Hepatic bile formation plays an essential role in lipid digestion and absorption, cholesterol homeostasis, and excretion of lipid soluble metabolites and xenobiotics. Bile is a complex, lipid-rich micellar solution composed primarily of water, inorganic solutes, and organic solutes such as amphipathic conjugated bile acids (BAs), the membrane phospholipid phosphatidylcholine (PC), cholesterol, bile pigments, and endogenous metabolites (1). The major organic solutes, BAs, phospholipids, and cholesterol are termed “biliary lipids” and their secretion into bile is mediated by three distinct canalicular membrane ABC transporters, ABCB11(BSEP), ABCB4 (MDR3) (Abcb4/Mdr2 in rodents), and ABCG5/ABCG8, respectively (1). ABCB4 functions to flop PC from the canalicular membrane into bile, where it forms stable mixed micelles with BAs and cholesterol. In doing so, PC acts to protect the biliary epithelium from the toxic detergent properties of BAs and to solubilize cholesterol, thereby preventing gallstone formation. Loss of Abcb4 in mice effectively blocks PC secretion into bile and leads to bile duct injury and progressive cholestatic liver disease (2, 3). In humans, ABCB4 mutations are responsible for progressive familial intrahepatic cholestasis type 3 (PFIC3), a severe form of liver disease that develops early in childhood. In addition to PFIC3, homozygous or heterozygous genetic variants in ABCB4 are associated with a spectrum of cholestatic diseases, including low phospholipid associated cholestasis and intrahepatic cholestasis of pregnancy (4, 5). With only limited therapeutic options currently available for ABCB4-associated disease, new strategies to rescue the activity of defective ABCB4 variants or to increase ABCB4 expression may have clinical benefit in these patients. To date, ABCB4 expression has been shown to be induced in human and animal models by BAs and fibrates, acting via the nuclear receptors farnesoid X receptor (FXR) and PPARα, respectively. The study by Gautherot and colleagues (6) in this issue of the Journal of Lipid Research adds the nuclear receptor thyroid hormone receptor-β (THRβ), and its isoform THRβ1, to the list of potential targets that increase ABCB4 levels.
Thyroid hormones act via the nuclear receptors, THRα (NR1A1; chromosome 17) and THRβ (NR1A2; chromosome 3), and exert profound effects on development, growth and metabolism. Although expression of both receptors can be detected in a wide variety of tissues, THRα shows the highest levels of expression in heart, brain, lung, and bone, whereas THRβ is the major THR in the liver. It has been known for many years that overproduction of thyroid hormone (hyperthyroidism) is associated with potential beneficial effects on lipid metabolism. However, the plethora of adverse effects of excess thyroid hormone on heart, bone, and muscle stymied further development of thyroid hormone as therapy. More recently, the identification of distinct THR isoforms, expressed in a tissue-specific fashion, the finding that THRα and THRβ differentially regulated hepatic lipid metabolism, and the development of THR isoform-selective agonists has focused renewed interest in therapeutically targeting the major liver isoform, THRβ1 (7, 8). With regard to biliary lipid secretion, studies in rat (9) and later in mouse (10) models demonstrated that thyroid hormone stimulates hepatic BA synthesis and biliary secretion of lipids, particularly that of cholesterol and phospholipid. Whereas the stimulation of biliary cholesterol secretion was shown to be dependent upon induction of ABCG5/ABCG8 expression, details of the mechanisms by which thyroid hormone stimulates biliary phospholipid secretion has not to date been systematically examined.
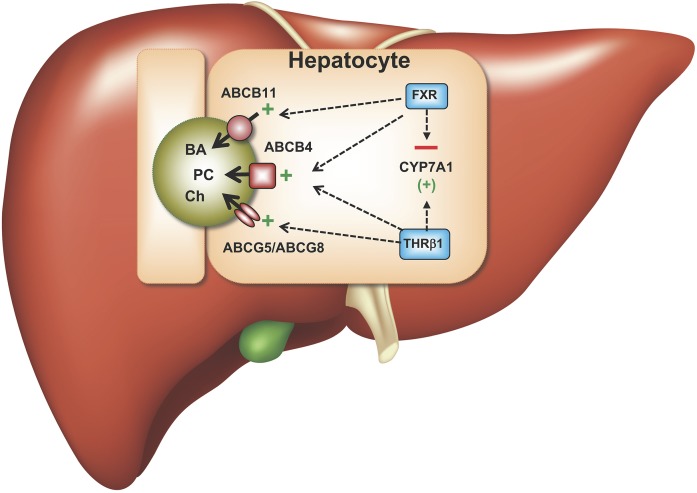
In the study by Gautherot et al. (6), thyroid hormone T3 was compared with FXR and PPARα agonists for its ability to induce ABCB4 mRNA and protein expression in human hepatocyte cell lines. Notably, the addition of T3 induced ABCB4 expression approximately 2-fold, an effect similar to that observed with the synthetic FXR agonist, GW4064. Further studies in cell culture revealed dose, time-course, and inhibition effects consistent with direct transcriptional activation of ABCB4 by thyroid hormone. In silico analysis of the promoter and 5′-flanking sequence identified several thyroid hormone response elements that were conserved in the human and mouse ABCB4 locus and shown to bind THRβ1 using in vitro DNA gel shift assays. In agreement with the tissue expression pattern for the thyroid hormone receptor isoforms, addition of THRβ1-selective agonists such as GC-1 (sobetirome), CGS-2345, KB-2115 (eprotirome), and particularly KB-141 induced the expression of ABCB4 mRNA and protein in Huh-7 cells. In vivo, administration of KB-141 in the diet for 7 days to WT C57Bl/6 male mice significantly increased bile flow (~1.8-fold) and the biliary output of PC (5.4-fold), cholesterol (3.3-fold), and BAs (2.9-fold). Additional tissue culture-based studies of ABCB4 expression in human hepatocyte cell lines demonstrated that T3 and FXR agonists have independent stimulatory effects. Interestingly, whereas T3 stimulated CYP7A1 expression in cultured hepatocytes, addition of GW4064 or coaddition of T3 and GW4046 significantly reduced expression of CYP7A1, suggesting that the FXR-mediated repression is dominant (summarized in Fig. 1).
Fig. 1.
Model for effects of THRβ1 and FXR agonism on BA synthesis, expression of the major biliary lipid transporters, and hepatic lipid secretion. BA, bile acid; CYP7A1, cholesterol 7-α hydroxylase; Ch, cholesterol; FXR, farnesoid X receptor; PC, phosphatidylcholine; THRβ, thyroid hormone receptor-β.
The referenced study extends earlier findings reporting the effects of thyroid hormone on biliary lipid secretion and is notable for providing evidence of direct transcriptional regulation of ABCB4 expression by THRβ1. In addition, as proposed by the authors, by boosting PC secretion while decreasing BA synthesis, concomitant THRβ1 and FXR stimulation may have potential therapeutic benefit in ABCB4-related liver disease and, potentially, other forms of cholestasis. A limitation of this work is that the current observations were primarily confined to tissue culture cell-based models and the in vivo effects on bile secretion and other hepatic functions remain to be determined. For example, with regard to plasma lipids, THRβ1 simulation in humans decreases LDL cholesterol (for review, see Ref. 7), whereas FXR stimulation elicits an unfavorable increase (11). As such, additional studies in appropriate models will be needed to understand the spectrum of in vivo effects observed with THRβ1 agonism or dual stimulation along with FXR. Although recent studies of thyromimetic drugs have demonstrated potential beneficial effects for metabolic diseases involving the liver (8), promising therapeutic strategies targeting nuclear receptors have shown relevant limitations when further evaluated clinically (12). Moreover, while THRβ1 agonists do not, or minimally, affect the hypothalamic-pituitary axis, long-term treatment may determine a state of hyperthyroidism in THRβ-dependent tissues and a state of normo- or hypothyroidism in THRα-dependent tissues; a peculiar condition for which the resulting effects on general homeostasis are unknown.
Footnotes
This work was supported by National Institutes of Health research Grant DK047987 (P.A.D). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the National Institutes of Health.
References
- 1.Boyer J. L. 2013. Bile formation and secretion. Compr. Physiol. 3: 1035–1078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Smit J. J., Schinkel A. H., Oude Elferink R. P., Groen A. K., Wagenaar E., van Deemter L., Mol C. A., Ottenhoff R., van der Lugt N. M., van Roon M. A., et al. 1993. Homozygous disruption of the murine mdr2 P-glycoprotein gene leads to a complete absence of phospholipid from bile and to liver disease. Cell. 75: 451–462. [DOI] [PubMed] [Google Scholar]
- 3.Fickert P., Fuchsbichler A., Wagner M., Zollner G., Kaser A., Tilg H., Krause R., Lammert F., Langner C., Zatloukal K., et al. 2004. Regurgitation of bile acids from leaky bile ducts causes sclerosing cholangitis in Mdr2 (Abcb4) knockout mice. Gastroenterology. 127: 261–274. [DOI] [PubMed] [Google Scholar]
- 4.Dixon P. H., Wadsworth C. A., Chambers J., Donnelly J., Cooley S., Buckley R., Mannino R., Jarvis S., Syngelaki A., Geenes V., et al. 2014. A comprehensive analysis of common genetic variation around six candidate loci for intrahepatic cholestasis of pregnancy. Am. J. Gastroenterol. 109: 76–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dröge C., Bonus M., Baumann U., Klindt C., Lainka E., Kathemann S., Brinkert F., Grabhorn E., Pfister E. D., Wenning D., et al. 2017. Sequencing of FIC1, BSEP and MDR3 in a large cohort of patients with cholestasis revealed a high number of different genetic variants. J. Hepatol. 67: 1253–1264. [DOI] [PubMed] [Google Scholar]
- 6.Gautherot J., Claudel T., Cuperus F., Fuchs C. D., Falguieres T., and Trauner M.. 2018. Thyroid hormone receptor beta1 stimulates ABCB4 to increase biliary phosphatidylcholine excretion in mice. J. Lipid Res. 59: 1610–1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jakobsson T., Vedin L. L., and Parini P.. 2017. Potential role of thyroid receptor beta agonists in the treatment of hyperlipidemia. Drugs. 77: 1613–1621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sinha R. A., Singh B. K., and Yen P. M.. 2018. Direct effects of thyroid hormones on hepatic lipid metabolism. Nat. Rev. Endocrinol. 14: 259–269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gebhard R. L., and Prigge W. F.. 1992. Thyroid hormone differentially augments biliary sterol secretion in the rat. II. The chronic bile fistula model. J. Lipid Res. 33: 1467–1473. [PubMed] [Google Scholar]
- 10.Bonde Y., Plosch T., Kuipers F., Angelin B., and Rudling M.. 2012. Stimulation of murine biliary cholesterol secretion by thyroid hormone is dependent on a functional ABCG5/G8 complex. Hepatology. 56: 1828–1837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pencek R., Marmon T., Roth J. D., Liberman A., Hooshmand-Rad R., and Young M. A.. 2016. Effects of obeticholic acid on lipoprotein metabolism in healthy volunteers. Diabetes Obes. Metab. 18: 936–940. [DOI] [PubMed] [Google Scholar]
- 12.Kirchgessner T. G., Sleph P., Ostrowski J., Lupisella J., Ryan C. S., Liu X., Fernando G., Grimm D., Shipkova P., Zhang R., et al. 2016. Beneficial and adverse effects of an LXR agonist on human lipid and lipoprotein metabolism and circulating neutrophils. Cell Metab. 24: 223–233. [DOI] [PubMed] [Google Scholar]