Abstract
Diabetes is caused by combined abnormalities in insulin production and action. The pathophysiology of these defects has been studied extensively and is reasonably well understood. Their causes are elusive and their manifestations pleiotropic, likely reflecting the triple threat of genes, environment, and lifestyle. Treatment, once restricted to monotherapy with secretagogues or insulin, now involves complex combinations of expensive regimens that stem the progression but do not fundamentally alter the underlying causes of the disease. As advances in our understanding of insulin action and β-cell failure reach a critical stage, here I draw on lessons learned from our research on insulin regulation of gene expression and pancreatic β-cell dedifferentiation to address the question of how we can translate this exciting biology into mechanism-based interventions to reverse the course of diabetes.
Introduction
The goal of our work is to understand the integrated physiology of insulin resistance and β-cell dysfunction to define potential sites and modes of intervention against diabetes (1). The impetus to do so is that effective, durable, and safe treatment requires mechanism-based approaches. To wit, treatment of autoimmune or oncologic disease—for example—has been transformed by targeting therapeutics based on a level of mechanistic engagement still lacking in metabolic disease, largely but not exclusively by means of biologics such as monoclonal antibodies. In recent years, new classes of antidiabetes drugs have been introduced into medical practice, including some that show promise to decrease macrovascular disease, the major cause of diabetes-related deaths (2). Paradoxically, this progress has had a blowback effect on research and development of new diabetes therapeutics, owing to the perception that intractable science, burdensome regulatory constraints, and payers’ diminishing appetite for costlier and potentially more complex regimens no longer justify the hefty up-front investment required to succeed. This review attempts to disabuse us of this notion by emphasizing research areas that have the potential to deliver transformative diabetes treatments.
After Insulin Binds
Since its introduction in the literature, insulin resistance has stood the test of time as a necessary predisposing factor to diabetes, even as its definition continues to evolve (3). In addition, evidence that restoring insulin sensitivity can prevent, delay, or improve diabetes and its complications is compelling (4). Although a systematic assessment of the cellular underpinnings of insulin action, as recently reviewed (5), is outside the purview of this discussion, it should be noted that insulin signaling, once a “black box,” has now been characterized extensively. This is a remarkable achievement by the scientific community. All modern diabetes treatments, from thiazolidinediones to glucagon-like peptide 1 analogs/dipeptidyl peptidase 4 and sodium–glucose cotransporter 2 inhibitors have either directly arisen and/or substantially benefited from a wealth of information available as a result of this endeavor. Why hasn’t a disease-modifying treatment emerged from this work? The primary reason is that the key mediators of insulin signaling either are poor drug target candidates due to their structure or are shared in common with processes that regulate cell growth. In addition, the several subphenotypes subsumed under the rubric of “insulin resistance” do not easily conform to a single treatment modality (6). In my opinion, we need a greater level of target specificity within the integrative and cellular modalities of insulin signaling to achieve safe and lasting therapeutic effects.
INSULIN ACTION AND THE DISCOVERY OF Foxo
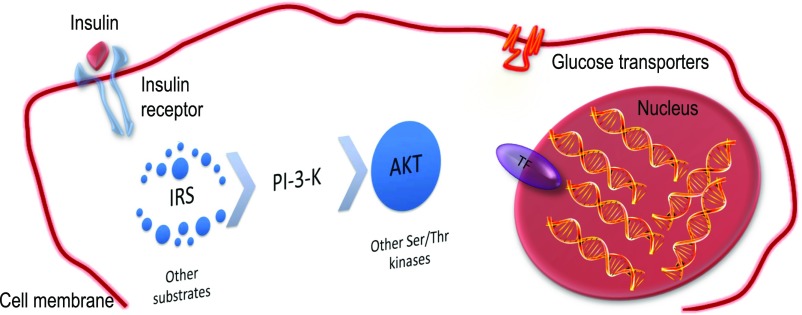
A watershed in insulin action research was the discovery of a family of insulin-responsive transcription factors in nematodes, the Foxo proteins (7). I refer to the three isoforms (Foxo1, 3a, and 4) collectively as Foxo. I use the specific isoform notation when the information available is limited to that specific isoform, e.g., Foxo1. Prior to it, research on the biochemistry of insulin action focused on the early steps following activation of the insulin receptor, while the pathophysiology of insulin resistance emphasized the impairment of insulin-dependent glucose transport and utilization in muscle and the role of glucose transporters in that process (8). Our contribution to this field was to shift the focus of investigation from the cell surface to the nucleus (Fig. 1). We were attracted to the idea that the integrated effects of insulin on multiple cellular functions could best be explained by its ability to regulate many genes at once through a transcription factor. Although the effects of insulin and glucagon on gene expression were widely recognized (9), the extent of their metabolic impact was not fully realized until genetic experiments revealed the startling reversal of diabetes through inhibition of Foxo1 (10–13). Moreover, Foxo’s ability to link a ubiquitous biochemical signal (Akt phosphorylation) to distinct sets of transcriptional targets in different cell types is important for the diversification of insulin signaling in different organs and has allowed the two cardinal features of diabetes, insulin resistance and pancreatic β-cell dysfunction, to be subsumed under a unifying Foxo-dependent mechanism (14,15).
Figure 1.
Shifting focus in insulin action research. Insulin acts through a single cell surface receptor, activating its tyrosine phosphorylation. This signal is converted through adaptor proteins (Irs) into a lipid phosphorylation signal (PI-3-kinase), which is then engaged to activate a subset of serine-threonine kinases, the most important of which is Akt. This leads to various biologic effects, including translocation of glucose transporters and inactivation of Foxo. TF, transcription factor.
PLEIOTROPIC FUNCTIONS OF Foxo IN METABOLISM
The functions of mammalian Foxo are more diverse and complex than foreshadowed from the nematode work. There are 3 Foxo isoforms, 1, 3a, and 4, exerting additive effects on gene expression. Their overarching physiologic role is to enable metabolic flexibility, i.e., the ability to switch from glucose to lipid utilization depending on nutrient availability (16,17). However, at a more granular level, this general property morphs into a more nuanced mode of action. Thus, in the central nervous system, Foxo integrates energy intake with energy expenditure through its actions on neuropeptide production, processing, and signaling in the hypothalamus (18–20). In the liver, it regulates hepatic glucose and lipid production (21–23), and in adipocytes, free fatty acid turnover (24). In the vasculature, Foxo regulates nitric oxide production and inflammatory responses (25–27). In addition, it carries out seemingly distinct functions in tissue differentiation and lineage stability that are especially evident in pancreatic and enteroendocrine cells (15,28–31). It does so through an elegant mechanism of phosphorylation-induced nuclear exclusion. Thus, in the postabsorptive state, Foxo is located in the nucleus and regulates gene expression. In response to meals, it undergoes insulin-dependent phosphorylation and is transferred from the nucleus to the cytoplasm, where it lies quiescent until nutrient levels fall again (7).
The key conceptual advance of this work is the extent to which the actions of insulin can be accounted for by changes in gene transcription. Experimental animal studies show that blocking insulin action by incapacitating any of its key upstream components (insulin receptor and its substrates, PI-3-kinase, or Akt) leads to Foxo activation, with ensuing insulin resistance and diabetes. This effect can be reversed by concurrent inactivation of Foxo, reestablishing metabolic control (10–13). Similar epistasis can be demonstrated to regulate fiber type composition and mass in skeletal muscle (32,33), as well as β-cell mass (15). These phenotypes are surprising to the extent that it had long been assumed that the primary actions of insulin occurred by regulating enzyme activities rather than gene expression (34). The data should not be construed to indicate that this signaling module is redundant but simply that its local actions in any given tissue can be compensated by insulin acting indirectly through substrate fluxes from other organs (11).
Foxo AND HEPATIC INSULIN RESISTANCE
Insulin integrates hepatic glucose and lipid metabolism, directing nutrients to glycolysis, glycogen storage, and lipogenesis. The diabetic liver overflows with glucose and atherogenic lipoproteins such VLDL triglycerides and small dense LDL, resulting in lower HDL. Thus, hepatic insulin resistance can be viewed as a causal factor in both microvascular (hyperglycemia-driven) and macrovascular (lipoprotein-driven) complications of diabetes (34).
Decreasing hepatic glucose production has long been viewed as a therapeutic target in diabetes. There appear to be at least three different modalities to regulate hepatic glucose production: 1) indirect regulation of nutrient flow from other tissues; 2) direct, Foxo-dependent actions in the hepatocyte, in part by its regulation of glucogenic enzymes glucose-6-phosphatase (G6pc) and glucokinase (Gck) and in part through yet undefined mechanisms; and 3) direct, Foxo-independent actions (34). I will not attempt to summarize, let alone reconcile, the many different opinions on these phenomena. No animal model can be said to fully recapitulate the human pathophysiology, in part because the latter is likely comprised of several distinct subphenotypes whose relative pathophysiologic contribution changes over the course of the disease. Which experimental lessons can be generalized and contribute to new approaches to treatment? First, suppression of adipocyte lipolysis by insulin is required to suppress hepatic glucose production in humans as well as rodents. Whether central nervous system–derived signals are also important in this process remains unclear because of species and methodologic differences (34). Somewhat overlooked, but worthy of consideration, is the notion that paracrine mechanisms modulate glucose production through nitric oxide generated in hepatic endothelial cells (35).
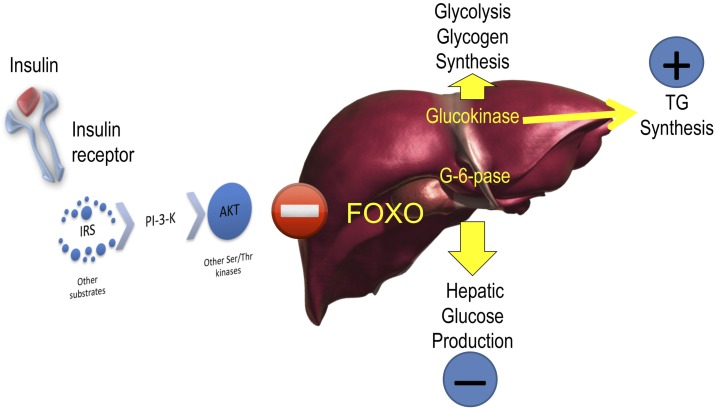
The Foxo pathway provides evidence that regulation of glucose and lipid production in the liver are mechanistically intertwined. Specifically, Foxo is the key factor controlling the coordinate induction of G6pc and suppression of Gck in the postabsorptive state (Fig. 2) (16). In normal physiology, the ratio of G6pc to Gck is key to maintaining plasma glucose levels within a narrow range. The tight linkage between the ability of insulin to suppress G6pc and activate Gck means that in insulin resistance, as insulin levels rise to maintain normal glucose production by inhibiting Foxo-dependent G6pc, they also promote Gck expression, predisposing to hepatic triglyceride accumulation and secretion. (Incidentally, this explains why G6pc levels are normal in patients with diabetes receiving treatment.) Thus, an atherogenic lipoprotein profile can be viewed as the price to be paid to suppress glucose production during the euglycemic phase of insulin resistance, as well as the early stages of diabetes. At a more fundamental level, this observation allowed us to propose that Foxo has arisen in evolution as a mechanism to control metabolic flexibility (16,17).
Figure 2.
Foxo as the single checkpoint for G6pc and Gck. Insulin activates Gck and inhibits G6pc through the Akt pathways. Surprisingly, it does so through a single mediator, Foxo. The implication of this discovery is that suppression of glucose release through inhibition of G6pc is bound to increase flux through Gck, with the potential to increase lipogenesis and glucose oxidation and storage. TG, triglyceride.
Bile Acids and Insulin Sensitivity
Another discovery from our studies is that insulin regulates bile acid pool composition through Cyp8b1, the 12α-hydroxylase responsible for the generation of cholic acid and deoxycholic acid. This has important implications as a mechanism to bypass the G6pc/Gck switch described above. Bile acids play important roles in triglyceride and cholesterol homeostasis and affect glucose levels. They act through different receptors, including the nuclear receptor Fxr, the cell surface receptor Tgr5, and nonreceptor-mediated mechanisms (36). Although there has been much interest in assessing total bile acid levels, less attention has been paid to specific types of bile acids made in normal and disease conditions. Initially synthesized as chenodeoxycholic acid and its derivative lithocholic acid, bile acids undergo conversion by gut microbes to yield cholic acid and deoxycholic acid. We discovered that insulin controls the 12α-hydroxylase enzyme necessary for the synthesis of chenodeoxycholic versus lithocholic acid, encoded by the Cyp8b1 gene, through Foxo1 (37). The consequences of this control are important to insulin sensitivity because 12α-hydroxylated bile acids differentially influence intestinal lipid absorption and the activity of bile acid receptors. Thus, one would predict that insulin resistance would increase 12α-hydroxylase activity and promote the formation of bile acids that increase triglyceride and cholesterol levels. This idea found support in studies of a cohort of euglycemic individuals stratified according to their insulin sensitivity, showing that levels of 12α-hydroxylated bile acids increase as a function of the quartile of insulin resistance. In addition, principal component analyses showed a strong correlation between 12α-hydroxylated bile acids and insulin, triglyceride, and LDL cholesterol, consistent with the idea that this constellation of bile acids is proatherogenic (38). Cyp8b1 remains an attractive target for drug development.
Designing Selective Insulin Sensitizers
The ability of Foxo to coordinate the dynamic relationship of G6pc and Gck during the fasting-to-feeding transition recapitulates the challenges underlying treatments for insulin resistance. In this respect, augmentation of insulin sensitivity would inhibit glucose production (lowering G6pc), potentially benefiting fasting hyperglycemia, but as hepatic glucose utilization increases, so does lipid synthesis, potentially predisposing to steatosis and release of atherogenic lipoproteins. This is a daily conundrum in clinical practice.
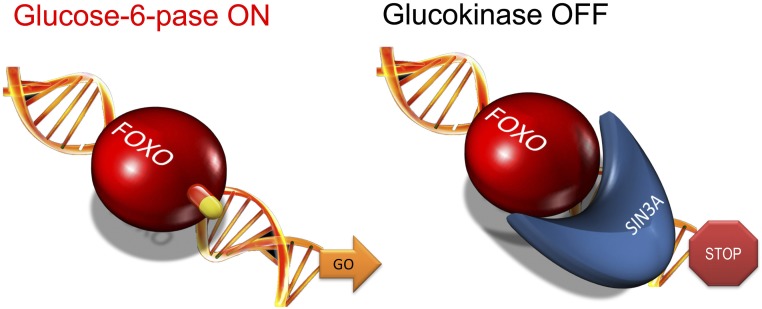
Our approach to this problem was to further dissect the Foxo regulation, i.e., how does Foxo turn on one gene (G6pc) and off another one (Gck)? We discovered the transcriptional coregulator Sin3a as the elusive Foxo-dependent Gck corepressor (Fig. 3) (39). This discovery raised the possibility that the two functions of Foxo1—as an activator and as a repressor of genes—could be independently modulated. Thus, we reckoned that if we selectively blocked the activating function of Foxo and left the repressor function intact, we could reduce the excessive hepatic glucose production characteristic of diabetes without increasing triglyceride synthesis. Screening of a library of small molecules led us to identify two series of Foxo inhibitors that were pared down through a combination of biochemical, computational, and toxicology analyses to a small number that could be tested in primary hepatocytes for their ability to differentially modulate G6pc and Gck (39).
Figure 3.
Activating and repressing function of Foxo1. Many transcription factors have dual effects on gene expression, turning some genes on and others off. In the case of Foxo regulation of G6pc and Gck, the activation mechanism is based on direct DNA binding through the protein’s namesake, forkhead domain. The repressor mechanism is based on the recruitment of Sin3a, a large component of the gene repressor complex, as the Foxo corepressor of Gck. This opens up the possibility of identifying drugs that act selectively on a specific function.
The expectation was that full inhibitors of Foxo1 would decrease G6pc and increase Gck, as indeed they did. In contrast, selective Foxo1 inhibitors would lower G6pc without increasing Gck. These results were borne out by functional assays of glucose production and de novo lipogenesis in hepatocytes (39). This work provided proof of principle that it is possible to modulate critical nodes of insulin signaling to dial up or down individual biologic responses to the hormone. Moreover, it showed that unliganded transcription factors, long the bane of drug development, can be targeted pharmacologically, leading to selective reversal of insulin resistance. Although fraught with uncertainty, the road ahead is ripe for transformative drug discovery.
β-Cell Dedifferentiation and β-Cell Failure
An intrinsic susceptibility of the β-cell to functional exhaustion, or “β-cell failure,” sets apart those individuals who go on to develop diabetes from those that, given the same level of insulin resistance, do not (40). And yet, we know from clinical experience that β-cell failure can be reversed, albeit partially, for years on end, even after the onset of hyperglycemia (41). There are at least three abnormalities of islet cell function in diabetes: impaired insulin response to stimulus, reduced number of β-cells, and inappropriate glucagon response (40).
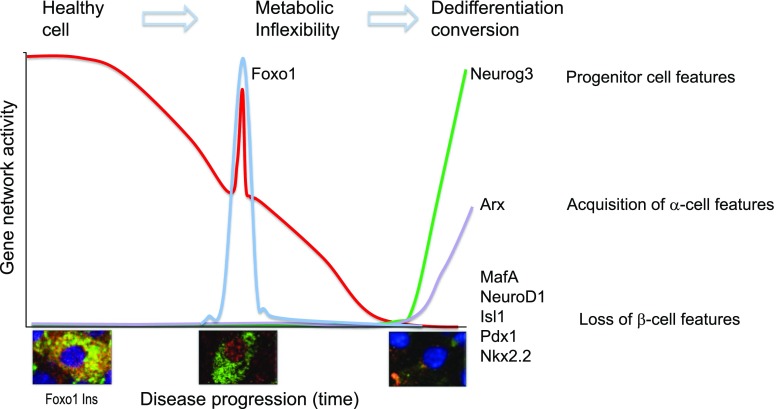
Our interest in this area was triggered by the striking regulation of Foxo in β-cells: present but inactive in healthy β-cells, Foxo is activated in early diabetes by moving into the nucleus; however, in advanced diabetes, it disappears from β-cells even as the latter lose their insulin content (29,30,42). Our research showed that these three features correspond to three stages in the development of β-cell failure: 1) healthy basal state, 2) impaired coupling of cellular metabolism with insulin secretion (metabolic inflexibility), and 3) loss of features as a mature, hormone-laden cell (dedifferentiation) with conversion to other endocrine cell types (Fig. 4) (43). In response to the stress associated with the growing metabolic demand for insulin, β-cells respond by activating Foxo (29). This activation boosts a network of homeostatic genes, including the maturity-onset diabetes of the young genes Hnf4α, Hnf1α, and Pdx1, to stave off failure (17) as long as the inciting causes are removed. Otherwise, there is loss of β-cell features, appearance of progenitor-like features, and acquisition of α-cell properties.
Figure 4.
Model of the progression of β-cell failure. The graph shows a model of changes in gene expression during three phases of progression of β-cell dysfunction, indicated at the top. Metabolic inflexibility is a state in which the pairing of ATP generation with insulin secretion is impaired. Dedifferentiation is a state in which features of mature β-cells are lost. Shown at the bottom is representative immunohistochemistry of Foxo1 translocation from the cytoplasm to the nucleus of the β-cell in response to changes in glucose, lipid, and cytokine levels. Foxo is detected by red fluorescence, insulin by green fluorescence. Nuclear translocation is associated with the activation of a stress response that aims to prevent loss of β-cell features through the genes indicated on the right-hand column. Nuclear Foxo1 is degraded if hyperglycemia is not reversed, paving the way for the activation of progenitor-like features and conversion into α-like–cells.
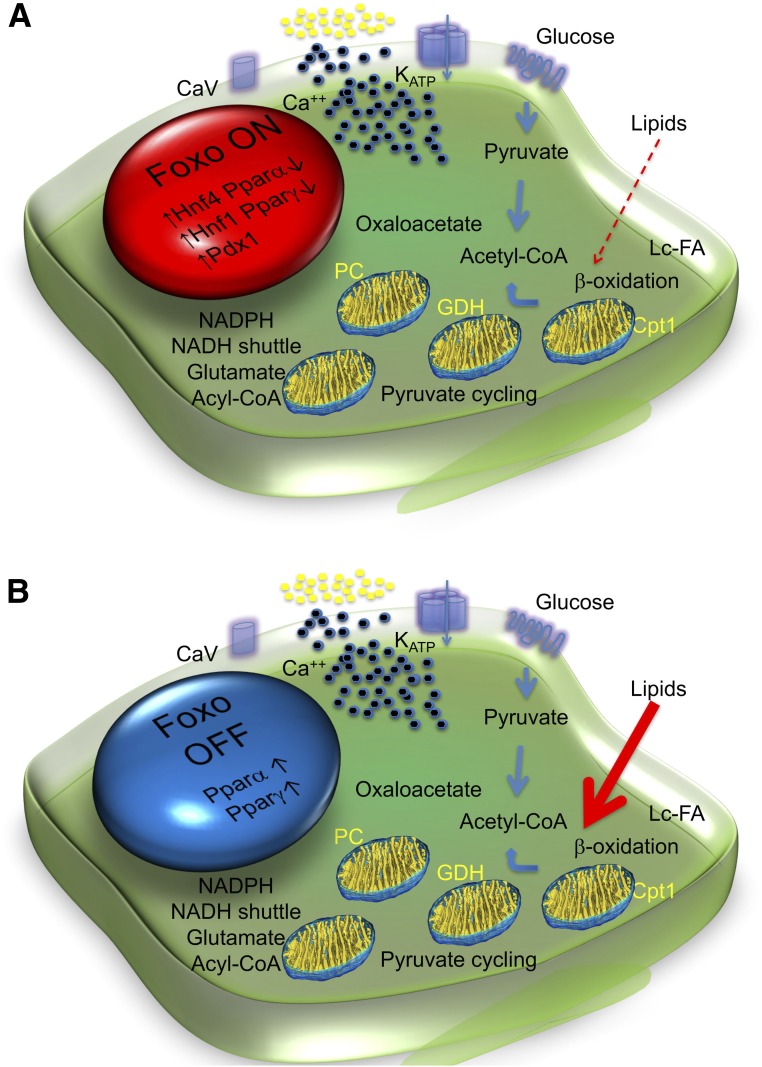
This model can best be understood in the physiologic context of insulin secretion (Fig. 5). The first phase is dependent on glucose phosphorylation, closure of ATP-dependent K channels, and secretion of stored, readily releasable insulin granules, whereas the second phase requires, in addition to energy generated through mitochondrial oxidative phosphorylation, various second messengers, including glutamate, NADPH, and acyl-CoA (44). The main source of substrate for mitochondrial oxidative phosphorylation is glucose, but amino acids and lipids also provide acetyl-CoA. We discovered that Foxo activation in response to metabolic challenges curtails fatty acid oxidation (Fig. 5A) (17,45,46). However, activated Foxo is rapidly degraded, leading to its depletion if the metabolic challenge does not subside (29). Once Foxo becomes “spent,” lipid oxidation rises. Increased lipid oxidation does not occur at the expense of glucose oxidation, which quantitatively accounts for >90% of ATP generation. Rather, the constitutive rise in lipid oxidation leads to the generation of toxic intermediates, with impaired ATP production, calcium mobilization, and insulin secretion (Fig. 5B). The exact nature of these “toxic” mediators remains to be determined, but there are many candidates in the literature (44).
Figure 5.
Metabolic inflexibility and β-cell failure. A: In response to factors requiring increased β-cell insulin secretion (transient hyperglycemia, increased lipids, inflammation, cytokines) in the early phases of diabetes, Foxo1 translocates to the nucleus to enforce a gene expression network that includes several maturity-onset diabetes of the young genes (e.g., Hnf4α, Hnf1α, Pdx1), allowing glucose flux into mitochondria for ATP production (thick arrows), while suppressing Pparα to limit lipid oxidation (thin dotted arrow). B: As Foxo1 becomes functionally exhausted, expression of maturity-onset diabetes of the young genes decreases and Pparα increases. β-Cells become transcriptionally blindsided to the effects of glucose, increasing lipid oxidation. Interestingly, Pparγ is also activated with Pparα. We interpret this as an attempt by the β-cell to divert carbons into lipid synthesis rather than oxidation. These data provide a potential explanation for the direct beneficial effects of thiazolidinediones on β-cell function. Cpt1, carnitine palmitoyl-transferase; GDH, glutamate dehydrogenase; Lc-FA, long chain fatty acids; PC, pyruvate carboxylase.
As a long-term consequence of metabolic inflexibility, β-cells gradually lose their terminally differentiated features. When we lineage-traced β-cells during diabetes progression, we obtained evidence that they do not die (at least, not immediately) but reverse their developmental pathway and come to resemble progenitor cells. Moreover, we learned that some of these former β-cells convert to glucagon-producing, α-like–cells, providing a potential explanation for the hyperglucagonemia of diabetes (30). We have now identified a subset of candidate genes that mediate the transition from a metabolically inflexible to a dedifferentiated β-cell (47).
We were immediately attracted to the notion that β-cells become dedifferentiated because it dovetails with the daily clinical reality of treating patients with diabetes. A slow-onset, demand-triggered process, β-cell dysfunction can be partly reversed for years on end after the onset of hyperglycemia (41,48). This revival is hardly compatible with a permanent loss of β-cells, although this is likely to occur after decades, when insulin secretion can no longer be restored. In fact, the β-cell’s regenerative abilities are as marginal as its ability to adopt different cell fates is remarkable (43). From an evolutionary standpoint, dedifferentiation serves the useful purpose of preserving precious and potentially irreplaceable β-cells “by stealth” (a concept that finds an interesting corollary in type 1 diabetes) rather than by the inefficient replication route. In a specific animal model, we could in fact recapitulate the beneficial effects of diet on β-cell dedifferentiation but failed—to our surprise—to recapitulate the expected effect of various drugs (49). This means that further mechanistic work is necessary to tackle dedifferentiation pharmacologically.
Dedifferentiation cannot be demonstrated in humans with the same level of mechanistic certainty as in genetically manipulated experimental animals (a truism that sadly bears repeating). With this proviso, we marshaled evidence of dedifferentiation as well as cell type conversion by analyzing human pancreata with a combination of immunohistochemical markers that demonstrate the presence of endocrine cells that no longer produce hormones and express markers of progenitor cells, as well as ectopic transcription factors. These cells are about fourfold more frequent than in nondiabetic pancreata (50). These data are consistent with an independent assessment of islet cell composition using single-cell gene expression profiling (J. Son and D.A., unpublished data).
Gut Is the New Pancreas
The introduction of gut biology into diabetes pathogenesis and treatment is among the important advances in recent metabolic research (51). Our insulin action research intersected with the biology of enteroendocrine hormones in a serendipitous manner. In the course of studies on the role of Foxo in endocrine progenitors (28,52), we discovered that genetically ablating Foxo1 from intestinal endocrine cells gave rise to β-like–cells that not only make insulin but also secrete it in a remarkably similar fashion to pancreatic β-cells. We showed that the newly arisen intestinal insulin-producing cells have all the markings of a “real” β-cell and that they can take over the functions of pancreatic β-cells after the latter have been destroyed with streptozotocin. These converted β-like–cells can effectively “cure” diabetes in mice (31). In hindsight, this observation should not have surprised us. Gut and pancreatic endocrine cells arise from Neurogenin3-expressing progenitors and give rise to similar types of endocrine cells in both organs, in addition to organ-specific cell types, such as α- and β-cells. Thus, enteroendocrine cells are the closest relatives of pancreatic endocrine cells.
An often-raised question is how can Foxo1 inactivation be possibly conducive to formation of β-cells in the gut and to loss of β-cells in the pancreas. Aren't these two functions incompatible? The answer is that these are two distinct processes. Physiologically, Foxo1 quiescence (i.e., cytoplasmic localization) is permissive for β-cell differentiation. And forced activation of Foxo1 promotes differentiation of endocrine progenitors into α-cells at the expense of β-cells (28). Thus, by genetic knockout of Foxo1, gut endocrine progenitors behave like pancreatic progenitors, giving rise to β-cells. But once β-cells have been made, Foxo1 preserves their stability. In theory, one would predict that gut-derived β-like–cells are less “resilient” than pancreatic β-cells. But this hardly matters given their short life span, as gut-derived β-like–cells are shed in the gut lumen with high turnover rates along with the gut epithelium in which they are embedded (31).
Human Relevance
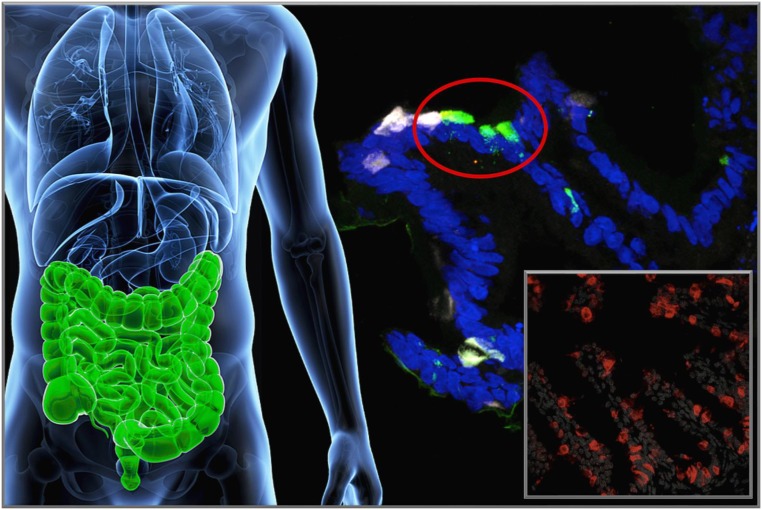
To demonstrate the feasibility of this approach in humans, we used human-induced pluripotent stem cell–derived gut organoid cultures, a system with substantial anatomical and functional similarities with the human gut, to test the conversion of enteroendocrine cells into insulin-producing cells. We showed that treating human gut organoid cultures with dominant-negative or shRNA Foxo1 inhibitors yields cells with β-like–cell features that make insulin and release it when challenged with glucose or sulfonylureas (Fig. 6) (53). Thus, the ability of gut endocrine cells to become insulin-producing cells is not confined to rodents, but it is a shared property of human gut cells. The human work also enabled us to pinpoint the likely cell type that undergoes conversion into β-like–cells. Of the nearly 20 different types of hormone-producing cells in the human gut, Foxo1 is expressed in serotonin-producing cells. (There is also a small [∼4%] percentage of somatostatin-producing cells that express Foxo1.) Interestingly, Foxo1 inhibition results in the loss of serotonin content in a majority of cells undergoing conversion into β-like–cells (53). We do not know if the two events are related, but it is intriguing that pancreatic insulin-producing β-cells also make serotonin (54), effectively making gut serotonin-producing cells the closest cell type to a pancreatic β-cell. This puts the original observation in rodents in an interesting perspective. It is possible that gut serotonin-producing cells are the evolutionary residue of insulin-producing cells in the pancreas and that we have restored an ancient function to these cells by inhibiting Foxo1 expression. We should note that we have not formally demonstrated (through lineage-tracing experiments) that conversion of gut endocrine cells to insulin-producing cells occurs at the terminal differentiation stage, when cells are already producing serotonin. The alternative explanation is that reprogramming occurs in Neurogenin3-positive endocrine progenitor cells (which also express Foxo1, but are much more difficult to track) to give rise to insulin-producing cells. This latter hypothesis would also be consistent with the murine data.
Figure 6.
Enteroendocrine cell conversion. Foxo1 inhibition leads to the generation of β-like–cells in the intestine, likely arising from the conversion of either serotonin-producing or endocrine progenitor cells. The main picture shows human-induced pluripotent stem–derived gut organoid cultures after treatment with a dominant-negative Foxo1. White fluorescence corresponds to serotonin, green fluorescence to insulin immunohistochemistry. The inset shows mouse intestinal cells converted into insulin-producing cells (red) after genetic knockout of Foxo1. Blue corresponds to DAPI nuclear staining.
Converting Enteroendocrine Cells to Treat Type 1 Diabetes
Converting gut endocrine cells into β-like–cells represents an approach to leverage intrinsic properties of an existing cell type in order to restore insulin production in patients lacking insulin, primarily people with type 1 diabetes but not excluding those with type 2 diabetes. We envision it not as a cell replacement or transplant approach but as a quasi-physiologic intervention, to the extent that we seek to tweak the features of an existing cell type to make it acquire those of a closely related cell type. Were one able to effect this change pharmacologically, there would be certain advantages to this approach that can be leveraged to treat type 1 diabetes. Enteroendocrine cells are plentiful and, unlike pancreatic endocrine cells, regenerate throughout life. In fact, unlike slow-turnover pancreatic endocrine cells, gut cells are replenished every 10–15 days. Enteroendocrine cells are poised to make and release hormones because they already possess the machinery required to sense nutrients and to make, process, and release peptides. Thus, they do not need to be reprogrammed from the ground up in order to generate β-like–cells. In addition, gut cells are readily accessible to orally administered drugs and can be targeted with minimal or no systemic exposure. Once converted into insulin-producing cells, our prediction is that these cells will be less immunogenic than the patient’s own pancreatic β-cells, owing to the gut immune privilege, which is partly stem cell niche mediated, and to their anatomical isolation and rapid turnover, which may make it easier for them to escape or outlast immune attack. We are testing these hypotheses while exploring this fascinating science in mechanistic detail. Our goal is to develop an orally available Foxo1 inhibitor that can convert gut endocrine cells in glucose-responsive insulin-producing β-like–cells.
Endgame
Research into the science of insulin action and pancreatic β-cell failure has yielded extraordinary results. We envision that they can be harnessed for the ambitious therapeutic goal of reversing and foreclosing diabetes, consigning the disease with its appurtenances to the museum of medical curiosities.
Article Information
Acknowledgments. The author is indebted to his mentors, Simeon I. Taylor, University of Maryland, Baltimore, MD, and Jesse Roth, Hofstra Northwell School of Medicine and Albert Einstein College of Medicine, Bronx, NY; to generations of students, fellows, and collaborators who have contributed to this work; and to the funders that have underwritten it: the American Diabetes Association, JDRF, the Russell Berrie Foundation, and the U.S. National Institutes of Health, especially the National Institute of Diabetes and Digestive and Kidney Diseases. The author thanks his colleague Rebecca Haeusler, Columbia University, New York, NY, for her critical reading of the manuscript. The list of references is meant to summarize the author’s laboratory contributions rather than conveying a comprehensive account of the literature. The author regrets the omissions.
Duality of Interest. D.A. is a cofounder, director, and chairman of the board of advisors of Forkhead BioTherapeutics, Inc., a company that aims to develop Foxo1 inhibitors as a diabetes treatment. No other potential conflicts of interest relevant to this article were reported.
Footnotes
The Banting Medal for Scientific Achievement Lecture was presented at the American Diabetes Association’s 77th Scientific Sessions in San Diego, CA, on Sunday, 11 June 2017.
References
- 1.Pajvani UB, Accili D. The new biology of diabetes. Diabetologia 2015;58:2459–2468 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cefalu WT, Kaul S, Gerstein HC, et al. Cardiovascular outcomes trials in type 2 diabetes: where do we go from here? Reflections from a Diabetes Care Editors’ Expert Forum. Diabetes Care 2018;41:14–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Samuel VT, Shulman GI. The pathogenesis of insulin resistance: integrating signaling pathways and substrate flux. J Clin Invest 2016;126:12–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Davidson MA, Mattison DR, Azoulay L, Krewski D. Thiazolidinedione drugs in the treatment of type 2 diabetes mellitus: past, present and future. Crit Rev Toxicol 2018;48:52–108 [DOI] [PubMed] [Google Scholar]
- 5.Haeusler RA, McGraw TE, Accili D. Biochemical and cellular properties of insulin receptor signalling. Nat Rev Mol Cell Biol 2018;19:31–44 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kim-Muller JY, Accili D. Cell biology. Selective insulin sensitizers. Science 2011;331:1529–1531 [DOI] [PubMed] [Google Scholar]
- 7.Accili D, Arden KC. FoxOs at the crossroads of cellular metabolism, differentiation, and transformation. Cell 2004;117:421–426 [DOI] [PubMed] [Google Scholar]
- 8.White MF. Insulin signaling in health and disease. Science 2003;302:1710–1711 [DOI] [PubMed] [Google Scholar]
- 9.O’Brien RM, Granner DK. Regulation of gene expression by insulin. Physiol Rev 1996;76:1109–1161 [DOI] [PubMed] [Google Scholar]
- 10.Dong XC, Copps KD, Guo S, et al. Inactivation of hepatic Foxo1 by insulin signaling is required for adaptive nutrient homeostasis and endocrine growth regulation. Cell Metab 2008;8:65–76 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Matsumoto M, Pocai A, Rossetti L, Depinho RA, Accili D. Impaired regulation of hepatic glucose production in mice lacking the forkhead transcription factor Foxo1 in liver. Cell Metab 2007;6:208–216 [DOI] [PubMed] [Google Scholar]
- 12.Kubota N, Kubota T, Itoh S, et al. Dynamic functional relay between insulin receptor substrate 1 and 2 in hepatic insulin signaling during fasting and feeding. Cell Metab 2008;8:49–64 [DOI] [PubMed] [Google Scholar]
- 13.Lu M, Wan M, Leavens KF, et al. Insulin regulates liver metabolism in vivo in the absence of hepatic Akt and Foxo1. Nat Med 2012;18:388–395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nakae J, Biggs WH 3rd, Kitamura T, et al. Regulation of insulin action and pancreatic beta-cell function by mutated alleles of the gene encoding forkhead transcription factor Foxo1. Nat Genet 2002;32:245–253 [DOI] [PubMed] [Google Scholar]
- 15.Kitamura T, Nakae J, Kitamura Y, et al. The forkhead transcription factor Foxo1 links insulin signaling to Pdx1 regulation of pancreatic beta cell growth. J Clin Invest 2002;110:1839–1847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Haeusler RA, Hartil K, Vaitheesvaran B, et al. Integrated control of hepatic lipogenesis versus glucose production requires FoxO transcription factors. Nat Commun 2014;5:5190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim-Muller JY, Zhao S, Srivastava S, et al. Metabolic inflexibility impairs insulin secretion and results in MODY-like diabetes in triple FoxO-deficient mice. Cell Metab 2014;20:593–602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kitamura T, Feng Y, Kitamura YI, et al. Forkhead protein FoxO1 mediates Agrp-dependent effects of leptin on food intake. Nat Med 2006;12:534–540 [DOI] [PubMed] [Google Scholar]
- 19.Plum L, Lin HV, Dutia R, et al. The obesity susceptibility gene Cpe links FoxO1 signaling in hypothalamic pro-opiomelanocortin neurons with regulation of food intake. Nat Med 2009;15:1195–1201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ren H, Orozco IJ, Su Y, et al. FoxO1 target Gpr17 activates AgRP neurons to regulate food intake. Cell 2012;149:1314–1326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pajvani UB, Qiang L, Kangsamaksin T, Kitajewski J, Ginsberg HN, Accili D. Inhibition of Notch uncouples Akt activation from hepatic lipid accumulation by decreasing mTorc1 stability. Nat Med 2013;19:1054–1060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pajvani UB, Shawber CJ, Samuel VT, et al. Inhibition of Notch signaling ameliorates insulin resistance in a FoxO1-dependent manner. Nat Med 2011;17:961–967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Matsumoto M, Han S, Kitamura T, Accili D. Dual role of transcription factor FoxO1 in controlling hepatic insulin sensitivity and lipid metabolism. J Clin Invest 2006;116:2464–2472 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chakrabarti P, Kandror KV. FoxO1 controls insulin-dependent adipose triglyceride lipase (ATGL) expression and lipolysis in adipocytes. J Biol Chem 2009;284:13296–13300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tsuchiya K, Banks AS, Liang CP, Tabas I, Tall AR, Accili D. Homozygosity for an allele encoding deacetylated FoxO1 protects macrophages from cholesterol-induced inflammation without increasing apoptosis. Arterioscler Thromb Vasc Biol 2011;31:2920–2928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tsuchiya K, Tanaka J, Shuiqing Y, et al. FoxOs integrate pleiotropic actions of insulin in vascular endothelium to protect mice from atherosclerosis. Cell Metab 2012;15:372–381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tsuchiya K, Westerterp M, Murphy AJ, et al. Expanded granulocyte/monocyte compartment in myeloid-specific triple FoxO knockout increases oxidative stress and accelerates atherosclerosis in mice. Circ Res 2013;112:992–1003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kitamura T, Kitamura YI, Kobayashi M, et al. Regulation of pancreatic juxtaductal endocrine cell formation by FoxO1. Mol Cell Biol 2009;29:4417–4430 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kitamura YI, Kitamura T, Kruse JP, et al. FoxO1 protects against pancreatic beta cell failure through NeuroD and MafA induction. Cell Metab 2005;2:153–163 [DOI] [PubMed] [Google Scholar]
- 30.Talchai C, Xuan S, Lin HV, Sussel L, Accili D. Pancreatic β cell dedifferentiation as a mechanism of diabetic β cell failure. Cell 2012;150:1223–1234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Talchai C, Xuan S, Kitamura T, DePinho RA, Accili D. Generation of functional insulin-producing cells in the gut by Foxo1 ablation. Nat Genet 2012;44:406-412 [DOI] [PMC free article] [PubMed]
- 32.O’Neill BT, Lee KY, Klaus K, et al. Insulin and IGF-1 receptors regulate FoxO-mediated signaling in muscle proteostasis. J Clin Invest 2016;126:3433–3446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kitamura T, Kitamura YI, Funahashi Y, et al. A Foxo/Notch pathway controls myogenic differentiation and fiber type specification. J Clin Invest 2007;117:2477–2485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lin HV, Accili D. Hormonal regulation of hepatic glucose production in health and disease. Cell Metab 2011;14:9–19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tsuchiya K, Accili D. Liver sinusoidal endothelial cells link hyperinsulinemia to hepatic insulin resistance. Diabetes 2013;62:1478–1489 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Staels B, Handelsman Y, Fonseca V. Bile acid sequestrants for lipid and glucose control. Curr Diab Rep 2010;10:70–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Haeusler RA, Pratt-Hyatt M, Welch CL, Klaassen CD, Accili D. Impaired generation of 12-hydroxylated bile acids links hepatic insulin signaling with dyslipidemia. Cell Metab 2012;15:65–74 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Haeusler RA, Astiarraga B, Camastra S, Accili D, Ferrannini E. Human insulin resistance is associated with increased plasma levels of 12α-hydroxylated bile acids. Diabetes 2013;62:4184–4191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Langlet F, Haeusler RA, Linden D, et al. Selective inhibition of FOXO1 activator/repressor balance modulates hepatic glucose handling. Cell 2017;171:824-835.e18 [DOI] [PMC free article] [PubMed]
- 40.Ferrannini E. The stunned beta cell: a brief history. Cell Metab 2010;11:349–352 [DOI] [PubMed] [Google Scholar]
- 41.Lean ME, Leslie WS, Barnes AC, et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet 2018;391:541–551 [DOI] [PubMed] [Google Scholar]
- 42.Lin HV, Ren H, Samuel VT, et al. Diabetes in mice with selective impairment of insulin action in Glut4-expressing tissues. Diabetes 2011;60:700–709 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Accili D, Talchai SC, Kim-Muller JY, et al. When β-cells fail: lessons from dedifferentiation. Diabetes Obes Metab 2016;18(Suppl. 1):117–122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Seino S, Sugawara K, Yokoi N, Takahashi H. β-Cell signalling and insulin secretagogues: a path for improved diabetes therapy. Diabetes Obes Metab 2017;19(Suppl. 1):22–29 [DOI] [PubMed] [Google Scholar]
- 45.Kim-Muller JY, Kim YJ, Fan J, et al. Foxo1 deacetylation decreases fatty acid oxidation in β-cells and sustains insulin secretion in diabetes. J Biol Chem 2016;291:10162–10172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Buteau J, Shlien A, Foisy S, Accili D. Metabolic diapause in pancreatic beta-cells expressing a gain-of-function mutant of the forkhead protein Foxo1. J Biol Chem 2007;282:287–293 [DOI] [PubMed] [Google Scholar]
- 47.Kim-Muller JY, Fan J, Kim YJ, et al. Aldehyde dehydrogenase 1a3 defines a subset of failing pancreatic β cells in diabetic mice. Nat Commun 2016;7:12631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Savage PJ, Bennion LJ, Flock EV, et al. Diet-induced improvement of abnormalities in insulin and glucagon secretion and in insulin receptor binding in diabetes mellitus. J Clin Endocrinol Metab 1979;48:999–1007 [DOI] [PubMed] [Google Scholar]
- 49.Ishida E, Kim-Muller JY, Accili D. Pair feeding, but not insulin, phloridzin, or rosiglitazone treatment, curtails markers of β-cell dedifferentiation in db/db mice. Diabetes 2017;66:2092–2101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cinti F, Bouchi R, Kim-Muller JY, et al. Evidence of β-Cell dedifferentiation in human type 2 diabetes. J Clin Endocrinol Metab 2016;101:1044–1054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Drucker DJ. The cardiovascular biology of glucagon-like peptide-1. Cell Metab 2016;24:15–30 [DOI] [PubMed] [Google Scholar]
- 52.Talchai SC, Accili D. Legacy effect of Foxo1 in pancreatic endocrine progenitors on adult β-cell mass and function. Diabetes 2015;64:2868–2879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bouchi R, Foo KS, Hua H, et al. FOXO1 inhibition yields functional insulin-producing cells in human gut organoid cultures. Nat Commun 2014;5:4242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kim H, Toyofuku Y, Lynn FC, et al. Serotonin regulates pancreatic beta cell mass during pregnancy. Nat Med 2010;16:804–808 [DOI] [PMC free article] [PubMed] [Google Scholar]