Abstract
Acute hepatopancreatic necrosis disease (AHPND) has extended rapidly, causing alarming shrimp mortalities. Initially, the only known causative agent was Vibrio parahaemolyticus carrying a plasmid coding for the mortal toxins PirVP. Recently, it has been found that the plasmid and hence the disease, could be transferred among members of the Harveyi clade. The current study performs a genomic characterization of an isolate capable of developing AHPND in shrimp. Mortality studies and molecular and histopathological analyses showed the infection capacity of the strain. Multilocus sequence analysis placed the bacteria as a member of the Orientalis clade, well known for containing commensal and even probiotic bacteria used in the shrimp industry. Further whole genome comparative analyses, including Vibrio species from the Orientalis clade, and phylogenomic metrics (TETRA, ANI and DDH) showed that the isolate belongs to a previously unidentified species, now named Vibrio punensis sp. nov. strain BA55. Our findings show that the gene transfer capacity of Vibrio species goes beyond the clade classification, demonstrating a new pathogenic capacity to a previously known commensal clade. The presence of these genes in a different Vibrio clade may contribute to the knowledge of the Vibrio pathogenesis and has major implications for the spread of emerging diseases.
Introduction
Shrimp production in Southeast Asia and Mexico had increased steadily in recent years, until the outbreak of a new disease, called acute hepatopancreatic necrosis disease (AHPND) or early mortality syndrome (EMS). This disease caused severe harm to the industry, resulting in significant economic loss to shrimp producers in China (2009), Vietnam (2010), Malaysia (2011), Thailand (2012), Mexico (2013) and Philippines (2015)1,2. The disease can affect both species of cultured penaeid shrimp, Penaeus monodon and Penaeus (Litopenaeus) vannamei. Mass mortality in cultured ponds has been observed 35 days after stocking, reaching up to 100 percent mortality within a few days after the first appearance of disease symptoms3. Clinical symptoms include lethargy, slow growth, empty stomach or midgut, and a pale to white atrophied hepatopancreas, with dead shrimp on the pond bottom4.
Members of the genus Vibrio comprise currently 14 recognized clades5, where the species are defined as clusters of strains with high phenotypic and genotypic similarities. Several Vibrio species are important causative agents of diseases in marine animals and humans. The pathogen identified by Tran et al.6 as the causing agent of AHPND correspond to a Vibrio parahaemolyticus strain. These authors reproduced the disease by infecting healthy shrimp with pathogenic bacteria. After verifying the Koch’s postulates, they concluded that AHPND has a bacterial etiology, identifying all isolates as members of the Vibrionaceae family and being closely related to V. parahaemolyticus. The genome of the pathogenic isolates of V. parahaemolyticus differs from non-pathogenic isolates because of the presence of two homologous genes related to the insecticidal toxin genes, PirA and PirB (PirVP), described for the first time in Photorhabdus sp.6. The toxin coding genes are located on a plasmid (pVAHPND) and the plasmid has been identified as being present in all AHPND-causing bacteria7.
Until now, V. parahaemolyticus strains containing plasmids with the genes for putative virulence have been described in Thailand8, Mexico9, China10, Vietnam8 and South America11. Although there is genetic variability in the plasmid sequences, which has led to a description of a Mexican type, characterized by a 4,243 bp Tn3-like transposon insertion and a 9 bp small sequence repeat (SSR), both features being absent from the Asian type8. However, all plasmids contain the toxin genes12 and a group of transposase-coding sequence related with horizontal gene transference (HGT)7.
Sawabe et al.13 argued that the HGT within the Vibrio genus might occur between sister species, such as V. cholerae and V. mimicus (Cholerae clade) or V. harveyi and V. campbellii (Harveyi clade). To date, no evidence of HGT has been observed between species belonging to distantly related clades. Recently, the presence of the pVAHPND has been discovered not only among V. parahaemolyticus strains, but also in other related species, in particular: V. harveyi, V. owensii and V. campbellii6,14–16, all species belonging to the Harveyi clade. For this reason, this clade is known to be the pathogenic clade for AHPND.
Most Vibrio species in the Orientalis clade are not causative agents of diseases, except V. tubiashii and V. sinaloensis, which have been associated with mortalities of bivalves and fishes17,18. In particular, there is no record of pathogens for crustaceans within this clade. The current study describes a new Vibrio species, Vibrio punensis sp. nov. strain BA55, belonging to the Orientalis clade and closely related to V. orientalis and V. hepatarius. Moreover, V. punensis sp. nov. strain BA55 carries the plasmid containing the toxin genes shown to be the cause of AHPND in shrimp. Our findings show that the gene acquisition capacity of Vibrio species goes beyond the clade boundaries that have been previously shown, demonstrating new infective capacity to a previously known commensal clade. The presence of these genes in a Vibrio clade not previously reported as pathogenic to crustaceans may contribute to the understanding of the Vibrio pathogenesis and has major implications for the spread of emerging diseases.
Results
PirVP gene detection, histopathologic analysis and typing using RAPDs
Shrimp collected during a mortality event in 2015 in a South American farm showed the symptomatology of AHPND, with external signs of empty stomach, lethargy, discoloration of the hepatopancreas, with a white membrane of smooth consistency expanding through it (Fig. 1A,B). In the longitudinal sections of the shrimp, a whitish coloration was present in the hepatopancreas, indicating a degenerative disease in this organ (Fig. 1A).
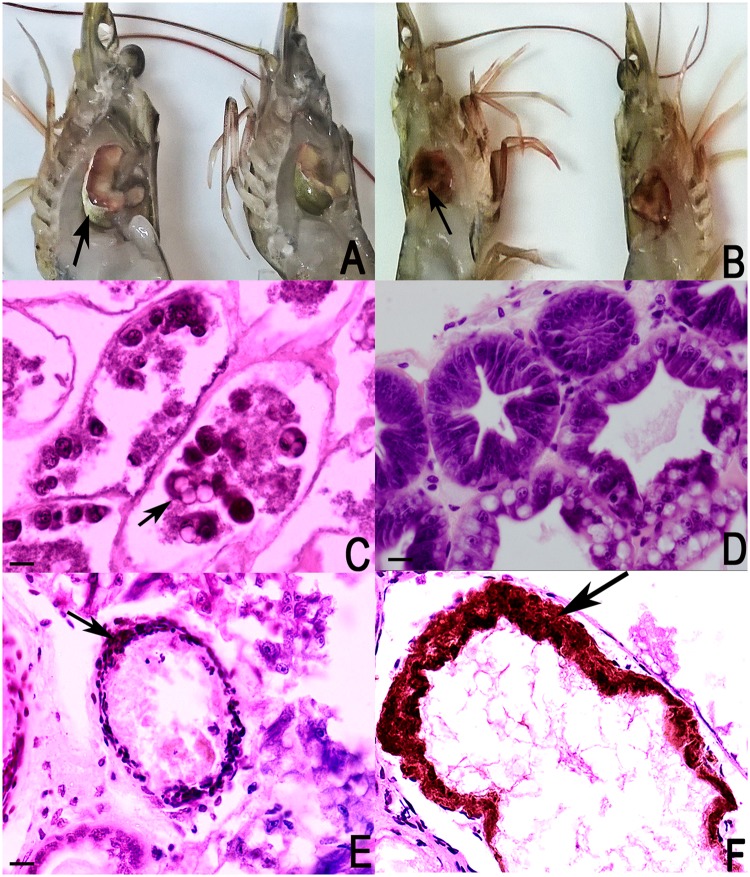
Figure 1.
Photographs from healthy P. vannamei shrimp and shrimp infected with BA55 strain carrying PirVP genes associated with AHPND. (A) Left shrimp view. Gross signs of AHPND-infected shrimp (left): pale, atrophied hepatopancreas surrounded with a white membrane with smooth consistency (black arrow). Normal shrimp (right): normal size hepatopancreas with brownish color. (B) Right shrimp view. Gross signs of AHPND-infected shrimp (left): complete hepatopancreas destruction (black arrow). Normal shrimp (right): normal size hepatopancreas with brownish color. (C) Hematoxylin and eosin-stained histological sections of the hepatopancreas of P. vannamei from sick shrimps. AHPND pathology characterized by sloughing of hepatopancreatic tubule epithelial cells (black arrow). (D) Hematoxylin and eosin-stained histological sections of the normal shrimp hepatopancreas. (E) Histological sections show severe necrosis of the hepatopancreas tubules (black arrow), making it impossible to discriminate the different types of cells. (F) Histological sections show cellular detachment caused by a bacterial infection, evidencing the formation of melanized haemocytic nodules (black arrow).
Histopathologic evaluation showed severe necrosis of the hepatopancreas tubules, with detachment of the epithelial cells from the membrane to the lumen (Fig. 1C), making it impossible to discriminate between the distinct types of cells that are affecting the integrity of the tubules (Fig. 1E). A marked difference was observed when compared to hepatopancreas tubules of healthy shrimp, where it was possible to discriminate between embryonic and secretory cells, both appearing to be in good condition, without any detachment and maintaining the tubule structure without deformities (Fig. 1D). Severe hepatopancreas necrosis limited the observation of lipid cells due to the cellular detachment caused by a bacterial infection, evidencing the formation of melanized haemocytic nodules in the middle part of the hepatopancreas tubules (Fig. 1F).
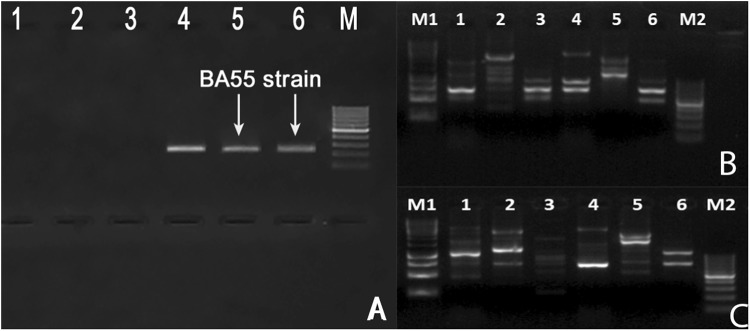
Bacterial recovery from infected animals was dominated by a single morphotype, named strain BA55, which was confirmed to contain the PirVP genes associated with AHPND (Fig. 2A). RAPD (randomly amplified polymorphic DNA), a simple and cost-efficient assay to evaluate genetic variability, was used to discriminate between related Vibrio species. A unique pattern for the identified BA55 strain was observed when compared to different isolates of Vibrio spp. previously characterized at CENAIM (Fig. 2B,C), suggesting that the disease was caused by a strain with unique molecular characteristics different to previously isolated bacteria from the Harveyi clade. Furthermore, full 16S Sanger sequencing confirmed that the isolated strain did not belonged to the Harveyi clade (Fig. S1).
Figure 2.
Identification of an isolate BA55 strain carrying PirVP genes associated with AHPND in P. vannamei. (A) Detection of PirVP genes. Lane 1: V. parahaemolyticus not containing the PirVP genes; lane 2: V. harveyi not containing the PirVP genes and lane 3: non-template control; lane 4: V. parahaemolyticus containing the PirVP genes; lane 5 and 6: PCR results from two independent DNA extractions from strain BA55; lane M: DL2000 DNA marker. (B) UBC101 amplicon profiles from the different isolates, lane 1: V. parahaemolyticus; lane 2: V. harveyi; lane 3: V. campbellii; lane 4: V. orientalis; lane 5: V. hepatarius; lane 6: BA55 strain. UBC 457 amplicon profiles from the different isolates, lane 1: V. parahaemolyticus; lane 2: V. harveyi; lane 3: V. campbellii; lane 4:V. orientalis; lane 5: V. hepatarius; lane 6: BA55 strain; lane M1: DL 10000 DNA marker; lane M and M2: DL1000 DNA marker.
Bacterial bioassay shows BA55 strain causes AHPND in shrimp
The BA55 strain was used in vivo to challenge susceptible shrimp in triplicates starting at 2 × 106 CFU ml−1. Shrimp mortality was observed after 10 h post infection, whereas the V. parahaemolyticus BA94C2 strain, at the same concentration, caused mortality after 8 h (Fig. 3). No mortalities were reported for the negative controls. Shrimp cumulative mortalities at 70 h post infection in both infected treatments were high (BA55: 95.2 ± 0.8%; BA94C2: 99.8 ± 0.7%), with a significant difference between treatments (P = 0.005, Kruskal‐Wallis test). Moribund shrimp from the BA55 infected treatment exhibited the same symptoms as observed on diseased P. vannamei collected from the shrimp farm. Moribund shrimp from different replicates infected with the BA55 strain were used for isolating the predominant Vibrio strains and recovering them in pure culture. The isolated strains were then further tested with the RAPD assay. The DNA banding pattern in these colonies was the same as those obtained from the BA55 strain isolated from the diseased cultured shrimp (Fig. 4) and used as an inoculum, thus confirming Koch’s postulates and confirming that the BA55 strain is a causative agent of AHPND. Additionally, the PirA and PirB toxin genes were detected by PCR in the isolated colonies recovered from moribund shrimp during the challenge, whereas the macerates of the negative controls remained negative (Fig. S2).
Figure 3.
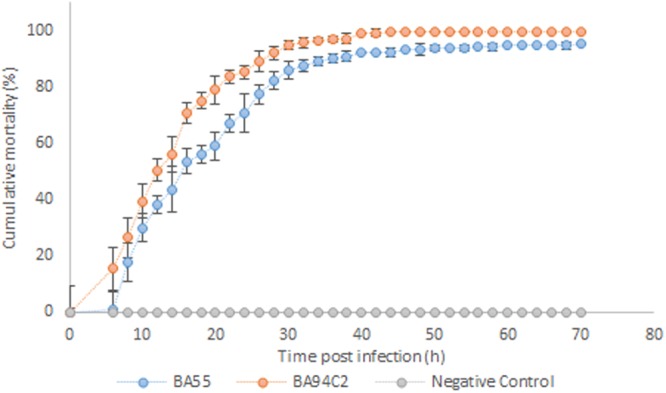
Shrimp cumulative mortality after being challenged with BA55 and Vibrio parahaemolyticus BA94C2 strains. Negative control was TSB 2% NaCl. Bars indicate standard deviations.
Figure 4.
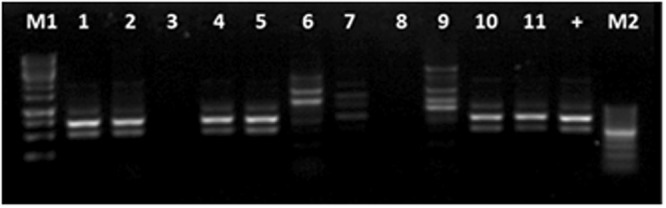
DNA banding pattern of bacterial isolates obtained from challenged shrimps infected with the pathogenic BA55 and V. parahaemolyticus BA94C2 strains. From challenged shrimp infected with the BA55 strain; Shrimp 1, lane 1: Large size and convex isolated (LCI); lane 2: Small size and convex isolated (SCI); lane 3: Large and plain isolated (LPI). Shrimp 2, lane 4: LCI; lane 5: SCI; lane 6: LPI. From challenged shrimp infected with the V. parahaemolyticus BA94C2 strain; Shrimp 3, lane 7: Large size isolated (LI); lane 8: Medium size isolated (MI); lane 9: Small size isolated (SI). Inoculum controls from BA55 strain, lane 10 and lane 11. Positive control, lane +. Lane M1: DL 10000 DNA marker; lane M2: DL1000 DNA marker.
De novo assembly of the BA55 strain genome sequence
Total DNA extracted from the isolated strain was used for de novo genome sequencing and assembled using 19,434,590 Illumina PE reads. A total of 75 contigs were obtained. Annotation and comparison to reference Vibrio spp. genomes showed evidence of two chromosomes (Chr 1: 3.2 Mbp long and Chr 2: 1.5 Mbp long) and a plasmid (pVAHPND; ~69 Kbp long) containing the toxin Pir, with two subunits: PirA and PirB. The average coverage was 46.3×, G + C content was 44.5%, 4,494 genes were predicted, and the total genome size was 4.7 Mbp (Table 1). An initial evaluation using BLAST among the predicted genes and a set of 76 other reference Vibrio genomes available in public databases (Table S1) showed no significant difference (less than 1% in the percent identity) among the scores obtained from the different Vibrio species, suggesting that this strain is equally distant from the previously known Vibrio species, potentially constituting an undescribed species.
Table 1.
Genome characteristics of the strain BA55 draft genome.
| Attributes | Values |
|---|---|
| Assembly size (bp) | 4,727,353 |
| Total number of contigs | 75 |
| Contig N50 (bp) | 206,237 |
| L50 | 10 |
| GC content % | 44.5 |
| Number of CDS | 4,494 |
| Number of rRNAs | 20 |
| Number of tRNAs | 142 |
Phylogenetic analysis of the BA55 strain
The Maximum Parsimony (MP), Maximum Likelihood (ML) and Neighbor Joining (NJ) analysis of 16S rDNA showed a high identity between all Vibrio sequences analyzed and low support for the branches in all the inference methods used, suggesting a low resolution to resolve intra and inter-specific phylogenetic relationships within the Vibrio genus. We found that the closest neighbor of the strain BA55 was V. brasiliensis strain LMG 20546 (75% bootstrap values; Fig. S3) from the Orientalis clade. Importantly, although the 16S rDNA gene is commonly used for phylogenetic analyses, the sequence identities among Vibrio species tend to be very high making this gene an unsuitable marker for phylogenetic analysis within the genus, further confirmed by the low branching support and the incongruence on the clustering with the traditional Vibrio clades.
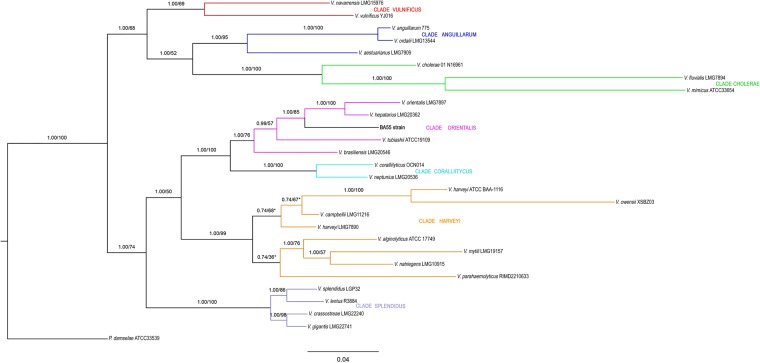
In order to gain a higher phylogenetic resolution, the phylogenetic trees derived from the concatenated nucleotide sequences of six commonly used MLST genes (ftsZ, gapA, gyrB, mreB, topA, and 16S rRNA genes) showed that the strain BA55 branched deep within the Orientalis clade, indicating a sister group relationship with V. orientalis and V. hepatarius. In fact, these three species form a monophyletic group within the Orientalis clade, distant from other known AHPND-causing bacteria and all the Vibrio within the Harveyi clade (Fig. 5). In addition, MP, ML, NJ and Bayes analyses revealed consistent phylogenetic tree topologies. The BA55 strain was phylogenetically distant from the Anguillarum clade (89.01% ± 3%), Vulnificus clade (87.69% ± 1%) and Cholerae clade (81.46% ± 3%), and more closely related to members of the Orientalis clade: V. hepatarius (96.32%), V. orientalis (95.57%) and V. tubiashii (89.85%). The closest clades to the BA55 strain were the Coralliilyticus and the Harveyi clades (90.12% ± 2% and 89.23% ± 4%), the latter being the pathogenic clade for AHPND, containing V. parahaemolyticus, V. harveyi, V. owensii and V. campbellii, the species where the presence of the pVAHPND plasmid has been previously reported6,14–16. Percentages shown are the average sequence similarity, calculated with the proportion of nucleotide sites that differ between sequences and divided by the total number of sites for the comparison; in our case, based on the six protein-coding housekeeping genes of the BA55 strain.
Figure 5.
Phylogenetic concatenated reconstruction based on ftsZ, gapA, gyrB, mreB, topA, and 16S rRNA genes. A Bayesian based phylogeny on a concatenated multiple sequence alignment using the TIM + G evolutionary model is shown. Numbers above the branches represent the posterior probabilities and percent bootstrap values (one thousand bootstrap replicates). Reference sequences used were described by Sawabe et al.5 and sequences extracted from complete genomes. An independent Maximum Parsimony and Maximum Likelihood phylogenetic analyses were performed. Branches that were not conserved among the three topologies are shown with an asterisk. Scale bar represents number of substitutions per site. Color lines represent each Vibrio clade as follows: Vulnificus clade in red; Anguillarum clade in blue; Cholerae clade in green; Orientalis clade in purple; Coralliilyticus clade in light blue; Harveyi clade in orange; Splendidus clade in gray. Our results show evidence that the strain BA55 is related to V. hepatarius and V. orientalis. The outgroup used was Photobacterium damselae.
Also, we show the phylogenetic reconstruction supporting the taxonomic position of the strain BA55, well-differentiated within the Orientalis clade (Fig. S4). Phylogenetic trees based on the concatenated partial sequences of the five housekeeping genes reconstructed by the NJ, ML and Bayes methods supported the independent and robust position of the strain BA55 within the Orientalis clade, especially the closest position relatives to V. orientalis and V. hepatarius and maintaining a low percentage of similarity with V. tubiashii, V. bivalvicida and V. europeaus. V. crossai maintains its position with V. brasiliensis.
For calibration of the evolutionary rates and estimation of the divergence times, we used a set of representative genomes from the Vibrio genus and analyzed the rate of amino acid substitutions in the six housekeeping genes. The lowest radiation time value for our strain BA55 was obtained with V. hepatarius (Table 2), revealing that these species are closely related. The different phylogenetic trees reconstructed from species of the Harveyi and Orientalis clades showed a consistent branch pattern, in particular for the relative position of the BA55 strain within Orientalis clade (Fig. 5).
Table 2.
Clades and subclades used for MLST analysis.
| Clade | Species included | No. of species | GC (mol%)a | MLST (%)b | AAI (%)c | Phi test (P value)d | Habitate |
|---|---|---|---|---|---|---|---|
| Anguillarum | V. anguillarum, V. aestuarianus and V. ordalii | 3 | 43–46 | 95.5 | 99 | 87.0–98.3 95.8–99.8 | Brackish water, seawater and fish |
| Cholerae | V. cholerae, V. mimicus, V. furnissii, V. fluvialis, V. metschnikovii and V. cincinnatiensis | 6 | 44–50 | 87 | 96.3 | 83.4–94.4 93.5–99.8 | Brackish water, seawater |
| Coralliilyticus | V. coralliilyticus and V. neptunis | 2 | 45–46 | 97 | 99.7 | 94.2 99.5 | Seawater, bivalves and rotifers |
| Harveyi | V. harveyi, V. campbellii, V. parahaemolyticus, V. alginolyticus, V. mytili, V. natriegens, and V. rotiferianus | 7 | 42–48 | 91.3 | 97.8 | 86.7–96.0 96.9–99.9 | Seawater, salt marsh mud and marine animals |
| Orientalis | V. orientalis, V. hepatarius, V. brasiliensis, V. sinaloensis, V. tubiashii and BA55 strain | 6 | 43–46 | 95.9 | 99.2 | 100 | Brackish water and seawater |
| Splendidus | V. splendidus, V. crassostrea, V. cyclitrophicus, V. chagasii, V. fortis, V. kanaloaei, V. lentus, V. gigantis, V. pelagius, V. tasmaniensis and V. pomeroyi | 11 | 39–47 | 98.2 | 99.8 | 88.8–93.7 97.1–98.9 | Seawater and marine animals |
| Vulnificus | V. vulnificus and V. navarrensis | 2 | 45–48 | 93.6 | 98.8 | Sewage, seawater and oyster | |
| Damselae | P. damselae | 1 | 42 | Seawater and fish |
aGC content calculated based on six genes.
bMLST percentage of concatenate similarity between sequences.
cAAI Raw average amino acid identity (10).
dPhi test. The phi test was conducted for clades that included at least four species.
eHabitat described for Sawabe et al.13.
Sequence analysis of pVAHPND in the strain BA55
The plasmid pVAHPND found in the strain BA55 was assembled in a single 69,163 bp contig, with a mean GC content of 44.9% and 53 putative ORFs. A homologue of the insecticidal Photorhabdus insect-related binary toxin PirA and PirB was also identified in this plasmid. The toxin genes are flanked by an arrangement of SWAT-3 type transposases (VSWAT3 like transposases), suggesting a potential mechanism of HGT between different Vibrio populations11. Importantly, no homologous sequences for the transposases were identified in any of the genomic contigs and evidence for a robust assembly of the plasmid was supported by pair-end reads agreement and an average coverage of 1,200X throughout the plasmid sequence. Xiao et al.19 reported that similar Tn903-like transposons were detected in either the plasmid (V. owensii, V. harveyi and V. parahaemolyticus) or within the chromosomal genome (V. anguillarum and V. campbellii) of species belonging to the Harveyi clade, but they have not been reported in the Orientalis clade. The comparison of the plasmid sequences from the V. parahaemolyticus (accession number: KM067908), V. owensii (accession number: KX268305), both from the Harveyi clade, and the strain BA55 showed a high percentage of similarity (99.97 average percent identity) demonstrating that they are closely related to each other (Fig. 6). It is important to note that no similar plasmid has been described within species from the Orientalis clade.
Figure 6.
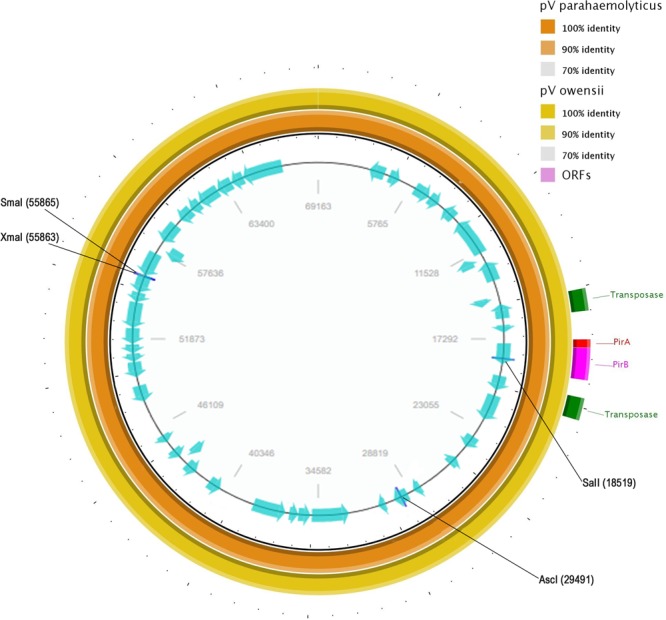
Sequence map of the AHPND-associated plasmid pVAHPND in strain BA55. First two outer circles represent the BLAST comparison with the plasmid of V. parahaemolyticus (orange) and V. owensii (yellow). The internal ring represents the predicted open reading frames (ORFs) shown as arrows in blue, direction of arrowhead represents the transcriptional orientation. The transposase genes in green, red and purple represent the PirA and PirB genes correspondingly.
Comparative genome analysis
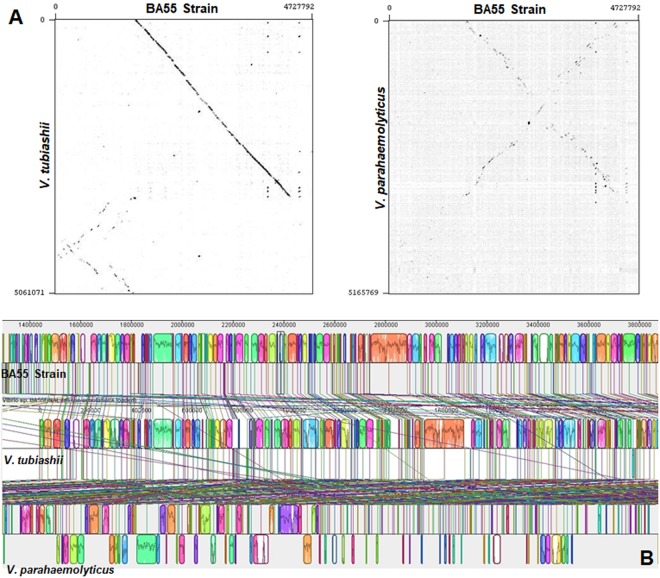
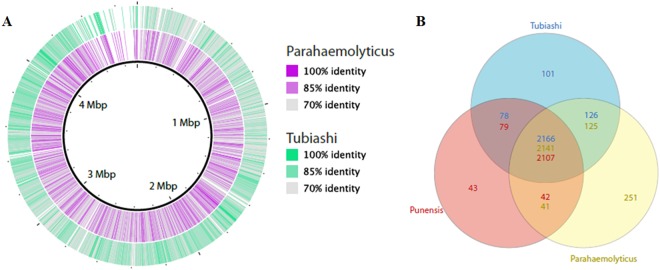
The comparative genomic analyses supported the hypothesis of the strain BA55 as a member of the Orientalis clade. The evidence came from multiple genome alignments and the evaluation of genomic architectures using Blast. The pairwise genome alignment between the genomes of the strain BA55 with V. tubiashii (Orientalis clade) and V. parahaemolyticus (Harveyi clade) suggested a closer relationship to V. tubiashii than to V. parahaemolyticus (Fig. 7A) as there was a higher number of rearrangements with the V. parahaemolyticus strain (Fig. 7B). The closer similarity was observed both when comparing the genomes at the nucleotide and amino acid level. Blast analysis showed a higher average percent identity with V. tubiashii (91%) compared to V. parahaemolyticus (88%) (Fig. 8A). At the amino acid level, the shared genes with a RAST annotated function between V. parahaemolyticus and the strain BA55 contained mainly essential genes that are needed for growth or viability (that were also shared with V. tubiashii and are likely part of a Vibrio core genome) and only 42 genes uniquely shared with V. parahaemolyticus, whereas V. tubiashii and the strain BA55 shared genes that are necessary for adaptation, including the genes for growth in different environments, which are associated with biofilm formation and bacterial communication like the Acetyltransferase SypM (Fig. 8B inset). This suggests the existence of gene clusters unique to the Orientalis strains distinct from the other Vibrio’s core genes. Thirty-two genomic islands (GIs) were detected for the strain BA55 (Table S2) including sequences coding for mobile genetic elements, as prophages and transposons, and several recombination-related proteins as integrases, recombinases and transposases. The gene comparison represented by the Venn diagram showed different number of shared genes between pair of genomes, which is due to homologs within the same genome (paralogs) (Fig. 8B inset).
Figure 7.
Whole genome alignments between V. punensis strain BA55, V. tubiashii strain ATCC 19109 and V. parahaemolyticus RIMD 2210633. (A) Dot-plots of nucleotide identities of the BA55 strain against V. tubiashii strain ATCC 19109 (left) and BA55 strain against V. parahaemolyticus RIMD 2210633 (right). (B) Nucleotide-based multiple genome alignment of the same genomes. Homologous blocks are shown as identically colored regions and linked across the genomes. Rearrangements are shown as homologous regions in different genomic locations.
Figure 8.
Comparative analysis of BA55 strain with Vibrio tubiashii ATCC 19109 and Vibrio parahaemolyticus RIMD 2210633. (A) Circles 1 and 2 show the BLAST comparison with V. tubiashii (green) and V. parahaemolyticus (purple). (B) Venn diagram showing unique and shared genes based on the RAST subsystems annotation among the three Vibrio species used before (BA55 strain, V. tubiashii and V. parahaemolyticus). Intersects show different number of genes for each genome due to potential paralogs within the genomes.
PirVP gene causing acute hepatopancreatic necrosis disease identified in a novel species from the Orientalis clade
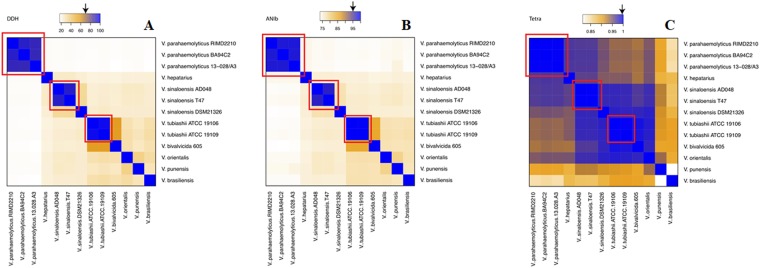
In order to calculate the distances among the genomes on seven members of the Orientalis clade (V. hepatarius, V. sinaloensis, V. tubiashii, V. bivalvicida, V. orientalis, V. brasiliensis and BA55 strain) and one member of the Harveyi clade (V. parahaemolyticus) we used the Genome-to-Genome Distance Calculator (GGDC 2.1)20. The first evaluated method was the in silico DDH (DNA-DNA hybridization) analysis. Our results show that the BA55 strain displayed DDH values below 70% with every other strain analyzed. This cut-off has been traditionally used for the delineation of species in prokaryotes, for both in silico21 and experimental analysis22 (Fig. 9A). For all pairwise comparisons made, the highest value obtained was 36% with V. orientalis (Table S3). In addition, the percentage of differences between G + C content was >1%, corresponding to values obtained traditionally between distinct species20.
Figure 9.
Heatmaps representing metrics for the evaluation of species circumscription among Vibrio strains. The extent of nucleotide identity was calculated by different indices for species circumscription: DDH (A), ANIb (B) and Tetra (C) as illustrated. The key color was constituted by orange and blue as indicated. Arrows shows the reported cut-off for species a boundary. Red boxes shows clusters of genomes within the reported cut-off.The species and strain number of samples are shown.
ANI calculations were performed using MUMmer (ANIm) and BLAST (ANIb) algorithms23. Previous studies using ANIm and ANIb for species classification in the Orientalis clade had suggested a species boundary of 95–96%20. Our results are consistent with those observations given, that all known strains within a given species showed pairwise ANIm values above the threshold, except for V. sinaloensis strain DSM21326, concordant to the DDH results (Fig. 9B, Table S3) furthermore, the ANIb results confirm that BA55 is a species distinct from others available, with an ANIb value of <79% and an ANIm of <86% (Fig. 9B, Table S3). The discrepancy between the ANIb and ANIm values obtained is consistent with previous reports from Li et al.24 and Dubert et al.25 where they demonstrated the lack of agreement between ANIb and ANIm when the compared genomes show low correlation values (ANIb < 75% and <77%, respectively), in our case the ANIb value is <79%.
Furthermore, it has been argued that the oligonucleotide frequencies carry a species-specific signal26. For this reason, statistical calculation of oligonucleotide frequencies (TETRA) from pairwise comparisons was performed. TETRA values indicated that the strain BA55 is more related to V. orientalis strain CIP102891 (Fig. 9C). When comparing with the DDH and ANI cut-offs for species demarcation, TETRA values between relative species were above 99.5%, supporting their affiliation to the same species and between the Orientalis clade, this value can be proposed as a plausible cut-off. In contrast, the highest values for the BA55 strain were 93% for the comparison with V. orientalis and 92.4% with V. sinaloensis (Table S3). Together with the results based on in silico DDH and ANI we also suggest that V. sinaloensis strain DSM21326 should be reclassified, as it does not comply with the species described cut-offs as the other strains of V. sinaloensis does.
Based on all previously reported evidence and the nucleotide composition values, we proposed the strain BA55 as a new specie named Vibrio punensis sp. nov. strain BA55, belonging to the Orientalis clade. This novel species carries the plasmid pVAHPND giving it the pathogenic characteristics that had been only previously reported on the Harveyi clade.
Discussion
The visible symptoms observed in dying shrimp collected during the challenge test were similar to those observed in diseased shrimp collected from farms. The results of our challenge test indicated that the isolated BA55 strain triggered a high mortality, although lower than the strain BA94C2, and similar to other AHPND strains reported by Tran et al.6 and Nunan et al.4, where shrimp reached 100% mortality at 48 hours post infection trough immersion assays in experimental tanks, at an approximate bacterial density of 2 × 106 CFU ml −1. The results obtained from the challenge test using the BA55 strain compared with the control, and the fact that it contains the PirA and PirB genes suggest that the toxin, as is the case in other AHPND-causing Vibrios, is responsible for the mortality of the infected shrimp.
Current microbial taxonomy is being revolutionized by the generation of full-length microbial genomes from uncultured microorganisms and full genome assembly from metagenomic projects27 requiring a full set of new taxonomical tools and standards. To date, DNA-DNA hybridization (DDH) has been the “gold standard” for bacterial species demarcation, as it provides a numerical threshold for a species boundary28. Nevertheless, some authors suggest the necessity of a new approach due to the labor-intensive and error-prone nature of DDH experiments20–28. According to Richter and Rosselló-Móra et al.23 the scientific community needs methods that offer a similar resolution of DDH and simultaneously allow the construction of databases to retrieval of any information for comparative purposes. Therefore, the average nucleotide identity (ANI) method was developed and it seems to be the best alternative for a gold standard method29. Richter and Rosselló-Móra23; Ramasamy et al.27 and Meier-Kolthoff et al.20 among others, have made efforts to validate ANI as a mirror for DDH and that in conjunction with TETRA values, has been proposed as a plausible substitute for DDH, although further testing should be performed in particular clades. Given the mounting evidence, the recent use of such metrics for proposing other Vibrio clades and species13–15, and the results shown in our study, we propose the BA55 strain as a novel species, named Vibrio punensis sp. BA55.
Researchers have proposed that HGT drives evolution by incorporating genetic material from one organism to another. In this specific case, it is plausible that through HGT, the Vibrio punensis sp. nov. strain BA55 had become pathogenic. This shows that the gene transfer and the capacity of this new species to use these foreign genes is offering a new infective capacity to a species and a clade that has not previously been reported as a pathogen in crustaceans. Previously, it was only reported that species of the Harveyi clade present the ability to use PirVP genes, carried in plasmids, to cause AHPND. The current phylogenetic study revealed that these genes could be found in new species and thus enhance their infective capacity, thereby potentially increasing the complexity of causative agents of AHPND and aggravating the threat to the cultured shrimp industry.
Current developments in sequencing technologies and microbial ecology are changing the perception that we had about commensals and pathogens and blurring the lines separating them. Common aquatic environments are composed of thousands of different bacterial species displaying complex interactions and where selective pressures award those capable of gaining new properties that enhance the fitness of the surviving species. The Vibrio genus is a large and complex taxon that includes free-living bacteria found in aquatic environments but with the potential of residing in different animal hosts. The specific interactions with the host ranges from beneficial, as those described in V. hepatarius used as probiotic in shrimp30 to pathogenic, such as V. cholerae, V. parahaemolyticus, V. vulnificus and V. alginolyticus31–33. Once related species with different adaptation strategies to an environment collocate within a confined space under strong selection (such as different Vibrio species within the gut of a crustacean animal), it constitutes the perfect setup for the occurrence of HGT. This scenario could be the cause for a previously commensal bacterium to become pathogenic. The same principle applies to any other environment where commensal or symbiotic microorganisms potentially interact with pathogens, thus, increasing the chances of horizontal gene transfer of such virulence markers, which not necessarily limited to toxins, but also to potential antibiotic resistance in environments such as the human gut.
Materials and Methods
Isolation of bacterial strains
On July 2015, stomachs and hepatopancreas from AHPND positive shrimp collected during a mortality event in 2015 in a South American farm were removed and separately macerated to obtain mixed cultures. These cultures were subdued to subculture on TCBS (thiosulfate citrate bile salts sucrose) agar plates to obtain individual colonies and were further purified on TSA (tryptic soy agar) plates and pure isolates were then cryopreserved at −80 °C. A total of 39 bacterial strains were isolated from P. vannamei juvenile shrimp. Bacterial strains were characterized using morphological and biochemical criteria following the schemes of Alsina & Blanch34 and bacterial typing were conducted by 16S rRNA sequencing. Amongst those, a predominant bacterial strain coded as BA55 was selected for further analysis.
PirVP gene detection
Bacterial samples were centrifuged at 4,000 g for 10 minutes and decanted. The resulting pellet was resuspended in 500 µl extraction STE buffer (10 mM Tris-HCl, 1 mM EDTA and 100 mM NaCl, and 0.2 mg/ml proteinase K; Invitrogen, Carlsbad, CA) and incubated at 55 °C for 2 hours. After cell lysis, purification was carried out with phenol-chloroform and precipitation with ethanol, following procedures by Maniatis et al.31 with minor modifications. Each supernatant was removed and extraction with an equal volume of phenol-chloroform-isoamyl alcohol (25:24:1) was performed, followed by extraction with chloroform-isoamyl alcohol (24:1). DNA was recovered by the addition of 1 volume of isopropanol followed by centrifugation at 10,000 g for 10 min. The DNA pellet was washed with 70% ethanol, dried, and resuspended in 50 µl of ultrapure water pH 7.0. DNA extracts were stored at −20 °C for further analysis. DNA concentrations were determined using a NanoDrop 8000 Spectrophotometer (NanoDrop 2000, Thermo Fisher Scientific Inc., Wilmington, USA).
The presence of the main genes associated to AHPND (PirVP) were tested on all 39 isolated bacterial strains using a nested PCR method, with primers previously described, to obtain amplified products of 1,269 and 230 bp (Table S4). Likewise, PirA and PirB genes were amplified separately with specific primers aimed at generating PCR products of 284 and 392 bp for PirA and PirB, respectively (Table S412).
RAPD analysis
Two primers (UBC101, 5′-GCGCCTGGAG-3′and UBC 457, 5′- CGACGCCCTG -3′) were selected from an initial screening with 10 decanucleotide primers, for the evaluation of RAPD pattern bands of the bacterial strains: BA55, AHPND V. parahaemolyticus BA94C211 and pathogenic non-AHPND strains (V. parahaemolyticus, V. harveyi, V. campbellii, acquired from the Microbiology Laboratory, Universidad Santiago de Compostela and V. hepatarius, acquired from the Microbiology Laboratory, CENAIM-ESPOL). All strains were grown overnight at 28 °C on TSA. One colony of each strain was suspended in 200 µl STE buffer and the DNA extraction was performed as described above. PCR was carried out in a 10 ml reaction volume containing 10 mM Tris-HCl, pH 8.3, 50 mM KCl, 3 mM MgCl2, 100 mM of each dNTP, 0.4 mM of the arbitrary primer, 2 µl of genomic DNA and 1 Taq DNA polymerase (Invitrogen). Amplifications were performed using a Thermo Fisher thermoblock under the following conditions: 4 min initial denaturation at 94 °C, followed by 39 cycles of 5 sec denaturation at 94 °C, 45 sec annealing at 46 °C, and 1 min elongation at 72 °C, with a final elongation step at 72 °C for 5 min. The PCR products (5 μl) were analyzed on a 1.5% agarose gel stained with SYBR Safe DNA Gel Stain (Thermo Fisher), which was then visualized under UV transillumination and photographed using an E-Gel Imager (Thermo Fisher).
Immersion challenge test
The pathogenicity of the BA55 strain was evaluated by an immersion challenge test as described by Tran et al.6, with minor modifications. Briefly, a single colony of the BA55 strain was picked and re-suspended in 30 ml of sterile TSB (tryptic soy broth culture medium with 2% NaCl). The culture was incubated for 18 h at 28 °C on a rotary shaker (200 rpm) and the bacterial density was determined by a microplate luminometer (Varioskan™, Thermo Fisher Scientific). One ml of this solution was transferred to 50 ml of TSB medium and cultured as described above for 10 h. The suspension was plated onto TSA after serial dilutions to determine the colony-forming units of the isolate (CFU ml−1). The V. parahaemolyticus BA94C211 strain was used as positive control under the same conditions.
For the infection, each strain was cultured in TSB for 12 h at 28 °C, to reach 2 × 109 CFU ml−1. Then, a 15 min immersion was performed on flasks containing 40 healthy shrimps (2.5 ± 0.5 g) per flask and a solution of bacterial suspension with saline water, to achieve a bacterial density of approximately 2 × 108 CFU ml−1. Immersed animals were transferred into experimental aquariums filled with 40 L of filtered and UV sterilized seawater and continuously aerated. After transfer, a solution of bacterial suspension was immediately added directly to the experimental tanks containing clean seawater (filtered and UV sterilized), to reach an approximate bacterial density of 2 × 106 CFU ml−1. A negative control (40 shrimp immersed in sterile TSB, without bacterial strains) was also included. All treatments, including the control, had 10 replicates. During the assay, shrimp were monitored for mortality every 2 h. Difference of cumulative mortality at 70 h post infection among infected treatments was analyzed with the non-parametric Kruskal-Wallis test by using the R software35.
Re-isolation of bacteria
Moribund shrimp from infected treatments were collected 12 h post infection. Samples from the hepatopancreas and stomachs were removed and macerated in sterile saline solution. One hundred microliter of serial dilutions (10−1, 10−2 and 10−3) containing macerated hepatopancreas and stomachs were inoculated on TCBS agar and incubated at 28 °C for 18 h, and the three most predominant colonies were purified on TSA plates (Table S5). Following this, the DNA of the colonies was extracted and stored for subsequent comparisons with the tested isolates using RAPD assay36, and PirVP genes detection, as described above. Macerates of shrimp hepatopancreas and stomachs of negative controls were also analyzed for PirVP genes detection. Shrimps were analyzed individually.
Histopathologic analysis
Shrimp samples were preserved with Davidson AFA fixative for histopathology analysis. Tissues were processed in accordance with the histological routine procedure37. Sections were cut at 5 μm and stained with Mayer-Bennet hematoxylin and eosin. Tissues were examined for histopathological changes.
Genome sequencing and de novo assemble
The genome of the predominant bacteria strain, BA55, was sequenced using Illumina MiSeq PE150 platform. The library was constructed with Illumina Paired-End DNA Sample Kit (Illumina Cambridge Ltd, UK). Raw Illumina FASTQ files were demultiplexed and quality-filtered using FastQC38. Any reads containing ambiguous base calls or barcode errors were discarded. De novo assembly was performed using Spades39. The generated contigs were used as seeds into Contiguator40 to polish it, through an alignment of raw reads on contigs by BLAST41. The complete genome sequence was deposited at DDBJ/EMBL/GenBank under the accession PRJNA412371.
Genome annotation
The genome of the BA55 strain was annotated using GLIMMER42 and NCBI Prokaryotic Genome Annotation Pipeline43. In addition, other 76 Vibrio genomes from the GenBank database (Table S1) were used for multilocus sequence typing (MLST) phylogeny. These 76 genomes were re-annotated by RAST44 and the RNA regions were characterized using RNAmmer45. Then, all the predicted ORFs were translated and compared by a homology search (BLAST) against non-redundant protein databases from NCBI (E-value threshold = 106 and minimal alignment length ≥80%), Swiss-Prot44, Clusters of Orthologous Groups (COG46), Kyoto Encyclopedia of Genes and Genomes (KEGG47) and Gene Ontology (GO48). To evaluate the functional and evolutionary relationship between the 76 Vibrio genomes and the assembled genome of the BA55 strain, a comparison among those genomes was performed to evaluate sequence similarities.
16S rDNA and multilocus sequence typing phylogeny
An initial phylogenetic analysis was performed with a dataset of 862 16S rDNA sequences obtained from the NCBI Reference Sequence Database (RefSeq), together with the 16S rDNA sequences extracted from the 77 complete Vibrio genomes (including the genome of the BA55 strain). Before performing all the phylogenetic analyses, a dereplication of the sequences by species was performed, retaining only one representative sequence per specie, obtaining a final set of 216 sequences (Fig. S5). All non-redundant sequences were aligned with MEGA 6.049 using the MUSCLE algorithm. Phylogenetic trees were constructed using Maximum Parsimony (MP), Maximum Likelihood (ML) and Neighbor Joining (NJ) methods. We use JModeltest 2.050 to test the evolution models based on the hierarchical likelihood ratio test, determining that the TIM + G model best fit the data51. The nucleotide substitution model chosen was consistent with previous reported models of Vibrio evolution13. The evolutionary distances between the species of Vibrios were estimated with MR. Bayes, considering the branch-length and divergence time for model selection across a wide range of phylogenetic and evolutionary models52.
To establish a more robust inference of the evolutionary history of the Vibrio genus, the same phylogenetic analyses described before were repeated, using a MLST analysis, by evaluating partial sequences of six commonly used, phylogenetic marker genes: ftsZ, gapA, gyrB, mreB, topA, and 16S rRNA from Vibrio species (70 sequences) reported by Sawabe et al.5, complemented by the 77 genomes used for re-annotation (including the genome of the BA55 strain). GenBank accession numbers are listed in Tables S1 and S6. We used the TCS program53 to estimate a phylogenetic network estimation among all Vibrio species used for MLST analysis, to choose the 21 more related to the BA55 strain.
Recombination was evaluated with the Phi test using the concatenated sequences of Vibrios, reported by Sawabe et al.13 Recombination was detected in gyrB (P = 6.4 × 10−3), gapA (P = 6.8 × 10−7) and topA (P = 4.5 × 10−2) in agreement with the conflicting phylogenetic splits (Table 2). The analysis was performed with SplitsTree454, a framework for building phylogenetic trees and networks. Detecting recombination is basically a statistical endeavor and ideally in vitro experimental work should be carried out in order to confirm the ability of Vibrios to carry out recombination in the loci analyzed13.
Comparative genome analysis
MAUVE software55 was used to perform whole genome alignments and comparisons. The genome of the BA55 strain was aligned to the two most cured, well-annotated and complete genomes of the Harveyi and Orientalis clades: V. parahaemolyticus RIMD 2210633 and V. tubiashii ATCC 19109, respectively, to evaluate gene conservation among the Harveyi and Orientalis clades. Genes previously predicted, were mapped in this comparison, for the identification of genomic islands (GIs) and several common characteristics of the genome, such as abnormal sequence composition or the presence of mobile genetic elements using Pfam. The gene comparison was represented by a Venn diagram (Fig. 6 inset) only on the genes that belong to a subsystem annotation in RAST.
Dot plots were generated by Gepard56 using default parameters with the ordered contigs produced by MAUVE for each genome.
Genomic islands identification
Genomic islands were predicted in the draft genome of BA55 strain using IslandPick, IslandPath. DIMOB and SIGI-HMM algorithms, performed in an integrated interface from IslandViewer57.
Average nucleotide identity, correlation indexes and DDH estimates
Current bioinformatics methods allow replacement of the wet-lab DNA:DNA hybridization (DDH) estimates by in-silico genome-to-genome comparison and transforms the distance from high-scoring segments pairs to values analogous to DDH using a generalized linear model58, this logistic regression model is inferred from an empirical reference dataset comprising real DDH values and pairwise genome sequences comparison. DDH in silico was used to obtain a better resolution of discrimination and simultaneously allow the construction of databases that permit the retrieval of any information for comparative purposes59.
In addition to DDH in silico, the metrics ANIb, ANIm and TETRA have been recently developed and are widely used as metrics to measure the distance between closely related species, generating boundaries and differentiating isolates of the same species to those of different species. The average nucleotide identity (ANI) was measured between pairs of genomes, based on the BLAST algorithm (ANIb39), as well as on the MUMmer aligning tool (ANIm60). In addition, we calculated the tetranucleotide frequencies (TETRA), a statistical method which is an alignment-free process. The values for ANIb, ANIm and TETRA for the Vibrio genome comparisons were calculated for all possible bacterial pairs, including all species in Orientalis clade (five members), one species of Harveyi clade (V. parahaemolyticus) and our BA55 strain. All comparisons were represented as heatmaps using R statistical software35.
Equipment and adjustments
Histological Images:
Biological Microscope OLYMPUS CX31, UIS (Universal Infinity System) optical system.
Infinity capture imaging software Lumenera Corporation, Trademark INFINITY 2.
Size: 1616 × 1216 pixels
Resolution: 96 DPI
Gel Images:
Equipment ENDUROtm GDS TOUCH Labnet
The Endurotm GDS Touch Aquisition Software.
Size: 768 × 526 pixels
Resolution: 96 DPI
Organism Images:
Ilumina Nokia 630
Size: 2592 × 1956 pixels
Resolution: 72 DPI.
The Fig. 1 is a merge of captures using photoshop CC 2015. The images were left in the same size 750 × 578 pixels. The text, arrows and contrast were modified in photoshop and the final merge was left in 300 DPI.
The Fig. 2 is a merge of captures using photoshop CC 2015. The images were left in the same size 2400 × 1644 pixels. The text, arrows and contrast were modified in photoshop. The final figure was left in 300 DPI.
Electronic supplementary material
Table S1, Table S2, Table S3, Table S4, Table S5 and Table S6
Acknowledgements
We thank Betsy Andrade and Karina Reyes for their technical assistance in this study. We would like to thank Dr. Andres Pinzón for his help in the preliminary analysis and Dr. Stanislaus Sonnenholzner and Nicolas Malo for their kind reviewing of the manuscript and suggestions. Additionally, we acknowledge the High-Performance Computing Service at Universidad de los Andes in Bogotá, Colombia, (http://hpc.uniandes.edu.co) for providing HPC resources that have contributed to the research results reported in this work.This work was funded by the Secretaria de Educación Superior, Ciencia, Tecnología e Innovación (SENESCYT) in the framework of the PIC-14-CENAIM-003 Project “Desarrollo e implementación de métodos de control y prevención de enfermedades en especies acuáticas de uso comercial y uso potencial en maricultura y repoblación”.
Author Contributions
A.R. designed this study. L.R. assembled the sequences and performed related experiments. L.R. analyzed the data and S.A. performed the phylogenetic analysis. L.R., L.B., I.B. and F.P. performed laboratory experiments. A.R. modified the manuscript. L.R. and B.B. wrote the manuscript. Contributed reagents/materials/analysis tools and acquired funding: B.B. and A.R. All authors reviewed the manuscript.
Data Availability Statement
The genome sequences used in the current study are available on the NCBI Genome Database under the accession number PRJNA412371.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-30903-x.
References
- 1.Hong X, Lu L, Xu D. Progress in research on acute hepatopancreatic necrosis disease (AHPND) Aquaculture International. 2016;24:577–593. doi: 10.1007/s10499-015-9948-x. [DOI] [Google Scholar]
- 2.De La Peña LD, et al. Acute hepatopancreatic necrosis disease (AHPND) outbreaks in Penaeus vannamei and P. monodon cultured in the Philippines. Dis. Aquat. Organ. 2015;116:251–254. doi: 10.3354/dao02919. [DOI] [PubMed] [Google Scholar]
- 3.Soto-Rodriguez SA, Gomez-Gil B, Lozano-Olvera R, Betancourt-Lozano M, Morales-Covarrubias MS. Field and experimental evidence of Vibrio parahaemolyticus as the causative agent of acute hepatopancreatic necrosis disease of cultured shrimp (Litopenaeus vannamei) in northwestern Mexico. Appl. Environ. Microbiol. 2015;81:1689–1699. doi: 10.1128/AEM.03610-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nunan L, Lightner D, Pantoja C, Gomez-Jimenez S. Detection of acute hepatopancreatic necrosis disease (AHPND) in Mexico. Dis. Aquat. Organ. 2014;111:81–86. doi: 10.3354/dao02776. [DOI] [PubMed] [Google Scholar]
- 5.Sawabe, T. et al. Updating the Vibrio clades defined by multilocus sequence phylogeny: Proposal of eight new clades, and the description of Vibrio tritonius sp. nov. Front. Microbiol. 4 (2013). [DOI] [PMC free article] [PubMed]
- 6.Tran L, et al. Determination of the infectious nature of the agent of acute hepatopancreatic necrosis syndrome affecting penaeid shrimp. Dis. Aquat. Organ. 2013;105:45–55. doi: 10.3354/dao02621. [DOI] [PubMed] [Google Scholar]
- 7.Han JE, Tang KFJ, Tran LH, Lightner DV. Photorhabdus insect-related (Pir) toxin-like genes in a plasmid of Vibrio parahaemolyticus, the causative agent of acute hepatopancreatic necrosis disease (AHPND) of shrimp. Dis. Aquat. Organ. 2015;113:33–40. doi: 10.3354/dao02830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Han JE, Tang KFJ, Lightner DV. Genotyping of virulence plasmid from Vibrio parahaemolyticus isolates causing acute hepatopancreatic necrosis disease in shrimp. Dis. Aquat. Organ. 2015;115:245–251. doi: 10.3354/dao02906. [DOI] [PubMed] [Google Scholar]
- 9.Gomez-Gil B, Soto-Rodríguez S, Lozano R, Betancourt-Lozano M. Draft Genome Sequence of Vibrio parahaemolyticus Strain M0605, Which Causes Severe Mortalities of Shrimps in Mexico. Genome Announc. 2014;2:e00055–14. doi: 10.1128/genomeA.00055-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yang Y, et al. Draft Genome Sequences of Four Strains of Vibrio parahaemolyticus, Three of Which Cause Early Mortality Syndrome/Acute. Genome. 2014;2:2–3. doi: 10.1128/genomeA.00816-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Restrepo L, Bayot B, Betancourt I, Pinzon A. Draft genome sequence of pathogenic bacteria Vibrio parahaemolyticus strain Ba94C2, associated with acute hepatopancreatic necrosis disease isolate from South America. Genomics Data. 2016;9:143–144. doi: 10.1016/j.gdata.2016.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Waterfield N, Kamita SG, Hammock BD, Ffrench-Constant R. The Photorhabdus Pir toxins are similar to a developmentally regulated insect protein but show no juvenile hormone esterase activity. FEMS Microbiol. Lett. 2005;245:47–52. doi: 10.1016/j.femsle.2005.02.018. [DOI] [PubMed] [Google Scholar]
- 13.Sawabe T, Kita-Tsukamoto K, Thompson FL. Inferring the evolutionary history of Vibrios by means of multilocus sequence analysis. J. Bacteriol. 2007;189:7932–7936. doi: 10.1128/JB.00693-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kondo H, Van PT, Dang LT. Draft Genome Sequence of Non-Vibrio parahaemolyticus Acute Diseased Shrimp in Vietnam. Genome Announc. 2015;3:2014–2015. doi: 10.1128/genomeA.00978-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu L, et al. Draft Genome Sequence of Vibrio owensii Strain SH-14, which causes shrimp acute hepatopancreatic necrosis disease. Genome Announc. 2015;3:3354. doi: 10.1128/genomeA.01395-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dong, X. et al. Complete genome sequence of Vibrio campbellii strain 20130629003S01 isolated from shrimp with acute hepatopancreatic necrosis disease. Gut Pathog. 9 (2017). [DOI] [PMC free article] [PubMed]
- 17.Hada HS, West PA, Lee JV, Stemmler J, Colwell RR. Vibrio tubiashii sp. nov., a pathogen of bivalve mollusks. Int J of Syst Bacteriol. 1984;34:1–4. doi: 10.1099/00207713-34-1-1. [DOI] [Google Scholar]
- 18.Gomez-Gil B, et al. Vibrio sinaloensis sp. nov., isolated from the spotted rose snapper, Lutjanus guttatus Steindachner, 1869. International journal of systematic and evolutionary microbiology. 2008;58:1621–1624. doi: 10.1099/ijs.0.65719-0. [DOI] [PubMed] [Google Scholar]
- 19.Xiao J, et al. Shrimp AHPND-causing plasmids encoding the PirAB toxins as mediated by pirAB-Tn903 are prevalent in various Vibrio species. Sci. Rep. 2017;7:42177. doi: 10.1038/srep42177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC bioinformatics. 2013;14:60. doi: 10.1186/1471-2105-14-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Meier-Kolthoff JP, Klenk HP, Göker M. Taxonomic use of DNA G+C content and DNA-DNA hybridization in the genomic age. Int J Syst Evol Microbiol. 2014;64:352–356. doi: 10.1099/ijs.0.056994-0. [DOI] [PubMed] [Google Scholar]
- 22.Prado S, Dubert J, Barja JL. Characterization of pathogenic vibrios isolated from bivalve hatcheries in Galicia, NW Atlantic coast of Spain. Description of Vibrio tubiashii subsp. europaensis subsp. nov. Syst Appl Microbiol. 2015;38:26–29. doi: 10.1016/j.syapm.2014.11.005. [DOI] [PubMed] [Google Scholar]
- 23.Richter M, Rosselló-Móra R. Shifting the genomic gold standard for the prokaryotic species definition. Proceedings of the National Academy of Sciences. 2009;45:19126–19131. doi: 10.1073/pnas.0906412106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Li X., Huang Y., Whitman W. B. The relationship of the whole genome sequence identity to DNA hybridization varies between genera of prokaryotes. Antonie van Leeuwenhoek 107241–249 (2015). [DOI] [PubMed]
- 25.Dubert J, et al. Reclassification of the larval pathogen for marine bivalves Vibrio tubiashii subsp. europaeus as Vibrio europaeus sp. Nov. International Journal of Systematic and Evolutionary Microbiology. 2016;66:4791–4796. doi: 10.1099/ijsem.0.001431. [DOI] [PubMed] [Google Scholar]
- 26.Richter M, Rossello-Mora R. Shifting the genomic gold standard for the prokaryotic species definition. Proc. Natl. Acad. Sci. 2009;106:19126–19131. doi: 10.1073/pnas.0906412106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ramasamy D, et al. A polyphasic strategy incorporating genomic data for the taxonomic description of novel bacterial species. International journal of systematic and evolutionary microbiology. 2014;64:384–391. doi: 10.1099/ijs.0.057091-0. [DOI] [PubMed] [Google Scholar]
- 28.Meier-Kolthoff JP, Göker M, Spröer C, Klenk HP. When should a DDH experiment be mandatory in microbial taxonomy? Archives of Microbiology. 2013;195:413–418. doi: 10.1007/s00203-013-0888-4. [DOI] [PubMed] [Google Scholar]
- 29.Kim M, Oh HS, Park SC, Chun J. Towards a taxonomic coherence between average nucleotide identity and 16S rRNA gene sequence similarity for species demarcation of prokaryotes. International journal of systematic and evolutionary microbiology. 2014;64:346–351. doi: 10.1099/ijs.0.059774-0. [DOI] [PubMed] [Google Scholar]
- 30.Thompson FL, et al. Vibrio fortis sp. nov. and Vibrio hepatarius sp. nov., isolated from aquatic animals and the marine environment. Int. J. Syst. Evol. Microbiol. 2003;53:1495–1501. doi: 10.1099/ijs.0.02658-0. [DOI] [PubMed] [Google Scholar]
- 31.Heidelberg JF, et al. DNA sequence of both chromosomes of the cholera pathogen Vibrio cholerae. Nature. 2000;406:477–483. doi: 10.1038/35020000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Makino K, et al. Genome sequence of Vibrio parahaemolyticus: A pathogenic mechanism distinct from that of V cholerae. Lancet. 2003;361:743–749. doi: 10.1016/S0140-6736(03)12659-1. [DOI] [PubMed] [Google Scholar]
- 33.Oliver JD. Wound infections caused by Vibrio vulnificus and other marine bacteria. Epidemiol. Infect. 2005;133:383–391. doi: 10.1017/S0950268805003894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Alsina M, Blanch AR. A set of keys for biochemical identification of environmental Vibrio species. J. Appl. Bacteriol. 1994;76:79–85. doi: 10.1111/j.1365-2672.1994.tb04419.x. [DOI] [PubMed] [Google Scholar]
- 35.Kembel SW, et al. Picante: R tools for integrating phylogenies and ecology. Bioinformatics. 2010;26:1463–1464. doi: 10.1093/bioinformatics/btq166. [DOI] [PubMed] [Google Scholar]
- 36.Tang S, et al. Genetic heterogeneity of the tropical abalone (Haliotis asinina) revealed by RAPD and microsatellite analyses. BMB Reports. 2005;38:182–190. doi: 10.5483/BMBRep.2005.38.2.182. [DOI] [PubMed] [Google Scholar]
- 37.Bell, T. A.A handbook of normal penaeid shrimp histology. World Aquaculture Society,Baton Rouge, LA 114 (1988).
- 38.Andrews, S. FQC: A quality control tool for high throughput sequence data. http://www.Bioinformatics.Babraham.Ac.Uk/Projects/Fastqc/http://www.bioinformatics.babraham.ac.uk/projects/ citeulike-article-id:11583827 (2010).
- 39.Bankevich A, et al. SPAdes: A New Genome Assembly Algorithm and Its Applications to Single-Cell Sequencing. J. Comput. Biol. 2012;19:455–477. doi: 10.1089/cmb.2012.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Galardini M, Biondi EG, Bazzicalupo M, Mengoni A. CONTIGuator: a bacterial genomes finishing tool for structural insights on draft genomes. Source Code Biol. Med. 2011;6:11. doi: 10.1186/1751-0473-6-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Altschul SF, et al. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Research. 1997;25:3389–3402. doi: 10.1093/nar/25.17.3389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Delcher AL, Bratke KA, Powers EC, Salzberg SL. Identifying bacterial genes and endosymbiont DNA with Glimmer. Bioinformatics. 2007;23:673–679. doi: 10.1093/bioinformatics/btm009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tatusova T, et al. NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res. 2016;44:6614–6624. doi: 10.1093/nar/gkw569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Aziz RK, et al. The RAST Server: Rapid Annotations using Subsystems Technology. BMC Genomics. 2008;9:75. doi: 10.1186/1471-2164-9-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lagesen K, et al. RNAmmer: Consistent and rapid annotation of ribosomal RNA genes. Nucleic Acids Res. 2007;35:3100–3108. doi: 10.1093/nar/gkm160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tatusov RL. The COG database: a tool for genome-scale analysis of protein functions and evolution. Nucleic Acids Res. 2000;28:33–36. doi: 10.1093/nar/28.1.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kanehisa M. The KEGG resource for deciphering the genome. Nucleic Acids Res. 2004;32:277D–280. doi: 10.1093/nar/gkh063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ashburner M, et al. Gene Ontology: tool for the unification of biology. Nat. Genet. 2000;25:25–29. doi: 10.1038/75556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013;30:2725–2729. doi: 10.1093/molbev/mst197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Darriba D, Taboada GL, Doallo R, Posada D. jModelTest 2: more models, new heuristics and parallel computing. Nat. Methods. 2012;9:772–772. doi: 10.1038/nmeth.2109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Posada, D. Using MODELTEST and PAUP* to Select a Model of Nucleotide Substitution. In Current Protocols in Bioinformatics 10.1002/0471250953.bi0605s00 (2003). [DOI] [PubMed]
- 52.Huelsenbeck JP, Ronquist F. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics. 2001;17:754–755. doi: 10.1093/bioinformatics/17.8.754. [DOI] [PubMed] [Google Scholar]
- 53.Clement M, Posada D, Crandall KA. TCS: A computer program to estimate gene genealogies. Mol. Ecol. 2000;9:1657–1659. doi: 10.1046/j.1365-294x.2000.01020.x. [DOI] [PubMed] [Google Scholar]
- 54. Huson, D. H., & Bryant, D. Estimating phylogenetic trees and networks using SplitsTree 4. Manuscript in preparation, software available from www. splitstree. Org (2005).
- 55.Darling ACE, Mau B, Blattner FR, Perna NT. Mauve: Multiple alignment of conserved genomic sequence with rearrangements. Genome Res. 2004;14:1394–1403. doi: 10.1101/gr.2289704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Krumsiek J, Arnold R, Rattei T. Gepard: a rapid and sensitive tool for creating dotplots on genome scale. Bioinformatics. 2007;23:1026–1028. doi: 10.1093/bioinformatics/btm039. [DOI] [PubMed] [Google Scholar]
- 57.Langille MG, Brinkman FS. IslandViewer: an integrated interface for computational identification and visualization of genomic islands. Bioinformatics. 2009;25:664–665. doi: 10.1093/bioinformatics/btp030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Auch AF, von Jan M, Klenk H-P, Göker M. Digital DNA-DNA hybridization for microbial species delineation by means of genome-to-genome sequence comparison. Stand. Genomic Sci. 2010;2:117–134. doi: 10.4056/sigs.531120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Stackebrandt E, et al. Report of the ad hoc committee for the re-evaluation of the species definition in bacteriology. Int. J. Syst. Evol. Microbiol. 2002;52:1043–1047. doi: 10.1099/00207713-52-3-1043. [DOI] [PubMed] [Google Scholar]
- 60.Kurtz S, et al. Versatile and open software for comparing large genomes. Genome Biol. 2004;5:R12. doi: 10.1186/gb-2004-5-2-r12. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1, Table S2, Table S3, Table S4, Table S5 and Table S6
Data Availability Statement
The genome sequences used in the current study are available on the NCBI Genome Database under the accession number PRJNA412371.