Abstract
The present study aimed to verify the expression and investigate the role of microRNA (miR)-148a in intervertebral disc degeneration (IDD) and explore the associated underlying mechanisms. Reverse transcription-quantitative polymerase chain reaction (RT-qPCR) was performed to investigate levels of miR-148a in the peripheral blood mononuclear cells (PBMCs) of patients with IDD. To investigate the role of miR-148a in IDD, a stable miR-148a-overexpression/underexpression human nucleus pulposus (NP) cell line was generated by transfection with miR-148a mimic/inhibitor. Then, NP cells were treated with LPS (10 µM) to induce inflammation. The mRNA expression level of miR-148a in NP cells was determined by RT-qPCR and the expression levels of p38 and p-p38 were measured using western blotting. The mRNA expression and supernatant level of pro-inflammatory cytokines, tumor necrosis factor (TNF-α), interleukin (IL)-1β and IL-6, was evaluated by RT-qPCR and ELISA, respectively. The results indicated that miR-148a was significantly downregulated in the PBMCs of IDD patients compared with healthy controls. In vitro upregulation of miR-148a in LPS-stimulated NP cells, by transfection with miR-148a mimic, resulted in inhibition of p-p38 expression; however, inhibition of miR-148a led to overexpression of p-p38. Meanwhile, the production of pro-inflammatory cytokines (TNF-α, IL-1β and IL-6) was significantly reduced in miR-148a-overexpressing LPS-stimulated NP cells and significantly increased in miR-148a-underexpressing NP cells. In conclusion, miR-148a inhibits pro-inflammatory cytokines released by intervertebral disc cells via regulation of the p38/mitogen-activated protein kinase pathway.
Keywords: microRNA-148a, intervertebral disc degeneration, pro-inflammatory cytokines, mitogen-activated protein kinase
Introduction
Intervertebral disc degeneration (IDD) is a major risk factor for lower back pain that is a worldwide problem. It badly affects human life and causes a serious socioeconomic burden (1–4). An imbalance between anabolic and catabolic genes expressed by chondrocytes that belong to the nucleus pulposus (NP) result in IDD. The etiology of IDD is very complicated. Numerous cellular events, including cell apoptosis, inflammation, autophagy and increased matrix catabolism, have been confirmed to be involved in the progress of IDD (5,6). It has previously been reported that NP cells, which share the common features of chondrocytes, play critical roles in maintaining integrity of intervertebral discs (IVDs) (7,8). One of the major features of IDD is the loss of proteoglycan (PG) content from IVDs. Lipopolysaccharide (LPS), a strong promoter of inflammation, can reduce PG content and cause the occurrence of IDD (9,10). Furthermore, pro-inflammatory factors such as interleukin (IL)-1β, IL-6 and tumor necrosis factor (TNF)-α also serve critical functions in IDD. However, the underlying molecular and cellular mechanisms of IDD remain unclear.
MicroRNAs (miRs) are small (20–22 nucleotides in length), non-coding, single-stranded RNAs that serve important functions in post-transcriptionally regulating gene expression (11,12). Previous studies have demonstrated that miRs are critical in cancer progress, as well as in inflammatory, neurodegenerative and most degenerative disorders (7,8). Previous reports have demonstrated that miRs serve important functions in degenerative disc diseases, such as IDD (13–16). It has also been indicated that miR-148a is downregulated in IDD compared with spinal cord injury (17), but the underlying mechanisms are still unknown.
Thus, the aim of the present study was to verify the expression and investigate the role of miR-148a in IDD and explore the underlying mechanisms.
Materials and methods
Materials
LPS was purchased from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany). ELISA kits for TNF-α (cat no. E-CL-M0047c), IL-1β (cat no. E-EL-M0037c) and IL-6 (cat no. E-EL-M0044c) were obtained from Elabscience Biotechnology Co., Ltd. (Wuhan, China). Antibodies against p38 (cat no. 9212)/p-p38 (cat no. 4631) and GAPDH (cat no. 97166) and secondary antibodies (anti-rabbit IgG, horseradish peroxidase (HRP)-linked antibody; cat no. 7074; anti-mouse IgG HRP-linked antibody; cat no. 7076) were supplied by Cell Signaling Technology, Inc. (Danvers, MA, USA). miR-148a mimic (cat no. miR10000243-1-5)/inhibitor (cat no. miR20000243-1-5) were purchased from Guangzhou Ribobio Co., Ltd. (Guangzhou, China); all other chemicals and reagents were purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China).
Patients
The study was approved by the Human Ethics Committees Review Board at the Affiliated Hospital of Xuzhou Medical College (Xuzhou, China) and written informed consent was obtained from each patient prior to enrollment. A total of 30 patients with IDD and 30 healthy volunteers were enrolled from the Department of Spinal Surgery at the Affiliated Hospital of Xuzhou Medical College (Xuzhou, China) from January 2014 to January 2015. Peripheral blood mononuclear cells (PBMCs) were obtained from the peripheral blood of patients and healthy volunteers as described previously (18). Peripheral blood was centrifuged at 650 × g for 25 min at room temperature. Middle phases containing Human PBMCs were further centrifuged (650 × g for 10 min at room temperature) by density using Lymphoprep (Stemcell Technologies, Inc., Beijing, China) (19).
Cell culture
Human NP cells were purchased from ScienCell Research Laboratories, Inc. (San Diego, CA, USA; cat. no. 4800) and cultured in Dulbecco's Modified Eagle's medium (DMEM; Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) containing 10% fetal bovine serum (Gibco), 1% L-glutamine (Gibco), 100 mg/ml streptomycin and 100 U/ml penicillin and then incubated in a 5% CO2 incubator at 37°C. The culture medium was changed every 2 days and cells were passaged until they reached 90% confluence.
Cell treatment
Human NP cells (5×104 cells per well) were added to a 6-well plate and transiently transfected with miR-148a mimic (50 nM)/inhibitor (100 nM) using Lipofectamine with Plus reagent (Thermo Fisher Scientific, Inc.) and control transfections were performed lacking miR-148a. The nucleotide sequences used were as follows: miR-148a mimic forward, 5′-UCAGUGCAUGACAGAACUUGG-3′ and reverse, 5′-AAGTTCUGUCAUGCACUGAUU-3′; NC forward, 5′-UUCUCCGAACGUGUCACGUTT-3′ and reverse, 5′-ACGUGACACGUUCGGAGAATT-3′; and miR-148a inhibitor, 5′-ACAAAGUUCUGUAGUGCACUGA-3′. At 24 h following the transfection, 5×104 cells were stimulated with LPS (10 µM) in serum-free medium (DMEM) for 24 h at 37°C under 5% CO2. The suspensions were then centrifuged at 1,000 × g for 10 min at 4°C and collected 24 h after the initiation of each treatment. Cells were then harvested for subsequent experimentation. The groups were divided into the following: A control group (NP cells without any treatment); an NC group (NP cells transfected with NC oligonucleotides and stimulated with LPS); a mimic group (NP cells transfected with miR148a mimic and then stimulated with LPS) and an inhibitor group (NP cells transfected with miR148a inhibitor and then stimulated with LPS).
RNA isolation and reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted from human PBMCs and human NP cells using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. Each sample (1 µg RNA) was then reverse-transcribed to cDNA using a reverse-transcription (RT) reagent kit (Takara Biotechnology Co., Ltd., Dalian, China). A 7500 real-time PCR instrument (Applied Biosystems; Thermo Fisher Scientific, Inc.) was used to quantify miR-148a, TNF-α, IL-1β and IL-6 mRNA levels. Amplification conditions were as follows: 16°C for 30 min, 42°C for 30 min, 85°C for 5 min and hold at 4°C. qPCR was performed using the QuantiTect SYBR-Green PCR kit (Takara Biotechnology Co., Ltd.). All reactions were performed in triplicate with the following conditions: 95°C for 10 min, followed by 37 cycles at 95°C for 15 sec and 72°C for 30 sec. All quantitative miRNA data were normalized to U6 expression and quantitative mRNA data were normalized to GAPDH. The comparative Cq method was used for the relative amounts of each transcript (20). The primers for qPCR were as presented in Table I.
Table I.
Primer sequences for polymerase chain reaction analysis.
| Gene | Forward primer (5′-3′) | Reverse primer (5′-3′) |
|---|---|---|
| TNF-α | CCTGTCTCTTCCTACCCAACC | GCAGGAGTGTCCGTGTCTTC |
| IL-1β | CTGTGACTCGTGGGATGATG | AGGGATTTTGTCGTTGCTTG |
| IL-6 | GTGCTCCTGGTATTGCTGGT | GGCTCCTCGTTTTCCTTCTT |
| miR-148a | ATGCTCAGTGCACTACAGAA | GTGCAGGGTCCGAGGT |
| U6 | CTCGCTTCGGCAGCACA | AACGCTTCACGAATTTGCGT |
| GAPDH | CTTTGGTATCGTGGAAGGACTC | GTAGAGGCAGGGATGATGTTCT |
TNF, tumor necrosis factor; IL, interleukin; miR, microRNA.
Western blot analysis
In brief, total cellular protein from NP cells was extracted using RIPA buffer (Cell Signaling Technology, Inc.) and separated by SDS-PAGE analysis. Sample protein concentrations were determined using the BCA protein assay (Thermo Fisher Scientific, Inc.). Each lane was loaded with 25 µg protein and resolved using a 10% SDS-PAGE gel and transferred to a polyvinylidene fluoride membrane. Membranes were then blocked using Tris-buffered saline with 0.1% Tween-20 (TBST) containing 5% non-fat milk for 1 h at room temperature. Membranes were subsequently incubated overnight at 4°C with primary antibodies against p-p38 and p38 (1:1,000) or GAPDH (1:2,000). Membranes were incubated with the correspondent secondary antibody (1:5,000) at room temperature for 2 h. Protein bands were observed with enhanced chemiluminescence and imaged.
ELISA assay
In order to determine levels of TNF-α, IL-1β and IL-6 in the NP cellular supernatant, ELISA was performed with ELISA kits, following the manufacturer's protocol.
Statistical analysis
Data are presented as the mean ± SD. All statistical analyses were performed using SPSS 17.0 statistical software (SPSS, Inc., Chicago, IL, USA). A t-test or one-way analysis of variance followed by Tukey's test was used to evaluate differences between groups. P<0.05 was considered to indicate a statistically significant difference.
Results
Downregulation of miR-148a in IDD
To investigate the potential role of miR-148a in IDD, the relative expression of miR-148a in PBMCs separated from IDD patients and control subjects was compared. miR-148a expression level was also detected in NP cells with or without LPS stimulation. As shown in Fig. 1A, the expression level of miR-148a was significantly decreased in PBMCs of patients with IDD compared with control subjects. Furthermore, miR-148a expression level in LPS-stimulated NP cells was significantly lower compared with the untreated NP cells (Fig. 1B). These data suggested that miR-148a is downregulated in IDD.
Figure 1.
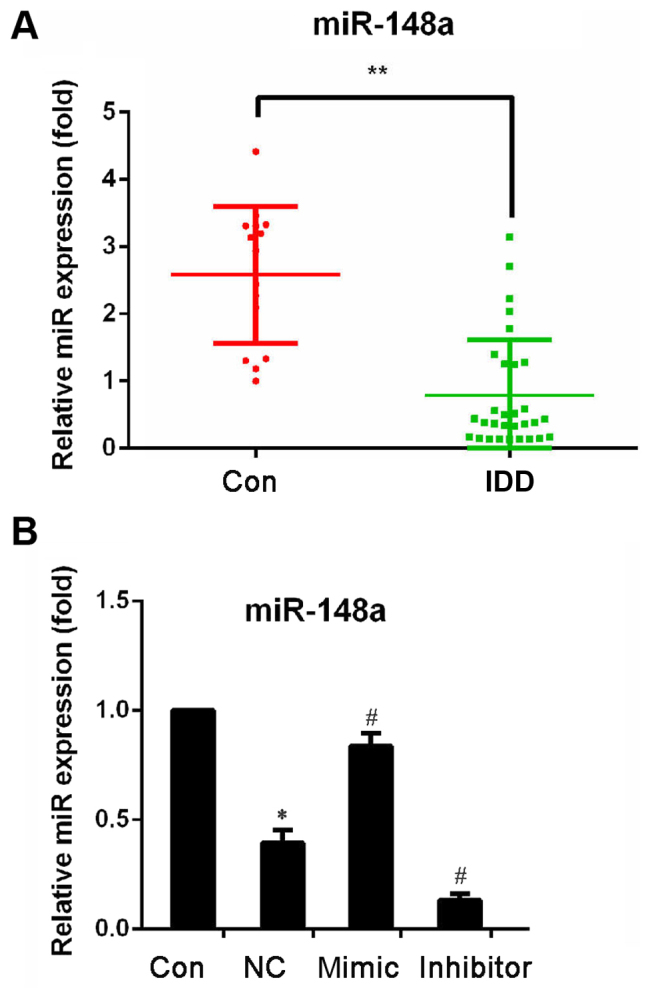
miR-148a expression detection. miR-148a expression level was measured by reverse transcription-quantitative polymerase chain reaction and data were normalized to U6 expression. (A) Expression level of miR-148a in peripheral blood mononuclear cells of patients with IDD and healthy controls. **P<0.01 vs. Con. (B) Expression level of miR-148a in LPS-stimulated NP cells. Experiments were performed in triplicate. *P<0.05 vs. Con; #P<0.05 vs. NC. Con, control (normal NP cells); NC, negative control (NP cells transfected with NC oligonucleotides and stimulated with LPS); mimic, NP cells transfected with miR-148a mimic and stimulated with LPS; inhibitor, NP cells transfected with miR-148a inhibitor and stimulated with LPS; NP, nucleus pulposus; LPS, lipopolysaccharide; miR, microRNA; IDD, intervertebral disc degeneration.
miR-148a affects pro-inflammatory cytokines levels in LPS-stimulated NP cells
In order to evaluate the effects of miR-148a in IDD, miR-148a expression was induced in LPS-stimulated NP cells using miR-148a mimic and inhibited using miR-148a inhibitor, while negative control (NC) oligonucleotides were used as the control. As shown in Fig. 1B, compared with the NP cells transfected with NC oligonucleotides, miR-148a expression was significantly upregulated by miR-148a mimic transfection and significantly inhibited by miR-148a inhibitor transfection. Then, the mRNA expression level and quantity of pro-inflammatory cytokines TNF-α, IL-1β and IL-6 was measured by RT-qPCR and ELISA, respectively. The results indicated that, compared with the NC group, the mRNA expression level of TNF-α, IL-1β and IL-6 significantly decreased in NP cells transfected with miR-148a mimic (Fig. 2A) and significantly increased in NP cells transfected with miR-148a inhibitor (Fig. 2B). In addition, the ELISA results indicated that, compared with the NC group, cellular supernatant TNF-α, IL-1β and IL-6 levels significantly decreased in NP cells transfected with miR-148a mimic and significantly increased in NP cells transfected with miR-148a inhibitor (Fig. 3). These results indicated that miR-148a inhibits pro-inflammatory cytokine levels in LPS-stimulated NP cells. This suggests that miR-148a may inhibit pro-inflammatory factors released by IVD cells.
Figure 2.
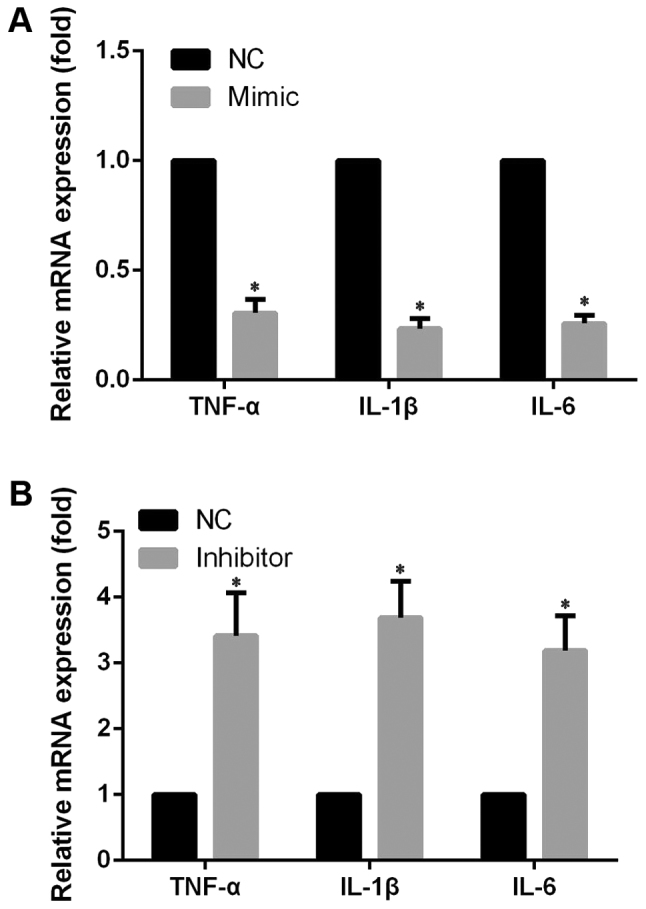
mRNA expression level of pro-inflammatory cytokines. The mRNA expression level of TNF-α, IL-1β and IL-6 was measured by reverse transcription-quantitative polymerase chain reaction and data were normalized to GAPDH expression. (A) mRNA expression level of TNF-α, IL-1β and IL-6 in NP cells transfected with miR-148a mimic and stimulated with LPS. (B) mRNA expression level of TNF-α, IL-1β and IL-6 in NP cells transfected with miR-148a inhibitor and stimulated with LPS. Experiments were performed in triplicate. *P<0.05 vs. NC. NC, negative control (NP cells transfected with NC oligonucleotides and stimulated with LPS); TNF, tumor necrosis factor; IL, interleukin; NP, nucleus pulposus; miR, microRNA; LPS, lipopolysaccharide.
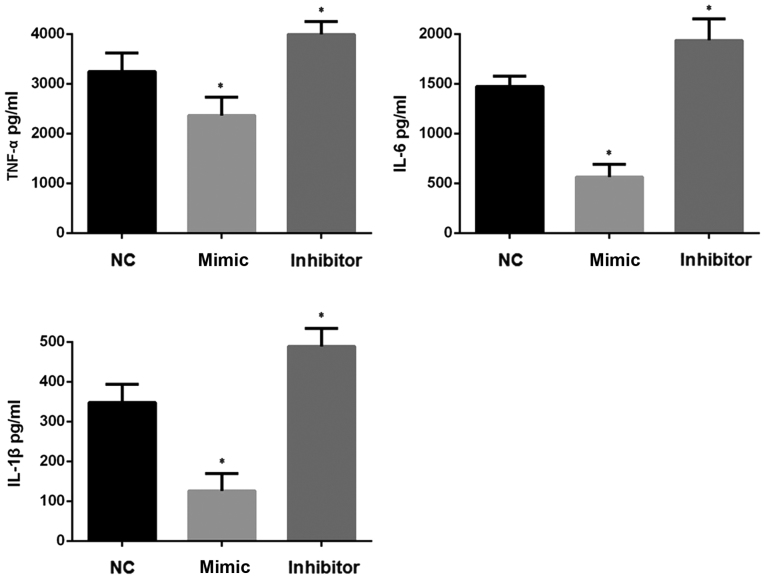
Figure 3.
Levels of pro-inflammatory cytokines in NP cellular supernatant. ELISA was performed to detect the expression level of TNF-α, IL-1β and IL-6 in the cellular supernatant of NP cells stimulated with LPS and transfected with miR-148a mimic or inhibitor. Experiments were performed in triplicate. *P<0.05 vs. NC. NC, negative control (NP cells transfected with NC oligonucleotides and stimulated with LPS); TNF, tumor necrosis factor; IL, interleukin; NP, nucleus pulposus; miR, microRNA; LPS, lipopolysaccharide.
miR-148a affects activation of the p38/mitogen-activated protein kinase (MAPK) pathway in LPS-stimulated NP cells
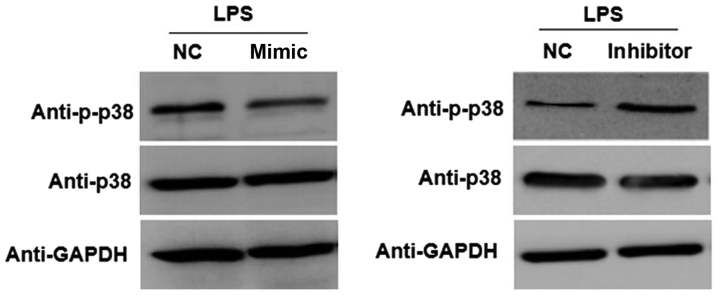
To investigate the underlying mechanism by which miR-148a affects the inflammatory response, the involvement of the p38/MAPK pathway was evaluated. As shown in Fig. 4, compared with the NC group, overexpression of miR-148a markedly reduced the protein expression level of p-p38 in LPS-stimulated NP cells transfected with miR-148a mimic. Furthermore, transfection with miR-148a inhibitor resulted in a marked increase in the expression of p-p38 in LPS-stimulated NP cells compared with the NC group. These results indicated that miR-148a overexpression inhibits activation of the p38/MAPK pathway in LPS-stimulated NP cells.
Figure 4.
Expression level of p38 and p-p38 in NP cells stimulated with LPS. NP cells were transfected with miR-148a mimic, miR-148a inhibitor or NC. At 24 h after the transfection, the cells were stimulated with LPS for 24 h. Then, western blot analysis was performed to detect protein expression levels. NC, negative control (NP cells transfected with NC oligonucleotides and stimulated with LPS); NP, nucleus pulposus; miR, microRNA; LPS, lipopolysaccharide.
Discussion
Numerous studies have indicated that miRs serve key functions in the development of IDD (13,14,16,21). However the expression and role of miR-148a in IDD remains unknown. In the present study, the role of miR-148a in IDD was investigated and the pathological links between miR-148a, IDD and inflammatory pathways were investigated.
In order to evaluate the role of miR-148a in IDD, the expression level of miR-148a was measured in PBMCs isolated from IDD patients and control subjects. miR-148a expression level was also detected in NP cells with or without LPS stimulation. The findings suggested that miR-148a was significantly downregulated in IDD patients compared with the healthy controls, as well as in the LPS-stimulated NP cells compared with non-stimulated cells. Similar to these results, previous studies on miRNA expression in degenerative discs have reported that numerous miRNAs are abnormally expressed in IDD (22–25).
To investigate the underlying mechanism of the role of miR-148a in IDD, a stable miR-148a-overexpression/underexpression human NP cell line was established by transfection with miR-148a mimic/inhibitor and an IDD cell model was established by LPS stimulation. A previous study confirmed that inflammation drives the degeneration of IVD (26). The increased expression level of pro-inflammatory cytokines secreted by IVD cells is the major characteristic of IDD and these cytokines promote extracellular matrix degradation, chemokine production and changes in IVD cell phenotype (27). Thus, in the present study, the mRNA expression level of pro-inflammatory cytokines (TNF-α, IL-1β and IL-6) was evaluated in NP cells. The results revealed that miR-148a mimic reduced the mRNA expression level of pro-inflammatory cytokines in LPS-stimulated NP cells and miR-148a inhibitor presented the opposite effect. Furthermore, the cellular supernatant levels of TNF-α, IL-1β and IL-6 were detected by ELISA and the results indicated the same trends as mRNA expression levels.
Finally, in order to investigate whether the p38/MAPK pathway is involved in the regulation of inflammatory responses by miR-148a in IDD, activation of the p38/MAPK pathway was evaluated in the present study. It was identified that upregulation of miR-148a in LPS-stimulated NP cells decreased the phosphorylation of p38, while miR-148a inhibitor increased the phosphorylation of p38. These results indicated that activation of the p38/MAPK pathway is associated with the function of miR-148a in regulating inflammatory response in LPS-stimulated NP cells.
In conclusion, the present findings suggest that miR-148a serves a function in IDD progress and miR-148a overexpression suppresses pro-inflammatory factors released by IVD cells though regulating the p38/MAPK pathway. Thus, miR-148a may have potential therapeutic applications in the treatment of IDD.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Millecamps M, Tajerian M, Naso L, Sage EH, Stone LS. Lumbar intervertebral disc degeneration associated with axial and radiating low back pain in ageing SPARC-null mice. Pain. 2012;153:1167–1179. doi: 10.1016/j.pain.2012.01.027. [DOI] [PubMed] [Google Scholar]
- 2.Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, Shibuya K, Salomon JA, Abdalla S, Aboyans V, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: A systematic analysis for the global burden of disease study 2010. Lancet. 2012;380:2163–2196. doi: 10.1016/S0140-6736(12)61729-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gore M, Sadosky A, Stacey BR, Tai KS, Leslie D. The burden of chronic low back pain: Clinical comorbidities, treatment patterns and health care costs in usual care settings. Spine (Phila Pa 1976) 2012;37:E668–E677. doi: 10.1097/BRS.0b013e318241e5de. [DOI] [PubMed] [Google Scholar]
- 4.Takatalo J, Karppinen J, Taimela S, Niinimäki J, Laitinen J, Sequeiros RB, Samartzis D, Korpelainen R, Näyhä S, Remes J, Tervonen O. Association of abdominal obesity with lumbar disc degeneration-a magnetic resonance imaging study. PLoS One. 2013;8:e56244. doi: 10.1371/journal.pone.0056244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kepler CK, Ponnappan RK, Tannoury CA, Risbud MV, Anderson DG. The molecular basis of intervertebral disc degeneration. Spine J. 2013;13:318–330. doi: 10.1016/j.spinee.2012.12.003. [DOI] [PubMed] [Google Scholar]
- 6.Risbud MV, Shapiro IM. Role of cytokines in intervertebral disc degeneration: Pain and disc content. Nat Rev Rheumatol. 2014;10:44–56. doi: 10.1038/nrrheum.2013.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Teague EM, Print CG, Hull ML. The role of microRNAs in endometriosis and associated reproductive conditions. Hum Reprod Update. 2010;16:142–165. doi: 10.1093/humupd/dmp034. [DOI] [PubMed] [Google Scholar]
- 8.Croce CM. Causes and consequences of microRNA dysregulation in cancer. Nat Rev Genet. 2009;10:704–714. doi: 10.1038/nrg2634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ellman MB, Kim JS, An HS, Chen D, KC R, An J, Dittakavi T, van Wijnen AJ, Cs-Szabo G, Li X, et al. Toll-like receptor adaptor signaling molecule MyD88 on intervertebral disk homeostasis: In vitro, ex vivo studies. Gene. 2012;505:283–290. doi: 10.1016/j.gene.2012.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iwata M, Ochi H, Asou Y, Haro H, Aikawa T, Harada Y, Nezu Y, Yogo T, Tagawa M, Hara Y. Variations in gene and protein expression in canine chondrodystrophic nucleus pulposus cells following long-term three-dimensional culture. PLoS One. 2013;8:e63120. doi: 10.1371/journal.pone.0063120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell. 1993;75:843–854. doi: 10.1016/0092-8674(93)90529-Y. [DOI] [PubMed] [Google Scholar]
- 12.Bartel DP. MicroRNAs: Genomics, biogenesis, mechanism and function. Cell. 2004;116:281–297. doi: 10.1016/S0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 13.Yu X, Li Z, Shen J, Wu WK, Liang J, Weng X, Qiu G. MicroRNA-10b promotes nucleus pulposus cell proliferation through RhoC-Akt pathway by targeting HOXD10 in intervetebral disc degeneration. PLoS One. 2013;8:e83080. doi: 10.1371/journal.pone.0083080. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 14.Ohrt-Nissen S, Døssing KB, Rossing M, Lajer C, Vikeså J, Nielsen FC, Friis-Hansen L, Dahl B. Characterization of miRNA expression in human degenerative lumbar disks. Connect Tissue Res. 2013;54:197–203. doi: 10.3109/03008207.2013.781594. [DOI] [PubMed] [Google Scholar]
- 15.Liu G, Cao P, Chen H, Yuan W, Wang J, Tang X. MiR-27a regulates apoptosis in nucleus pulposus cells by targeting PI3K. PLoS One. 2013;8:e75251. doi: 10.1371/journal.pone.0075251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Liu H, Huang X, Liu X, Xiao S, Zhang Y, Xiang T, Shen X, Wang G, Sheng B. miR-21 promotes human nucleus pulposus cell proliferation through PTEN/AKT signaling. Int J Mol Sci. 2014;15:4007–4018. doi: 10.3390/ijms15034007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhao B, Yu Q, Li H, Guo X, He X. Characterization of microRNA expression profiles in patients with intervertebral disc degeneration. Int J Mol Med. 2014;33:43–50. doi: 10.3892/ijmm.2013.1543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Diaz-Morales N, Iannantuoni F, Escribano-Lopez I, Bañuls C, Rovira-Llopis S, Sola E, Rocha M, Hernandez-Mijares A, Victor VM. Does metformin modulate endoplasmic reticulum stress and autophagy in type 2 diabetic peripheral blood mononuclear cells? Antioxid Redox Signal. 2017 doi: 10.1089/ars.2017.7409. (Epub ahead of print) [DOI] [PubMed] [Google Scholar]
- 19.Nakanishi R, Kitao H, Kiniwa M, Morodomi Y, Iimori M, Kurashige J, Sugiyama M, Nakashima Y, Saeki H, Oki E, Maehara Y. Monitoring trifluridine incorporation in the peripheral blood mononuclear cells of colorectal cancer patients under trifluridine/tipiracil medication. Sci Rep. 2017;7:16969. doi: 10.1038/s41598-017-17282-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 21.Wang HQ, Yu XD, Liu ZH, Cheng X, Samartzis D, Jia LT, Wu SX, Huang J, Chen J, Luo ZJ. Deregulated miR-155 promotes Fas-mediated apoptosis in human intervertebral disc degeneration by targeting FADD and caspase-3. J Pathol. 2011;225:232–242. doi: 10.1002/path.2931. [DOI] [PubMed] [Google Scholar]
- 22.Gruber HE, Hoelscher GL, Ingram JA, Hanley EN., Jr Genome-wide analysis of pain-, nerve- and neurotrophin-related gene expression in the degenerating human annulus. Mol Pain. 2012;8:63. doi: 10.1186/1744-8069-8-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xu YQ, Zhang ZH, Zheng YF, Feng SQ. Dysregulated miR-133a mediates loss of typeII collagen by directly targeting matrix metalloproteinase 9 (MMP9) in human intervertebral disc degeneration. Spine (Phila Pa 1976) 2016;41:E714–E724. doi: 10.1097/BRS.0000000000001375. [DOI] [PubMed] [Google Scholar]
- 24.Wang B, Wang D, Yan T, Yuan H. MiR-138-5p promotes TNF-α-induced apoptosis in human intervertebral disc degeneration by targeting SIRT1 through PTEN/PI3K/Akt signaling. Exp Cell Res. 2016;345:199–205. doi: 10.1016/j.yexcr.2016.05.011. [DOI] [PubMed] [Google Scholar]
- 25.Ma JF, Zang LN, Xi YM, Yang WJ, Zou D. MiR-125a Rs12976445 Polymorphism is associated with the apoptosis status of nucleus pulposus cells and the risk of intervertebral dis degeneration. Cell Physiol Biochem. 2016;38:295–305. doi: 10.1159/000438630. [DOI] [PubMed] [Google Scholar]
- 26.Teixeira GQ, Pereira CL, Ferreira JR, Maia AF, Gomez-Lazaro M, Barbosa MA, Neidlinger-Wilke C, Goncalves RM. Immunomodulation of human mesenchymal stem/stromal cells in intervertebral disc degeneration: insights from a proinflammatory/degenerative ex vivo model. Spine (Phila Pa 1976) 2017 doi: 10.1097/BRS.0000000000002494. (Epub ahead of print) [DOI] [PubMed] [Google Scholar]
- 27.Maidhof R, Jacobsen T, Papatheodorou A, Chahine NO. Inflammation induces irreversible biophysical changes in isolated nucleus pulposus cells. PLoS One. 2014;9:e99621. doi: 10.1371/journal.pone.0099621. [DOI] [PMC free article] [PubMed] [Google Scholar]