Abstract
Understanding of vitamin D physiology is important because about half of the population is being diagnosed with deficiency and treated with supplements. Clinical guidelines were developed based on observational studies showing an association between low serum levels and increased cardiovascular risk. However, new randomized-controlled trials have failed to confirm any cardiovascular benefit from supplementation in the general population. A major concern is that excess vitamin D is known to cause calcific vasculopathy and valvulopathy in animal models. For decades, administration of vitamin D has been used in rodents as a reliable experimental model of vascular calcification. Technically, vitamin D is a misnomer. It is not a true vitamin because it can be synthesized endogenously through ultraviolet exposure of the skin. It is a steroid hormone that comes in three forms that are sequential metabolites produced by hydroxylases. As a fat-soluble hormone, the vitamin D-hormone metabolites must have special mechanisms for delivery in the aqueous blood stream. Importantly, endogenously synthesized forms are carried by a binding protein, whereas dietary forms are carried within lipoprotein particles. This may result in distinct bio-distributions for sunlight-derived vs. supplement-derived vitamin D-hormones. Since the cardiovascular effects of vitamin D-hormones are not straightforward, both toxic and beneficial effects may result from current recommendations.
Keywords: Cholecalciferol, vitamin D, calcitriol, cardiovascular, calcification
On November 30, 2010, at the request of the Canadian and U.S. Governments, the Institute of Medicine provided a report addressing conflicting information on vitamin D.1 This report took into consideration more than 1,000 studies and reports and considered testimony from scientists and “stakeholders.” Outcomes included bone and cardiovascular diseases as well as cancer, diabetes, inflammation, neuropsychological function, physical performance, pre-eclampsia and reproduction. The overall conclusion of this report was that “the majority of Americans and Canadians are receiving adequate amounts of both calcium and vitamin D,” and that “too much of these nutrients may be harmful.” It further noted that “information about health benefits beyond those for bone -- benefits often reported in the media -- were from studies with mixed and inconclusive results that could not be considered reliable.”2 Even before this report, Towler had noted that the effects of vitamin D on cardiovascular health are complex and biphasic, with direct and indirect actions mediating its vasculotropic actions.3 There is not yet evidence from a randomized controlled trial showing cardiovascular benefit of vitamin D supplementation.4
Until recently, hormonal regulation of calcium-phosphate metabolism by vitamin D metabolites and the parathyroid gland were of little interest to cardiovascular scientists and clinicians. But with new clinical guidelines and media attention, awareness of vitamin D physiology is necessary, especially given that, despite the conclusions of the Institute of Medicine, routine vitamin D testing and supplementation are widely recommended by physicians. As commonly occurs with supplements, it is often used in doses far beyond those directed. Given its extensive actions in human metabolism - both beneficial and harmful - the biochemistry, physiology, and financial motivations surrounding vitamin D warrant attention.
Forms of vitamin D
From a technical standpoint, the term “vitamin D” is a misnomer. It is not a true vitamin because the human body has the capacity to synthesize its own cholecalciferol (D3), except in rare instances of complete lack of ultraviolet radiation. It is more accurate to view it as a steroid hormone or an oxysterol. The International Union of Pure and Applied Chemistry’s Commission on the Nomenclature of Biological Chemistry defines vitamin D3 as a steroid or secosteroid. Its chemical name is 9,10-secocholesta-5,7,10(19)-trien-3beta-ol. Six different steroid hormones go by the name “vitamin D,” with varying degrees of activity: the endogenous precursor, cholecalciferol (D3), which is derived from cholesterol; its hydroxylated derivative, calcidiol [25(OH)D3], which has partial activity; and its hydroxylated derivative, the “active” dihydroxy form, calcitriol [1,25(OH)2D3]. In addition, there is a plant-derived form, ergocalciferol (D2), which also has the corresponding monohydroxy and dihydroxy metabolites. Since 25(OH)D3 is longer lasting, it is the level of this hormone -- not that of the more active 1,25(OH)2D3 -- that is used to diagnose clinical deficiency. Notably, levels of 25(OH)D3 tend to vary inversely with the levels of the active form, possibly due to displacement of the active metabolite from D-binding protein (DBP).5 In this article, we will use the term “vitamin D-hormones” for all six types of steroid hormones and to emphasize their true physiological nature.
Sources of vitamin D-hormones and biodistribution
Sources of vitamin D-hormones include exposure to ultraviolet light, certain foods, and dietary supplements. As one of the four fat-soluble vitamins (A, D, E, and K), its lipophilicity requires special mechanisms to pass through the aqueous environment of blood to reach tissues and cells. Separate mechanisms are used for the endogenous D3 synthesized in sun-exposed skin vs. exogenous D3 obtained from diet or supplements. This may result in distinct pharmacokinetic volumes and targets of distribution (Figure 1).
Figure 1. Vitamin D-hormone metabolism, carriers and distribution.
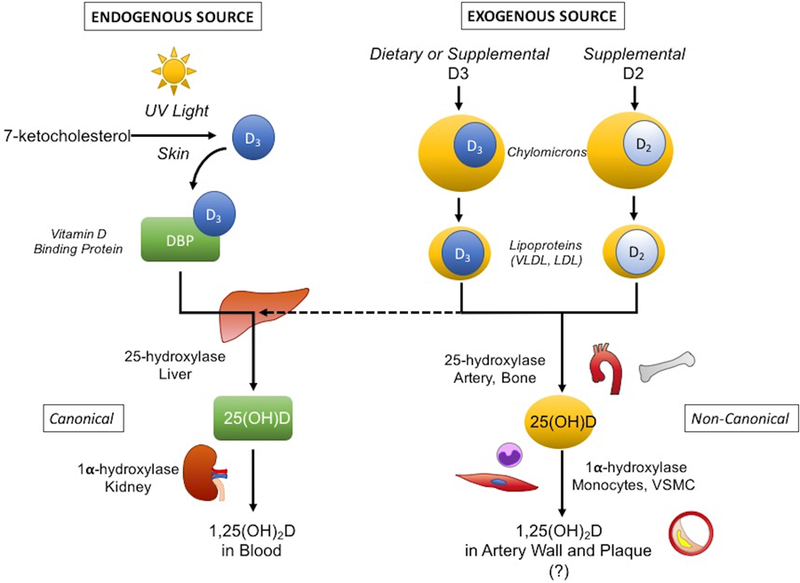
Due to its lipophilic nature, endogenously produced D3 is carried in the aqueous environment of blood by D-binding protein (DBP), whereas exogenous (dietary and supplemental) D3 and D2, absorbed from the intestines, are transported within chylomicrons, which are further processed to lipoproteins (e.g., VLDL and LDL), many of which continue to carry the exogenous D. The conventional sites of vitamin D-hormone metabolism are the liver and proximal tubules of the kidney, where hydroxylases convert it to its active form. But hydroxylase activity is also found in parenchymal and immune cells, including VSMC and monocytes, in other tissues. This raises the potential for accumulation of vitamin D-hormone within LDL in the subendothelial space, where it may undergo activation in atherosclerotic plaque and possibly influence ectopic differentiation and calcification. Abbreviations: LDL, low-density lipoprotein; VLDL, very low-density lipoprotein; VSMC, vascular smooth muscle cells; UV, ultraviolet.
Endogenous vitamin D-hormone synthesis, transport and activation
Endogenous vitamin D–hormone synthesis occurs by ultraviolet light exposure of 7-dehydrocholesterol within the microvessels of the skin resulting in its conversion into cholecalciferol (D3). But, as a fat-soluble oxysterol, D3 must be carried in the blood by DBP, a liver-derived apoprotein and a member of the albumin gene family.6 For light-skinned individuals, sun exposure of the face and arms for just 15 minutes per week may produce tens of thousands of units of cholecalciferol. This endogenous production from sun exposure had been the major source for most humans for centuries. The fact that sun-derived D3 is carried on DBP is a key difference from exogenous D3, because of its potential influence on bio-distribution.
Exogenous vitamin D-hormone sources and delivery
Exogenous sources of vitamin D-hormones include diet (eggs, fish, liver, and marine mammal fat) and supplements. A cup of milk provides about 100 international units (IU) and a serving of salmon contains about 400 IU of D3. While the dietary sources may be in the D3 or D2 form, supplements typically derive from the plant-derived hormone, ergocalciferol (D2). A key feature of dietary or supplemental sources is that D3 taken orally is absorbed from the intestinal tract via chylomicrons,7 which pass into the lymphatic circulation before returning to the central venous circulation via the thoracic duct. Eventually, about 35% of ingested D3 is carried in lipoproteins,8 rather than DBP.
Activation by sequential hydroxylation of D3
For both endogenous and exogenous sources, the D3 carried in the bloodstream on either DBP or lipoproteins undergoes a two-step sequential hydroxylation to active metabolites. First, it is converted by 25-hydroxylase to the monohydroxy- derivative, 25(OH)D3, the metabolite that is measured for “vitamin D levels.” This occurs primarily in the liver, but may take place in other tissues as well. Next, 25(OH)D3 is further hydroxylated by 1-alpha hydroxylase to the active, dihydroxy- form, 1,25(OH)2D3.8 This occurs primarily in the capillaries surrounding the proximal convoluted tubules of kidney, but, importantly, the enzyme producing the active form is also found in vascular cells and monocytes among other tissues and cells (Table).
Table.
Partial literature summary of vitamin D-hormone actions
| Vitamin D-hormone effects | Targets |
|---|---|
| Cells that metabolize D3 | • Hepatocytes,66 renal cells,67 endothelial cells,68 smooth muscle cells,69 monocytes,70 skeletal muscle cells71 |
| Pathophysiology affected | • Osteomalacia,72 vascular calcification,56, 57,73,28 renal and myocardial calcification,73 atherosclerosis,74 heart failure,75 thrombosis76 |
| Tissue content and function affected |
• Calcium balance,77 aortic elastin,78 bile acid synthesis,79 vasoconstrictor response to norepinephrine,80 lipogenesis,81 insulin sensitivity,82 endothelial-dependent contraction83 |
| Cellular functions | • Osteoblastogenesis,84 osteoclastogenesis,85 chondrogenesis86 • Myogenesis,87adipogenesis,88 hematopoiesis,89 nerve growth,90 • Smooth muscle cell (SMC) dedifferentiation,91 SMC migration,92 SMC contraction,93 • Tumor cell differentiation94 • Macrophage cholesterol uptake,95 T-lymphocytes96 • Apoptosis,97 DNA synthesis,98 arachidonic acid turnover99 • Oscillation of inositol phosphate 3 and diacylglycerol production100 • Prostaglandin production,101, 102 superoxide anion production103 • Nitric oxide synthase104 |
| Signaling pathways | • Protein kinase C-alpha,105 cyclic AMP,106 p38 MAPK,107 c-myc,108 c-fos,85 NFAT1,109 Wnt,110 nuclear factor-kappa B111 |
| Known interactions | • Retinoic acid receptors,112 retinoid X receptors,113 glucocortoid,114 • Runx2,115 transforming growth factor-beta,116 insulin-like growth factor binding protein-5,117 vitamin K,118 estrogen,119 |
| Protein synthesis and serum levels regulated |
•Low-density lipoprotein (LDL),120 prostaglandin synthesis,101 endothelin receptor91 |
| Gene expression | • Bone morphogenetic protein −2,121 alkaline phosphatase,122 bone sialoprotein,123 collagen I,124 osteopontin,124 osteocalcin115 tissue plasminogen activator,125 parathyroid hormone,126 fibroblast growth factor-23127 • Receptor activator of nuclear factor kappa B ligand128 • Muscle segment homeobox-containing gene Msx-2 (Hox-8),129 • Peroxisome proliferator activated receptor-gamma130 • Integrins,131 fibronectin,132 laminin receptor133 • Collagenase,134 matrix metalloproteinase135 • Granulocyte-macrophage colony stimulating factor,109, 114 macrophage-colony stimulating factor136 tumor necrosis factor-alpha,137 interleukin 8138 • Insulin receptor139 • Vascular endothelial growth factor,140 nerve growth factor,141 • Type A natriuretic peptide receptor142 • Aromatase143 • Nephrin112 • CYP7A1,144 CYP19A1,143 CYP24A1145 • p450 cytochrome,146 Mullerian-inhibiting substance147 |
Guidelines for vitamin D-hormone assessment and supplementation
Different criteria for vitamin D deficiency have been proposed by the Endocrine Society, Osteoporosis Society, and Institute of Medicine. Normal reference values shown by individual clinical laboratories are not standardized. Conservative definitions define vitamin D deficiency as levels of 25(OH)D3 < 20 ng/ml (< 50 nmol/L), and vitamin D insufficiency as 20 – 30 ng/ml (50 – 75 nmol/L).9 The Institute of Medicine chooses cut-off values of < 12 ng/ml and > 50 ng/ml as levels with increased risk of deficiency and excess, respectively.1 The Institute of Medicine does not recommend specific doses, but, based on bone health indicators, their analysis suggests that the daily use of vitamin D is 600 IU for individuals from 1 to 70 years of age, and 800 IU for individuals 71 and older, some or all of which may be achieved by ordinary sun exposure.1 They further suggest a “safe upper limit” of dietary vitamin D intake as 4000 IU daily, a level at which risk for toxicity begins to increase. Yet, the Institute of Medicine emphasizes that this upper limit “should not be misunderstood as amounts people need or should strive to consume.”1
The use of the terms “daily” and “per day” in these recommendations may give the false impression that a day without sunshine requires a dose of supplement. Even though adults may use a given amount of cholecalciferol each day, such daily use does not necessarily require daily replacement. 25(OH)D3 has a half-life of 2 weeks10 to 3 months,11 and is stored primarily in adipose tissue12, 13 and, to a lesser extent, in the liver.14 Presumably, this stored source of vitamin D is available for release back into the plasma, as indicated by a long-term study in Norwegians.15 Moreover, cholecalciferol recycles in the enterohepatic circulation.16 Thus, vitamin D-hormones may not require daily, weekly or even monthly replenishment. Summer sun exposure may provide enough for the winter.17 Major institutions have used dosing schedules as infrequent as once every 1–4 months.18, 19 It may be more correct to refer to a monthly requirement, and this requirement may vary depending on age (< 70 years or > 70 years) or season (i.e., summer vs. winter).17
Personalized approach to vitamin D-hormones
A personalized medicine approach is important in considering vitamin D-hormone supplementation because of the influence of differences in body composition, environmental factors, and genetic variations in D binding protein as well as variations in the intracellular vitamin D receptor. Although darkly pigmented individuals are believed to require more sun exposure to generate the same amount of vitamin D-hormones, they have genetic polymorphisms of the vitamin D binding protein,20 which change the bioavailability of vitamin D, counteracting the decrease in synthesis.21 The half-life of 25(OH)D3 in the bloodstream is influenced by genotype of the DBP. Based on in vitro studies, the intracellular vitamin D receptor (VDR), which binds and translocates 1,25(OH)2D3 to the nucleus, has approximately 7% readthrough efficiencies, producing VDR proteoforms that have reduced binding.22 This phenomenon may vary among individuals. High body fat content may decrease availability of fat-soluble 25(OH)D3 due to sequestration in adipose tissue.23 Conversely, high skeletal muscle content also modulates vitamin D-hormone availability; muscle cells internalize D-binding protein and expose it, allowing extensive intracellular uptake and retention of 25(OH)D3.24 The elderly may have lower levels due to less outdoor activity and sun exposure.17 Polymorphisms arising in the Inuit Eskimo background limit intestinal D3 uptake protect against excess D3 ingestion from the ancestral diet of whale blubber rich in vitamin D hormones. These genetic variations, phenotypic differences, and environmental influences underscore the importance of tailoring any recommendations for vitamin D supplementation to individualized needs.
Vitamin D-hormones and cardiovascular health
Evidence for cardiovascular protection by vitamin D-hormones was almost entirely inferred from observational studies. Confounders that may adversely affect results of these studies include obesity, which alters the storage of vitamin D, skeletal muscle content, physical exercise, which corresponds with time outdoors in the sun, and illness, which corresponds with time indoors.
Widespread supplementation became a guideline25 based on observational studies without adequate randomized controlled trials.2 Although it is widely cited as showing an inverse relation between 25(OH)D3 levels and cardiovascular risk, a close look at data in the Offspring Cohort of the Framingham Heart Study26 shows a U-shaped relationship between 25(OH)D3 levels and cardiovascular risk (Figure 226). The apparent minimum risk occurs at a serum level of approximately 20 ng/ml, far below the level considered sufficient. A U-shaped curve was also found for the relationship between 25(OH)D3 and all-cause mortality in the NHANES III population.27 Preclinical studies support this relationship with both deficiency and excess of 25(OH)D3 increasing atherosclerotic calcification.28 Yet, public education campaigns continue to describe the relationship as inverse.
Figure 2.

U-shape of the multivariable-adjusted relation between baseline serum 25(OH)D3 levels and incident cardiovascular events reported by Wang et al. (from Circulation 117:503–511, with permission [pending]). Solid lines show the estimated relation of adjusted hazard ratios (with 95% confidence limits) and 25(OH)D3 levels when time to cardiovascular event is modeled as a function of penalized regression splines of 25(OH)D3 levels with adjustment for all other covariates. Hatched lines on the horizontal axis represent cardiovascular events (top axis) and individuals (bottom axis). This relationship suggests increased cardiovascular risk at both low and high levels.
Although observational studies show associations between 25(OH)D3 levels and cardiovascular risk, the few randomized controlled trials available have failed to confirm any cardiovascular benefit of supplementation29 with the exception of patients with chronic kidney disease, where the kidney’s 1alpha-hydroxylase activity and capacity to produce active 1,25(OH)2D3 are greatly diminished. One study found no reduction in cardiovascular risk factors in patients randomized to supplementation.30 Another study of over 5000 patients found no reduction in cardiovascular mortality in patients randomized to supplementation, even though the treatment increased 25(OH)D3 levels by an average of 20 ng/ml.18 In contrast, patients with renal insufficiency and/or dialysis, where vitamin D-hormone deficiency is prevalent,31 vitamin D-hormone supplementation improved vascular function32–34 without affecting plasma levels of calcium and phosphate.35, 36 Overall, only the observational studies showed reductions in all-cause and cardiovascular mortality.37–39
Accumulation and activation of LDL-associated vitamin D-hormones in the artery wall
Both mono- and dihydroxy- forms of vitamin D-hormones are delivered to cells either by DBP,40 where entry is mediated by the endocytic receptors, cubilin/megalin,41 or by lipoproteins,8 where entry is mediated by the low-density lipoprotein (LDL) receptor.42 However, during pathogenesis of atherosclerosis, vitamin D-hormones that are consumed in the diet may accompany LDL into the subendothelial space of the artery wall where atherosclerotic lesions form.43 In peripheral tissues that express lipoprotein lipase, the chylomicron metabolizing enzyme, a fraction of vitamin D-hormones can be taken up by the tissues. Since 1-alpha hydroxylase is present in tissues and cells, including vessel walls and monocyte-derived cells, the active form may be produced locally within the artery wall and, conceivably, within monocyte-laden atherosclerotic plaque.44
Targets of vitamin D-hormones
Cellular and molecular effects of vitamin D-hormones are extensive. In addition to homodimerization, VDR heterodimerizes with the retinoid X receptor to activate transcription of a wide range of genes. As steroid hormones, they are related to estrogen, testosterone, mineralocorticoids, and glucocorticoids. Even our limited search of the literature (Table) reveals hundreds of diverse genomic and non-genomic targets of vitamin D-hormones, affecting a vast array of physiological functions. Adding further complexity, vitamin D-hormones have significant cross-talk with steroid and nuclear hormones and their receptors.45 For instance, vitamin D3 may affect actions of glucocorticoids.46,47 Conversely, steroid and xenobiotic receptors48 as well as peroxisome proliferator-activated receptor gamma49 inhibit VDR-mediated CYP24 (24-hydroxylase) promoter activity.
Effects of vitamin D-hormones in the vasculature
Given that diet-derived 25(OH)D3 is carried in lipoproteins, and that lipoproteins accumulate in the subendothelial space of arteries leading to atherosclerotic lesions, it is likely that diet-derived 25(OH)D3 also accumulates in the neointima artery wall and atherosclerotic plaque. Given that vascular smooth muscle cells and monocytes both produce 1-alpha hydroxylase, it follows that 1,25(OH)2D3 may also accumulate in artery walls and atherosclerosis. Potential effects of this accumulation remain to be determined. One possibility is acceleration of both atherosclerosis and cardiovascular calcification, based on studies showing that vitamin D-receptor deficiency significantly reduces calcific atherosclerosis in hyperlipidemic mice.50 Vitamin D-hormones are known to stimulate smooth muscle cell proliferation51 and induce expression of fibroblast growth factor-23 (FGF-23), high levels of which are linked to adverse cardiovascular events.52 With respect to mineralization, effects of vitamin D-hormones are double-edged. Although there is convincing evidence that supplements increase bone density,53 any benefit to bone may be at the cost of cardiovascular morbidity and mortality due to calcific vasculopathy and valvulopathy. Cardiovascular calcification has been shown to occur by many of the same cellular and molecular processes as bone mineralization,54 including induction of osteogenic factors by vitamin D-hormones.55 Indeed, high dose vitamin D supplements used for several decades as an experimental model reproducibly induces severe aortic calcification, acutely and chronically, over a wide range of conditions in a variety of species in the hands of many different investigators.56,57 The dramatic vascular calcification seen in patients with chronic kidney disease may be due in part to local induction of 1-alpha hydroxylase in the artery wall.58 The extensive immunomodulatory effects of vitamin D have been reviewed elsewhere.59
Vitamin D-hormone toxicity and benefits
Overuse of vitamin D-hormone supplements carries significant risks that have been known for decades, and these risks have traditionally been associated with those of the resulting hypercalcemia that can occur at 25(OH)D3 plasma concentrations of > 150 ng/ml (> 375 nmol/L). Thus, the traditional clinical manifestations of vitamin D-hormone toxicity are those of hypercalcemia, which include generalized (fatigue, weakness), neurological (altered mental status, irritability, coma), gastrointestinal (nausea, vomiting, constipation), and endocrinological (polyuria, polydipsia) symptoms. Additionally, renal injury as well as the development of kidney stones may occur. As such, studies evaluating the safety of various dosing regimens typically use measurements of serum and urinary calcium to monitor the safety of the administered doses.60, 61
However, given the number of cell types and tissues that possess 25-hydroxylase, vitamin D-hormones may have effects on these systems without necessarily affecting the serum or urinary calcium levels, and all of the biological processes listed in the Table may be deranged by excess intake. A daily intake of 25(OH)D3 up to 4,000 IU is deemed to be the upper limit of safety,62 as the risk of harm appears to increase above this level. Yet as discussed above, variations in vitamin D-hormone production and metabolism may depend significantly on individual genotype, phenotype, and environmental conditions; thus, a universal upper limit of safety and a universal lower limit of sufficiency for all patients may not necessarily be accurate. Additionally, excess vitamin D-hormone supplements also displace the active form from binding sites, making it more available even when not appropriate.5 Further, given the cross-talk with other steroid hormone receptors, vitamin D-hormones in excess may have physiological effects similar to those of glucocorticoids, estrogen, or even those of anabolic steroids.63 Nonetheless, in general, it is difficult to categorize any one of the numerous effects of vitamin D as necessarily beneficial or toxic, given the dependence on location as well as physiological and pathological contexts. For instance, osteoblastogenesis may be beneficial in osteoporosis but hazardous in calcific vasculopathy and valvulopathy.
Concluding remarks
Widely used guidelines for monitoring and supplementing vitamin D3 hormones resembles the ill-fated call, years ago, for widespread use of another steroid hormone, estradiol, for post-menopausal women based on observational studies. The impact of confounding environmental factors was not recognized. Even after the Women’s Health Initiative showed increased cardiovascular risk in postmenopausal women randomized to hormone replacement therapy,19 recommendations were slow to change. Observational studies are not sufficient to recommend widespread hormonal supplementation, and the same applies to vitamin D-hormones. The ongoing randomized clinical trial, Vitamin D and Omega-3 Trial (VITAL), will be helpful in determining whether vitamin D-hormone supplementation provides any benefit in the primary prevention of cancer and cardiovascular disease.64
Decades ago, the pioneering Johns Hopkins cardiologist, Dr. Helen Taussig, anticipated the need for a personalized approach to D3 supplements: “As is so common, the popular belief was that ‘if some is good, more is better.’ The result was the overdosing with vitamin D and adding it to various foods. Then came the recognition of vitamin D intoxication... we are coming to appreciate that there exists an inborn variation in man’s ability to metabolize vitamin D and that some individuals may be injured by doses of vitamin D which are safe for others.”65 For health reasons, many Americans pay extra for bread free of preservatives (such as antioxidants) and meats that are free of steroid hormones. In the next aisle of the store, they buy bottles of antioxidant preservatives and steroid hormones in pill form, labeled as nutritional supplements, including D3 hormones. Scientists need to use their knowledge of molecular, cellular, and integrative physiology to advocate for rational use of vitamin D-hormone supplements to prevent adverse consequences to cardiovascular health by overenthusiastic guidelines followed by well-meaning physicians.
Acknowledgements
Without express permission, the authors wish to acknowledge the late Hywel Davies MD FRCP (1924–2017), whose writing and input contributed to this review.
SOURCES OF FUNDING
This work was supported in part by funding from the National Institutes of Health (HL114709, HL121019, HL007895), the Claude D. Pepper Older American Independence Center (OAIC) at UCLA, and an award from the UCLA Specialty Training and Advanced Research (STAR) Program.
Nonstandard Abbreviations and Acronyms:
- 25(OH)D3
Calcidiol
- 1,25(OH)2D3
Calcitriol
- DBP
(Vitamin) D-binding protein
- IU
International unit
- D2
Ergocalciferol
- VDR
Vitamin D receptor
- LDL
Low-density lipoprotein
- VLDL
Very low-density lipoprotein CYP24 (24-hydroxylase)
- FGF-23
Fibroblast growth factor-23
- SMC
Smooth muscle cell
- VSMC
Vascular smooth muscle cell
- Hox-8
Muscle segment homeobox-containing gene Msx-2
- UV
Ultraviolet
- NFAT1
Nuclear factor of activated T cells 1
- MAPK
Mitogen-activated protein kinase
- c-myc
Cellular homolog of the oncogene of avian myelocytomatosis virus strain 29
- Runx2
Runt related transcription factor-2
Footnotes
DISCLOSURES:
None.
REFERENCES
- 1.Committee IoM. In: Ross AC, Taylor CL, Yaktine AL, Del Valle HB, eds. Dietary reference intakes for calcium and vitamin d Washington (DC: ); 2011. [PubMed] [Google Scholar]
- 2.Shapses SA, Manson JE. Vitamin d and prevention of cardiovascular disease and diabetes: Why the evidence falls short. JAMA 2011;305:2565–2566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Towler DA. Calciotropic hormones and arterial physiology: “D”-lightful insights. J Am Soc Nephrol 2007;18:369–373 [DOI] [PubMed] [Google Scholar]
- 4.Veloudi P, Jones G, Sharman JE. Effectiveness of vitamin d supplementation for cardiovascular health outcomes. Pulse (Basel) 2017;4:193–207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fraser DR. Vitamin d. Lancet 1995;345:104–107 [DOI] [PubMed] [Google Scholar]
- 6.Constans J Group-specific component is not only a vitamin-d-binding protein. Exp Clin Immunogenet 1992;9:161–175 [PubMed] [Google Scholar]
- 7.Bays HE, Neff D, Tomassini JE, Tershakovec AM. Ezetimibe: Cholesterol lowering and beyond. Expert Rev Cardiovasc Ther 2008;6:447–470 [DOI] [PubMed] [Google Scholar]
- 8.Haddad JG, Matsuoka LY, Hollis BW, Hu YZ, Wortsman J. Human plasma transport of vitamin d after its endogenous synthesis. J Clin Invest 1993;91:2552–2555 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Holick MF. Vitamin d deficiency. N Engl J Med 2007;357:266–281 [DOI] [PubMed] [Google Scholar]
- 10.Jones KS, Assar S, Harnpanich D, Bouillon R, Lambrechts D, Prentice A, Schoenmakers I. 25(oh)d2 half-life is shorter than 25(oh)d3 half-life and is influenced by dbp concentration and genotype. J Clin Endocrinol Metab 2014;99:3373–3381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Oliveri B, Mastaglia SR, Brito GM, Seijo M, Keller GA, Somoza J, Diez RA, Di Girolamo G. Vitamin d3 seems more appropriate than d2 to sustain adequate levels of 25ohd: A pharmacokinetic approach. Eur J Clin Nutr 2015;69:697–702 [DOI] [PubMed] [Google Scholar]
- 12.Heaney RP, Recker RR, Grote J, Horst RL, Armas LA. Vitamin d(3) is more potent than vitamin d(2) in humans. J Clin Endocrinol Metab 2011;96:E447–452 [DOI] [PubMed] [Google Scholar]
- 13.Rosenstreich SJ, Rich C, Volwiler W. Deposition in and release of vitamin d3 from body fat: Evidence for a storage site in the rat. J Clin Invest 1971;50:679–687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dueland S, Holmberg I, Berg T, Pedersen JI. Uptake and 25-hydroxylation of vitamin d3 by isolated rat liver cells. J Biol Chem 1981;256:10430–10434 [PubMed] [Google Scholar]
- 15.Martinaityte I, Kamycheva E, Didriksen A, Jakobsen J, Jorde R. Vitamin d stored in fat tissue during a 5-year intervention affects serum 25-hydroxyvitamin d levels the following year. J Clin Endocrinol Metab 2017;102:3731–3738 [DOI] [PubMed] [Google Scholar]
- 16.Arnaud SB, Goldsmith RS, Lambert PW, Go VL. 25-hydroxyvitamin d3: Evidence of an enterohepatic circulation in man. Proc Soc Exp Biol Med 1975;149:570–572 [DOI] [PubMed] [Google Scholar]
- 17.Lawson DE, Paul AA, Black AE, Cole TJ, Mandal AR, Davie M. Relative contributions of diet and sunlight to vitamin d state in the elderly. Br Med J 1979;2:303–305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Scragg R, Stewart AW, Waayer D, Lawes CMM, Toop L, Sluyter J, Murphy J, Khaw KT, Camargo CA Jr. Effect of monthly high-dose vitamin d supplementation on cardiovascular disease in the vitamin d assessment study : A randomized clinical trial. JAMA Cardiol 2017;2:608–616 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Manson JE, Hsia J, Johnson KC, Rossouw JE, Assaf AR, Lasser NL, Trevisan M, Black HR, Heckbert SR, Detrano R, Strickland OL, Wong ND, Crouse JR, Stein E, Cushman M, Women’s Health Initiative I. Estrogen plus progestin and the risk of coronary heart disease. N Engl J Med 2003;349:523–534 [DOI] [PubMed] [Google Scholar]
- 20.Kamboh MI, Ferrell RE. Ethnic variation in vitamin d-binding protein (gc): A review of isoelectric focusing studies in human populations. Hum Genet 1986;72:281–293 [DOI] [PubMed] [Google Scholar]
- 21.Sinotte M, Diorio C, Berube S, Pollak M, Brisson J. Genetic polymorphisms of the vitamin d binding protein and plasma concentrations of 25-hydroxyvitamin d in premenopausal women. Am J Clin Nutr 2009;89:634–640 [DOI] [PubMed] [Google Scholar]
- 22.Loughran G, Jungreis I, Tzani I, Power M, Dmitriev RI, Ivanov IP, Kellis M, Atkins JF. Stop codon readthrough generates a c-terminally extended variant of the human vitamin d receptor with reduced calcitriol response. J Biol Chem 2018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gangloff A, Bergeron J, Lemieux I, Despres JP. Changes in circulating vitamin d levels with loss of adipose tissue. Curr Opin Clin Nutr Metab Care 2016;19:464–470 [DOI] [PubMed] [Google Scholar]
- 24.Abboud M, Rybchyn MS, Ning YJ, Brennan-Speranza TC, Girgis CM, Gunton JE, Fraser DR, Mason RS. 1,25-dihydroxycholecalciferol (calcitriol) modifies uptake and release of 25-hydroxycholecalciferol in skeletal muscle cells in culture. J Steroid Biochem Mol Biol 2018;177:109–115 [DOI] [PubMed] [Google Scholar]
- 25.Pludowski P, Holick MF, Grant WB, Konstantynowicz J, Mascarenhas MR, Haq A, Povoroznyuk V, Balatska N, Barbosa AP, Karonova T, Rudenka E, Misiorowski W, Zakharova I, Rudenka A, Lukaszkiewicz J, Marcinowska-Suchowierska E, Laszcz N, Abramowicz P, Bhattoa HP, Wimalawansa SJ. Vitamin d supplementation guidelines. J Steroid Biochem Mol Biol 2018;175:125–135 [DOI] [PubMed] [Google Scholar]
- 26.Wang TJ, Pencina MJ, Booth SL, Jacques PF, Ingelsson E, Lanier K, Benjamin EJ, D’Agostino RB, Wolf M, Vasan RS. Vitamin d deficiency and risk of cardiovascular disease. Circulation 2008;117:503–511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Melamed ML, Michos ED, Post W, Astor B. 25-hydroxyvitamin d levels and the risk of mortality in the general population. Arch Intern Med 2008;168:1629–1637 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ellam T, Hameed A, ul Haque R, Muthana M, Wilkie M, Francis SE, Chico TJ. Vitamin d deficiency and exogenous vitamin d excess similarly increase diffuse atherosclerotic calcification in apolipoprotein e knockout mice. PLoS One 2014;9:e88767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pilz S, Verheyen N, Grubler MR, Tomaschitz A, Marz W. Vitamin d and cardiovascular disease prevention. Nat Rev Cardiol 2016;13:404–417 [DOI] [PubMed] [Google Scholar]
- 30.Seibert E, Lehmann U, Riedel A, Ulrich C, Hirche F, Brandsch C, Dierkes J, Girndt M, Stangl GI. Vitamin d3 supplementation does not modify cardiovascular risk profile of adults with inadequate vitamin d status. Eur J Nutr 2017;56:621–634 [DOI] [PubMed] [Google Scholar]
- 31.Cupisti A, Vigo V, Baronti ME, D’Alessandro C, Ghiadoni L, Egidi MF. Vitamin d status and cholecalciferol supplementation in chronic kidney disease patients: An italian cohort report. Int J Nephrol Renovasc Dis 2015;8:151–157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chitalia N, Ismail T, Tooth L, Boa F, Hampson G, Goldsmith D, Kaski JC, Banerjee D. Impact of vitamin d supplementation on arterial vasomotion, stiffness and endothelial biomarkers in chronic kidney disease patients. PLoS One 2014;9:e91363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kumar V, Yadav AK, Lal A, Kumar V, Singhal M, Billot L, Gupta KL, Banerjee D, Jha V. A randomized trial of vitamin d supplementation on vascular function in ckd. J Am Soc Nephrol 2017;28:3100–3108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Levin A, Tang M, Perry T, Zalunardo N, Beaulieu M, Dubland JA, Zerr K, Djurdjev O. Randomized controlled trial for the effect of vitamin d supplementation on vascular stiffness in ckd. Clin J Am Soc Nephrol 2017;12:1447–1460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bhan I, Dobens D, Tamez H, Deferio JJ, Li YC, Warren HS, Ankers E, Wenger J, Tucker JK, Trottier C, Pathan F, Kalim S, Nigwekar SU, Thadhani R. Nutritional vitamin d supplementation in dialysis: A randomized trial. Clin J Am Soc Nephrol 2015;10:611–619 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Garcia-Lopes MG, Pillar R, Kamimura MA, Rocha LA, Canziani ME, Carvalho AB, Cuppari L. Cholecalciferol supplementation in chronic kidney disease: Restoration of vitamin d status and impact on parathyroid hormone. Ann Nutr Metab 2012;61:74–82 [DOI] [PubMed] [Google Scholar]
- 37.Lu RJ, Zhu SM, Tang FL, Zhu XS, Fan ZD, Wang GL, Jiang YF, Zhang Y. Effects of vitamin d or its analogues on the mortality of patients with chronic kidney disease: An updated systematic review and meta-analysis. Eur J Clin Nutr 2017;71:683–693 [DOI] [PubMed] [Google Scholar]
- 38.Mann MC, Hobbs AJ, Hemmelgarn BR, Roberts DJ, Ahmed SB, Rabi DM. Effect of oral vitamin d analogs on mortality and cardiovascular outcomes among adults with chronic kidney disease: A meta-analysis. Clin Kidney J 2015;8:41–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zheng Z, Shi H, Jia J, Li D, Lin S. Vitamin d supplementation and mortality risk in chronic kidney disease: A meta-analysis of 20 observational studies. BMC Nephrol 2013;14:199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Haddad JG. Plasma vitamin d-binding protein (gc-globulin): Multiple tasks. J Steroid Biochem Mol Biol 1995;53:579–582 [DOI] [PubMed] [Google Scholar]
- 41.Christensen EI, Birn H. Megalin and cubilin: Multifunctional endocytic receptors. Nat Rev Mol Cell Biol 2002;3:256–266 [DOI] [PubMed] [Google Scholar]
- 42.Teramoto T, Endo K, Ikeda K, Kubodera N, Kinoshita M, Yamanaka M, Ogata E. Binding of vitamin d to low-density-lipoprotein (ldl) and ldl receptor-mediated pathway into cells. Biochem Biophys Res Commun 1995;215:199–204 [DOI] [PubMed] [Google Scholar]
- 43.Navab M, Fogelman AM, Berliner JA, Territo MC, Demer LL, Frank JS, Watson AD, Edwards PA, Lusis AJ. Pathogenesis of atherosclerosis. Am J Cardiol 1995;76:18C–23C [DOI] [PubMed] [Google Scholar]
- 44.Hsu JJ, Tintut Y, Demer LL. Vitamin d and osteogenic differentiation in the artery wall. Clin J Am Soc Nephrol 2008;3:1542–1547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Schwartz N, Verma A, Bivens CB, Schwartz Z, Boyan BD. Rapid steroid hormone actions via membrane receptors. Biochim Biophys Acta 2016;1863:2289–2298 [DOI] [PubMed] [Google Scholar]
- 46.Homme M, Schmitt CP, Himmele R, Hoffmann GF, Mehls O, Schaefer F. Vitamin d and dexamethasone inversely regulate parathyroid hormone-induced regulator of g protein signaling-2 expression in osteoblast-like cells. Endocrinology 2003;144:2496–2504 [DOI] [PubMed] [Google Scholar]
- 47.Obradovic D, Gronemeyer H, Lutz B, Rein T. Cross-talk of vitamin d and glucocorticoids in hippocampal cells. J Neurochem 2006;96:500–509 [DOI] [PubMed] [Google Scholar]
- 48.Zhou C, Assem M, Tay JC, Watkins PB, Blumberg B, Schuetz EG, Thummel KE. Steroid and xenobiotic receptor and vitamin d receptor crosstalk mediates cyp24 expression and drug-induced osteomalacia. J Clin Invest 2006;116:1703–1712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Alimirah F, Peng X, Yuan L, Mehta RR, von Knethen A, Choubey D, Mehta RG. Crosstalk between the peroxisome proliferator-activated receptor gamma (ppargamma) and the vitamin d receptor (vdr) in human breast cancer cells: Ppargamma binds to vdr and inhibits 1alpha,25-dihydroxyvitamin d3 mediated transactivation. Exp Cell Res 2012;318:2490–2497 [DOI] [PubMed] [Google Scholar]
- 50.Shamsuzzaman S, Onal M, St John HC, Jeffery JJ, Pike JW. Absence of the vitamin d receptor inhibits atherosclerotic plaque calcification in female hypercholesterolemic mice. J Cell Biochem 2017;118:1050–1064 [DOI] [PubMed] [Google Scholar]
- 51.Tukaj C, Trzonkowski P, Kubasik-Juraniec J, Mysliwski A. Quantifying division of aortal smooth muscle cells in culture stimulated by 1,25(oh)2d3. J Steroid Biochem Mol Biol 2007;103:525–528 [DOI] [PubMed] [Google Scholar]
- 52.Parker BD, Schurgers LJ, Brandenburg VM, Christenson RH, Vermeer C, Ketteler M, Shlipak MG, Whooley MA, Ix JH. The associations of fibroblast growth factor 23 and uncarboxylated matrix gla protein with mortality in coronary artery disease: The heart and soul study. Ann Intern Med 2010;152:640–648 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Reid IR, Horne AM, Mihov B, Gamble GD, Al-Abuwsi F, Singh M, Taylor L, Fenwick S, Camargo CA, Stewart AW, Scragg R. Effect of monthly high-dose vitamin d on bone density in community-dwelling older adults substudy of a randomized controlled trial. J Intern Med 2017;282:452–460 [DOI] [PubMed] [Google Scholar]
- 54.Tintut Y, Parhami F, Bostrom K, Jackson SM, Demer LL. Camp stimulates osteoblast-like differentiation of calcifying vascular cells. Potential signaling pathway for vascular calcification. J Biol Chem 1998;273:7547–7553 [DOI] [PubMed] [Google Scholar]
- 55.Jono S, Nishizawa Y, Shioi A, Morii H. 1,25-dihydroxyvitamin d3 increases in vitro vascular calcification by modulating secretion of endogenous parathyroid hormone-related peptide. Circulation 1998;98:1302–1306 [DOI] [PubMed] [Google Scholar]
- 56.Price PA, Buckley JR, Williamson MK. The amino bisphosphonate ibandronate prevents vitamin d toxicity and inhibits vitamin d-induced calcification of arteries, cartilage, lungs and kidneys in rats. J Nutr 2001;131:2910–2915 [DOI] [PubMed] [Google Scholar]
- 57.Shi Y, Lu W, Hou Y, Fu K, Gan F, Liu J. Fibroblast growth factor 21 ameliorates vascular calcification by inhibiting osteogenic transition in vitamin d3 plus nicotine-treated rats. Biochem Biophys Res Commun 2018;495:2448–2455 [DOI] [PubMed] [Google Scholar]
- 58.Torremade N, Bozic M, Panizo S, Barrio-Vazquez S, Fernandez-Martin JL, Encinas M, Goltzman D, Arcidiacono MV, Fernandez E, Valdivielso JM. Vascular calcification induced by chronic kidney disease is mediated by an increase of 1alpha-hydroxylase expression in vascular smooth muscle cells. J Bone Miner Res 2016;31:1865–1876 [DOI] [PubMed] [Google Scholar]
- 59.Colotta F, Jansson B, Bonelli F. Modulation of inflammatory and immune responses by vitamin d. J Autoimmun 2017;85:78–97 [DOI] [PubMed] [Google Scholar]
- 60.Hollis BW, Johnson D, Hulsey TC, Ebeling M, Wagner CL. Vitamin d supplementation during pregnancy: Double-blind, randomized clinical trial of safety and effectiveness. J Bone Miner Res 2011;26:2341–2357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hollis BW, Wagner CL. Clinical review: The role of the parent compound vitamin d with respect to metabolism and function: Why clinical dose intervals can affect clinical outcomes. J Clin Endocrinol Metab 2013;98:4619–4628 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Committee IoM. Dietary reference intakes for calcium and vitamin d 2011 [PubMed] [Google Scholar]
- 63.Davies H Coronary heart disease: The significance of coronary pathology in infancy and the role of mitogens such as vitamin d. Med Hypotheses 1989;30:179–185 [DOI] [PubMed] [Google Scholar]
- 64.Manson JE, Bassuk SS, Lee IM, Cook NR, Albert MA, Gordon D, Zaharris E, Macfadyen JG, Danielson E, Lin J, Zhang SM, Buring JE. The vitamin d and omega-3 trial (vital): Rationale and design of a large randomized controlled trial of vitamin d and marine omega-3 fatty acid supplements for the primary prevention of cancer and cardiovascular disease. Contemp Clin Trials 2012;33:159–171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Taussig HB. Possible injury to the cardiovascular system from vitamin d. Ann Intern Med 1966;65:1195–1200 [DOI] [PubMed] [Google Scholar]
- 66.Hosseinpour F, Ibranovic I, Tang W, Wikvall K. 25-hydroxylation of vitamin d3 in primary cultures of pig hepatocytes: Evidence for a role of both cyp2d25 and cyp27a1. Biochem Biophys Res Commun 2003;303:877–883 [DOI] [PubMed] [Google Scholar]
- 67.Chen ML, Boltz MA, Armbrecht HJ. Effects of 1,25-dihydroxyvitamin d3 and phorbol ester on 25-hydroxyvitamin d3 24-hydroxylase cytochrome p450 messenger ribonucleic acid levels in primary cultures of rat renal cells. Endocrinology 1993;132:1782–1788 [DOI] [PubMed] [Google Scholar]
- 68.Zehnder D, Bland R, Chana RS, Wheeler DC, Howie AJ, Williams MC, Stewart PM, Hewison M. Synthesis of 1,25-dihydroxyvitamin d(3) by human endothelial cells is regulated by inflammatory cytokines: A novel autocrine determinant of vascular cell adhesion. J Am Soc Nephrol 2002;13:621–629 [DOI] [PubMed] [Google Scholar]
- 69.Somjen D, Weisman Y, Kohen F, Gayer B, Limor R, Sharon O, Jaccard N, Knoll E, Stern N. 25-hydroxyvitamin d3–1alpha-hydroxylase is expressed in human vascular smooth muscle cells and is upregulated by parathyroid hormone and estrogenic compounds. Circulation 2005;111:1666–1671 [DOI] [PubMed] [Google Scholar]
- 70.Stoffels K, Overbergh L, Giulietti A, Verlinden L, Bouillon R, Mathieu C. Immune regulation of 25-hydroxyvitamin-d3–1alpha-hydroxylase in human monocytes. J Bone Miner Res 2006;21:37–47 [DOI] [PubMed] [Google Scholar]
- 71.Girgis CM, Clifton-Bligh RJ, Mokbel N, Cheng K, Gunton JE. Vitamin d signaling regulates proliferation, differentiation, and myotube size in c2c12 skeletal muscle cells. Endocrinology 2014;155:347–357 [DOI] [PubMed] [Google Scholar]
- 72.Prentice A Vitamin d deficiency: A global perspective. Nutr Rev 2008;66:S153–164 [DOI] [PubMed] [Google Scholar]
- 73.Long GG. Acute toxicosis in swine associated with excessive dietary intake of vitamin d. J Am Vet Med Assoc 1984;184:164–170 [PubMed] [Google Scholar]
- 74.Szeto FL, Reardon CA, Yoon D, Wang Y, Wong KE, Chen Y, Kong J, Liu SQ, Thadhani R, Getz GS, Li YC. Vitamin d receptor signaling inhibits atherosclerosis in mice. Mol Endocrinol 2012;26:1091–1101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Chen S, Law CS, Grigsby CL, Olsen K, Hong TT, Zhang Y, Yeghiazarians Y, Gardner DG. Cardiomyocyte-specific deletion of the vitamin d receptor gene results in cardiac hypertrophy. Circulation 2011;124:1838–1847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Aihara K, Azuma H, Akaike M, Ikeda Y, Yamashita M, Sudo T, Hayashi H, Yamada Y, Endoh F, Fujimura M, Yoshida T, Yamaguchi H, Hashizume S, Kato M, Yoshimura K, Yamamoto Y, Kato S, Matsumoto T. Disruption of nuclear vitamin d receptor gene causes enhanced thrombogenicity in mice. J Biol Chem 2004;279:35798–35802 [DOI] [PubMed] [Google Scholar]
- 77.Suda T, Masuyama R, Bouillon R, Carmeliet G. Physiological functions of vitamin d: What we have learned from global and conditional vdr knockout mouse studies. Curr Opin Pharmacol 2015;22:87–99 [DOI] [PubMed] [Google Scholar]
- 78.Norman P, Moss I, Sian M, Gosling M, Powell J. Maternal and postnatal vitamin d ingestion influences rat aortic structure, function and elastin content. Cardiovasc Res 2002;55:369–374 [DOI] [PubMed] [Google Scholar]
- 79.Jiang W, Miyamoto T, Kakizawa T, Nishio SI, Oiwa A, Takeda T, Suzuki S, Hashizume K. Inhibition of lxralpha signaling by vitamin d receptor: Possible role of vdr in bile acid synthesis. Biochem Biophys Res Commun 2006;351:176–184 [DOI] [PubMed] [Google Scholar]
- 80.Shimosawa T, Ando K, Fujita T. Enhancement of vasoconstrictor response by a noncalcemic analogue of vitamin d3. Hypertension 1993;21:253–258 [DOI] [PubMed] [Google Scholar]
- 81.Kang EJ, Lee JE, An SM, Lee JH, Kwon HS, Kim BC, Kim SJ, Kim JM, Hwang DY, Jung YJ, Yang SY, Kim SC, An BS. The effects of vitamin d3 on lipogenesis in the liver and adipose tissue of pregnant rats. Int J Mol Med 2015;36:1151–1158 [DOI] [PubMed] [Google Scholar]
- 82.Karadag C, Yoldemir T, Yavuz DG. Effects of vitamin d supplementation on insulin sensitivity and androgen levels in vitamin-d-deficient polycystic ovary syndrome patients. J Obstet Gynaecol Res 2018;44:270–277 [DOI] [PubMed] [Google Scholar]
- 83.Wong MS, Delansorne R, Man RY, Vanhoutte PM. Vitamin d derivatives acutely reduce endothelium-dependent contractions in the aorta of the spontaneously hypertensive rat. Am J Physiol Heart Circ Physiol 2008;295:H289–296 [DOI] [PubMed] [Google Scholar]
- 84.Sooy K, Sabbagh Y, Demay MB. Osteoblasts lacking the vitamin d receptor display enhanced osteogenic potential in vitro. J Cell Biochem 2005;94:81–87 [DOI] [PubMed] [Google Scholar]
- 85.Takasu H, Sugita A, Uchiyama Y, Katagiri N, Okazaki M, Ogata E, Ikeda K. C-fos protein as a target of anti-osteoclastogenic action of vitamin d, and synthesis of new analogs. J Clin Invest 2006;116:528–535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Tsonis PA. 1,25-dihydroxyvitamin d3 stimulates chondrogenesis of the chick limb bud mesenchymal cells. Dev Biol 1991;143:130–134 [DOI] [PubMed] [Google Scholar]
- 87.Wagatsuma A, Sakuma K. Vitamin d signaling in myogenesis: Potential for treatment of sarcopenia. Biomed Res Int 2014;2014:121254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Blumberg JM, Tzameli I, Astapova I, Lam FS, Flier JS, Hollenberg AN. Complex role of the vitamin d receptor and its ligand in adipogenesis in 3t3-l1 cells. J Biol Chem 2006;281:11205–11213 [DOI] [PubMed] [Google Scholar]
- 89.Bunce CM, Brown G, Hewison M. Vitamin d and hematopoiesis. Trends Endocrinol Metab 1997;8:245–251 [DOI] [PubMed] [Google Scholar]
- 90.Chabas JF, Stephan D, Marqueste T, Garcia S, Lavaut MN, Nguyen C, Legre R, Khrestchatisky M, Decherchi P, Feron F. Cholecalciferol (vitamin d(3)) improves myelination and recovery after nerve injury. PLoS One 2013;8:e65034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Wu-Wong JR, Nakane M, Ma J, Ruan X, Kroeger PE. Effects of vitamin d analogs on gene expression profiling in human coronary artery smooth muscle cells. Atherosclerosis 2006;186:20–28 [DOI] [PubMed] [Google Scholar]
- 92.Rebsamen MC, Sun J, Norman AW, Liao JK. 1alpha,25-dihydroxyvitamin d3 induces vascular smooth muscle cell migration via activation of phosphatidylinositol 3-kinase. Circ Res 2002;91:17–24 [DOI] [PubMed] [Google Scholar]
- 93.Bukoski RD, DeWan P, McCarron DA. 1,25 (oh)2 vitamin d3 modifies growth and contractile function of vascular smooth muscle of spontaneously hypertensive rats. Am J Hypertens 1989;2:553–556 [DOI] [PubMed] [Google Scholar]
- 94.Shan NL, Wahler J, Lee HJ, Bak MJ, Gupta SD, Maehr H, Suh N. Vitamin d compounds inhibit cancer stem-like cells and induce differentiation in triple negative breast cancer. J Steroid Biochem Mol Biol 2017;173:122–129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Oh J, Weng S, Felton SK, Bhandare S, Riek A, Butler B, Proctor BM, Petty M, Chen Z, Schechtman KB, Bernal-Mizrachi L, Bernal-Mizrachi C. 1,25(oh)2 vitamin d inhibits foam cell formation and suppresses macrophage cholesterol uptake in patients with type 2 diabetes mellitus. Circulation 2009;120:687–698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Correale J, Ysrraelit MC, Gaitan MI. Vitamin d-mediated immune regulation in multiple sclerosis. J Neurol Sci 2011;311:23–31 [DOI] [PubMed] [Google Scholar]
- 97.Wagner KD, Wagner N, Sukhatme VP, Scholz H. Activation of vitamin d receptor by the wilms’ tumor gene product mediates apoptosis of renal cells. J Am Soc Nephrol 2001;12:1188–1196 [DOI] [PubMed] [Google Scholar]
- 98.Marie PJ, Connes D, Hott M, Miravet L. Comparative effects of a novel vitamin d analogue mc-903 and 1,25-dihydroxyvitamin d3 on alkaline phosphatase activity, osteocalcin and DNA synthesis by human osteoblastic cells in culture. Bone 1990;11:171–179 [DOI] [PubMed] [Google Scholar]
- 99.Schwartz Z, Swain LD, Ramirez V, Boyan BD. Regulation of arachidonic acid turnover by 1,25-(oh)2d3 and 24,25-(oh)2d3 in growth zone and resting zone chondrocyte cultures. Biochim Biophys Acta 1990;1027:278–286 [DOI] [PubMed] [Google Scholar]
- 100.Grosse B, Bourdeau A, Lieberherr M. Oscillations in inositol 1,4,5-trisphosphate and diacyglycerol induced by vitamin d3 metabolites in confluent mouse osteoblasts. J Bone Miner Res 1993;8:1059–1069 [DOI] [PubMed] [Google Scholar]
- 101.Liu X, Nelson A, Wang X, Farid M, Gunji Y, Ikari J, Iwasawa S, Basma H, Feghali-Bostwick C, Rennard SI. Vitamin d modulates prostaglandin e2 synthesis and degradation in human lung fibroblasts. Am J Respir Cell Mol Biol 2014;50:40–50 [DOI] [PubMed] [Google Scholar]
- 102.Schwartz Z, Swain LD, Kelly DW, Brooks B, Boyan BD. Regulation of prostaglandin e2 production by vitamin d metabolites in growth zone and resting zone chondrocyte cultures is dependent on cell maturation. Bone 1992;13:395–401 [DOI] [PubMed] [Google Scholar]
- 103.Polidoro L, Properzi G, Marampon F, Gravina GL, Festuccia C, Di Cesare E, Scarsella L, Ciccarelli C, Zani BM, Ferri C. Vitamin d protects human endothelial cells from h(2)o(2) oxidant injury through the mek/erk-sirt1 axis activation. J Cardiovasc Transl Res 2013;6:221–231 [DOI] [PubMed] [Google Scholar]
- 104.Andrukhova O, Slavic S, Zeitz U, Riesen SC, Heppelmann MS, Ambrisko TD, Markovic M, Kuebler WM, Erben RG. Vitamin d is a regulator of endothelial nitric oxide synthase and arterial stiffness in mice. Mol Endocrinol 2014;28:53–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Bissonnette M, Tien XY, Niedziela SM, Hartmann SC, Frawley BP Jr., Roy HK, Sitrin MD, Perlman RL, Brasitus TA. 1,25(oh)2 vitamin d3 activates pkc-alpha in caco-2 cells: A mechanism to limit secosteroid-induced rise in [ca2+]i. Am J Physiol 1994;267:G465–475 [DOI] [PubMed] [Google Scholar]
- 106.Berg JP, Haug E. Vitamin d: A hormonal regulator of the camp signaling pathway. Crit Rev Biochem Mol Biol 1999;34:315–323 [DOI] [PubMed] [Google Scholar]
- 107.Pardo VG, Boland R, de Boland AR. 1alpha,25(oh)(2)-vitamin d(3) stimulates intestinal cell p38 mapk activity and increases c-fos expression. Int J Biochem Cell Biol 2006;38:1181–1190 [DOI] [PubMed] [Google Scholar]
- 108.Piek E, Sleumer LS, van Someren EP, Heuver L, de Haan JR, de Grijs I, Gilissen C, Hendriks JM, van Ravestein-van Os RI, Bauerschmidt S, Dechering KJ, van Zoelen EJ. Osteo-transcriptomics of human mesenchymal stem cells: Accelerated gene expression and osteoblast differentiation induced by vitamin d reveals c-myc as an enhancer of bmp2-induced osteogenesis. Bone 2010;46:613–627 [DOI] [PubMed] [Google Scholar]
- 109.Towers TL, Staeva TP, Freedman LP. A two-hit mechanism for vitamin d3-mediated transcriptional repression of the granulocyte-macrophage colony-stimulating factor gene: Vitamin d receptor competes for DNA binding with nfat1 and stabilizes c-jun. Mol Cell Biol 1999;19:4191–4199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Fretz JA, Zella LA, Kim S, Shevde NK, Pike JW. 1,25-dihydroxyvitamin d3 regulates the expression of low-density lipoprotein receptor-related protein 5 via deoxyribonucleic acid sequence elements located downstream of the start site of transcription. Mol Endocrinol 2006;20:2215–2230 [DOI] [PubMed] [Google Scholar]
- 111.Chen Y, Zhang J, Ge X, Du J, Deb DK, Li YC. Vitamin d receptor inhibits nuclear factor kappab activation by interacting with ikappab kinase beta protein. J Biol Chem 2013;288:19450–19458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Okamura M, Takano Y, Saito Y, Yao J, Kitamura M. Induction of nephrin gene expression by selective cooperation of the retinoic acid receptor and the vitamin d receptor. Nephrol Dial Transplant 2009;24:3006–3012 [DOI] [PubMed] [Google Scholar]
- 113.Zou A, Elgort MG, Allegretto EA. Retinoid x receptor (rxr) ligands activate the human 25-hydroxyvitamin d3–24-hydroxylase promoter via rxr heterodimer binding to two vitamin d-responsive elements and elicit additive effects with 1,25-dihydroxyvitamin d3. J Biol Chem 1997;272:19027–19034 [DOI] [PubMed] [Google Scholar]
- 114.Zhang Y, Leung DY, Goleva E. Vitamin d enhances glucocorticoid action in human monocytes: Involvement of granulocyte-macrophage colony-stimulating factor and mediator complex subunit 14. J Biol Chem 2013;288:14544–14553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Paredes R, Arriagada G, Cruzat F, Olate J, Van Wijnen A, Lian J, Stein G, Stein J, Montecino M. The runx2 transcription factor plays a key role in the 1alpha,25-dihydroxy vitamin d3-dependent upregulation of the rat osteocalcin (oc) gene expression in osteoblastic cells. J Steroid Biochem Mol Biol 2004;89-90:269–271 [DOI] [PubMed] [Google Scholar]
- 116.Rohe B, Safford SE, Nemere I, Farach-Carson MC. Regulation of expression of 1,25d3-marrs/erp57/pdia3 in rat iec-6 cells by tgf beta and 1,25(oh)2d3. Steroids 2007;72:144–150 [DOI] [PubMed] [Google Scholar]
- 117.Schedlich LJ, Muthukaruppan A, O’Han MK, Baxter RC. Insulin-like growth factor binding protein-5 interacts with the vitamin d receptor and modulates the vitamin d response in osteoblasts. Mol Endocrinol 2007;21:2378–2390 [DOI] [PubMed] [Google Scholar]
- 118.Miyake N, Hoshi K, Sano Y, Kikuchi K, Tadano K, Koshihara Y. 1,25-dihydroxyvitamin d3 promotes vitamin k2 metabolism in human osteoblasts. Osteoporos Int 2001;12:680–687 [DOI] [PubMed] [Google Scholar]
- 119.Cheema C, Grant BF, Marcus R. Effects of estrogen on circulating “free” and total 1,25-dihydroxyvitamin d and on the parathyroid-vitamin d axis in postmenopausal women. J Clin Invest 1989;83:537–542 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Ponda MP, Dowd K, Finkielstein D, Holt PR, Breslow JL. The short-term effects of vitamin d repletion on cholesterol: A randomized, placebo-controlled trial. Arterioscler Thromb Vasc Biol 2012;32:2510–2515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Fu B, Wang H, Wang J, Barouhas I, Liu W, Shuboy A, Bushinsky DA, Zhou D, Favus MJ. Epigenetic regulation of bmp2 by 1,25-dihydroxyvitamin d3 through DNA methylation and histone modification. PLoS One 2013;8:e61423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kim HS, Zheng M, Kim DK, Lee WP, Yu SJ, Kim BO. Effects of 1,25-dihydroxyvitamin d3 on the differentiation of mc3t3-e1 osteoblast-like cells. J Periodontal Implant Sci 2018;48:34–46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Sodek J, Li JJ, Kim RH, Ogata Y, Yamauchi M. Characterization of the bone sialoprotein (bsp) gene promoter. Connect Tissue Res 1996;35:23–31 [DOI] [PubMed] [Google Scholar]
- 124.Kuroki Y, Shiozawa S, Kano J, Chihara K. Competition between c-fos and 1,25(oh)2 vitamin d3 in the transcriptional control of type i collagen synthesis in mc3t3-e1 osteoblastic cells. J Cell Physiol 1995;164:459–464 [DOI] [PubMed] [Google Scholar]
- 125.Merchiers P, Bulens F, Stockmans I, De Vriese A, Convents R, Bouillon R, Collen D, Belayew A, Carmeliet G. 1,25-dihydroxyvitamin d(3) induction of the tissue-type plasminogen activator gene is mediated through its multihormone-responsive enhancer. FEBS Lett 1999;460:289–296 [DOI] [PubMed] [Google Scholar]
- 126.Liu SM, Koszewski N, Lupez M, Malluche HH, Olivera A, Russell J. Characterization of a response element in the 5’-flanking region of the avian (chicken) pth gene that mediates negative regulation of gene transcription by 1,25-dihydroxyvitamin d3 and binds the vitamin d3 receptor. Mol Endocrinol 1996;10:206–215 [DOI] [PubMed] [Google Scholar]
- 127.Tang WJ, Wang LF, Xu XY, Zhou Y, Jin WF, Wang HF, Gao J. Autocrine/paracrine action of vitamin d on fgf23 expression in cultured rat osteoblasts. Calcif Tissue Int 2010;86:404–410 [DOI] [PubMed] [Google Scholar]
- 128.Kim S, Yamazaki M, Zella LA, Meyer MB, Fretz JA, Shevde NK, Pike JW. Multiple enhancer regions located at significant distances upstream of the transcriptional start site mediate rankl gene expression in response to 1,25-dihydroxyvitamin d3. J Steroid Biochem Mol Biol 2007;103:430–434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Hodgkinson JE, Davidson CL, Beresford J, Sharpe PT. Expression of a human homeobox-containing gene is regulated by 1,25(oh)2d3 in bone cells. Biochim Biophys Acta 1993;1174:11–16 [DOI] [PubMed] [Google Scholar]
- 130.Lin YM, Sun HY, Chiu WT, Su HC, Chien YC, Chong LW, Chang HC, Bai CH, Young KC, Tsao CW. Calcitriol inhibits hcv infection via blockade of activation of ppar and interference with endoplasmic reticulum-associated degradation. Viruses 2018;10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Wietrzyk J, Filip B, Milczarek M, Klopotowska D, Maciejewska M, Dabrowska K, Kurzepa A, Dzimira S, Madej J, Kutner A. The influence of 1,25-dihydroxyvitamin d3 and 1,24-dihydroxyvitamin d3 on alphavbeta3 integrin expression in cancer cell lines. Oncol Rep 2008;20:941–952 [PubMed] [Google Scholar]
- 132.Polly P, Carlberg C, Eisman JA, Morrison NA. Identification of a vitamin d3 response element in the fibronectin gene that is bound by a vitamin d3 receptor homodimer. J Cell Biochem 1996;60:322–333 [DOI] [PubMed] [Google Scholar]
- 133.Kuhne H, Schutkowski A, Weinholz S, Cordes C, Schierhorn A, Schulz K, Konig B, Stangl GI. Vitamin d receptor regulates intestinal proteins involved in cell proliferation, migration and stress response. Lipids Health Dis 2014;13:51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Lacraz S, Dayer JM, Nicod L, Welgus HG. 1,25-dihydroxyvitamin d3 dissociates production of interstitial collagenase and 92-kda gelatinase in human mononuclear phagocytes. J Biol Chem 1994;269:6485–6490 [PubMed] [Google Scholar]
- 135.Miyashita M, Koga K, Izumi G, Sue F, Makabe T, Taguchi A, Nagai M, Urata Y, Takamura M, Harada M, Hirata T, Hirota Y, Wada-Hiraike O, Fujii T, Osuga Y. Effects of 1,25-dihydroxy vitamin d3 on endometriosis. J Clin Endocrinol Metab 2016;101:2371–2379 [DOI] [PubMed] [Google Scholar]
- 136.Song L, Papaioannou G, Zhao H, Luderer HF, Miller C, Dall’Osso C, Nazarian RM, Wagers AJ, Demay MB. The vitamin d receptor regulates tissue resident macrophage response to injury. Endocrinology 2016;157:4066–4075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Hakim I, Bar-Shavit Z. Modulation of tnf-alpha expression in bone marrow macrophages: Involvement of vitamin d response element. J Cell Biochem 2003;88:986–998 [DOI] [PubMed] [Google Scholar]
- 138.Tang X, Pan Y, Zhao Y. Vitamin d inhibits the expression of interleukin-8 in human periodontal ligament cells stimulated with porphyromonas gingivalis. Arch Oral Biol 2013;58:397–407 [DOI] [PubMed] [Google Scholar]
- 139.Maestro B, Davila N, Carranza MC, Calle C. Identification of a vitamin d response element in the human insulin receptor gene promoter. J Steroid Biochem Mol Biol 2003;84:223–230 [DOI] [PubMed] [Google Scholar]
- 140.Anisiewicz A, Pawlik A, Filip-Psurska B, Turlej E, Dzimira S, Milczarek M, Gdesz K, Papiernik D, Jarosz J, Klopotowska D, Kutner A, Mazur A, Wietrzyk J. Unfavorable effect of calcitriol and its low-calcemic analogs on metastasis of 4t1 mouse mammary gland cancer. Int J Oncol 2018;52:103–126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Cornet A, Baudet C, Neveu I, Baron-Van Evercooren A, Brachet P, Naveilhan P. 1,25-dihydroxyvitamin d3 regulates the expression of vdr and ngf gene in schwann cells in vitro. J Neurosci Res 1998;53:742–746 [DOI] [PubMed] [Google Scholar]
- 142.Chen S, Ni XP, Humphreys MH, Gardner DG. 1,25 dihydroxyvitamin d amplifies type a natriuretic peptide receptor expression and activity in target cells. J Am Soc Nephrol 2005;16:329–339 [DOI] [PubMed] [Google Scholar]
- 143.Krishnan AV, Swami S, Peng L, Wang J, Moreno J, Feldman D. Tissue-selective regulation of aromatase expression by calcitriol: Implications for breast cancer therapy. Endocrinology 2010;151:32–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Quach HP, Noh K, Hoi SY, Bruinsma A, Groothuis GMM, Li AP, Chow ECY, Pang KS. Alterations in gene expression in vitamin d-deficiency: Down-regulation of liver cyp7a1 and renal oat3 in mice. Biopharm Drug Dispos 2018;39:99–115 [DOI] [PubMed] [Google Scholar]
- 145.Meyer MB, Goetsch PD, Pike JW. A downstream intergenic cluster of regulatory enhancers contributes to the induction of cyp24a1 expression by 1alpha,25-dihydroxyvitamin d3. J Biol Chem 2010;285:15599–15610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Hahn CN, Kerry DM, Omdahl JL, May BK. Identification of a vitamin d responsive element in the promoter of the rat cytochrome p450(24) gene. Nucleic Acids Res 1994;22:2410–2416 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Malloy PJ, Peng L, Wang J, Feldman D. Interaction of the vitamin d receptor with a vitamin d response element in the mullerian-inhibiting substance (mis) promoter: Regulation of mis expression by calcitriol in prostate cancer cells. Endocrinology 2009;150:1580–1587 [DOI] [PMC free article] [PubMed] [Google Scholar]


