Abstract
Objective
The primary purpose of the present study was to survey the quality of life (QoL) in primary Sjögren’s syndrome (pSS) and to analyze the relationships between disease activity, anxiety/depression, fatigue, pain, age, oral disorders, impaired swallowing, sicca symptoms, and QoL.
Patients and methods
A survey was conducted on 185 pSS patients and 168 healthy individuals using the Short Form 36 health survey for QoL. Disease activity was assessed using the European League Against Rheumatism Sjögren’s Syndrome Disease Activity Index. We examined these data using independent samples t-tests, Mann–Whitney U test, chi squared analysis, and linear regression.
Results
The result for each domain in Short Form 36 health survey was lower in pSS patients than in healthy controls, especially the score in the dimension of role physical function. In the bivariate analysis, age, pain, fatigue, disease activity, disease complication, anxiety/depression, oral disorders, and impaired swallowing correlated with QoL. Also, in the linear regression model, pain, fatigue, disease activity, impaired swallowing, and anxiety/depression remained the main predictors of QoL.
Conclusion
pSS patients had a considerably impaired QoL compared to the controls, and pSS could negatively affect the QoL of patients. Measuring QoL should be considered as a vital part of the comprehensive evaluation of the health status of pSS patients, which could contribute some valuable clues in improving the management of disease and treatment decisions.
Keywords: primary Sjögren’s syndrome, quality of life, disease activity, depression
Background
Primary Sjögren’s syndrome (pSS) is a systemic autoimmune disease characterized by chronic inflammation of exocrine glands such as the lachrymal and salivary glands, leading to dry eyes and dry mouth.1 A majority of pSS patients also suffer from extraglandular symptoms including fatigue, mental disorders, musculoskeletal, and neurological symptoms.2 In addition, pSS patients exhibit a 44 times higher relative risk for lymphoma development compared to the general population.3 This disease predominantly affects middle-aged women (women:men ratio 9:1), but can also be detected in children and the aged.4 pSS is a common autoimmune rheumatic condition, second only in frequency to rheumatoid arthritis, with a prevalence between 0.1% and 4.8% in various populations.5 pSS has a negative influence on health-related quality of life (HR-QoL)6 and is associated with high health care costs,7 pain,8 fatigue,9 work disability,10 and anxiety/depression.11 Moreover, dry mouth in pSS may lead to oral uncomfortableness, trouble talking, impaired swallowing, tooth decay, and continual oral infections. Though these complications are not life-threatening, the complication and chronicity of symptoms can lead to heavy debility and a reduction in patient’s quality of life (QoL).12 Individuals with pSS also experience voice disturbances and specific voice-related manifestations that are related to decreased QoL.13
QoL is defined as the general well-being of individuals and societies, based on the individual’s culture and life values with respect to that individual’s objectives, expectation, and standards. HR-QoL is the impact of disease and its treatment of the individual’s ability to function based on physical, mental, and social well-being.14 Variables that may affect the QoL of pSS patients include disease activity, fibromyalgia,15 comorbidity,2 medical therapy,16 pain, fatigue, dryness, and anxiety/depression. In the literature, there are various studies investigating fatigue and discomfort, oral HR-QoL,17 employment and disability,18 and psychological status of pSS patients. However, to our best knowledge, there are a few previous studies focusing on how these variables might affect the HR-QoL in pSS, especially in China. The objective of the present study was to evaluate QoL in pSS and to explore the relationships between disease activity, fatigue, anxiety/depression, oral disorders, impaired swallowing, pain, age, dryness, and QoL.
Patients and methods
Participants
The study was carried out in the Affiliated Hospital of Nantong University from July 2016 to July 2017. pSS patients were outpatients or inpatients from the Department of Rheumatology; age- and sex-matched controls were randomly selected from the general population. The Ethics Committee of the Affiliated Hospital of Nantong University approved this study (approval number 2017-K003), and all participants met the American–European Consensus Group Criteria for pSS and gave written informed consent. All study participants were asked to complete the related questionnaires. Medical assessments also were recorded. There was no significant difference between the two groups in terms of age, sex, education level, occupation, and income/year.
Inclusion criteria and exclusion criteria
Inclusion criteria consisted of pSS diagnosis, age over 18 years, no known cognitive deficits, and Chinese speaking. Exclusion criteria consisted of secondary Sjögren syndrome, having serious systemic diseases lately (lymphoma, similar renal/pulmonary involvement, myositis, or vasculitis), and participants with a physical or psychological problem (eg, cancer, psychiatric disorders) which may confound the outcome.
Methods
This study included 185 pSS patients according to the American–European Consensus Group criteria and 168 healthy controls. Sicca symptoms were assessed by the dryness domain of the European League Against Rheumatism Sjögren’s Syndrome Patient-Reported Index.19 Disease activity was assessed using the European League Against Rheumatism Sjögren’s Syndrome Disease Activity Index (ESSDAI).20 Mental state was assessed through Hospital Anxiety and Depression Scale; it had acceptable internal consistency and test–retest reliability, with a Cronbach’s alpha of 0.85 and intraclass correlation coefficient of 0.90.21 Independent samples t-tests, chi-squared analyses, Mann–Whitney U test, and linear regression modeling were used to analyze the data.
Demographics and clinical characteristics
Demographic characteristics and clinical variables were examined. Demographic variables included age, sex, education, marital status, and socioeconomic status. Next, differences in mean Hospital Anxiety and Depression Scale scores and QoL scores between pSS patients and control groups were analyzed. Clinical data such as disease duration and medication use were obtained by asking the patients or searching their electronic medical records.
The medical outcomes study, 36-item short-form health survey (SF-36)
The SF-36 instrument is aimed to rate HR-QoL within the previous 4 weeks. It includes 36 statements, with eight scales assessing two dimensions. The first dimension is physical health function and includes the following four specific scores: physical functioning (the extent to which health interferes with various activities), physical role functioning (the extent to which health interferes with usual daily activities), bodily pain, and general health. These physical scores are summarized by the physical composite score (PCS). The second dimension is the mental health function, which includes the following four specific scores: vitality, social functioning, emotional role functioning (limitations due to emotional problems), and mental health. These four mental scores are summarized by the mental composite score (MCS). Each scale gives a standardized score that ranges from 0 to 100, with 0 illustrating the worst possible health state and 100 illustrating the best possible health state.22,23 In addition, this scale has been translated and validated for use in Hong Kong.24,25
Convergent validity and discriminant validity were satisfactory for all except the social functioning scale. Cronbach’s α coefficients ranged from 0.72 to 0.88 except 0.39 for the social functioning scale and 0.66 for the vitality scale. Two weeks test-retest reliability coefficients ranged from 0.66 to 0.94.26
Fatigue severity scale
Fatigue was measured by using the fatigue severity scale. It is a self-reported instrument developed to explore the impact and severity of fatigue. It involves nine questions to assess the severity of fatigue, as it relates to daily activities such as physical functioning, exercise, work, and family/social life. The scores range from 1 to 7 for each item, with a lower score signifying less fatigue (the best condition is 9 points and the worst condition is 63 points). We used a cut-off score ≥4 to define fatigue.27
Other related scales
Pain was investigated by the visual analog scale to identify pain severity. Patients were asked to estimate their experiences of pain during the last week, each on a visual analog scale of 0–10, with a higher score implying more severe pain.28 The MD Anderson Dysphagia Inventory29 measures patient’s swallowing disorders symptoms (score ranges from 0 to 100, with higher scores indicating more severe symptoms), and the Oral Health Impact Profile 1430 was used to measure oral distress.
Data analysis
Data were analyzed by IBM SPSS 22.0 package program. Continuous variables were expressed as mean±SD. Before the continuous variables were compared, it was checked whether the parametric test assumptions were met or not. Qualitative variables were presented as numbers and percentages. The distinction between independent groups in terms of the qualitative variables was evaluated by using chi-squared test. While conformity to normal distribution was evaluated by using Shapiro–Wilk test, the homogeneity of the variances was examined by using the Levene’s test. The difference between independent groups in terms of numerical variables was investigated by using the independent samples t-test in cases where the parametric test assumptions were met and by using the Mann–Whitney U test in cases where the said assumptions were not met. The presence of a correlation between the numerical variables was determined by Spearman’s correlation coefficient. Multicollinearity between variables was checked by computing variance inflation factors before inclusion into the model. A backward procedure was used, removing variables with a P-value of >0.05 in the multivariable model in order of significance, until the best-fitting model was identified. While we regarded P-values <0.05 in each study as significant, we set stringent significant levels of P-values in the combined analysis based on Bonferroni’s correction.
Written informed consent
Written informed consent was obtained from all individual participants included in the study.
Results
Patient characteristics
Of the 203 eligible cases, 185 patients fully completed the provided questionnaires, with a rough response rate of 91%. In pSS patients (n=185), the mean age was 50.12±12.13 years, and 94.6% of the subjects were women. Demographic, clinical, and laboratory characteristics of the pSS patients and controls are presented in Table 1. Furthermore, the impact of disease-related variables on severity of depression and anxiety was assessed. Whether depression, anxiety, and disease-related variables can be determinants of QoL among affected individuals was also investigated. There was no significant difference in age, sex, education, occupation, and income/year between the pSS individuals and the controls (P>0.05). Tests of collinearity indicated good content validity.
Table 1.
Demographic, clinical, and psychological characteristics of the pSS patients and controls
| Clinical characteristics | pSS (n=185) | Controls (n=168) | t/χ2/z | P-value |
|---|---|---|---|---|
| Age, mean±SD, years | 50.12±12.13 | 50.25±11.17 | −0.105 | 0.916 |
| Sex, female, n (%) | 175 (94.6) | 160 (95.2) | 0.075 | 0.784 |
| Marital status, n (%) | 5.590 | 0.018 | ||
| Married | 172 (93) | 165 (98.2) | ||
| Single/divorced/bereaved | 13 (7) | 3 (1.8) | ||
| Education, mean±SD, years | 12.44±4.19 | 11.99±4.64 | 0.945 | 0.345 |
| Employment status, n (%) | 0.931 | 0.335 | ||
| Employed | 83 (44.9) | 84 (50) | ||
| Unemployed | 102 (55.1) | 84 (50) | ||
| Income/person/year, yuan, n (%) | −0.671 | 0.502 | ||
| <15,000 yuan | 55 (29.7) | 49 (29.2) | ||
| 15,000–33,000 yuan | 55 (29.7) | 61 (36.3) | ||
| >33,000 yuan | 75 (40.5) | 58 (34.5) | ||
| Type of medical insurance, n (%) | 3.582 | 0.167 | ||
| With basic medical insurance | 170 (91.9) | 164 (96.5) | ||
| Self-pay | 14 (7.6) | 6 (3.6) | ||
| Other | 1 (0.5) | 0 (0) | ||
| HADS-A, mean±SD | 6.43±3.99 | 3.04±2.78 | 9.315 | 0.000 |
| HADS-D, mean±SD | 6.63±4.16 | 3.89±2.8 | 7.318 | 0.000 |
| Oral disorders, n (%) | 165 (89.2) | 101 (60.1) | 8.246 | 0.000 |
| Impaired swallowing, mean±SD | 75.52±18.04 | – | ||
| Disease duration, mean±SD, years | 4.34±5.24 | – | ||
| VAS pain (range 0–10), mean±SD | 2.15±2.56 | – | ||
| ESR, mean±SD mm/h | 29.16±23.99 | – | ||
| Hospitalization, yes, n (%) | 124 (67) | – | ||
| Comorbid conditions, yes, n (%) | 112 (60.5) | – | ||
| ESSDAI, mean±SD | 9.56±7.3 | – | ||
| ESSPRI, mean±SD | 4.18±1.89 | – | ||
| Impaired swallowing, n (%) | 51 (27.57) | – | ||
| Medicine use, n (%) | 153 (82.7) | – | ||
| Glucocorticoids, yes, n (%) | 56 (30.3) | – | ||
| Hydroxychloroquine, yes, n (%) | 140 (75.7) | – | ||
| Others, yes, n (%) | 143 (77.3) | – |
Abbreviations: ESR, erythrocyte sedimentation rate; ESSPRI, European League Against Rheumatism Patient Reported Index; ESSDAI, European League Against Rheumatism Sjögren’s Syndrome Disease Activity Index; HADS, Hospital Anxiety and Depression Scale; HADS-A, Hospital Anxiety Scale; HADS-D, Hospital Depression Scale; pSS, primary Sjögren’s syndrome; VAS, visual analog scale.
QoL of pSS patients and controls
The scores of global SF-36, including PCS and MCS of pSS patients and the controls, are presented in Figure 1. Eight domains of SF-36 of the pSS patients and controls are shown in Table 2. The average global SF-36 score of pSS patients was 56.01±18.13, ranging from 13.94 to 98.13. Compared with the controls, all SF-36 domains and total scores were much lower in pSS patients (P<0.01).
Figure 1.
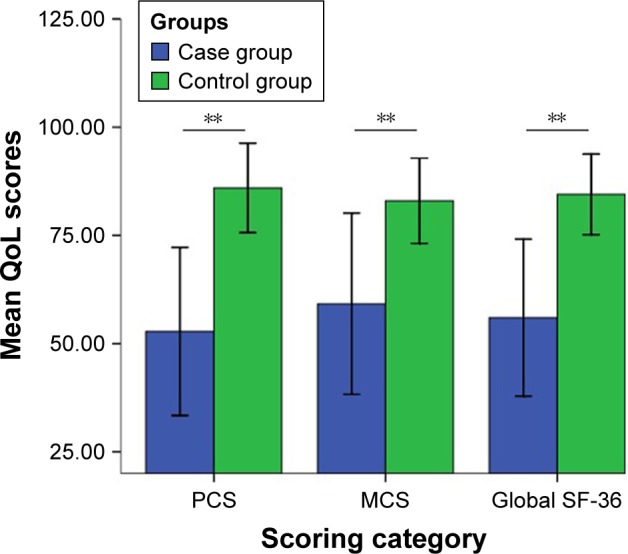
Global SF-36, PCS, and MCS among pSS patients and controls.
Note: **P<0.01.
Abbreviations: MCS, mental composite score; PCS, physical composite score; pSS, primary Sjögren’s syndrome; QoL, quality of life; SF-36, Short Form 36 health survey.
Table 2.
Eight domains in SF-36 of patients with pSS and the controls
| pSS (n=185) | Controls (n=168) | P-value | |
|---|---|---|---|
| Physical function | 76.51±23.10 | 92.97±11.28 | <0.001 |
| Role physical | 32.56±38.62 | 92.56±19.83 | <0.001 |
| Body pain | 74.51±23.55 | 88.34±15.79 | <0.001 |
| General health | 41.19±19.67 | 69.95±16.05 | <0.001 |
| Vitality | 55.16±15.35 | 74.08±10.99 | <0.001 |
| Social function | 69.12±21.93 | 87.35±12.16 | <0.001 |
| Role emotional | 42.88±45.43 | 92.06±21.96 | <0.001 |
| Mental health | 66.98±13.26 | 78.43±8.53 | <0.001 |
| Physical composite score | 52.80±19.41 | 85.96±10.33 | <0.001 |
| Mental composite score | 59.21±20.92 | 82.98±9.85 | <0.001 |
Note: Data are presented as mean±SD.
Abbreviations: pSS, primary Sjögren’s syndrome; SF-36, Short Form 36 health survey.
Correlations between fatigue, disease activity etc, and QoL in pSS patients
As shown in Table 3, we found signifcant correlations between erythrocyte sedimentation rate, comorbidity, hospitalization, and European League Against Rheumatism Sjögren’s Syndrome Patient-Reported Index and QoL.
Table 3.
Correlations between disease activity, anxiety/depression, fatigue, pain, age, and QoL in pSS patients
| Domains of SF-36 | ESSDAI
|
VAS
|
HADS-A
|
HADS-D
|
FSS
|
Age
|
||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R | P-value | R | P-value | R | P-value | R | P-value | R | P-value | R | P-value | |
| PF | −0.232 | 0.001** | −0.334 | 0.000** | −0.253 | 0.001** | −0.363 | 0.000** | −0.355 | 0.000** | −0.368 | 0.000** |
| RP | −0.354 | 0.000** | −0.251 | 0.001** | −0.415 | 0.000** | −0.403 | 0.003** | −0.453 | 0.000** | −0.308 | 0.000** |
| BP | −0.304 | 0.000** | −0.697 | 0.000** | −0.216 | 0.003** | −0.250 | 0.001** | −0.312 | 0.001** | −0.081 | 0.270 |
| GH | −0.361 | 0.000** | −0.250 | 0.001** | −0.495 | 0.000** | −0.480 | 0.000** | −0.420 | 0.000** | −0.203 | 0.005** |
| VT | −0.274 | 0.000** | −0.274 | 0.000** | −0.463 | 0.000** | −0.497 | 0.000** | −0.542 | 0.000** | −0.271 | 0.000** |
| SF | −0.428 | 0.000** | −0.342 | 0.000** | −0.400 | 0.000** | −0.493 | 0.000** | −0.467 | 0.000** | −0.238 | 0.001** |
| RE | −0.299 | 0.000** | −0.230 | 0.002** | −0.475 | 0.000** | −0.463 | 0.000** | −0.404 | 0.000** | −0.166 | 0.000** |
| MH | −0.249 | 0.001** | −0.254 | 0.000** | −0.648 | 0.000** | −0.672 | 0.000** | −0.337 | 0.000** | −0.195 | 0.008** |
| PCS | −0.287 | 0.000** | −0.413 | 0.000** | −0.461 | 0.000** | −0.493 | 0.000** | −0.388 | 0.000** | −0.294 | 0.000** |
| MCS | −0.391 | 0.000** | −0.324 | 0.000** | −0.560 | 0.000** | −0.586 | 0.000** | −0.530 | 0.000** | −0.231 | 0.002** |
Note:
P<0.01.
Abbreviations: BP, bodily pain; ESSDAI, European League Against Rheumatism Sjögren’s Syndrome Disease Activity Index; FSS, Fatigue Severity Scale; GH, general health; HADS-A, Hospital Anxiety Scale; HADS-D, Hospital Depression Scale; MCS, mental composite score; MH, mental health; PCS, physical composite score; PF, physical functioning; pSS, primary Sjögren’s syndrome; QoL, quality of life; RE, role emotional; RP, role physical; SF, social functioning; SF-36, Short Form 36 health survey; VAS, visual analog scale; VT, vitality.
Stepwise linear regression analysis for the QoL
Stepwise linear regression analysis revealed depression, pain, and impaired swallowing were notably associated with the SF-36 PCS in pSS patients. Moreover, depression, anxiety, disease activity, fatigue, and impaired swallowing significantly accounted for the SF-36 MCS (Table 4). Finally, these variables explained 65.5% and 72.5% of the variance in PCS and MCS, respectively.
Table 4.
Result of analysis of forward stepwise ordered linear regression models in SF-36
| Predictors | β | SE | P-value | B (95% CI) |
|---|---|---|---|---|
| MCS | ||||
| Depression | −0.260 | 0.392 | 0.001 | −1.309 (−2.083, −0.535) |
| FSS | −0.251 | 0.725 | 0.000 | −3.053 (−4.483, −1.623) |
| Impaired | 0.171 | 0.068 | 0.004 | 0.198 (0.064, 0.332) |
| swallowing | ||||
| ESSDAI | −0.181 | 0.160 | 0.001 | −0.525 (−0.916, −0.280) |
| Anxiety | −0.165 | 0.411 | 0.037 | −0.866 (−1.677, −0.055) |
| PCS | ||||
| Impaired | 0.322 | 0.065 | 0.000 | 0.346 (0.217, 0.475) |
| swallowing | ||||
| Depression | −0.327 | 0.282 | 0.000 | −1.526 (−2.082, −0.970) |
| Pain | −0.260 | 0.444 | 0.000 | −1.969 (−2.846, −1.093) |
Abbreviations: ESSDAI, European League Against Rheumatism Sjögren’s Syndrome Disease Activity Index; FSS, Fatigue Severity Scale; MCS, mental composite score; PCS, physical composite score; SE, standard error; SF-36, Short Form 36 health survey.
Discussion
pSS patients experience lower QoL compared to age-and sex-matched controls. Impaired QoL is associated with symptoms such as fatigue, pain, anxiety/depression, oral disorders, impaired swallowing, as well as disease activity, exemplifying the value of optimal management of all aspects of the disease. The underlying pathogenic process between depression and pSS remains unknown. Some studies attempted to explore depression in pSS patients; depression was reported as a relative factor of reduced QoL in Moroccan pSS patients.31 Segal et al32 stated that somatic fatigue was the main predictor of physical function and general health; additionally, depression was the key predictor of emotional well-being in the US. In agreement with the findings of previous studies, our study found that the strong correlation between MCS of the SF-36 and anxiety/depression in pSS patients emphasized the significance of the psychological dimension of SF-36. Consequently, recognition of determinants of depression and anxiety and their impact on QoL has noticeable clinical value, especially in China.
Fatigue is another essential symptom and has been reported to be associated with a reduction in HR-QoL in pSS.33 A study from Germany reported that patients with pSS frequently suffer from fatigue that increases the risk of work disability and requires treatment by numerous health care specialists.34 As expected, bivariate analysis showed that fatigue correlated with all domains of the SF-36. Fatigue and pain are the most common extraglandular symptoms,35 and pain severity explained 14% of the variance in disability of daily activities in Korean patients with pSS.36 Our results were in line with previous studies reporting that pain is a major symptomatic problem related to low physical functioning and health outcomes. Therefore, systematic monitoring of pain treatment seems essential.
The results showed that pSS patients had a moderate level of disease activity, with a mean ESSDAI score of 9.56. Similarly, the mean ESSDAI score of pSS patients from multicenter clinics of France was 9.04.37 The comparison to age- and sex-matched healthy controls suggests that the disease-related symptoms of pSS patients are not attributed to the natural process of aging, but age was truly thought to play a crucial role in dysregulation of the neuroendocrine system.38 We found that patient-perceived swallowing disorders were relatively common in pSS and increased with disease severity, which was uniquely associated with reduced QoL. Since eating and drinking form an important part of social interaction, dysphagic pSS patients often eat in seclusion because of shame. Also, they worry choking on their food or develop aspiration pneumonia. Frequent worries can decrease the QoL even more and aggravate anxiety. Therefore, health care professionals need to pay more attention to pSS patients who manifest swallowing symptoms and to strengthen the assessment of swallowing function of the pSS patients in clinical practice.
Notably, a significant difference was found in the marital status between the groups. The possible explanation is that women with pSS often suffer from vaginal dryness. Many pSS patients were unable to continue regular marital relationship due to sexual difficulties. Disease severity and disease duration may cause an increase in the divorce rate.
There are several limitations of this study. First, this study is cross-sectional in design, which does not allow for following up the progress of the examined variables during the course of the disease, and thus, we are unable to comment on the causal relationships. Second, it fails to make a distinction between men and women because of the great sex disparity. Third, all subjects were from a single hospital and the scales were all self-administered, which might give rise to possible biases of the outcomes. Even so, we still consider that our results may provide important insights into this complex process by identifying the associations between variables and QoL in pSS patients.
Conclusion
Patients with pSS had a considerable impaired QoL in comparison with the healthy subjects. pSS could have an unfavorable influence on the QoL of patients. Measuring QoL should be considered as a vital part of the comprehensive evaluation of the health status of pSS patients, which could contribute some valuable clues to improving the management of disease and treatment decisions.
Acknowledgments
This study was funded by the Chinese National Natural Science Foundation (Grant no 81671616 and 81471603), Jiangsu Provincial Commission of Health and Family Planning Foundation (Grant no H201317 and H201623), the Science Foundation of Nantong City (Grant no MS32015021, MS2201564, MS22016028, and MS22016019), and the Science and Technology Foundation of Nantong City (Grant no HS2014071 and HS2016003).
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Xiao F. Neuromyotonia as an unusual neurological complication of primary Sjögren’s syndrome: case report and literature review. Clin Rheumatol. 2017;36(2):481–484. doi: 10.1007/s10067-016-3499-z. [DOI] [PubMed] [Google Scholar]
- 2.Lackner A, Ficjan A, Stradner MH, et al. It’s more than dryness and fatigue: The patient perspective on health-related quality of life in primary Sjögren’s syndrome – a qualitative study. PLoS One. 2017;12(2):e1720562017-02-09. doi: 10.1371/journal.pone.0172056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Papageorgiou A, Voulgarelis M, Tzioufas AG. Clinical picture, outcome and predictive factors of lymphoma in Sjögren syndrome. Autoimmun Rev. 2015;14(7):641–649. doi: 10.1016/j.autrev.2015.03.004. [DOI] [PubMed] [Google Scholar]
- 4.Brito-Zero NPB. Sjogren syndrome. Nat Rev Dis Primers. 2016;2:16047. doi: 10.1038/nrdp.2016.47. [DOI] [PubMed] [Google Scholar]
- 5.Gupta S, Gupta N. Sjögren Syndrome and Pregnancy: A Literature Review. Perm J. 2017;21:16–047. doi: 10.7812/TPP/16-047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cornec D, Devauchelle-Pensec V, Mariette X, et al. Severe health-related quality of life impairment in active primary Sjögren’s syndrome and patient-reported outcomes: data from a large therapeutic trial. Arthritis Care Res. 2017;69(4):528–535. doi: 10.1002/acr.22974. [DOI] [PubMed] [Google Scholar]
- 7.Milin M, Cornec D, Chastaing M, et al. Sicca symptoms are associated with similar fatigue, anxiety, depression, and quality-of-life impairments in patients with and without primary Sjögren’s syndrome. Joint Bone Spine. 2016;83(6):681–685. doi: 10.1016/j.jbspin.2015.10.005. [DOI] [PubMed] [Google Scholar]
- 8.Koh JH, Kwok SK, Lee J, et al. Pain, xerostomia, and younger age are major determinants of fatigue in Korean patients with primary Sjögren’s syndrome: a cohort study. Scand J Rheumatol. 2017;46(1):49–55. doi: 10.3109/03009742.2016.1153142. [DOI] [PubMed] [Google Scholar]
- 9.Karageorgas T, Fragioudaki S, Nezos A, Karaiskos D, Moutsopoulos HM, Mavragani CP. Fatigue in primary Sjögren’s syndrome: clinical, laboratory, psychometric, and biologic associations. Arthritis Care Res. 2016;68(1):123–131. doi: 10.1002/acr.22720. [DOI] [PubMed] [Google Scholar]
- 10.Mandl T, Jørgensen TS, Skougaard M, Olsson P, Kristensen LE. Work disability in newly diagnosed patients with primary Sjögren syndrome. J Rheumatol. 2017;44(2):209–215. doi: 10.3899/jrheum.160932. [DOI] [PubMed] [Google Scholar]
- 11.Koçer B, Tezcan ME, Batur HZ, et al. Cognition, depression, fatigue, and quality of life in primary Sjögren’s syndrome: correlations. Brain Behav. 2016;6(12):e586. doi: 10.1002/brb3.586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Leung KC, Mcmillan AS, Wong MC, Leung WK, Mok MY, Lau CS. The efficacy of cevimeline hydrochloride in the treatment of xerostomia in Sjögren’s syndrome in southern Chinese patients: a randomised double-blind, placebo-controlled crossover study. Clin Rheumatol. 2008;27(4):429–436. doi: 10.1007/s10067-007-0723-x. [DOI] [PubMed] [Google Scholar]
- 13.Tanner K, Pierce JL, Merrill RM, Miller KL, Kendall KA, Roy N. The quality of life burden associated with voice disorders in Sjögren’s syndrome. Ann Otol Rhinol Laryngol. 2015;124(9):721–727. doi: 10.1177/0003489415579911. [DOI] [PubMed] [Google Scholar]
- 14.Abushakra M. Quality of Life, Coping and Depression in Systemic Lupus Erythematosus. Isr Med Assoc J. 2016;18(3–4):144–145. [PubMed] [Google Scholar]
- 15.El-Rabbat MS, Mahmoud NK, Gheita TA. Clinical significance of fibromyalgia syndrome in different rheumatic diseases: relation to disease activity and quality of life. Rheumatol Clin. 2017 doi: 10.1016/j.reuma.2017.02.008. [DOI] [PubMed] [Google Scholar]
- 16.Jiang Q, Zhang H, Pang R, Chen J, Liu Z, Zhou X. Acupuncture for primary Sjögren syndrome (pSS) on symptomatic improvements: study protocol for a randomized controlled trial. BMC Complement Altern Med. 2017;17(1):61. doi: 10.1186/s12906-017-1559-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Catanzaro J, Dinkel S. Sjögren’s syndrome: the hidden disease. Medsurg Nurs. 2014;23(4):219–223. [PubMed] [Google Scholar]
- 18.Meijer JM, Meiners PM, Huddleston Slater JJ, et al. Health-related quality of life, employment and disability in patients with Sjogren’s syndrome. Rheumatology. 2009;48(9):1077–1082. doi: 10.1093/rheumatology/kep141. [DOI] [PubMed] [Google Scholar]
- 19.Seror R, Bowman SJ, Brito-Zeron P, et al. EULAR Sjögren’s syndrome disease activity index (ESSDAI): a user guide. RMD Open. 2015;1(1):e22. doi: 10.1136/rmdopen-2014-000022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Seror R, Ravaud P, Mariette X, et al. EULAR Sjogren’s Syndrome Patient Reported Index (ESSPRI): development of a consensus patient index for primary Sjogren’s syndrome. Ann Rheum Dis. 2011;70(6):968–972. doi: 10.1136/ard.2010.143743. [DOI] [PubMed] [Google Scholar]
- 21.Wang W, Chair SY, Thompson DR, Twinn SF. A psychometric evaluation of the Chinese version of the Hospital Anxiety and Depression Scale in patients with coronary heart disease. J Clin Nurs. 2009;18(17):2436–2443. doi: 10.1111/j.1365-2702.2009.02807.x. [DOI] [PubMed] [Google Scholar]
- 22.Dassouki T, Benatti FB, Pinto AJ, et al. Objectively measured physical activity and its influence on physical capacity and clinical parameters in patients with primary Sjögren’s syndrome. Lupus. 2017;26(7):690–697. doi: 10.1177/0961203316674819. [DOI] [PubMed] [Google Scholar]
- 23.Ware JE, Gandek B, Kosinski M, et al. The equivalence of SF-36 summary health scores estimated using standard and country-specific algorithms in 10 countries: results from the IQOLA project. International Quality of Life Assessment. J Clin Epidemiol. 1998;51(11):1167–1170. doi: 10.1016/s0895-4356(98)00108-5. [DOI] [PubMed] [Google Scholar]
- 24.Lam CL, Gandek B, Ren XS, Chan MS. Tests of scaling assumptions and construct validity of the Chinese (HK) version of the SF-36 health survey. J Clin Epidemiol. 1998;51(11):1139–1147. doi: 10.1016/s0895-4356(98)00105-x. [DOI] [PubMed] [Google Scholar]
- 25.Lam CL, Tse EY, Gandek B, Fong DY. The SF-36 summary scales were valid, reliable, and equivalent in a Chinese population. J Clin Epidemiol. 2005;58(8):815–822. doi: 10.1016/j.jclinepi.2004.12.008. [DOI] [PubMed] [Google Scholar]
- 26.Li L, Wang HM, Shen Y. Chinese SF-36 health survey: translation, cultural adaptation, validation, and normalisation. J Epidemiol Community Health. 2003;57(4):259–263. doi: 10.1136/jech.57.4.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Krupp LB, Larocca NG, Muir-Nash J, Steinberg AD. The fatigue severity scale. Application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol. 1989;46(10):1121–1123. doi: 10.1001/archneur.1989.00520460115022. [DOI] [PubMed] [Google Scholar]
- 28.Sokka T, Kankainen A, Hannonen P. Scores for functional disability in patients with rheumatoid arthritis are correlated at higher levels with pain scores than with radiographic scores. Arthritis Rheum. 2000;43(2):386–389. doi: 10.1002/1529-0131(200002)43:2<386::AID-ANR19>3.0.CO;2-Z. [DOI] [PubMed] [Google Scholar]
- 29.Pierce JL, Tanner K, Merrill RM, Miller KL, Kendall KA, Roy N. Swallowing disorders in Sjögren’s syndrome: prevalence, risk factors, and effects on quality of life. Dysphagia. 2016;31(1):49–59. doi: 10.1007/s00455-015-9657-7. [DOI] [PubMed] [Google Scholar]
- 30.Enger TB, Palm Ø, Garen T, Sandvik L, Jensen JL. Oral distress in primary Sjögren’s syndrome: implications for health-related quality of life. Eur J Oral Sci. 2011;119(6):474–480. doi: 10.1111/j.1600-0722.2011.00891.x. [DOI] [PubMed] [Google Scholar]
- 31.Ibn Yacoub Y, Rostom S, Laatiris A, Hajjaj-Hassouni N. Primary Sjögren’s syndrome in Moroccan patients: characteristics, fatigue and quality of life. Rheumatol Int. 2012;32(9):2637–2643. doi: 10.1007/s00296-011-2009-5. [DOI] [PubMed] [Google Scholar]
- 32.Segal B, Bowman SJ, Fox PC, et al. Primary Sjögren’s syndrome: health experiences and predictors of health quality among patients in the United States. Health Qual Life Outcomes. 2009;7(46):1. doi: 10.1186/1477-7525-7-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cho HJ, Yoo JJ, Yun CY, et al. The EULAR Sjogren’s syndrome patient reported index as an independent determinant of health-related quality of life in primary Sjogren’s syndrome patients: in comparison with non-Sjogren’s sicca patients. Rheumatology. 2013;52(12):2208–2217. doi: 10.1093/rheumatology/ket270. [DOI] [PubMed] [Google Scholar]
- 34.Westhoff G, Dörner T, Zink A. Fatigue and depression predict physician visits and work disability in women with primary Sjögren’s syndrome: results from a cohort study. Rheumatology. 2012;51(2):262–269. doi: 10.1093/rheumatology/ker208. [DOI] [PubMed] [Google Scholar]
- 35.Segal B, Thomas W, Rogers T, et al. Prevalence, severity, and predictors of fatigue in subjects with primary Sjögren’s syndrome. Arthritis Rheum. 2008;59(12):1780–1787. doi: 10.1002/art.24311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shim E-J, Hahm B-J, Go DJ, et al. Modeling quality of life in patients with rheumatic diseases: the role of pain catastrophizing, fear-avoidance beliefs, physical disability, and depression. Disabil Rehabil. 2018;40(13):1509–1516. doi: 10.1080/09638288.2017.1300691. [DOI] [PubMed] [Google Scholar]
- 37.Seror R, Ravaud P, Bowman SJ, et al. EULAR Sjogren’s syndrome disease activity index: development of a consensus systemic disease activity index for primary Sjogren’s syndrome. Ann Rheum Dis. 2010;69(6):1103–1109. doi: 10.1136/ard.2009.110619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Oxenkrug GF. Metabolic syndrome, age-associated neuroendocrine disorders, and dysregulation of tryptophan-kynurenine metabolism. Ann N Y Acad Sci. 2010;1199(1):1–14. doi: 10.1111/j.1749-6632.2009.05356.x. [DOI] [PubMed] [Google Scholar]