Abstract
The aim of the current study was to investigate the effect of SB202190, a specific inhibitor of p38 MAPK signaling pathway, on the expression levels of IL-6 and NF-κB in flap ischemia-reperfusion injury. Healthy Sprague-Dawley rats were randomly divided into four groups of 12 each. For the ischemia-reperfusion group, the flap was constructed and then sutured after 8 h of ischemia. For the saline group, rats were intraperitoneally infused with saline at regular intervals after flap ischemia-reperfusion. For the inhibitor group, rats were intraperitoneally infused with SB202190 at regular intervals after flap ischemia-reperfusion. For the control group, the flap was constructed and then sutured immediately. The flap survival rate of each group was measured after 7 days. The concentration of IL-6 in serum was measured by ELISA kit. The mRNA and protein expression levels of IL-6 and NF-κB in the flap were measured using RT-PCR and western blot analysis, respectively. In the ischemia-reperfusion group and the saline group, the flap survival rates were much lower than that in the control group (P<0.05). By contrast, the mRNA and protein expression levels of IL-6 and NF-κB in the flap and the concentration of IL-6 in serum were much higher (P<0.05). In the inhibitor group, the flap survival rate was significantly higher than those in the ischemia-reperfusion and saline groups (P<0.05). By contrast, the concentration of IL-6 in serum and the mRNA and protein expression levels of NF-κB and IL-6 in the flap were significantly decreased (P<0.05). The results show that, SB202190 played a role in the protection of the flap by reducing the inflammatory response in flap ischemia-reperfusion injury.
Keywords: flap, ischemia-reperfusion injury, inflammatory response, IL-6, NF-κB, p38 MAPK signaling pathway inhibitor SB202190
Introduction
In orthopedic surgery, flap grafting is the most commonly used method for organ reconstruction or repair of tissue defect and deformity. However, in this process part or whole of the flap may undergo necrosis, which is a serious consequence due to ischemia-reperfusion injury (1).
Although the exact mechanism underlying ischemia-reperfusion injury has not been confirmed thus far, a variety of protective therapies against ischemia-reperfusion injury were reported, such as applications of low temperature, hydrogen, or antioxidants (2–5). In addition, it was found that activation of the p38 MAPK signaling pathway led to increased expression of NF-κB and promoted the secretion and release of inflammatory factors (such as IL-6, and TNF-α), which played an important role in the occurrence and progression of ischemia-reperfusion injury (6,7). SB202190 is a specific inhibitor of the P38 MAPK signaling pathway (8).
In this study, the mechanism underlying flap ischemia-reperfusion injury was explored by treatment of flap ischemia-reperfusion injury rat models with SB202190. The mRNA and protein expression levels of NF-κB and IL-6 in the flap, as well as the IL-6 concentration in serum were measured.
Materials and methods
Materials
The following materials were purchased from different sources: 48 healthy specific pathogen-free (SPF) male Sprague-Dawley rats weighing 250–300 g from Jiangsu University Animal Center; the enzyme-linked immunosorbent assay reader from BioTek Instruments, Inc. (Winooski, VT, USA); the optical microscopy and image acquisition system from Olympus Corporation, Tokyo, Japan; the rat IL-6 enzyme-linked immunosorbent assay (ELISA) kit and RT-PCR kit from R&D Systems, Inc. (Minneapolis, MN, USA); the internal reference β-actin, NF-κB p65, IL-6 primary and secondary antibodies from Abcam (Cambridge, UK); the cDNA synthesis kit from Tiangen Biotech Co. Ltd. (Beijing, China); the BCA kit from Beyotime Biotechnology (Shanghai, China); SB202190 from Cayman Chemical Company (Ann Arbor, MI, USA); and TRIzol and RIPA lysate from Invitrogen; Thermo Fisher Scientific, Inc., (Waltham, MA, USA).
Methods
Establishment of animal models
Rats were anesthetized via an intraperitoneal injection of ketamine (100 mg/kg), followed by fixation on the operating plate. After treatment of abdominal hair removal and alcohol disinfection, the superficial inferior epigastric arteriovenous flap was constructed according to literature (9). A rectangular flap with a size of 3×6 cm was created by dissection of the whole skin layer to the fascia on the abdominal wall with a scalpel. Attention was paid not to injure the superficial inferior epigastric artery pedicle, because the superficial inferior epigastric artery and vein were the only vessels for blood circulation in the flap.
Experimental grouping and treatments
Forty-eight rats were randomly divided into four groups of 12 each. In the control group, after a flap was created the incision was sutured in situ with the 3-0 silk thread. In the ischemia-reperfusion group, after a flap was created the superficial inferior epigastric artery and vein were clamped with microvascular clips for 8 h of ischemia. Following removal of the vessel clips and restoration of normal blood flow, the flap incision was sutured in situ. In the saline group, the rats received an intraperitoneal injection of normal saline (2 ml/kg) on the 2nd, 4th and 6th day after undergoing the same treatment as the ischemia-reperfusion group. In the inhibitor group, the rats received an intraperitoneal injection of SB202190 (2 µg/kg) on the 2nd, 4th and 6th day after undergoing the same treatment as the ischemia-reperfusion group. This study was approved by the Animal Ethics Committee of Jinan Municipal Hospital of Traditional Chinese Medicine (Jinan, China).
Observed indicators
Flap survival rate
On the 7th day after surgery, an image of the flap was taken with a digital camera for rats in all the groups. The flap survival rate was obtained by analysis of the image with the Image-Pro Plus. V6.0 software (Olympus) using the equation: survival rate = flap survival area / total area × 100%.
IL-6 concentration in serum
On the 7th day after surgery, 2 ml of blood was extracted from the superficial inferior epigastric vein for rats in all the groups. The serum was obtained after centrifugation at 3,000 × g for 10 min at 27°C, in which the IL-6 concentration was measured using enzyme-linked immunosorbent assay.
Measurement of NF-κB and IL-6 levels in flap tissue
On the 7th day after operation, the appropriate amount of flap tissue was harvested from rats in each group, and was treated with the appropriate amount of TRIzol reagent to extract total RNA. Then cDNA was synthesized from RNA using cDNA synthesis kit. Amplification of IL-6, NF-κB and internal reference β-actin was achieved by RT-PCR. The RT-PCR reaction conditions were: 95°C for 2 min; then 40 cycles of 95°C for 20 sec, 56°C for 20 sec, 72°C for 20 sec; 72°C for 5 min; 95°C for 15 sec; 60°C for 15 sec; 60–95°C for 20 min; and finally 95°C for 15 sec. The mRNA expression levels of NF-κB and IL-6 were calculated by using the amplification curve. The primer sequences of each gene are listed in Table I. In a separate experiment, an appropriate amount of flap tissue was harvested and homogenized in appropriate amount of RIPA lysis butter. Total protein was extracted from the homogenate, and the protein concentration was measured using a BCA kit. A sample containing 100 µg of proteins was applied to SDS-PAGE gel electrophoresis and transferred to the PVDF membrane using a wet system. The rabbit anti-rat NF-κB, IL-6, β-actin primary polyclonal antibodies (1:500; cat. nos. 14220-1-AP, 21865-1-AP, 20536-1-AP, respectively; ProteinTech Group, Inc., Wuhan, China) was added after the membrane was blocked with 5% BSA at room temperature for 2 h followed by incubation at 4°C overnight. The membrane was rinsed a few times, followed by the addition of the goat anti-rabbit secondary polyclonal antibody (1:2,000; cat. no. SA00001-2; ProteinTech Group, Inc.) and incubation at room temperature for 2 h. After three rinses, the ECL developer was applied to the membrane in a dark chamber. Images were obtained using fluorescence imaging techniques. The images were scanned, and the gray value as well as area of each band was analyzed using the Image Pro Plus 6.0 software.
Table I.
Primer sequences of each gene.
| Gene | Primer sequence |
|---|---|
| IL-6 | Up: 5′-GACTGATGTTGCTGACAGCCACTGC-3′ |
| Down: 5′-TAGCCACTGCTTCTGTGACTCTAACT-3′ | |
| NF-κB | Up: 5′-CCAGGCGGACATCTACAA-3′ |
| Down: 5′-CAAGGCCAAATGAAAGGA-3′ | |
| β-actin | Up: 5′-CCTAAGGCCAACCGTGAA-3′ |
| Down: 5′-CTGGAAGGTGGCAGTGAG-3′ |
Statistical analysis
The SPSS 19.0 software (Ndtimes, Beijing, China) was used in the statistical analysis. A comparison of multiple sets of quantitative samples was made using variance analysis. SNK-q test was used for comparison between groups. Correlation analysis was performed using Pearson's product-moment correlation coefficient. The difference was statistically significant when P<0.05.
Results
Comparison of flap survival rates
The flap survival rates in each group were obtained by image analysis (Fig. 1). The differences were statistically significant, as indicated by variance analysis (F = 8.214, P<0.05). The flap survival rates in the control group [(93.56+3.15)%] and the inhibitor group [(81.83+2.44)%] were much higher than those in the saline group [(49.61+2.55)%] and the ischemia-reperfusion group [(41.47+3.28)%] (P<0.05). There was also a significant difference between the control group and the inhibitor group (P<0.05).
Figure 1.
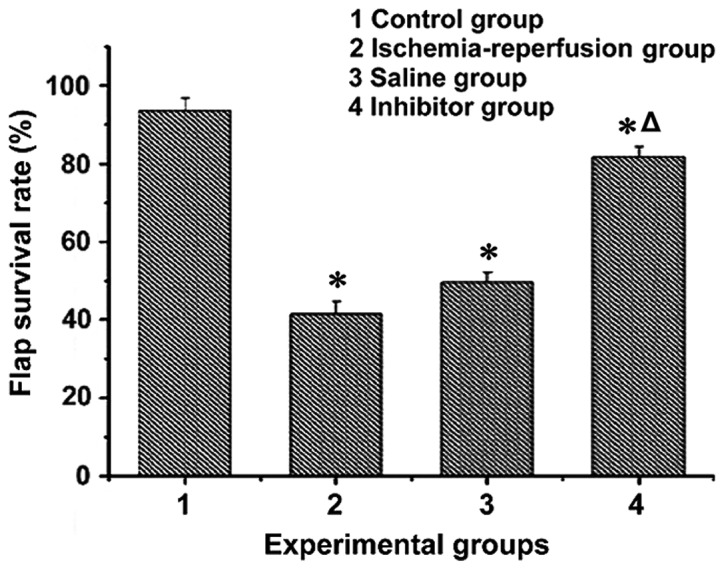
Flap survival rates of rats in the four groups (*P<0.05, compared with the control group; ΔP<0.05, compared with the ischemia-reperfusion and saline groups).
Serum IL-6 concentration
The serum concentrations of IL-6 in the ischemia-reperfusion group (91.18±3.52 ng/l) and the saline group (82.82±3.25 ng/l) were nearly double the concentration in the control group (47.45±2.85 ng/l) (P<0.05). The serum IL-6 concentration in the inhibitor group (59.66±2.27 ng/l) was much lower than those in the ischemia-reperfusion group and the normal saline group (P<0.05), but higher than that in the control group (P<0.05). The detailed data were listed in Table II.
Table II.
Serum IL-6 concentrations in the four groups (mean ± SD).
| Groups | ||||
|---|---|---|---|---|
| Variables | Control | Ischemia-reperfusion | Saline | Inhibitor |
| n | 12 | 12 | 12 | 12 |
| IL-6 concentration (ng/l) | 47.45±2.85 | 91.18±3.52a | 82.82±3.25a | 59.66±2.27a,b |
P<0.05, compared with the control group
P<0.05, compared with the ischemia-reperfusion group and the saline group.
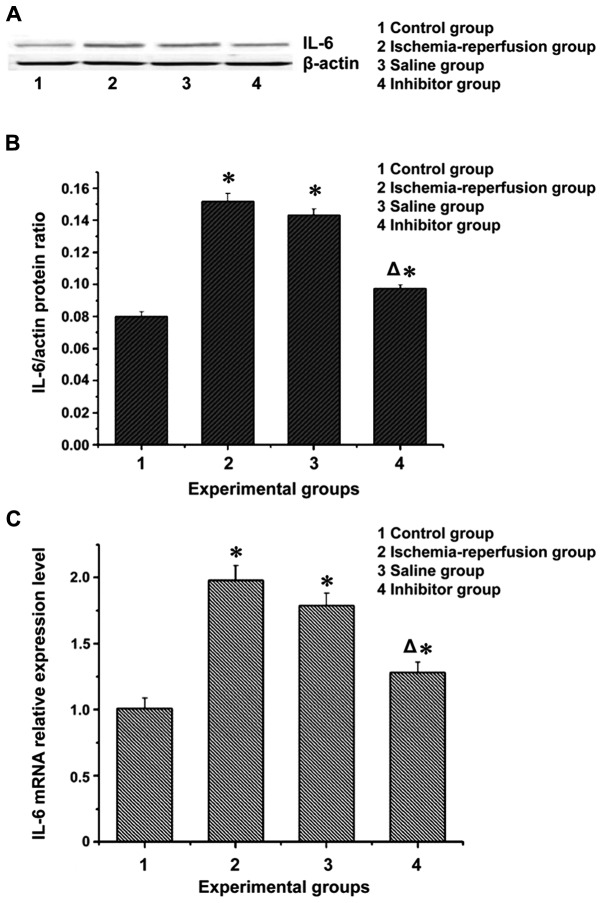
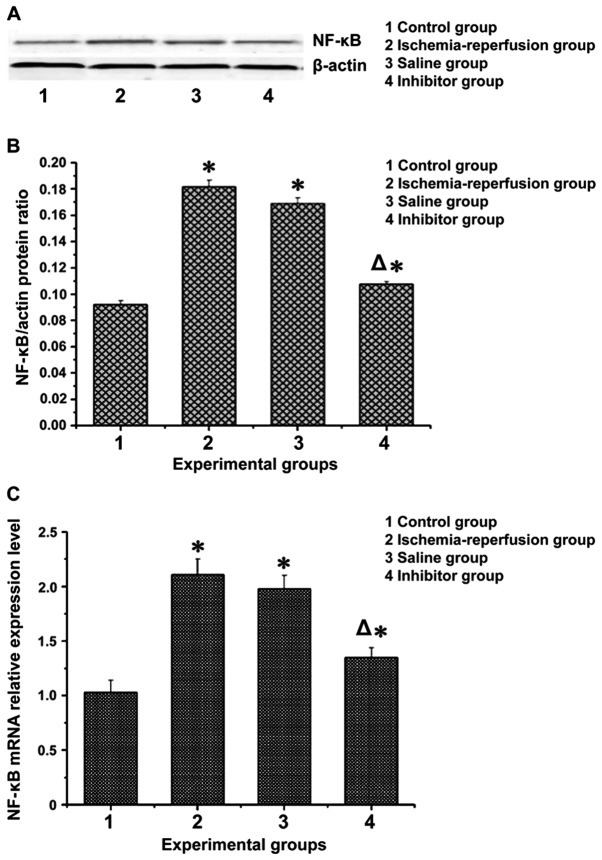
mRNA and protein expression levels of NF-κB and IL-6 in flap tissues
The expression levels of inflammatory factor IL-6 and nuclear transcription factor NF-κB in the flap of each group were processed and are shown in Figs. 2 and 3. Apparently, the mRNA and protein expression levels of IL-6 and NF-κB in the ischemia-reperfusion group and the saline group were significantly higher than those in the control group (P<0.05). These levels in the inhibitor group were lower than those in the ischemia-reperfusion group and the saline group (P<0.05), but higher than those in the control group (P<0.05).
Figure 2.
The mRNA and protein expression levels of IL-6 in the flap tissues. (A) Western blotting measurement of IL-6 expression. (B) IL-6 gray value analysis. (C) RT-PCR measurement of mRNA expression of IL-6: the mRNA and protein expression levels of inflammatory factor IL-6 were much higher in the ischemia-reperfusion group and the saline group than those in the control group and the inhibitor group (P<0.05). *P<0.05, compared with the control group; ΔP<0.05, compared with the ischemia-reperfusion group and the saline group.
Figure 3.
The mRNA and protein expression levels of NF-κB in the flap tissues. (A) Western blot analysis measurement of NF-κB expression. (B) NF-κB gray value analysis. (C) RT-PCR measurement of mRNA expression of NF-κB: the mRNA and protein expression levels of NF-κB were much higher in the ischemia-reperfusion group and the saline group than those in the control group and the inhibitor group (P<0.05). *P<0.05, compared with the control group; ΔP<0.05, compared with the ischemia-reperfusion group and the saline group.
Discussion
Flap ischemia-reperfusion injury is the leading cause of necrosis after flap transplantation. It was found that cell inflammatory factors played an important role in ischemia-reperfusion injury, which can lead to tissue morphological damage and dysfunction (10,11). Therefore, anti-inflammatory response is a major research focus aiming to alleviate ischemia-reperfusion injury and improve flap survival rate (12). Studies have shown that the MAPK pathways that promote occurrence and progression of inflammation were activated in ischemia-reperfusion injuries of a variety of tissues and organs (13). The p38 MAPK pathway, an important member in the MAPK family, played a major role in regulation of intracellular inflammatory response (14,15). In order to understand the role of p38 MAPK signaling pathway in flap ischemia-reperfusion injury and the underlying mechanism, a successfully constructed flap ischemia-reperfusion injury animal model was treated with SB202190, a specific inhibitor of the p38 MAPK pathway. Relevant indicators were measured in the flap, which were used to demonstrate if inhibition of the p38 MAPK pathway had a protective effect in flap ischemia and reperfusion injury.
IL-6 is one of the most common immune cytokines. It is also an important inflammatory mediator of ischemia-reperfusion injury, which can lead to inflammatory injury of tissues and organs. The concentration of IL-6 in serum is an important clinical reference to diagnose inflammatory injury and determine its severity (16–18). Nuclear transcription factor NF-κB is a transcriptional regulator of a variety of cytokine genes. It is also an important transcriptional regulator of the intracellular inflammatory gene, and plays an important role in the inflammatory response (19). Activation of the p38 MAPK pathway leads to elevated expression of NF-κB, which binds to DNA on specific binding sites after entering into the nucleus and upregulate the expression of some inflammatory factors, such as IL-6 and TNF-α (7,20). In this study it was found that in the ischemia-reperfusion model the flap survival rate was lower, but the concentration of IL-6 in serum and the mRNA and protein expression levels of NF-κB and IL-6 in the flap tissue were all higher than the corresponding indicators in the control group. This result was similar to previous reports (17,20). Inflammation leads to an increase in serum IL-6 concentration and an increase in expression levels of NF-κB and IL-6 in tissues of ischemia-reperfusion injury. The flap survival rate in the inhibitor group which was treated with SB202190 was significantly higher than that in the ischemia-reperfusion and saline groups. The concentration of IL-6 in serum and the mRNA and protein concentrations of NF-κB and IL-6 in the flap tissues were significantly decreased. This is similar to previous experimental results (6,14). When the p38 MAPK signaling pathway was inhibited, the expression levels of inflammatory factors decreased, alleviating the inflammatory response in the flap. This finding suggests that SB202190 has a protective effect on flap ischemia-reperfusion injury, which may be achieved by reducing the inflammatory response during the process.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
KG and JM designed the study. WL established the animal models. KG and JM performed western blotting. KG prepared the manuscript. All authors have read and approved the final manuscript.
Ethics approval and consent to participate
This study was approved by the Animal Ethics Committee of Jinan Municipal Hospital of Traditional Chinese Medicine (Jinan, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Cheng HF, Feng Y, Jiang DM, Tao KY, Kong MJ. Protective function of tocilizumab in human cardiac myocytes ischemia reperfusion injury. Asian Pac J Trop Med. 2015;8:48–52. doi: 10.1016/S1995-7645(14)60186-3. [DOI] [PubMed] [Google Scholar]
- 2.Khalil AA, Aziz FA, Hall JC. Reperfusion injury. Plast Reconstr Surg. 2006;117:1024–1033. doi: 10.1097/01.prs.0000204766.17127.54. [DOI] [PubMed] [Google Scholar]
- 3.Uchida Y, Freitas MC, Zhao D, Busuttil RW, Kupiec-Weglinski JW. The protective function of neutrophil elastase inhibitor in liver ischemia/reperfusion injury. Transplantation. 2010;89:1050–1056. doi: 10.1097/TP.0b013e3181d45a98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kanemoto S, Matsubara M, Noma M, Leshnower BG, Parish LM, Jackson BM, Hinmon R, Hamamoto H, Gorman JH, III, Gorman RC. Mild hypothermia to limit myocardial ischemia-reperfusion injury: Importance of timing. Ann Thorac Surg. 2009;87:157–163. doi: 10.1016/j.athoracsur.2008.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Liu YQ, Liu YF, Ma XM, Xiao YD, Wang YB, Zhang MZ, Cheng AX, Wang TT, Li JL, Zhao PX, et al. Hydrogen-rich saline attenuates skin ischemia/reperfusion induced apoptosis via regulating Bax/Bcl-2 ratio and ASK-1/JNK pathway. J Plast Reconstr Aesthet Surg. 2015;68:e147–e156. doi: 10.1016/j.bjps.2015.03.001. [DOI] [PubMed] [Google Scholar]
- 6.Ashraf MI, Ebner M, Wallner C, Haller M, Khalid S, Schwelberger H, Koziel K, Enthammer M, Hermann M, Sickinger S, et al. A p38MAPK/MK2 signaling pathway leading to redox stress, cell death and ischemia/reperfusion injury. Cell Commun Signal. 2014;12:6. doi: 10.1186/1478-811X-12-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Uemura T, Tsujii M, Akeda K, Iino T, Satonaka H, Hasegawa M, Sudo A. Transfection of nuclear factor-kappaB decoy oligodeoxynucleotide protects against ischemia/reperfusion injury in a rat epigastric flap model. J Gene Med. 2012;14:623–631. doi: 10.1002/jgm.2677. [DOI] [PubMed] [Google Scholar]
- 8.Grossi V, Liuzzi M, Murzilli S, Martelli N, Napoli A, Ingravallo G, Del Rio A, Simone C. Sorafenib inhibits p38α activity in colorectal cancer cells and synergizes with the DFG-in inhibitor SB202190 to increase apoptotic response. Cancer Biol Ther. 2012;13:1471–1481. doi: 10.4161/cbt.22254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Manson PN, Anthenelli RM, Im MJ, Bulkley GB, Hoopes JE. The role of oxygen-free radicals in ischemic tissue injury in island skin flaps. Ann Surg. 1983;198:87–90. doi: 10.1097/00000658-198307000-00017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Al-Juboori MJ. Flap designs for implant-related surgical procedures: A review. Implant Dent. 2016;25:845–854. doi: 10.1097/ID.0000000000000460. [DOI] [PubMed] [Google Scholar]
- 11.Silva JJ, Pompeu DG, Ximenes NC, Duarte AS, Gramosa NV, Carvalho KM, Brito GA, Guimarães SB. Effects of kaurenoic acid and arginine on random skin flap oxidative stress, inflammation, and cytokines in rats. Aesthetic Plast Surg. 2015;39:971–977. doi: 10.1007/s00266-015-0559-8. [DOI] [PubMed] [Google Scholar]
- 12.Joshi YB, Praticò D. The 5-lipoxygenase pathway: Oxidative and inflammatory contributions to the Alzheimer's disease phenotype. Front Cell Neurosci. 2015;8:436. doi: 10.3389/fncel.2014.00436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Patten RD, Pourati I, Aronovitz MJ, Baur J, Celestin F, Chen X, Michael A, Haq S, Nuedling S, Grohe C, et al. 17beta-estradiol reduces cardiomyocyte apoptosis in vivo and in vitro via activation of phospho-inositide-3 kinase/Akt signaling. Circ Res. 2004;95:692–699. doi: 10.1161/01.RES.0000144126.57786.89. [DOI] [PubMed] [Google Scholar]
- 14.Ju J, Wu J, Hou R. Role of the p38 mitogen-activated protein kinase signaling pathway in estrogen-mediated protection following flap ischemia-reperfusion injury. Cell Biochem Funct. 2016;34:522–530. doi: 10.1002/cbf.3226. [DOI] [PubMed] [Google Scholar]
- 15.Han HH, Lim YM, Park SW, Lee SJ, Rhie JW, Lee JH. Improved skin flap survival in venous ischemia-reperfusion injury with the use of adipose-derived stem cells. Microsurgery. 2015;35:645–652. doi: 10.1002/micr.22522. [DOI] [PubMed] [Google Scholar]
- 16.Denes A, Thornton P, Rothwell NJ, Allan SM. Inflammation and brain injury: Acute cerebral ischaemia, peripheral and central inflammation. Brain Behav Immun. 2010;24:708–723. doi: 10.1016/j.bbi.2009.09.010. [DOI] [PubMed] [Google Scholar]
- 17.Suzuki S, Tanaka K, Suzuki N. Ambivalent aspects of interleukin-6 in cerebral ischemia: Inflammatory versus neurotrophic aspects. J Cereb Blood Flow Metab. 2009;29:464–479. doi: 10.1038/jcbfm.2008.141. [DOI] [PubMed] [Google Scholar]
- 18.Clark WM, Rinker LG, Lessov NS, Hazel K, Hill JK, Stenzel-Poore M, Eckenstein F, Haley EC. Lack of interleukin-6 expression is not protective against focal central nervous system ischemia. Stroke. 2000;31:1715–1720. doi: 10.1161/01.STR.31.7.1715. [DOI] [PubMed] [Google Scholar]
- 19.Liu SF, Malik AB. NF-kappa B activation as a pathological mechanism of septic shock and inflammation. Am J Physiol Lung Cell Mol Physiol. 2006;290:L622–L645. doi: 10.1152/ajplung.00477.2005. [DOI] [PubMed] [Google Scholar]
- 20.Wang J, Min P, Grassetti L, Lazzeri D, Zhang YX, Nicoli F, Innocenti M, Torresetti M, Levin LS, Persichetti P. Preliminary outcomes of distal IMAP and SEAP flaps for the treatment of unstable keloids subject to recurrent inflammation and infections in the lower sternal and upper abdominal areas. J Reconstr Microsurg. 2015;31:621–630. doi: 10.1055/s-0035-1556078. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.