Abstract
Purpose
To evaluate liver cancer incidence rates and risk factor correlations in non-Hispanic AI/AN populations for the years 1999–2009.
Methods
We linked data from 51 central cancer registries with the Indian Health Service patient registration databases to improve identification of the AI/AN population. Analyses were restricted to non-Hispanic persons living in Contract Health Service Delivery Area counties. We compared age-adjusted liver cancer incidence rates (per 100,000) for AI/AN to white populations using rate ratios. Annual percent changes (APCs) and trends were estimated using joinpoint regression analyses. We evaluated correlations between regional liver cancer incidence rates and risk factors using Pearson correlation coefficients.
Results
AI/AN persons had higher liver cancer incidence rates than whites overall (11.5 versus 4.8, RR = 2.4, 95% CI 2.3– 2.6). Rate ratios ranged from 1.6 (Southwest) to 3.4 (Northern Plains and Alaska). We observed an increasing trend among AI/AN persons (APC 1999–2009 = 5%). Rates of distant disease were higher in the AI/AN versus white population for all regions except Alaska. Alcohol use (r = 0.84) and obesity (r = 0.79) were correlated with liver cancer incidence by region.
Conclusions
Findings highlight disparities in liver cancer incidence between AI/AN and white populations and emphasize opportunities to decrease liver cancer risk factor prevalence.
Keywords: Cancer incidence, American Indian, Alaska Native, Liver cancer, Health disparity
Introduction
Primary liver cancer incidence is increasing worldwide, including in the United States (US) [1]. Between the years 2003 and 2012, liver cancer incidence rates increased significantly, with an average annual percent change (AAPC) of 3.5 for all races and ethnicities [1]. Liver cancer incidence rates in the US varied by racial and ethnic population groups, with the highest rates in non-Hispanic American Indians/Alaska Natives (AI/AN) [1]. Age-adjusted liver cancer incidence rates (per 100,000) were higher for non-Hispanic AI/AN compared to non-Hispanic whites (NHW) for men (21.0 versus 9.3) and women (9.9 versus 3.2) [1]. Previous reports also provide evidence for differences in AI/AN cancer rates by region and by cancer type across regions [2, 3] suggesting that nationally aggregated statistics do not adequately describe liver cancer burden across the AI/AN population. This variation is potentially attributed to differences in AI/AN culture, tribal ancestry, and cancer risk factor prevalence [2].
In the United States, chronic infections with hepatitis B or hepatitis C virus (HBV, HCV) are important risk factors for hepatocellular carcinoma (HCC), the most common histologic subtype of liver cancer [4–6]. Other risk factors include cirrhosis of the liver due to alcohol consumption, hemochromatosis, non-alcoholic fatty liver disease (NAFLD), non-alcoholic steatohepatitis (NASH), and primary biliary cirrhosis [2]. Tobacco use, obesity, and diabetes have also been linked with increased liver cancer risk [7].
The present study provides a comprehensive overview of liver cancer incidence in non-Hispanic AI/AN in six previously defined Indian Health Service (IHS) regions in the US. We also describe correlations between liver cancer incidence rates and associated risk factors assessed through the Behavioral Risk Factor Surveillance System (BRFSS). We utilized the most recently available cancer incidence data (1999–2009) from central cancer registries that have been linked with the IHS patient registration databases according to previously established techniques [8] for reducing racial misclassification. The comparison group is non-Hispanic whites living in the same regions of the US. This study focuses on AI/ANs living in IHS Contract Health Service Delivery Areas (CHSDA), defined as counties containing or abutting federally recognized reservations or tribal lands, for whom IHS is responsible for medical services.
Materials and methods
Cancer cases
We utilized data from population-based registries, which participate in the National Program of Cancer Registries of the Centers for Disease Control and Prevention (CDC) and Surveillance, Epidemiology and End Results (SEER) program of the National Cancer Institute (NCI) [9, 10]. During the period covered by this study, tumor histology, tumor behavior, and primary cancer site were classified according to the Third Edition of the International Classification of Disease for Oncology (ICD-O-3).
For this study, incident cases of primary cancer of the liver and intrahepatic bile ducts were identified as invasive primary cancers using ICD-O-3 codes C22.0-C.22.1, excluding lymphomas originating in the lymphatic tissue of the liver, mesothelioma, and Kaposi’s sarcoma. Cancer cases in this study period (1999–2009) were from statewide and regional registries that provided permission and have met the standard for high-quality data according to the United States Cancer Statistics. Analyses of histologic subtype were limited to microscopically confirmed cases only. Cancer cases diagnosed between 2001 and 2009 were staged using the 2000 SEER summary staging system [11]. Analyses included all primary liver cancers (instead of HCC only) due to small numbers in AI/AN populations.
Previous data showed that racial misclassification can result in an underestimation of AI/AN cancer incidence rates; efforts to reduce racial misclassification have been described elsewhere [8]. Briefly, all case records from each state were linked with the IHS registration database to identify AI/AN cases misclassified as non-AI/AN. These linkages were conducted using LinkPlus, a probabilistic software program developed by the CDC that utilizes key patient identifiers (social security number, first name, last name, date of birth, etc.) [12]. In an effort to further improve race classification, the present analyses focus on IHS Contract Health Service Delivery Area (CHSDA) counties [13]. These counties generally contain or are located adjacent to federally recognized lands where AI/AN are more likely to access IHS services.
During previous analyses, it was discovered that the updated bridged intercensal population estimates substantially overestimated AI/AN populations of Hispanic origin [14]. To avoid underestimating rates in AI/AN populations, we limited all analyses to non-Hispanic AI/AN populations. Non-Hispanic white was chosen as the reference population. Therefore, all the analyses in the present study are limited to non-Hispanic populations. The term “non-Hispanic” will henceforth be omitted when discussing both groups in this study.
Risk factor data
We analyzed several health behavior characteristics in the AI/AN population by IHS region (CHSDA counties only) associated with liver cancer, using the BRFSS data. Briefly, the BRFSS, administered by CDC in collaboration with state health departments in the 50 states and the District of Colombia, is an annual, state-based, random-digit-dialed telephone survey of the non-institutionalized civilian adult population [15]. The BRFSS questionnaire contains a core set of questions that are asked annually and two sets of questions (rotating core) that are alternated biannually. There are also optional modules that were not included in the present analyses [15].
We included hypertension, diagnosed diabetes, smoking, obesity, and alcohol consumption in our correlation analyses of liver cancer incidence and population level risk factor data. The exact text of each question can be found on the CDC website [15]. BRFSS creates calculated variables for the most commonly used measures. We used these calculated variables when possible, merging them over time for compatibility. Risk factors were dichotomized and analyses were stratified by sex. Hypertension and diabetes were identified if respondents reported being told they had high blood pressure or diabetes (outside pregnancy for women). Ever versus never smokers were defined as current and former smokers versus never smokers. We used body mass index (BMI: measured as kilograms divided by meters squared) to calculate overweight and obese (BMI 25+) versus neither overweight nor obese (BMI < 25). Due to changes in the module of the survey, binge drinking was defined as having 5 or more drinks on 1 occasion for the years 1999–2005, and ≥ 4 drinks for women and ≥ 5 drinks for men for the years 2006–2009 with 2 categories: (1) no binge drinking; (2) binge drinking. Unknown or “Don’t Know” responses were excluded. Because of the small number of AI/AN cases, we combined responses from all the available years for each characteristic. Analysis for smoking, diabetes, and obesity included all 11 years since the modules were on the core. Analysis for alcohol consumption (binge drinking) excluded the year 2000 because limited data were available for the AI/AN population for this year. We included odd years for hypertension since this module is on the rotating core.
Population estimates
The NCI makes refinements to population estimates produced by the Census Bureau regarding race, county geographic codes, and adjustments for population shifts because of Hurricanes Katrina and Rita in 2005, and provided public access to these for calculation of incidence rates [16]. Population estimates available from the SEER website for the period 1999–2009 were used as the denominators for the rate calculations.
Statistical analyses
For these analyses, we restricted to contract health service delivery area (CHSDA) counties only. These counties, in general, contain or are adjacent to federally recognized tribal lands [17]. Previous linkage studies have suggested more accurate race classification for the AI/AN population in these counties [17]. Additional information regarding cases and population coverage have been detailed elsewhere [8]. Information regarding IHS regions and CHSDA counties are shown in Supplementary Fig. 1.
Cancer incidence rates are expressed per 100,000 persons and are adjusted by 19 age groups (< 1, 1–4, 5–0,...,80–84, 85+ years) to the 2000 US standard population by use of the direct method [18]. Percent distributions are also age-adjusted. Rate ratios with 95% confidence intervals (CI) are provided for regional comparison of incidence rates between AI/AN and white populations, overall and by sex, age group, histologic subtype, and stage. Incidence rates, rate ratios, and 95% CI are generated by use of SEER*Stat Software version 8.3.2 [18]. Comparisons of the distribution of histological subtype between the AI/AN and white populations overall were performed using Chi-square tests.
Temporal trends in age-adjusted liver cancer incidence rates were estimated by joinpoint regression using software developed by the NCI (Joinpoint Regression Program version 4.3.10) [19, 20]. Annual Percent Change (APC) was used to describe fixed interval trends from 1999 to 2009 [21].
We used the Pearson correlation coefficient to evaluate the linear association between CHSDA liver cancer incidence rates and the estimated prevalence of each of the risk factors by region (see Supplementary Fig. 1) and gender. Correlations were calculated using SAS version 9.3 (Research Triangle Institute, Research Triangle Park, NC).
Results
Cancer incidence by region and sex
Age-adjusted liver cancer incidence rates for AI/AN persons and whites (expressed per 100,000), stratified by region and sex for the years 1999–2009 are presented in Table 1. The incidence rate among AI/AN males was more than twice the rate of AI/AN females. Incidence rates among both AI/ AN males (16.1) and females (7.7) were more than twice the rates of white males and females (RR=2.2 (95% CI 2.0–2.4) and 2.9 (95% CI 2.7–3.3), respectively). We found considerable regional variation in incidence rates for AI/AN males, ranging from 10.3 (95% CI 7.2–14.3) in the East to 20.4 (95% CI 17.6–23.5) in the Pacific Coast. Incidence rates for AI/AN females ranged from 5.8 (95% CI 3.5–8.9) in the East to 8.1 (95% CI 6.1–10.5) in the Northern Plains and 8.1 (95% CI 6.8–9.6) in the Southwest. Incidence rates for white males and females were much lower.
Table 1.
Liver and Intrahepatic Bile Duct Cancer Incidence by Indian Health Service Region and Sex for American Indians/Alaska Natives and Whitesa, United States, Contract Health Service Delivery Areas (CHSDA) only, 1999–2009
| AI/AN rateb (95% CI) | AI/AN count | White ratec (95% CI) | White count | AI/AN:White RR (95% CI) | ||
|---|---|---|---|---|---|---|
| IHS region | ||||||
| Northern Plains | Overall | 12.5 (10.7–14.6) | 198 | 3.8 (3.7–3.9) | 3,687 | 3.3d (2.8–3.9) |
| Male | 17.6 (14.4–21.3) | 137 | 5.6 (5.3–5.8) | 2,448 | 3.2d (2.6–3.9) | |
| Female | 8.1 (6.1–10.5) | 61 | 2.3 (2.2–2.4) | 1,239 | 3.5d (2.7–4.6) | |
| Alaska | Overall | 10.0 (7.7–12.7) | 75 | 5.6 (4.8–6.4) | 241 | 1.8d (1.3–2.4) |
| Male | 13.4 (9.3–18.6) | 46 | 8.3 (7.0–9.9) | 182 | 1.6d (1.1–2.3) | |
| Female | 7.3 (4.8–10.5) | 29 | 2.9 (2.1–3.8) | 59 | 2.5d (1.5–4.1) | |
| Southern Plains | Overall | 10.9 (9.6–12.3) | 270 | 4.9 (4.7–5.2) | 1,914 | 2.2d (1.9–2.5) |
| Male | 14.9 (12.7–17.4) | 176 | 7.2 (6.8–7.6) | 1,260 | 2.1d (1.7–2.5) | |
| Female | 7.3 (5.8–8.9) | 94 | 3.0 (2.8–3.3) | 654 | 2.4d (1.9–3.0) | |
| Southwest | Overall | 11.0 (9.8–12.3) | 343 | 4.5 (4.4–4.7) | 4,005 | 2.4d (2.2–2.7) |
| Male | 14.7 (12.6–17) | 208 | 6.8 (6.6–7.1) | 2,842 | 2.2d (1.8–2.5) | |
| Female | 8.1 (6.8–9.6) | 135 | 2.5 (2.3–2.6) | 1,163 | 3.2d (2.7–4.0) | |
| Pacific Coast | Overall | 13.7 (12.2–15.5) | 320 | 5.1 (5.0–5.2) | 9,388 | 2.7d (2.4–3.0) |
| Male | 20.4 (17.6–23.5) | 232 | 7.7 (7.5–7.9) | 6,614 | 2.6d (2.3–3.1) | |
| Female | 7.9 (6.3–9.9) | 88 | 2.8 (2.7–2.9) | 2,774 | 2.8d (2.2–3.5) | |
| East | Overall | 8.2 (6.1–10.6) | 63 | 5.0 (4.9–5.1) | 9,357 | 1.6d (1.2–2.1) |
| Male | 10.3 (7.2–14.3) | 42 | 8.0 (7.8–8.2) | 6,665 | 1.3 (0.9–1.8) | |
| Female | 5.8 (3.5–8.9) | 21 | 2.5 (2.4–2.6) | 2,692 | 2.3d (1.4–3.5) | |
| Total US | Overall | 11.5 (10.9–12.2) | 1,269 | 4.8 (4.7–4.8) | 28,592 | 2.4d (2.3–2.6) |
| Male | 16.1 (14.9–17.3) | 841 | 7.3 (7.2–7.4) | 20,011 | 2.2d (2.0–2.4) | |
| Female | 7.7 (7.0–8.5) | 428 | 2.6 (2.6–2.7) | 8,581 | 2.9d (2.7–3.3) |
Source: Cancer Registries in the National Program of Cancer Registries (NCPR) of the Centers for Disease Control and Prevention and the Surveillance, Epidemiology, and End Results program of the National Cancer Institute
CHSDA contract health service delivery areas, IHS Indian Health Service, AI/AN American Indian/Alaska Natives
IHS Regions are defined as follows: Alaska; Northern Plains (IL, IN,* IA,* MA,* MN,* MT,* NE,* ND,* SD,* WI,* WY*); Southern Plains (OK,* KS,* TX*); Southwest (AZ,* CO,* NV,* NM,* UT*); Pacific Coast (CA,* ID,* OR,* WA,* HI); East (AL,* AR, CT,* DE, FL,* GA, KY, LA,* ME,* MD, MA,* MS,* MO, NH, NJ, NY,* NC,* OH, PA,* RI,* SC,* TN, VT, VA, WV, DC); *Identifies states with at least 1 country designated as CHSDA. Percent regional coverage of AI/AN persons in CHSDA counties to AI/AN persons in all counties: Alaska = 100%; East = 14.9%; Northern Plains = 55.0%; Southern Plains = 58.0%; Pacific Coast = 54.1%; Southwest = 84.8%
AI/AN race is reported by NPCR and SEER registries or through linkage with the IHS patient registration database. The updated bridged inter-censal population estimates significantly overestimate AI/AN populations of Hispanic origin. All analyses are limited to non-Hispanic AI/AN populations. Non-Hispanic White was chosen as the reference population. The term “non-Hispanic” is omitted when discussing both groups
Rates are per 100,000 and age-adjusted to the 2000 US Std Population (19 age groups Census P25–1130) standard; Confidence Intervals (Tiwari model) are 95% for rates and ratios
The rate ratio indicates that the rate is significantly different than the rate for Whites (p < 0.05)
Histologic subtype
Out of the 1,269 cases in AI/AN, 823 were histologically confirmed (64.8%), and 19,965 out of the 28,592 white cases (70%) were histologically confirmed. The most common histologic classifications in the AI/AN population were HCC (76.2%) and cholangiocarcinoma (8.7%) (Table 2). These histologic classifications were also the most common among whites (69.2 and 13.1%, respectively). These data suggest a higher proportion of HCC among AI/AN compared to whites overall (p < 0.001.)
Table 2.
Invasive liver and intrahepatic bile duct cancer percentage histology distribution among microscopically confirmed cases by Indian Health Service region for American Indians/Alaska Natives and Whitesa CHSDA Counties, United States, 1999–2009
| IHS region | Adenocarcinoma NOSb |
Cholangiocarcinomac |
Combined hepatocellular and cholangiocarcinomad |
Hepatocellular carcinome |
Other adenocarcinomasf |
Other malignant histologies |
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AI/AN (%)g | White (%)g | AI/AN (%)g | White (%)g | AI/AN (%)g | White (%)g | AI/AN (%)g | White (%)g | AI/AN (%)g | White (%)g | AI/AN (%)g | White (%)g | |
| (N = 36) | (N = 1.458) | (N=72) | (N=2.621) | (N = 6) | (N=243) | (N = 627) | (N= 13.808) | (N=24) | (N= 537) | (N = 58) | (N= 1,298) | |
| Northern Plains | 5.6 | 10.4 | 12.7 | 16.5 | 0.7 | 1.4 | 71.8 | 62.5 | 3.5 | 2.8 | 5.6 | 6.5 |
| Alaska | 0.0 | 5.1 | 9.8 | 13.6 | 0.0 | 2.3 | 74.5 | 67.6 | 5.9 | 2.3 | 9.8 | 9.1 |
| Southern Plains | 7.3 | 10.8 | 5.7 | 11.5 | 1.0 | 0.8 | 73.1 | 60.8 | 3.1 | 3.6 | 9.8 | 12.4 |
| Southwest | 4.5 | 6.4 | 9.4 | 10.1 | 0.5 | 1.1 | 73.3 | 73.1 | 4.0 | 2.8 | 8.4 | 6.6 |
| Pacific Coast | 2.1 | 6.2 | 9.0 | 13.5 | 0.5 | 1.0 | 84.6 | 70.9 | 0.0 | 2.8 | 3.7 | 5.7 |
| East | 2.1 | 6.8 | 4.3 | 12.9 | 2.1 | 1.4 | 83.0 | 70.5 | 4.3 | 2.3 | 4.3 | 6.0 |
| Total US | 4.4 | 7.3 | 8.7 | 13.1 | 0.7 | 1.2 | 76.2 | 69.2 | 2.9 | 2.7 | 7.0 | 6.5 |
| Chi-2 p value | 0.001 | 0.002 | 0.2 | <0.001 | 0.69 | 0.53 | ||||||
If no cases were reported, then percent distribution could not be calculated
Source: Cancer Registries in the National Program of Cancer Registries (NCPR) of the Centers for Disease Control and Prevention and the Surveillance, Epidemiology, and End Results program of the National Cancer Institute; see Table 1 for states included
CHSDA contract health service delivery areas, IHS Indian Health Service, AH AN American Indian/Alaska Natives, Chi-2 Chi-squared
AI/AN race is reported by NPCR and SEER registries or through linkage with the IHS patient registration database. The updated bridged intercensal population estimates significantly overestimate AI/AN populations of Hispanic origin. All analyses are limited to non-Hispanic AI/AN populations. Non-Hispanic white was chosen as the reference population. The term “non-Hispanic” is omitted when discussing both groups
Includes histology 8140
Includes histology 8160
Includes histology 8180
Includes histologies 8170–9175
Includes histologies 8141–8159, 8161–8169, 8176–8179, 8181–8389, 8401, 8408, 8410, 8441, 8450, 8460, 8470, 8480–8482, 8490, 8500, 8503, 8504, 8510, 8520, 8525, 8530, 8571–8574, 8576, 8650, 9070
Percentages in the histology distribution are age-adjusted to the 2000 US standard population and may not add to 100% due to rounding
Age at diagnosis
Age-adjusted liver cancer incidence rates by age group are shown in Table 3. Forty-four percent of liver cancer cases in AI/AN occurred at ages < 60 years while only 20.6% occurred among AI/AN adults aged ≥ 75 years. The corresponding percentages among white persons were 34.6 and 30.2%, respectively. Among regions, the Pacific Coast had the highest percentage of AI/AN persons diagnosed with liver cancer at age younger than 60 years (55.6%) compared with 39% of whites. Rates in the overall population were significantly higher among AI/AN persons than among white persons for each age group. The rate ratio (RR) ranged from 1.7 (95% CI 1.3–2.1) among persons aged < 45 years to 2.8 (95% CI 2.4–3.1) among those aged 75+. Incidence rates varied by age group and among regions. AI/AN persons in most age groups in the different regions had significantly higher incidence rates than white persons. Significant differences in RR ranged from 1.5 (95% CI 1.0–2.4) among persons aged 60–74 years in the East to 3.7 (95% CI 2.9–4.7) among persons in the same age group in the Northern Plains.
Table 3.
Invasive liver and intrahepatic bile duct cancer incidence rates and distribution by age (%) and Indian Health Service Region for American Indians/Alaska Natives and Whitesa, CHSDA Counties, United States, 1999–2009
| Years | AI/AN |
White |
AI/AN:White RR (95% CI) | |||||
|---|---|---|---|---|---|---|---|---|
| Count | % of casesb | Ratec | Count | % of casesb | Ratec | |||
| Northern Plains | < 45 | 9 | 4.5 | 0.5 | 164 | 4.4 | 0.3 | 1.2 (0.7–2.9) |
| 45–59 | 79 | 39.9 | 19.1 | 929 | 25.2 | 5.1 | 3.7d (2.9–4.7) | |
| 60–74 | 76 | 38.4 | 45.5 | 1,332 | 36.1 | 13.2 | 3.4d (2.7–4.4) | |
| 75+ | 34 | 17.2 | 65.5 | 1,262 | 34.2 | 20.9 | 3.1d (2.2–4.4) | |
| Alaska | < 45 | 7 | 9.3 | 0.9 | 23 | 9.5 | 0.7 | 1.2 (0.4–3.0) |
| 45–59 | 29 | 38.7 | 15.5 | 115 | 47.7 | 9.5 | 1.6d (1.1–2.5) | |
| 60–74 | 24 | 32.0 | 29.7 | 69 | 28.6 | 17.4 | 1.7d (1.0–2.8) | |
| 75+ | 15 | 20.0 | 57 | 34 | 14.1 | 25.7 | 2.2d (1.1–4.2) | |
| Southern Plains | < 45 | 12 | 4.4 | 0.6 | 76 | 4.0 | 0.4 | 1.4 (0.7–2.5) |
| 45–59 | 88 | 32.6 | 14.6 | 560 | 29.3 | 8.1 | 1.8d (1.4–2.3) | |
| 60–74 | 104 | 38.5 | 37.6 | 662 | 34.6 | 15.4 | 2.4d (2.0–3.0) | |
| 75+ | 66 | 24.4 | 64.4 | 616 | 32.2 | 26 | 2.5d (1.9–3.2) | |
| Southwest | < 45 | 25 | 7.3 | 0.8 | 172 | 4.3 | 0.4 | 1.8d (1.1–2.7) |
| 45–59 | 110 | 32.1 | 14.1 | 1,208 | 30.2 | 7.7 | 1.8d (1.5–2.2) | |
| 60–74 | 116 | 33.8 | 33.8 | 1,518 | 37.9 | 15.1 | 2.2d (1.8–2.7) | |
| 75+ | 92 | 26.8 | 72.6 | 1,107 | 27.6 | 21.2 | 3.4d (2.7–4.2) | |
| Pacific Coast | < 45 | 16 | 5.0 | 0.8 | 378 | 4.0 | 0.4 | 2.0d (1.1–3.2) |
| 45–59 | 162 | 50.6 | 28.1 | 3,291 | 35.1 | 9.4 | 3.0d (2.6–3.5) | |
| 60–74 | 102 | 31.9 | 44.8 | 3,107 | 33.1 | 16.5 | 2.7d (2.2–3.3) | |
| 75+ | 40 | 12.5 | 55.4 | 2,612 | 27.8 | 23.3 | 2.4d (1.7–3.2) | |
| East | < 45 | e | 4.8 | 0.4 | 363 | 3.9 | 0.4 | 1.1 (0.2–3.1) |
| 45–59 | 24 | 38.1 | 11.7 | 2,627 | 28.1 | 7.9 | 1.5 (0.9–22) | |
| 60–74 | 22 | 34.9 | 26.5 | 3,354 | 35.8 | 17.1 | 1.5d (1.0–2.4) | |
| 75+ | 14 | 22.2 | 48.6 | 3,013 | 32.2 | 24.9 | 2.0d (1.1–3.3) | |
| Total US | < 45 | 72 | 5.7 | 0.7 | 1,176 | 4.1 | 0.4 | 1.7d (1.3–2.1) |
| 45–59 | 492 | 38.8 | 17.8 | 8,730 | 30.5 | 7.9 | 2.3d (2.1–2.5) | |
| 60–74 | 444 | 35.0 | 37.6 | 10,042 | 35.1 | 15.9 | 2.4d (2.1–2.6) | |
| 75+ | 261 | 20.6 | 64.1 | 8,644 | 30.2 | 23.3 | 2.8d (2.4–3.1) | |
Source: Central Cancer Registries in the National Program of Cancer Registries (NCPR) of the Centers for Disease Control and Prevention and the Surveillance, Epidemiology, and End Results program of the National Cancer Institute; see Table 1 for states included CHSDA contract health service delivery areas, IHS Indian Health Service
AI/AN race is reported by NPCR and SEER registries or through linkage with the IHS patient registration database. The updated bridged intercensal population estimates substantially overestimate AI/AN populations of Hispanic origin. All analyses are limited to non-Hispanic AI/AN populations. Non-Hispanic White was chosen as the reference population. The term “non-Hispanic” is omitted when discussing both groups
Percentages may not add to 100% due to rounding
Rates are per 100,000 persons and are age-adjusted to the 2000 U.S standard population (19 age groups, Census P25–1130)
Rate ratio indicates that AI/AN rate is statistically significantly higher than the White rate (p < 0.05)
Counts less than 6 are suppressed
Cancer stage
Age-adjusted liver cancer incidence rates (years 2001–2009) stratified by stage (localized, regional, distant, unstaged) are shown in Table 4. Overall, the rates were approximately two times higher for the AI/AN compared to the white population, regardless of stage at diagnosis. Percent distribution for regional and distant disease varied by region, with distant stage higher in AI/AN population compared to whites in Alaska (30.0 versus 16.2%), and the Southwest (13.8 versus 11.5%). Overall, rate ratios for the AI/AN population were significantly higher than the white population for each of the reported stages. Rates for the AI/AN population compared to the white ranged from 20% to over 300% higher than the white population (Southwest RR = 1.6, 95% CI 2.8–6.5, Northern Plains RR = 4.4, 95% CI 2.8–6.5) for regional disease. For distant disease, rates for the AI/AN population were nearly two to three times higher than rates for the white population (Northern Plains RR = 1.8, 95% CI 0.8–3.2, East RR = 1.8, 95% CI 0.5–4.1, Alaska RR = 2.6, 95% CI 1.1–5.5).
Table 4.
Invasive liver and intrahepatic bile duct cancer incidence rates and distribution by SEER summary stage (%) and Indian Health Service Region for American Indians/ Alaska Natives and Whitesa, CHSDA Counties, 2001–2009
| SEER summary stage | AI/AN |
White |
AI/AN:White RR (95% CI) | ||||
|---|---|---|---|---|---|---|---|
| Count | Rateb | % of casesc | Count | Rateb | % of casesc | ||
| Localized | |||||||
| Northern Plains | 27 | 2.1 | 25 | 578 | 0.7 | 26.9 | 2.8d (1.8–4.3) |
| Alaska | 15 | 2.2 | 37.5 | 61 | 1.5 | 33.0 | 1.5 (0.7–2.7) |
| Southern Plains | 43 | 1.9 | 22.5 | 378 | 1.2 | 30.8 | 1.6d (1.1–2.2) |
| Southwest | 38 | 1.4 | 25 | 542 | 0.7 | 23.9 | 1.9d (1.3–2.6) |
| Pacific Coast | 64 | 2.9 | 37.7 | 1,538 | 1.0 | 33.3 | 2.9d (2.1–3.7) |
| East | 14 | 1.6 | 30.4 | 1,445 | 0.9 | 31.7 | 1.7 (0.9–3.0) |
| Total US | 201 | 2.0 | 28.4 | 4,542 | 0.9 | 30.3 | 2.2d (1.9–2.5) |
| Regional | |||||||
| Northern Plains | 31 | 2.4 | 28.7 | 432 | 0.5 | 20.1 | 4.4d (2.8–6.5) |
| Alaska | 9 | 1.4 | 22.5 | 43 | 1.2 | 23.2 | 1.2 (0.4–2.5) |
| Southern Plains | 31 | 1.4 | 16.2 | 165 | 0.5 | 13.4 | 2.7d (1.8–4.1) |
| Southwest | 30 | 1.1 | 19.7 | 275 | 0.4 | 12.1 | 2.9d (1.9–4.3) |
| Pacific Coast | 48 | 2.2 | 28.2 | 1,165 | 0.8 | 25.2 | 2.9d (2.1–3.9) |
| East | 7 | 1.2 | 15.2 | 841 | 0.6 | 18.4 | 2.1 (0.8–4.4) |
| Total US | 156 | 1.6 | 22.1 | 2,921 | 0.6 | 19.5 | 2.7d (2.3–3.2) |
| Distant | |||||||
| Northern Plains | 12 | 0.9 | 11.1 | 396 | 0.5 | 18.4 | 1.8 (0.8–3.2) |
| Alaska | 12 | 2.2 | 30.0 | 30 | 0.8 | 16.2 | 2.6d (1.1–5.5) |
| Southern Plains | 25 | 1.3 | 13.1 | 173 | 0.5 | 14.1 | 2.3d (1.4–3.6) |
| Southwest | 21 | 0.8 | 13.8 | 260 | 0.4 | 11.5 | 2.3d (1.4–3.6) |
| Pacific Coast | 23 | 1.3 | 13.5 | 801 | 0.4 | 17.4 | 2.3d (1.4–3.7) |
| East | e | 0.9 | 10.9 | 736 | 0.5 | 16.1 | 1.8 (0.5–4.1) |
| Total US | 98 | 1.1 | 13.9 | 2,396 | 0.5 | 16.0 | 2.3d (1.8–2.8) |
| Unstaged | |||||||
| Northern Plains | 38 | 3.0 | 35.2 | 742 | 0.9 | 34.5 | 3.4d (2.3–4.8) |
| Alaska | e | 0.8 | 10.0 | 51 | 1.5 | 27.6 | 0.6 (0.1–0.3) |
| Southern Plains | 92 | 4.6 | 48.2 | 513 | 1.6 | 41.7 | 2.8d (2.2–3.6) |
| Southwest | 63 | 2.5 | 41.5 | 1,190 | 1.6 | 52.5 | 1.6d (1.2–2.0) |
| Pacific Coast | 35 | 2.0 | 20.6 | 1,111 | 0.7 | 24.1 | 2.7d (1.8–3.9) |
| East | 20 | 3.3 | 43.5 | 1,543 | 1.0 | 33.8 | 3.4d (2.0–5.4) |
| Total US | 252 | 2.9 | 35.6 | 5,150 | 1.0 | 34.3 | 2.9d (2.5–3.3) |
Source: Central Cancer registries in the National Program of Cancer Registries of the Centers for Disease Control and Prevention and the Surveillance, Epidemiology, and End Results Program of the National Cancer Institute; see Table 1 for states included
CHSDA contract health service delivery areas, IHS Indian Health Service
AI/AN race is reported by NPCR and SEER registries or through linkage with the IHS patient registration database. The updated bridged intercensal population estimates significantly overestimate AI/AN populations of Hispanic origin. All analyses are limited to non-Hispanic AI/AN populations. Non-Hispanic White was chosen as the reference population. The term “non-Hispanic” is omitted when discussing both groups
Rates are per 100,000 persons and are age-adjusted to the 2000 U.S standard population (19 age groups, Census P25–1130)
Percentages may not add to 100% due to rounding
Rate ratio indicates that AI/AN rate is statistically significantly higher than the White rate (p < 0.05)
Counts less than 6 are suppressed
Trends
Trends in liver cancer incidence rates and APC for the years 1999–2009 are shown in Supplementary Fig. 2a, b, and Table 5. Among persons less than 60 years of age, AI/AN males had the highest incidence across all years (Supplementary Fig. 2b). Overall liver cancer incidence rates increased by 5% for AI/AN persons and 3.6% for whites (Table 5).Rates of liver cancer among AI/AN males increased by 4.8 and 4% in white males. For AI/AN females, overall rates of liver cancer increased by 5.2 and 2.2% in white females. However, AI/AN liver cancer trends varied by region. Overall, the Southern Plains and Pacific Coast saw significant increases in liver cancer rates (8.1 and 6.6%, respectively) and for men (7.8 and 7.5%, respectively). For AI/AN women, significant annual percent increases in liver cancer incidence rates were observed in the Southern Plains only (8.7%). No significant differences in trends were observed comparing the AI/AN population to the white population overall or by region.
Table 5.
Invasive liver and intrahepatic bile duct cancer incidence annual percent change (APC) by year and Indian Health Service Region for American Indians/Alaska Natives and Non-Hispanic Whitesa, CHSDA Counties, United States, 1999–2009
| IHS region | Both sexes |
Males |
Females |
|||
|---|---|---|---|---|---|---|
| AI/AN APC | White APC | AI/AN APC | White APC | AI/AN APC | White APC | |
| Northern Plains | 4.8 (−0.8 to 10.7) | 1.4 (−0.1 to 14.5) | 5.4 (−3.5 to 15.2) | 1.5 (0.0–3.1) | 5.7 (−6.6 to 19.5) | 0.6 (−1.9 to 3.1) |
| Alaska | −1.2 (−11.9 to 10.9) | −0.5 (−4.8 to 4.0) | −3.0 (−17.2 to 13.5) | −1.8 (−7.9 to 4.7) | c | c |
| Southern Plains | 8.1b (3.8–12.6) | 4.8b (2.5–7.3) | 7.8b (2.2–13.7) | 5.0b (2.4–7.7) | 8.7b (3.0–14.8) | 4.0 (−0.2 to 8.3) |
| Southwest | 2.1 (−1.0 to 5.3) | 2.9b (1.4–4.4) | 0.8 (−2.6 to 4.4) | 3.1b (1.7–4.5) | 2.3 (−5.2 to 10.5) | 1.8 (−0.9 to 4.5) |
| Pacific Coast | 6.6b (3.0–10.4) | 4.6b (3.9–5.2) | 7.5b (3.3–12.0) | 5.1b (4.4–5.9) | 4.1 (−4.5 to 13.6) | 2.8b (2.4–8.2) |
| East | c | 3.7b (2.7–4.7) | c | 4b (3.0–5.0) | c | 2.2 (0.0–4.4) |
| Total US | 5.0b (2.7–7.3) | 3.6b (3.0–4.3) | 4.8b (2.0–7.8) | 4b (3.3–4.6) | 5.2b (0.8–9.9) | 2.2b (0.9–3.6) |
Source: Cancer registries in the National Program of Cancer Registries of the Centers for Disease Control and Prevention and the Surveillance, Epidemiology, and End Results Program of the National Cancer Institute. Rates are per 100,000 persons and are age-adjusted to the 2000 US standard population (19 age groups Census P25–1130)
CHSDA contract health service delivery areas, IHS Indian Health Service, AI/AN American Indians/Alaska Natives, APC annual percent change
AI/AN race is reported by NPCR and SEER registries or through linkage with the IHS patient registration database. The updated bridged intercensal population estimates significantly overestimate AI/AN populations of Hispanic origin. All analyses are limited to non-Hispanic AI/AN populations. Non-Hispanic White was chosen as the reference population. The term “non-Hispanic” is omitted when discussing both groups
APC is significantly different from zero (p < 0.05)
APC cannot be calculated
Correlations between liver cancer incidence rates and prevalence of risk factors in the AI/AN population
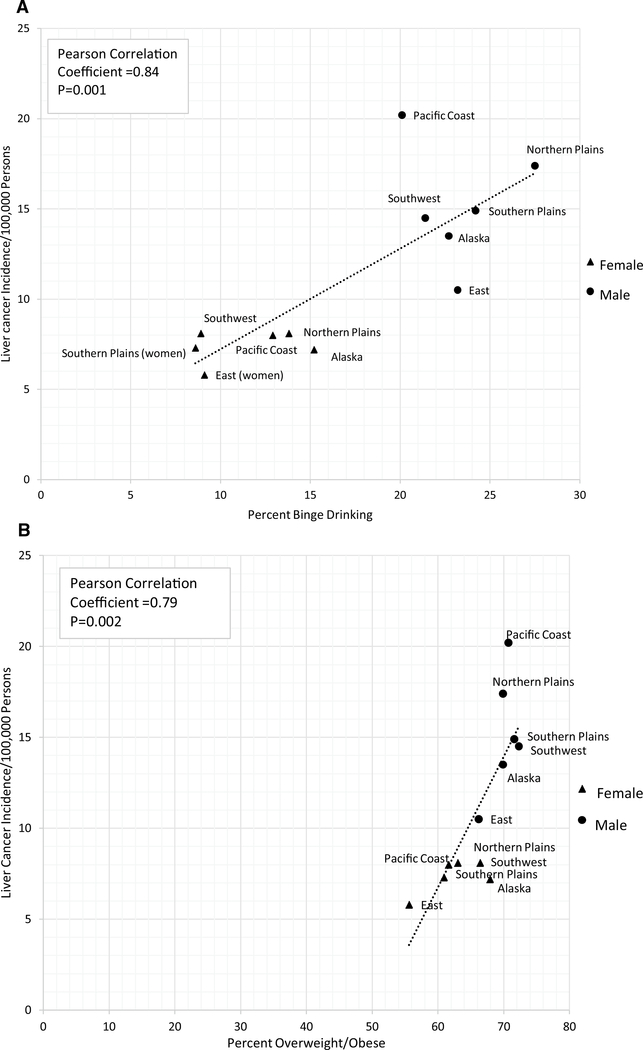
Prevalence of risk factors including smoking, diabetes, obesity, binge drinking, and hypertension are shown in Supplementary Table 1. The prevalence of these risk factors varied widely for AI/AN populations across IHS regions. The overall prevalence of obesity ranged from nearly 56% (AI/AN women in the East) to over 72% (AI/AN men in the Southwest). The overall prevalence of binge drinking was over 10% in AI/AN females and 23% in AI/AN males, with the highest prevalence found in AI/AN men in the Northern Plains (27.5%) and AI/AN women in Alaska (15.2%). We found a strong correlation between liver cancer incidence rates and prevalence of binge drinking (r = 0.84, p = 0.001) and overweight/obesity (r = 0.79, p = 0.002) (Fig. 1a, b) by region. We did not observe any statistically significant correlations between the liver cancer incidence rates and the remaining risk factors (Supplementary Table 1).
Fig. 1.
a Correlation between prevalence of binge drinking (1999–2009, excluding 2000) and liver cancer incidence rates (1999–2009) by IHS regions and sex (AI/AN population). b Correlation between prevalence of overweight/obesity and liver cancer incidence rates (1999–2009) by IHS region and sex (AI/AN population). Note: AI/AN race is reported by NPCR and SEER registries or through linkage with the IHS patient registration database. The updated bridged intercensal population estimates significantly overestimate AI/ AN populations of Hispanic origin. All analyses are limited to non-Hispanic AI/AN populations. Non-Hispanic White was chosen as the reference population. The term “non-Hispanic” is omitted when discussing both groups
Discussion
This study provides a comprehensive examination of liver cancer incidence and regional variation of these rates in AI/ AN populations. The inclusion of data from US central cancer registries strengthens the stability of regional data and linkage to the IHS patient registration database improves on racial classification. These data highlight the disproportionate burden of liver cancer incidence in AI/AN populations compared to white populations. The highest liver cancer incidence rates were found in AI/AN males, and the lowest in white females. This study confirmed variation in liver cancer incidence rates by IHS region, with the lowest rates in the East and the highest in the Northern Plains. We also observed regional variation for the AI/AN population overall and by age of diagnosis, histology, and stage of disease.
In a recent review of case series published in the United States, 65% of all HCC cases were seropositive for either hepatitis B surface antigen (HBsAg) or HCV antibodies, with 50% of HCC cases seropositive for HCV antibodies alone [22]. Drug regimens for HCV now have extremely high rates of sustained virologic clearance, so treatment may represent the greatest opportunity for liver cancer prevention. Compared to the national average, AI/AN persons have nearly a twofold higher rate of acute HCV incidence and HCV associated mortality. IHS has implemented facility-level interventions including electronic health record clinical decision support tools and clinical trainings to improve HCV screening coverage among IHS participants born between 1945 and 1965 [23], but access to treatment continues to be a challenge [24].
In 1984, an HBV control program was implemented through the efforts of the Liver Disease and Hepatitis Program of the Alaska Native Tribal Health Consortium and the IHS. At that time, HBV was the exclusive viral infection associated with primary liver cancer [25, 26] in the Alaskan Native (AN) population. This program included universal infant immunization, population-wide screening, vaccination for HBsAg seronegative individuals, an active surveillance program, and administration of antiviral therapy where warranted [26]. No cases of HBV have occurred in AN children since 1992 and no cases of HCC have occurred in AN children since 1999 [25]. Despite the successful HBV control program, however, many remain at risk for HBV or HCV, and hepatitis-associated liver cancers have increased significantly, as have liver cancers due to other, undetermined causes [25]. Therefore, despite these successful prevention efforts, more targeted strategies are needed to reduce the prevalence of HCV and other liver cancer-associated risk factors in the AI/AN population.
Another factor that may contribute to the observed racial and regional differences in liver cancer are differences in obesity prevalence. The present study suggests a correlation between prevalence of obesity in the AI/AN population and liver cancer incidence rates. These correlations are descriptive and ecologic in nature, and therefore cannot be used to determine causality. However, they provide important descriptive information that can be used to generate hypotheses for future research. A recent study estimated that the population-attributable fraction for liver cancer of obesity and diabetes was nearly 37%, and that the elimination of these two risk factors would reduce the incidence of liver cancer more than any other risk factor [27]. The high observed prevalence of obesity and diabetes in AI/AN populations suggest that this is an important area of focus for cancer prevention programs. Efforts to address these issues, such as the National Center for Chronic Disease Prevention and Health Promotion’s “Good Health and Wellness in Indian Country” program is one such effort aimed improving nutrition and physical activity, which will have a broad impact on chronic disease prevention including cancer [28].
AI/AN men and women have higher prevalence of obesity than their white counterparts (33.9 versus 23.3% for men and 35.5 versus 21.0% for women, respectively) [29]. The prevalence of diagnosed diabetes is more than double for AI/ AN individuals overall, compared to whites [29]. A recent study utilizing data from 14 U.S. based prospective studies reported significant associations between BMI and waist circumference and liver cancer, and diabetes was associated with over a 2.5-fold risk of liver cancer [30]. The increased liver cancer risk associated with excess body weight may be caused by NAFLD, which represents a spectrum of changes in the liver tissue, ranging from accumulation of fat in the liver, to non-alcoholic steatohepatitis NASH and cirrhosis [30, 31]. Type 2 diabetes may be associated with liver cancer risk via obesity and NAFLD [32] and has been shown to increase risk of HCV infections [33].
We assessed the prevalence of various liver cancer risk factors through the BRFSS and found that many liver cancer-associated risk factors, including obesity, are higher in the AI/AN population compared to the white population. This suggests that the disproportionate liver cancer burden in AI/ AN communities is unlikely to abate in the coming years. Alcohol and tobacco use are important contributing factors for cirrhosis, chronic liver disease CLD, and liver cancer and there is an elevated risk of liver cancer in heavy drinkers also infected with viral hepatitis [2, 34, 35]. Although there are no substantial differences in estimates of heavy alcohol use or binge drinking between AI/AN and white populations [29], alcohol-related morbidity and mortality remains a significant health concern for many tribal groups [2, 36] and the observed regional variation in alcohol consumption may play an important role in variation in liver cancer incidence rates. Smoking prevalence estimates in the AI/AN population are nearly double those in whites [29]. Smoking has also been shown to have an independent association with liver cancer and may potentially interact with other risk factors including alcohol use and obesity [7].
This study found that AI/AN adults were diagnosed with liver cancer at later stages compared to whites. Many AI/ AN persons live in remote rural areas far from specialty care; rural and urban AI/AN populations also tend to have lower household incomes than the general US population [29]. We conducted a BRFSS analysis that showed that AI/AN individuals were more likely to not see a physician due to cost (over 18 versus 10% in whites, Supplementary Table 2). Access to care issues are, therefore, both financial and geographic. The highest rates of distant disease for the AI/AN population were observed in Alaska which may be due, in part, to long travel distances and increased barriers to access to care resulting in delayed diagnosis for this population. Efforts to address access to care for HCV in rural and underserved communities through the Extension for Community Healthcare outcomes (ECHO) program have been successful [37]. This program implements teleconsulting and telementoring partnerships between specialists and providers in rural and underserved communities [38]. These programs provide a viable mechanism for improving access to best practices for cancer and risk factor prevention, early detection, and treatment in the AI/AN community.
There are several limitations to this report. We used data from population-based central cancer registries linked with IHS patient registration databases to minimize racial misclas-sification in AI/AN. However, this methodology does not take into account individuals who are not members of federally recognized tribes, not eligible for IHS services, or for those who have not previously accessed IHS health services. Individuals living in urban, non-CHSDA areas are under-represented in these data and therefore these results may not be generalizable to all AI/AN in the United States or in individual IHS regions. Although the exclusion of Hispanic AI/AN persons from the analyses reduced the overall AI/AN incidence rates by less than 5%, this exclusion may disproportionally impact some states and regions. The BRFSS sampling frame may be potentially problematic in the AI/AN communities where several families may not have a landline or cellular phone or the single landline phone may serve several families. BRFSS data are self-reported, and therefore subject to reporting bias. Finally, the correlations presented here between risk factor prevalence and liver cancer rates are descriptive in nature. Due to the long period for liver cancer to develop, these correlations are not temporally plausible and therefore must be interpreted with caution.
In summary, AI/AN populations have higher liver cancer incidence and a higher burden of liver cancer risk factors than whites. Interactions between these factors and HBV/HCV infection may result in disproportionately higher rates of liver cancer among AI/AN men and women. Future research is needed to understand variations in HBV/HCV prevalence and behavioral risk factors that could be associated with regional differences in liver cancer incidence observed in the AI/AN population. These data underscore the importance of targeted interventions and resource allocation to prevent liver cancer and associated conditions including CLD, cirrhosis, and HBV/ HCV infections. Strategies for prevention in AI/AN communities could include improving vaccination coverage for HBV, screening for HCV [39], use of new and effective HCV therapies, the promotion of culturally appropriate programs to address obesity and type 2 diabetes, excessive alcohol use, and tobacco cessation efforts. Integrated and comprehensive approaches hold promise for reducing liver cancer disparities among AI/AN persons in the United States.
Supplementary Material
Acknowledgments
This study was supported by the Centers for Disease Control and Prevention (CDC). We thank Brian McMahon, Brenna Simons-Petrusa, and Sarah H. Nash from the Alaska Native Tribal Health Consortium for their input in this article.
Footnotes
Disclaimer The findings and conclusions in this report are those of the authors and do not necessarily reflect the official position of the Centers for Disease Control and Prevention.
Compliance with ethical standards
Conflict of interest No conflicts of interest to disclose.
Ethical approval The study did not involve human participants, institutional review board approval was not necessary.
Electronic supplementary material The online version of this article (https://doi.org/10.1007/s10552-018-1059-3) contains supplementary material, which is available to authorized users.
References
- 1.Ryerson AB, Eheman CR, Altekruse SF et al. (2016) Annual Report to the Nation on the Status of Cancer, 1975–2012, featuring the increasing incidence of liver cancer. Cancer 122:1312–1337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jim MA, Perdue DG, Richardson LC et al. (2008) Primary liver cancer incidence among American Indians and Alaska Natives, US, 1999–2004. Cancer 113:1244–1255 [DOI] [PubMed] [Google Scholar]
- 3.White MC, Espey DK, Swan J, Wiggins CL, Eheman C, Kaur JS (2014) Disparities in cancer mortality and incidence among American Indians and Alaska Natives in the United States. Am J Public Health 104(Suppl 3):S377–S387 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Di Bisceglie AM, Lyra AC, Schwartz M et al. (2003) Hepatitis C-related hepatocellular carcinoma in the United States: influence of ethnic status. Am J Gastroenterol 98:2060–2063 [DOI] [PubMed] [Google Scholar]
- 5.Armstrong GL, Wasley A, Simard EP, McQuillan GM, Kuhnert WL, Alter MJ (2006) The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med 144:705–714 [DOI] [PubMed] [Google Scholar]
- 6.Perz JF, Armstrong GL, Farrington LA, Hutin YJ, Bell BP (2006) The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J Hepatol 45:529–538 [DOI] [PubMed] [Google Scholar]
- 7.Marrero JA, Fontana RJ, Fu S, Conjeevaram HS, Su GL, Lok AS (2005) Alcohol, tobacco and obesity are synergistic risk factors for hepatocellular carcinoma. J Hepatol 42:218–224 [DOI] [PubMed] [Google Scholar]
- 8.Espey DK, Wiggins CL, Jim MA, Miller BA, Johnson CJ, Becker TM (2008) Methods for improving cancer surveillance data in American Indian and Alaska Native populations. Cancer 113:1120–1130 [DOI] [PubMed] [Google Scholar]
- 9.Hankey BF, Ries LA, Edwards BK (1999) The surveillance, epidemiology, and end results program: a national resource. Cancer Epidemiol Biomarkers Prev 8:1117–1121 [PubMed] [Google Scholar]
- 10.Thoburn KK, German RR, Lewis M, Nichols PJ, Ahmed F, Jack-son-Thompson J (2007) Case completeness and data accuracy in the Centers for Disease Control and Prevention’s National Program of Cancer Registries. Cancer 109:1607–1616 [DOI] [PubMed] [Google Scholar]
- 11.Young JL, Roffers SD, Ries LAG, Fritz AG, Hurlbut AA (eds) (2001) SEER Summary Sating Manual-2000: Codes and Coding Instructions. NIH Publication No. 01–4969 National Cancer Institute, Bethesda [Google Scholar]
- 12.National Center for Chronic Disease Prevention and Health Promotion (2005) Link Plus: A suite of publicly available software programs for collecting and processing cancer registry data Atlanta Ga: Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, U.S. Department of Health and Human Services; http://www.cdc.gov/cancer/npcr/tools/registryplus/lp.htm. Accessed on 18 Oct 2016 [Google Scholar]
- 13.U.S. Department of Health and Human Services (2017) Indian health service. Purchased/Referred Care. Contract Health Service Delivery Areas (CHSDA). https://www.ihs.gov/prc/index.cfm?module=chs_requirements_chsda. Accessed Nov 2017
- 14.Arias E, Schauman WS, Eschbach K, Sorlie PD, Backlund E (2008) The validity of race and Hispanic origin reporting on death certificates in the United States. Vital Health Stat 2:1–23 [PubMed] [Google Scholar]
- 15.Behavioral Risk Factor Surveillance System Survey Data. Atlanta, Georgia: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, 1999–2009. https://www.cdc.gov/brfss/annual_data/
- 16.National Cancer Institute. Surveillance, Epidemiology, and End Results (SEER) Program. Statistical Resources. U.S. population data 1969–2011 http://seer.cancer.gov/popdata/. Accessed on 10 June 2013
- 17.Jim MA, Arias E, Seneca DS et al. (2014) Racial misclassification of American Indians and Alaska Natives by Indian health service contract health service delivery area. Am J Public Health 104(Suppl 3):S295–S302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Surveillance Research Program, National Cancer Institute. SEER*Stat Software, Latest Release: 8.3.2 (2016) http://seer.cancer.gov/seerstat. Accessed Oct 2016
- 19.Kim HJ, Fay MP, Feuer EJ, Midthune DN (2000) Permutation tests for joinpoint regression with applications to cancer rates. Stat Med 19:335–351 [DOI] [PubMed] [Google Scholar]
- 20.National Cancer Institute. Joinpoint regression program, Version 4.3.1.0 (2016) Bethesda, MD: National Cancer Institute. http://surveillance.cancer.gov/joinpoint.. Accessed 15 Nov 2016
- 21.Tiwari RC, Clegg LX, Zou Z (2006) Efficient interval estimation for age-adjusted cancer rates. Stat Methods Med Res 15:547–569 [DOI] [PubMed] [Google Scholar]
- 22.de Martel C, Maucort-Boulch D, Plummer M, Franceschi S (2015) World-wide relative contribution of hepatitis B and C viruses in hepatocellular carcinoma. Hepatology 62:1190–1200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Reilley B, Leston J, Hariri S et al. (2016) Birth cohort testing for hepatitis C virus - Indian health service 2012–2015. MMWR Morb Mortal Wkly Rep 65:467–469 [DOI] [PubMed] [Google Scholar]
- 24.Leston J, Finkbonner J (2016) The need to expand access to hepatitis C virus drugs in the Indian Health Service. JAMA 316:817–818 [DOI] [PubMed] [Google Scholar]
- 25.McMahon BJ, Bulkow LR, Singleton RJ et al. (2011) Elimination of hepatocellular carcinoma and acute hepatitis B in children 25 years after a hepatitis B newborn and catch-up immunization program. Hepatology 54:801–807 [DOI] [PubMed] [Google Scholar]
- 26.Connelly M, Bruce MG, Bulkow L, Snowball M, McMahon BJ (2016) The changing epidemiology and aetiology of hepatocellular carcinoma from 1969 through 2013 in Alaska Native people. Liver Int 36:1829–1835 [DOI] [PubMed] [Google Scholar]
- 27.Welzel TM, Graubard BI, Quraishi S et al. (2013) Population-attributable fractions of risk factors for hepatocellular carcinoma in the United States. Am J Gastroenterol 108:1314–1321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.CDC Chronic Disease Prevention and Health Promotion, Tribal Resources. Atlanta, Georgia: U.S Department of Health and Human Services, Centers for Disease Control and Prevention https ://www.cdc.gov/chronicdisease/tribal/factsheet.htm. Accessed Feb 2017
- 29.Cobb N, Espey D, King J (2014) Health behaviors and risk factors among American Indians and Alaska Natives, 2000–2010. Am J Public Health 104(Suppl 3):S481–S489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Larsson SC, Wolk A (2007) Overweight, obesity and risk of liver cancer: a meta-analysis of cohort studies. Br J Cancer 97:1005–1008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Neuschwander-Tetri BA, Caldwell SH (2003) Nonalcoholic steatohepatitis: summary of an AASLD Single Topic Conference. Hepatology 37:1202–1219 [DOI] [PubMed] [Google Scholar]
- 32.Connolly GC, Safadjou S, Chen R et al. (2012) Diabetes mellitus is associated with the presence of metastatic spread at disease presentation in hepatocellular carcinoma. Cancer Invest 30:698–702 [DOI] [PubMed] [Google Scholar]
- 33.White DL, Ratziu V, El-Serag HB (2008) Hepatitis C infection and risk of diabetes: a systematic review and meta-analysis. J Hepatol 49:831–844 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tanaka K, Hirohata T, Takeshita S et al. (1992) Hepatitis B virus, cigarette smoking and alcohol consumption in the development of hepatocellular carcinoma: a case-control study in Fukuoka, Japan. Int J Cancer 51:509–514 [DOI] [PubMed] [Google Scholar]
- 35.Yuan JM, Govindarajan S, Arakawa K, Yu MC (2004) Synergism of alcohol, diabetes, and viral hepatitis on the risk of hepatocellular carcinoma in blacks and whites in the U.S. Cancer 101:1009–1017 [DOI] [PubMed] [Google Scholar]
- 36.Landen M, Roeber J, Naimi T, Nielsen L, Sewell M (2014) Alcohol-attributable mortality among American Indians and Alaska Natives in the United States, 1999–2009. Am J Public Health 104(Suppl 3):S343–S349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Arora S, Thornton K, Murata G et al. (2011) Outcomes of treatment for hepatitis C virus infection by primary care providers. N Engl J Med 364:2199–2207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Arora S, Kalishman S, Thornton K et al. (2016) Project ECHO (project extension for community healthcare outcomes): a national and global model for continuing professional development. J Contin Educ Health Prof 36(Suppl 1):S48–S49 [DOI] [PubMed] [Google Scholar]
- 39.Recommendations for prevention and control of hepatitis C virus (HCV) infection and HCV-related chronic disease (1998) Centers for disease control and prevention. MMWR Recomm Rep 47:1–39 [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.