Abstract
Morphological organ regeneration following acute tissue loss is common among lower vertebrates, but is rarely observed in mammalian postnatal life. Adult liver regeneration after 70% partial hepatectomy results in hepatocyte hypertrophy with some replication in remaining lobes with restoration of metabolic activity, but with permanent loss of the injured lobe's morphology and architecture. Here, we detail a new surgical method in the neonate that leaves a physiologic environment conducive to regeneration. This model involves amputation of the left lobe apex and a subsequent conservative management regimen, and lacks the necessity for ligation of major liver vessels or chemical injury, leaving a physiologic environment where regeneration may occur. We extend this protocol to amputations on juvenile (P7-14) mice, during which the injured liver transitions from organ regeneration to compensatory growth by hypertrophy. The presented, brief 30 min protocol provides a framework to study the mechanisms of regeneration, its age-associated decline in mammals, and the characterization of putative hepatic stem or progenitors.
Keywords: Developmental Biology, Issue 135, Partial Lobular Hepatectomy, Hepatectomy, Liver, Liver Resection, Liver Amputation, Regeneration
Introduction
The ability to regenerate an organ, or to restore form and function, has been thought to be mostly lost over evolutionary time. The regenerative potential of the adult mammalian liver after acute chemical or physical injury has been found to involve the mobilization of all remaining hepatocytes resulting in waves of hypertrophy and few rounds of cell division, resulting in a functional but architecturally different organ1,2,3,4,5. Recently, studies have begun to characterize the regenerative response of neonatal mammalian organs to injury within the first week of life6,7,8. These studies have shown that when injured during neonatal development, certain mammalian organs respond with morphological regeneration instead of compensatory growth or fibrosis7,8.
Recent studies have shown that regeneration of both global structure and function occurs during the early neonatal period6,7,8. Established liver injury protocols involve chemical injury or administration of ethanol9,10,11, acetaminophen12,13,14,15, carbon tetrachloride16,17,18,19, 70% partial hepatectomy4,20,21, or removal of the left and median lobes. Chemical administration leads to hepatocyte cell death, but often leaves micro- and macro-structures intact. Morphologic regeneration cannot be readily studied in this context, as the overall hepatic architecture was not obliterated. The 70% partial hepatectomy involves suture ligation of the major vessels, which is necessary to stop bleeding, but leaves a non-physiologic environment with permanent disruption of vasculature. Furthermore, this method has only been used on adult rodents, and its application to neonates is technically extremely difficult. With this in mind, we developed a method in which 20-30% of the apex of the left lobe is removed in a newborn P0 mouse (Figure 1A-1B). This method is surgically conservative, minimally invasive, not technically challenging, and leads to gross loss of morphology without the ligation of vasculature, leaving room for regeneration to occur. The resulting step-by-step protocol, described below, allows for any researcher to perform a partial lobular hepatectomy on neonatal mice in order to study mammalian neonatal regeneration in the early stages of post-natal life. This method also has clear applications to comparative studies in regenerative medicine and stem cell biology, as it can be used in the liver during later stages of life.
The most common acute liver injury studies are chemically-induced damage, adult liver amputation, or 70% partial hepatectomy. Chemical damage often involves intravenous, intraperitoneal or oral administration of acetaminophen, carbon tetrachloride, or ethanol, and is a relatively easy and non-invasive injury model. As previously discussed, chemical damage results in hepatocyte cell death, but often leaves stroma and parenchyma structures intact, making it difficult to make claims about morphologic regeneration. Chemical damage often centers on hepatic vessels, making it a useful technique to study site and cell-specific injury, but also makes it difficult to interrogate, at the whole organ level, other populations that may be situated further from vessels and that may contribute to regeneration. Despite these limitations, chemical damage still remains a useful and highly physiologically relevant injury model.
Adult 70% partial hepatectomy involves the removal of the left and median lobes following ligation of hepatic vasculature. The response to hepatectomy has been well characterized: the amputated liver 14 days post 70% partial hepatectomy develops a grossly different architecture from that of the original undamaged lobe, as the hepatocytes of the remaining right and caudate lobes undergo hypertrophy and a few rounds of cell division4,5. This makes up lost mass and function, but fails to regenerate the two amputated lobes, and therefore does not replace gross morphology. As a result, the injury response to 70% partial hepatectomy is useful to study compensatory growth mechanisms with limited regeneration.
Here, we fully describe a protocol for a neonatal partial lobular hepatectomy. The procedure involves appropriate animal selection and preparation, surgical field preparation, surgery, and recovery. Optimization and adaptation of each of these steps may be required for different applications of the protocol.
We have extensively performed and optimized this protocol on wild type C57BL/6J pups (JAX 000664), however, to study different cell populations and mechanisms of regeneration, we also used various transgenic animals including mice harboring various Cre and CreERT2 transgenes and/or knock-ins (Axin2CreERT2 JAX 018867, and Sox9CreERT2 JAX 018829) in combination with fluorescent reporters, such as the Rainbow and mTmG systems (R26VT2/GK3, R26mT/mG)22,23. We found no need to change this methodology for different mouse strains, as no differences in survival outcomes or regenerative potential were observed.
In addition to using different animal strains, we also performed partial lobular hepatectomies on neonatal mice treated with small molecules, such as 4-hydroxy-tamoxifen and 5-ethynyl-2′-deoxyuridine (EdU). Dimethyl sulfoxide (DMSO) and ethanol were used as solvents, as it was found that corn oil was a significant cause of morbidity. We otherwise found that intraperitoneal administration of small molecules did not affect survival or regenerative outcomes. We predict that this protocol will be adapted for use with other small molecules to interrogate various aspects of regeneration.
Neonatal mouse surgeries can be technically challenging and may require special expertise in animal handling and microscopic dissection. Animal husbandry expertise is necessary to avoid maternal cannibalism following surgery and during the immediate recovery period.
Protocol
All animal experiments were carried out in strict accordance with the guidelines set forth by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) and Stanford University's Administrative Panel on Laboratory Animal Care (APLAC), (Protocol number #10266) and in the United States, or the European Animal Welfare Act, Directive 2010/63/EU. The protocol was approved by the Committee on the Ethics of Animal Experiments of the Government of Bavaria, Germany, and received the permission No: 55.2-1-54-2532-150-2015.
1. Animal Preparation
Prepare an empty cage with appropriate bedding on a heating pad.
Prior to touching animals with gloves, rub the mother's bedding onto the gloves.
Remove all pups from their mother and place into the empty cage. Remove some of the mother's bedding and place it in the empty cage with the pups.
Place the mother in a separate, clean, dry cage away from the surgical field.
Don new, clean sterile gloves prior to surgical field preparation.
2. Surgical Field Preparation
Using a 10-mL pipette, add 10 mL of phosphate buffered saline (PBS) to a 10-cm Petri dish.
Pipette 2 mL of betadine or equivalent anti-septic solution in the 10-cm Petri dish.
Place a dissecting scope on the surgical field. Turn on dissecting scope lamp and adjust the level of light to the surgeon's comfort. A pup can be placed in the surgical field underneath the dissecting scope lamp. Adequate light can be confirmed by the absence of shadows on the ventral surface of the pup.
Prepare the isoflurane anesthesia chamber (see Table of Materials) with a nose cone. The chamber should be cleaned without evidence of urine or feces. Place the nose cone and associated tubing under the dissecting scope in the surgical field. Divert the flow of oxygen and isoflurane solely to the nose cone.
Prepare a post-operative recovery area with a heating pad set at approximately 37 °C. The post-operative recovery should consist of 4 pieces of gauze placed on top of the heating pad. If possible, rub gauze pads in the mother's bedding, feces, and urine to preserve the mother's scent. NOTE: It is ill-advised to use a heat lamp, as this makes it difficult to control the temperature. Elevated temperatures will result in the death of neonatal mice.
Prepare and sterilize all surgical instruments with an autoclave. The tools needed include: micro-dissecting scissors, micro-dissecting forceps, gauze, hemostat, and any 6-0 monofilament non-absorbable suture. Surgical instruments can be resterilzed with any glass bead sterilizer.
3. Partial Lobular Hepatectomy
- Anesthetize the pup by placing it in the nose cone on its back and gently taping its feet and hands in place. The pup should be receiving 5% isoflurane in oxygen.
- Allow the pup to sit for 5 min or until adequately anesthetized, which can be verified by a toe pinch test.
- Inject 5 mg/kg of carprofen subcutaneously prior to incision. NOTE: The entire surgery should take no longer than 30 min. Poorer outcomes may be observed in neonates that are under general anesthesia for over 30 min. Take precautions to minimize the length of surgery through thorough field preparation and wetting of the skin prior to closure to minimize suture induced skin tears.
Gently clean off the posterior abdominal wall with a small gauze pad wet with betadine. Allow the betadine to dry for 1 min.
Make a left mid-clavicular 0.5 cm incision immediately below the rib cage with the micro-dissecting scissors and forceps. Gently separate the skin using forceps and make a second deeper incision into the peritoneal cavity (See Figure 1C, left and center)
Gently apply lateral pressure from both sides of the abdomen using the back, blunt ends of the micro-dissecting scissors and forceps to force the apex of the left lobe out of the peritoneal cavity. The left apex of the left lobe should be easily visualized (Figure 1C, right).
- From the apex, amputate and weigh the amount of tissue to be removed.
- Using the micro-dissection scissors, gently amputate the desired amount of tissue from the apex of the left lobe.
- Place the amputated tissue into a 1.5-mL tube filled with PBS. Weigh the amputated tissue using an analytical balance (see Table of Materials). NOTE: Place a piece of gauze or paper on the balance and tare it. Then place the amputated tissue on the gauze or paper and measure its mass.
- Fix the amputated area in 2% paraformaldehyde at room temperature for 1 h and place in optimal cutting temperature (OCT) compound over dry ice for frozen section analysis. Analyze frozen sections by cutting 7-10 µm sections using any standard cryostat.
Using a rolled piece of gauze, gently replace the left lobe into the peritoneal cavity. Leave the gauze in the cavity until the bleeding stops.
Remove the gauze and wet the surgical site and the surrounding area with gauze soaked with PBS.
Close the surgical site with 6-0 monofilament, non-absorbable sutures with a running stitch. The peritoneum and skin should be closed separately. NOTE: Gently wetting the skin with gauze soaked with PBS may minimize suture induced tears.
Gently but thoroughly clean the pup with gauze soaked with PBS and ensure no blood or betadine remains. Roll the soaked gauze pad and gently scrub over the wound sign to clean off any blood or betadine. NOTE: This is especially important as the mother may cannibalize the pups if they are cleaned inadequately.
Remove the pup from the nose cone and place it on the recovery area that includes the gauze pads exposed to the mother's feces and urine. Once the pup recovers, replace the pup in the empty cage with the mother's bedding. NOTE: Do not use a heat lamp. The use of a heat lamp can cause the neonate to overheat.
Repeat the procedure on the desired number of pups. Although all pups can be used, it is generally advisable to leave a few pups not operated on to be replaced together with the operated pups.
Replace all pups simultaneously with their mother's bedding in the mother's cage.
4. Recovery and Analysis
- Follow up on the mice daily.
- Check that the wound remains closed and remove any dead pups, if present.
- If the wound reopens, prepare the surgical site and recovery area as previously described, and repeat steps 3.8 to 3.12.
- Inject 5 mg/kg of carpofen subcutaneously 24 and 48 h following the procedure.
- Follow Pups for 56 days or more.
- Euthanize the animals after the desired amount of post-operative days by carbon dioxide (CO2) exposure and cervical dislocation.
- Place mice in the induction / euthanasia chamber and turn on the CO2 until mice stop breathing.
- Ensure euthanasia by cervical dislocation. Push down on the dorsal neck of the mouse using the fore-finger and thumb and using the other hand, pull down on tail.
- Remove the entire liver en bloc and weigh it.
- Carefully separate each lobe and weigh separately. NOTE: Remove the liver by careful dissection of the diaphragm and hepatic and portal vessels. Separate the lobes from each other by carefully dissecting each lobe at its proximal attachment.
Determine the extent of regeneration by comparing the mass of the amputated left lobe to the mass of the whole liver. An uninjured left lobe is approximately 30% of the whole liver.
Representative Results
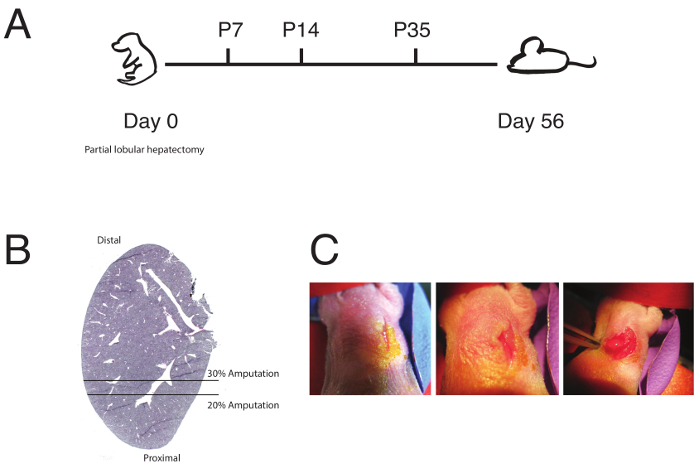
Figure 1A details a general timeline of the neonatal partial lobular hepatectomy (schematic in Figure 1B), and the expected length of time to wait until regeneration is observed. Subtle regeneration of the left lobe can be observed 7-14 days post surgery. Full regeneration was often observed after 56 days post surgery. Mice should show no signs of physiologic abnormalities after surgery.
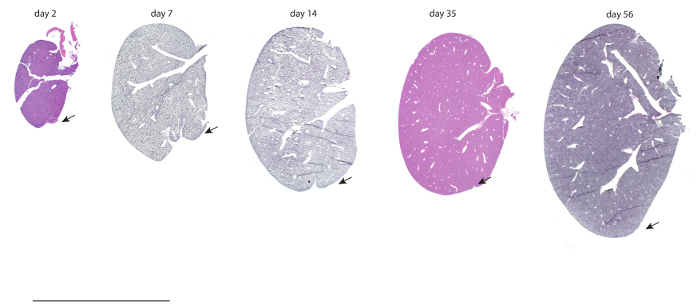
Mice undergoing partial lobular hepatectomies were allowed to recover for 2, 7, 14, 35, and 56 days. Hematoxylin and eosin (H&E) of injured left lobes from these mice after recovery are shown in Figure 2. Notably, after 56 days, the amputated left lobe may look indistinguishable from control, uninjured lobes. Surgeries done on P14 juvenile mice were done for comparison and allowed to recover for 7, 14, and 56 days post surgery (Figure S1).
To characterize neonatal regeneration, 45 mice underwent partial lobular hepatectomy at day 0 and the masses of all their lobes were taken 56 days post surgery. The mass of the injured left lobe underwent an increased change in mass when compared to the other uninjured median, right, and caudate lobes (Figure 3A) and uninjured controls, nearing the mass of an uninjured left lobe at P56. This indicates that regeneration following neonatal liver injury is localized to the left lobe. Surgeries done on P14 juvenile mice were done for comparison, which showed decreased regeneration in the left lobe and increased compensation from the uninjured lobes (Figure 3B), indicating that by 14 days, the injury response to acute resection switched from lobe specific regeneration to global compensation. Further characterization was done by staining areas of the left lobe from injured mice at post-operative day 56 with filamentous actin (f-actin) to visualize cell membranes (Figure 4A). Areas distal and proximal to the area of injury were compared to uninjured controls and adult lobes 14 days following 70% partial hepatectomies. Hepatocytes were found to have similar areas as uninjured controls, about 1.5-2x less than adult mice undergoing regeneration following classical 70% partial hepatectomy (Figure 4B). This suggests that hypertrophy does not play a role in regeneration. Finally, neonatal mice were injected with 0.025 mg of 5-ethynyl-2'-deoxyuridine (EdU) in 90% PBS and 10% ethanol and 1, 3, 5, 7, and 14 days following surgery. The number of EdU positive cells were counted from mice allowed to recover for 7 days following surgery (Figure 4C). A significant increase in the number of EdU positive cells were found in the injured/regenerating left lobe when compared to uninjured controls, indicating that cell proliferation contributes to neonatal regeneration.
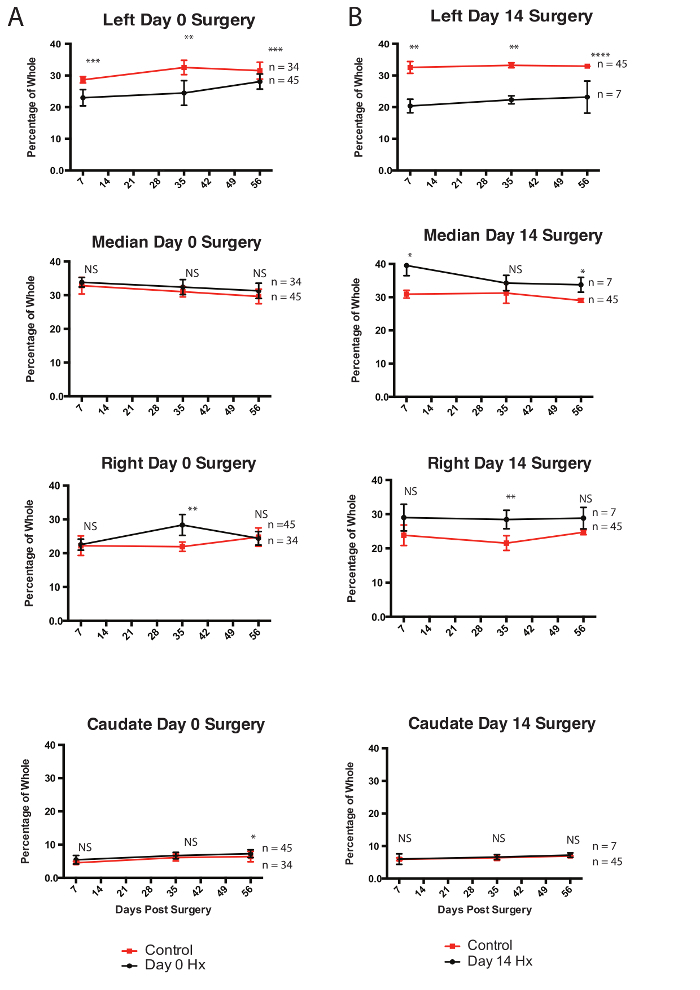
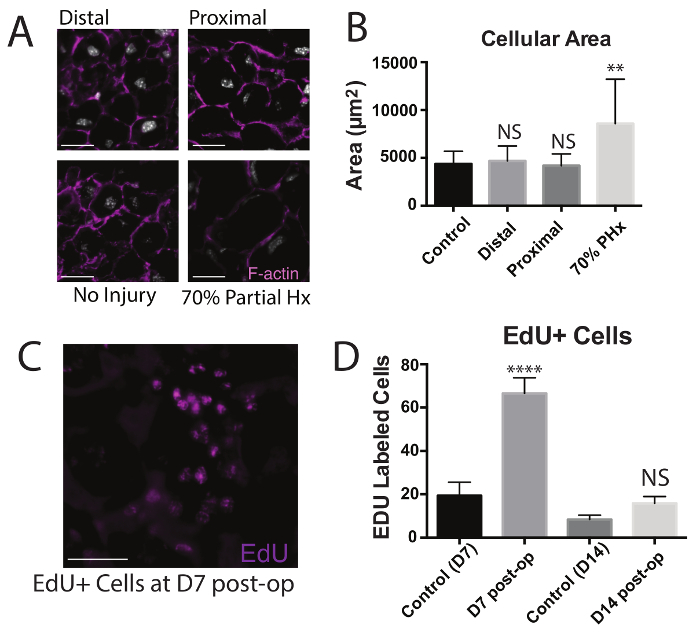
Figure 1: Partial Lobular Hepatectomy Overview. (A)A general schematic and timeline of the partial lobular hepatectomy is shown with neonatal liver resection done at P0. Analyses were done at P7, P14, P35, or P56. Resections were also tried at P7 and P14. (B) A schematic of the extent of resection of the left lobe is shown, demarcating 20 and 30% resections. (This figure has been modified from Tsai et al.)6. (C) Images from neonatal surgeries showing: right sided mid-clavicular incision (left, center) and exposure of the left lobe apex (right). Please click here to view a larger version of this figure.
Figure 2: Regeneration Following Partial Lobular Hepatectomy. Mice undergoing partial lobular hepatectomies at P0 were followed for 2, 7, 14, 35, and 56 days. Livers were fixed and stained with H&E, and the extent of regeneration of the left apex was noted. Arrows denote areas where regeneration occurred in P0 mice. Scale bar is 1 cm. This figure was modified from Tsai et al.6 Please click here to view a larger version of this figure.
Figure 3: Regeneration per Lobe Following Partial Lobular Hepatectomy. (A) Mice undergoing partial lobular hepatectomies at P0 were analyzed at 7, 35, and 56 days post-operatively. Mice were euthanized and masses of all lobes from injured mice (red) were taken and compared to age matched masses of uninjured control (red). (B)Mice undergoing partial lobular hepatectomies at P14 were analyzed at 7, 35, and 56 days post-operatively. Masses of all lobes from injured mice (red) were taken and compared to masses of uninjured control (red). * = p < 0.05, ** = p < 0.005, *** = p < 0.0005, NS = not significant. This figured has been modified from Tsai et al.6 Please click here to view a larger version of this figure.
Figure 4: Characterization of Regeneration Post Hepatectomy. (A) Mice undergoing partial lobular hepatectomies at P0 were analyzed 56 days after resection and stained for F-actin. Images are shown of stains from areas proximal and distal to the area of amputation, as well as from age-matched uninjured controls, and from adult mice 14 days after 70% partial hepatectomy. Scale bars are 100 µm. (B) Areas of hepatocytes following injury at areas proximal or distal to the resection site were compared to areas of hepatocytes from uninjured controls and adult 70% partial hepatectomies.* = p < 0.05, ** = p < 0.005, *** = p < 0.0005, **** = p < 0.00005, NS = not significant. (C) Mice undergoing lobular hepatectomies at P0 were treated with EdU and were analyzed 7 days following resection. EdU+ cells in the left lobe are shown. (Scale bar, 100 µm). (D) Quantification of EdU+ cells in mice treated with EdU 7 and 14 days following partial lobular hepatectomy compared to controls. Values are means ± SEM. This figured been has modified from Tsai et al.6 Please click here to view a larger version of this figure.
Figure S1: Incomplete Regeneration of Juvenile Mice. Mice undergoing partial lobular hepatectomies at P14 were followed for 7, 35, and 56 days. Livers were fixed and stained with H&E and the extent of regeneration of the left apex was noted. Arrows denote areas where regeneration occurred in P0 mice. Scale bar is 1 cm. This figured was modified from Tsai et al.6 Please click here to download this file.
Discussion
Acute hepatic injury has traditionally been studied using chemical (acetaminophen, ethanol, carbon tetrachloride), or surgical models (70% partial hepatectomy). The regenerative response after 70% partial hepatectomy has been characterized to involve global hepatocyte hypertrophy and multiple rounds of cell division4,5. To stop hemorrhaging, however, this model is limited, as the major vessels must be ligated leaving an abnormal environment for regeneration. Many studies have therefore employed other less invasive models of acute injury through chemical damage, leaving the gross architecture in place for regeneration to occur. Recently, Porrello et al. and Chang et al. have demonstrated a markedly different neonatal regenerative response after acute injury in the heart, digit tips, and ears7,8. Their results parallel presented conclusions that the liver also undergoes a distinct regenerative phenomenon in neonatal life6. With multiple similar findings in major organs, regeneration in the early stages of post-natal development is an emerging field with potential implications for stem cell biology.
Early mortality from neonatal partial lobular hepatectomies often comes from inadequate recovery, major hemorrhage, or maternal neglect. As stated previously, the use of a higher intensity heat source such as a heat lamp for recovery, may lead to death following surgery. Neonatal mice are dependent on their mother for at least the first two weeks of life. At the same time, the mother will often neglect and or cannibalize her young if she senses an abnormality (such as the scent of blood or other chemicals)24,25. It is therefore highly important that the neonate is cleaned thoroughly post-operatively and rubbed with maternal bedding to mask any offensive scents. If these issues are adequately addressed, survival can reach up to 100%. If maternal cannibalism becomes an issue, the pups can be placed into a cage with a surrogate mother with some of her own pups. If this is the case, use the surrogate mother's bedding in the previous steps.
The resection of 20-30% of the neonatal left lobe and subsequent regeneration is likely not inherent to only the left lobe. Currently, this method has only been tested on the left, as exposing the median and more posterior right and caudate lobes would necessitate a larger laparotomy, resulting in a higher risk of hemorrhage and, indirectly, a higher risk of maternal cannibalism for the neonate. However, whether the mechanisms of neonatal regeneration are heterogeneous within the liver is an important question to be addressed, and therefore surgical adjustments to this protocol should be made to interrogate the other hepatic lobes.
The results from these neonatal hepatectomy studies have shown a time period (P0-P7) during which regeneration is able to occur. Similar hepatic resections have been done on juvenile mice (P7, P10, P14) and do not result in full regeneration with demonstrated scar and fibrosis, marking a clear area where the amputation occurred. Although the injury response in juvenile mice following partial lobular hepatectomy was not the focus of an initial study, the discrepancy in regenerative potential between neonatal and juvenile mice, and the loss of the ability to reconstitute organ and tissue architecture, will be essential to understanding by what mechanism stem or progenitor cell neonatal regeneration occurs.
We have previously demonstrated that neonatal regenerated livers not only appear the same in architecture and structure, but are also indistinguishable by function. Immunofluorescence stains for functional hepatic enzymes such as glutamine synthetase (GS), carbamoylphosphate synthetase (CPS), and cytochrome p450 2E1 show a similar distribution within regenerated areas when compared to uninjured lobes. However, the secondary regenerative potential of a regenerated neonate has not been tested. As neonatal mice allowed to recovery for 56 days are physiologically indistinguishable from uninjured controls, it is likely that the classical regenerative response following adult 70% partial hepatectomy would occur. However, this liver regeneration is often limited by hepatocyte exhaustion, and therefore serial hepatectomies following partial lobular hepatectomies would be an important study.
Disclosures
The authors have no disclosures.
Acknowledgments
We thank P. Chu for performing H&E and histology; and C. Wang and A. McCarty for helpful discussions. Research was supported through funding from the Virginia and D. K. Ludwig Fund for Cancer Research; the National Heart, Lung, and Blood Institute (R01HL058770 and U01HL099999); and the California Institute for Regenerative Medicine (RC1 00354) grants to I.W. Y.R. was supported by the Human Frontier Science Program Career Development Award (CDA00017), the German Research Foundation (RI 2787/1), the Siebel Stem Cell Institute, and the Thomas and Stacey Siebel Foundation (1119368-104-GHBJI). J.M.T. was supported by the NIH (T32GM007365), the National Research Service Award (1F30DK108561), and the Paul and Daisy Soros Fellowship for New Americans.
References
- Michalopoulos GK, DeFrances MC. Liver Regeneration. Science. 1997;276(80):60–66. doi: 10.1126/science.276.5309.60. [DOI] [PubMed] [Google Scholar]
- Ponfick VA. Surgery of the Liver. Lancet. 1890;1:881. [Google Scholar]
- Higgins G, Anderson RM. Experimental Pathology of the liver. Restoration of the liver of the white rat following partial surgical removal. Arch. Pathol. 1931;12:186–202. [Google Scholar]
- Miyaoka Y, et al. Hypertrophy and unconventional cell division of hepatocytes underlie liver regeneration. Curr. Biol. 2012;22:1166–1175. doi: 10.1016/j.cub.2012.05.016. [DOI] [PubMed] [Google Scholar]
- Miyaoka Y, Miyajima A. To divide or not to divide: revisiting liver regeneration. Cell Div. 2013;8:8. doi: 10.1186/1747-1028-8-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai JM, et al. Localized hepatic lobular regeneration by central-vein-associated lineage-restricted progenitors. Proc. Natl. Acad. Sci. U. S. A. 2017;114:3654–3659. doi: 10.1073/pnas.1621361114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porrello ER, et al. Transient regenerative potential of the neonatal mouse heart. Science. 2011;331:1078–1080. doi: 10.1126/science.1200708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shyh-Chang N, et al. Lin28 enhances tissue repair by reprogramming cellular metabolism. Cell. 2013;155:778–792. doi: 10.1016/j.cell.2013.09.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin M, et al. Essential role of tumor necrosis factor alpha in alcohol-induced liver injury in mice. Gastroenterology. 1999;117:942–952. doi: 10.1016/s0016-5085(99)70354-9. [DOI] [PubMed] [Google Scholar]
- Gao B, Bataller R. Alcoholic liver disease: pathogenesis and new therapeutic targets. Gastroenterology. 2011;141:1572–1585. doi: 10.1053/j.gastro.2011.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uesugi T, Froh M, Arteel GE, Bradford BU, Thurman RG. Toll-like receptor 4 is involved in the mechanism of early alcohol-induced liver injury in mice. Hepatology. 2001;34:101–108. doi: 10.1053/jhep.2001.25350. [DOI] [PubMed] [Google Scholar]
- Coen M, et al. An integrated metabonomic investigation of acetaminophen toxicity in the mouse using NMR spectroscopy. Chem. Res. Toxicol. 2003;16:295–303. doi: 10.1021/tx0256127. [DOI] [PubMed] [Google Scholar]
- Oz HS, et al. Diverse antioxidants protect against acetaminophen hepatotoxicity. J. Biochem. Mol. Toxicol. 2004;18:361–368. doi: 10.1002/jbt.20042. [DOI] [PubMed] [Google Scholar]
- Ruepp SU, Tonge RP, Shaw J, Wallis N, Pognan F. Genomics and proteomics analysis of acetaminophen toxicity in mouse liver. Toxicol. Sci. 2002;65:135–150. doi: 10.1093/toxsci/65.1.135. [DOI] [PubMed] [Google Scholar]
- Gunawan BK, et al. c-Jun N-terminal kinase plays a major role in murine acetaminophen hepatotoxicity. Gastroenterology. 2006;131:165–178. doi: 10.1053/j.gastro.2006.03.045. [DOI] [PubMed] [Google Scholar]
- Manibusan MK, Odin M, Eastmond Da. Postulated carbon tetrachloride mode of action: a review. J. Environ. Sci. Health. C. Environ. Carcinog. Ecotoxicol. Rev. 2007;25:185–209. doi: 10.1080/10590500701569398. [DOI] [PubMed] [Google Scholar]
- Recknagel RO, Glende Ea, Dolak Ja, Waller RL. Mechanisms of carbon tetrachloride toxicity. Pharmacol. Ther. 1989;43:139–154. doi: 10.1016/0163-7258(89)90050-8. [DOI] [PubMed] [Google Scholar]
- Sell S. Heterogeneity and plasticity of hepatocyte lineage cells. Hepatology. 2001;33:738–750. doi: 10.1053/jhep.2001.21900. [DOI] [PubMed] [Google Scholar]
- Malato Y, et al. Fate tracing of mature hepatocytes in mouse liver homeostasis and regeneration. J. Clin. Invest. 2011;121:4850–4860. doi: 10.1172/JCI59261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greene AK, Puder M. Partial hepatectomy in the mouse: technique and perioperative management. J. Invest. Surg. 2003;16:99–102. [PubMed] [Google Scholar]
- Kan NG, Junghans D, Belmonte JCI. Compensatory growth mechanisms regulated by BMP and FGF signaling mediate liver regeneration in zebrafish after partial hepatectomy. FASEB J. 2009;23:3516–3525. doi: 10.1096/fj.09-131730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Red-Horse K, Ueno H, Weissman IL, Krasnow MA. Coronary arteries form by developmental reprogramming of venous cells. Nature. 2010;464:549–553. doi: 10.1038/nature08873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muzumdar MD, Tasic B, Miyamichi K, Li L, Luo L. A Global Double-Fluorescent Cre Reporter Mouse. Genesis. 2007;605:593–605. doi: 10.1002/dvg.20335. [DOI] [PubMed] [Google Scholar]
- Poley W. Emotionality related to maternal cannibalism in BALB and C57BL mice. Anim. Learn. Behav. 1974;2:241–244. [Google Scholar]
- Smotherman WP, Bell RW, Starzec J, Elias J, Zachman TA. Maternal responses to infant vocalizations and olfactory cues in rats and mice. Behav. Biol. 1974;12:55–66. doi: 10.1016/s0091-6773(74)91026-8. [DOI] [PubMed] [Google Scholar]


