Abstract
The contribution of nuclear medicine to management of melanoma patients is increasing. In intermediate-thickness N0 melanomas, lymphoscintigraphy provides a roadmap for sentinel node biopsy. With the introduction of single-photon emission computed tomography images with integrated computed tomography (SPECT/CT), 3D anatomic environments for accurate surgical planning are now possible. Sentinel node identification in intricate anatomical areas (pelvic cavity, head/neck) has been improved using hybrid radioactive/fluorescent tracers, preoperative lymphoscintigraphy and SPECT/CT together with modern intraoperative portable imaging technologies for surgical navigation (free-hand SPECT, portable gamma cameras). Furthermore, PET/CT today provides 3D roadmaps to resect 18F-fluorodeoxyglucose-avid melanoma lesions. Simultaneously, in advanced-stage melanoma and recurrences, 18F-fluorodeoxyglucose-PET/CT is useful in clinical staging and treatment decision as well as in the evaluation of therapy response. In this article, we review new insights and recent nuclear medicine advances in the management of melanoma patients.
Keywords: : 18F-FDG-PET/CT, intraoperative technologies, radioguided surgery, sentinel node biopsy, SPECT/CT
Practice points.
Lymphoscintigraphy for sentinel node biopsy (SNB) offers a precise roadmap for the surgeon.
Multicenter Selective Lymphadenectomy Trial-I results enhanced the paper of early nodal assessment and treatment of melanoma based on SNB: the tumor status of the sentinel node (SN) is the single most important prognostic factor and, even though no differences in overall survival were demonstrated, SNB with immediate regional lymphadenectomy results in an improvement in melanoma-specific survival among patients with intermediate thickness melanoma and tumor-positive SN.
However, Multicenter Selective Lymphadenectomy Trial-II results showed that immediate complete lymphadenectomy after SNB did not increase melanoma-specific survival compared with nodal observation with ultrasonography and was associated with higher rate of complications.
Innovative technological developments, especially in the field of hybrid fluorescent-radioactive tracers, enable better depiction of the tumor's lymphatic drainage mapping and accurate intraoperative identification of SN in real-time.
The use of 18F-fluorodeoxyglucose (FDG)-PET is not warranted for staging patients with positive SNB but could be justified in those cases with clinically palpable lymph nodes before excision, due to its potential modification on treatment decisions.
FDG-PET is a precise tool for whole-body staging assessment in patients with advanced-stage melanoma and those with recurrence suspicion. PET/CT may constitute a 3D roadmap for surgical resection in patients with oligometastases.
The use of new inmunotherapeutic-targeted therapeutic agents in metastatic melanoma is changing FDG-PET/CT criteria for therapy response assessment, introducing concepts as ‘heterogeneous response’ and ‘immune flare response’ or ‘pseudoprogression’.
Since the incorporation of lymphoscintigraphy for lymphatic mapping in the sentinel node biopsy (SNB) procedure in the early 1990s of the past century, an important body of evidence regarding the role of nuclear medicine in the management of early-staged melanoma patients has been accumulated. Together with new insights based on valuable clinical data, recent studies have validated important technical advances like single-photon emission computed tomography images with integrated computed tomography (SPECT/CT) and intraoperative portable imaging devices to search SN in complex anatomical sites. In the same period, PET/CT using 18F-fluorodeoxyglucose (18F-FDG) has definitively been incorporated for restaging in the case of recurrences and its role is being explored in staging of advanced-stage melanoma. With respect to a previous review [1], we will discuss the most relevant advances and insights concerning nuclear medicine in melanoma in this paper.
A high degree of accuracy for staging purposes is important in case of an advanced melanoma lesion as this will give proper information about prognosis facilitating the election of the most adequate treatment [2]. Nuclear medicine procedures for SNB and PET/CT have remarkably improved the accuracy of melanoma staging. Concerning locoregional staging, the latest results of the first Multicenter Selective Lymphadenectomy Trial (MSLT-I) confirmed the tumor status of the SN as the single most important prognostic factor and simultaneously proved that, even though no differences in overall survival (OS) were demonstrated, SNB coupled with immediate regional lymph node dissection improves melanoma-specific survival among patients with intermediate thickness melanoma (the American Joint Committee on Cancer stage I or II, between 1.2 and 3.5 mm Breslow thickness) and tumor-positive SN [3–6]. Because of its prognostic significance, SNB has been included in the staging classification of the American Joint Committee on Cancer and the Union International Contre le Cancer since 2001 [7,8].
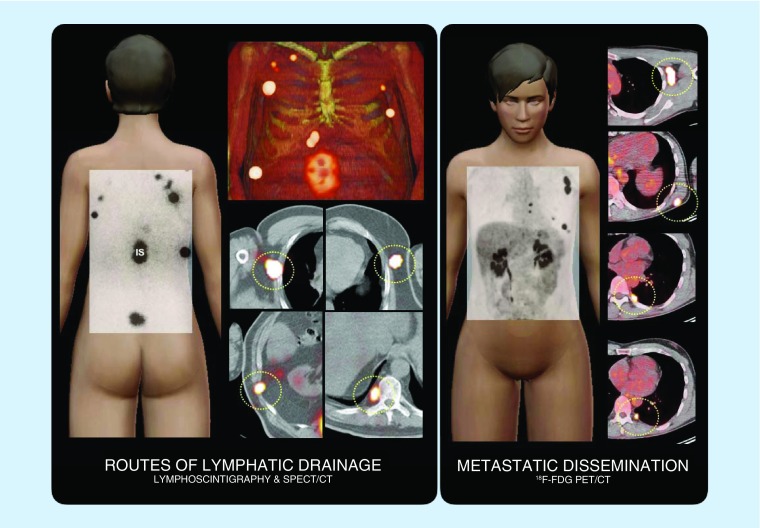
Melanoma can spread to lymphatic basins (lymph nodes) and visceral organs, but also has a propensity to disseminate through the lymph vessels as satellite or in-transit metastasis. In this context, many of the lymphatic drainage routes assessed by lymphoscintigraphy and SPECT/CT correspond with sites of lymph node metastases when PET/CT is used to establish dissemination of the disease (Figure 1).
Figure 1. . Routes of lymphatic drainage and metastatic dissemination.
On the left, schematic illustration of a case showing lymphatic drainage from the injections (IS) around the site of the primary melanoma on the back to intercostal, axillary and paravertebral (circles) sentinel lymph nodes as assessed by lymphoscintigraphy and SPECT/CT. On the right, another case where PET/CT shows FDG-avid lymph node metastases in left axilla, left scapular area and left paravertebral (circles).
FDG: 18F-fluorodeoxyglucose; IS: Injection site; SPECT/CT: Single-photon emission computed tomography images with integrated computed tomography.
Hybrid fluorescent-radioactive tracers in combination with intraoperative imaging technologies such as free-hand SPECT and portable gamma cameras have emerged as a valid tool for SN localization in cases of complex anatomical areas, enabling to perform a high-quality precise preoperative roadmap for the surgeon, especially when complemented with SPECT/CT.
Meticulous evaluation of the diverse spreading patterns conducts to an improvement of the decision-making process and PET/CT with 18F-fluorodeoxyglucose (FDG-PET) is a paramount tool in the appraisal of advanced and recurrent disease. FDG-PET is effective in detecting occult metastatic disease and its utility for the clinical staging and treatment decision-making has been reported as trustworthy. In particular for patients with oligometastastic disease [9], PET/CT can potentially be used as a 3D roadmap for image-guided resection of the FDG-avid lesions. Furthermore, the revolution in the treatment of advanced-stage melanoma caused by the implementation of new targeted therapeutic agents is changing the so-far used criteria in FDG-PET for therapy response assessment, introducing concepts as ‘heterogeneous response’, ‘immune flare’ or ‘pseudoprogression’.
Lymphoscintigraphy improves the accuracy of sentinel lymph node staging
Concept & relevance of SNB
The regional lymph nodes are the most common site of metastatic dissemination from melanoma [10]. 20% of the patients with a melanoma of Breslow thickness over 1 mm presented with metastasis in their regional lymphatic basins. Randomized studies revealed no clear advantages when a prophylactic regional lymph node dissection was carried out [11]. The SNB procedure relies on the assumption of the stepwise spreading of melanoma through the lymphatic system and the SN is defined as the node on a direct lymphatic drainage pathway from the primary tumor [12].
First developed by Morton et al. [13] for melanoma patients, SNB is currently an important component of routine staging in various neoplastic diseases [14–18]. The classical SNB concept implied that in the case of no SN metastatic involvement the rest of the lymph nodes in the regional basin could be considered as disease-free, sparing these patients for a regional lymphadenectomy, which was reserved only for patients with SN-containing metastases. However, recent results from MSLT-II and DeCOG-SLT trials showed no melanoma-specific survival benefits between immediate lymphadenectomy and active ultrasonography surveillance in patients with metastatic SN [19–21].
Preoperative lymphoscintigraphy is able to depict all lymph node stations that might be at risk for metastases, providing a roadmap to guide the intervention. In the last decades, this approach has been extended to other radioguided applications and was the basis to elaborate the Guided intraoperative Scintigraphic Tumor Targeting (GOSTT) concept that is not just limited to lymphatic studies following interstitial radiotracer administration (SNB) but also includes a wide gamma of applications related to systemic and intralesional administration of radiopharmaceuticals [22]. GOSTT concept was introduced with the objective of agglutinating the whole of possibilities of basic and advanced nuclear medicine procedures that can aid in the intraoperative identification of targeted tissues with the aid of portable portable gamma cameras or handheld SPECT (Figure 2) [23]. Hence, GOSTT techniques improve the accuracy and reproducibility of lymphatic mapping [24–27] and offer a suitable solution for several objectives: to ascertain the lymphatic basins at risk of metastatic involvement, to depict the number of SNs, to help distinguish SNs from second-tier nodes, to detect SNs in uncommon and nonpredictable sites [24,25,28–30], to mark the precise localization of the SN on the skin projection, and to guide intraoperative identification and excision (Figure 3).
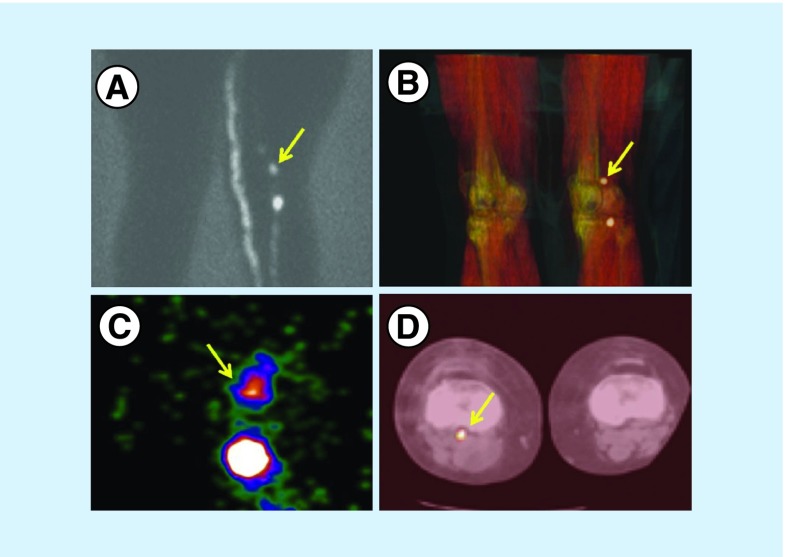
Figure 2. . Patient with a melanoma on the dorsum of the right foot.
(A) The planar posterior view shows two lymphatic channels. One goes up to the groin and the other to the popliteal fossa where two nodes are depicted, while there is a hint of a third node downstream. (B) The 3D volume-rendered reconstructed image provides the anatomical habitat of the two nodes and confirms the presence of the faint third node. (C) The intraoperative image with a portable gamma camera shows the first two popliteal nodes. SPECT/CT is a very useful tool for planning the surgical approach. (D) In this case, a very deep popliteal node is demonstrated in the axial slice, requiring the surgeon to make a long incision.
SPECT/CT: Single-photon emission computed tomography images with integrated computed tomography.
Reproduced with permission from [1] © Future Medicine Ltd. (2014).
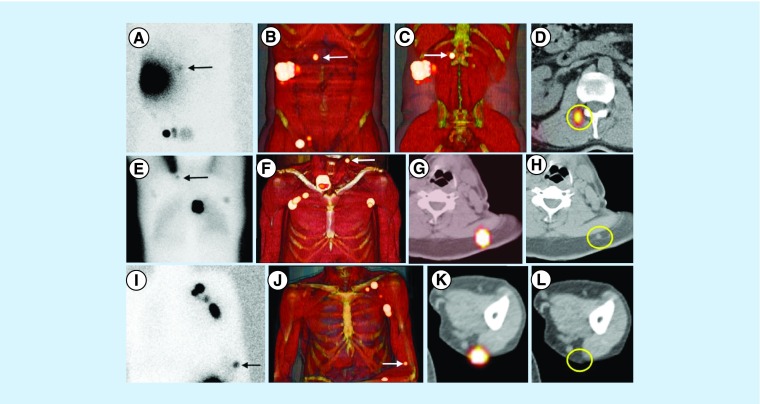
Figure 3. . Sentinel nodes in unexpected areas of lymphatic drainage.
(A) Anterior planar image of a patient with a melanoma in the right flank showing not only drainage to the right groin but also to the medial area of the trunk (arrow). Note that the sentinel node is clearly defined on (B & C) 3D imaging and (D) transversal SPECT/CT in the paravertebral muscle on the right. (E) In another patient with a melanoma of the back, posterior planar imaging shows drainage to both axillae and the left extracurricular region (arrow). This sentinel node is anatomically indicated on (F) 3D imaging and transversal (G) SPECT/CT and corresponds on (H) CT with a slightly enlarged node (circle), which contained metastases at histopathology. (I) In another patient with a melanoma of the left underarm, anterior planar imaging shows drainage to the left axilla and to the area of the left elbow (arrow). (J–L) On SPECT/CT, this sentinel node is seen as a dorsal subcutaneous node.
SPECT/CT: Single-photon emission computed tomography images with integrated computed tomography.
Reproduced with permission from [1] © Future Medicine Ltd. (2014).
Effectiveness & value of SNB
The SN is identified and excised in approximately 100% of the melanoma patients. The probability of finding metastatic SNs grows accordingly with the increase in T stage of the lesion [31]. Furthermore, a study recently showed a direct correlation between smoking and SN node metastasis [32]. Long-term postsurgical complications are presented in up to 18% of cases, the most remarkable being lymphedema of the lower limb after SNB in the groin [33]. The reported false-negative rates (i.e., patients that present a positive second echelon lymph node in basins with a negative SN at histopathology) ranged from 0 to 38% [6,34–36]. The high false-negative values of some series may be partially explained by the complexity of the lymphatic drainage. In the head and neck area and the posterior trunk, these values tend to be higher than in other sites [37]. In an extensive meta-analysis including 71 studies and 25,240 patients, the average false-negative rate was 12.5%, varying from 0% (limbs) to 34% (head and neck) in complex regions. The overall estimated risk of nodal recurrence after a negative SNB was found to be 5% or less [38]. Another important factor associated with increased risk of nodal metastases has been described for melanomas with multiple lymphatic basin drainage. Although this has already been observed for head/neck melanomas, truncal melanomas with multiple drainage routes have a less favorable survival when compared with a single draining lymphatic pattern [39].
Even though some researchers discuss whether all microscopic metastases will progress into clinical nodal disease [40], there is an increasing amount of publications supporting the issue that, when left to observation, clinically hidden lymph node metastases have a tendency to grow and spread to other regional nodes and systemically, decreasing the chance of prolonged survival [2–5,41–43].
Final results from the MSLT-1 trial confirmed SN status as the most important prognostic factor and that patients who underwent SNB had fewer recurrences than patients who underwent nodal observation. Moreover, a 20% melanoma-specific survival benefit was observed in SN-positive patients who underwent immediate lymph node dissection compared with patients in which delayed lymphadenectomy was done due to recurrence detected in regional lymphatic basins after clinical observation [4,5,19]. The SNB is now considered to be standard care in the majority of clinical guidelines [2,4,6–8,44–50] and in cases with challenging SNB procedure and failure to remove the SN, ultrasound follow-up of the lymph node stations at risk identified by lymphoscintigraphy is strongly recommended as management strategy [51].
Nevertheless, recent evidence has emerged about the use of immediate completion of lymph node dissection after positive SNB [19,20]. MSLT-II and DeCOG-SLT trial results showed no melanoma-specific survival benefits when comparing immediate lymphadenectomy versus active surveillance of the nodal basin with ultrasonography in patients with metastatic SN. Even though immediate completion lymph-node dissection has proved to be of value regarding staging and ameliorated rate of regional disease control, the high incidence of complications (24 vs 6% of lymphedema) is considered prohibitive taking into account the lack of survival benefit. Considering both MSLT-I and MSLT-II results, it seems fair to recognize some potential therapeutic value of the SNB procedure as increase in survival occurred exclusively among patients with disease limited to the SN [52]. On the other hand, patients with non-SN metastases can still undergo complete nodal basin dissection but not immediately after SNB; active surveillance of the nodal basin seems to be a safe and efficient way to identify patients who are most likely to benefit from delayed, selective, node-directed treatment [53].
After obtaining these new data, SNB relevance has therefore been reinforced. Now more than ever, a careful and precise identification of the SN is required to grant in one single procedure valuable prognostic information as well as specific survival benefit until initial nodal recurrence.
Clinical indications
The different guidelines agree that it is appropriate to offer SNB to those patients presenting a clinically localized melanoma with a significant risk of nodal involvement, depending on histopathologic characteristics of the melanoma [7,50]: in those cases where primary lesion Breslow's thickness is ≤0.75 mm, SNB is not recommended, as the risk of lymph node involvement is around 1%. When thickness varies from 0.75 to 1 mm, some authors thoroughly recommend SNB since the risk for regional lymph node metastases is nearly 5% [54]; others reserve it for patients with presence of ulceration in melanoma or high mitotic rate [7,55] in whom the risk of nodal involvement is higher than 10% [7,34]. The indication in these patients is warranted because of the need for staging and obtaining prognostic information in this type of patients. SNB is generally offered to all patients with a melanoma lesion thickness from 1 to 4 mm due to the 8–30% risk of nodal metastasis and, based on MSLT-I results, because their early treatment improves the survival rate [56]. Patients with melanoma thicker than 4 mm present a possible nodal involvement in around 40% and a higher risk of distant metastasis; however, metastatic lymph nodes are usually clinically nonpalpable, so SNB may constitute important prognostic information which may be relevant to prevent regional recurrence [57].
A multidisciplinary approach
SNB is a procedure that involves a multidisciplinary team requiring a close cooperation between nuclear medicine staff, surgical specialists and nurses as well as pathologists for accurate presurgical and intraoperative SN localization and harvesting and subsequent pathological assessment [37]. In order to ascertain which nodes are eligible to be considered SNs, both the administration of a radioactive tracer and a subsequent pre- or intraoperative procedure of SN identification are needed.
The original blue dye approach (‘open and see’), described by Morton et al. to identify SNs in expected areas of lymphatic drainage was based on the visualization of blue-colored lymphatic channels and lymph nodes directly draining from the tumor site immediately after skin incision.
Blue dyes
The blue dye (isosulfan blue, patent blue V or methylene blue) is intradermally administered 10–20 min prior to skin incision in a volume ranging from 0.5 to 1 ml surrounding the primary melanoma or biopsy scar. The dye quickly spreads through the lymphatic channels and enters into the SNs without being trapped. The washout time is approximately 45 min [58]. The blue-stained channels are identified in the regional lymphatic basin and dissected downstream to the SN [50,59]. The main drawback of these kinds of dye is that they do not allow preoperative lymphatic mapping. Another cosmetic disadvantage is that blue dye might cause, in some cases, a long-term tattooing of the wide excision skin limits; this is especially relevant in cases of head and neck melanoma patients. As blue dyes are invisible from the skin surface they are usually injected in the operation room and combined with previous administration of radiocolloid tracers.
Recently, the use of blue dye in the SN procedure has been questioned because of its negligible additional value, calculated in 0.9% on the basis of 681 evaluated melanoma patients, to detect nonradioactive blue SNs during the surgical act [60].
Radioactive tracers
The advent of radiotracers in the SNB approach permitted visualization of the physiologic process of lymph drainage using gamma cameras. The images provided by preoperative lymphoscintigraphy transformed the original ‘open and see’ paradigm into a ‘see and open’ approach because it was able to visualize the melanoma's lymphatic drainage pattern, accurately improving the surgical plan. Lymphatic mapping should be performed, where possible, before wide local excision of the primary melanoma, because excision may disrupt lymphatic channels causing migration of the tracer to other lymph nodes that initially do not receive direct drainage from the melanoma [24].
The radiotracers utilized for lymphoscintigraphy are usually 99mTc-colloids that travel through the lymphatic vessels at different speed depending on their particle size and are then trapped by macrophages in the lymph node.
As previously mentioned, the radiotracer is intradermally injected around the primary lesion or the scar of the resected melanoma (two to four depots). Early dynamic imaging is recommended as it allows the visualization of the radiotracer's passage through the lymphatic ducts and its arrival and trapping in the first SN(s). Subsequently, early and late planar static images of all potential receiving drainage basins are acquired for 3–5 min. During image acquisition, a 57Co or 99mTc flood source or point source can be used to delineate the body contour. Finally, the location of each SN is indicated on the skin using a pointer and an indelible ink marker [50].
A new radiotracer (99mTc-tilmanocept) has been introduced first in trials and recently for clinical practice. 99mTc-tilmanocept (Lymphoseek®) accumulation does not depend on particle size (diameter: 7.1 nm), its chemical structure and small size enable 99mTc-tilmanocept to exit the injection site rapidly and quickly accumulate in first-echelon nodes. The interactions between the mannose moieties of 99mTc-tilmanocept and CD206 macrophage receptors enable avid binding to the target receptor and retention for up to 30 h, limiting transit to second-echelon nodes. It has been demonstrated that SNB with 99mTc-filtered sulfur colloid and with 99mTc-tilmanocept performed similarly in detection of SNs. However, lymphatic channel visualization was different and these structures were seen more often with 99mTc-filtered sulfur colloid than with 99mTc-tilmanocept [61].
This tracer was approved by the US FDA in 2013 and by the EMA in 2014, but it was not introduced to some European countries until mid-2017. Although several multicentric trials in the USA have been conducted with this new radiotracer, no head-to-head comparison against other available radiotracers has been performed. However, preliminary results in 156 patients with melanoma showed an SN identification rate of 100% without any false-negative cases nor any adverse reactions directly related to 99mTc-tilmanocept [62].
Available data suggest that 99mTc-tilmanocept has the potential to replace current colloidal radiotracers due to the aforementioned properties, however, future cost–effectiveness studies must be performed.
Fluorescent agents
The implementation of near-infrared (NIR) dyes allows the intraoperative visualization of lymphatic vessels and identification of the SNs, with the potential to reduce surgical time and improve lymph node detection with reduction of incision size [63]. Although real-time transcutaneous visualization (up to 1 cm depth) has also been described, this is limited to a minority of patients [64]. Indocyanine green (ICG, Verdye Diagnostic Green, Aschheim-Dornach, Germany) is the fluorescent agent most frequently used for SNB with application in neoplasms like breast, colon or gynecological tumors [63,65–67].
The NIR fluorescent light is practically invisible to the human eye and therefore special intraoperative imaging devices are needed to excite the NIR tracer in the lymphatic system, collect the subsequently emitted photons and fuse the acquired information with the real-time video image of the surgical field [65,68]. Different groups have reported that for effective intraoperative (NIR) fluorescence imaging, the lights in the operating room have to be dimmed or switched off for accurate fluorescence signal visualization [65,69]. Nowadays, however, there are new devices that allow the surgeon to excise the lesion of interest under real-time fluorescence guidance in ambient-light conditions [70].
Even though published results on fluorescent dyes in dermatologic tumors are promising [71–73], most of studies do not recommend an independent use due to their limitations to identify deep-located SNs because of a limited tissue penetration, a short diagnostic window due to the diffusion of the agent and the very limited ability to perform preoperative lymphatic imaging for surgical planning [74–76].
It should be noted that while blue dyes or fluorescent tracers are able to depict lymphatic vessels and nodes for a limited window of time (‘open and see’), radioactive tracers have the particular feature of being retained within the lymph nodes due to macrophage phagocytosis. Hence, the ‘see and open’ approach simultaneously allows a careful presurgical examination of the lymphatic drainage as well as a suitable tool for intraoperative identification of the SN with the injection of a radiotracer in the same procedure.
SPECT/CT anatomic & functional imaging fusion
When lymphatic drainage is unclear or the SNs are situated profoundly under the surface or closely placed next to the injection site, conventional planar lymphoscintigraphy can sometimes be challenging for SNs identification. Modern gamma cameras are able to acquire SPECT/CT and fuse the functional SPECT images with the anatomical data from CT in three dimensions [77]. This modality implements scatter- and attenuation-corrected of the nuclear images obtaining superior contrast and higher resolution images significantly enhancing the ‘see and open’ approach. They provide better anatomical localization and are capable of modifying the evaluation of planar images in terms of number and localization of SNs. Furthermore, SPECT/CT can discriminate precisely the activity arising from two closely placed nodes that are usually depicted as a single hot spot on planar images. In line with numerous studies [78–83], a prospective multicenter trial has proved the additional value of SPECT/CT in melanoma as well as in other neoplastic diseases such as breast and pelvic cancer, recommending to perform SPECT/CT in all patients with melanoma of the head and neck and trunk and in all melanoma patients with unexpected drainage on planar images. In this study, SPECT/CT information not only led to the detection of more SNs, but also to a 37% surgical adjustment [84]. Furthermore, one study [85] showed that although the addition of SPECT/CT to the SNB procedure in patients with cutaneous melanoma increases the preoperative imaging costs slightly, it was associated to a 30% reduction in costs achieved by decreasing operative time, hospital stay duration and also contributing to a more frequent use of local anesthesia compared with general anesthesia.
This hybrid imaging technique can accurately depict lymph nodes placed in the vicinity of one another and also SNs adjacent to the injection site where visualization is often hampered by the scatter activity. Additionally, SPECT/CT can contribute to the recognition of non-nodal tracer accumulation and also provide important information in cases of unclear drainage patterns with inconclusive interpretation from the conventional images (e.g., nonvisualization or unclear location of the nodes). SPECT/CT is of special value in cases where conventional planar images are not able to discriminate between an interval SN and a lymphatic lake, lymphangioma or contamination of the skin [28,77,78,86–88].
SPECT/CT facilitates the appraisal of the number and size of SNs allowing correlation of focal uptakes to morphological structures. The assignment of different colors to the diverse anatomical structures in volumetric rendering techniques provides a better 3D recognition of the anatomical landmarks and the SN location. This facilitates the SN resection by allowing a proper planning of the localization and size of the surgical incision and provides a better understanding of the lymphoscintigraphic information [87].
Alvarez Paez et al. stated that in 50% of patients with melanoma of the trunk, SPECT/CT hybrid-fused images provided clinically relevant information and visualized additional SNs in 23% of the cases. Moreover, the surgical approach was modified in 35% of such patients by demonstration of metastases in SNs depicted exclusively by the SPECT/CT, contributing to a more accurate staging and a reduction of potential false-negative results [87]. The study of Stoffels et al. [89] revealed that in the head and neck area and in obese patients with melanoma, the identification of the SNs was improved by the SPECT/CT when compared with planar lymphosintigraphy. Furthermore, this study, based on the retrospective evaluation of two historical cohorts, showed that a higher frequency of metastatic involvement and a higher rate of disease-free survival were associated with the use of SPECT/CT-aided SNB compared with SNB without SPECT/CT assessment.
All available studies support that the aforementioned advantages of SPECT/CT overcome the reported disadvantages of the technique (increased dosimetry, additional costs and exploration time).
Despite all its virtues, SPECT/CT should always be performed in combination with planar lymphoscintigraphy. SPECT/CT performed independently is not able to replace the sequential information provided by dynamic lymphoscintigraphy and subsequent planar imaging, which is the standard technique to identify the individual lymphatic collectors reaching the SN, an essential aspect to distinguish an SN from a secondary radioactive lymph node [90].
Handheld gamma probe
The gamma ray detection probe detects the photons emitted by the radiocolloid tracer. The handheld probe consists of a radiation detector surrounded by metal shielding as collimation in order to obtain a restricted field of view that allows a precise orientation and spatial discrimination between different, one another closely placed, focal activities [50]. The radioactive signal is often represented as an audible pitch or volume variation sound along with the visual display, adding an acoustic element to the ‘open and see’ approach.
The information provided by the lymphoscintigraphy and the mark placed on the surface of the skin, in combination with the ability of a handheld gamma probe to identify the radioactive signal of the SN also from the skin surface, allows the surgeon to place the incision more precisely. The gamma detection probe guides the surgeon to dissect all tissues required to identify the radioactive SN. The surgical bed should always be examined to confirm complete removal of all SNs and ex vivo count rate activity of the SNs has to be documented. The statement that node resection is recommended until residual surgical bed activity is less than 10% of the hottest SN [91] should not be taken as a rigid rule when an adequate use of preoperative lymphoscintigraphy and eventually SPECT/CT is performed for SN identification. If the SN concept is clearly understood and a strict technique followed, virtually all metastatic nodes can be identified [59]. Mastering this technique requires a learning curve and is operator-dependent.
Portable gamma cameras
Thanks to the anatomical landmarks provided by SPECT/CT, the ‘see and open’ paradigm has gradually evolved into a ‘see, open, and recognize’ paradigm. This approach has been reinforced by the intraoperative incorporation of modern portable imaging technologies (PIT) and the development of new hybrid radioactive-fluorescent tracers. In centers with advanced portable technologies, the original role of blue dye to visually identify SNs into the operating room is gradually taken over by the use of PIT devices and hybrid tracers, especially in areas of complex anatomy or tumors with deep lymphatic drainage.
SPECT/CT combined with PIT allows a more reliable use of the gamma probe, mostly in situations where SNs are located close to the injection site and in cases of complex lymphatic drainage areas. To grant its usefulness in the operation room, these types of devices need to fulfill several specific requirements. They should be of stable design, be compact and facilitating portability, small enough for maneuvering during the operation, enabling an adequate spatial resolution with generation of fast real-time imaging as well as to be able to allow the possibility to change and adjust their position to obtain images from special angle projections.
The majority of the systems consist of an articulated arm holding 2D arrays of scintillation crystals or solid-state semiconductor photodetectors connected to photomultipliers. Collimation of this system is crucial for a precise detection and visual representation of the activity emitted from the 99mTc colloids. Recently, a portable gamma camera equipped with a pin-hole collimator was able to detect SNs at a distance of at least 3 mm from the injection site [92]. This device is able to discriminate between two different energy peaks and display them separately on the screen, allowing the use of external pointers such as 153Gd, 133Ba or 125I. Systems such as Sentinella S102 (Oncovision-GEM Imaging, Valencia, Spain), LumaGEM® (Gamma Medica Ideas, CA, USA) and eZ-SCOPE (Anzai Medical, Tokio, Japan) are all particularly usable in problematic cases detected during presurgical assessment or even when the lymphoscintigraphy is not available [93,94]. In the operation room, the portable gamma camera's detector arm can be placed above the previously marked skin locations, thanks to the help of a cross-shaped laser beam projected on the skin of the patient, simultaneously with display on the screen of the portable gamma camera. The matching of these two signals shows that the exact location of the SN can be identified by the matching of this information with an external pointer, such as a surgical pen, that can be used as a spatial reference.
Recently developed devices have managed to successfully combine scintigraphic and optical images in real-time with the addition of an optical camera to the portable gamma camera, providing an anatomic reference within a more friendly an intuitive environment [95–97]. An example is shown on Figure 4.
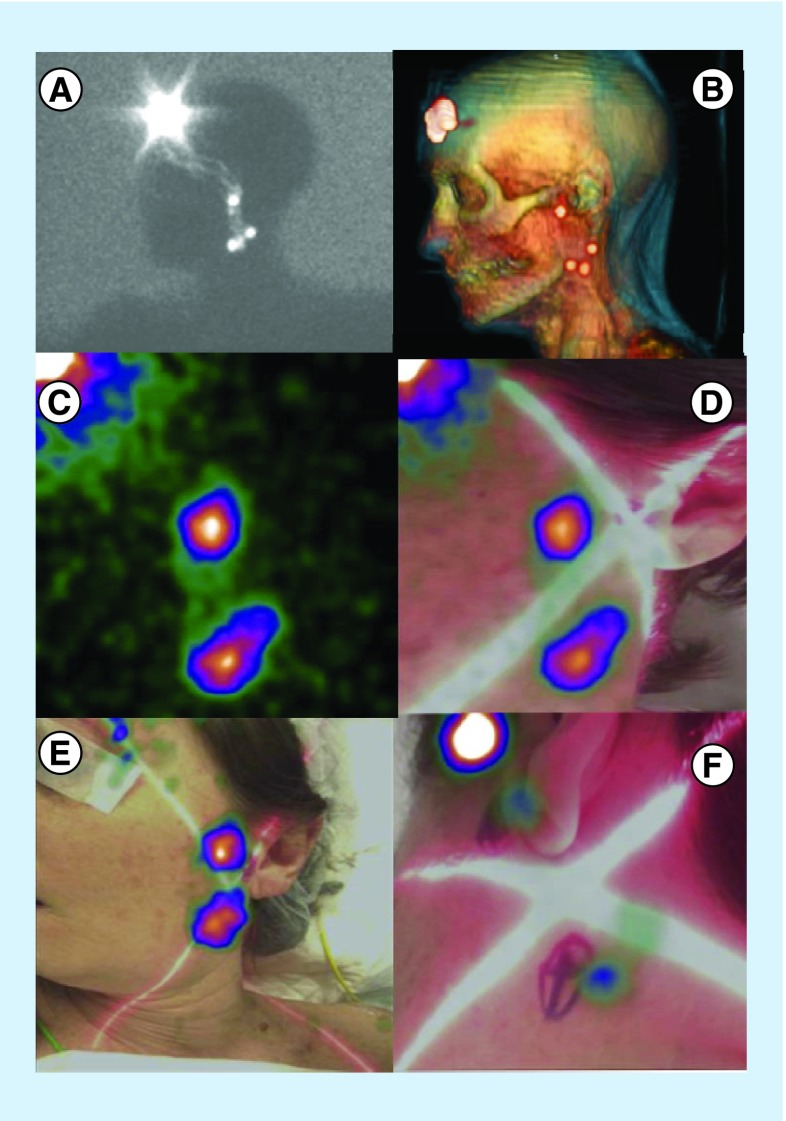
Figure 4. . Patient with frontal melanoma.
Fourty-eight-year-old female with a left-sided frontal melanoma (2.5 mm Breslow thickness). Lateral view planar image showed several lymphatic channels aiding to different SNs (A). SPECT/CT data with volume rendering showed a preauricular SN and other SNs in left cervical level II situation (B). Portable gamma camera demonstrated significant activity in those areas but without any anatomical environment (C). Hybrid gamma camera (with and optical component) provided a more convenient anatomical landscape (D). Placing the hybrid gamma camera at different distances allows the nuclear medicine physician or surgeon to better depict and overview of the SN distribution (E) and to mark very precisely the hot nodes on the skin (F).
SN: Sentinel node; SPECT/CT: Single-photon emission computed tomography images with integrated computed tomography.
Free-hand SPECT
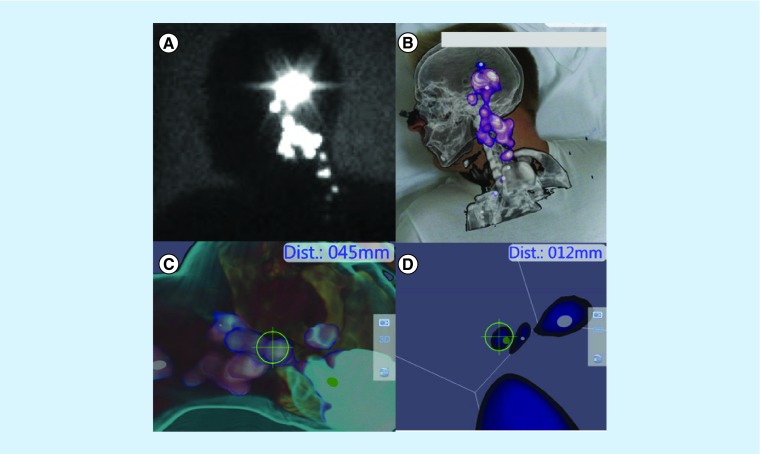
A few years ago, an innovative free-hand SPECT device (DeclipseSPECT, SurgicEye, München, Germany) was introduced, facilitating the readings in counts per second supplied by a standard handheld gamma probe with SPECT image generation of the target lymph node, thanks to the registration of all the spatial movements of the probe by the free-hand SPECT tracking system. For spatial positioning, infrared technology is used to track two fixed detachable fiducial markers: one placed on the patient's body and another one on the probe. Then, when all data are collected, a reconstruction algorithm generates a 3D image that is finally coregistered in real time with the image captured by a video camera focused on the surgical field. Situation and depth of scanned radiolabeled tissues can be easily checked on the screen by the surgical team. This novel tool permits the generation of intraoperative 3D images of the radioactive SN, also offering the possibility to load and fuse them in real-time with tracked SPECT/CT studies previously acquired. Following the concept of mixed reality, all this information can be visually superimposed on the patient's body using incorporated augmented reality devices. Sulzbacher et al. studied the clinical usefulness of free-hand SPECT in melanoma patients and found that it was able to not only provide precise anatomical information of the SN location but also revealed additional SNs compared with SPECT/CT [98]. The use of this kind of device can enhance SN identification and resection enabling an extended use of radioguided surgery to other fields such as nonpalpable lesion localization and parathyroid resection [99,100]. The next step of this approach is the use of a tracked portable gamma camera aimed to obtain a higher sensitivity than the gamma probe. Some efforts have been made in this way and this approach, compared with the conventional lymphoscintigraphy, showed highly reproducible images [101]. An example of this type of devices is shown on Figure 5.
Figure 5. . Free-hand single-photon emission computed tomography.
Delayed lateral planar image belongs to a 34-year-old male patient with a melanoma (3.2 mm Breslow thickness) in his left ear. Several hot spots are depicted from preauricular area to left cervical (A). After loading SPECT/CT-tracked data volume, rendering reconstruction clearly depicts the lymph nodes in the anatomical environment of level II–III in the neck (B). Using tracked free-hand SPECT acquisition navigation to the nodes is possible (C) and every node can be observed as a 3D virtual spot. The distance from the tip of the gamma probe (12 mm) is depicted in real-time (D).
SPECT/CT: Single-photon emission computed tomography images with integrated computed tomography.
Hybrid tracers
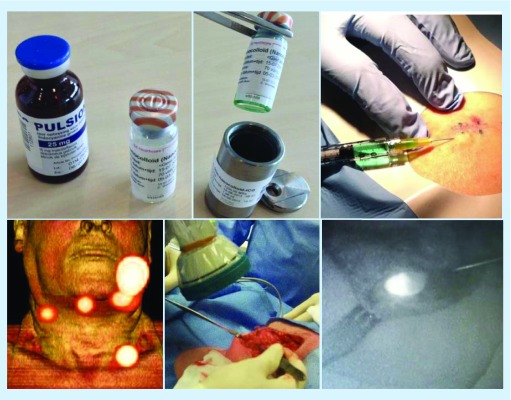
Radiolabeled nanocolloid has been combined with ICG (ICG-99mTc-Nanocoll®) to assist the intraoperative identification of SNs located in areas of difficult surgical access such as pelvic nodes from prostate and vulvar cancer or lymph nodes situated in complex locations as in cases of head and neck cancer. These hybrid agents for bimodal imaging combine radioactivity and fluorescence in one signature, providing the best of both worlds to overcome the limitations in which the independent use of fluorescent dyes presents to add preoperative lymphatic mapping to intraoperative detection [102]. There is evidence that hybrid tracers are capable of reproducing the exact pattern of lymphatic drainage as the parental 99mTc-colloid-radioactive tracer, allowing the combined use of presurgical lymphoscintigraphic planning and intraoperative NIR optical detection (Figure 6) [100,103–105]. The use of hybrid tracer has been proposed as the optimal approach for SNB in melanoma with particular application in complex cases such as in the head and neck region where SNs are placed in close proximity to site of the injection [75,76]. The radioactive component of the hybrid tracer allows to perform the preoperative lymphoscintigraphy with SPECT/CT, while at the same time, the fluorescent component is not hampered by the background signal coming from the site of injection. Therefore, the SN can stand out among a cluster of lymph nodes and the surrounding structures during visual identification [106].
Figure 6. . Nanocolloid and indocyanine green hybrid radiotracer.
After addition of ICG to radioactive nanocolloid the hybrid tracer ICG-99mTc-nanocolloid is injected intracutaneously around the excision scar of a melanoma (above). On SPECT/CT (below on the left), drainage from the primary lesion in the left cheek to four sentinel lymph nodes in both sides of the neck is observed. Subsequently, a dedicated fluorescence camera (middle) enables intraoperative sentinel lymph node identification and excision (on the right).
ICG: Indocyanine green; SPECT/CT: Single-photon emission computed tomography images with integrated computed tomography.
Lately, studies with a recently developed hybrid optonuclear probe have been reported [107,108]. This new device allows the detection of both radioactive and fluorescent signals through integration of two optical fibers (one for excitation and the other for emission signal collection) into the handheld gamma probe. In this manner, it is possible to get a quantitative feel of the fluorescent signal intensity by means of acoustic readout in a very similar way to the one of conventional gamma probes. However, further studies must define the potential role of this type of devices, as the use of radioactive tracers is still required because of the short penetration depth of fluorescent dyes that could severely limit the surgical guidance. Current efforts are being made with the aim to develop a device that can integrate both scintigraphic and fluorescent imaging modalities in the operation room [109].
Although blue dye or a radioactive tracer alone can provide excellent results, the most preferred approach is the use of a combination of the available techniques [44]. The array of nuclear medicine tools for SNB in melanoma has been refined with the recent addition of fluorescent/radioactive hybrid tracers. However, before these hybrid tracers can become available for clinical practice in many European countries, regulatory approval of hybrid tracers is still needed. Also further financial evaluation is required, because even though the cost of fluorescent tracers vial is relatively low, purchasing NIR camera systems implies higher additional costs [75].
Based on the upgraded approach ‘see, open and recognize,’ it is possible to add navigation technologies to the combined use of SPECT/CT and PIT. Thanks to the implementation of all the technological advances in a context of augmented reality, an adequate combination in the use of multiple portable devices such as NIR cameras, gamma probes and gamma cameras can guide surgical exploration by fusing all information in real-time images. Substantial efforts are currently being made in combining all available features into one single hybrid device, integrating the information via intuitive screens or other portable devices such as glasses or head-mounted displays with the ability to visualize virtual elements merged with the real objects onto the field of view.
Irrespective of the chosen method for surgical SN identification, it is recommended that surgeons always palpate the nodal basins in search of suspicious lymph nodes unable to incorporate the tracer due to blockage by metastatic tumor [50].
In conclusion, despite OS benefit has not been demonstrated, SNB is recommended in patients with thick and intermediate-thickness melanomas because it is the single most important prognostic factor, due to the described melanoma-specific survival improvement when complete lymphadenectomy or active ultrasonography surveillance was performed immediately following SNB in patients with metastatic nodal involvement and also because it is a low morbidity procedure that can identify patients candidates for adjuvant therapy.
Role of positron emission tomography
Positron emission tomography, as diagnostic modality, is able to depict metabolic changes that precede anatomic alterations. The modality was introduced in the last decades of the past century but in this millennium has gained in importance, thanks to its integration with CT and more recently with MRI. The most used radiotracer for PET in oncology is FDG because of its capability to represent the neoplastic hypermetabolism due to the incremented glucose consumption of cancerous lesions. Melanoma lesions are highly avid for FDG, allowing the use of FDG-PET/CT in selected melanoma patients. In this part, FDG-PET/CT applications will be discussed with special emphasis on their uprising part in therapy response assessment regarding the introduction of new targeted therapeutic agents for advanced-stage melanoma.
FDG-PET/CT in early-stage melanoma
Most cases with clinically localized melanoma show no evidence of disseminated disease and if they do, metastases are often of minimal size. In this type of patients, the yield of FDG-PET/CT is hindered by its limited spatial resolution in contrast with the previously mentioned SNB, which benefits from microscopic examination [110,111]. The lack of usefulness of FDG-PET/CT has already been described for metastasis detection in cases of clinically localized melanoma [109] and the superiority of SNB has been advocated in a systematic review [114]. Moreover, in a study including 64 T2–T4 melanoma patients, preoperative FDG-PET/CT was positive in only two out of 19 patients with subsequently confirmed metastases in surgically removed SNs [110]. However, in individual clinical situations assuming a high risk of metastasis (Breslow > 4 mm, presence of ulceration and high mitotic index and trunk and upper extremities), some authors consider that this technique could provide useful information [111].
An important effort is currently being made to improve PET's capacity for microscopic metastatic disease detection. Even though spatial resolution has substantially incremented from the first positron emission tomographer, there is no evidence that PET/CT using 18F-FDG might play a role in the staging and surgical management of early-stage melanoma patients.
FDG-PET/CT in advanced-stage melanoma
PET/CT with 18F-FDG may play an important role in determining which lesions should be surgically removed in cases of lymph node metastases in stage III patients and in cases of of limited quantity of distant organ metastases in stage IV patients [115].
It has been shown that FDG-PET/CT is more sensitive and, at least as specific as other imaging techniques like ultrasound, CT and MRI. FDG-PET has significantly enhanced the detection of lung metastases, thanks to the addition of CT as part of multimodal imaging. Additionally, some authors state that for the diagnosis of bone metastases PET/CT may be the most accurate study, outdoing bone scintigraphy [7]. In a comprehensive review including 28 studies, a pooled sensitivity of 83% and a pooled specificity of 84% confirmed the superiority of FDG-PET/CT in comparison with other imaging modalities [115]. The same systematic review involving 2905 patients suggested that FDG-PET/CT was associated with 33% disease management changes (range: 15–64). Even though small brain, lung or liver lesions can sometimes be missed, FDG-PET/CT is able to perform a whole-body evaluation of the patient (Figure 7), with the benefit of detecting second incidental neoplasms [117]. Although inclusion of lower extremities and head in FDG-PET/CT scans increases by only 15 min the acquisition time, Niederkohr et al. have described that the inclusion of these regions is not justified in terms of changes to clinical management for patients with unknown or suspected primary or metastatic melanoma in these parts of the body [118]. In a large meta-analysis based on 74 studies, PET/CT with a sensitivity of 86% appeared to be better than PET (82%) and CT (63%) alone [119].
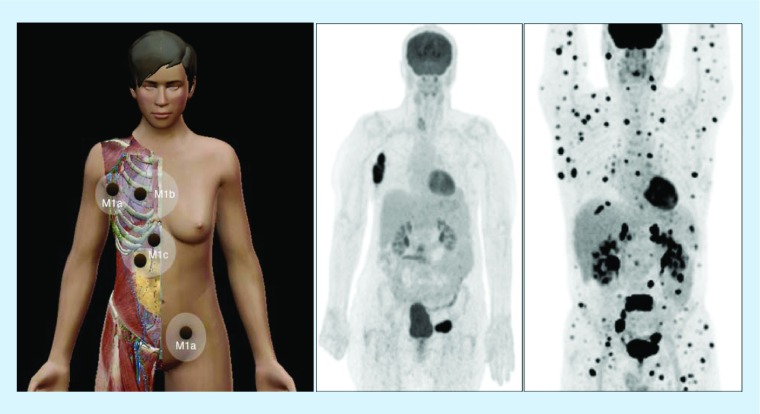
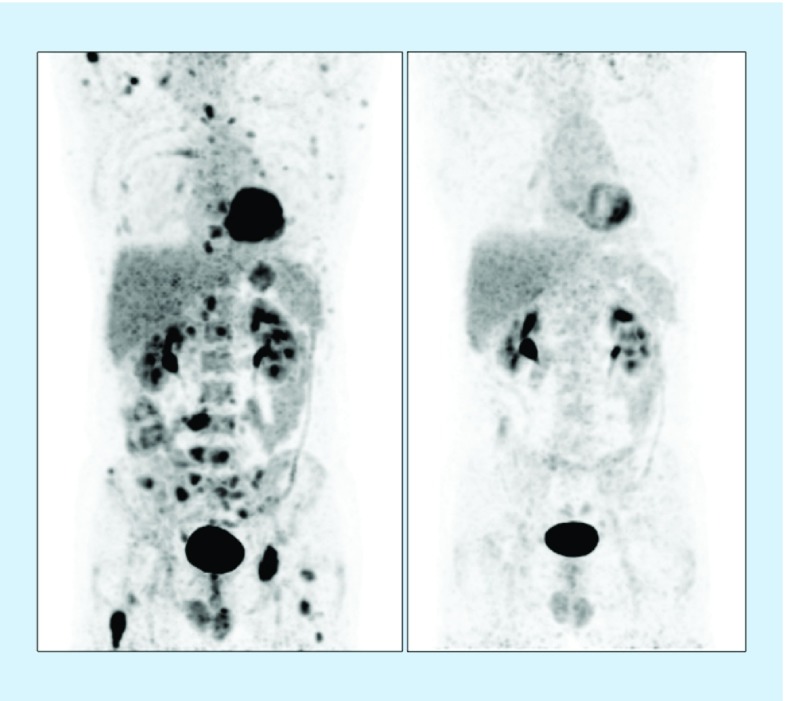
Figure 7. . Melanoma dissemination.
On the left, scheme illustrating disseminated disease in melanoma with distant metastases in lymph nodes (M1a), lung (M1b) and liver/bowel (M1c). Middle, 18F-FDG-avid lymph node metastases in right axilla and left groin as displayed on PET with maximum intensity projection. On the right, multiple FDG-metastases in another patient with widespread disease.
FDG: 18F-fluorodeoxyglucose.
When surgical treatment is considered, an accurate identification of all sites of metastatic spread is of paramount importance [120]. It has been described that complete resection of metastases can be achieved in 70–80% of the patients with M1a disease (skin, subcutaneous or distant lymph node metastases) [121] and one study revealed that surgical candidacy can be achieved in over half of the patients with stage IV for resection of recurrence lesions [122]. Despite some lack of consensus regarding patient's selection criteria and the standardization of surgical procedures [123], a careful selection of candidates for metastasectomy is needed in order to identify those patients with slow growing oligometastases following a long disease-free interval after treatment of their primary melanomas. For that purpose, evaluation with PET/CT and brain MRI in conjunction with an exhaustive preoperative workup is needed to determine the extent of the disease [124]. Consequently, as described earlier, GOSTT techniques can offer a variety of reliable tools for the presurgical planning and intraoperative identification of the metastatic lesions to be resected.
Although intraoperative use of systemic-administered FDG in combination with gamma detection probe technology has been advocated to manage cases of metastatic melanoma, further development and evaluation of this technique is in progress [125]. A more practical approach within the spectrum of GOSTT procedures is the intralesional administration of a hybrid tracer with the aim of guiding isolated melanoma metastases resection, according to the original radioguided occult lesion localisation (ROLL) principle developed for breast cancer (Figure 8). Additionally to the radioguided detection, the fluorescence component of the hybrid tracer enables high-resolution intraoperative visualization of the target lesion [22,126].
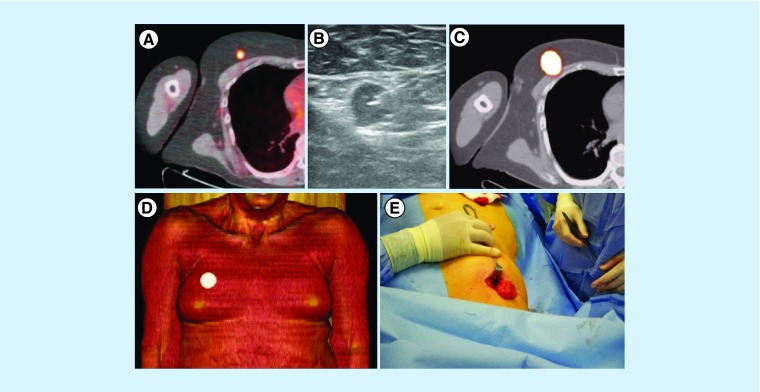
Figure 8. . Radioguided surgery of isolated melanoma metastases.
(A) In a melanoma patient, PET/CT showed a solitary 18F-FDG-avid lesion in the right breast. (B) After ultrasound-guided injection of a radioactive tracer, the lesion was indicated by (C & D) SPECT/CT and subsequently (E) resected by the surgeon under guidance of a portable gamma-ray device.
FDG: 18F-fluorodeoxyglucose; SPECT/CT: Single-photon emission computed tomography images with integrated computed tomography.
Reproduced with permission from [1] © Future Medicine Ltd. (2014).
Even though FDG-PET is considered not indicated as a routine study for staging patients with positive SNB [127], the technique might be justified in cases of palpable lymph nodes before resection because of its potential repercussions on decision management [128,129].
FDG-PET/CT in follow-up & melanoma recurrence
Based on the available data, there is not enough evidence to state that surveillance of melanoma patients improves regional control, survival and quality of life of patients with melanoma. A small number of metastatic lesions are detected by routine imaging studies or blood testing and the amount of melanoma patients who survived because they underwent these tests is even lower. Far more often, needless concern due to false-positive results occurs, and can lead to further avoidable testing that may even result in unnecessary surgery. On the other hand, out of 110 asymptomatic stage IIB–IIIB patients recurrence was detected with a single PET/CT in 10% of the cases, whereas 31% of the patients with a negative PET/CT subsequently developed recurrences at a median time of 19 months [130]. Although FDG-PET/CT is not recommended for routine follow-up, it constitutes an excellent tool for recurrence assessment [128]. In a single whole-body scan, this hybrid whole-body imaging technique allows evaluation of extent of disease, with a higher sensitivity than conventional imaging studies [131]. In the specific group of patients where valuable additional information can influence management decisions, FDG-PET/CT can be recommended, especially to confirm suspicious lesions detected by other studies, in patients with increased serum S-100B during follow-up [132] or even in the absence of an elevation of this serum protein [133], cases of suspected or at high risk of nodal or distant metastatic disease, and for restaging of patients before surgical metastasectomy [111,134].
FDG-PET/CT in the assessment of therapy response
FDG activity of neoplastic lesions should decrease when there is destruction of tumor cells due to effective therapy, as FDG uptake is supposed to reflect the amount of viable tumor cell numbers. Nevertheless, it should be borne in mind that sometimes an increase of standard FDG uptake value can be seen in relation to the inflammatory reaction associated with tumor necrosis. Standard FDG uptake value reduction is to be expected in responding patients after effective chemotherapy, no significant uptake reduction variations in nonresponders and uptake increment when progressing disease [135]. The significance of FDG-PET/CT for the assessment of therapy response has gained in relevance with the definition of Positron Emission Tomography Response Criteria In Solid Tumours [136], but the implementation of new targeted therapeutic is challenging the current body of knowledge introducing novel concepts for a new immune-related response criteria.
Generally, melanoma is considered mostly insensitive to chemotherapy. Only high doses infused during isolated regional perfusion can obtain considerable remissions. Although PET has proved its usefulness for monitoring the effect of this kind of therapy, in many cases minimal remaining tumor rests cannot be detected [137]. Two new forms of systemic therapy have recently become available with promising results: molecular targeted therapy and immunotherapy.
Molecular-targeted therapy
Vemurafenib, dabrafenib or encorafenib are BRAF inhibitors, a gene whose mutation is found in 40–50% of metastatic melanomas. Several studies have shown improvement of progression-free survival and OS [138–140], but while responses happen very quickly (Figure 9), most patients recur due to acquired resistance [141,142].
Figure 9. . Therapy response assessment with PET.
Patient with multiple FDG-avid metastases on baseline PET (on the left) with complete response on PET after therapy with BRAF-inhibitors (on the right).
FDG: 18F-fluorodeoxyglucose.
It has been described that early reduction of lesion's metabolic activity displayed on the FDG-PET/CT could be associated with OS [143,144] despite a lack of reduction in tumor dimension, also absence of early reduction in FDG uptake could indicate primary refractory disease sites.
Resistance to treatment can be described as persistence or reactivation of FDG uptake and some patients may even show what is considered as an ‘heterogeneous response’, which could be defined as a situation of complete or partial metabolic response with simultaneous progressing or new lesions, or even a situation of response with more than 10% of lesions having ‘stable metabolic disease’. Heterogeneous responses were associated with shorter time to progression but not with OS [145].
It is important to bear in mind other possible causes before classifying a FDG-PET/CT study as ‘progression’. Sometimes new hypermetabolic cutaneous lesions (squamous cell carcinomas and keratoacanthomas) can be developed as a secondary effect of BRAF-targeted therapies. Additionally, an immune flare response should also be considered when increased or ongoing FDG uptake in the context of progressive tumor shrinkage or when accompanied by increased activity in the spleen [145].
Immunotherapy
Ipilimumab, nivolumab and pembrolizumab are monoclonal antibodies that block physiological inhibitory checkpoints of immune activation and their use has demonstrated survival benefit with durable response [146]. Combination of these monoclonal antibodies seems to be associated with longer progression-free survival but also with higher toxicity [147]. More frequent and severe immune-related adverse events are associated with ipilimumab; these include enterocolitis, dermatitis, hepatitis and endocrinopathies. Some of these can be spotted on FDG-PET/CT imaging, being autoimmune thyroiditis the most common but the pituitary fossa, adrenals, lungs, liver, pancreas and bowel can all demonstrate increased FDG uptake [145].
Like molecular therapy, an ‘Immune Flare’ pattern is usually seen early after treatment introduction (6–8 weeks) [148] and should be considered when not associated with progression at prior sites of disease, especially when most disease sites are on regression [149].
Wong et al. recommend a follow-up study before defining progression when patterns of inflammatory response are present, being symmetric hilar and mediastinal nodal uptake similar to sarcoidosis (usually seen in patients with pulmonary metastasis), reactive nodal uptake in the drainage basin of metastases (e.g., porta hepatitis nodal activity in the setting of hepatic metastases) and diffuse splenic uptake [145].
Lesions that have very similar uptake before and after treatment are more suspicious than those that vary during treatment; in fact, the ‘pseudoprogression’ concept has been introduced to define an atypical response that consists of an increment in the lesion's size before a subsequent reduction. Pseudoprogression has to be considered to prevent premature cessation of treatment [150].
While early reports were promising, not all subsequent studies support the use [151] of FDG-PET/CT and there is no doubt that additional research will be required. Nonetheless, FDG-PET/CT can play an important role for the evaluation of these innovative treatments, providing important information regarding early discrimination between responders and refractory disease and also enabling detection of immune-related toxicities prior to clinical manifestations [135,145,152,153].
PET/MRI
No studies have reported a diagnostic contribution of PET/MRI for staging. Some studies compared whole-body PET/MRI with PET/CT for melanoma staging [154,155], showing a good diagnostic performance (sensitivity 82–84% and specificity 87–97%), especially when MRI with diffusion weighted imaging was used versus 73–80% and 93% for PET/CT, respectively.
The use of diffusion-weighted imaging (DWI) techniques for the detection of extracranial lesions is a promising tool and there might be a role of PET/MRI in research but data are still limited [156–158].
Conclusion
In recent decades, nuclear medicine has increased its implication in the management of melanoma patients and its use has been recommended by various European Associations [159,160]. For lymph node status evaluation of early-stage melanoma patients, SNB is now routinely employed and lymphoscintigraphy combined with SPECT/CT is the preferred roadmap for surgeons. Also in the operating room, the role of nuclear medicine technology to identify the SN is quickly expanding through the incorporation of hybrid tracers and navigation techniques. FDG-PET/CT is increasingly being used in patients with more advanced disease for staging and therapy response assessment following the introduction of new targeted therapeutic agents. In an interventional approach FDG-PET/CT may also be useful as roadmap to guide metastasectomy in melanoma patients with slow growing oligometastases. A continuous refinement of the evolution of nuclear medicine techniques has been witnessed through the interaction with other medical disciplines and the ongoing technological advances. Thanks to its capability to combine these advances with new insights about lymphatic drainage and dissemination routes of metastases, nuclear medicine will continue to play an essential role in the diagnosis and management of melanoma patients.
Future perspective
Following a period of increasing implication in many aspects regarding management of melanoma patients during the last two decades, new advances of nuclear medicine have reinforced this contribution. In addition to the standardization of lymphoscintigraphy as a roadmap for SN identification in the operating room, the validation of SPECT/CT has enabled not only the identification of SN in complex anatomical sites but has also been associated with a better disease-free survival and significant cost-saving implications in melanoma patients with indication for SNB. New possibilities such as intraoperative imaging with hybrid tracers are gaining acceptance and in the coming years, they will contribute to consolidate the paradigm ‘see, open and recognize’ in the search of SN on the basis of the synergistic use of both established preoperative SPECT/CT and novel intraoperative navigation tools.
Also FDG-PET/CT has increased its relevance in staging and therapy response assessment of advanced-stage melanoma patients. PET/CT technology can offer an unparalleled opportunity to study the continuous evolvement of the metastatic molecular mechanisms [161]. Even though further improvement is certainly still needed, the above considerations highlight the urge to bring together all available techniques with the aim to produce a positive impact on management of melanoma patients while assuring high-quality assessment of melanoma patients at reasonable costs. Finally, PET/CT may also play a role in radioguided surgery of FDG-avid lesions in patients with oligometastatic melanoma.
Footnotes
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
Open access
This work is licensed under the Attribution-Non Commercial-No Derivatives 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
References
Papers of special note have been highlighted as: • of interest; •• of considerable interest
- 1.Perissinotti A, Vidal-Sicart S, Nieweg O, Olmos RV. Melanoma and nuclear medicine. Melanoma Manag. 2014;1(1):57–74. doi: 10.2217/mmt.14.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Balch CM, Soong SJ, Gershenwald JE, et al. Prognostic factors analysis of 17,600 melanoma patients: validation of the American Joint Committee on Cancer melanoma staging system. J. Clin. Oncol. 2001;19(16):3622–3634. doi: 10.1200/JCO.2001.19.16.3622. [DOI] [PubMed] [Google Scholar]
- 3.Morton DL, Cochran AJ, Thompson JF, et al. Sentinel node biopsy for early-stage melanoma: accuracy and morbidity in MSLT-I, an international multicenter trial. Ann. Surg. 2005;242(3):302–311. doi: 10.1097/01.sla.0000181092.50141.fa. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Prospective multicenter study that confirmed the value of early nodal evaluation and treatment of melanoma based on sentinel node biopsy (SNB).
- 4.Morton DL, Thompson JF, Cochran AJ, et al. Sentinel-node biopsy or nodal observation in melanoma. N. Engl. J. Med. 2006;355(13):1307–1317. doi: 10.1056/NEJMoa060992. [DOI] [PubMed] [Google Scholar]
- 5.Morton DL, Thompson JF, Cochran AJ, et al. Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N. Engl. J. Med. 2014;370(7):599–609. doi: 10.1056/NEJMoa1310460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Testori A, De Salvo GL, Montesco MC, et al. Clinical considerations on sentinel node biopsy in melanoma from an Italian multicentric study on 1,313 patients (SOLISM-IMI) Ann. Surg. Oncol. 2009;16(7):2018–2027. doi: 10.1245/s10434-008-0273-8. [DOI] [PubMed] [Google Scholar]
- 7.Balch CM, Gershenwald JE, Soong S-J, et al. Final version of 2009 AJCC melanoma staging and classification. J. Clin. Oncol. 2009;27(36):6199–6206. doi: 10.1200/JCO.2009.23.4799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mohr P, Eggermont AMM, Hauschild A, Buzaid A. Staging of cutaneous melanoma. Ann. Oncol. 2009;20(Suppl. 6):vi14–vi21. doi: 10.1093/annonc/mdp256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Reyes DK, Pienta KJ. The biology and treatment of oligometastatic cancer. Oncotarget. 2015;6(11):8491–8524. doi: 10.18632/oncotarget.3455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Reintgen D, Cruse CW, Wells K, et al. The orderly progression of melanoma nodal metastases. Ann. Surg. 1994;220(6):759–767. doi: 10.1097/00000658-199412000-00009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Surgery R. Complete lymph node dissection in melanoma: a systematic review and meta-analysis. Anticancer Res. 2017;37(12):6825–6829. doi: 10.21873/anticanres.12143. [DOI] [PubMed] [Google Scholar]
- 12.Nieweg OE, Tanis PJ, Kroon BB. The definition of a sentinel node. Ann. Surg. Oncol. 2001;8(6):538–541. doi: 10.1007/s10434-001-0538-y. [DOI] [PubMed] [Google Scholar]
- 13.Morton DL, Wen DR, Wong JH, et al. Technical details of intraoperative lymphatic mapping for early stage melanoma. Arch. Surg. 1992;127(4):392–399. doi: 10.1001/archsurg.1992.01420040034005. [DOI] [PubMed] [Google Scholar]
- 14.Giuliano AE, Kirgan DM, Guenther JM, Morton DL. Lymphatic mapping and sentinel lymphadenectomy for breast cancer. Ann. Surg. 1994;220(3):391–398. doi: 10.1097/00000658-199409000-00015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Broglie MA, Stoeckli SJ. Relevance of sentinel node procedures in head and neck squamous cell carcinoma. Q. J. Nucl. Med. Mol. Imaging. 2011;55(5):509–520. [PubMed] [Google Scholar]
- 16.de Hullu JA, Doting E, Piers DA, et al. Sentinel lymph node identification with technetium-99m-labeled nanocolloid in squamous cell cancer of the vulva. J. Nucl. Med. 1998;39(8):1381–1385. [PubMed] [Google Scholar]
- 17.Torné A, Pahisa J, Vidal-Sicart S, et al. Transvaginal ultrasound-guided myometrial injection of radiotracer (TUMIR): a new method for sentinel lymph node detection in endometrial cancer. Gynecol. Oncol. 2013;128(1):88–94. doi: 10.1016/j.ygyno.2012.10.008. [DOI] [PubMed] [Google Scholar]
- 18.Horenblas S, Jansen L, Meinhardt W, Hoefnagel CA, de Jong D, Nieweg OE. Detection of occult metastasis in squamous cell carcinoma of the penis using a dynamic sentinel node procedure. J. Urol. 2000;163(1):100–104. [PubMed] [Google Scholar]
- 19.Faries MB, Thompson JF, Cochran AJ, et al. Completion dissection or observation for sentinel-node metastasis in melanoma. N. Engl. J. Med. 2017;376(23):2211–2222. doi: 10.1056/NEJMoa1613210. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Prospective multicenter study showing that complete lymphadenectomy after SNB did not increase melanoma-specific survival compared with nodal observation with ultrasonography.
- 20.Leiter U, Stadler R, Mauch C, et al. Complete lymph node dissection versus no dissection in patients with sentinel lymph node biopsy positive melanoma (DeCOG-SLT): a multicentre, randomised, Phase 3 trial. Lancet Oncol. 2016;17(6):757–767. doi: 10.1016/S1470-2045(16)00141-8. [DOI] [PubMed] [Google Scholar]; •• Evaluation of complete lymphadenectomy versus observation with ultrasonography after SNB in line with Multicenter Selective Lymphadenectomy Trial-II.
- 21.Wong SL, Faries MB, Kennedy EB, et al. Sentinel lymph node biopsy and management of regional lymph nodes in melanoma: American Society of Clinical Oncology and Society of Surgical Oncology clinical practice guideline update. J. Clin. Oncol. 2018;36(4):399–413. doi: 10.1200/JCO.2017.75.7724. [DOI] [PubMed] [Google Scholar]
- 22.Valdés Olmos RA, Vidal-Sicart S, Manca G, et al. Advances in radioguided surgery in oncology. Q. J. Nucl. Med. Mol. Imaging. 2017;61(3):247–270. doi: 10.23736/S1824-4785.17.02995-8. [DOI] [PubMed] [Google Scholar]
- 23.Zaknun JJ, Giammarile F, Olmos RAV, Vidal-Sicart S, Mariani G. Changing paradigms in radioguided surgery and intraoperative imaging: the GOSTT concept. Eur. J. Nucl. Med. Mol. Imaging. 2012;39(1):1–3. doi: 10.1007/s00259-011-1951-5. [DOI] [PubMed] [Google Scholar]
- 24.Uren RF, Howman-Giles R, Thompson JF. Patterns of lymphatic drainage from the skin in patients with melanoma. J. Nucl. Med. 2003;44(4):570–582. [PubMed] [Google Scholar]
- 25.Statius Muller MG, Hennipman FA, van Leeuwen PAM, Pijpers R, Vuylsteke RJ, Meijer S. Unpredictability of lymphatic drainage patterns in melanoma patients. Eur. J. Nucl. Med. Mol. Imaging. 2002;29(2):255–261. doi: 10.1007/s00259-001-0670-8. [DOI] [PubMed] [Google Scholar]
- 26.Thompson JF, Uren RF. Lymphatic mapping in management of patients with primary cutaneous melanoma. Lancet Oncol. 2005;6(11):877–885. doi: 10.1016/S1470-2045(05)70423-X. [DOI] [PubMed] [Google Scholar]
- 27.Vidal M, Vidal-Sicart S, Torrents A, et al. Accuracy and reproducibility of lymphoscintigraphy for sentinel node detection in patients with cutaneous melanoma. J. Nucl. Med. 2012;53(8):1193–1199. doi: 10.2967/jnumed.112.104463. [DOI] [PubMed] [Google Scholar]
- 28.Ortín-Pérez J, Vidal-Sicart S, Doménech B, Rubí S, Lafuente S, Pons F. In-transit sentinel lymph nodes in malignant melanoma. What is their importance? Rev. Española Med. Nucl. 2008;27(6):424–429. [PubMed] [Google Scholar]
- 29.Vidal-Sicart S, Pons F, Fuertes S, et al. Is the identification of in-transit sentinel lymph nodes in malignant melanoma patients really necessary? Eur. J. Nucl. Med. Mol. Imaging. 2004;31(7):945–949. doi: 10.1007/s00259-004-1485-1. [DOI] [PubMed] [Google Scholar]
- 30.Uren RF, Howman-Giles R, Thompson JF, et al. Interval nodes: the forgotten sentinel nodes in patients with melanoma. Arch. Surg. 2000;135(10):1168–1172. doi: 10.1001/archsurg.135.10.1168. [DOI] [PubMed] [Google Scholar]
- 31.Rousseau DL, Ross MI, Johnson MM, et al. Revised American Joint Committee on Cancer staging criteria accurately predict sentinel lymph node positivity in clinically node-negative melanoma patients. Ann. Surg. Oncol. 2003;10(5):569–574. doi: 10.1245/aso.2003.09.016. [DOI] [PubMed] [Google Scholar]
- 32.Jones MS, Jones PC, Stern SL, et al. The impact of smoking on sentinel node metastasis of primary cutaneous melanoma. Ann. Surg. Oncol. 2017 doi: 10.1245/s10434-017-5775-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Estourgie SH, Nieweg OE, Valdés Olmos RA, Hoefnagel CA, Kroon BBR. Review and evaluation of sentinel node procedures in 250 melanoma patients with a median follow-up of 6 years. Ann. Surg. Oncol. 2003;10(6):681–688. doi: 10.1245/aso.2003.01.023. [DOI] [PubMed] [Google Scholar]
- 34.Kesmodel SB, Karakousis GC, Botbyl JD, et al. Mitotic rate as a predictor of sentinel lymph node positivity in patients with thin melanomas. Ann. Surg. Oncol. 2005;12(6):449–458. doi: 10.1245/ASO.2005.04.027. [DOI] [PubMed] [Google Scholar]
- 35.van Akkooi ACJ, Voit CA, Verhoef C, Eggermont AMM. New developments in sentinel node staging in melanoma: controversies and alternatives. Curr. Opin. Oncol. 2010;22(3):169–177. doi: 10.1097/CCO.0b013e328337aa78. [DOI] [PubMed] [Google Scholar]
- 36.Nieweg OE. False-negative sentinel node biopsy. Ann. Surg. Oncol. 2009;16(8):2089–2091. doi: 10.1245/s10434-009-0540-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bagaria S. Sentinel node biopsy in melanoma: technical considerations of the procedure as performed at the John Wayne Cancer Institute. J. Surg. Oncol. 2010;101(8):669–676. doi: 10.1002/jso.21581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Valsecchi ME, Silbermins D, de Rosa N, Wong SL, Lyman GH. Lymphatic mapping and sentinel lymph node biopsy in patients with melanoma: a meta-analysis. J. Clin. Oncol. 2011;29(11):1479–1487. doi: 10.1200/JCO.2010.33.1884. [DOI] [PubMed] [Google Scholar]
- 39.Jimenez RE, Panageas K, Busam KJ, Brady MS. Prognostic implications of multiple lymphatic basin drainage in patients with truncal melanoma. J. Clin. Oncol. 2005;23(3):518–524. doi: 10.1200/JCO.2005.00.075. [DOI] [PubMed] [Google Scholar]
- 40.van Akkooi ACJ, Bouwhuis MG, de Wilt JHW, Kliffen M, Schmitz PIM, Eggermont AMM. Multivariable analysis comparing outcome after sentinel node biopsy or therapeutic lymph node dissection in patients with melanoma. Br. J. Surg. 2007;94(10):1293–1299. doi: 10.1002/bjs.5814. [DOI] [PubMed] [Google Scholar]
- 41.Kretschmer L, Hilgers R, Möhrle M, et al. Patients with lymphatic metastasis of cutaneous malignant melanoma benefit from sentinel lymphonodectomy and early excision of their nodal disease. Eur. J. Cancer. 2004;40(2):212–218. doi: 10.1016/j.ejca.2003.07.003. [DOI] [PubMed] [Google Scholar]
- 42.Cascinelli N, Morabito A, Santinami M, MacKie RM, Belli F. Immediate or delayed dissection of regional nodes in patients with melanoma of the trunk: a randomised trial. WHO Melanoma Programme. Lancet. 1998;351(9105):793–796. doi: 10.1016/s0140-6736(97)08260-3. [DOI] [PubMed] [Google Scholar]
- 43.Ross MI, Gershenwald JE. How should we view the results of the Multicenter Selective Lymphadenectomy Trial-1 (MSLT-1)? Ann. Surg. Oncol. 2008;15(3):670–673. doi: 10.1245/s10434-008-9828-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cochran AJ, Balda BR, Starz H, et al. The Augsburg Consensus. Techniques of lymphatic mapping, sentinel lymphadenectomy, and completion lymphadenectomy in cutaneous malignancies. Cancer. 2000;89(2):236–241. [PubMed] [Google Scholar]
- 45.Chakera AH, Hesse B, Burak Z, et al. EANM-EORTC general recommendations for sentinel node diagnostics in melanoma. Eur. J. Nucl. Med. Mol. Imaging. 2009;36(10):1713–1742. doi: 10.1007/s00259-009-1228-4. [DOI] [PubMed] [Google Scholar]
- 46.Crippa F, Leutner M, Belli F, et al. Which kinds of lymph node metastases can FDG PET detect? A clinical study in melanoma. J. Nucl. Med. 2000;41(9):1491–1494. [PubMed] [Google Scholar]
- 47.Wong SL, Balch CM, Hurley P, et al. Sentinel lymph node biopsy for melanoma: American Society of Clinical Oncology and Society of Surgical Oncology Joint Clinical Practice Guideline. J. Clin. Oncol. 2012;30(23):2912–2918. doi: 10.1200/JCO.2011.40.3519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Australian Cancer Network Melanoma Guidelines Revision Working Party. Cancer Council Australia/Australian Cancer Network; Wellington, New Zealand: 2008. Clinical practice guidelines for the management of melanoma in Australia and New Zealand.www.cancer.org.au/content/pdf/HealthProfessionals/ClinicalGuidelines/ClinicalPracticeGuidelines-ManagementofMelanoma.pdf [Google Scholar]
- 49.Garbe C, Peris K, Hauschild A, et al. Diagnosis and treatment of melanoma: European consensus-based interdisciplinary guideline. Eur. J. Cancer. 2010;46(2):270–283. doi: 10.1016/j.ejca.2009.10.032. [DOI] [PubMed] [Google Scholar]
- 50.Bluemel C, Herrmann K, Giammarile F, et al. EANM practice guidelines for lymphoscintigraphy and sentinel lymph node biopsy in melanoma. Eur. J. Nucl. Med. Mol. Imaging. 2015;42(11):1750–1766. doi: 10.1007/s00259-015-3135-1. [DOI] [PubMed] [Google Scholar]; • Very complete revision and current guideline for sentinel node in Europe.
- 51.Ipenburg NA, Nieweg OE, Uren RF, Thompson JF. Outcome of melanoma patients who did not proceed to sentinel node biopsy after preoperative lymphoscintigraphy. Ann. Surg. Oncol. 2017;24(1):117–126. doi: 10.1245/s10434-016-5458-y. [DOI] [PubMed] [Google Scholar]
- 52.Geimer T, Sattler EC, Flaig MJ, et al. The impact of sentinel node dissection on disease-free and overall tumour-specific survival in melanoma patients: a single centre group-matched analysis of 1192 patients. J. Eur. Acad. Dermatol. Venereol. 2017;31(4):629–635. doi: 10.1111/jdv.13939. [DOI] [PubMed] [Google Scholar]
- 53.Coit D. The enigma of regional lymph nodes in melanoma. N. Engl. J. Med. 2017;376(23):2280–2281. doi: 10.1056/NEJMe1704290. [DOI] [PubMed] [Google Scholar]
- 54.Ortega Candil A, Rodríguez Rey C, Carreras Delgado JL. Malignant melanoma. ISRN Dermatol. 2012 doi: 10.5402/2012/308279. Epub ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wright BE, Scheri RP, Ye X, et al. Importance of sentinel lymph node biopsy in patients with thin melanoma. Arch. Surg. 2008;143(9):892–899. doi: 10.1001/archsurg.143.9.892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Mays MP, Martin RCG, Burton A, et al. Should all patients with melanoma between 1 and 2 mm Breslow thickness undergo sentinel lymph node biopsy? Cancer. 2010;116(6):1535–1544. doi: 10.1002/cncr.24895. [DOI] [PubMed] [Google Scholar]
- 57.Gajdos C, Griffith KA, Wong SL, et al. Is there a benefit to sentinel lymph node biopsy in patients with T4 melanoma? Cancer. 2009;115(24):5752–5760. doi: 10.1002/cncr.24660. [DOI] [PubMed] [Google Scholar]
- 58.Rietbergen DD, van den Berg NS, van Leeuwen FW, Valdés Olmos RA. Hybrid techniques for intraoperative sentinel lymph node imaging: early experiences and future prospects. Imaging Med. 2013;5(2):147–159. [Google Scholar]
- 59.Nieweg OE, Jansen L, Kroon BB. Technique of lymphatic mapping and sentinel node biopsy for melanoma. Eur. J. Surg. Oncol. 1998;24(6):520–524. doi: 10.1016/s0748-7983(98)93428-x. [DOI] [PubMed] [Google Scholar]
- 60.van der Ploeg IMC, Madu MF, van der Hage JA, et al. Blue dye can be safely omitted in most sentinel node procedures for melanoma. Melanoma Res. 2016;26(5):464–468. doi: 10.1097/CMR.0000000000000271. [DOI] [PubMed] [Google Scholar]
- 61.Moncayo VM, Alazraki AL, Alazraki NP, Aarsvold JN. Sentinel lymph node biopsy procedures. Semin. Nucl. Med. 2017;47(6):595–617. doi: 10.1053/j.semnuclmed.2017.06.004. [DOI] [PubMed] [Google Scholar]
- 62.Sondak VK, King DW, Zager JS, et al. Combined analysis of Phase III trials evaluating [99mTc]tilmanocept and vital blue dye for identification of sentinel lymph nodes in clinically node-negative cutaneous melanoma. Ann. Surg. Oncol. 2013;20(2):680–688. doi: 10.1245/s10434-012-2612-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Schaafsma BE, Mieog JSD, Hutteman M, et al. The clinical use of indocyanine green as a near-infrared fluorescent contrast agent for image-guided oncologic surgery. J. Surg. Oncol. 2011;104(3):323–332. doi: 10.1002/jso.21943. [DOI] [PMC free article] [PubMed] [Google Scholar]; • Introduces near-infrared imaging and its application in sentinel node mapping and tumor imaging.
- 64.Stoffels I, Dissemond J, Pöppel T, Schadendorf D, Klode J. Intraoperative fluorescence imaging for sentinel lymph node detection: prospective clinical trial to compare the usefulness of indocyanine green vs technetium Tc 99m for identification of sentinel lymph nodes. JAMA Surg. 2015;150(7):617–623. doi: 10.1001/jamasurg.2014.3502. [DOI] [PubMed] [Google Scholar]
- 65.Crane LMA, Themelis G, Pleijhuis RG, et al. Intraoperative multispectral fluorescence imaging for the detection of the sentinel lymph node in cervical cancer: a novel concept. Mol. Imaging Biol. 2011;13(5):1043–1049. doi: 10.1007/s11307-010-0425-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Rossi EC, Ivanova A, Boggess JF. Robotically assisted fluorescence-guided lymph node mapping with ICG for gynecologic malignancies: a feasibility study. Gynecol. Oncol. 2012;124(1):78–82. doi: 10.1016/j.ygyno.2011.09.025. [DOI] [PubMed] [Google Scholar]
- 67.Hirche C, Mohr Z, Kneif S, et al. Ultrastaging of colon cancer by sentinel node biopsy using fluorescence navigation with indocyanine green. Int. J. Colorectal Dis. 2012;27(3):319–324. doi: 10.1007/s00384-011-1306-5. [DOI] [PubMed] [Google Scholar]
- 68.Gioux S, Choi H, Frangioni J. Image-guided surgery using invisible near-infrared light: fundamentals of clinical translation. Mol. Imaging. 2010;9(5):237–255. [PMC free article] [PubMed] [Google Scholar]
- 69.Mieog JSD, Troyan SL, Hutteman M, et al. Toward optimization of imaging system and lymphatic tracer for near-infrared fluorescent sentinel lymph node mapping in breast cancer. Ann. Surg. Oncol. 2011;18(9):2483–2491. doi: 10.1245/s10434-011-1566-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.van den Berg NS, Miwa M, KleinJan GH, et al. (Near-Infrared) fluorescence-guided surgery under ambient light conditions: a next step to embedment of the technology in clinical routine. Ann. Surg. Oncol. 2016;23:2586–2595. doi: 10.1245/s10434-016-5186-3. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Prospective study evaluating the use of hybrid tracer in a large population of melanoma patients.
- 71.van der Vorst JR, Schaafsma BE, Verbeek FPR, et al. Dose optimization for near-infrared fluorescence sentinel lymph node mapping in patients with melanoma. Br. J. Dermatol. 2013;168(1):93–98. doi: 10.1111/bjd.12059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Stoffels I, von der Stück H, Boy C, et al. Indocyanine green fluorescence-guided sentinel lymph node biopsy in dermato-oncology. J. Dtsch. Dermatol. Ges. 2012;10(1):51–57. doi: 10.1111/j.1610-0387.2011.07843.x. [DOI] [PubMed] [Google Scholar]
- 73.Fujiwara M, Mizukami T, Suzuki A, Fukamizu H. Sentinel lymph node detection in skin cancer patients using real-time fluorescence navigation with indocyanine green: preliminary experience. J. Plast. Reconstr. Aesthet. Surg. 2009;62(10):e373–e378. doi: 10.1016/j.bjps.2007.12.074. [DOI] [PubMed] [Google Scholar]
- 74.Namikawa K, Tsutsumida A, Tanaka R, Kato J, Yamazaki N. Limitation of indocyanine green fluorescence in identifying sentinel lymph node prior to skin incision in cutaneous melanoma. Int. J. Clin. Oncol. 2014;19(1):198–203. doi: 10.1007/s10147-013-0524-y. [DOI] [PubMed] [Google Scholar]
- 75.van den Berg NS, Brouwer OR, Schaafsma BE, et al. Multimodal surgical guidance during sentinel node biopsy for melanoma: combined gamma tracing and fluorescence imaging of the sentinel node through use of the hybrid tracer indocyanine green–99m Tc-nanocolloid. Radiology. 2015;275(2):521–529. doi: 10.1148/radiol.14140322. [DOI] [PubMed] [Google Scholar]
- 76.Stoffels I, Leyh J, Pöppel T, Schadendorf D, Klode J. Evaluation of a radioactive and fluorescent hybrid tracer for sentinel lymph node biopsy in head and neck malignancies: prospective randomized clinical trial to compare ICG-99mTc-nanocolloid hybrid tracer versus 99mTc-nanocolloid. Eur. J. Nucl. Med. Mol. Imaging. 2015;42(11):1631–1638. doi: 10.1007/s00259-015-3093-7. [DOI] [PubMed] [Google Scholar]
- 77.Even-Sapir E, Lerman H, Lievshitz G, et al. Lymphoscintigraphy for sentinel node mapping using a hybrid SPECT/CT system. J. Nucl. Med. 2003;44(9):1413–1420. [PubMed] [Google Scholar]
- 78.van der Ploeg IMC, Nieweg OE, Kroon BBR, et al. The yield of SPECT/CT for anatomical lymphatic mapping in patients with breast cancer. Eur. J. Nucl. Med. Mol. Imaging. 2009;36(6):903–909. doi: 10.1007/s00259-008-1050-4. [DOI] [PubMed] [Google Scholar]
- 79.Khafif A, Schneebaum S, Fliss DM. Lymphoscintigraphy for sentinel node mapping using a hybrid single photon emission CT (SPECT)/CT system in oral cavity squamous cell carcinoma. Head Neck. 2006;28(10):874–879. doi: 10.1002/hed.20434. [DOI] [PubMed] [Google Scholar]
- 80.Vidal-Sicart S, Brouwer OR, Valdés-Olmos R. Evaluation of the sentinel lymph node combining SPECT/CT with the planar image and its importance for the surgical act. Rev. Esp. Med. Nucl. 2011;30(5):331–337. doi: 10.1016/j.remn.2011.05.009. [DOI] [PubMed] [Google Scholar]
- 81.Vermeeren L, Meinhardt W, Bex A, et al. Paraaortic sentinel lymph nodes: toward optimal detection and intraoperative localization using SPECT/CT and intraoperative real-time imaging. J. Nucl. Med. 2010;51(3):376–382. doi: 10.2967/jnumed.109.071779. [DOI] [PubMed] [Google Scholar]
- 82.Perissinotti A, Paredes P, Vidal-Sicart S, et al. Use of SPECT/CT for improved sentinel lymph node localization in endometrial cancer. Gynecol. Oncol. 2013;129(1):42–48. doi: 10.1016/j.ygyno.2013.01.022. [DOI] [PubMed] [Google Scholar]
- 83.Martínez A, Zerdoud S, Mery E, Bouissou E, Ferron G, Querleu D. Hybrid imaging by SPECT/CT for sentinel lymph node detection in patients with cancer of the uterine cervix. Gynecol. Oncol. 2010;119(3):431–435. doi: 10.1016/j.ygyno.2010.08.001. [DOI] [PubMed] [Google Scholar]
- 84.Jimenez-Heffernan A, Ellmann A, Sado H, et al. Results of a prospective multicenter international atomic energy agency sentinel node trial on the value of SPECT/CT over planar imaging in various malignancies. J. Nucl. Med. 2015;56(9):1338–1344. doi: 10.2967/jnumed.114.153643. [DOI] [PubMed] [Google Scholar]; •• International multicenter study that provides evidence in favor of the use of single-photon emission computed tomography images with integrated computed tomography in various malignancies.
- 85.Stoffels I, Müller M, Geisel MH, et al. Cost-effectiveness of preoperative SPECT/CT combined with lymphoscintigraphy vs. lymphoscintigraphy for sentinel lymph node excision in patients with cutaneous malignant melanoma. Eur. J. Nucl. Med. Mol. Imaging. 2014;41(9):1723–1731. doi: 10.1007/s00259-014-2771-1. [DOI] [PubMed] [Google Scholar]
- 86.Vermeeren L, Valdés Olmos RA, Meinhardt W, et al. Value of SPECT/CT for detection and anatomic localization of sentinel lymph nodes before laparoscopic sentinel node lymphadenectomy in prostate carcinoma. J. Nucl. Med. 2009;50(6):865–870. doi: 10.2967/jnumed.108.060673. [DOI] [PubMed] [Google Scholar]
- 87.Alvarez Paez AM, Brouwer OR, Veenstra HJ, et al. Decisive role of SPECT/CT in localization of unusual periscapular sentinel nodes in patients with posterior trunk melanoma. Melanoma Res. 2012;22(3):278–283. doi: 10.1097/CMR.0b013e32835312b1. [DOI] [PubMed] [Google Scholar]
- 88.Veenstra HJ, Vermeeren L, Olmos RAV, Nieweg OE. The additional value of lymphatic mapping with routine SPECT/CT in unselected patients with clinically localized melanoma. Ann. Surg. Oncol. 2012;19(3):1018–1023. doi: 10.1245/s10434-011-2031-6. [DOI] [PubMed] [Google Scholar]
- 89.Stoffels I, Boy C, Pöppel T, Kuhn J. Association between sentinel lymph node excision with or without preoperative SPECT/CT and metastatic node detection and disease-free survival in melanoma. JAMA. 2012;308(10):1007–1014. doi: 10.1001/2012.jama.11030. [DOI] [PubMed] [Google Scholar]; • High-ranking publication that compares the use of conventional lymphoscintigraphy with single-photon emission computed tomography images with integrated computed tomography in melanoma patients analyzing the incidence of metastatic nodes and disease-free survival.
- 90.Uren RF. SPECT/CT Lymphoscintigraphy to locate the sentinel lymph node in patients with melanoma. Ann. Surg. Oncol. 2009;16(6):1459–1460. doi: 10.1245/s10434-009-0463-z. [DOI] [PubMed] [Google Scholar]
- 91.McMasters KM, Reintgen DS, Ross MI, et al. Sentinel lymph node biopsy for melanoma: how many radioactive nodes should be removed? Ann. Surg. Oncol. 2001;8(3):192–197. doi: 10.1007/s10434-001-0192-4. [DOI] [PubMed] [Google Scholar]
- 92.Hellingman D, de Wit-van der Veen LJ, Klop WMC, Olmos RAV. Detecting near-the-injection-site sentinel nodes in head and neck melanomas with a high-resolution portable gamma camera. Clin. Nucl. Med. 2015;40(1):e11–e16. doi: 10.1097/RLU.0000000000000370. [DOI] [PubMed] [Google Scholar]
- 93.Vidal-Sicart S, Paredes P, Zanón G, et al. Added value of intraoperative real-time imaging in searches for difficult-to-locate sentinel nodes. J. Nucl. Med. 2010;51(8):1219–1225. doi: 10.2967/jnumed.110.074880. [DOI] [PubMed] [Google Scholar]
- 94.Stoffels I, Poeppel T, Boy C, et al. Radio-guided surgery: advantages of a new portable γ-camera (Sentinella) for intraoperative real time imaging and detection of sentinel lymph nodes in cutaneous malignancies. J. Eur. Acad. Dermatol. Venereol. 2012;26(3):308–313. doi: 10.1111/j.1468-3083.2011.04057.x. [DOI] [PubMed] [Google Scholar]
- 95.Vidal-Sicart S, Rioja ME, Paredes P, Keshtgar MR, Valdés Olmos RA. Contribution of perioperative imaging to radioguided surgery. Q. J. Nucl. Med. Mol. Imaging. 2014;58(2):140–160. [PubMed] [Google Scholar]
- 96.Hellingman D, Vidal-Sicart S, de Wit-van der Veen LJ, Paredes P, Valdés Olmos RA. A new portable hybrid camera for fused optical and scintigraphic imaging: first clinical experiences. Clin. Nucl. Med. 2016;41(1):e39–e43. doi: 10.1097/RLU.0000000000000874. [DOI] [PubMed] [Google Scholar]
- 97.Haneishi H, Onishi Y, Shimura H, Hayashi H. Simultaneous acquisition and image synthesis of gamma cameras and optical cameras for sentinel lymph node identification during radioguided surgery. IEEE Trans. Nucl. Sci. 2007;54(5):1703–1709. [Google Scholar]
- 98.Sulzbacher L, Klinger M, Scheurecker C, et al. Clinical usefulness of a novel freehand 3D imaging device for radio-guided intraoperative sentinel lymph node detection in malignant melanoma. Clin. Nucl. Med. 2015;40(9):e436–e440. doi: 10.1097/RLU.0000000000000882. [DOI] [PubMed] [Google Scholar]
- 99.Wendler T, Hartl A, Lasser T, et al. Towards intra-operative 3D nuclear imaging: reconstruction of 3D radioactive distributions using tracked gamma probes. Med. Image Comput. Comput. Assist. Interv. 2007;10(Pt 2):909–917. doi: 10.1007/978-3-540-75759-7_110. [DOI] [PubMed] [Google Scholar]
- 100.Brouwer OR, Buckle T, Bunschoten A, et al. Image navigation as a means to expand the boundaries of fluorescence-guided surgery. Phys. Med. Biol. 2012;57(10):3123–3136. doi: 10.1088/0031-9155/57/10/3123. [DOI] [PubMed] [Google Scholar]
- 101.Engelen T, Winkel BM, Rietbergen DD, et al. The next evolution in radioguided surgery: breast cancer related sentinel node localization using a freehandSPECT-mobile gamma camera combination. Am. J. Nucl. Med. Mol. Imaging. 2015;5(3):233–245. [PMC free article] [PubMed] [Google Scholar]
- 102.Chin PTK, Buckle T, Aguirre de Miguel A, Meskers SCJ, Janssen RAJ, van Leeuwen FWB. Dual-emissive quantum dots for multispectral intraoperative fluorescence imaging. Biomaterials. 2010;31(26):6823–6832. doi: 10.1016/j.biomaterials.2010.05.030. [DOI] [PubMed] [Google Scholar]
- 103.Brouwer OR, Buckle T, Vermeeren L, et al. Comparing the hybrid fluorescent-radioactive tracer indocyanine green-99mTc-nanocolloid with 99mTc-nanocolloid for sentinel node identification: a validation study using lymphoscintigraphy and SPECT/CT. J. Nucl. Med. 2012;53(7):1034–1040. doi: 10.2967/jnumed.112.103127. [DOI] [PubMed] [Google Scholar]; • Validates the hybrid tracer use of indocyanine green-99mTc-nanocolloid for sentinel node identification by comparison study with the conventional 99mTc-nanocolloid tracer.
- 104.van der Poel HG, Buckle T, Brouwer OR, Valdés Olmos RA, van Leeuwen FWB. Intraoperative laparoscopic fluorescence guidance to the sentinel lymph node in prostate cancer patients: clinical proof of concept of an integrated functional imaging approach using a multimodal tracer. Eur. Urol. 2011;60(4):826–833. doi: 10.1016/j.eururo.2011.03.024. [DOI] [PubMed] [Google Scholar]
- 105.Frontado LM, Brouwer OR, van den Berg NS, et al. Added value of the hybrid tracer indocyanine green-99mTc-nanocolloid for sentinel node biopsy in a series of patients with different lymphatic drainage patterns. Rev. Esp. Med. Nucl. Imagen. Mol. 2013;32(4):227–233. doi: 10.1016/j.remn.2013.02.004. [DOI] [PubMed] [Google Scholar]
- 106.Brouwer OR, Klop WMC, Buckle T, et al. Feasibility of sentinel node biopsy in head and neck melanoma using a hybrid radioactive and fluorescent tracer. Ann. Surg. Oncol. 2012;19(6):1988–1994. doi: 10.1245/s10434-011-2180-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.van den Berg NS, Simon H, Kleinjan GH, et al. First-in-human evaluation of a hybrid modality that allows combined radio- and (near-infrared) fluorescence tracing during surgery. Eur. J. Nucl. Med. Mol. Imaging. 2015;42(11):1639–1647. doi: 10.1007/s00259-015-3109-3. [DOI] [PubMed] [Google Scholar]
- 108.Poumellec M-A, Dejode M, Figl A, et al. Détection du ganglion sentinelle dans le cancer du sein par sonde opto-nucléaire après injection de vert indocyanine et de technétium 99m. Gynécologie Obs. Fertil. 2016;44(4):207–210. doi: 10.1016/j.gyobfe.2016.02.012. [DOI] [PubMed] [Google Scholar]
- 109.KleinJan GH, Hellingman D, van den Berg NS, et al. Hybrid surgical guidance: does hardware integration of γ- and fluorescence imaging modalities make sense? J. Nucl. Med. 2017;58(4):646–650. doi: 10.2967/jnumed.116.177154. [DOI] [PubMed] [Google Scholar]
- 110.Jiménez-Requena F, Delgado-Bolton RC, Fernández-Pérez C, et al. Meta-analysis of the performance of (18)F-FDG PET in cutaneous melanoma. Eur. J. Nucl. Med. Mol. Imaging. 2010;37(2):284–300. doi: 10.1007/s00259-010-1476-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Belhocine T, Scott A. Role of nuclear medicine in the management of cutaneous malignant melanoma. J. Nucl. Med. 2006;47(6):957–967. [PubMed] [Google Scholar]
- 112.Yancovitz M, Finelt N, Warycha MA, et al. Role of radiologic imaging at the time of initial diagnosis of stage T1b–T3b melanoma. Cancer. 2007;110(5):1107–1114. doi: 10.1002/cncr.22868. [DOI] [PubMed] [Google Scholar]
- 113.Clark PB, Soo V, Kraas J, Shen P, Levine EA. Futility of fluorodeoxyglucose F 18 positron emission tomography in initial evaluation of patients with T2 to T4 melanoma. Arch. Surg. 2006;141(3):284–288. doi: 10.1001/archsurg.141.3.284. [DOI] [PubMed] [Google Scholar]
- 114.El-Maraghi RH, Kielar AZ. PET vs sentinel lymph node biopsy for staging melanoma: a patient intervention, comparison, outcome analysis. J. Am. Coll. Radiol. 2008;5(8):924–931. doi: 10.1016/j.jacr.2008.02.022. [DOI] [PubMed] [Google Scholar]
- 115.Petersen H, Holdgaard PC, Madsen PH, et al. FDG PET/CT in cancer: comparison of actual use with literature-based recommendations. Eur. J. Nucl. Med. Mol. Imaging. 2016;43(4):695–706. doi: 10.1007/s00259-015-3217-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Krug B, Crott R, Lonneux M, Baurain J-F, Pirson A-S, Vander Borght T. Role of PET in the initial staging of cutaneous malignant melanoma: systematic review. Radiology. 2008;249(3):836–844. doi: 10.1148/radiol.2493080240. [DOI] [PubMed] [Google Scholar]
- 117.Ishimori T, Patel P, Wahl R. Detection of unexpected additional primary malignancies with PET/CT. J. Nucl. Med. 2005;46(5):752–757. [PubMed] [Google Scholar]
- 118.Niederkohr RD, Rosenberg J, Shabo G, Quon A. Clinical value of including the head and lower extremities in 18F-FDG PET/CT imaging for patients with malignant melanoma. Nucl. Med. Commun. 2007;28(9):688–695. doi: 10.1097/MNM.0b013e32827420cc. [DOI] [PubMed] [Google Scholar]
- 119.Xing Y, Bronstein Y, Ross MI, et al. Contemporary diagnostic imaging modalities for the staging and surveillance of melanoma patients: a meta-analysis. J. Natl. Cancer Inst. 2011;103(2):129–142. doi: 10.1093/jnci/djq455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Pfannenberg C, Aschoff P, Schanz S, et al. Prospective comparison of 18F-fluorodeoxyglucose positron emission tomography/computed tomography and whole-body magnetic resonance imaging in staging of advanced malignant melanoma. Eur. J. Cancer. 2007;43(3):557–564. doi: 10.1016/j.ejca.2006.11.014. [DOI] [PubMed] [Google Scholar]
- 121.Ollila DW, Gleisner AL, Hsueh EC. Rationale for complete metastasectomy in patients with stage IV metastatic melanoma. J. Surg. Oncol. 2011;104(4):420–424. doi: 10.1002/jso.21961. [DOI] [PubMed] [Google Scholar]
- 122.Howard JH, Thompson JF, Mozzillo N, et al. Metastasectomy for distant metastatic melanoma: analysis of data from the first multicenter selective lymphadenectomy trial (MSLT-I) Ann. Surg. Oncol. 2012;19(8):2547–2555. doi: 10.1245/s10434-012-2398-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ollila DW. Complete metastasectomy in patients with stage IV metastatic melanoma. Lancet Oncol. 2006;7(11):919–924. doi: 10.1016/S1470-2045(06)70938-X. [DOI] [PubMed] [Google Scholar]
- 124.Lasithiotakis K, Zoras O. Metastasectomy in cutaneous melanoma. Eur. J. Surg. Oncol. 2017;43(3):572–580. doi: 10.1016/j.ejso.2016.11.001. [DOI] [PubMed] [Google Scholar]
- 125.Povoski SP, Neff RL, Mojzisik CM, et al. A comprehensive overview of radioguided surgery using gamma detection probe technology. World J. Surg. Oncol. 2009;7:11. doi: 10.1186/1477-7819-7-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.KleinJan GH, Brouwer OR, Mathéron HM, et al. Hybrid radioguided occult lesion localization (hybrid ROLL) of 18F-FDG-avid lesions using the hybrid tracer indocyanine green-99mTc-nanocolloid. Rev. Esp. Med. Nucl. Imagen. Mol. 2016;35(5):292–297. doi: 10.1016/j.remn.2016.04.001. [DOI] [PubMed] [Google Scholar]
- 127.Horn J, Sjøstrand H, Lock-Andersen J, Loft A. PET scanning for malignant melanoma and positive sentinel node diagnostics. Ugeskr. Laeger. 2010;172(15):1126–1130. [PubMed] [Google Scholar]
- 128.Aukema TS, Valdés Olmos RA, Wouters MWJM, et al. Utility of preoperative 18F-FDG PET/CT and brain MRI in melanoma patients with palpable lymph node metastases. Ann. Surg. Oncol. 2010;17(10):2773–2778. doi: 10.1245/s10434-010-1088-y. [DOI] [PubMed] [Google Scholar]
- 129.Bastiaannet E, Wobbes T, Hoekstra OS, et al. Prospective comparison of [18F]fluorodeoxyglucose positron emission tomography and computed tomography in patients with melanoma with palpable lymph node metastases: diagnostic accuracy and impact on treatment. J. Clin. Oncol. 2009;27(28):4774–4780. doi: 10.1200/JCO.2008.20.1822. [DOI] [PubMed] [Google Scholar]
- 130.Koskivuo I, Kemppainen J, Giordano S, et al. Whole body PET/CT in the follow-up of asymptomatic patients with stage IIB-IIIB cutaneous melanoma. Acta Oncol. 2016;55(11):1355–1359. doi: 10.1080/0284186X.2016.1213879. [DOI] [PubMed] [Google Scholar]
- 131.Fuster D, Chiang S, Johnson G, Schuchter LM, Zhuang H, Alavi A. Is 18F-FDG PET more accurate than standard diagnostic procedures in the detection of suspected recurrent melanoma? J. Nucl. Med. 2004;45(8):1323–1327. [PubMed] [Google Scholar]
- 132.Aukema TS, Olmos RAV, Korse CM, et al. Utility of FDG PET/CT and brain MRI in melanoma patients with increased serum S-100B level during follow-up. Ann. Surg. Oncol. 2010;17(6):1657–1661. doi: 10.1245/s10434-010-0963-x. [DOI] [PubMed] [Google Scholar]
- 133.Wieder HA, Tekin G, Rosenbaum-Krumme S, et al. 18FDG-PET to assess recurrence and long term survival in patients with malignant melanoma. Nuklearmedizin. 2013;52(5):198–203. doi: 10.3413/Nukmed-0584-13-05. [DOI] [PubMed] [Google Scholar]
- 134.Reinhardt MJ, Joe AY, Jaeger U, et al. Diagnostic performance of whole body dual modality 18F-FDG PET/CT imaging for N- and M-staging of malignant melanoma: experience with 250 consecutive patients. J. Clin. Oncol. 2006;24(7):1178–1187. doi: 10.1200/JCO.2005.03.5634. [DOI] [PubMed] [Google Scholar]; • Evaluates [18F]-Fluorodeoxyglucose-PET/CT diagnostic performance for N- and M-staging, suggesting its use for whole-body tumor staging in melanoma patients, especially for detection or exclusion of distant metastases.
- 135.Engell-Noerregaard L, Hendel HW, Johannesen HH, Alslev L, Svane IM. FDG PET scans as evaluation of clinical response to dendritic cell vaccination in patients with malignant melanoma. Cancer Immunol. Immunother. 2013;62(1):17–25. doi: 10.1007/s00262-012-1306-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J. Nucl. Med. 2009;1(Suppl. 50)(5):122S–150S. doi: 10.2967/jnumed.108.057307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.van Ginkel RJ, Kole AC, Nieweg OE, et al. L-[1–11C]-tyrosine PET to evaluate response to hyperthermic isolated limb perfusion for locally advanced soft-tissue sarcoma and skin cancer. J. Nucl. Med. 1999;40(2):262–267. [PubMed] [Google Scholar]
- 138.McArthur GA, Chapman PB, Robert C, et al. Safety and efficacy of vemurafenib in BRAF(V600E) and BRAF(V600K) mutation-positive melanoma (BRIM-3): extended follow-up of a Phase 3, randomised, open-label study. Lancet. Oncol. 2014;15(3):323–332. doi: 10.1016/S1470-2045(14)70012-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Robert C, Karaszewska B, Schachter J, et al. Improved overall survival in melanoma with combined dabrafenib and trametinib. N. Engl. J. Med. 2015;372(1):30–39. doi: 10.1056/NEJMoa1412690. [DOI] [PubMed] [Google Scholar]
- 140.Ascierto PA, McArthur GA, Dréno B, et al. Cobimetinib combined with vemurafenib in advanced BRAF(V600)-mutant melanoma (coBRIM): updated efficacy results from a randomised, double-blind, Phase 3 trial. Lancet. Oncol. 2016;17(9):1248–1260. doi: 10.1016/S1470-2045(16)30122-X. [DOI] [PubMed] [Google Scholar]
- 141.Van Allen EM, Wagle N, Sucker A, et al. The genetic landscape of clinical resistance to RAF inhibition in metastatic melanoma. Cancer Discov. 2014;4(1):94–109. doi: 10.1158/2159-8290.CD-13-0617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Rizos H, Menzies AM, Pupo GM, et al. BRAF inhibitor resistance mechanisms in metastatic melanoma: spectrum and clinical impact. Clin. Cancer Res. 2014;20(7):1965–1977. doi: 10.1158/1078-0432.CCR-13-3122. [DOI] [PubMed] [Google Scholar]
- 143.McArthur GA, Puzanov I, Amaravadi R, et al. Marked, homogeneous, and early [18F]Fluorodeoxyglucose–positron emission tomography responses to vemurafenib in BRAF-mutant advanced melanoma. J. Clin. Oncol. 2012;30(14):1628–1634. doi: 10.1200/JCO.2011.39.1938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.McArthur GA, Callahan J, Ribas A, et al. Metabolic tumor burden for prediction of overall survival following combined BRAF/MEK inhibition in patients with advanced BRAF mutant melanoma. J. Clin. Oncol. 2014;32(Suppl. 5):9006. [Google Scholar]
- 145.Wong ANM, McArthur GA, Hofman MS, Hicks RJ. The advantages and challenges of using FDG PET/CT for response assessment in melanoma in the era of targeted agents and immunotherapy. Eur. J. Nucl. Med. Mol. Imaging. 2017;44(Suppl. 1):67–77. doi: 10.1007/s00259-017-3691-7. [DOI] [PubMed] [Google Scholar]; •• Very complete review of the role of [18F]-fluorodeoxyglucose-PET/CT for response assessment with new therapies.
- 146.Schadendorf D, Hodi FS, Robert C, et al. Pooled analysis of long-term survival data from Phase II and Phase III trials of ipilimumab in unresectable or metastatic melanoma. J. Clin. Oncol. 2015;33(17):1889–1894. doi: 10.1200/JCO.2014.56.2736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Larkin J, Chiarion-Sileni V, Gonzalez R, et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N. Engl. J. Med. 2015;373(1):23–34. doi: 10.1056/NEJMoa1504030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Weber JS, Dummer R, de Pril V, Lebbé C, Hodi FS. MDX010–20 Investigators. Patterns of onset and resolution of immune-related adverse events of special interest with ipilimumab: detailed safety analysis from a Phase 3 trial in patients with advanced melanoma. Cancer. 2013;119(9):1675–1682. doi: 10.1002/cncr.27969. [DOI] [PubMed] [Google Scholar]
- 149.Hofman MS, Hicks RJ. How we read oncologic FDG PET/CT. Cancer Imaging. 2016;16(1):35. doi: 10.1186/s40644-016-0091-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Hodi FS, Hwu WJ, Kefford R, et al. Evaluation of immune-related response criteria and RECIST v1.1 in patients with advanced melanoma treated with pembrolizumab. J. Clin. Oncol. 2016;34(13):1510–1517. doi: 10.1200/JCO.2015.64.0391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Kruijff S, Bastiaannet E, Speijers MJ, Kobold AC, Brouwers AH, Hoekstra HJ. The value of pre operative S-100B and SUV in clinically Stage III melanoma patients undergoing therapeutic lymph node dissection. Eur. J. Surg. Oncol. 2011;37(3):225–232. doi: 10.1016/j.ejso.2010.12.013. [DOI] [PubMed] [Google Scholar]
- 152.Beasley GM, Parsons C, Broadwater G, et al. A multicenter prospective evaluation of the clinical utility of F-18 FDG-PET/CT in patients with AJCC Stage IIIB or IIIC extremity melanoma. Ann. Surg. 2012;256(2):350–356. doi: 10.1097/SLA.0b013e318256d1f5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Kraeber-Bodéré F, Carlier T, Naegelen VM, et al. Differences in the biologic activity of 2 novel MEK inhibitors revealed by 18F-FDG PET: analysis of imaging data from two Phase I trials. J. Nucl. Med. 2012;53(12):1836–1846. doi: 10.2967/jnumed.112.109421. [DOI] [PubMed] [Google Scholar]
- 154.Jouvet JC, Thomas L, Thomson V, et al. Whole-body MRI with diffusion-weighted sequences compared with 18 FDG PET-CT, CT and superficial lymph node ultrasonography in the staging of advanced cutaneous melanoma: a prospective study. J. Eur. Acad. Dermatol. Venereol. 2014;28(2):176–185. doi: 10.1111/jdv.12078. [DOI] [PubMed] [Google Scholar]
- 155.Laurent V, Trausch G, Bruot O, Olivier P, Felblinger J, Régent D. Comparative study of two whole-body imaging techniques in the case of melanoma metastases: advantages of multi-contrast MRI examination including a diffusion-weighted sequence in comparison with PET-CT. Eur. J. Radiol. 2010;75(3):376–383. doi: 10.1016/j.ejrad.2009.04.059. [DOI] [PubMed] [Google Scholar]
- 156.Buchbender C, Heusner TA, Lauenstein TC, Bockisch A, Antoch G. Oncologic PET/MRI, Part 2: bone tumors, soft-tissue tumors, melanoma, and lymphoma. J. Nucl. Med. 2012;53(8):1244–1252. doi: 10.2967/jnumed.112.109306. [DOI] [PubMed] [Google Scholar]
- 157.Lee IS. Musculoskeletal applications of PET/MR. Semin. Musculoskelet. Radiol. 2014;18(2):203–216. doi: 10.1055/s-0034-1371021. [DOI] [PubMed] [Google Scholar]
- 158.Schwenzer NF, Pfannenberg AC. PET/CT, MR, and PET/MR in lymphoma and melanoma. Semin. Nucl. Med. 2015;45(4):322–331. doi: 10.1053/j.semnuclmed.2015.03.006. [DOI] [PubMed] [Google Scholar]
- 159.Dummer R, Hauschild A, Guggenheim M, Jost L, Pentheroudakis G. Melanoma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2010;21(Suppl. 5):v194–v197. doi: 10.1093/annonc/mdq188. [DOI] [PubMed] [Google Scholar]
- 160.Garbe C, Peris K, Hauschild A, et al. Diagnosis and treatment of melanoma. European consensus-based interdisciplinary guideline – update 2016. Eur. J. Cancer. 2016;63:201–217. doi: 10.1016/j.ejca.2016.05.005. [DOI] [PubMed] [Google Scholar]
- 161.Nathanson SD, Shah R, Rosso K. Sentinel lymph node metastases in cancer: causes, detection and their role in disease progression. Semin. Cell Dev. Biol. 2015;38:106–116. doi: 10.1016/j.semcdb.2014.10.002. [DOI] [PubMed] [Google Scholar]