Abstract
Background:
Bacterial resistance to antibiotics limits treatment options, increases morbidity and mortality, and raises the risk of antibiotic-associated adverse events. Antibacterial resistance emerges rapidly following an increase in the consumption of antibiotics against infectious diseases. The spread of ESBL producing strains has a limiting factor based on antibiotic function for the treatment of infections particularly caused by Acinetobacter baumannii (A. baumannii).
Objective:
This study was conducted to evaluate the prevalence of antimicrobial resistance and distribution of blaTEM, blaCTX, and blaSHV genes among A. baumannii strains isolated from clinical samples at a major hospital in Teheran, Iran.
Methods:
A. baumannii strains were isolated and identified using standard microbiological methods. The disc diffusion and combined discs methods were used for testing antimicrobial susceptibility and to identify the strains producing Extended-Spectrum Beta-Lactamases (ESBL), respectively. DNA extraction was done by boiling method. Finally, the frequency of resistant genes including blaTEM, blaCTX, and blaSHV in ESBL producing isolates was studied by PCR.
Results:
Gender distribution in this study was 53 (53%) samples for men and 47 (47%) for women. Totally, one hundred A. baumannii strains were isolated. More than 93% of the isolates were multi drug resistant. The highest to lowest antibiotic resistance was observed against amoxicillin/clavulanic acid (98%), ceftriaxone (96%), cefotaxime (94%), and ceftazidime (93%), respectively. The frequency of positive phenotypic test of ESBL was 19% and 16% for CAZ-C and CTX-C, respectively. The frequency of blaTEM, blaCTX, and blaSHV genes was 52.1, 43.4, and 21.7, respectively.
Conclusion:
A. baumannii isolates exhibited an extremely worrying level of antibiotic resistance, and a high percentage of the isolates showed MDR in this study. This is a serious warning because ESBLs are a major threat to the effectiveness of antibiotics that are currently available for medical uses. The frequency of genes encoded ESBL isolates of A. baumannii may be due to overuse and misuse of antibiotics.
Keywords: Antibiotic, Bacterial resistance, A. baumannii, ESBL, PCR, Clinical samples
1. INTRODUCTION
Bacterial resistance to antibiotics limits treatment options increases mortality and the risk of side effects of antibiotics. Resistance to excessive antibiotic use is increasing rapidly [1]. Extended-spectrum beta-lactamases (ESBL) is responsible for creating resistant strains of bacteria against antibiotics [2-4]. Expansion of ESBL producing strains restricted the antimicrobial agents to treat patients effectively and raised concerns for control of infections caused by A. baumannii. The misuse of antimicrobial drugs in hospitals can lead to the development of antibiotic-resistant bacteria such as A. baumannii. Acinetobacter's hospital strains are usually resistant to several drugs at the same time. A. baumannii is more common than other Acinetobacter strains and is the most common cause of bacterial infection. This opportunistic bacteria can survive on a variety of surfaces, including patient skin and medical equipment. Therefore, prolonged hospitalization in the intensive care unit, severe illnesses, immunodeficiency conditions, burn injuries, long-term use of antimicrobial agents, and catheter use are some of the most important risk factors for Acinetobacter infection [5]. A. baumannii is an important opportunistic pathogen responsible for a variety of nosocomial infections; including ventilator-associated pneumonia, bacteremia, surgical site infections, secondary infections of burn patients, secondary meningitis, and urinary tract infections in the Middle East region [6, 7] and other parts of the world [8, 9]. Hydrolysis of the β-lactam ring structure by ESBL causes an abnormal antibiotic function. ESBL enzymes are able to target a wide range of antibiotics, such as penicillins and their complexes, monobactams, and new generation cephalosporins [10]. There are hundred types of β-lactamase enzymes, each of the ESBL bacteria may have genes for one or more of these enzymes. Similar bla genes, similar to the same structure within these gene families, are grouped according to phylogenetic and are often detected and diagnosed by PCR [11]. Transmission of resistance genes between bacteria is easy to do since these genes are encoded on mobile vectors (plasmids and transposons) [12, 13]. β-lactamases are inactivating enzymes that are present in all types of bacteria. The efficacy of beta-lactam antibiotics in eradicating infection in different places in the body can be reduced by the beta-lactamase producing organisms [14]. By using DNA-based technologies, a new era in the diagnostic and molecular epidemiology of an antibiotic-resistant bacterium opens. The use of PCR-based techniques has revolutionized the rapid diagnosis of determinants of resistance, such as ESBLs [15, 16].
We have previously investigated ESBLs genes in some high resistant bacterial species particularly those were isolated from clinical cases [17-22].
In this study, we aimed to determine the prevalence of antimicrobial resistance and distribution of blaTEM, blaCTX, and blaSHV genes among A. baumannii strains isolated from clinical samples at a major hospital in Teheran, Iran.
2. MATERIALS AND METHODS
In a cross-sectional descriptive study, from January until the end of June 2017, clinical samples (endotracheal tubes, blood, sputum, urine, catheters, wounds, and various fluids) were recovered from patients admitted to a major Hospital in Tehran.
2.1. Bacterial Strains Identification
The samples were transported to the laboratory then were cultured on blood and MacConkey agar for 24 hours at 37°C was done. To identify the isolates of A. baumannii, the preliminary conventional phenotypic tests including growth on MacConkey agar, sugar fermentation, motility, catalase and oxidase tests, and other standard recommended tests [23, 24]. For definitive identification of these isolates to the species level, molecular methods were used in the next step.
2.2. Antibiotics Susceptibility Testing
According to the instructions of the clinical and laboratory standards institute, antibiotic resistance of the isolates was determined using the disk diffusion antibiotic sensitivity test [25]. Antibiotic discs used in this study were including cefotaxime, ceftazidime, amoxicillin/clavulanic acid and ceftriaxone. A. baumannii suspension was expanded after incubation at 37°C on Mueller-Hinton agar culture medium and compared to McFarland standard using carpet culture. Then the discs were placed on them. After incubation for 24 hours, the growth inhibition diameter was measured and compared with reference values.
2.3. Detection of ESBL Producing Strains
The phenotypic detection of the different beta-lactamases was performed on Mueller-Hinton medium using discs containing ceftazidime, ceftazidime/clavulanic acid (CAZ-C), cefotaxime, and cefotaxime/clavulanic acid (CTX-C) as previously described [26].
The DNA was extracted from isolates by the boiling method and frequency of TEM, SHV and CTX using specific primers was evaluated, as shown in Table 1 [26, 27]. PCR test was performed using Master Mix, primer, bacterial DNA, and distilled water to obtain a final volume of 25 mL. Except for annealing temperature, the reaction temperature of the PCR for all three beta-lactamase genes was the same. It contained an initial denaturating for 4 minutes at 94°C, followed by 35 repetitions for 1 minute at 94°C, amplification at 72°C for 40 seconds, and final amplification at 72°C for 5 minutes (Table 1). PCR products were electrophoresed on 1.5% agarose gel containing ethidium bromide at 80 V for 1 h.
Table 1. Primers used to amplify the genes of beta-lactamase.
| Target type of β-lactamase Genes | Primer Sequence | Size of Product (bp) |
AT (ºC) |
|---|---|---|---|
| blaTEM | 5´-ATG AGT ATT CAA CAT TTC CG-3´ (F) | 867 | 52.2 |
| 5´-CTG ACA GTT ACC AAT GCT TA-3´ (R) | |||
| blaCTX | 5´-TTT GCG ATG TGC AGT ACC AGT AA- 3´ (F) | 560 | 60 |
| 5´-CTC CGC TGC CGG TTT TAT -3´ (R) | |||
| blaSHV | 5´-TTA ACT CCC TGT TAG CCA-3´ (F) | 860 | 51.2 |
| 5´-GAT TTG CTG ATT TCG CCC-3´ (R) |
F: Forward; R: Reverse; AT: Annealing temperature.
3. RESULTS
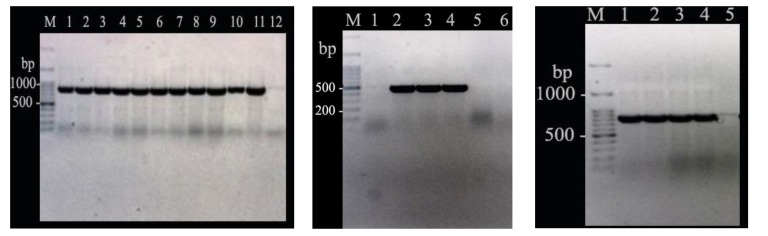
Totally, one hundred A. baumannii strains were isolated. Gender distribution in this study was 53 (53%) samples from men and 47 (47%) of women. The percentage of antibiotic resistance was: amoxicillin/ clavulanic acid (98%), ceftriaxone (96%), cefotaxime (94%), and ceftazidime (93%). Of these, 93% of the clinical samples showed multiple antibiotic resistance. The frequency of positive phenotypic test of ESBL was 19% and 16% for CAZ-C and CTX-C, respectively. The frequency of resistance genes was 52.1, 43.4, and 21.7 in A. baumannii isolates based on the detection of ESBLs genes including blaTEM, blaCTX, and blaSHV, respectively (Fig. 1).
Fig. (1).
PCR amplification products of blaTEM, blaCTX, and blaSHV (In order from left to right) in some representative Acinetobacter baumannii strains.
4. DISCUSSION
In this study, antimicrobial susceptibility testing of A. baumannii isolates originally showed highly significant resistance to different types of antibiotics. This resistance can be due to the presence of specific genes of ESBL such as blaTEM, blaCTX, and blaSHV. Knowing the types and frequency of these genes helps us to make a good decision for the treatment process of patients effectively. High level of multi-drug resistance including Cefotaxime (CTX), Ceftazidime (CAZ), Amoxicillin/clavulanic acid (AMC) and Ceftriaxone (CRO) was observed among the isolates under the study.
Various factors, including the abuse of antibiotics, the spread of clonal resistant microorganisms, can cause the release of highly resistant pathogens to the environment and clinical setting. Previous researches showed that the prevalence of A. baumannii MDR isolates ranged from 32.7% to 100% [28-32].
Among the mechanisms that create resistance to drugs, ESBLs play an important role in resistance to conventional antibiotics such as penicillin and cephalosporins. ESBL genes, due to the widespread diffusion of pathogens from the hospital through plasmids and integrons, can further increase drug resistance, including MDR isolates [34, 35].
Jaggi et al. reported that the prevalence of some cephalosporins antibiotics group resistance in the A. baumannii strains of clinical samples against cefepime, ceftazidime, and ciprofloxacin was 90.3%, 92.1%, 91.6%, respectively [36]. Similar findings have been reported from different countries [37-40].
Several studies have reported the frequency of ESBL gene in A. baumannii ranging from 25% to 93.5% [41-44]. In the present study, of the 100 isolates of A. baumannii, 39% of the ESBL producer was consistent with the results of previous studies. Safari et al. reported that SHV (58%) and TEM (20%) were the highest numbers of ESBL genes in their study [34]. Azhar et al. based on a study that conducted in Iraq, reported that SHV (25%) was the most frequently detected ESBL gene [45], while in this study, TEM (52.1) was the most. Khalilzadegan and colleagues identified that CTX-M and TEM have most ESBL genes [31]. There is some reason to observe the differences in resistance patterns and the prevalence of A. baumannii in various investigations including abuse and misuse of antibiotics, differences in the type of antibiotics used, long-term hospitalization, type of samples taken, differences in diagnostic methods used to identify genes, geographical conditions, gender and age of patients, and so on [46, 47].
This study showed high levels of antibiotic resistance in A. baumannii isolates recovered from clinical samples. This is a serious warning because ESBLs are a major threat to the effectiveness of antibiotics that are currently available for medical uses. In this paper, we presented evidence of a great frequency of multi-drug resistant bacteria in clinical samples. Resistance to antimicrobials increasingly undermines the effectiveness of the use of b-lactam antibiotics and other antibacterial drugs that have been effective in the past. To combat antibiotic resistance and develop knowledge-based interventions, a comprehensive understanding of the mechanisms of resistance and the regulation of genes that cause resistance to the material is needed. Molecular methods particularly DNA-based detection techniques help us in the field of diagnostic and molecular epidemiology of antibiotic resistance genes [48-50].
CONCLUSION
A. baumannii isolates exhibited an extremely worrying level of antibiotic resistance, and a high percentage of the isolates showed MDR in this study. This is a serious warning because ESBLs are a major threat to the effectiveness of antibiotics that are currently available for medical uses. The frequency of genes encoded ESBL isolates of A. baumannii may be due to overuse and misuse of antibiotics.
ACKNOWLEDGEMENTS
We would like to thank from the “Clinical Research Development Center of Baqiyatallah hospital” for their kindly cooperation. This study was supported in part by a grant from “Clinical Research Development Center of Baqiyatallah hospital”.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are the basis of this research.
CONSENT FOR PUBLICATION
Not applicable.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
REFERENCES
- 1.Zhang Y., Marrs C.F., Simon C., Xi C. Wastewater treatment contributes to selective increase of antibiotic resistance among Acinetobacter spp. Sci. Total Environ. 2009;407(12):3702–3706. doi: 10.1016/j.scitotenv.2009.02.013. [DOI] [PubMed] [Google Scholar]
- 2.Cantón R., Novais A., Valverde A., Machado E., Peixe L., Baquero F., Coque T.M. Prevalence and spread of extended-spectrum β-lactamase-producing Enterobacteriaceae in Europe. Clin. Microbiol. Infect. 2008;14(Suppl. 1):144–153. doi: 10.1111/j.1469-0691.2007.01850.x. [DOI] [PubMed] [Google Scholar]
- 3.Hawkey P.M. Prevalence and clonality of extended-spectrum β-lactamases in Asia. Clin. Microbiol. Infect. 2008;14(Suppl. 1):159–165. doi: 10.1111/j.1469-0691.2007.01855.x. [DOI] [PubMed] [Google Scholar]
- 4.Picozzi S.C., Casellato S., Rossini M., Paola G., Tejada M., Costa E., Carmignani L. Extended-spectrum beta-lactamase-positive Escherichia coli causing complicated upper urinary tract infection: Urologist should act in time. Urol. Ann. 2014;6(2):107–112. doi: 10.4103/0974-7796.130536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ellis D., Cohen B., Liu J., Larson E. Risk factors for hospital-acquired antimicrobial-resistant infection caused by Acinetobacter baumannii. Antimicrob. Resist. Infect. Control. 2015;4:40. doi: 10.1186/s13756-015-0083-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ranjbar R., Sadeghifard N., Ahmadi A., Izadi M., Zaeimi-Yazdi J., et al. Antimicrobial Susceptibility and AP-PCR Typing of Acinetobacter Spp. Strains. Iran. J. Public Health. 2007;36(4):50–56. [Google Scholar]
- 7.Sadeghifarda N., Ranjbarb R., Zaeimic J., Alikhanid M.Y., Ghafouryana S., Raftarie M., et al. Antimicrobial susceptibility, plasmid profiles, and RAPD-PCR typing of Acinetobacter bacteria. Asian Biomed. 2010;4(6):901–911. doi: 10.2478/abm-2010-0118. [DOI] [Google Scholar]
- 8.Bergogne-Bérézin E., Towner K.J. Acinetobacter spp. as nosocomial pathogens: Microbiological, clinical, and epidemiological features. Clin. Microbiol. Rev. 1996;9(2):148–165. doi: 10.1128/cmr.9.2.148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Khoshnood S, Eslami G, Hashemi A, Bahramian A, Heidary M, Yousefi N, et al. Distribution of aminoglycoside resistance genes among A. baumannii strains isolated from burn patients in Tehran, Iran. 2017;5(3) [Google Scholar]
- 10.Livermore D.M. beta-Lactamases in laboratory and clinical resistance. Clin. Microbiol. Rev. 1995;8(4):557–584. doi: 10.1128/cmr.8.4.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Naas T., Zerbib M., Girlich D., Nordmann P. Integration of a transposon Tn1-encoded inhibitor-resistant β-lactamase gene, bla(TEM-67) from Proteus mirabilis, into the Escherichia coli chromosome. Antimicrob. Agents Chemother. 2003;47(1):19–26. doi: 10.1128/AAC.47.1.19-26.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Schink A.K., Kadlec K., Schwarz S. Analysis of bla(CTX-M)-carrying plasmids from Escherichia coli isolates collected in the BfT-GermVet study. Appl. Environ. Microbiol. 2011;77(20):7142–7146. doi: 10.1128/AEM.00559-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bailey J.K., Pinyon J.L., Anantham S., Hall R.M. Distribution of the blaTEM gene and blaTEM-containing transposons in commensal Escherichia coli. J. Antimicrob. Chemother. 2011;66(4):745–751. doi: 10.1093/jac/dkq529. [DOI] [PubMed] [Google Scholar]
- 14.Ranjbar R., Masoudimanesh M., Dehkordi F.S., Jonaidi-Jafari N., Rahimi E. Shiga (Vero)-toxin producing Escherichia coli isolated from the hospital foods; virulence factors, o-serogroups and antimicrobial resistance properties. Antimicrob. Resist. Infect. Control. 2017;6(1):4. doi: 10.1186/s13756-016-0163-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Behzadi P., Najafi A., Behzadi E., Ranjbar R. Microarray long oligo probe designing for Escherichia coli: An in-silico DNA marker extraction. Cent. European J. Urol. 2016;69(1):105–111. doi: 10.5173/ceju.2016.654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Iversen A., Kühn I., Franklin A., Möllby R. High prevalence of vancomycin-resistant enterococci in Swedish sewage. Appl. Environ. Microbiol. 2002;68(6):2838–2842. doi: 10.1128/AEM.68.6.2838-2842.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ranjbar R., Giammanco G.M., Aleo A., Plano M.R., Naghoni A., Owlia P., Mammina C. Characterization of the first extended-spectrum beta-lactamase-producing nontyphoidal Salmonella strains isolated in Tehran, Iran. Foodborne Pathog. Dis. 2010;7(1):91–95. doi: 10.1089/fpd.2009.0382. [DOI] [PubMed] [Google Scholar]
- 18.Ranjbar R., Ghazi F.M., Farshad S., Giammanco G.M., Aleo A., Owlia P., Jonaidi N., Sadeghifard N., Mammina C. The occurrence of extended-spectrum β-lactamase producing Shigella spp. in Tehran, Iran. Iran. J. Microbiol. 2013;5(2):108–112. [PMC free article] [PubMed] [Google Scholar]
- 19.Ghafourian S., Bin Sekawi Z., Sadeghifard N., Mohebi R., Kumari Neela V., Maleki A., Hematian A., Rhabar M., Raftari M., Ranjbar R. The Prevalence of ESBLs Producing Klebsiella pneumoniae Isolates in Some Major Hospitals, Iran. Open Microbiol. J. 2011;5:91–95. doi: 10.2174/1874285801105010091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ranjbar R., Sami M. Genetic investigation of Beta-Lactam associated antibiotic resistance among Escherichia coli strains isolated from water sources. Open Microbiol. J. 2017;11:203–210. doi: 10.2174/1874285801711010203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Abdi S., Ranjbar R., Vala M.H., Jonaidi N., Bejestany O.B., Bejestany F.B. Frequency of bla TEM, bla SHV, bla CTX-M, and qnrA among Escherichia coli isolated from urinary tract infection. Arch. Clin. Infect. Dis. 2014;9(1):e18690. doi: 10.5812/archcid.18690. [DOI] [Google Scholar]
- 22.Ranjbar R., Ahmadnezhad B., Jonaidi N. The prevalance of beta lactamase producing Escherichia coli strains isolated from the urine samples in Valiasr Hospital. Biomed. Pharmacol. J. 2014;7(2):425–431. doi: 10.13005/bpj/507. [DOI] [Google Scholar]
- 23.Forbes B.A., Sahm D.F., Weissfeld A.S. Bailey & Scott’s diagnostic microbiology. 12th ed. St Louis, MO: Mosby; 2007. [Google Scholar]
- 24.Golanbar G.D., Lam C.K., Chu Y.M., Cueva C., Tan S.W., Silva I., Xu H.H. Phenotypic and molecular characterization of Acinetobacter clinical isolates obtained from inmates of California correctional facilities. J. Clin. Microbiol. 2011;49(6):2121–2131. doi: 10.1128/JCM.02373-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Turton J.F., Kaufmann M.E., Glover J., Coelho J.M., Warner M., Pike R., Pitt T.L. Detection and typing of integrons in epidemic strains of Acinetobacter baumannii found in the United Kingdom. J. Clin. Microbiol. 2005;43(7):3074–3082. doi: 10.1128/JCM.43.7.3074-3082.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hajjar Soudeiha M., Dahdouh E., Daoud Z., Sarkis D.K. Phenotypic and genotypic detection of β-lactamases in Acinetobacter spp. isolates recovered from Lebanese patients over a 1-year period. J. Glob. Antimicrob. Resist. 2018;12:107–112. doi: 10.1016/j.jgar.2017.09.016. [DOI] [PubMed] [Google Scholar]
- 27.Aliakbarzade K., Farajnia S., Karimi Nik A., Zarei F., Tanomand A. Prevalence of aminoglycoside resistance genes in Acinetobacter baumannii isolates. Jundishapur J. Microbiol. 2014;7(10):e11924. doi: 10.5812/jjm.11924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Agodi A., Barchitta M., Quattrocchi A., Maugeri A., Aldisio E., Marchese A.E., Mattaliano A.R., Tsakris A. Antibiotic trends of Klebsiella pneumoniae and Acinetobacter baumannii resistance indicators in an intensive care unit of Southern Italy, 2008-2013. Antimicrob. Resist. Infect. Control. 2015;4:43. doi: 10.1186/s13756-015-0087-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mirnejad R., Vafaei S. Antibiotic resistance patterns and the prevalence of ESBLs among strains of A. baumannii isolated from clinical specimens. J Gen Microbes Immunity. 2013;2013:1–8. doi: 10.5899/2013/jgmi-00002. [DOI] [Google Scholar]
- 30.Kheltabadi RF, Moniri R, Shajari GR, Nazem Shirazi MH, Musavi SGA, Ghasemi A, et al. Antimicrobial susceptibility patterns and the distribution of resistance genes among acinetobacter species isolated from patients in shahid Beheshti hospital, Kashan. Feyz J Kashan Univ Med Sci. 2009;12(4) [Google Scholar]
- 31.Khalilzadegan S., Sade M., Godarzi H., Eslami G., Hallajzade M., Fallah F., Yadegarnia D. Beta-lactamase encoded genes blaTEM and blaCTX among Acinetobacter baumannii species isolated from medical devices of intensive care units in Tehran hospitals. Jundishapur J. Microbiol. 2016;9(5):e14990. doi: 10.5812/jjm.14990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shakibaie M.R., Adeli S., Salehi M.H. Antibiotic resistance patterns and extended-spectrum β-lactamase production among Acinetobacter spp. isolated from an intensive care Unit of a hospital in Kerman, Iran. Antimicrob. Resist. Infect. Control. 2012;1(1):1. doi: 10.1186/2047-2994-1-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Azizi M, Mortazavi SH, Etemadimajed M, Gheini S, Vaziri S, Alvandi A, et al. Prevalence of extended-spectrum β-lactamases and antibiotic resistance patterns in A. baumannii isolated from clinical samples in kermanshah, Iran. 2017;10(12) [Google Scholar]
- 34.Safari M., Mozaffari Nejad A.S., Bahador A., Jafari R., Alikhani M.Y. Prevalence of ESBL and MBL encoding genes in Acinetobacter baumannii strains isolated from patients of intensive care units (ICU). Saudi J. Biol. Sci. 2015;22(4):424–429. doi: 10.1016/j.sjbs.2015.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ribera A., Vila J., Fernández-Cuenca F., Martínez-Martínez L., Pascual A., Beceiro A., Bou G., Cisneros J.M., Pachón J., Rodríguez-Baño J., Spanish Group for Nosocomial Infection (GEIH) Type 1 integrons in epidemiologically unrelated Acinetobacter baumannii isolates collected at Spanish hospitals. Antimicrob. Agents Chemother. 2004;48(1):364–365. doi: 10.1128/AAC.48.1.364-365.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jaggi N., Sissodia P., Sharma L. A baumannii isolates in a tertiary care hospital: Antimicrobial resistance and clinical significance. J Microbiol Infect Dis. 2012;12:57–63. doi: 10.5799/ahinjs.02.2012.02.0043. [DOI] [Google Scholar]
- 37.Văduva D.B., Muntean D., Lonescu G., Licker M., Văduva M.B., Velimirovici D., Rădulescu M., Dumitraşcu V., Crăciunescu M., Dugăeşescu D., Horhat F., Piluţ C., Bădiţoiu L., Moldovan R. Antibiotic resistance patterns in Acinetobacter spp. strains isolated from hospital environment. Bacteriol. Virusol. Parazitol. Epidemiol. 2008;53(2):103–107. [PubMed] [Google Scholar]
- 38.Mirnejad R, Vafaei S. Antibiotic resistance patterns and the prevalence of ESBLs among strains of A. baumannii isolated from clinical specimens. Journal of Genes, Microbes and Immunity. 2013:1–8. [Google Scholar]
- 39.Reguero M.T., Medina O.E., Hernández M.A., Flórez D.V., Valenzuela E.M., Mantilla J.R. Antibiotic resistance patterns of Acinetobacter calcoaceticus-A. baumannii complex species from Colombian hospitals. Enferm. Infecc. Microbiol. Clin. 2013;31(3):142–146. doi: 10.1016/j.eimc.2012.07.013. [DOI] [PubMed] [Google Scholar]
- 40.Zhao S.Y., Jiang D.Y., Xu P.C., Zhang Y.K., Shi H.F., Cao H.L., Wu Q. An investigation of drug-resistant Acinetobacter baumannii infections in a comprehensive hospital of East China. Ann. Clin. Microbiol. Antimicrob. 2015;14:7. doi: 10.1186/s12941-015-0066-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gupta V., Garg R., Garg S., Chander J., Attri A.K. Coexistence of extended spectrum beta-lactamases, AmpC beta-lactamases and metallo-beta-lactamases in Acinetobacter baumannii from burns patients: A report from a tertiary care centre of India. Ann. Burns Fire Disasters. 2013;26(4):189–192. [PMC free article] [PubMed] [Google Scholar]
- 42.Shrivastava G., Bajpai T., Bhatambare G.S., Patel K.B. Sensitivity profile of multidrug resistant Acinetobacter Spp. isolated at ICUs of tertiary care hospital. Int J Health Sys Disaster Manag. 2013;1(4):200. doi: 10.4103/2347-9019.130731. [DOI] [Google Scholar]
- 43.Banerjee M., Chaudhary B., Shukla S. Prevalence of ESBL and MBL in acinetobacter species isolated from clinical samples in tertiary care hospital. Int. J. Sci. Res. (Ahmedabad) 2015;4(6):1183–1186. [Google Scholar]
- 44.Alyamani E.J., Khiyami M.A., Booq R.Y., Alnafjan B.M., Altammami M.A., Bahwerth F.S. Molecular characterization of extended-spectrum beta-lactamases (ESBLs) produced by clinical isolates of Acinetobacter baumannii in Saudi Arabia. Ann. Clin. Microbiol. Antimicrob. 2015;14:38. doi: 10.1186/s12941-015-0098-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.AL-Thahab AA Molecular detection of extended-spectrum betalactamases in clinical isolates of A. baumannii. J Biol Agric Healthc. 2013;3(7):32–39. [Google Scholar]
- 46.Sedighi I., Arabestani M.R., Rahimbakhsh A., Karimitabar Z., Alikhani M.Y. Dissemination of extended-spectrum β-lactamases and quinolone resistance genes among clinical isolates of uropathogenic Escherichia coli in children. Jundishapur J. Microbiol. 2015;8(7):e19184. doi: 10.5812/jjm.19184v2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Darvishi M. Virulence factors profile and antimicrobial resistance of A. baumannii strains isolated from various infections recovered from immunosuppressive patients. 2016;9(3):1057–1062. [Google Scholar]
- 48.Ranjbar R., Karami A., Farshad S., Giammanco G.M., Mammina C. Typing methods used in the molecular epidemiology of microbial pathogens: A how-to guide. New Microbiol. 2014;37(1):1–15. [PubMed] [Google Scholar]
- 49.Khakabimamaghani S., Najafi A., Ranjbar R., Raam M. GelClust: A software tool for gel electrophoresis images analysis and dendrogram generation. Comput. Methods Programs Biomed. 2013;111(2):512–518. doi: 10.1016/j.cmpb.2013.04.013. [DOI] [PubMed] [Google Scholar]
- 50.Ranjbar R., Naghoni A., Yousefi S., Ahmadi A., Jonaidi N., Panahi Y. The study of genetic relationship among third generation cephalosporin-resistant salmonella enterica strains by ERIC-PCR. Open Microbiol. J. 2013;7:142–145. doi: 10.2174/1874285801307010142. [DOI] [PMC free article] [PubMed] [Google Scholar]