Abstract
Myelodysplastic syndromes (MDS) are characterized by ineffective hematopoiesis and may progress to acute myeloid leukemia (AML). MicroRNAs (miRNA/miRs) as oncogenes or tumor suppressors regulate a number of biological processes including cell proliferation, cell cycle and apoptosis in different types of cancer cells. Recently, it has been reported that miR-21 as an oncogene is overexpressed and directly targets SMAD-7 in MDS. However, little is known about the mechanism of miR-21 in the progression of MDS. In the present study, the role of miR-21 in the proliferation and apoptosis of SKM-1 cells, an acute myeloid leukemia cell line established in the AML/MDS leukemic phase was investigated. The present results demonstrated that downregulation of miR-21 inhibited proliferation, induced apoptosis and caused G1 phase cell cycle arrest of SKM-1 cells. In addition, the expression levels of apoptosis regulator Bcl-2 (bcl2), cyclinD1 and phosphorylated-protein kinase B (AKT) were significantly decreased in SKM-1 cells transfected with the miR-21 inhibitor, whilst the expression levels of phosphatase and tensin homolog (PTEN), bcl-associated protein X (bax) and cleaved caspase 3 were significantly elevated. Furthermore, knockdown of Akt by small interfering (si)RNA significantly increased the expression of bax, cleaved caspase 3 and reduced the expression of bcl2 and cyclinD1 in SKM-1 cells. Taken together, these data indicate that miR-21 targets the PTEN/AKT pathway in the pathogenesis of MDS and could be a potential target for MDS therapy.
Keywords: micro-RNA-21, phosphatase and tensin homolog/protein kinase B pathway, apoptosis, proliferation, myelodysplastic syndrome, SKM-1 cells
Introduction
Myelodysplastic syndromes (MDS), as clonal malignant diseases of the hematopoietic stem cells, are characterized by ineffective hemopoiesis of stem or progenitor cells, which leads to peripheral blood cytopenias and may progress to acute myeloid leukemia (AML) in 30% of MDS patients (1). Although numerous types of therapy including immunomodulatory agents, low-dose chemotherapy and hematopoietic stem cell transplantation have been developed based on the molecular mechanisms of MDS, the current treatment methods only alleviate the symptoms, and, therefore, an in-depth understanding of the disease pathogenesis as well as biological alterations are necessary for the treatment of patients with MDS.
microRNAs (miRNA/miRs) are short single-stranded RNAs that serve roles in regulation of gene expression at the post-transcriptional level (2). In recent years, increasing number of studies have demonstrated that miRNAs may act as oncogenes or tumor suppressors regulating numerous biological processes including cell proliferation, cell cycle and apoptosis (3–5). In addition, it has been reported that dysregulation of miRNAs is involved in the pathogenesis of MDS (6). For example, miR-22 as a potent proto-oncogene is upregulated in MDS and contributes to the onset of hematological malignancies by negatively regulating the expression of methylcytosine dioxygenase 2 (7). Kuang et al (8) has reported that miR-378 inhibits cell growth and enhances apoptosis in human MDS. In addition, miR-21 has been demonstrated to be dysregulated in many types of cancer acting as an oncogene promoting cell proliferation, migration and invasion (9,10). Furthermore, miR-21 is overexpressed and directly targets mothers against decapentaplegic (SMAD)-7 in MDS (11). Therefore, the expression levels of SMAD-7 are markedly reduced which leads to ineffective hematopoiesis by overactivation of transforming growth factor-β signaling in MDS. To date, the majority of functional analyses of miR-21 focused on various human cancers, including colon (12), renal (13), lung (14) and cervical cancers (15). However, the mechanism underlying miR-21-mediated regulation of cell proliferation and apoptosis in MDS/AML remains to be elucidated.
In the present study, downregulation of miR-21 expression inhibited cell proliferation, induced G1 arrest and promoted apoptosis in SKM-1 cells. Furthermore, phosphatase and tensin homolog (PTEN) is a downstream target of miR-21 and miR-21 inhibitor inhibited cell proliferation, induced G1 arrest and promoted cell apoptosis by modulating the PTEN/protein kinase B (AKT) pathway. These results suggest that miR-21 could be a potential target for MDS therapy.
Materials and methods
Cell culture
SKM-1, SH-SY5Y, SRA01/04 and Kasumi-1 cell lines were purchased from Cell Bank of Chinese Academy of Sciences (Shanghai, China). The SKM-1 and SH-SY5Y cells were maintained in Dulbecco's modified Eagle's medium (DMEM; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.), 100 µg/ml streptomycin and 100 U/ml penicillin. All cells were incubated at 37°C with 5% CO2. The SRA01/04 cells were cultured in modified Eagle's medium (MEM; Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10% FBS and 1% Non-Essential Amino Acid Solution (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) at 37°C in a humidified atmosphere containing 5% CO2 and 95% air. The Kasumi-1 cells were cultured in RPMI-1640 (Gibco; Thermo Fisher Scientific, Inc.) supplemented with 15% FBS, 100 µg/ml streptomycin and 100 U/ml penicillin at 37°C in a humidified atmosphere containing 5% CO2 and 95% air.
Lentiviral vector construction and lentivirus transfection
To down-regulate miR-21 in SKM-1 cells, the inhibitor of hsa-miR-21 lentivirus gene transfer vector encoding green fluorescent protein (GFP) was constructed by Shanghai GenePharma Co., Ltd. (Shanghai, China). The sequence of the inhibitor of hsa-miR-21 5′-TAGCTTATCAGACTGATGTTGA-3′ was confirmed by sequencing (data not shown). The recombinant lentivirus of miR-21 inhibitor (LV-miR-21 inhibitor) and the control lentivirus (LV-NC, 5′-TTCTCCGAACGTGTCACGT-3′) were prepared and tittered to 1×108 transfection unit (TU)/ml. A total of ~0.5×105 SKM-1 cells were plated in each well in 24-well plates overnight at 37°C.
Following 24 h of culture, lentiviruses were diluted in 0.4 ml Iscove's Modified Dulbecco Medium (IMDM; Gibco; Thermo Fisher Scientific, Inc.) containing polybrene (5 µg/ml; Sigma-Aldrich; Merck KGaA) and added to the cells and incubated at 37°C for an additional 24 h, followed by incubation in 0.5 ml of fresh IMDM for another 24 h at 37°C, which was replaced with fresh IMDM and the cells were cultured for 48 h at 37°C. The lentivirus transduction efficiency of SKM-1 cells was determined by the detection of GFP signals by fluorescence microscopy (magnification, ×100) and flow cytometry (FACSCalibur; BD Biosciences, San Jose, CA, USA) CellQuest software version 2.0 (BD Biosciences) 96 h following transduction. Data were analysed using CellQuest software (BD Biosciences).
Cell viability assay
The proliferation of SKM-1 cells was measured using the cell counting kit-8 (CCK-8; Dojindo Molecular Technologies, Inc., Kumamoto, Japan) assay. In brief, cells at a density of 1×104 cells/well were seeded into a 96-well plate and incubated for 24 h at 37°C. Cells were transfected with LV-miR-21 inhibitor and LV-NC for a further 24 h, as described above. After 4 days of lentiviral infection, a total of 10 µl CCK-8 solution was added to each well and incubated for 4 h at 37°C. The absorbance value of each well was measured with a microplate reader (Multiskan MK3; Thermo Fisher Scientific, Inc.) at a wavelength of 450 nm. All experiments were performed thrice and the results are presented as the mean ± standard deviation.
Cell cycle assay
The cell cycle distribution of SKM-1 cells was analyzed by flow cytometry (BD Biosciences) using a Propidium Iodide staining kit (BD Biosciences). Briefly, SKM-1 cells were seeded at 5×105−1×106 cells/well in 6-well plates for 24 h at 37°C. Following 96 h of viral transfection, as described above, the cell groups were collected by centrifugation at 1,000 × g for 5 min at room temperature and fixed in ice-cold 70% ethanol overnight. Subsequently, RNase A (60 µg/ml) and propidium iodide (25 µg/ml) in PBS were added, and samples were incubated for 30 min in the dark at room temperature. Finally, cells were tested using flow cytometry (FACSCalibur) at 488 nm to determine DNA content. Data were analysed using CellQuest software version 2.0.
Measurement of apoptosis by flow cytometry
Cell apoptosis was assessed using a PE-Annexin-V/7-AAD apoptosis detection kit (BD Pharmingen, San Jose, CA) and the apoptotic rate was analyzed by a flow cytometry on FACS Calibur. Data were analysed using CellQuest software version 2.0. A total of 5×105−1×106 cells/well were seeded in 6-well plates for 24 h at 37°C. Following 96-h of viral transfection, as described above, cells were harvested and washed with PBS 2–3 times at 37°C for 5 min. Subsequently, cells were resuspended at a density of 1×106 cells/ml, stained with Annexin V-PE and counterstained with 7-AAD in binding buffer (included in kit) at room temperature for 15 min. The apoptotic cells were measured using a flow cytometer with 488 nm excitation and 578 nm emission for Annexin V-PE detection, and 488 nm excitation and 647 nm emission for 7-AAD detection.
RNA extraction and reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted from homogenized cell samples using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.) and treated with DNase (Promega Corporation, Madison, WI, USA). SKM-1 cells were analyzed following 4 days of culture post lentiviral transfection, as described above. For each sample, 2 µg of RNA was used for complementary (c)DNA synthesis with the All-in-One™ miRNA First-Strand cDNA Synthesis kit (GeneCopoeia, Inc., Rockville, MD, USA) containing universal qPCR primers, and was reverse transcribed at 37°C for 60 min. The primers used were as follows: miRNA-21, forward 5′-TAGCTTATCAGACTGATGTTGA-3′; U6, forward 5′-CTCGCTTCGGCAGCACA-3′ and reverse 5′-ACGCTTCACGAATTTGCGT-3′. were obtained from GeneCopoeia. The expression of miRNA-21 was determined using All-in-One miRNA qPCR Detection kit (GeneCopoeia, Inc., Rockville, MD, USA) and the U6 gene was used as a control for normalization. The PCR reaction was conducted at 95°C for 10 min, followed by 40 cycles of denaturing at 95°C for 10 sec, annealing at 60°C for 20 sec, and extension at 72°C for 10 sec. The relative level of miR-21 was calculated with the comparative Cq method (2−ΔΔCq) (16). qPCR was performed with SYBR Green I (included in kit) on ABI 7300 (Applied Biosystems; Thermo Fisher Scientific, Inc.).
Western blot analysis
Cells were seeded in 6-well plates at a density of 2×106 cells/well with 2 ml complete DMEM (10% FBS, 100 µg/ml streptomycin and 100 U/ml penicillin) for 24 h at 37°C. Following 96 h of viral transfection, as described above, the cells were washed with PBS and lysed with radioimmunoprecipitation assay lysis buffer (150 mmol/l NaCl, 50 mmol/l Tris-HCl, pH 7.4, 1% Triton X-100, 1% sodium deoxycholate, 0.1% SDS) with 1 mM sodium orthovanadate, 1 mM PMSF, and 1% cocktail of protease inhibitors (Sigma-Aldrich; Merck KGaA). The protein concentration was determined using the BCA method. Equal quantities of proteins (20 µg) were separated by 10–15% SDS-PAGE and transferred by electroblotting onto a nitrocellulose membrane. After blocking with 5% nonfat milk for 2 h at room temperature, membranes were incubated with various primary antibodies overnight at 4°C: AKT (1:1,000 dilution; cat. no. 9272), phosphorylated (p)-AKT (1:1,000 dilution; cat. no. 4060), cyclin D1 (1:1,000 dilution; cat. no. 2978), cleaved caspase 3 (1:1,000 dilution; cat. no. 9661), apoptosis regulator Bcl-2 (bcl-2; 1:1,000 dilution; ab182858), bcl-associated protein X (bax; 1:1,000 dilution; ab32503) or GAPDH (1:3,000 dilution; cat. no. 5174) followed by incubation with the corresponding horseradish peroxidase-conjugated secondary antibody (1:20,000 dilution; cat. no. 7074) for 2 h at room temperature. Primary antibodies and secondary antibodies were obtained from Abcam (Cambridge, UK) and Cell Signaling Technology, Inc. (Danvers, MA, USA), respectively. Visualization was achieved using SuperSignal West Pico chemiluminescent substrate (Pierce; Thermo Fisher Scientific, Inc.). Densitometry of western blots was performed using ImageJ version 1.38× software (National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
All the data were expressed as the mean ± standard deviation from three independent experiments. Differences between groups were determined by one-way analysis of variance followed by Dunnett's or Tukey's post hoc tests. P<0.05 was considered to indicate a statistically significant difference. Data were analyzed using GraphPad Prism 6.0 (GraphPad Software Inc., La Jolla, CA, USA).
Results
miR-21 is highly expressed in the SKM-1 cell line
In the present study, SKM-1, Kasumi-1 and SH-SY5Y cells were used as a model of cancer cells, and SRA01/04 cells as a model of non-cancer cells. The expression level of miR-21 was analyzed in SRA01/04, SKM-1, Kasumi-1 and SH-SY5Y four cell lines. miR-21 expression was relatively increased in SKM-1 cells compared with SRA01/04 and SH-SY5Y cells (Fig. 1). Kasumi-1 cells exhibited the highest expression levels of miR-21; however, this cell line is often used as a model of AMl. Therefore, based on the expression profile of the analysed cell lines, SKM-1 cells as a MDS cell model were selected for the following loss-of-function studies.
Figure 1.
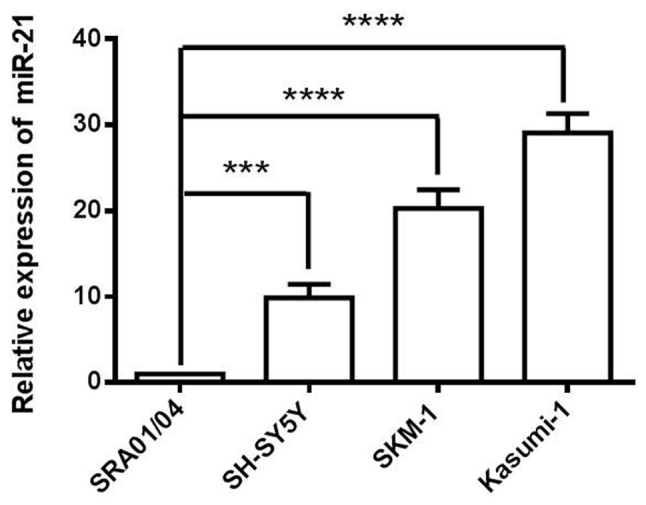
Reverse transcription-quantitative polymerase chain reaction analysis of miR-21 expression in four human normal and cancer cell lines (Kasumi-1, SRA01/04, SH-SY5Y and SKM-1 cells). ***P<0.001, ****P<0.0001 vs. SRA01/04. miR, microRNA.
miR-21 inhibitor downregulates miR-21 in SKM-1 cells
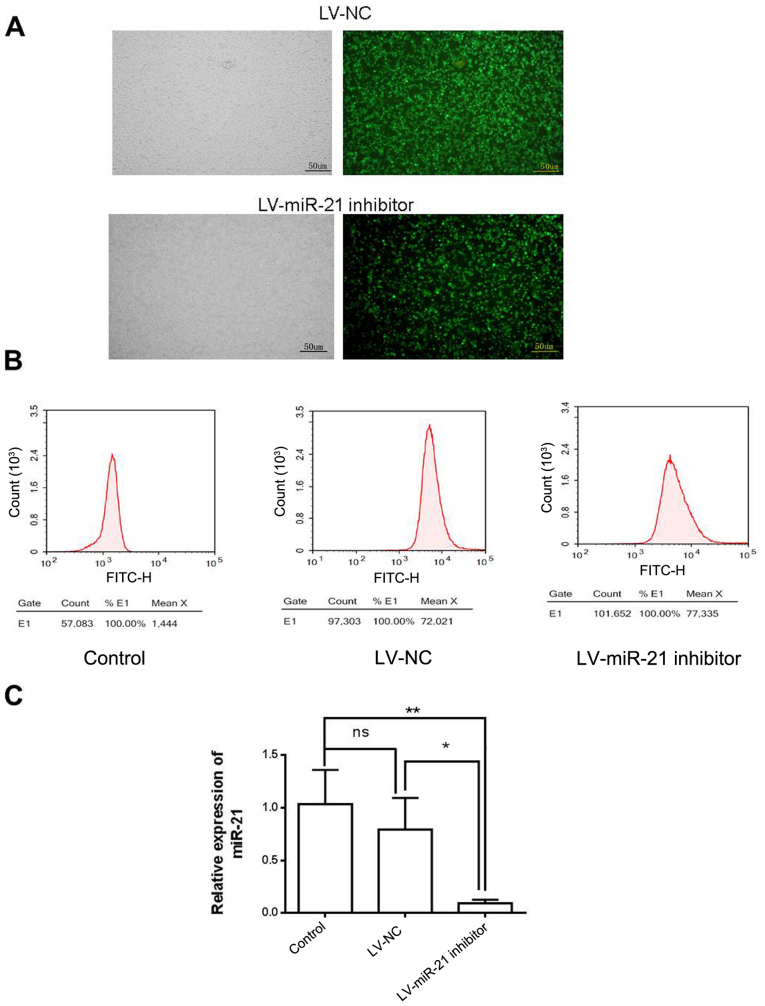
To investigate the molecular role of miR-21 in SKM-1 cells, a lentiviral vector system (LV-miR-21 inhibitor), which incorporated GFP as a reporter gene, was used to downregulate the expression of miR-21 (Fig. 2A). Following lentiviral transfection, a high percentage of cells in the LV-NC group (71.5%) and LV-miR-21 inhibitor group (73.5%) expressed GFP (Fig. 2B), indicating efficient transfection. RT-qPCR demonstrated that the expression level of miR-21 was similar in LV-NC and blank control groups. However, miR-21 expression level was lower in the LV-miR-21 inhibitor group compared with LV-NC and blank control groups, indicating that miR-21 was successfully downregulated in SKM-1 cells (Fig. 2C).
Figure 2.
LV-miR-21 inhibitor and LV-NC were successfully introduced into SKM-1 cells. (A) SKM-1 cells trnasfected with LV-miR-21 inhibitor and LV-NC were observed under the bright field and green fluorescent field of a fluorescence microscope (×40). (B) Infection efficiency was examined by flow cytometry. (C) Expression of miR-21 was significantly decreased in the LV-miR-21 inhibitor group compared with LV-NC and blank groups. *P<0.05. **P<0.01. LV, lentivirus; miR, microRNA; NC, negative control; ns, non-significant.
miR-21 inhibitor reduces proliferation of SKM-1 cells
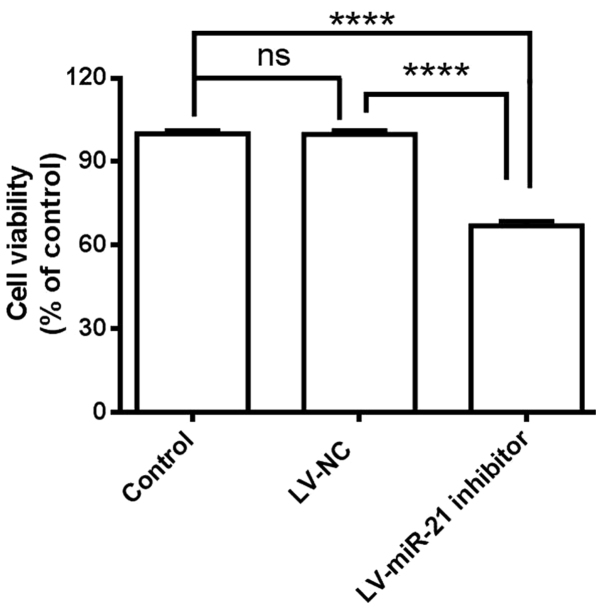
Based on the above results, an association between miR-21 and SKM-1 proliferation was hypothesized. miR-21 was inhibited in SKM-1 cells and cell viability was measured with CCK-8 assay. Suppression of miR-21 by transfection with the LV-miR-21 inhibitor significantly decreased the proliferation of SKM-1 cells compared with LV-NC and blank control cells (Fig. 3). These results indicated that miR-21 inhibitor exhibits inhibitory effects on the proliferation of SKM-1 cells.
Figure 3.
Downregulation of miR-21 inhibits cell proliferation in SKM-1 cells. Cell viability was determined by cell counting kit-8 assay in SKM-1 cells transfected with LV-miR-21 inhibitor or LV-NC. Data are presented as the mean ± standard deviation from three independent experiments. ****P<0.0001. LV, lentivirus; miR, microRNA; NC, negative control; ns, non-significant.
miR-21 inhibitor induces G1 arrest in SKM-1 cells
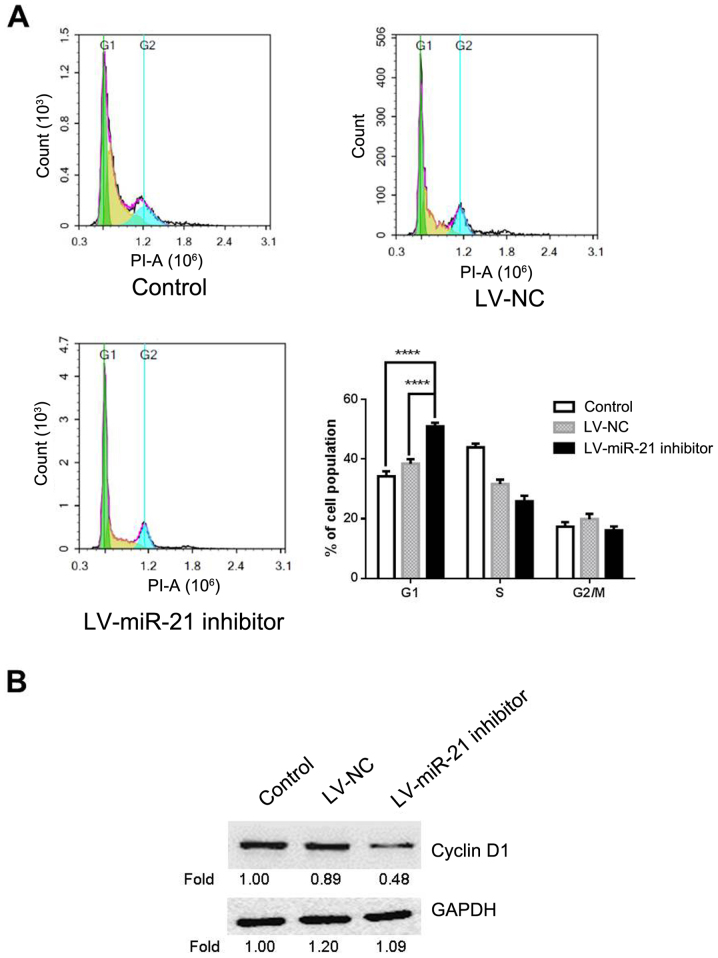
Given the suppressive effect of miR-21 inhibitor on cell proliferation, miR-21 inhibitor may affect cell cycle. Therefore, flow cytometry was carried out for cell cycle analysis in LV-miR-21 inhibitor, LV-NC and blank control groups. The results demonstrated that the percentage of cells at the G0/G1 phase in the LV-miR-21 inhibitor group (50.93%) significantly increased compared with the blank control (34.27%) and LV-NC groups (38.06%; Fig. 4A). To further elucidate the molecular mechanism underlying miR-21 inhibitor-induced G1 cell cycle arrest, the expression level of cyclin D1 in SKM-1 cells was measured. This demonstrated that LV-miR-21 inhibitor transfection markedly decreased the expression of cyclin D1 in SKM-1 cells (Fig. 4B) compared with the LV-NC and blank groups. The above results indicated that miR-21 inhibitor reduces proliferation possibly by inducing G0/G1 cell cycle arrest.
Figure 4.
Effects of downregulation of miR-21 on SKM-1 cell cycle distribution and the expression of cell cycle-associated protein cyclin D1. (A) Cell cycle was analyzed by flow cytometry in SKM-1 cells transfected with LV-miR-21 inhibitor or LV-NC and in blank control cells. (B) Protein expression levels of cyclin D1 were assayed by western blotting in LV-miR-21 inhibitor, LV-NC and blank control groups. ****P<0.0001. LV, lentivirus; miR, microRNA; NC, negative control; ns, negative control.
miR-21 inhibitor induces apoptosis in SKM-1 cells
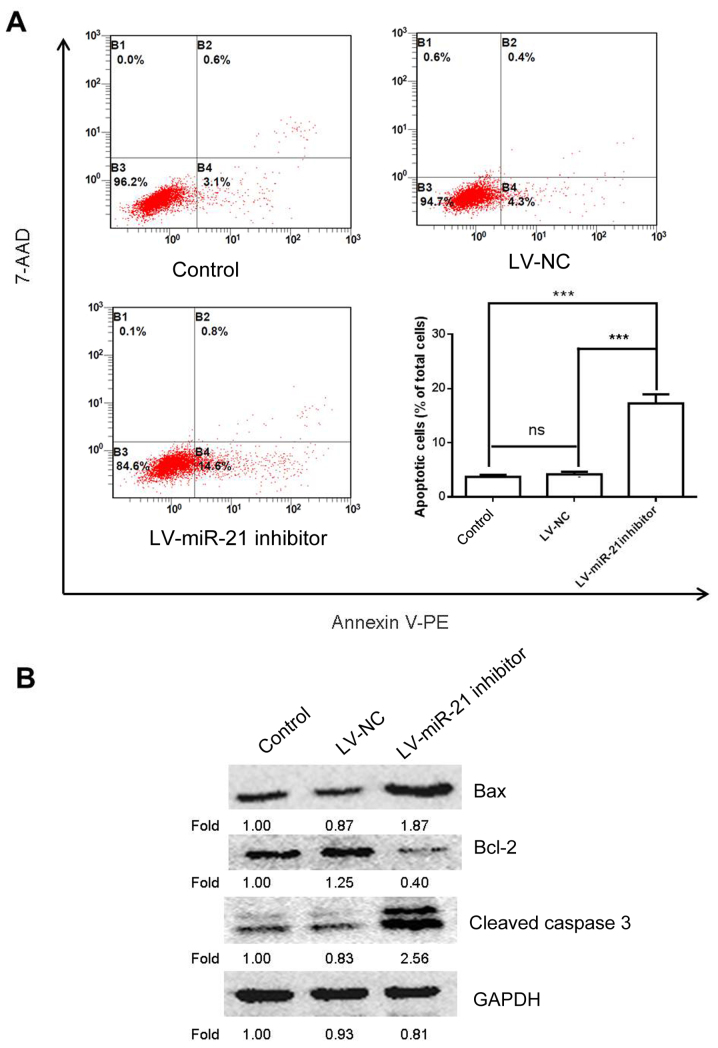
To further study the effects of miR-21 on SKM-1 cell apoptosis a flow cytometry experiment was conducted. The results demonstrated that cells transfected with the LV-miR-21 inhibitor exhibited increased apoptosis compared with the blank and NC groups (Fig. 5A). To confirm these findings, western blotting was also performed to examine the protein expression of bcl-2, bax and cleaved caspase 3, which are apoptosis-associated markers (17). As expected, transfection with the LV-miR-21 inhibitor elevated the expression level of bax and cleaved caspase 3 and decreased the expression level of bcl-2 (Fig. 5B). These results demonstrated proapoptotic effects of miR-21 inhibitor on SKM-1 cells.
Figure 5.
Effects of downregulation of miR-21 on SKM-1 cell apoptosis and the expression of cell apoptosis-associated proteins. (A) Cell apoptosis was analyzed by flow cytometry in SKM-1 cells transfected with LV-miR-21 inhibitor, or LV-NC and in the blank group group. (B) Protein expression levels of bcl-2, bax and cleaved caspase 3 were assayed by western blotting. ***P<0.001. Bax, bcl-associated protein X; bcl-2, apoptosis regulator Bcl-2; LV, lentivirus; miR, microRNA; NC, negative control; ns, negative control.
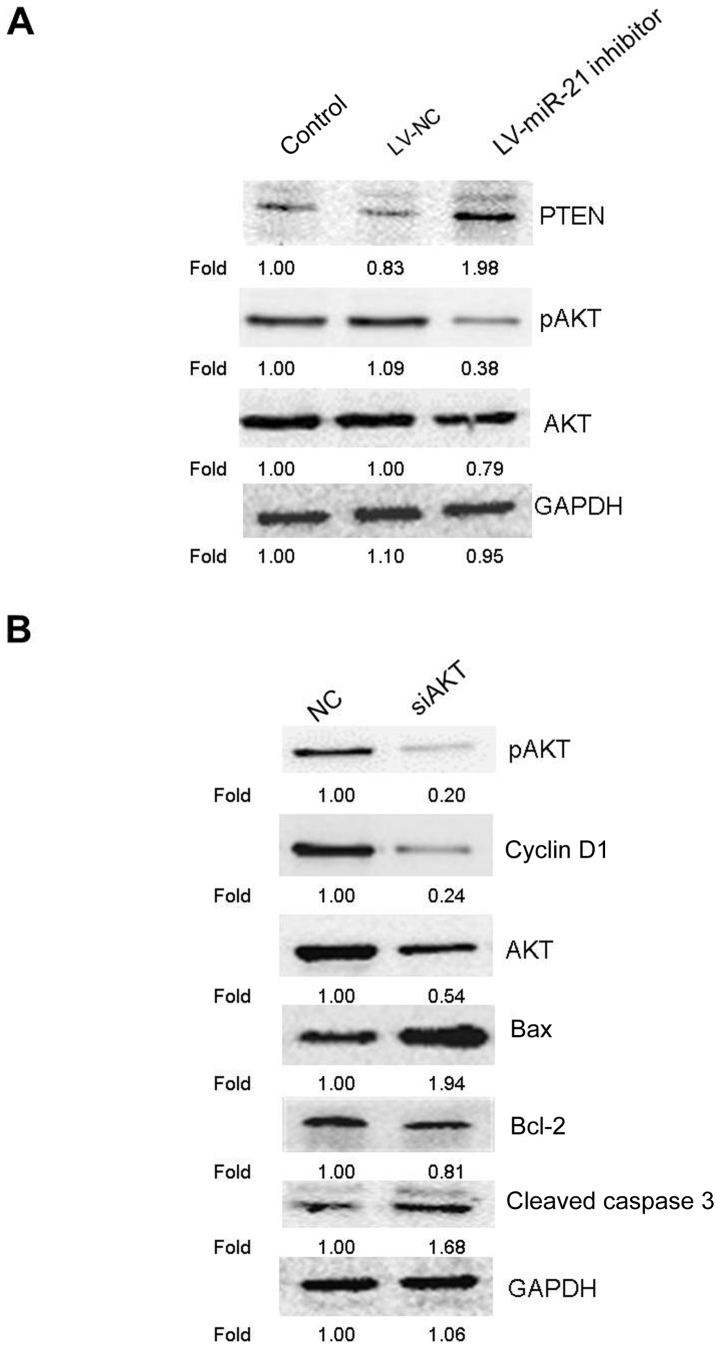
miR-21 inhibitor regulates the PTEN/AKT signaling pathway to induce apoptosis and cell cycle arrest
An increasing amount of evidence demonstrated that the PTEN/AKT pathway modulates tumor cell proliferation and apoptosis (17,18), and miR-21 has been demonstrated to control the expression and the activities of PTEN and AKT (19,20). Therefore, the effect of miR-21 inhibitor on the expression of PTEN and p-AKT was investigated. The result of western blotting revealed that, compared with the blank control and LV-NC groups, miR-21 inhibitor promoted the expression of PTEN, but suppressed the protein level of p-AKT (Fig. 6A). These results indicated that miR-21 inhibitor may regulate the PTEN/AKT pathway in SKM-1 cells. Subsequently, to further assess whether the PTEN/AKT pathway was involved in the anticancer effects of miR-21 inhibitor, the protein expression levels of cyclin D1 and apoptosis-associated proteins (bcl-2, bax and cleaved caspase 3) were examined by western blotting following treatment of SKM-1 cells with AKT siRNA. AKT knockdown markedly increased the expression of bax and cleaved caspase 3, and reduced the expression of cyclin D1, p-AKT, AKT and bcl-2 in SKM-1 cells (Fig. 6B). Numerous studies have revealed that the G0/G1 arrest may be association with reducing the expression of cyclin D1 (21–23). Taken together, these results (Fig. 6A and B) indicated that the PTEN/AKT pathway could serve an important role in regulating miR-21 inhibitor-induced apoptosis and G0/G1 cell cycle arrest in SKM-1 cells as reported previously (21–23).
Figure 6.
Apoptosis-associated proteins were detected by western blotting following AKT knockdown in SKM-1 cells. (A) Representative western blot images following knockdown of miR-21 affecting expression of PTEN, pAKT and AKT. (B) Following 48 h of transfection with siAKT or non-targeted NC into SKM-1 cells, SKM-1 cells were collected and subjected to western blot analysis for detection of apoptosis-associated protein levels. Bax, bcl-associated protein X; bcl-2, apoptosis regulator Bcl-2; LV, lentivirus; miR, microRNA; NC, negative control; ns, negative control; p, phosphorylated; si, small interference.
Discussion
Accumulated evidence has demonstrated that miRNAs are aberrantly expressed in various physiological and pathological processes, including carcinogenesis, and numerous miRNAs function as tumor suppressors or oncogenes (24–26). Previous studies have demonstrated that miR-21 is frequently upregulated and serves a role in tumorigenesis and tumor progression of glioblastoma (27), head and neck cancer (28), ovarian cancer (29), B-cell lymphoma (30), hepatocellular carcinoma (31), cervical cancer (32), prostate cancer (33), lung cancer (34) and leukemia (35). However, the potential role of miR-21 in MDS is relatively uncharacterized. SKM-1 is an acute myeloid leukemia cell line established in the leukemic phase during the progression of MDS to AML (MDS/AML) and has been used in numerous studies on myelodysplastic syndromes as a MDS cell model (8,36). In the present study, the SKM-1 cell line was used and the effects of miR-21 on cell proliferation, apoptosis, and cell cycle arrest were evaluated. SKM-1 cells were used as an in vitro model of MDS/AML to further investigate whether the cell viability and apoptosis of SKM-1 cells can be modulated by miR-21 inhibitor.
In the present study, for the first time to the best of our knowledge, it was demonstrated that miR-21 inhibitor exerts tumor suppressive function in SKM-1 cells. Functional experiments using SKM-1 cells further demonstrated that downregulation of miR-21 expression lead to suppression of cyclin D1 activity, cell cycle arrest at the G0/G1 checkpoint and inhibited cancer cell proliferation. Furthermore, suppression of miR-21 expression in SKM-1 cells could lead to upregulation of the bax expression and cleaved caspase 3 and downregulation of the bcl-2 expression, promoting cell apoptosis.
PTEN is located on human chromosome 10 in region 10q23 and functions as a tumor suppressor gene (37). It has been reported that PTEN is an important downstream target of miR-21 which regulates cancer development and progression by targeting PTEN (38). For instance, miR-21 promotes tumor growth and invasion by downregulation of PTEN in prostate cancer (19). Zheng et al (20) revealed that miR-21 modulates cisplatin sensitivity of gastric cancer by modulating the PTEN/PI3K/AKT pathway. Wang et al (39) revealed that downregulation of miR-21 enhanced imatinib-induced apoptosis, and that overexpression of miR-21 conferred imatinib resistance by modulating PTEN expression in acute lymphoblastic leukemia. In addition, loss of PTEN facilitates cell proliferation and inhibits apoptosis (40–43). It has been suggested that silencing of the PTEN gene in A549 cells significantly enhanced cell proliferation and inhibited cell apoptosis (40). Silencing PTEN expression may promote cell proliferation, decrease the rate of apoptosis of HCC827 cells and reduce the sensitivity of HCC827 cells to icotinib (41). Loss of PTEN can effectively inhibit glucocorticoid-induced apoptosis and induce resistance to glucocorticoid therapy in acute lymphoblastic leukemia (42). PTEN loss is also likely to result in cancer progression and relapse in T-cell acute lymphoblastic leukemia (43). To verify the association between miR-21 and PTEN in SKM-1 cells, western blot analysis was performed and the results demonstrated that down-regulation of miR-21 markedly elevated levels of PTEN in SKM-1 cells.
AKT is a serine/threonine kinase and AKT pathway has been widely reported to be associated with cell growth, proliferation and apoptosis in various types of tumor cells (44,45). It has also been reported that inactivation of PTEN results in constitutive activation of the PI3K/AKT pathway and increased proliferation and survival of cancer cells (18). Furthermore, up-regulation of PTEN protein inactivated the AKT signaling pathway, which further inhibited proliferation and induced apoptosis in human acute T cell leukemia cells (46). miR-21 expression was demonstrated to suppress cell proliferation and induce cell apoptosis in SKM-1 cells. Subsequently, whether AKT signaling pathway was involved in the regulation of cell proliferation and apoptosis was investigated. In response to inhibited miR-21 expression, p-AKT protein level was downregulated. AKT may regulate carcinogenesis by several downstream targets including cyclinD1, bax, bcl-2 and caspase 3 (47,48). To further confirm the role of the AKT signaling in cell proliferation and apoptosis, siAKT was used to inhibit the expression of p-AKT in SKM-1 cells. It was demonstrated that the protein levels of cyclin D1 and bcl-2 were reduced and the protein levels of bax and cleaved caspase 3 increased following transfection of SKM-1 cells with siAKT. These results indicated inactivation of Akt signaling pathway may involve in miR-21 inhibitor-mediated cell proliferation and apoptosis of SKM-1 cells.
In conclusion, the present study demonstrated that miR-21 inhibitor may act as a tumor suppressor in SKM-1 cells. Furthermore, downregulation of miR-21 expression inhibited SKM-1 cell proliferation and induced SKM-1 cell apoptosis by regulating the miR-21/PTEN/AKT axis. These results suggested that miR-21 may be a potential therapeutic target for the treatment of MDS; however, the present study was performed in vitro. Therefore the anti-tumor activity via the inhibitory effects of miR-21 requires further investigation in vivo.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Youth Natural Science Foundation of Shanxi (grant no. 2013JM4016).
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Authors' contributions
GL conducted the experiments and analyzed the data. YS made substantial contributions to the design of the present study and prepared the manuscript. GCL, JR, JX, YZ, FG, JM and JD performed the western blotting and analyzed the data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Aggerholm A, Holm MS, Guldberg P, Olesen LH, Hokland P. Promoter hypermethylation of p15INK4B, HIC1, CDH1 and ER is frequent in myelodysplastic syndrome and predicts poor prognosis in early-stage patients. Eur J Haematol. 2006;76:23–32. doi: 10.1111/j.1600-0609.2005.00559.x. [DOI] [PubMed] [Google Scholar]
- 2.Krol J, Loedige I, Filipowicz W. The widespread regulation of microRNA biogenesis, function and decay. Nat Rev Genet. 2010;11:597–610. doi: 10.1038/nrg2843. [DOI] [PubMed] [Google Scholar]
- 3.Massillo C, Dalton GN, Farre PL, De Luca P, De Siervi A. Implications of microRNA dysregulation in the development of prostate cancer. Reproduction. 2017;154:R81–R97. doi: 10.1530/REP-17-0322. [DOI] [PubMed] [Google Scholar]
- 4.Zhou K, Liu M, Cao Y. New insight into microRNA functions in cancer: Oncogene-microRNA-tumor suppressor gene network. Front Mol Biosci. 2017;4:46. doi: 10.3389/fmolb.2017.00046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sekar D, Krishnan R, Thirugnanasambantham K, Rajasekaran B, Islam VI, Sekar P. Significance of microRNA 21 in gastric cancer. Clin Res Hepatol Gastroenterol. 2016;40:538–545. doi: 10.1016/j.clinre.2016.02.010. [DOI] [PubMed] [Google Scholar]
- 6.Jang SJ, Choi IS, Park G, Moon DS, Choi JS, Nam MH, Yoon SY, Choi CH, Kang SH. MicroRNA-205-5p is upregulated in myelodysplastic syndromes and induces cell proliferation via PTEN suppression. Leuk Res. 2016;47:172–177. doi: 10.1016/j.leukres.2016.06.003. [DOI] [PubMed] [Google Scholar]
- 7.Song SJ, Ito K, Ala U, Kats L, Webster K, Sun SM, Jongen-Lavrencic M, Manova-Todorova K, Teruya-Feldstein J, Avigan DE, et al. The oncogenic microRNA miR-22 targets the TET2 tumor suppressor to promote hematopoietic stem cell self-renewal and transformation. Cell Stem Cell. 2013;13:87–101. doi: 10.1016/j.stem.2013.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kuang X, Wei C, Zhang T, Yang Z, Chi J, Wang L. miR-378 inhibits cell growth and enhances apoptosis in human myelodysplastic syndromes. Int J Oncol. 2016;49:1921–1930. doi: 10.3892/ijo.2016.3689. [DOI] [PubMed] [Google Scholar]
- 9.Pfeffer SR, Yang CH, Pfeffer LM. The role of miR-21 in cancer. Drug Dev Res. 2015;76:270–277. doi: 10.1002/ddr.21257. [DOI] [PubMed] [Google Scholar]
- 10.Selcuklu SD, Donoghue MT, Spillane C. miR-21 as a key regulator of oncogenic processes. Biochem Soc Trans. 2009;37:918–925. doi: 10.1042/BST0370918. [DOI] [PubMed] [Google Scholar]
- 11.Bhagat TD, Zhou L, Sokol L, Kessel R, Caceres G, Gundabolu K, Tamari R, Gordon S, Mantzaris I, Jodlowski T, et al. miR-21 mediates hematopoietic suppression in MDS by activating TGF-β signaling. Blood. 2013;121:2875–2881. doi: 10.1182/blood-2011-12-397067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lu Y, Han B, Yu H, Cui Z, Li Z, Wang J. Berberine regulates the microRNA-21-ITGBeta4-PDCD4 axis and inhibits colon cancer viability. Oncol Lett. 2018;15:5971–5976. doi: 10.3892/ol.2018.7997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.DeMaria AN. Clinical research in the United States-a threatened activity. J Am Coll Cardiol. 1989;13:508–510. doi: 10.1016/0735-1097(89)90533-0. [DOI] [PubMed] [Google Scholar]
- 14.Song Y, Zuo Y, Qian XL, Chen ZP, Wang SK, Song L, Peng LP. Inhibition of MicroRNA-21-5p promotes the radiation sensitivity of non-small cell lung cancer through HMSH2. Cell Physiol Biochem. 2017;43:1258–1272. doi: 10.1159/000481839. [DOI] [PubMed] [Google Scholar]
- 15.Xu L, Xu Q, Li X, Zhang X. MicroRNA-21 regulates the proliferation and apoptosis of cervical cancer cells via tumor necrosis factor-alpha. Mol Med Rep. 2017;16:4659–4663. doi: 10.3892/mmr.2017.7143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 17.Huang C, Hu G. Shikonin suppresses proliferation and induces apoptosis in endometrioid endometrial cancer cells via modulating miR-106b/PTEN/AKT/mTOR signaling pathway. Biosci Rep. 2018;38:BSR20171546. doi: 10.1042/BSR20171546. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 18.Di Cristofano A, Pandolfi PP. The multiple roles of PTEN in tumor suppression. Cell. 2000;100:387–390. doi: 10.1016/S0092-8674(00)80674-1. [DOI] [PubMed] [Google Scholar]
- 19.Yang Y, Guo JX, Shao ZQ. miR-21 targets and inhibits tumor suppressor gene PTEN to promote prostate cancer cell proliferation and invasion: An experimental study. Asian Pac J Trop Med. 2017;10:87–91. doi: 10.1016/j.apjtm.2016.09.011. [DOI] [PubMed] [Google Scholar]
- 20.Zheng P, Chen L, Yuan X, Luo Q, Liu Y, Xie G, Ma Y, Shen L. Exosomal transfer of tumor-associated macrophage-derived miR-21 confers cisplatin resistance in gastric cancer cells. J Exp Clin Cancer Res. 2017;36:53. doi: 10.1186/s13046-017-0528-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yang Y, Zhou W, Wu J, Yao L, Xue L, Zhang Q, Wang Z, Wang X, Dong S, Zhao J, Yin D. Antitumor activity of nimotuzumab in combination with cisplatin in lung cancer cell line A549 in vitro. Oncol Lett. 2018;15:5280–5284. doi: 10.3892/ol.2018.7923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yang L, Liu H, Long M, Wang X, Lin F, Gao Z, Zhang H. Peptide SA12 inhibits proliferation of breast cancer cell lines MCF-7 and MDA-MB-231 through G0/G1 phase cell cycle arrest. Onco Targets Ther. 2018;11:2409–2417. doi: 10.2147/OTT.S154337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Liu SL, Liu Z, Zhang LD, Zhu HQ, Guo JH, Zhao M, Wu YL, Liu F, Gao FH. GSK3β-dependent cyclin D1 and cyclin E1 degradation is indispensable for NVP-BEZ235 induced G0/G1 arrest in neuroblastoma cells. Cell Cycle. 2017;16:2386–2395. doi: 10.1080/15384101.2017.1383577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang B, Pan X, Cobb GP, Anderson TA. microRNAs as oncogenes and tumor suppressors. Dev Biol. 2007;302:1–12. doi: 10.1016/j.ydbio.2006.08.028. [DOI] [PubMed] [Google Scholar]
- 25.Zhou B, Wang D, Sun G, Mei F, Cui Y, Xu H. Effect of miR-21 on apoptosis in lung cancer cell through inhibiting the PI3K/Akt/NF-κB signaling pathway in vitro and in vivo. Cell Physiol Biochem. 2018;46:999–1008. doi: 10.1159/000488831. [DOI] [PubMed] [Google Scholar]
- 26.Yang C, Tabatabaei SN, Ruan X, Hardy P. The dual regulatory role of MiR-181a in breast cancer. Cell Physiol Biochem. 2017;44:843–856. doi: 10.1159/000485351. [DOI] [PubMed] [Google Scholar]
- 27.Masoudi MS, Mehrabian E, Mirzaei H. MiR-21: A key player in glioblastoma pathogenesis. J Cell Biochem. 2018;119:1285–1290. doi: 10.1002/jcb.26300. [DOI] [PubMed] [Google Scholar]
- 28.Wang AJ, Li ZW, Hu MX, Wang SD, Leng M. Ionic mechanism of noradrenaline-induced membrane potential changes of neurones in toad dorsal root ganglion. Sheng Li Xue Bao. 1989;41:145–152. (In Chinese) [PubMed] [Google Scholar]
- 29.Echevarria-Vargas IM, Valiyeva F, Vivas-Mejia PE. Upregulation of miR-21 in cisplatin resistant ovarian cancer via JNK-1/c-Jun pathway. PLoS One. 2014;9:e97094. doi: 10.1371/journal.pone.0097094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Munch-Petersen HD, Ralfkiaer U, Sjo LD, Hother C, Asmar F, Nielsen BS, Brown P, Ralfkiaer E, Grønbæk K. Differential expression of miR-155 and miR-21 in tumor and stroma cells in diffuse large B-cell lymphoma. Appl Immunohistochem Mol Morphol. 2015;23:188–195. doi: 10.1097/PAI.0000000000000073. [DOI] [PubMed] [Google Scholar]
- 31.He C, Dong X, Zhai B, Jiang X, Dong D, Li B, Jiang H, Xu S, Sun X. MiR-21 mediates sorafenib resistance of hepatocellular carcinoma cells by inhibiting autophagy via the PTEN/Akt pathway. Oncotarget. 2015;6:28867–28881. doi: 10.18632/oncotarget.4814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Peralta-Zaragoza O, Deas J, Meneses-Acosta A, De la O-Gómez F, Fernández-Tilapa G, Gómez-Cerón C, Benítez-Boijseauneau O, Burguete-García A, Torres-Poveda K, Bermúdez-Morales VH, et al. Relevance of miR-21 in regulation of tumor suppressor gene PTEN in human cervical cancer cells. BMC Cancer. 2016;16:215. doi: 10.1186/s12885-016-2231-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Reis ST, Pontes-Junior J, Antunes AA, Dall'Oglio MF, Dip N, Passerotti CC, Rossini GA, Morais DR, Nesrallah AJ, Piantino C, et al. miR-21 may acts as an oncomir by targeting RECK, a matrix metalloproteinase regulator, in prostate cancer. BMC Urol. 2012;12:14. doi: 10.1186/1471-2490-12-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xue X, Liu Y, Wang Y, Meng M, Wang K, Zang X, Zhao S, Sun X, Cui L, Pan L, Liu S. MiR-21 and MiR-155 promote non-small cell lung cancer progression by downregulating SOCS1, SOCS6 and PTEN. Oncotarget. 2016;7:84508–84519. doi: 10.18632/oncotarget.13022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jurkovicova D, Lukackova R, Magyerkova M, Kulcsar L, Krivjanska M, Krivjansky V, Chovanec M. microRNA expression profiling as supportive diagnostic and therapy prediction tool in chronic myeloid leukemia. Neoplasma. 2015;62:949–958. doi: 10.4149/neo_2015_115. [DOI] [PubMed] [Google Scholar]
- 36.Zeng W, Dai H, Yan M, Cai X, Luo H, Ke M, Liu Z. Decitabine-induced changes in human myelodysplastic syndrome cell line SKM-1 are mediated by FOXO3A activation. J Immunol Res. 2017;2017:4302320. doi: 10.1155/2017/4302320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li J, Yen C, Liaw D, Podsypanina K, Bose S, Wang SI, Puc J, Miliaresis C, Rodgers L, McCombie R, et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast and prostate cancer. Science. 1997;275:1943–1947. doi: 10.1126/science.275.5308.1943. [DOI] [PubMed] [Google Scholar]
- 38.Wu Y, Song Y, Xiong Y, Wang X, Xu K, Han B, Bai Y, Li L, Zhang Y, Zhou L. MicroRNA-21 (Mir-21) promotes cell growth and invasion by repressing tumor suppressor PTEN in colorectal cancer. Cell Physiol Biochem. 2017;43:945–958. doi: 10.1159/000481648. [DOI] [PubMed] [Google Scholar]
- 39.Wang WZ, Lin XH, Pu QH, Liu MY, Li L, Wu LR, Wu QQ, Mao JW, Zhu JY, Jin XB. Targeting miR-21 sensitizes Ph+ ALL Sup-b15 cells to imatinib-induced apoptosis through upregulation of PTEN. Biochem Biophys Res Commun. 2014;454:423–428. doi: 10.1016/j.bbrc.2014.10.107. [DOI] [PubMed] [Google Scholar]
- 40.Lu XX, Cao LY, Chen X, Xiao J, Zou Y, Chen Q. PTEN inhibits cell proliferation, promotes cell apoptosis and induces cell cycle arrest via downregulating the PI3K/AKT/hTERT pathway in lung adenocarcinoma A549 cells. Biomed Res Int. 2016;2016:2476842. doi: 10.1155/2016/2476842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhai Y, Zhang Y, Nan K, Liang X. Reduced expression levels of PTEN are associated with decreased sensitivity of HCC827 cells to icotinib. Oncol Lett. 2017;13:3233–3238. doi: 10.3892/ol.2017.5829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Piovan E, Yu J, Tosello V, Herranz D, Ambesi-Impiombato A, Da Silva AC, Sanchez-Martin M, Perez-Garcia A, Rigo I, Castillo M, et al. Direct reversal of glucocorticoid resistance by AKT inhibition in acute lymphoblastic leukemia. Cancer Cell. 2013;24:766–776. doi: 10.1016/j.ccr.2013.10.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Clappier E, Gerby B, Sigaux F, Delord M, Touzri F, Hernandez L, Ballerini P, Baruchel A, Pflumio F, Soulier J. Clonal selection in xenografted human T cell acute lymphoblastic leukemia recapitulates gain of malignancy at relapse. J Exp Med. 2011;208:653–661. doi: 10.1084/jem.20110105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lien EC, Dibble CC, Toker A. PI3K signaling in cancer: Beyond AKT. Curr Opin Cell Biol. 2017;45:62–71. doi: 10.1016/j.ceb.2017.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xue G, Zippelius A, Wicki A, Mandalà M, Tang F, Massi D, Hemmings BA. Integrated Akt/PKB signaling in immunomodulation and its potential role in cancer immunotherapy. J Natl Cancer Inst. 2015;107:djv171. doi: 10.1093/jnci/djv171. [DOI] [PubMed] [Google Scholar]
- 46.Wang Y, Chen B, Wang Z, Zhang W, Hao K, Chen Y, Li K, Wang T, Xie Y, Huang Z, Tong X. Marsdenia tenacissimae extraction (MTE) inhibits the proliferation and induces the apoptosis of human acute T cell leukemia cells through inactivating PI3K/AKT/mTOR signaling pathway via PTEN enhancement. Oncotarget. 2016;7:82851–82863. doi: 10.18632/oncotarget.12654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gao YH, Zhang HP, Yang SM, Yang Y, Ma YY, Zhang XY, Yang YM. Inactivation of Akt by arsenic trioxide induces cell death via mitochondrial-mediated apoptotic signaling in SGC-7901 human gastric cancer cells. Oncol Rep. 2014;31:1645–1652. doi: 10.3892/or.2014.2994. [DOI] [PubMed] [Google Scholar]
- 48.Gu Y, Li A, Sun H, Li X, Zha H, Zhao J, Xie J, Zeng Z, Zhou L. BCL6B suppresses proliferation and migration of colorectal carcinoma cells through inhibition of the PI3K/AKT signaling pathway. Int J Mol Med. 2018;41:2660–2668. doi: 10.3892/ijmm.2018.3451. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.