Abstract
Background
Recent work with long-term ethanol self-administration in non-human primate models has revealed a complex array of behavioral and physiological effects that closely mimic human alcohol abuse. Detailed neurophysiological analysis in these models suggests a myriad of pre- and postsynaptic neurobiological effects that may contribute to the behavioral manifestations of long-term ethanol drinking. The molecular mechanisms regulating presynaptic effects of this chronic ethanol exposure are largely unknown. To this end, we analyzed the effects of long-term ethanol self-administration on the levels of presynaptic SNARE complex proteins in Mucacca mullatta basolateral amygdala, a brain region known to regulate both aversive and reward-seeking behaviors.
Methods
Basolateral amygdala samples from control and ethanol-drinking male and female monkeys were processed. Total basolateral amygdala protein was analyzed by Western blotting using antibodies directed against both core-SNARE and SNARE-associated proteins. We also performed correlational analyses between protein expression levels and a number of ethanol drinking parameters, including life-time grams of ethanol consumed, preference, and blood ethanol concentration.
Results
Significant interactions or main effects of sex/drinking were seen for a number of SNARE core and SNARE-associated proteins. Across the range of ethanol drinking phenotypes, SNAP25 and Munc13-1 proteins levels were significantly different between males and females; and Munc13-2 levels were significantly lower in animals with a history of ethanol drinking. A separate analysis of very-heavy drinking individuals revealed significant decreases in Rab3c (females) and Complexin 2 (males).
Conclusions
Protein expression analysis of basolateral amygdala total protein from controls and animals following long-term ethanol self-administration suggest a number of alterations in core SNARE or SNARE-associated components that could dramatically alter presynaptic function. A number of proteins or multi-protein components were also correlated with ethanol drinking behavior, which suggest a potentially heritable role for presynaptic SNARE proteins.
Keywords: Macaque, Rab3a/c, Munc13-1/2, Complexin1/2, Munc18-1
Introduction
Non-human primate ethanol drinking models closely parallel many aspects of human alcohol use/misuse. These parallels include the enhanced vulnerability of late adolescent animals (Baker et al., 2017, Helms et al., 2014) and the ability of drinking behaviors during initial exposures to predict future consumption patterns (Baker et al., 2017). Although the neurophysiological mechanisms controlling these behaviors are poorly appreciated at present, neurophysiological studies from the brain slices of long-term, ethanol-drinking and control monkeys reveal a variety of alterations to neurotransmitter systems that parallel extensive work in rodents. Notably, many of these alterations are region specific but involve changes to both postsynaptic function and presynaptic neurotransmitter release. For example, ethanol drinking by adult male cynomolgus macaques increases dopamine release in the nucleus accumbens but not dorsolateral striatum/putamen (Siciliano et al., 2015). Ethanol-drinking likewise increases both the frequency and amplitude of spontaneous glutamatergic synaptic events in the orbitofrontal cortex (Nimitvilai et al., 2017) but appears to only upregulate glutamate release in the putamen (Cuzon Carlson et al., 2011). Data on GABAergic alterations following long-term drinking is more mixed with both increased and decreased GABA release reported in different brain regions (Cuzon Carlson et al., 2011, Pleil et al., 2016). Potential molecular mechanisms governing these presynaptic effects of long-term ethanol exposure are currently unknown in non-human primates and have been less frequently explored in other models.
Recent work in the rat lateral/basolateral amygdala suggests that ethanol exposure robustly modulates presynaptic function at glutamatergic and GABAergic synapses. The lateral/basolateral amygdala serves as the primary input nucleus for the extended amygdala. It receives sensory-, cognitive-, and memory-related glutamatergic inputs from numerous brain regions. Glutamatergic principal neurons in this region project both to well-studied components of the extended amygdala during regulation of innate anxiety and conditioned fear as well as to cognitive- and reward-related brain regions during reward-seeking behaviors (Janak and Tye, 2015). Principal neuron activity is tightly controlled by at least two populations of GABAergic interneurons that include ‘feedback’ cells scattered throughout the nuclei and ‘feedforward’ neurons found locally as well as concentrated with intercalated cell ‘islands’ found along the lateral boundary (Palomares-Castillo et al., 2012). Acute ethanol facilitates GABAergic synaptic function from these interneuron populations through distinct pre- (‘feedback’) and post-synaptic (‘feedforward’) mechanisms (Silberman et al., 2008). On the other hand, chronic ethanol reduces release probability from ‘feedforward’ GABA synapses without altering presynaptic function at inputs from ‘feedback’ interneurons (Diaz et al., 2011). Chronic ethanol also dramatically up-regulates presynaptic glutamate release onto lateral/basolateral amygdala neurons but only at stria terminalis synapses coursing into the region along the medial boundary (Christian et al., 2013). Recent work in this brain region shows that acute ethanol can modulate the recycling of presynaptic vesicles at glutamatergic presynaptic vesicles without dramatically altering basal release probability (Gioia and McCool, 2017). This process involves a Munc13 proteins (see below, (Gioia et al., 2017)) that likely reflects direct interactions between these proteins and ethanol (Das et al., 2013). Other than the role of Munc13 proteins, the effects of either acute or chronic ethanol on presynaptic release machinery have not been extensively explored.
The core SNARE complex mediates vesicular release at all ‘fast’ neurotransmitter release sites (Sudhof, 2013). Critical components within the complex include the syntaxins, which act as T-SNARE proteins associated with the presynaptic plasma membrane, and the VAMP proteins (also called synaptobrevins), which are intrinsic vesicular proteins (V-SNARE) that directly bind to syntaxins to anchor synaptic vesicles to release sites. SNAP 25, a syntaxin-associated protein, and the complexins mediate these T- and V-SNARE interactions during the fusion process by acting as priming/fusion co-factors. Importantly, proteomic analysis of orbitofrontal cortical samples identified significant increases in both the syntaxins and SNAP25 following long-term ethanol-drinking by adult male monkeys (Nimitvilai et al., 2017). An ancillary although critical protein in this core complex is Munc18-1 which binds to and converts syntaxin to a ‘release-competent’ form during the priming process. PKC-dependent phosphorylation of Munc18-1 facilitates this conversion process (Wierda et al., 2007). All of these proteins are widely expressed in the central nervous system although there is regional specificity for some isoforms. For example, VAMP1 has higher relative expression in thalamic, midbrain, and cerebellar structures compared to VAMP2 (Allen Brain Atlas).
Like vesicular fusion, vesicular priming at presynaptic release sites is a highly orchestrated process mediated by numerous protein-protein interactions (James and Martin, 2013). For example, Munc13 proteins mediate initial interactions between synataxin and vesicle membrane-associated Rab3 GTAases via interactions with the Rab3-binding protein, RIM1α (Lu et al., 2006). Munc13 proteins thus facilitate vesicle priming and stay associated with primed vesicles to modulate release via direct interactions with Munc18-1 and syntaxin. Munc13-1 is present in most synapses while Munc13-2 is more enriched in cortical structures (Augustin et al., 1999). There is a mixed literature on exact role of Munc13-2 showing both dominant-negative and positive roles for vesicle priming depending upon the synapse and brain region (Chen et al., 2013, Rosenmund et al., 2002, Breustedt et al., 2010). This disparity may reflect the relative expression and distinct roles of known splice variants for Munc13-2 that include the ubiquitous isoform (uMun13-2), which closely resembles Munc13-1 in both form and function, and the brain-specific isoform (bMunc13-2), which replaces important priming-promoting regions of the protein with negative regulatory sequences.
In summary, previous neurophysiological studies on brain slices from ethanol-drinking monkeys and chronic ethanol-exposed rodents suggest that modulation of presynaptic neurotransmitter release is a common finding across these models; and, there are suggestions that long-term ethanol drinking can shift the abundance of some SNARE-related proteins (Nimitvilai et al., 2017). The purpose of the current work is to offer a more detailed examination of the impact of ethanol drinking in both male and female non-human primates on the relative expression of vesicle priming and release proteins. We focus these efforts on monkey lateral amygdala given the central role of this brain region in regulating emotional behaviors related to aversion and reward-seeking.
Methods
Tissue Samples
Lateral amygdala tissue samples were obtained from the Monkey Alcohol Tissue Research Resource (www.matrr.com; (Daunais et al., 2014)). The dissections were performed using MRI-guided dissection (Daunais et al., 2010). The brain was blocked into 4 mm slabs beginning at the rostral tips of the temporal poles. At the level of the caudal caudate/putamen and globus pallidus internal and external segments, the amygdaloid complex is visible on the face of a single block. The lateral nucleus is clearly distinguished from surrounding structures in this complex; it is lateral and well ventral to the central nucleus and lateral to the basolateral/accessory basal nuclei which are clearly demarcated by robust white matter tracts within those subdivisions. Dissections thus stayed within the margin of the lateral nucleus. Male and female individuals from separate cohorts (Females – cohorts 6a and 6b; Males – cohorts 5 and 7a) chosen based on similar drinking experiences (below), ages of the individuals (4–6 years old at the beginning of the exposure), and broad representation of drinking phenotypes as defined previously for these cohorts (Baker et al., 2017). These phenotypes included binge drinkers (“BD”), light drinkers (“LD”), heavy drinkers (“HD”), and very heavy drinkers (“VHD”) which denote categorical classifications based on average daily intake (g/kg/day), stable intake patterns, and average blood ethanol concentration (BEC; mg/dL) (Baker et al., 2014). Experimentally naïve animals within each cohort served as controls; they consumed the same diet and self-administered water and an isocaloric maltose-dextrin solution under identical operant conditions (Grant et al., 2008, Baker et al., 2014).
Long-term ethanol self-administration was established as previously described (Grant et al., 2008). Briefly, monkeys experienced a 3 month-long induction period where banana-flavored food pellets were delivered on a fixed-time interval schedule along with the availability of water and then 4% (w/v) ethanol solution in water. Consumption was precisely limited such that total intake increased incrementally from 0.5 g/kg/day, 1.0 g/kg/day, and finally 1.5 g/kg/day over thirty consecutive days for each intake limit. Following this induction period, all cohorts were given 12 months continuous access (22h/day) of ethanol referred to as open-access self-administration. A period of 2–3 weeks followed the first six months of open-access to allow for MRI brain imaging and endocrine assessment profiling where ethanol continued to be available, but the sessions were shortened to accommodate the repeated blood sampling during this time (Porcu et al., 2006). The final 6 months of drinking was also followed by a period of shortened sessions for MRI and endocrine measures, and then a return to normal 22 h open-access sessions at least 3 full weeks prior to necropsy. The drinking data from the combined 6 and 12 months of open-access self-administration as well as categorical classifications for individual animals are shown in Table 1. Blood alcohol concentrations were determined from saphenous vein blood samples as previously described (Grant et al., 2008).
Table 1. Drinking Data.
Drinking data from individual monkeys used in the current work. (a) Cohort numbers, monkey IDs, and drinking data are from the Monkey Alcohol Tissue Research Resource (MATRR, www.matrr.com). (b) Voluntary intake represents total grams consumed during the entire 12month, free-choice, open-access period (“gE”, see text). (c) Blood draws for BAC determinations were performed approximately 7h into the drinking sessions as described previously (Vivian et al., 2001). (d) Drinking phenotype sas defined by Grant et al. 2008 and denoted on the MATRR website. “VHD”, very heavy drinker. “HD”, heavy drinker. “LD”, light drinker. “BD”, binge drinker. (e) Data for g/kg/day previously reported in Jimenez et al. 2015.
| Monkey ID#a | Voluntary Intakeb (g) | Average Daily Intake (g/kg) | Average BAC (mg/dL)c | Drinking Phenotyped |
|---|---|---|---|---|
| Females | ||||
| Cohort 6a | ||||
| 10069 | 1699.3 | 5.1 | 99.4 | VHD |
| 10070 | 1286.2 | 3.9 | 49.1 | VHD |
| 10078 | 1615.0 | 4.9 | 79.8 | VHD |
| 10079 | 1301.9 | 3.9 | 66.5 | VHD |
| 10080 | 1083.1 | 3.3 | 41.4 | VHD |
| 10081 | 1319.8 | 4.0 | 61.0 | VHD |
| Cohort 6b | ||||
| 10072e | 355.0 | 1.1 | 6.1 | LD |
| 10073e | 1342.7 | 4.0 | 111.2 | VHD |
| 10074e | 585.3 | 1.7 | 7.7 | LD |
| 10075e | 953.0 | 2.7 | 33.9 | HD |
| 10077e | 457.3 | 1.3 | 11.1 | LD |
| Males | ||||
| Cohort 5 | ||||
| 10060 | 911.3 | 2.6 | 90.1 | BD |
| 10061 | 1318.7 | 3.8 | 116.9 | VHD |
| 10062 | 1458.3 | 4.1 | 136.7 | VHD |
| 10063 | 1108.1 | 3.1 | 43.7 | VHD |
| 10064 | 977.25 | 2.7 | 98.1 | HD |
| 10065 | 1106.7 | 3.1 | 87.2 | HD |
| 10066 | 1160.5 | 3.3 | 141.2 | VHD |
| 10067 | 1077.5 | 3.1 | 103.0 | HD |
| Cohort 7a | ||||
| 10087 | 822.5 | 2.3 | 33.2 | BD |
| 10088 | 989.7 | 2.8 | 66.6 | HD |
| 10089 | 685.9 | 2.0 | 32.5 | LD |
| 10090 | 637.9 | 1.8 | 35.9 | LD |
| 10091 | 1085.2 | 3.1 | 73.0 | VHD |
| 10092 | 650.1 | 1.9 | 20.6 | LD |
| 10097 | 1055.3 | 3.0 | 74.8 | HD |
| 10098 | 1158.7 | 3.3 | 95.9 | VHD |
Sample Preparation
20 mg rhesus lateral amygdala tissue was homogenized in T-PER Tissue Protein Extraction Reagent (#78510; Thermo Fisher, Waltham, MA) containing protease (#P8340; Sigma, St. Louis, MO) and phosphatase inhibitors (P5726 and PP0040; Sigma, St. Louis, MO) using brief sonication. Following two hour incubation on a rotisserie mixer at 4° C, samples were centrifuged at 15,000 x g to isolate the supernatant. Protein yield was quantified using Pierce BCA protein assay kit (#PI23227; Thermo Fisher, St. Louis, MO). Optical densities from the BCA reaction were read on a SpectraMax 384 Plus Spectrophotometer and compared to known protein concentrations using SoftMax Pro Software (Molecular Devices, Sunnyvale, CA). Analysis of lateral amygdala samples was randomized across different sides of the brain in the current study.
Western Analysis
Five to fifteen micrograms of total protein was loaded onto 4 – 20% Criterion TGX 26-well precast gels (#567-1095; Bio-Rad, Hercules, CA) along with Precision Plus Protein Dual Xtra Standards (#161-0377; Bio-Rad, Hercules, CA), separated per manufacturer’s recommendations in a Bio-Rad Criterion Vertical Electrophoresis Cell. Samples were loaded in pseudorandom order with respect to control or ethanol drinking; and each gel contained representative cohorts from both sexes. Gels were transferred to nitrocellulose membrane using a semi-dry technique with a Thermo Fisher iBlot Gel Transfer Device and pre-assembled membrane stacks (#IB301001; Thermo Fisher, Waltham, MA). After transfer, membranes were incubated in blocking buffer containing Tris Buffered Saline, 0.05% Tween-20, and 5% non-fat milk or, for detection of phosphorylated proteins, Pierce Protein Free TBS Blocking Buffer (#0037570; Thermo Fisher, Waltham, MA) for 1 hour at room temperature with agitation. Subsequently, membranes were incubated with agitation at 4° C overnight in a 1% solution of blocking buffer containing primary antibody for specific protein targets (Table 2). Following extensive rinsing in Tris Buffered Saline/0.05% Tween-20, the membranes were exposed to peroxidase-labeled goat anti-rabbit or goat anti-mouse secondary antibody (#A4914 or #A8924 respectively; Sigma, St. Louis, MO). Detection of bound secondary antibody was performed using SuperSignal West Dura Extended Duration Substrate Enhanced Chemiluminescence (#34076; Thermo Fisher, Waltham, MA). To control for loading differences between samples, membranes were stripped with Restore Plus Western Blot Stripping Buffer (#46430; Thermo Fisher, Waltham, MA) and probed with mouse monoclonal antibody directed against β-actin, 1:10,000 dilution (#MAB1501; Millipore, Burlington, MA) followed by peroxidase-labeled goat anti-mouse secondary antibody, 1:10,000 dilution. Immunoreactive band intensity was captured from digital images with sequential exposure durations using a Bio-Rad Chemi-Doc XRS Imaging System and analyzed with Quantity One Analysis software (Bio-Rad, Hercules, CA). To normalize band intensity across different gels, band intensities within each sample were normalized to control samples within each sex and adjusted by simple algebraic scaling across different blots. Results are therefore reported as arbitrary units. Data are presented as mean±SEM.
Table 2. Antibodies Used.
Antibodies used in the Western analysis of SNARE complex and complex-associated proteins. Manufacturers, catalogue numbers, and conditions within the western analysis are noted.
| Target (Predicted Size) | Manufacturer / Cat# | 1° Ab Dilution | 2° Ab Dilution |
|---|---|---|---|
| Complexin 1,2 (15kD) | Synaptic Systems / #122 002 | 1:500 | 1:1500 |
| Munc13-1 (220kD) | Synaptic Systems / #126 102 | 1:1000 | 1:1500 |
| Mun18-1 (70kD) | Cell Signaling / #13414 | 1:5000 | 1:3000 |
| Munc18-1 (p-Ser 515) | Thermo Scientific /#PA1-4689 | 1:1000 | 1:1500 |
| Rab 3a (28kD) | Synaptic Systems / #107 111 | 1:2500 | 1:1500 |
| Rab3c (28kD) | Synaptic Systems / #107 203 | 1:1000 | 1:1500 |
| SNAP25 (25kD) | Synaptic Systems / #111 002 | 1:10000 | 1:5000 |
| Synataxin 1 (38kD) | Millipore /#AB5820 | 1:10000 | 1:5000 |
| uMunc13-2 (220kD) | Thermo Scientific /#PA5-33159 | 1:5000 | 1:3000 |
| VAMP1 (15kD) | Abcam / #ab3346 | 0.5μg/mL | 1:1500 |
| VAMP2 (18kD) | Abcam / #ab3347 | 1:1000 | 1:1500 |
| vGluT1 (66kD) | Synaptic Systems / #135 303 | 1:40000 | 1:7500 |
| vGluT2 (66kD) | Synaptic Systems / #135 403 | 1:5000 | 1:5000 |
Statistical Analysis
Standard two-way ANOVA was used to analyze effects of ethanol drinking on protein expression levels using drinking history (control or ethanol drinking across phenotypes) and sex (male or female) as the main factors. Bonferroni multiple comparison post-hoc tests were used to define significant effects of drinking within each sex (GraphPad Prism, GraphPad Software Inc.). Using this dataset, we also performed select comparisons between control and very heavy drinking individuals to compliment recent work showing that induction-phase consumption patterns predict the drinking phenotypes during free access (Baker et al., 2017, Baker et al., 2014) and the identification of differentially methylated genes in the nucleus accumbens across these phenotypes which are related to synaptic plasticity including several presynaptic proteins (Cervera-Juanes et al., 2017a). We continued to use two-way ANOVA across phenotype and sex as main factors along with Bonferroni multiple comparison post-hoc tests. For correlational analyses with individual proteins, standard linear regressions were performed within sex for expression level versus drinking variable (see results; GraphPad Prism). To identify specific drinking variables used in these correlations, Pearson product moment analysis for the different drinking variables was used (SigmaPlot, Systat Software Inc.). For correlations involving multiple proteins (see results), forward step-wise multiple regressions were performed within each drinking variable (SigmaPlot). For all analyses, p-values less than 0.05 were considered significant.
Results
Analysis of the Drinking Data
In our analysis of the drinking data, we focused on grams of ethanol voluntarily consumed across the experiment (total grams ethanol, gE), daily average consumption (g/kg), and average blood ethanol concentration measured in samples taken 7h after the start of the daily session (BEC, mg/dL). In ethanol-drinking females (n=11), Pearson product moment analysis across these drinking variables indicated significant correlations between all three variables: gE and BEC (r=0.901, p<0.001); gE and average daily g/kg (r=0.999, p<0.001); and BEC and average daily g/kg (r=0.889, p<0.001). There were also significant correlations between all three variables in ethanol-drinking males (n=16): gE and BEC (r=0.830, p<0.001); gE and average daily g/kg (r=0.999, p<0.001); and BEC and average daily g/kg (r=0.829, p<0.001). However, within the male drinkers, multiple linear regression analysis suggested that lifetime voluntary drinking strongly predicted average daily g/kg (p<0.001) but not BEC (p≫0.05). For this reason, we confined our subsequent correlation analysis between protein expression levels across both sexes to lifetime voluntary grams of ethanol (gE) and BEC (mg/dL).
Vesicle-priming Proteins
We artificially divided presynaptic proteins into those primarily associated with vesicle priming and those more directly involved with vesicle release (see Fig. 5). We subsequently used two-way analysis of variance with sex and drinking history as the main factors to assess the effects of long-term ethanol drinking on protein expression levels. For priming proteins, there was no significant interaction between the main factors and no significant main effects for either sex or drinking for Rab3c, vGluT1, and vGluT2 (Fig. 1B, 1E, and 1F). However, for both Rab3a (F=4.234, p<0.05, Fig. 1A) and Munc13-2 (F=4.985, p<0.05, Fig. 1D), there was a significant main effect of ethanol drinking history in both sexes. This suggests that long-term ethanol drinking by rhesus macaques reduces levels of these proteins relative to control monkeys. However, the effect sizes were relatively modest. Rab3a levels were reduced by only ~10% in both sexes; and, Munc13-2 levels were reduced by ~10% in male ethanol drinkers and by ~23% in females. Bonferroni’s multiple comparison post-test did not identify significant effects of ethanol drinking within either sex. In contrast to these significant main effects of ethanol drinking, there was significant main effects of sex for Munc13-1 (F=15.94, p<0.001; Fig. 1C) with protein levels in female controls being ~50% greater than that of male controls.
Figure 5. Functional relationships between presynaptic vesicle priming and release proteins from lateral amygdala samples examined in the current manuscript.
Parenthetical statements indicate significant main effects, either sex or ethanol drinking, sound in the western analysis. Asterisks denote proteins significantly correlated with either total voluntary ethanol consumption or average blood alcohol concentration (see text).
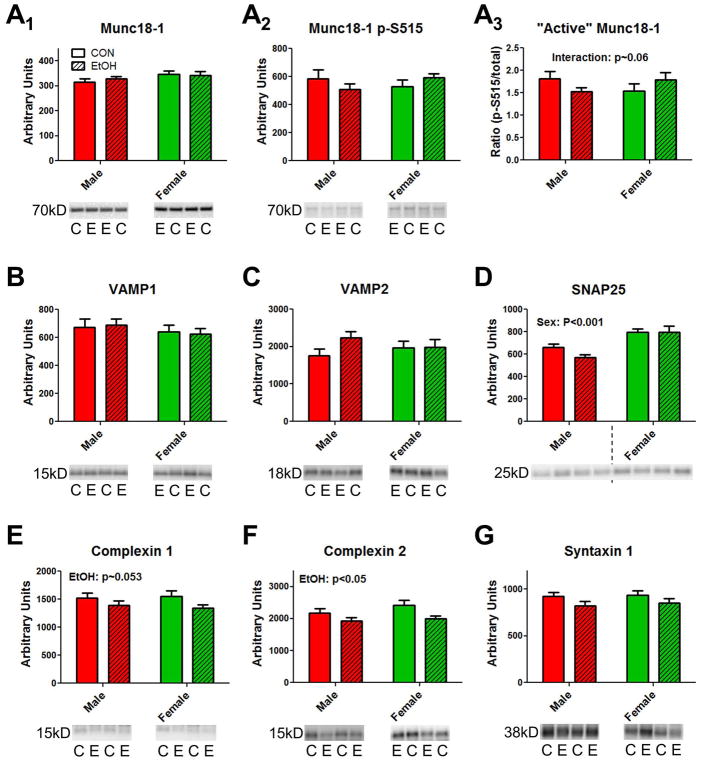
Figure 1. Ethanol drinking and sex alter expression levels for some proteins involved in vesicle priming.
Exemplar blots for each protein are shown beneath the bar graphs with approximate molecular weights of the target indicated to the left. “C” and “E” denote lateral amygdala samples prepared from non-drinking controls (also denoted as “CON”) and ethanol drinkers (also “EtOH”), respectively. Protein expression level is denoted as ‘arbitrary units’ (see Methods). (A) Rab3a protein expression is significantly reduced in samples from ethanol drinkers (two-way ANOVA; main effect of ethanol, p<0.05, see text) regardless of sex. (B) No significant effects of either ethanol drinking or sex on Rab3c protein expression. (C) Munc13-1 protein expression is significantly higher in samples prepared from females compared to males (main effect of sex, p<0.001, see text). Dashed line separates male and female samples in an exemplar blot. (D) Ethanol drinking significantly decreases protein expression of Munc13-2 (main effect of ethanol, p<0.05). (E) No significant effect of ethanol drinking or sex on vGluT1 protein expression. (F) No significant effect of ethanol drinking or sex on vGluT2 protein expression.
Recent work with non-human primate tissues collected following long-term ethanol self-administrations suggests unique cellular and molecular effects within tissues isolated from individuals with distinct ethanol drinking phenotypes (Cuzon Carlson et al., 2011, Cervera-Juanes et al., 2017b, Cervera-Juanes et al., 2017a). In the cohorts used in the current study, very heavy drinking phenotypes (VHD) are well represented among male and female drinkers (n=6 and 7, respectively; Table 1). We therefore performed a selective comparison between control (CON) and VHD individuals across the proteins associated with vesicle priming (Table 3). Expression of Rab3a, Munc13-2, vGluT1, and vGluT2 proteins yielded similar results to the two-way analysis across all drinking phenotypes. However, comparisons of Rab3c and Munc13-1 protein levels within CON and VHD individuals revealed novel effects of long-term ethanol self-administration in VHD individuals. For Rab3c, there were novel and significant main effects of both sex (F=9.41, P<0.01) and VHD phenotype (F=5.22, P<0.05) but no significant interaction between factors (F=1.39, P>0.05). Bonferroni’s multiple comparison tests across the CON and VHD groups revealed a significant decrease in Rab3c protein levels in females (~30% decrease, t=2.43, P<0.05) but not males (~8% decrease, t=0.79, P>0.05). The selective Munc13-1 analysis comparing CON and VHD individuals identified a significant difference in protein levels between males and females (F=14.5, P<0.001) in addition to a novel main effect of drinking in the VHD phenotype (F=5.29, P<0.05). Notably, these significant differences were not identified in the analysis combining all drinkers.
Table 3. Selected Comparisons Between Control and Very Heavy Drinking (VHD) Individualsa.
Results from the select comparisons between control and very heavy drinking phenotypes. Numbers represent mean±SEM band intensities from each treatment group (see Methods). Bold text denotes significant findings distinct from those identified across all ethanol drinking phenotypes (see Fig. 1 & 3). (a) Very heavy drinking (VHD) individuals were identified based criteria from Baker et al. 2014 (see text). (b) Expression levels represented as arbitrary units (see Methods for details). (c) Two-way ANOVA with sex and drinking phenotype (CON versus VHD) as main factors. Only statistically significant effects are indicated with “n.s.” indicating ‘not significant’. Asterisks (*) denote P<0.05 using Bonferroni multiple comparison post-test comparing control (CON) versus VHD. (d) Parentheses indicate number of individual animals represented in each group.
| Proteinb | Males | Females | Statisticsc | ||
|---|---|---|---|---|---|
| CON (9)d | VHD (6) | CON (6) | VHD (7) | ||
| Vesicle Priming | |||||
| Rab3a | 728.7±28.8 | 636.0±38.0 | 756.3±23.0 | 667.0±48.9 | VHD: P<0.05 |
| Rab3c | 774.0±51.6 | 703.8±50.6 | 662.4±56.3 | 518.8±71.1* |
Sex: P<0.01 VHD: P<0.05 |
| Munc13-1 | 524.6±86.1 | 336.4±69.5 | 787.8±80.2 | 636.0±22.2 |
Sex: P<0.001 VHD: P<0.05 |
| Munc13-2 | 709.1±58.5 | 654.7±37.2 | 781.5±75.6 | 738.3±77.1 | n.s. |
| vGluT1 | 614.1±87.2 | 611.3±31.4 | 663.3±41.7 | 669.7±115.2 | n.s. |
| vGluT2 | 3519±383 | 4047±548 | 4418±754 | 3415±487 | n.s. |
| Vesicle Release | |||||
| Munc18-1 | 316.3±12.5 | 323.0±12.5 | 347.8±13.7 | 350.8±17.2 | n.s. |
| pMunc18-1 (Ser515) | 584.3±65.5 | 486.1±47.0 | 527.0±49.8 | 597.2±39.1 | n.s. |
| “Active” Munc18-1 | 1.82±0.16 | 1.49±0.10 | 1.54±0.17 | 1.74±0.16 | n.s. |
| VAMP1 | 673.5±57.8 | 729.8±72.6 | 642.7±46.8 | 567.6±49.2 | n.s. |
| VAMP2 | 1759±175 | 2605±319* | 1966±177 | 1670±229 | Interaction: P<0.05 |
| SNAP25 | 659.9±30.0 | 573.8±49.0 | 793.9±29.9 | 713.2±42.4 |
Sex: P<0.01 VHD: P<0.05 |
| Complexin1 | 1526±89 | 1299±119 | 1556±101 | 1342±88 | VHD: P<0.05 |
| Complexin2 | 2178±129 | 1711±82* | 2419±162 | 2045±125 |
Sex: P<0.05 VHD: P<0.01 |
| Syntaxin1 | 924.5±41.8 | 826.8±99.6 | 937.1±45.5 | 825.9±93.4 | n.s. |
While ethanol drinking and sex had influenced expression levels for some proteins related to vesicle priming, it is also plausible that the relative expression levels of these proteins themselves contribute to drinking-related behaviors. We therefore performed correlation analysis between protein expression levels and both lifetime voluntary grams of ethanol and BEC. We restricted our analysis to proteins with no main effect of ethanol drinking across all drinking phenotypes. In female drinkers (green lines, Fig. 2), there was a modest negative correlation between the expression levels of Rab3c and voluntary ethanol consumption (g) during the experiment (R2=0.4, F=6.115, p<0.05, Fig. 2A1). In male drinkers (red lines, Fig. 2), there were also modest negative correlations between the average BEC (mg/dL) during the twelve month drinking period and the expression levels of Munc13-1 (R2=0.35, F=7.48, p<0.05, Fig. 2B2) and vGluT2 (R2=0.32, F=6.598, p<0.05, Fig. 2D2). There were no significant correlation between vGluT1 protein levels and either voluntary consumption (g) or BEC in either sex. These data suggest modest associations between the relative levels of Rab3c, Munc13-1, and vGluT2 and either lifetime consumption in females (Rab3c) or BEC in males (Munc13-1 and vGluT2).
Figure 2. Correlational analysis between protein expression and drinking variables for proteins involved with vesicle priming.
Correlations were performed between protein expression levels (arbitrary units) and lifetime ethanol intake during the voluntary consumption periods (in grams, left column; see Methods) or average blood alcohol concentration (mg%; right column) only for those proteins without a significant main effect of ethanol drinking (Fig. 1). Protein levels from male (red lines and symbols) and female samples (green lines and symbols) are shown separately. Statistically significant correlations are denoted by R2 and P values; and 90% confidence limits are indicated by the dashed lines. (A) Protein levels of Rab3c were significantly correlated (slope = −0.33±0.13, F=6.115) with total voluntary ethanol intake but not average BAC in females. (B) Munc13-1 protein expression levels were significantly correlated with average BAC (slope = −3.12±1.14, F=7.48) but not total voluntary intake in males. (C) There is no significant correlation between vGluT1 protein levels and either voluntary consumption or BAC. (D) vGluT2 protein levels are significantly correlated with BAC (slope=−27.2±10.6, F=6.6), but not ethanol intake, in lateral amygdala from male drinkers.
Fusion-complex Proteins
With respect to proteins more directly involved in vesicle release, two-way ANOVA revealed no significant interactions between the sex and ethanol drinking main factors for most of these proteins (Fig. 3). However, using the ratio of S515-phosphorylated Munc18-1 to total Munc18-1 protein levels as a proxy ‘active’ Munc18-1 (Sassa et al., 1996), we found a trend for an interaction between ethanol drinking and sex (F=4.079; p~0.06, Fig. 3A3) with ethanol drinking tending to decrease levels in males and increase levels in females. There was a significant main effect of drinking history for Complexin 2 (F=7.03, p<0.05, Fig. 3F) and a trend for Complexin 1 (F=4.003, p~0.053, Fig. 3E). Effect sizes were again modest with ethanol drinking decreasing Complexin 2 levels by ~12% in males and ~17% in females with no significant differences between control and ethanol drinking in either sex (Bonferroni multiple comparisons test, p>0.05). We also found a significant main effect of sex for SNAP25 (F=19.9, p<0.001, Fig. 3D) with females expressing higher levels than control males by ~20%. There was no significant main effect across either factor for Munc18-1, VAMP1/2, or Syntaxin 1 protein levels. Like priming-related proteins, these data suggest modest effects of ethanol drinking on Complexin 2 and potentially Complexin 1 and ‘active’ Munc18-1.
Figure 3. Ethanol drinking and sex alter the expression of lateral amygdala proteins involved with presynaptic vesicle release.
Notations for the exemplar blots are the same as for Figure 1. (A) Total protein levels for Munc18-1 (A1) and for Munc18-1 phosphorylated on Ser515 (A2) were not significantly altered by either sex or ethanol drinking. However, when ‘active’ Munc18-1 was calculated as the ratio between pSer515 Munc18-1 divided by total Munc18-1 (A3), there was a trend for an interaction between the ethanol drinking and sex main factors. (B & C) No significant effect of either sex or ethanol drinking on VAMP1 or VAMP2 protein levels. (D) SNAP25 protein levels were significantly higher in female lateral amygdala samples compared to male samples (two-way ANOVA, P<0.001 across the sex main factor). (E & F) Ethanol drinking significantly reduced protein levels for Complexin 2 (P<0.05) with a trend for Complexin 1 levels. (G) No significant effect of either sex or ethanol drinking on Syntaxin 1 protein levels in lateral amygdala samples.
Our selective comparison of expression level for vesicle release proteins across CON and VHD animals again revealed potentially novel effects of ethanol drinking among those individuals with the highest consumption levels. For example, the CON/VHD comparison revealed a significant interaction between sex and phenotype for the VAMP2 protein (Table 3, F=6.35, P<0.05). Compared to controls, VAMP 2 protein levels were significantly higher in male VHD individuals (~48%, t=2.72, P<0.05, Bonferroni’s multiple comparison post-test) and lower in female VHDs (~15% inhibition, t=0.90, P>0.05). This analysis also found significant, novel main effects of the VHD drinking phenotype for SNAP25 (F=4.76, P<0.05) and Complexin 1 (F=4.91, P<0.05); but these effect sizes were modest (10–15% decrease relative to CON) and were not significant in the post-test. The CON/VHD comparison also revealed a novel and significant main effect of sex on Complexin 2 protein levels (F=4.76, P<0.05) but no interaction between sex and drinking phenotype (F=0.12, P>0.05). Notably, the main effect of the VHD phenotype on Complexin 2 that was also present in the analysis across the range of drinking phenotypes was now characterized by a significant decrease in protein levels in male (~21%, t=2.58, P<0.05, Bonferroni post-test) but not female VHD individuals (~15% inhibition, t=1.96, P>0.05). This selective comparison between controls and VHD ethanol animals did not find any significant effects on protein expression for Munc18-1, Ser515-phosphorylated Munc18-1, VAMP 1, or Syntaxin 1.
To examine associations between protein expression levels and drinking behaviors, we again performed correlations for individual proteins not significantly altered by drinking history. In female drinkers (green lines, Fig. 4), there were no significant correlations between any of the fusion-related proteins and either total voluntary consumption (g) or BEC (mg/dL). However, in male drinkers (red lines, Fig. 4), there was a significant positive correlation between total voluntary consumption and VAMP2 proteins levels (R2=0.45, p<0.01, Fig. 4D1) as well as a modest negative correlation between BEC and Syntaxin 1 protein levels (R2=0.26, p<0.05, Fig. 4F2)
Figure 4. Correlational analyses for protein expression levels versus ethanol drinking variables reveals significant relationships for VAMP2 and Syntaxin 1.
(A & B) No significant relationship between total Munc18-1 protein levels or phosphorylated Munc18-1 and either total voluntary consumption or BAC. (C) No correlation between VAMP1 levels and either ethanol intake of BAC. (D) VAMP2 protein expression was significantly correlated with voluntary consumption (slope = 1.96±0.57, F=11.67), but not BAC, in lateral amygdala samples from male drinkers. (E) No correlation between SNAP25 protein levels and ethanol intake or BAC. (F) Level of Syntaxin 1 were significantly correlated with BAC (slope = −2.83±1.27, F = 4.934), but not voluntary intake, in male lateral amygdala.
Multiple Regression Analysis
Given that individual protein levels had some ability to predict total consumption or BEC within each of the sexes, we addressed whether combinations of proteins might together better predict drinking outcomes using a forward step-wise regression analysis. In male drinkers, a model containing only VAMP2 significantly contributed to the voluntary consumption data. This was also the only protein showing a significant relationship with drinking in the pairwise correlations as well. For the BEC data however, a model combining Munc13-1, Munc18-1, and VAMP1 was significantly correlated with the average mg/dL measures (adjusted R2=0.597, F8.396, p<0.01). Both Munc13-1 and VAMP1, each with negative correlation coefficients, together comprised 84% of the predicative power while Munc18-1, with a positive correlation coefficient, contributed the remaining 16%. This suggested a combination of lower Munc13-1 and VAMP1 protein levels, with modest contributions by higher Munc18-1 protein levels, predicted male BEC measured 7h into the drinking period.. Notably, this Munc13-1/Munc18-1/VAMP1 linear model predicted BEC to a greater degree (higher R2) than the individual proteins identified in the pair-wise analysis (Munc13-1 and vGluT2, see Fig. 2). A similar analysis in female drinkers showed that no combination of proteins reliably predicted BEC. For total voluntary consumption however, only Rab3c was significantly correlated, consistent with the pairwise correlation analysis for individual proteins (Fig. 2A1).
Discussion
A summary of our findings across all the proteins examined in our study is shown in Figure 5. For synaptic vesicle docking/priming proteins (Fig. 5, left), long-term ethanol self-administration reduced levels of both Rab3a and Munc13-2 in the lateral amygdala regardless of sex. Rab3 proteins are small, vesicle-associated GTPases that, in the GTP-bound form, coordinate juxtaposition of plasma membrane calcium channels with synaptic vesicles at release sites via coordination of a macromolecular complex consisting of Mun13 and channel-binding proteins, RIM, and RIM-BP (Sudhof, 2004, Sudhof, 2013). Recent evidence suggests that Munc13-2 levels modulate both intrinsic release probability at glutamate synapses (Gioia et al., 2016) as well as the acute sensitivity of vesicle recycling pathways to ethanol (Gioia et al., 2017). These distinct roles suggest that decreases in Munc13-2 in ethanol drinkers may facilitate presynaptic activity while decreases in Rab3A levels may represent a compensatory down-regulation of priming/release mechanisms following long-term ethanol drinking. Notably, females express higher levels of Munc13-1 than males. Given that greater levels of Munc13-1 relative to other Munc13 proteins increases presynaptic neurotransmitter release at central synapses (Gioia et al., 2016, Chen et al., 2013), increased Munc13-1 levels in females suggests they may have greater presynaptic function. We also note however that there was a significant cohort-effect in ethanol drinking males (cohorts 5 and 7a) for the Rab3A data. Given cohort effects were not consistently present across all proteins with significant main effects of ethanol drinking, it is likely any cohort-related differences in Rab3A levels represent the differential distribution of categorical drinking phenotypes across these cohorts (Table 1).
For proteins directly involved with vesicle release (Fig. 5, right), an ethanol drinking history reduces protein levels of both Complexin 1 and 2, again regardless of sex. These proteins facilitate calcium-dependent neurotransmitter release by stabilizing interactions between core SNARE complex proteins to promote priming (Xue et al., 2008, Trimbuch and Rosenmund, 2016); but, this stabilization also appears to reduce spontaneous release (Huntwork and Littleton, 2007). Given that chronic ethanol exposure in rodents facilitates presynaptic function at lateral/basolateral glutamate synapses; and this facilitation is characterized by both increased calcium-dependent release (Christian et al., 2013) and enhanced spontaneous release (Lack et al., 2007), complexin down-regulation may be a compensatory effect of long-term ethanol exposure (Lack et al., 2007). Presynaptic modulation at GABAergic synapses in the lateral/basolateral amygdala is less well documented (but see below) although chronic ethanol can down-regulate GABA release at some synapses (Diaz et al., 2011) which typically express relatively a high frequency of spontaneous neurotransmission. It is also notable that we found that females express higher levels of SNAP25 compared to males. SNAP25 is a central component of the core SNARE complex and is intimately involved with presynaptic release for a wide variety of neurotransmitters including acetylcholine, glutamate, and GABA. SNAP-25 increases the clustering of syntaxin at the plasma membrane, facilitates vesicle recruitment to release sites, and regulates the size of the pool of synaptic vesicles (Nagy et al., 2002, Nagy et al., 2004, Sorensen et al., 2003, Toft-Bertelsen et al., 2016). Like our findings with Munc13-1, these observations suggest females may express greater capacity for release within presynaptic compartments.
Our correlational analysis examined only proteins without any treatment effects across the entire population of ethanol drinkers. These results suggest that protein with significant relationships represent potentially heritable phenotypes contributing to specific aspects of drinking phenotype. In females for example, the only significant correlation was between Rab3c and total voluntary consumption with lower expression levels predicting higher intakes. Like Rab3a, Rab3c is abundant in the central nervous system (Cheng et al., 2002) where it has been localized to both glutamatergic and GABAergic terminals (Bragina et al., 2011), oligodendrocytes (Madison et al., 1996), and secretory endocrine tissue (Iezzi et al., 1999). However, it is noteworthy that our selective analysis between controls and VHD females also found significant effects of ethanol drinking Rab3c among this category of individuals that was not significant when including all phenotypes. This suggests that these particular correlations may have been substantially influenced by the decrease in Rab3c protein levels among VHD females. Given the relatively poor representation of HD and LD phenotypes in female drinkers for these specific cohorts, correlations within this sex should be interpreted with caution given potential confounds related to the ‘over-representation’ of VHD female samples. In males, VAMP 2 was the only protein correlated with total voluntary consumption with higher levels predicting higher consumption. VAMP2 levels are also correlated with dementia duration in Lewy-Body disorder, Parkinson’s disease, and Alzheimer’s disease (Vallortigara et al., 2016); and reduced levels of VAMP2 are found in transgenic mice that overexpress amyloid precursor protein (Pham et al., 2010). Notably, exposure of hippocampal neurons to amyloid-β peptide reduces presynaptic glutamate release in vitro via modulation of VAMP2 levels (Hu et al., 2015). These data suggest that positive relationships between VAMP2 levels and ethanol intake in males may reflect an ethanol-dependent up-regulation of vesicle release in this sex. A more complex picture emerged for relationships between SNARE protein levels and blood alcohol concentrations. In the current study, blood samples were taken every 5–7 days throughout the 12 month drinking period but only at 7 hours after session onset. Thus, BEC itself may be a phenotypic proxy for drinking patterns. Firstly, only male BECs were significantly correlated with any protein’s expression level. Individually, Syntaxin 1 and vGluT2 were each negatively correlated with average BEC again suggesting that individual differences in neurotransmitter release from presynaptic compartments dictate individual drinking patterns. Interestingly, a multiple regression model containing expression levels of Munc13-1, Munc18-1, and VAMP1 proteins was correlated with BEC to a greater degree than individual proteins. This suggests that the overall function of the SNARE complex is a better predictor of individual drinking.
There have been no studies to date on the synaptic neurophysiology of lateral amygdala in monkeys so integration of our expression analysis with the neurophysiological effects of ethanol drinking must be made in the context of other brain regions. For the glutamate system, chronic self-administration generally increases presynaptic function. This includes reports of increased frequency of miniature/spontaneous glutamatergic EPSCs recorded from both putamen medium spiney neurons and insular cortical neurons (Cuzon Carlson et al., 2011, Alexander et al., 2012) in male macaques and in the insular cortex where ethanol drinking increased apparent glutamate release probability as indicated by synaptic responses to pairs of juxtaposed electrical stimuli (Alexander et al., 2012). Similar findings have also been reported in the recordings from orbitofrontal cortical neurons of adult male cynomolgus macaques with an ethanol drinking history (Nimitvilai et al., 2017). Notably, the increased frequency of miniature EPSCs in the putamen was also associated with an increase in spine density suggesting increased numbers of synapses in ethanol drinkers as a potential mechanism (Cuzon Carlson et al., 2011). These data are consistent with a growing literature in rodents showing increased presynaptic glutamatergic function following chronic ethanol exposure (Christian et al., 2013, Lack et al., 2007, Morales et al., 2018, Sabria et al., 2003). Similar to these glutamate observations, total lifetime intake by ethanol-drinking female macaques was positively correlated with dopamine release in the nucleus accumbens (Siciliano et al., 2016). The effects of ethanol drinking on GABAergic presynaptic function are less frequent and include both increased (Pleil et al., 2016) and decreased (Cuzon Carlson et al., 2011) release in within distinct brain regions. Given the paucity of data related to the neurophysiological effects of ethanol drinking in macaques, the rich literature in rodents highlighting region-specific alterations in synaptic function points towards a similar diversity in non-human primates.
In summary, we found that an extensive history of ethanol self-administration in rhesus macaques altered expression of a number of proteins participating in presynaptic neurotransmitter release and vesicle priming/recycling. These alterations were protein-dependent and occurred independently in both male and female drinkers. Correlational analysis revealed significant proportional associations between proteins levels and aspects of ethanol intake BEC during the open-access self-administration for several proteins or groups of proteins. Given these proteins were not significantly altered by the ethanol self-administration across the drinking population, it is possible that individual differences in expression levels for these SNARE complex components represent potentially heritable phenotypes related to neurotransmitter homeostasis or release; and these phenotypes contribute to both total consumption and potentially to patterns of intake.
Acknowledgments
This work was supported by the National Institutes of Health [grant numbers R01 AA014445 (BAM), R01 AA023999 (BAM), P50 AA026117 (BAM, JBD), R24 AA019431 (KAG, JBD), and U01 AA013510 (KAG)]
References
- Alexander GM, Graef JD, Hammarback JA, Nordskog BK, Burnett EJ, Daunais JB, Bennett AJ, Friedman DP, Suomi SJ, Godwin DW. Disruptions in serotonergic regulation of cortical glutamate release in primate insular cortex in response to chronic ethanol and nursery rearing. Neuroscience. 2012;207:167–81. doi: 10.1016/j.neuroscience.2012.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Augustin I, Betz A, Herrmann C, Jo T, Brose N. Differential expression of two novel Munc13 proteins in rat brain. Biochem J. 1999;337:363–71. [PMC free article] [PubMed] [Google Scholar]
- Baker EJ, Farro J, Gonzales S, Helms C, Grant KA. Chronic alcohol self-administration in monkeys shows long-term quantity/frequency categorical stability. Alcohol Clin Exp Res. 2014;38:2835–43. doi: 10.1111/acer.12547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker EJ, Walter NA, Salo A, Rivas Perea P, Moore S, Gonzales S, Grant KA. Identifying future drinkers: Behavioral analysis of monkeys initiating drinking to intoxication is predictive of future drinking classification. Alcohol Clin Exp Res. 2017;41:626–636. doi: 10.1111/acer.13327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bragina L, Fattorini G, Giovedi S, Melone M, Bosco F, Benfenati F, Conti F. Analysis of Synaptotagmin, SV2, and Rab3 Expression in Cortical Glutamatergic and GABAergic Axon Terminals. Front Cell Neurosci. 2011;5:32. doi: 10.3389/fncel.2011.00032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breustedt J, Gundlfinger A, Varoqueaux F, Reim K, Brose N, Schmitz D. Munc13-2 differentially affects hippocampal synaptic transmission and plasticity. Cereb Cortex. 2010;20:1109–20. doi: 10.1093/cercor/bhp170. [DOI] [PubMed] [Google Scholar]
- Cervera-Juanes R, Wilhelm LJ, Park B, Grant KA, Ferguson B. Alcohol-dose-dependent DNA methylation and expression in the nucleus accumbens identifies coordinated regulation of synaptic genes. Transl Psychiatry. 2017a;7:e994. doi: 10.1038/tp.2016.266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cervera-Juanes R, Wilhelm LJ, Park B, Grant KA, Ferguson B. Genome-wide analysis of the nucleus accumbens identifies DNA methylation signals differentiating low/binge from heavy alcohol drinking. Alcohol. 2017b;60:103–113. doi: 10.1016/j.alcohol.2016.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z, Cooper B, Kalla S, Varoqueaux F, Young SM., Jr The Munc13 proteins differentially regulate readily releasable pool dynamics and calcium-dependent recovery at a central synapse. J Neurosci. 2013;33:8336–51. doi: 10.1523/JNEUROSCI.5128-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng H, Ma Y, Ni X, Jiang M, Luo Y, Ying K, Xie Y, Ma Y. Cloning, mapping, and characterization of the human Rab3C gene. Biochem Genet. 2002;40:263–72. doi: 10.1023/a:1019834901190. [DOI] [PubMed] [Google Scholar]
- Christian DT, Alexander NJ, Diaz MR, Mccool BA. Thalamic glutamatergic afferents into the rat basolateral amygdala exhibit increased presynaptic glutamate function following withdrawal from chronic intermittent ethanol. Neuropharmacology. 2013;65:134–142. doi: 10.1016/j.neuropharm.2012.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuzon Carlson VC, Seabold GK, Helms CM, Garg N, Odagiri M, Rau AR, Daunais J, Alvarez VA, Lovinger DM, Grant KA. Synaptic and morphological neuroadaptations in the putamen associated with long-term, relapsing alcohol drinking in primates. Neuropsychopharmacology. 2011;36:2513–28. doi: 10.1038/npp.2011.140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das J, Xu S, Pany S, Guillory A, Shah V, Roman GW. The pre-synaptic Munc13-1 binds alcohol and modulates alcohol self-administration in Drosophila. J Neurochem. 2013;126:715–26. doi: 10.1111/jnc.12315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daunais JB, Davenport AT, Helms CM, Gonzales SW, Hemby SE, Friedman DP, Farro JP, Baker EJ, Grant KA. Monkey alcohol tissue research resource: banking tissues for alcohol research. Alcohol Clin Exp Res. 2014;38:1973–81. doi: 10.1111/acer.12467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daunais JB, Kraft RA, Davenport AT, Burnett EJ, Maxey VM, Szeliga KT, Rau AR, Flory GS, Hemby SE, Kroenke CD, Grant KA, Friedman DP. MRI-guided dissection of the nonhuman primate brain: a case study. Methods. 2010;50:199–204. doi: 10.1016/j.ymeth.2009.03.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diaz MR, Christian DT, Anderson NJ, Mccool BA. Chronic ethanol and withdrawal differentially modulate basolateral amygdala paracapsular and local GABAergic synapses. J Pharmacol Exp Ther. 2011;337:162–170. doi: 10.1124/jpet.110.177121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gioia DA, Alexander NJ, Mccool BA. Differential expression of Munc13-2 produces unique synaptic phenotypes in the basolateral amygdala of C57BL/6J and DBA/2J mice. J Neurosci. 2016;36:10964–10977. doi: 10.1523/JNEUROSCI.1785-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gioia DA, Alexander NJ, Mccool BA. Ethanol mediated inhibition of synaptic vesicle recycling at amygdala glutamate synapses is dependent upon Munc13-2. Front Neurosci. 2017;11:424. doi: 10.3389/fnins.2017.00424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gioia DA, Mccool B. Strain-dependent effects of acute alcohol on synaptic vesicle recycling and post-tetanic potentiation in medial glutamate inputs to the mouse basolateral amygdala. Alcohol Clin Exp Res. 2017;41:735–746. doi: 10.1111/acer.13343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grant KA, Leng X, Green HL, Szeliga KT, Rogers LS, Gonzales SW. Drinking typography established by scheduled induction predicts chronic heavy drinking in a monkey model of ethanol self-administration. Alcohol Clin Exp Res. 2008;32:1824–38. doi: 10.1111/j.1530-0277.2008.00765.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helms CM, Rau A, Shaw J, Stull C, Gonzales SW, Grant KA. The effects of age at the onset of drinking to intoxication and chronic ethanol self-administration in male rhesus macaques. Psychopharmacology (Berl) 2014;231:1853–61. doi: 10.1007/s00213-013-3417-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu S, Wang H, Chen K, Cheng P, Gao S, Liu J, Li X, Sun X. MicroRNA-34c downregulation ameliorates amyloid-beta-induced synaptic failure and memory deficits by targeting VAMP2. J Alzheimers Dis. 2015;48:673–86. doi: 10.3233/JAD-150432. [DOI] [PubMed] [Google Scholar]
- Huntwork S, Littleton JT. A complexin fusion clamp regulates spontaneous neurotransmitter release and synaptic growth. Nat Neurosci. 2007;10:1235–7. doi: 10.1038/nn1980. [DOI] [PubMed] [Google Scholar]
- Iezzi M, Escher G, Meda P, Charollais A, Baldini G, Darchen F, Wollheim CB, Regazzi R. Subcellular distribution and function of Rab3A, B, C, and D isoforms in insulin-secreting cells. Mol Endocrinol. 1999;13:202–12. doi: 10.1210/mend.13.2.0228. [DOI] [PubMed] [Google Scholar]
- James DJ, Martin TF. CAPS and Munc13: CATCHRs that SNARE vesicles. Front Endocrinol. 2013;4:187. doi: 10.3389/fendo.2013.00187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janak PH, Tye KM. From circuits to behaviour in the amygdala. Nature. 2015;517:284–92. doi: 10.1038/nature14188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lack AK, Diaz MR, Chappell A, Dubois DW, Mccool BA. Chronic ethanol and withdrawal differentially modulate pre- and postsynaptic function at glutamatergic synapses in rat basolateral amygdala. J Neurophysiol. 2007;98:3185–96. doi: 10.1152/jn.00189.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu J, Machius M, Dulubova I, Dai H, Sudhof TC, Tomchick DR, Rizo J. Structural basis for a Munc13-1 homodimer to Munc13-1/RIM heterodimer switch. PLoS Biol. 2006;4:e192. doi: 10.1371/journal.pbio.0040192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madison DL, Kruger WH, Kim T, Pfeiffer SE. Differential expression of rab3 isoforms in oligodendrocytes and astrocytes. J Neurosci Res. 1996;45:258–68. doi: 10.1002/(SICI)1097-4547(19960801)45:3<258::AID-JNR7>3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- Morales M, Mcginnis MM, Robinson SL, Chappell AM, Mccool BA. Chronic intermittent ethanol exposure modulation of glutamatergic neurotransmission in rat lateral/basolateral amygdala is duration-, input-, and sex-dependent. Neuroscience. 2018 doi: 10.1016/j.neuroscience.2017.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagy G, Matti U, Nehring RB, Binz T, Rettig J, Neher E, Sorensen JB. Protein kinase C-dependent phosphorylation of synaptosome-associated protein of 25 kDa at Ser187 potentiates vesicle recruitment. J Neurosci. 2002;22:9278–86. doi: 10.1523/JNEUROSCI.22-21-09278.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagy G, Reim K, Matti U, Brose N, Binz T, Rettig J, Neher E, Sorensen JB. Regulation of releasable vesicle pool sizes by protein kinase A-dependent phosphorylation of SNAP-25. Neuron. 2004;41:417–29. doi: 10.1016/s0896-6273(04)00038-8. [DOI] [PubMed] [Google Scholar]
- Nimitvilai S, Uys JD, Woodward JJ, Randall PK, Ball LE, Williams RW, Jones BC, Lu L, Grant KA, Mulholland PJ. Orbitofrontal neuroadaptations and cross-species synaptic biomarkers in heavy-drinking macaques. J Neurosci. 2017;37:3646–3660. doi: 10.1523/JNEUROSCI.0133-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palomares-Castillo E, Hernandez-Perez OR, Perez-Carrera D, Crespo-Ramirez M, Fuxe K, Perez De La Mora M. The intercalated paracapsular islands as a module for integration of signals regulating anxiety in the amygdala. Brain Res. 2012;1476:211–34. doi: 10.1016/j.brainres.2012.03.047. [DOI] [PubMed] [Google Scholar]
- Pham E, Crews L, Ubhi K, Hansen L, Adame A, Cartier A, Salmon D, Galasko D, Michael S, Savas JN, Yates JR, Glabe C, Masliah E. Progressive accumulation of amyloid-beta oligomers in Alzheimer’s disease and in amyloid precursor protein transgenic mice is accompanied by selective alterations in synaptic scaffold proteins. FEBS J. 2010;277:3051–67. doi: 10.1111/j.1742-4658.2010.07719.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pleil KE, Helms CM, Sobus JR, Daunais JB, Grant KA, Kash TL. Effects of chronic alcohol consumption on neuronal function in the non-human primate BNST. Addict Biol. 2016;21:1151–1167. doi: 10.1111/adb.12289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porcu P, Rogers LS, Morrow AL, Grant KA. Plasma pregnenolone levels in cynomolgus monkeys following pharmacological challenges of the hypothalamic-pituitary-adrenal axis. Pharmacol Biochem Behav. 2006;84:618–27. doi: 10.1016/j.pbb.2006.05.004. [DOI] [PubMed] [Google Scholar]
- Rosenmund C, Sigler A, Augustin I, Reim K, Brose N, Rhee JS. Differential control of vesicle priming and short-term plasticity by Munc13 isoforms. Neuron. 2002;33:411–24. doi: 10.1016/s0896-6273(02)00568-8. [DOI] [PubMed] [Google Scholar]
- Sabria J, Torres D, Pasto M, Peralba JM, Allali-Hassani A, Pares X. Release of neurotransmitters from rat brain nerve terminals after chronic ethanol ingestion: differential effects in cortex and hippocampus. Addict Biol. 2003;8:287–94. doi: 10.1080/13556210310001602194. [DOI] [PubMed] [Google Scholar]
- Sassa T, Ogawa H, Kimoto M, Hosono R. The synaptic protein UNC-18 is phosphorylated by protein kinase C. Neurochem Int. 1996;29:543–52. doi: 10.1016/0197-0186(96)00009-5. [DOI] [PubMed] [Google Scholar]
- Siciliano CA, Calipari ES, Cuzon Carlson VC, Helms CM, Lovinger DM, Grant KA, Jones SR. Voluntary ethanol intake predicts kappa-opioid receptor supersensitivity and regionally distinct dopaminergic adaptations in macaques. J Neurosci. 2015;35:5959–68. doi: 10.1523/JNEUROSCI.4820-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siciliano CA, Calipari ES, Yorgason JT, Lovinger DM, Mateo Y, Jimenez VA, Helms CM, Grant KA, Jones SR. Increased presynaptic regulation of dopamine neurotransmission in the nucleus accumbens core following chronic ethanol self-administration in female macaques. Psychopharmacology (Berl) 2016;233:1435–43. doi: 10.1007/s00213-016-4239-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silberman Y, Shi L, Brunso-Bechtold JK, Weiner JL. Distinct mechanisms of ethanol potentiation of local and paracapsular GABAergic synapses in the rat basolateral amygdala. J Pharmacol Exp Ther. 2008;324:251–60. doi: 10.1124/jpet.107.128728. [DOI] [PubMed] [Google Scholar]
- Sorensen JB, Nagy G, Varoqueaux F, Nehring RB, Brose N, Wilson MC, Neher E. Differential control of the releasable vesicle pools by SNAP-25 splice variants and SNAP-23. Cell. 2003;114:75–86. doi: 10.1016/s0092-8674(03)00477-x. [DOI] [PubMed] [Google Scholar]
- Sudhof TC. The synaptic vesicle cycle. Annu Rev Neurosci. 2004;27:509–47. doi: 10.1146/annurev.neuro.26.041002.131412. [DOI] [PubMed] [Google Scholar]
- Sudhof TC. Neurotransmitter release: the last millisecond in the life of a synaptic vesicle. Neuron. 2013;80:675–90. doi: 10.1016/j.neuron.2013.10.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toft-Bertelsen TL, Ziomkiewicz I, Houy S, Pinheiro PS, Sorensen JB. Regulation of Ca2+ channels by SNAP-25 via recruitment of syntaxin-1 from plasma membrane clusters. Mol Biol Cell. 2016;27:3329–3341. doi: 10.1091/mbc.E16-03-0184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trimbuch T, Rosenmund C. Should I stop or should I go? The role of complexin in neurotransmitter release. Nat Rev Neurosci. 2016;17:118–25. doi: 10.1038/nrn.2015.16. [DOI] [PubMed] [Google Scholar]
- Vallortigara J, Whitfield D, Quelch W, Alghamdi A, Howlett D, Hortobagyi T, Johnson M, Attems J, O’brien JT, Thomas A, Ballard CG, Aarsland D, Francis PT. Decreased Levels of VAMP2 and Monomeric Alpha-Synuclein Correlate with Duration of Dementia. J Alzheimers Dis. 2016;50:101–10. doi: 10.3233/JAD-150707. [DOI] [PubMed] [Google Scholar]
- Wierda KD, Toonen RF, De Wit H, Brussaard AB, Verhage M. Interdependence of PKC-dependent and PKC-independent pathways for presynaptic plasticity. Neuron. 2007;54:275–90. doi: 10.1016/j.neuron.2007.04.001. [DOI] [PubMed] [Google Scholar]
- Xue M, Stradomska A, Chen H, Brose N, Zhang W, Rosenmund C, Reim K. Complexins facilitate neurotransmitter release at excitatory and inhibitory synapses in mammalian central nervous system. Proc Natl Acad Sci U S A. 2008;105:7875–80. doi: 10.1073/pnas.0803012105. [DOI] [PMC free article] [PubMed] [Google Scholar]