The Ebola outbreak
As a new outbreak of Ebola virus has recently taken place, it is vital that scientists take several steps towards its combat. The Ebola Hemorrhagic Fever outbreak of 2014 is listed as the biggest one in history with almost 9000 reported human cases and a 53% rate of fatality. The Ebola virus is classified in the genus of Ebolovirus, in the family of Filoviridae and order Mononegavirales (Kuhn et al. 2010). Five viruses of this class are known to be disease-causing in humans; Zaire ebolavirus (EBOV), Sudan ebolavirus (SUDV), Reston ebolavirus (RESTV), Côte d’Ivoire ebolavirus (TAFV) and Uganda ebolavirus (BDBV). In the case of the current outbreak, all listed cases have been infected by EBOV. The latter is the most lethal of all. It was named after the fact that the first formal record was made in Zaire in 1976, presenting an 88% rate of fatality; even though 2014’s outbreak is estimated to be the deadliest Ebola outbreak by far (Gire et al. 2014).
Since 1976, humanity has been compelled to face Ebola’s disease cases on an approximately annual basis. At the beginning of the appearance of the disease, there were cases caused by all viruses’ species. However, with the passage of time the viruses with the low fatality rate disappeared. Is this a phenomenon of natural selection or an artificial outcome? Can we diagnose any pattern in this statistical data?
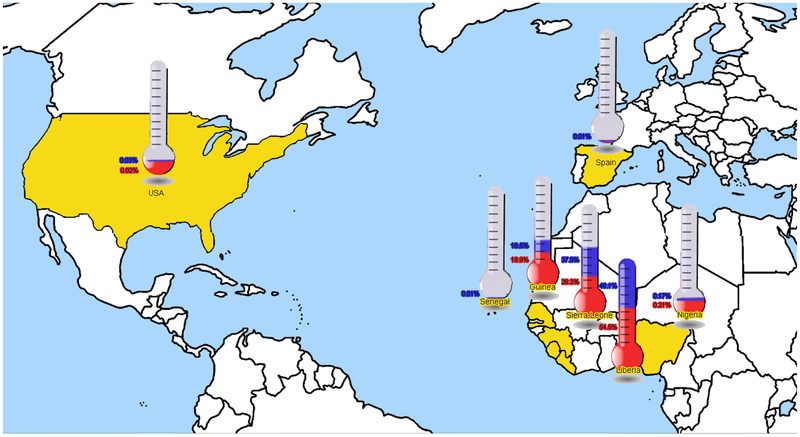
Additionally, the mortality rate has always increased in underdeveloped countries, probably due to the lack of an organized health care system. During all these years the number of human cases has been fluctuating between 1 and 500. The Ebola’s outbreak, which started in Guinea in December 2013 and spread among Liberia, Sierra Leone, Nigeria, Senegal and almost all the West African countries, admeasures 8469 human cases to date (Figure 1). The epidemic soon spread in the Democratic Republic of Congo and is still ongoing, with scientists predicting that the cases could reach the number of 1.4 million by January (Prevention, October 13, 2014).
Figure 1.
World prevalence of Ebola outbreak. Data are totals and only include confirmed cases (colored blue) and case deaths (colored red). Data are based on official information reported by the Ministries of Health up to October 14 2014 (WHO, 2014).
Europe and the United States have had the confidence that Ebola will not spread among their territory. Nevertheless, on September 30, the first Ebola case was diagnosed in Dallas, Texas (McCarthy 2014). As for the European countries, the first recorded human case with Ebola was a Spanish missionary who contracted the virus in Liberia (Gulland 2014). Germany has also recorded the third patient of this deadly disease and the first human death on October 14. According to the World Health Organization (WHO) in August 2014, 10241 travelers originating from Sierra Leone, Guinea and Liberia travelled to European countries. In addition, large numbers of migrant workers from the outbreak area are located within Europe and the US.
The epidemic has not made its presence felt in Greece yet. However, Reuters reported a scientific research that predicts a 75 percent chance of the virus being imported into France and a 50 percent chance into Great Britain by the end of October (Gomes, Sep 2014). Ebola outbreak is expected to accelerate in October 2014 with the number of projected cases touching the 14000.
Nevertheless, researchers are absolutely confident that Europe and America will not experience the nightmare of Ebola’s outbreak. This seems justifiable, as nearly all of the human cases in developed countries have been healed. Whoever seeks conspiracy theories may claim that these countries already have the cure and that what is taking place in Africa is merely an experiment. However, hereafter may be a more logical explanation to oppose these theories.
Nowadays the media are flooded with images of how humanity deals with the Ebola virus. It is obvious that the range of Ebola cases depends on the preventive and dealing measures (Muyembe-Tamfum et al. 1999). In developing countries, due to the lack of education, expertise and health care, there is a high transmission and mortality rate. Doctors there do not have the appropriate medical equipment. They are not able to run full tests. They do not have the expertise to treat this epidemic or even to control its spread. People diagnosed with Ebola should be kept in quarantine individually, but in West Africa there are barely any main hospital units. As a result of these, if scientists have the slightest suspicion of the virus infection in people “they imprison them in the same cage” (Fowler et al. 2014).
Culture and traditions can sometimes also play dramatic roles towards this direction. For instance, several funeral and/or burial rituals require physical contact with the body of the deceased, even if he/she has died from Ebola. Physical contact is a crucial communication code among west Africans (Dowell et al. 1999). Religious beliefs could also aid in this direction.
On the other hand, developed countries have all the resources to avoid this epidemic. People can get informed on how to protect themselves and disease symptoms are widely known. They have the necessary educational level in order to resort immediately to medical aid protecting their health and not putting others in risk. In Europe and America, there are special ized treatment and hemodialysis units in hospitals. These countries also have the financial background to support expensive patient medication (Nagata 2014).
The origin of Ebola
Another subject that may cause a plethora of arguments is that this virus may be a laboratory generated virus. It may be assumed that all these could be a conspiracy or scaremongering theory. Nevertheless, scientists do not know much about this virus except a few basic characteristics. The virus depletes the body’s immune cells and its organs (Sullivan et al. 2011). Notwithstanding the high fatality rates, there are some people who survive and recover completely from EVD’s infections. Furthermore, several research projects have led to the conclusion that some people may be explicitly resistant to Ebola infections due to a mutation in the NPC1 gene (Cote et al. 2011). Yet, we barely know anything about this virus.
For example, it is not completely clear how Ebola is transmitted. There is a conjecture that the virus is transmitted to people from wild animals. However, by reason of the high mortality among them, it is impossible that these animals are the reservoir host of EVD. The conviction is that fruit bats of the pteropodidae family are natural Ebola virus hosts even though the reservoir of EVD has not yet been identified (Groseth et al. 2007). It is also believed that, among humans, EVD can be disseminated through direct contact with blood or body fluids such as sweat, urine, breast milk, saliva and vomit of an infected person (Francesconi et al. 2003).
There are some not well known dark sides of EVD though, such as the potential use in biological warfare. This encompasses the use of biological infectious agents as weapons in order to nullify or exterminate humans, animals or plants. The target can range from one single individual to an entire population. Based on the merits, biological warfare represents an act of an invisible war. In these battlefields there is no need of troop reinforcements or heavy weapons. These wars do not require colossal financial resources, due to the fact that a biological weapon acts underground and it can be transferred easily through water, through the ground or through the air without being perceived by anybody.
So the ability to disseminate disease through aerosol or airborne small-droplet nuclei would render filoviruses a potential biologic warfare threat (Salvaggio & Baddley 2004). According to some researchers who have been involved with Soviet biological weapons, Ebola and some other viruses have been already weaponized in this manner by the former Soviet Union (Miller et al. 2002). Aerosol transmission of EVD has been demonstrated in experimental models involving nonhuman primates (Borio et al. 2002). During an experiment to evaluate the benefits of interferon treatment following infection with EBO-Z, two out of three control rhesus monkeys became ill. Researchers were unable to document direct or percutaneous contact through injections, and speculated that inoculation to the control animals occurred via pulmonary, nasopharyngeal, oral or conjunctival routes (Jaax et al. 1995). Definitive evidence that small-droplet nuclei pose a substantial transmission risk among humans during naturally occurring outbreaks is lacking. After making a quick comparison of the number of cases between this outbreak and previous ones, one could argue that something is different this time (Salvaggio & Baddley 2004). Is this epidemic disease a genuine natural disaster or something else?
Diagnosis, treatment and EBOV proteins
In accordance with what is known so far, the symptoms occur between 2 to 21 days after the infection. Typically, early symptoms include fever, headache, weakness, stomach disorders, joint and muscle pains (Bwaka et al. 1999). At a later stage, the patient may present cough, breathing difficulties, hiccups, bloodshot eyes and internal-external bleeding. There is no treatment or vaccines to date. Hence, every research project on this virus will constitute a significant progress towards the identification of Ebola’s mechanisms.
The diagnosis of Ebola’s infection is labor-intensive, as its symptoms are similar to other diseases, such as cholera and malaria. Even the symptoms of a common flu can be confused with those of Ebola. Diagnostic methods include CBC, electrolytes, blood tests, liver tests and virus-specific antibodies (Tigabu et al. 2009). The genome of EBOV is roughly 19 kb long. It encodes seven structural proteins; RNA polymerase, nucleoprotein (NP), polymerase cofactors VP35 and VP40, GP as well as transcription activators VP30 and VP24 (Huang et al. 2002). It has been shown from a plethora of research projects that this protein is an antagonist of type one interferons and that VP35 interacts with the IRF-3 kinases IKKε and TBK-1. Its function is to prevent the establishment of a cellular antiviral state by blocking virus-induced phosphorylation and triggering the activation of the interferon regulatory factor 3 (IRF3) (Basler et al. 2003). IRF3 is a transcription factor which plays a significant role in the induction of interferons alpha and beta (IFN-α/β).VP35 possesses a carboxy-terminal domain with a unique fold that allows double-stranded RNA (dsRNA) binding (Cardenas et al. 2006). This may be a necessary function for the inhibition of IFN-α/β production. Furthermore, VP35 mutants missing the coiled-coil motif or carrying a mutation designed to disrupt coiled-coil function were defective in the formation of an oligomer. Of note, VP35 is also related with the H1N1 and Margburg virus (Basler et al. 2000). In contrast, we do not know if VP35 is phosphorylated, although it is thought to share the function of the phosphorylated proteins of rhabdoviruses and paramyxoviruses due to its position in the genome.
Future directions
The full crystal structure of the Zaire EBOV VP35 is not available yet. However, only recently the structure of the Zaire EBOV VP35 interferon inhibitory domain (IID) has been established. In the free form the Zaire EBOV VP35 IID bounds to dsRNA. VP35 IID forms a unique fold with 2 basic residue clusters. One of these clusters is important for dsRNA binding (Leung et al. 2010). The cluster of dsRNA that function to bind is centered on Arg-312. It is a required conserved residue for IFN inhibition. The C-terminal cluster of basic amino acids of Ebola virus VP35 is a prerequisite in order to bind the dsRNA with the interferon regulatory factor 3 (IRF3) and inhibit its functionality. Finally, VP35 is assumed to block and reverse the activation of protein kinase R (PKR).
There is a dear need for a full structural 3D model of VP35 (Carvalho et al. 2013, Dalkas et al. 2013, Vlachakis et al. 2014, Vlachakis & Kossida, 2013). There are preliminary indications of allosteric sites on its structural domains, which could be exploited for the designing customized drug-like inhibitors (Vlachakis et al. 2014, Loukatou et al. 2014, Papangelopoulos et al. 2014, Papageorgiou et al. 2014, Sellis et al. 2009, Vangelatos et al. 2009). Funds must be allocated towards the functional and biochemical characterization of viral proteins such as VP35, which constitute necessary machinery for Ebola’s virulence. It is absolutely crucial to dig deeper, investigate and understand the molecular mechanisms, which underlie the rather complicated nature of this viral species.
Footnotes
Conficts of Interest
The authors declare no conflicts of interest.
References
- Basler CF, Mikulasova A, Martinez-Sobrido L, Paragas J, Muhlberger E, Bray M, Klenk HD, Palese P & Garcia-Sastre A 2003. The Ebola virus VP35 protein inhibits activation of interferon regulatory factor 3. J Virol 77 7945–7956 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basler CF, Wang X, Muhlberger E, Volchkov V, Paragas J, Klenk HD, Garcia-Sastre A & Palese P 2000. The Ebola virus VP35 protein functions as a type I IFN antagonist. Proc Natl Acad Sci U S A 97 12289–12294 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borio L, Inglesby T, Peters CJ, Schmaljohn AL, Hughes JM, Jahrling PB, Ksiazek T, Johnson KM, Meyerhoff A, O’Toole T, Ascher MS, Bartlett J, Breman JG, Eitzen EM Jr, Hamburg M, Hauer J, Henderson DA, Johnson RT, Kwik G, Layton M, Lillibridge S, Nabel GJ, Osterholm MT, Perl TM, Russell P, Tonat K & Working Group on Civilian Biodefense 2002. Hemorrhagic fever viruses as biological weapons: Medical and public health management. JAMA 287 2391–2405 [DOI] [PubMed] [Google Scholar]
- Bwaka MA, Bonnet MJ, Calain P, Colebunders R, De Roo A, Guimard Y, Katwiki KR, Kibadi K, Kipasa MA, Kuvula KJ, Mapanda BB, Massamba M, Mupapa KD, Muyembe-Tamfum JJ, Ndaberey E, Peters CJ, Rollin PE, Van den Enden E & Van den Enden E 1999. Ebola hemorrhagic fever in Kikwit, Democratic Republic of the Congo: clinical observations in 103 patients. J Infect Dis 179 S1–7 [DOI] [PubMed] [Google Scholar]
- Cardenas WB, Loo YM, Gale M Jr, Hartman AL, Kimberlin CR, Martinez-Sobrido L, Saphire EO & Basler CF 2006. Ebola virus VP35 protein binds double-stranded RNA and inhibits alpha/beta interferon production induced by RIG-I signalling. J Virol 80 5168–5178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carvalho CS, Vlachakis D, Tsiliki G, Megalooikonomou V & Kossida S 2013. Protein signatures using electrostatic molecular surfaces in harmonic space PeerJ 1 e185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cote M, Misasi J, Ren T, Bruchez A, Lee K, Filone CM, Hensley L, Li Q, Ory D, Chandran K & Cunningham J 2011. Small molecule inhibitors reveal Niemann-Pick C1 is essential for Ebola virus infection. Nature 477 344–348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dalkas GA, Vlachakis D, Tsagkrasoulis D, Kastania A & Kossida S 2013. State-of-the-art technology in modern computer-aided drug design. Brief Bioinform 14 745–752 [DOI] [PubMed] [Google Scholar]
- Dowell SF, Mukunu R, Ksiazek TG, Khan AS, Rollin PE & Peters CJ 1999. Transmission of Ebola hemorrhagic fever: a study of risk factors in family members, Kikwit, Democratic Republic of the Congo, 1995. Commission de Lutte contre les Epidemies a Kikwit. J Infect Dis 179 S87–91 [DOI] [PubMed] [Google Scholar]
- Fowler RA, Fletcher T, Fischer WA 2nd, Lamontagne F, Jacob S, Brett-Major D, Lawler JV, Jacquerioz FA, Houlihan C, O’Dempsey T, Ferri M, Adachi T, Lamah MC, Bah EI, Mayet T, Schieffelin J, McLellan SL, Senga M, Kato Y, Clement C, Mardel S, Vallenas Bejar De Villar RC, Shindo N & Bausch D 2014. Caring for critically ill patients with ebola virus disease. Perspectives from west Africa. Am J Respir Crit Care Med 190 733–737 [DOI] [PubMed] [Google Scholar]
- Francesconi P, Yoti Z, Declich S, Onek PA, Fabiani M, Olango J, Andraghetti R, Rollin PE, Opira C, Greco D & Salmaso S 2003. Ebola hemorrhagic fever transmission and risk factors of contacts, Uganda. Emerg Infect Dis 9 1430–1437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gire SK, Goba A, Andersen KG, Sealfon RS, Park DJ, Kanneh L, Jalloh S, Momoh M, Fullah M, Dudas G, Wohl S, Moses LM, Yozwiak NL, Winnicki S, Matranga CB, Malboeuf CM, Qu J, Gladden AD, Schaffner SF, Yang X, Jiang PP, Nekoui M, Colubri A, Coomber MR, Fonnie M, Moigboi A, Gbakie M, Kamara FK, Tucker V, Konuwa E, Saffa S, Sellu J, Jalloh AA, Kovoma A, Koninga J, Mustapha I, Kargbo K, Foday M, Yillah M, Kanneh F, Robert W, Massally JL, Chapman SB, Bochicchio J, Murphy C, Nusbaum C, Young S, Birren BW, Grant DS, Scheiffelin JS, Lander ES, Happi C, Gevao SM, Gnirke A, Rambaut A, Garry RF, Khan SH & Sabeti PC 2014. Genomic surveillance elucidates Ebola virus origin and transmission during the 2014 outbreak. Science 345 1369–1372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomes MFC, Pastore y Piontti A, Rossi L, Chao D, Longini I, Halloran ME & Vespignani A 2014. Assessing the International Spreading Risk Associated with the 2014 West African Ebola Outbreak. PLOS Cur Outbreak Edition 1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Groseth A, Feldmann H & Strong JE 2007. The ecology of Ebola virus. Trends Microbiol 15 408–416 [DOI] [PubMed] [Google Scholar]
- Gulland A 2014. Spanish authorities investigate how nurse contracted Ebola. BMJ 349 g6120. [DOI] [PubMed] [Google Scholar]
- Huang Y, Xu L, Sun Y & Nabel GJ 2002. The assembly of Ebola virus nucleocapsid requires virion-associated proteins 35 and 24 and posttranslational modification of nucleoprotein Mol Cell 10 307–316 [DOI] [PubMed] [Google Scholar]
- Jaax N, Jahrling P, Geisbert T, Geisbert J, Steele K, McKee K, Nagley D, Johnson E, Jaax G & Peters C 1995. Transmission of Ebola virus (Zaire strain) to uninfected control monkeys in a biocontainment laboratory. Lancet 346 1669–1671 [DOI] [PubMed] [Google Scholar]
- Kuhn JH, Becker S, Ebihara H, Geisbert TW, Johnson KM, Kawaoka Y, Lipkin WI, Negredo AI, Netesov SV, Nichol ST, Palacios G, Peters CJ, Tenorio A, Volchkov VE & Jahrling PB 2010. Proposal for a revised taxonomy of the family Filoviridae: classification, names of taxa and viruses, and virus abbreviations Arch Virol 155 2083–2103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung DW, Prins KC, Borek DM, Farahbakhsh M, Tufariello JM, Ramanan P, Nix JC, Helgeson LA, Otwinowski Z, Honzatko RB, Basler CF & Amarasinghe GK 2010. Structural basis for dsRNA recognition and interferon antagonism by Ebola VP35. Nat Struct Mol Biol 17 165–172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loukatou S, Papageorgiou L, Fakourelis P, Filtisi A, Polychronidou E, Bassis I, Megalooikonomou V, Makalowski W, Vlachakis D & Kossida S 2014. Molecular dynamics simulations through GPU video games technologies. J Mol Biochem 3 64–71 [PMC free article] [PubMed] [Google Scholar]
- McCarthy M 2014. Liberian man being treated for Ebola in Texas dies. BMJ 349 g6145. [DOI] [PubMed] [Google Scholar]
- Miller J, Engelberg S & Broad WJ 2002. Germs: Biological Weapons and America’s Secret War: Simon & Schuster [Google Scholar]
- Muyembe-Tamfum JJ, Kipasa M, Kiyungu C & Cole-bunders R 1999. Ebola outbreak in Kikwit, Democratic Republic of the Congo: discovery and control measures. J Infect Dis 179 S259–262 [DOI] [PubMed] [Google Scholar]
- Nagata N 2014. International Control of Epidemic Diseases from a Historical and Cultural Perspective In Herren M, ed, Networking the International System, pp. 73–88. Springer International Publishing. [Google Scholar]
- Prevention C. f. D. C. a. (October 13, 2014). Outbreaks Chronology: Ebola Virus Disease.
- Papangelopoulos N, Vlachakis D, Filtisi A, Fakourelis P, Papageorgiou L, Megalooikonomou V & Kossida S 2014. State of the art GPGPU applications in bioinformatics Int J Sys Biol Biomed Tech 2 24–48 [Google Scholar]
- Papageorgiou L, Vlachakis D, Koumandou VL, Papagelopoulos N & Kossida S 2014. Computer-aided drug design and biological evaluation of novel anti-Greek goat encephalitis agents. Int J Sys Biol Biomed Tech 2 1–16 [Google Scholar]
- Salvaggio MR & Baddley JW 2004. Other viral bioweapons: Ebola and Marburg hemorrhagic fever. Dermatol Clin 22 291–302 [DOI] [PubMed] [Google Scholar]
- Sellis D, Vlachakis D & Vlassi M 2009. Gromita: a fully integrated graphical user interface to gromacs 4. Bioinform Biol Ins 3 99–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan NJ, Hensley L, Asiedu C, Geisbert TW, Stanley D, Johnson J, Honko A, Olinger G, Bailey M, Geisbert JB, Reimann KA, Bao S, Rao S, Roederer M, Jahrling PB, Koup RA & Nabel GJ 2011. CD8+ cellular immunity mediates rAd5 vaccine protection against Ebola virus infection of nonhuman primates. Nat Med 17 1128–1131 [DOI] [PubMed] [Google Scholar]
- Tigabu B, Juelich T, Bertrand J & Holbrook MR 2009. Clinical evaluation of highly pathogenic tick-borne flavivirus infection in the mouse model. J Med Virol 81 1261–1269 [DOI] [PubMed] [Google Scholar]
- Vangelatos I, Vlachakis D, Sophianopoulou V & Dial-linas G 2009. Modelling and mutational evidence identify the substrate binding site and functional elements in APC amino acid transporters. Mol Membrane Biol 26 356–370 [DOI] [PubMed] [Google Scholar]
- Vlachakis D, Bencurova E, Papangelopoulos N & Kossida S 2014. Current State-of-the-Art Molecular Dynamics Methods and Applications. Adv Protein Chem Struct Biol 94 269–313 [DOI] [PubMed] [Google Scholar]
- Vlachakis D & Kossida S 2013. Molecular modeling and pharmacophore elucidation study of the Classical Swine Fever virus helicase as a promising pharmacological target. PeerJ 1 e85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vlachakis D, Pavlopoulou A, Roubelakis MG, Feidakis C, Anagnou NP & Kossida S 2014. 3D molecular modeling and evolutionary study of the Trypanosoma brucei DNA Topoisomerase IB, as a new emerging pharmacological target. Genomics 103 107–113. [DOI] [PubMed] [Google Scholar]