of the whole CNS using optimized passive clearing techniques
Optical clearance of mouse and rat whole CNS tissues was rapidly achieved using various passive clearing techniques (Figure 1). A schematic of tissue clearing over time is shown in Figure 2A. Unlike the original PACT method, psPACT (process-separate PACT) involves treating samples with 4% acrylamide (A4P0) and 0.25% VA-044 initiator solution in two separate steps in order to form the hydrogel hybrid. mPACT (modified PACT) further improves upon psPACT by supplementing the 8% SDS clearing solution with 0.5% α-thioglycerol. Samples processed via PACT, psPACT, and mPACT at Day 14 are compared and presented in Figure 2B.
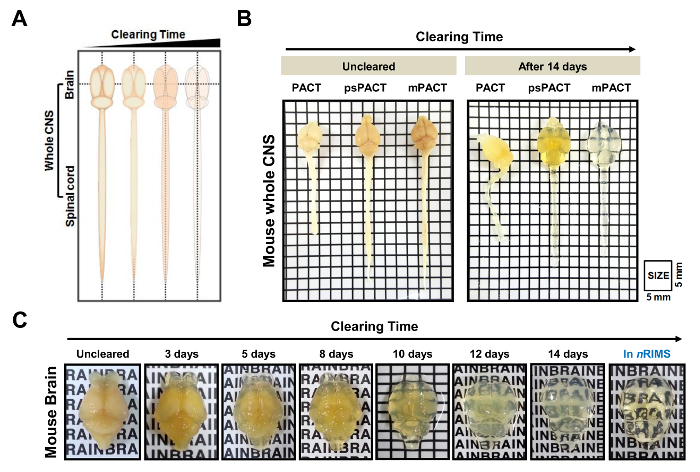
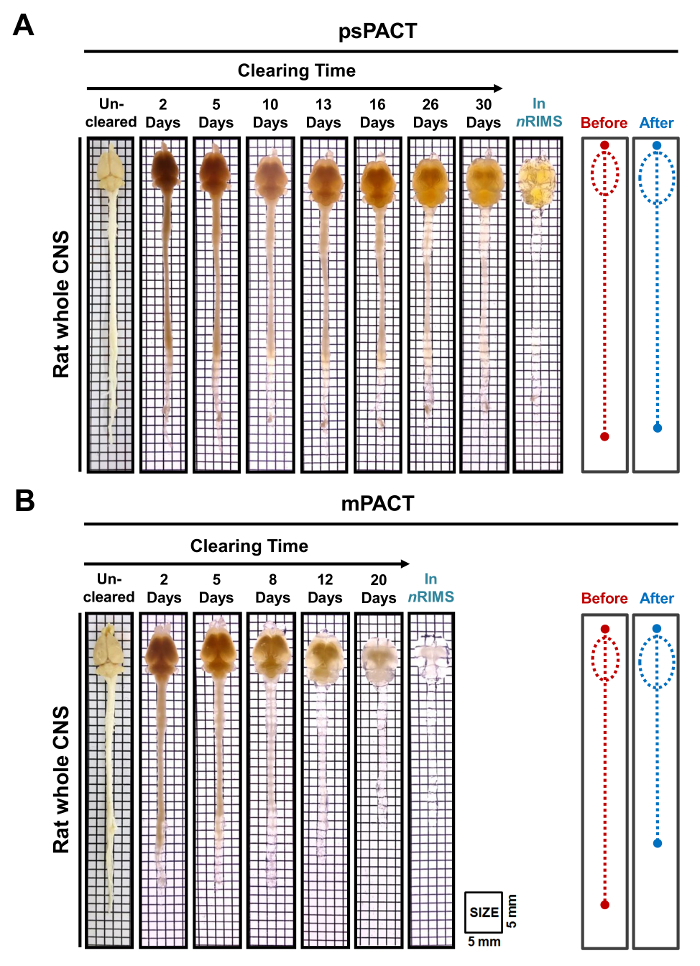
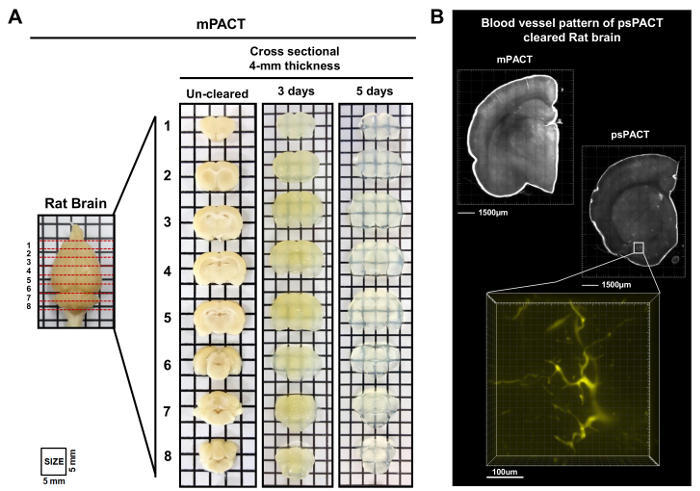
In a previous study, we reported that while the original PACT protocol achieved optical clearance in 23 days, psPACT and mPACT cleared the entire mouse CNS in just 20 and 14 days, respectively10. In the present study, when we treated mouse brain samples via the mPACT protocol, we found that they achieved optical transparency after 14 days (Figure 2B). Rat whole CNS samples processed via psPACT and mPACT also showed that mPACT achieved maximal clearance with the greatest efficiency, as tissues were cleared in 30 and 20 days, respectively (Figure 3A, B). Adult rat brains alone, as opposed to the entire CNS, were cleared with mPACT in a mere 5 days (Figure 4A). Blood vessel patterns of cleared rat brains were analyzed via immunofluorescence after psPACT and mPACT processing to demonstrate the utility of these clearing procedures for the anatomical and functional analysis of the brain (Figure 4B, mPACT data shown in Woo et al. 2016)10.
Using these optimized passive tissue clearing protocols, we were able to visualize intact, transparent models of the whole CNS, although the clearing times differed among the three protocols. Optimized passive clearing methods achieved organ clarity in a shorter length of time. Taken together, these results suggest that the mPACT clearing method can generate clear CNS more stably and rapidly than the previous passive clearing methods. Thus, mPACT holds great potential for use in future structural and anatomical analyses of mammalian organs and provides a significant advantage over existing methods in terms of both efficacy and safety.
Generation of optically cleared internal organs and embryos using mPACT
In addition to whole CNS tissues, the adult rat spleen and kidney were cleared with mPACT in 19 and 23 days, respectively (Figure 5A,B). Mouse embryos at E10.5 and E13.5 were also cleared after 3 days of processing via the mPACT protocol. Figure 5C-E shows the visualization of blood vessel patterns in the whole embryo, as well as the microvasculature of the tail region of the mouse embryo, rat cauda equine and the rat spleen. These results suggest that mPACT can be used to generate transparent models of a wide variety of tissues and organs for 3D anatomical analysis.
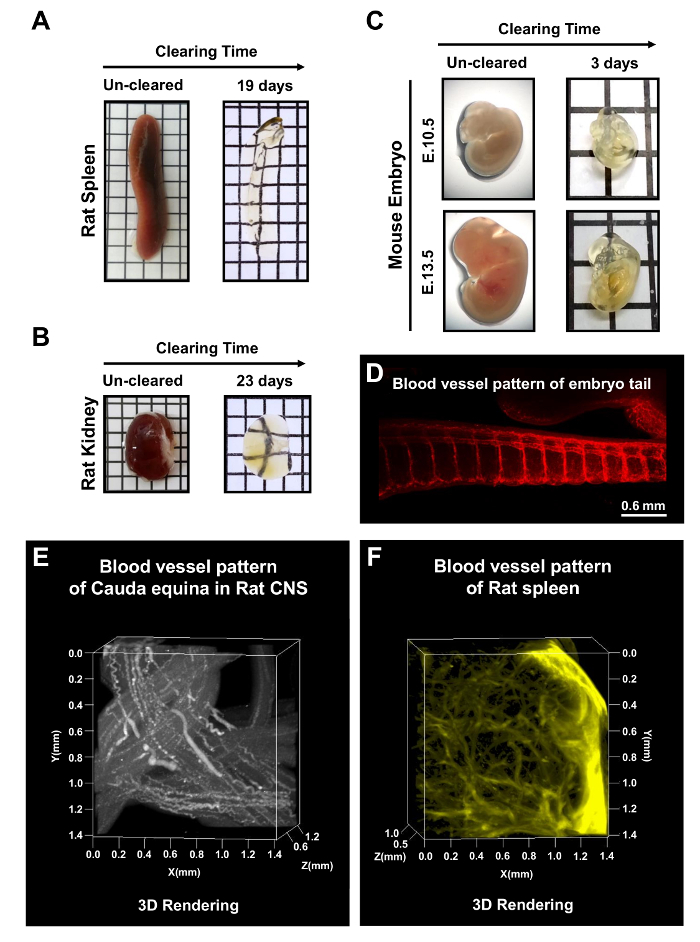
Figure 1: Schematic representation of optimized passive clearing techniques (PACT). An outline of the individual steps involved in each of the three PACT protocols is provided alongside the reagents required for each step. The original PACT protocol is tissue polymerization from hydrogel formation with a cold mixture of 4% PFA, 4% Acrylamide (A.A) solution and 0.25% VA-044 initiator solution. After tissue is polymerized using nitrogen gas, it is passively cleared with 8% SDS clearing solution. The psPACT (process-separate PACT) uses separate treatment processes of 4% A.A solution and 0.25% VA-044 initiator solution for the hydrogel formation of the tissue; these conditions differ from those of the original PACT protocol. The mPACT (modified PACT) additional adds 0.5% α-thioglycerol to the 8% SDS clearing solution of the psPACT protocol. For all three protocols, the intact tissues are incubated at >= 37 °C in a shaking incubator until maximal clearance iss achieved. Please click here to view a larger version of this figure.
Figure 2: Optical transparency of mouse whole central nervous system (CNS) by passive tissue clearing. (A) Schematization of whole CNS tissue clearing via PACT-based methods over time. (B) Comparison of the three passive clearing protocols (original PACT, psPACT, mPACT) in mouse whole CNS at 14 days after processing. (C) Optical clearance of the mouse brain subjected to mPACT processing over 14 days. This figure has been modified from Woo et al. 201610. The letters in 2C are 3 mm tall. Please click here to view a larger version of this figure.
Figure 3: Optical transparency of rat whole CNS tissues post-psPACT and mPACT. (A) Adult rat whole CNS tissue achieved maximal optical clearance with psPACT after 30 days. (B) Adult rat whole CNS tissue achieved maximal optical clearance with mPACT after 20 days. The before and after diagrams indicate the size of the whole CNS before and after treatment, as measured by eye. Please click here to view a larger version of this figure.
Figure 4: mPACT processing and microvasculature visualization of sectioned adult rat brain. (A) Comparison of 4-mm thick adult rat brain sections after mPACT processing at 3 and 5 days. (B) 3D projection of blood vessel patterns in the adult rat brain visualized with anti-CD31 antibody after psPACT processing. Scale bars, 1,500 µm and 100 µm. This figure has been modified from Woo et al. 201610. Please click here to view a larger version of this figure.
Figure 5: mPACT processing of rodent organs and whole mouse embryos. mPACT achieved optical transparency in the adult rat (A) spleen and (B) kidney in 19 and 23 days, respectively. (C) mPACT produced transparent E10.5 and E13.5 mouse embryos after 3 days. (D) Visualization of microvasculature with anti-CD31 in the tail region of the mouse embryo after clearing with mPACT. (E) 3D projection of vasculature in the rat cauda equine and (F) rat spleen with anti-CD31, after processing via mPACT. This figure has been modified from Woo et al. 201610. Please click here to view a larger version of this figure.
Discussion
While the passive, non-electrophoretic extraction methods employed in PACT significantly improved the consistency achieved with previous tissue clearing methods such as CLARITY2,3,4,7,8, the technique still bears several shortcomings, the most pressing of which is the length of time required to achieve maximal tissue clarity12. In the current study, we present modified PACT protocols that significantly reduce the time required for tissue clearing while still preserving tissue integrity and structure6,10. Furthermore, for the first time, we demonstrate the application of tissue clearing protocols in whole CNS tissues and in a higher-order rodent model, the rat10.
Specifically, psPACT involves sample processing in 4% acrylamide and 0.25% VA-044 in two separate steps during hydrogel formation, while mPACT further builds upon psPACT and supplements the 8% SDS clearing solution with 0.5% α-thioglycerol10. Separating the acrylamide and subsequent VA-044 step avoids the need for removing remaining unpolymerized hydrogel monomers that surround the tissue, which can compromise final tissue integrity after achieving transparency with PACT; additionally, this significantly reduces the time needed to produce maximal optical clearance. The addition of α-thioglycerol in the mPACT method further accelerates the clearing process13, as α-thioglycerol is an un-browning agent that helps clear regions of the tissue that are less accessible by using only the reagents in the original PACT method7,8,9.
A comparison of PACT, psPACT and mPACT on rodent tissue demonstrated that while psPACT improves optical transparency relative to the original PACT protocol, mPACT most efficiently allows for the clearing and subsequent high-resolution imaging of both whole mammalian organs and sectioned slices, as demonstrated in Figure 2, Figure 3, Figure 4. The α-thioglycerol-based lipid washout of processed organs in the mPACT method proved key to the differences in transparency, efficiency, and tissue stability in comparison to the other two clearing methods. This allowed for the rapid 3D analysis of processed organs, specifically with respect to blood vasculature morphology, via immunofluorescence and confocal microscopy (Figure 5D,E).
While the addition of α-thioglycerol to the SDS-based clearing solution is critical for accelerating optical transparency, it also slightly increases tissue fragility and makes it more susceptible to damage post-processing. The clear superiority of mPACT with regards to its efficiency outweighs this limitation; nevertheless, work is currently being done to ensure that maximal clearing can be achieved with minimal to no expense of tissue integrity.
Thus, our results establish mPACT as a powerful investigative tool for providing a 3D analysis of the functional and anatomical features of mammalian organs, including but not limited to neuronal networks, blood vasculature, and tissue matrix architecture. Moving forward, we believe mPACT could be especially useful in the investigation of higher-order biological mechanisms that contribute to the pathophysiology of disease, such as tissue injury, developmental malformations, cancer, and neurodegenerative disease14,15,16,17. Combined with a study of the finer cellular mechanisms involved in disease, mPACT has the potential to provide a more complete, multi-scale understanding of how various disparate disease mechanisms interact to give rise to the observed phenotype.
Disclosures
The authors have nothing to disclose.
Acknowledgments
This work was supported by the Brain Korea 21 PLUS Project for Medical Science, Yonsei University. In addition, this work was supported by a grant from the National Research Foundation of Korea (NRF-2017R1D1A1B03030315).
References
- Zhu X, Xia Y, Wang X, Si K, Gong W. Optical brain imaging: A powerful tool for neuroscience. Neurosci Bull. 2017;33(1):95–102. doi: 10.1007/s12264-016-0053-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung K, et al. Structural and molecular interrogation of intact biological systems. Nature. 2013;497(7449):332–337. doi: 10.1038/nature12107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomer R, Ye L, Hsueh B, Deisseroth K. Advanced CLARITY for rapid and high-resolution imaging of intact tissues. Nat Protoc. 2014;9(7):1682–1697. doi: 10.1038/nprot.2014.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng Y, et al. CLARITY reveals dynamics of ovarian follicular architecture and vasculature in three-dimensions. Sci Rep. 2017;7:44810. doi: 10.1038/srep44810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H, Park JH, Seo I, Park SH, Kim S. Improved application of the electrophoretic tissue clearing technology, CLARITY, to intact solid organs including brain, pancreas, liver, kidney, lung, and intestine. BMC Dev Biol. 2014;14:48. doi: 10.1186/s12861-014-0048-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen KHR, Berg RW. Advances and perspectives in tissue clearing using CLARITY. J Chem Neuroanat. 2017;86:19–34. doi: 10.1016/j.jchemneu.2017.07.005. [DOI] [PubMed] [Google Scholar]
- Treweek JB, et al. Whole-body tissue stabilization and selective extractions via tissue-hydrogel hybrids for high-resolution intact circuit mapping and phenotyping. Nat Protoc. 2015;10(11):1860–1896. doi: 10.1038/nprot.2015.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang B, et al. Single-cell phenotyping within transparent intact tissue through whole-body clearing. Cell. 2014;158(4):945–958. doi: 10.1016/j.cell.2014.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neckel PH, Mattheus U, Hirt B, Just L, Mack AF. Large-scale tissue clearing (PACT): Technical evaluation and new perspectives in immunofluorescence, histology, and ultrastructure. Sci Rep. 2016;6:34331. doi: 10.1038/srep34331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo J, Lee M, Seo JM, Park HS, Cho YE. Optimization of the optical transparency of rodent tissues by modified PACT-based passive clearing. Exp Mol Med. 2016;48(12):274. doi: 10.1038/emm.2016.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gage GJ, Kipke DR, Shain W. Whole animal perfusion fixation for rodents. J Vis Exp. 2012. [DOI] [PMC free article] [PubMed]
- Roberts DG, Johnsonbaugh HB, Spence RD, MacKenzie-Graham A. Optical clearing of the mouse central nervous system using passive CLARITY. J Vis Exp. 2016. [DOI] [PMC free article] [PubMed]
- Ke MT, Fujimoto S, Imai T. SeeDB: A simple and morphology-preserving optical clearing agent for neuronal circuit reconstruction. Nat Neurosci. 2013;16(8):1154–1161. doi: 10.1038/nn.3447. [DOI] [PubMed] [Google Scholar]
- Choi BR, et al. Increased expression of the receptor for advanced glycation end products in neurons and astrocytes in a triple transgenic mouse model of Alzheimer's disease. Exp Mol Med. 2014;46:75. doi: 10.1038/emm.2013.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang DJ, et al. Contralaterally transplanted human embryonic stem cell-derived neural precursor cells (ENStem-A) migrate and improve brain functions in stroke-damaged rats. Exp Mol Med. 2013;45:53. doi: 10.1038/emm.2013.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim TK, et al. Analysis of differential plaque depositions in the brains of Tg2576 and Tg-APPswe/PS1dE9 transgenic mouse models of Alzheimer disease. Exp Mol Med. 2012;44(8):492–502. doi: 10.3858/emm.2012.44.8.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinameri E, et al. Prdm proto-oncogene transcription factor family expression and interaction with the Notch-Hes pathway in mouse neurogenesis. PLoS One. 2008;3(12):e3859. doi: 10.1371/journal.pone.0003859. [DOI] [PMC free article] [PubMed] [Google Scholar]


