Abstract
Exposure to certain environmental chemicals in human and animals has been found to cause cellular damage of the pancreatic β cells which will lead to the development of type 2 diabetes mellitus (T2DM). Although the mechanisms for the chemical-induced β cell damage were unclear and likely to be complex, one recurring finding is that these chemicals induce oxidative stress leading to the generation of excessive reactive oxygen species (ROS) which induce damage to the β cell. To identify potential diabetogenic environmental chemicals, we isolated pancreatic islet cells from C57BL/6 mice and cultured islet cells in 96-well cell culture plates; then, the islet cells were dosed with chemicals and the ROS generation was detected by 2',7'-dichlorofluorescein (DCFH-DA) fluorescent dye. Using this method, we found that bisphenol A (BPA), Benzo[a]pyrene (BaP), and polychlorinated biphenyls (PCBs), could induce high levels of ROS, suggesting that they may potentially induce damage in islet cells. This method should be useful for screening diabetogenic xenobiotics. In addition, the cultured islet cells may also be adapted for in vitro analysis of chemical-induced toxicity in pancreatic cells.
Keywords: Immunology and Infection, Issue 136, Pancreatic islets, T2DM, reactive oxygen species, diabetogenic chemicals, ROS, xenobiotics
Introduction
Increases in the prevalence of T2DM have become a global health crisis in recent years posing a serious threat to public health1. Many factors have been found to be causally linked to the development of T2DM, among which, recurring findings suggest that one common convergent point for these factors is the induction of oxidative stress which leads to the generation of excessive ROS2,3.
A wide spectrum of environmental chemicals including PCBs, dioxins, and BaP has been found to induce oxidative stress, which may impair the function of pancreatic β cells and lead to insulin resistance and T2DM4. Although the physiological level of ROS plays an important role in cellular functions, exposure to ROS that exceeds the capacity of the antioxidant system results in the damage to cells/tissues and leads to diseases5. Pancreatic β cells express a low level of antioxidant enzyme, and thus are a sensitive target for the oxidative stress-mediated damage6,7. Chronic exposure to high levels of ROS has been shown to cause stress-induced pancreatic cell dysfunction5 as well as insulin resistance in the liver and adipose tissue8.
The overall goal of this project is to develop a cell-based assay to screen chemicals for their diabetogenic potentials based on their induction of ROS in pancreatic cells. The pancreas lacks metabolic detoxification and is a sensitive target for xenobiotic-induced damage6,7. Therefore, by directly measuring the ROS generated in the pancreatic cells, this assay should provide a direct approximation of the chemical-induced injury in the pancreas. To develop this method, we isolated mouse pancreatic islets, cultured the isolated islet under cell culture condition with chemicals, and utilized the chemical-induced ROS generation as the readout. This procedure is simple and effective in identifying ROS-inducing chemicals in the isolated islet; it can be further developed for investigation of the mechanisms of toxicity that are specific to the pancreas in vitro.
Protocol
All animal experiments were executed in compliance with all relevant guidelines, regulations and regulatory agencies. The protocol being demonstrated was performed under the guidance and approval of the Institutional Animal Care and Use Committee (IACUC) of the Texas A&M Institute for Genomic Medicine.
1. Solution Preparation
Dilute 10x Hank's balanced salt solution to 1x with double distilled H2O, and store at 4 °C.
Prepare the isolation solution by adding HEPES (10 mM), MgCl2 (1 mM), and glucose (5 mM). Adjust the pH to 7.4 and keep on ice.
Prepare the collagenase solution from collagenase Type 4. Note: Specifically, collagenase Type 4 contains 160 U per mg of dry weight. 1 g of collagenase Type 4 is diluted in 5 mL double distilled H2O with a concentration of 200 mg/mL. 2 mL of the diluted Collagenase is added to 30 mL ice cold isolation solution, and 6 mL of collagenase solution is transferred to a 50 mL tube for each mouse.
Prepare the wash solution by adding 1 mM CaCl2 to the isolation solution.
Prepare the purification solution with cold polysucrose/sodium diatrizoate solution (1.1119 g/mL, 5 mL).
Prepare 500 mL of islet culture medium by adding L-glutamine (20 mM), penicillin (100 U/mL), streptomycin (100 µg/mL), and fetal bovine serum (FBS) (10%) into RPMI medium.
Prepare the DCFH-DA stock solution by dissolving the powder in distilled water at 200 µM and store at -20 °C. Note: The ROS staining solution is freshly made by diluting DCFH-DA stock solution to the final concentration of 5 µM in RPMI. This reagent should be protected from light.
2. Surgical Preparation
Intraperitoneally inject (I.P.) a mouse with avertin (2.5% avertin, 0.4 mL/20 g). After the mouse is completely anesthetized and no longer responds to hind foot pinches, euthanize the mouse and move to the biological hood.
Place the euthanized mouse with the abdominal side facing up and spray the skin with 70% ethanol for sterilization.
Open the abdominal cavity with a 1 cm U-incision upon the upper abdomen and retract the skin in the rostral direction over the chest to expose the abdominal cavity.
3. Pancreas Perfusion and Removal
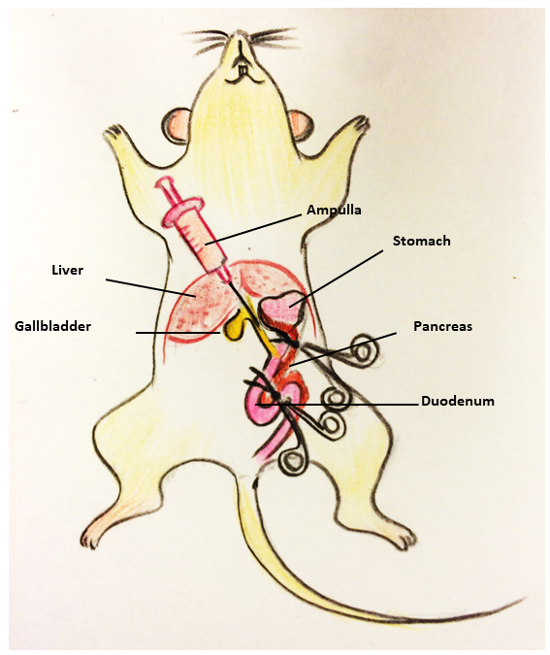
As illustrated in Figure 1, find the location of the ampulla. Use a pair of curved forceps to clamp the duodenum close to the position of sphincter of Oddi. Use another pair of curved forceps to clamp the duodenum wall to block the bile pathway in the duodenum. Note: The ampulla has not been inserted in this step. This step is meant to block the bile pathway in the duodenum.
Reposition the mouse with the head proximal and the feet distal with regards to the researcher.
Find the common bile duct and slowly inject 3 mL of collagenase solution with a 30 G needle and 5 mL syringe. Make sure the common bile duct and the pancreas are inflated after the injection.
Remove the distended pancreas using the curved forceps and fine scissors to separate it from the descending colon, intestines, stomach, and spleen. Place the pancreas in a 50-mL tube containing 3 mL of collagenase solution and keep it on ice.
4. Pancreas Digestion
Place each 50-mL tube into a 37 °C water-bath. Incubate for 15 - 20 min (time varies with the strain and age of the mice).
Vibrate the tube using a vortex.
Pool 4 - 5 digested pancreas in a large sterile sink strainer and use a scoop to make the pancreas fall apart thoroughly. Use a 23 G needle and 10 mL syringe containing wash solution to forcefully pipette the digested cells off the screen. Collect the wash solution in a 50-mL tube.
Centrifuge the tubes at 97 x g at 4 °C for 1 min and decant the supernatant.
Add 25 mL of wash solution to tubes and re-suspend the tissue by gentle vortexing. Spin down the tissues at 97 x g at 4 °C for 1 min and decant the supernatant. Repeat for 3 times. Be careful not to dislodge the pelleted tissue.
5. Purification of Islets
Add 10 mL of purification solution to the washed tissue pellet and gently resuspend the contents.
Gently overlay 5 mL of isolation solution on the purification solution. Be careful to keep a sharp liquid interface between the two solutions.
Centrifuge the tube at 560 x g at 4 °C for 15 min with slow acceleration and no brake. Insert the pipette into the interface and collect the islets layer into new 50 mL tubes.
Wash the islets as described in step 3.5.
6. Plating and Treatment of Islet Cells
Resuspend the islets in 10 mL of islets culture solution, gently mix well, and place 100 µL/well with a multichannel pipette to a 96-well plate. Place approximately 5 islets per 96 well.
Place the plate in the tissue culture incubator for 12 h.
Centrifuge (112 x g) the 96-well plate and remove the culture medium.
Dilute the environmental chemicals in the islets culture medium. Add 100 µL of each of the chemical diluents into separate wells of the plate. Resuspend the islets in each well and incubate for 12 h at 37 °C (the concentration varies for the different environmental chemicals).
7. ROS Staining and Measurement
After islets treatment, wash with 1x PBS (200 µL for each 96-well). Note: The concentration of the chemicals is as follow: AFB1: 3 µM, Atrazine: 100 µM, BaP: 10 µM, BPA: 100 µM, β-estrogen, PCB126: 10 µM, Nonylphenol, and BPA: 100 µM.
Use N-Acetyl Cysteine (NAC) as an antioxidant to quench ROS. Add 5 mM of NAC to the ROS-induced chemical diluents and incubate for 12 h at 37 °C.
Centrifuge the 96-well plate at 112 x g and incubate with 5 µM solution of fluorescent probe (H2-DCFH-DA dissolved in RPMI media) at 37 °C for 60 min.
Wash the treated islet cells 3 times with PBS and measure the fluorescence with a microplate reader at 488 nm. Note: The accumulation of DCF in cells may be measured by an increase in fluorescence at 530 nm when the sample is excited at 488 nm.
Perform the glucose-stimulated insulin secretion (GSIS) assay9 on ROS-inducing xenobiotic treated islets and untreated islets to measure the xenobiotic effects on the physiological function of the pancreatic islets.
Representative Results
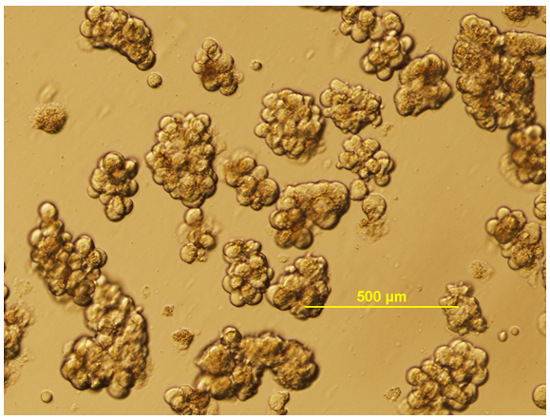
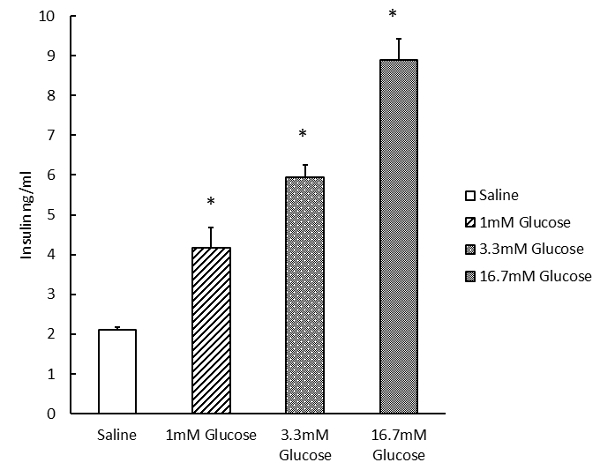
A micrograph of the healthy Isolated islet is shown in Figure 2, in which islets have a round or oval shape with relatively uniform size (although size uniformity can vary from strain to strain). We next investigated the pancreatic islet functions in an in vitro assay by isolating the islet and stimulating the insulin secretion in the culture islets. Figure 3 shows our typical analysis of the GSIS assay from C57BL/6 mouse isolated islets induced by 3.3 mM and 16.7 mM glucose9.
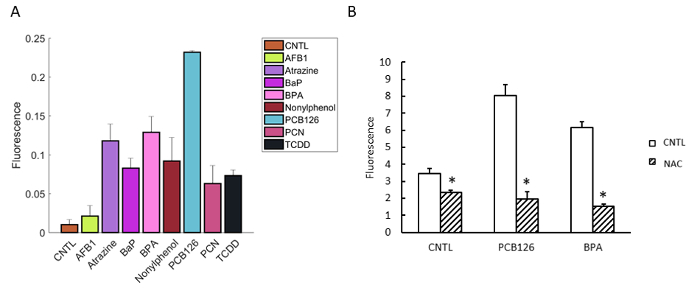
Using the plate format screen method, we have identified several xenobiotics that caused significant induction of ROS in islet cells (Figure 4). We treated the isolated islets with different chemicals for 12 h and found that Atrazine, BPA, BaP, and PCB126, and β-estrogen could cause significant induction of ROS in the isolated islets10,11,12,13.
Although ROS is not equal to the diabetogenic induction, there is ample evidence indicating the important role of ROS in causing damage to the physiological functions of pancreatic β cells, which leads to T2DM8. Therefore, the value of the work is to identify these ROS inducing chemical as the initial screen. Furthermore, the compounds identified can be further analyzed in vitro in the isolated pancreatic islet cell culture system described in the manuscript. We have shown the effects of ROS-inducing xenobiotics on glucose-stimulating insulin secretion of the isolated islets, which briefly provide diabetogenic data for these tested compounds. Pancreatic islets pre-treated with PCB126 and BPA showed a significant decrease in insulin secretion than the control group (Figure 5).
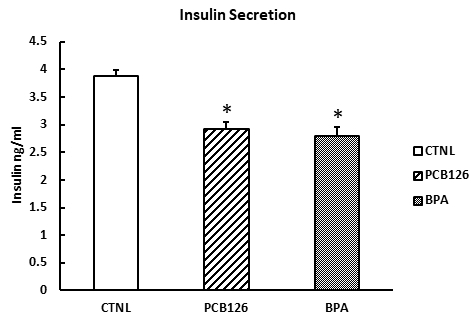
Figure 1: Procedure of mouse pancreas perfusion. The procedure consists of the following steps: (1)Find the ampulla and use two pairs of curved forceps to clamp the ampulla. (2) Insert the needle through the common bile. (3) Slowly inject 3 mL of collagenase solution stock in the 5 mL syringe. (4) Collect the distended pancreas starting from the duodenum.
Figure 2: Isolated mouse islets. Isolated mouse pancreatic islets in culture medium are shown. Scale bar of 500 µm.
Figure 3: Glucose-stimulated insulin secretion of isolated pancreatic islets. Dose-dependent glucose-stimulated insulin secretion (1, 3.3, and 16.7 mM, treated for 15 min) was measured in isolated islets (average 5 islets per well), using a mouse insulin ELISA kit. Error bars show standard deviations. *p <0.05.
Figure 4: Induction of ROS by xenobiotics in isolated islets from C57BL/6 mice. (A) Isolated islets were treated for 12 h and stained with fluorescence dye 2',7'-dichlorofluorescein (DCFH-DA) for 1 h. The fluorescence was determined by a fluorescence plate reader. (B) The chemical-induced ROS were quenched by NAC (5 mM). (AFB1: 3 µM, Atrazine: 100 µM, BaP: 10 µM, BPA: 100 µM, PCB126: 10 µM, Nonylphenol, BPA: 100 µM, and NAC: 5 mM). Error bars show standard deviations. *p <0.05. Please click here to view a larger version of this figure.
Figure 5: Xenobiotic effects on glucose-stimulated insulin secretion of isolated islets. Isolated islets were pre-treated with PCB126, β-estrogen, and BPA (4 h) and kept in islet culture medium for 1 h. The supernatant of the islets was collected for measurement of insulin by a commercially available mouse insulin ELISA assay. Error bars show standard deviations. *p <0.05.
Discussion
Accumulating evidence suggests that exposure to environmental chemicals plays an important role in the development of T2DM. Xenobiotics-induced ROS has been recognized as a potential etiological factor contributing to the development of T2DM. Humans are exposed to a wide range of xenobiotic chemicals and there is a great need for novel research techniques to effectively identify the pancreatic toxicants and to investigate the mechanism of toxicity specific to the pancreatic cells.
In this study, based on published procedures14, we have developed a protocol to isolate pancreatic islets from mouse and screen the cells with xenobiotic chemicals for oxidative stress-induced damage to the pancreatic cells. We also use this procedure to investigate the pancreatic specific toxic responses induced by the xenobiotic compounds. To obtain the pancreas from mouse, we prefer to use avertin over other anesthesia because it does not alter the blood glucose levels and does not affect the vasculature of the pancreas15,16,17. We found that the density gradient centrifugation step is critical for isolation of the islet with high purity. Care should be taken to obtain the distinct layer between the polysucrose/sodium and isolation buffer phase to obtain a pure islets sample, devoid of debris and exocrine cells/tissues. The method can be used for in vitro analysis of environmental chemical effects on pancreatic islet physiology, such as the GSIS assay (as shown in Figure 3). The islets should be carefully dispersed with repeated pipetting into separate cells to ensure uniformed cell number in the 96-cell culture plate. The method described here combined the isolation of the islet and a rapid assay for ROS generation and therefore, is a simple and effective initial screen for potential pancreas-damaging chemicals.
We used insulin secretion in response to glucose challenge to confirm the identity of the isolated pancreatic islets. The procedure is highly adaptable for analysis of changes in parameters, including insulin secretion (Figure 3) and cAMP generation, in response to xenobiotic treatments18.
Disclosures
The authors declare that they have no competing financial interests.
Acknowledgments
This work was supported by a pilot project grant from CREH center sponsored by NIEHS and by National Natural Science Foundation of China (No. 31572626).
References
- Maruthur NM. The growing prevalence of type 2 diabetes: increased incidence or improved survival? Current diabetes reports. 2013;13(6):786–794. doi: 10.1007/s11892-013-0426-4. [DOI] [PubMed] [Google Scholar]
- Houstis N, Rosen ED, Lander ES. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature. 2006;440(7086):944–948. doi: 10.1038/nature04634. [DOI] [PubMed] [Google Scholar]
- Ma ZA, Zhao Z, Turk J. Mitochondrial dysfunction and beta-cell failure in type 2 diabetes mellitus. Exp Diabetes Res. 2012. p. 703538. [DOI] [PMC free article] [PubMed]
- Valavanidis A, Vlahogianni T, Dassenakis M, Scoullos M. Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicology and environmental safety. 2006;64(2):178–189. doi: 10.1016/j.ecoenv.2005.03.013. [DOI] [PubMed] [Google Scholar]
- Robertson RP, Harmon J, Tran PO, Tanaka Y, Takahashi H. Glucose toxicity in β-cells: type 2 diabetes, good radicals gone bad, and the glutathione connection. Diabetes. 2003;52(3):581–587. doi: 10.2337/diabetes.52.3.581. [DOI] [PubMed] [Google Scholar]
- Kaneto H, et al. Oxidative stress induces p21 expression in pancreatic islet cells: possible implication in beta-cell dysfunction. Diabetologia. 1999;42(9):1093–1097. doi: 10.1007/s001250051276. [DOI] [PubMed] [Google Scholar]
- Maechler P, Jornot L, Wollheim CB. Hydrogen peroxide alters mitochondrial activation and insulin secretion in pancreatic beta cells. Journal of Biological Chemistry. 1999;274(39):27905–27913. doi: 10.1074/jbc.274.39.27905. [DOI] [PubMed] [Google Scholar]
- Gao D, et al. The effects of palmitate on hepatic insulin resistance are mediated by NADPH Oxidase 3-derived reactive oxygen species through JNK and p38MAPK pathways. Journal of Biological Chemistry. 2010;285(39):29965–29973. doi: 10.1074/jbc.M110.128694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Efendić S, et al. Pancreastatin and islet hormone release. Proceedings of the National Academy of Sciences. 1987;84(20):7257–7260. doi: 10.1073/pnas.84.20.7257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian Y, Ke S, Denison MS, Rabson AB, Gallo MA. Ah receptor and NF-κB interactions, a potential mechanism for dioxin toxicity. Journal of Biological Chemistry. 1999;274(1):510–515. doi: 10.1074/jbc.274.1.510. [DOI] [PubMed] [Google Scholar]
- Cui H, et al. Pregnane X receptor regulates the AhR/Cyp1A1 pathway and protects liver cells from benzo-[α]-pyrene-induced DNA damage. Toxicology Letters. 2017;275:67–76. doi: 10.1016/j.toxlet.2017.03.028. [DOI] [PubMed] [Google Scholar]
- Li LA, Wang PW. PCB126 induces differential changes in androgen, cortisol, and aldosterone biosynthesis in human adrenocortical H295R cells. Toxicological Sciences. 2005;85(1):530–540. doi: 10.1093/toxsci/kfi105. [DOI] [PubMed] [Google Scholar]
- Asahi J, et al. Bisphenol A induces endoplasmic reticulum stress-associated apoptosis in mouse non-parenchymal hepatocytes. Life sciences. 2010;87(13):431–438. doi: 10.1016/j.lfs.2010.08.007. [DOI] [PubMed] [Google Scholar]
- Szot GL, Koudria P, Bluestone JA. Murine pancreatic islet isolation. JoVE (Journal of Visualized Experiments) 2007. p. e255. [DOI] [PMC free article] [PubMed]
- Kirstetter P, Lagneau F, Lucas O, Krupa Y, Marty J. Role of endothelium in the modulation of isoflurane-induced vasodilatation in rat thoracic aorta. British journal of anaesthesia. 1997;79(1):84–87. doi: 10.1093/bja/79.1.84. [DOI] [PubMed] [Google Scholar]
- Brown E, Umino Y, Loi T, Solessio E, Barlow R. Anesthesia can cause sustained hyperglycemia in C57/BL6J mice. Visual neuroscience. 2005;22(5):615–618. doi: 10.1017/S0952523805225105. [DOI] [PubMed] [Google Scholar]
- Vaupel D, McCoun D, Cone EJ. Phencyclidine analogs and precursors: rotarod and lethal dose studies in the mouse. Journal of Pharmacology and Experimental Therapeutics. 1984;230(1):20–27. [PubMed] [Google Scholar]
- Neuman JC, Truchan NA, Joseph JW, Kimple ME. A method for mouse pancreatic islet isolation and intracellular cAMP determination. Journal of visualized experiments: JoVE. 2014. [DOI] [PMC free article] [PubMed]


