Abstract
Background
Uremic pruritus is a common and frustrating symptom among patients receiving peritoneal dialysis (PD). This study aimed to examine the prognostic importance of uremic pruritus and to identify the determinants for higher pruritus intensity in PD patients.
Methods
We conducted a prospective cohort study of patients receiving maintenance PD. A visual analogue scale (VAS) score was used to measure the intensity of uremic pruritus. The composite endpoint of PD technique failure or all-cause death was assessed using a multivariable Cox proportional hazards model. The determinants for the VAS score of uremic pruritus was assessed using a multivariable linear regression model.
Results
Among the 85 PD patients, 24 (28%) had uremic pruritus. During a median follow-up of 28.0 months, 12 patients experienced technique failure, and 7 died. We found that a higher VAS score of pruritus intensity was an independent risk factor for technique failure or death (hazard ratio, 1.64; 95% confidence interval, 1.18 to 2.28; P = 0.003) after adjusting for a variety of confounding factors. We also found that a weekly total Kt/V of less than 1.88, a longer duration of dialysis, a higher dietary protein intake, and higher blood levels of intact parathyroid hormone and high-sensitivity C-reactive protein were independent determinants of higher VAS scores of pruritus intensity.
Conclusions
Our results show that uremic pruritus is an independent risk factor of technique failure and death in patients receiving PD. We also found that a weekly total Kt/V < 1.88 is associated with higher intensity of uremic pruritus in PD patients.
Introduction
Uremic pruritus is one of the most common and frustrating symptoms among patients with end-stage renal disease (ESRD), with prevalence ranging from 10% to 70% among patients receiving peritoneal dialysis (PD) [1,2]. Patients with pruritus suffer from sleep disturbance, depression, anxiety, uncontrollable itching, and impaired quality of life [3,4]. In patients receiving hemodialysis (HD), divalent ions, calcium-phosphate products, hyperparathyroidism, and interleukin-31 have been reported to be associated with uremic pruritus [5–9]. However, the pathophysiology of uremic pruritus remains obscure, and the therapeutic options are limited and unsatisfactory to PD patients.
Our previous study showed that the dialysis modality affects the prevalence and severity of uremic pruritus [10], suggesting that the pathogenesis of pruritus may differ between patients receiving PD and those on HD. Many predictive factors of uremic pruritus, such as age, comorbid diseases, nutritional status, solute clearance, and peritoneal transport properties, affect outcome in dialysis patients, and some factors are distinct in PD patients [11–16]. Although uremic pruritus is associated with mortality in patients receiving HD [9,17], the impact of uremic pruritus on technique failure and mortality in PD patients has never been investigated. Moreover, the relationships between uremic pruritus, biochemical parameters, and dialysis adequacy in patients receiving PD have rarely been reported.
Here, we report a prospective cohort study that examined the prognostic importance of uremic pruritus in patients receiving PD, and also identified the determinants for higher pruritus intensity in the cross-sectional part of the study.
Materials and methods
Study participants
We conducted a prospective cohort study of patients receiving maintenance PD at the Far Eastern Memorial Hospital, a tertiary medical center in Taiwan. There were 98 patients receiving maintenance PD. Patients were excluded if they were aged less than 20 years, had communication problems, were diagnosed with other primary skin disorders, or refused to participate in the study. We also excluded those with an active infection, an active malignancy, a psychotic illness, or a pregnancy. After excluding 12 patients who refused to participate and 1 patient with a psychotic illness, a total of 85 patients were enrolled and assessed in May 2013, and followed until August 2015.
Ethics
This study protocol complied with the Declaration of Helsinki. The Institutional Review Board of the Far Eastern Memorial Hospital, New Taipei City, Taiwan, approved this prospective cohort study, and all participants provided written informed consent.
Patient characteristics
Patient data, including age, sex, body mass index (BMI), comorbid diseases, etiology of ESRD, results of blood laboratory tests, PD assessments, and duration of dialysis therapy, were recorded for each participant. Systolic and diastolic blood pressure levels were measured during routine visits. Hypertension was defined as a systolic blood pressure ≥ 140 mmHg and/or a diastolic blood pressure ≥ 90 mmHg [18,19]. BMI was calculated using the following formula: [weight (kg) / height (m)2 ]. Venous blood was sampled after an overnight fast exceeding 8 hours. Patients with a positive test result for hepatitis B virus surface antigen were considered hepatitis B carriers, and those with a positive result for hepatitis C virus antibody were considered hepatitis C carriers. All laboratory tests were performed by the hospital’s central laboratory, and auto-analyzers were used to determine the biochemical data.
Pruritus assessment
The patients were considered to have uremic pruritus if they had (1) at least three episodes of pruritus during a period of less than 2 weeks, with the symptoms occurring a few times a day, lasting at least a few minutes, and troubling the patient; or (2) the regular occurrence of pruritus during a period of 6 months but with a lower frequency than in (1) [2,20]. A visual analogue scale (VAS) score, which is a 10-cm line on which patients indicate the intensity of pruritus by marking the line at the point that corresponds to the severity of their pruritus, was reported from 0 to 10 (0 = no itching, 10 = worst imaginable itching) [21].
Dialysis adequacy of solute clearance
The PD regimen and modality for each patient were evaluated and prescribed in the PD unit during monthly follow-ups. Dialysis solute clearance was assessed based on the Kt/V parameter (amount of dialysis delivered: K = clearance of urea, t = time on dialysis, V = estimated total body water) using urea kinetic modeling. The adequacy of solute clearance was assessed based on the weekly total Kt/V (the sum of peritoneal Kt/V and renal Kt/V) and should be at least 1.7 or above [22,23]. The transport properties of the peritoneal membrane were determined using the standard peritoneal equilibration test, which measures net fluid removal and the ratio of dialysate creatinine to blood creatinine after 4 hours [24]. Dietary protein intake was assessed based on protein nitrogen appearance and was normalized to the ideal body weight [25].
Outcomes
This study had two aims. Firstly, a prospective cohort study design was used to examine the prognostic importance of VAS score of uremic pruritus. Secondly, a cross-sectional study design was used to explore the determinants associated with higher pruritus intensity based on the clinical and biochemical parameters of participants and their pruritus assessments at baseline. The status of the patients was recorded as permanent transfer to hemodialysis, death, renal transplantation, transfer to other hospitals, or no change in treatment status as of the last recorded follow-up date. Permanent transfer to hemodialysis was considered PD technique failure. The primary outcome was the composite endpoint of PD technique failure or all-cause death, and renal transplantation and hospital transfer were regarded as right-censoring events in the cohort study. The secondary outcome was the determinants for the VAS score of uremic pruritus using the cross-sectional baseline data.
Statistical analysis
Statistical analysis was performed using R 2.14.1 software (R Foundation for Statistical Computing, Vienna, Austria). The data are expressed as the mean ± standard deviation for normally distributed continuous variables, as the median (1st and 3rd quartiles) for non-normally distributed continuous variables, and as numbers (percentages) for categorical variables. Continuous data were tested for normality by the Shapiro–Wilk test. For the descriptive analysis, univariable analyses were conducted using the independent two-sample t-test, the Wilcoxon rank-sum test, and Pearson’s chi-square test, respectively. The Kaplan-Meier method and the log-rank test were used to compare event-free survival between patients with and without pruritus. Multivariable Cox proportional hazards models were used to estimate the hazard ratio (HR) and 95% confidence interval (CI) of the VAS score of uremic pruritus for the composite endpoint of technique failure or all-cause death. Multivariable linear regression analysis was conducted to identify the determinants for the VAS score of uremic pruritus.
To find the parsimonious regression models that fitted the observed data well for effect estimation and ensure a good quality of analysis, basic model-fitting techniques for (1) variable selection, (2) goodness-of-fit assessment, and (3) regression diagnostics were used in the regression analyses. Specifically, the stepwise variable selection procedure (with iterations between the forward and backward steps) was applied to obtain the best candidate final regression model. All the significant and non-significant covariates were put on the variable list to be selected. The significance levels for entry (SLE) and for stay (SLS) were set to 0.15 for being conservative. Then, with the aid of clinical evidence, the best candidate final regression model was identified by dropping the covariates with P value > 0.05 one at a time until all regression coefficients were significantly different from 0. Since the statistical testing at each step of the stepwise variable selection procedure was conditioning on the other covariates in the regression model, the multiple testing problem was not of concern.
The generalized additive models (GAMs) were fitted to detect nonlinear effects of continuous covariates and identify appropriate cutoff points for discretizing continuous covariates, if necessary, during the stepwise variable selection procedure. Computationally, the vgam() function (with the default values of smoothing parameters) of the VGAM package [26–28] was used to fit GAMs for continuous, binary, and count responses in R. Since GAMs were originally developed for smoothing the effects of continuous covariates in generalized linear models (GLMs), we fitted GAMs of binary responses for survival outcomes. Alternatively, we specified the smoothing option pspline (for the smoothing splines using a “p-spline” basis) inside the coxph() function of the survival package to smooth the effects of continuous covariates on the survival outcome of Cox’s proportional hazards models in R [29]. The concordance and the R2 were examined to assess the goodness-of-fit of the Cox proportional hazards model and the linear regression model, respectively. Statistical tools for regression diagnosis, including verification of proportional hazards assumption, residual analysis, the detection of influential cases, and test for multicollinearity by checking variance inflation factors, were used to assess problems within the model or data. A two-sided P value ≤ 0.05 was considered statistically significant.
Results
Patient characteristics
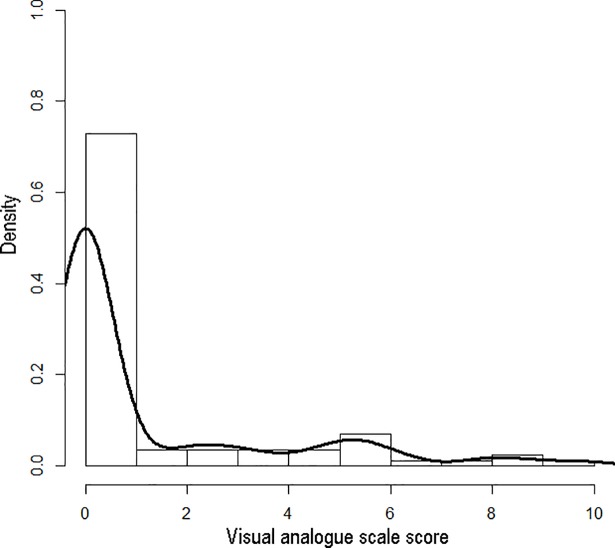
A total of 85 patients receiving maintenance PD completed this study. During a median follow-up period of 28.0 (1st quartile 27.5, 3rd quartile 28.0) months, 12 patients had technique failure, and 7 died. The baseline characteristics of the participants are summarized in Table 1. The study participants had a mean age of 55.5 years (65% were female) and a median duration of dialysis of 2.3 years. Among the study participants, 24 (28.2%) were classified as having uremic pruritus and had a median VAS score of 4.6. The distribution of the severity of uremic pruritus in the study participants are shown in Fig 1. Comparisons of the clinical and laboratory parameters of participants with uremic pruritus (VAS>0) and without uremic pruritus (VAS = 0) are shown in Table 2. Dietary protein intake was higher and the prevalence of diabetes mellitus was lower among patients with uremic pruritus.
Table 1. Demographic and clinical characteristics of the study participants.
| Baseline characteristics | Statistics | ||
|---|---|---|---|
| Participant number | 85 | ||
| Sex (female: male) | 55 (65%) | : | 30 (35%) |
| Age (year) | 55.5 | ± | 12.0 |
| Duration of dialysis (year) | 3.1 | ± | 2.7 |
| Etiology of end stage renal disease | |||
| Glomerulonephritis | 45 (52.9%) | ||
| Diabetes mellitus | 23 (27.1%) | ||
| Systemic lupus erythematosus | 5 (5.9%) | ||
| Hypertension | 2 (2.4%) | ||
| Interstitial nephritis | 2 (2.4%) | ||
| Polycystic kidney disease | 2 (2.4%) | ||
| Obstructive uropathy | 1 (1.2%) | ||
| Others | 5 (5.9%) | ||
The data are expressed as the mean ± standard deviation or the number (percentage).
Fig 1. Histogram and density plot of visual analogue scale score of pruritus intensity in the study population (n = 85).
The density of vertical axis represents the percentage of study participants.
Table 2. Demographic and clinical characteristics of participants with and without pruritus.
| Variable | With pruritus | Without pruritus | P value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Participant number | 24 | 61 | ||||||||||
| Visual analogue scale score of pruritus intensity | 4.6 | ( | 2.5, 5.9 | ) | 0.0 | ( | 0, 0 | ) | <0.001 | * | ||
| Age (year) | 57.1 | ± | 13.3 | 54.9 | ± | 11.5 | 0.46 | |||||
| Female | 18 | ( | 75.0% | ) | 37 | ( | 60.7% | ) | 0.21 | |||
| Duration of dialysis (year) | 2.7 | ( | 0.8, 5.9 | ) | 2.1 | ( | 1.1, 3.7 | ) | 0.63 | |||
| Body mass index (kg/m2) | 22.8 | ± | 4.2 | 24.2 | ± | 3.3 | 0.10 | |||||
| Diabetes mellitus | 2 | ( | 8.3% | ) | 21 | ( | 34.4% | ) | 0.02 | * | ||
| Heart failure | 1 | ( | 4.2% | ) | 5 | ( | 8.2% | ) | 0.51 | |||
| Viral hepatitis B | 1 | ( | 4.2% | ) | 7 | ( | 11.5% | ) | 0.30 | |||
| Viral hepatitis C | 2 | ( | 8.3% | ) | 3 | ( | 4.9% | ) | 0.55 | |||
| Daily urine amount (L/day) | 0.19 | ( | 0, 0.33 | ) | 0.40 | ( | 0, 0.70 | ) | 0.17 | |||
| Weekly total Kt/V | 2.11 | ± | 0.27 | 2.07 | ± | 0.31 | 0.60 | |||||
| Weekly renal Kt/V | 0.11 | ( | 0, 0.3 | ) | 0.15 | ( | 0, 0.4 | ) | 0.44 | |||
| Weekly peritoneal Kt/V | 1.91 | ± | 0.30 | 1.81 | ± | 0.28 | 0.12 | |||||
| Total creatinine clearance (L/week/1.73 m2) | 55.2 | ( | 52.9, 63.5 | ) | 58.0 | ( | 49.9, 68.7 | ) | 0.64 | |||
| Renal creatinine clearance (L/week/1.73 m2) | 6.8 | ( | 0, 23.1 | ) | 8.2 | ( | 0, 23.9 | ) | 0.69 | |||
| Peritoneal creatinine clearance (L/week/1.73 m2) | 49.7 | ± | 7.8 | 45.9 | ± | 8.5 | 0.06 | |||||
| Normalized protein nitrogen appearance (g/kg/day) | 1.01 | ( | 0.97, 1.27 | ) | 0.94 | ( | 0.81, 1.13 | ) | 0.05 | * | ||
| Hematocrit (%) | 30.5 | ± | 3.7 | 30.5 | ± | 4.2 | 1.00 | |||||
| Platelets (103/μL) | 219 | ± | 72 | 236 | ± | 69 | 0.32 | |||||
| Albumin (g/dL) | 3.7 | ± | 0.4 | 3.7 | ± | 0.5 | 0.60 | |||||
| Fasting glucose (mg/dL) | 103.5 | ( | 97.0, 118.0 | ) | 100.0 | ( | 90.0, 124.0 | ) | 0.64 | |||
| Total cholesterol (mg/dL) | 195.7 | ± | 58.7 | 185.8 | ± | 38.5 | 0.45 | |||||
| Triglycerides (mg/dL) | 129.0 | ( | 91.0, 227.5 | ) | 113.0 | ( | 74.0, 190.0 | ) | 0.39 | |||
| Creatinine (mg/dL) | 11.0 | ± | 3.8 | 11.9 | ± | 3.2 | 0.25 | |||||
| Uric acid (mg/dL) | 6.7 | ( | 5.5, 7.9 | ) | 6.6 | ( | 6.1, 7.5 | ) | 0.74 | |||
| Alanine transaminase (U/L) | 15.0 | ( | 10.0, 20.0 | ) | 14.0 | ( | 10.0, 19.0 | ) | 0.83 | |||
| Total bilirubin (mg/dL) | 0.2 | ( | 0.2, 0.3 | ) | 0.2 | ( | 0.2, 0.3 | ) | 0.64 | |||
| Ca × P** (mg/dL × mg/dL) | 50.1 | ± | 13.1 | 50.1 | ± | 12.0 | 1.00 | |||||
| Intact parathyroid hormone (pg/mL) | 251.2 | ( | 100.8, 792.7 | ) | 264.1 | ( | 122.5, 462.6 | ) | 0.72 | |||
| High-sensitivity C-reactive protein (mg/L) | 0.3 | ( | 0.1, 0.6 | ) | 0.3 | ( | 0.1, 0.7 | ) | 0.73 | |||
The data are expressed as the mean ± standard deviation for normally distributed continuous variables; as the median (first and third quartiles) for non-normally distributed continuous variables; and as the number (percentage) for categorical variables. P values are calculated using the two-sample t-test, the Wilcoxon rank-sum test, and the χ2 test, respectively.
*P ≤ 0.05.
** Ca × P = Product of albumin-adjusted serum calcium (Ca) and serum phosphorus (P) levels.
Prognostic importance of uremic pruritus
During follow-up, 19 patients (22.4%) had reached the composite endpoint of PD technique failure or all-cause death. The events among these patients included 6 technique failures and 1 death among those with uremic pruritus (14.8 events per 100 person-years) compared with 6 technique failures and 6 deaths among the patients without pruritus (9.8 events per 100 person-years). The Kaplan-Meier cumulative survival curve in S1 Fig show that compared with nonpruritic patients, those with pruritus showed a trend of lower event-free survival, but the difference did not reach statistical significance (P = 0.38).
The results of the multivariable Cox proportional hazards model are shown in Table 3. With the assistance of generalized additive models, we found that a higher VAS score of pruritus intensity was an independent risk factor for technique failure or death (HR, 1.64; 95% CI, 1.18 to 2.28; P = 0.003) after adjusting for daily urine amount, peritoneal solute clearance, peritoneal transport, body mass index, heart failure, fasting glucose, platelets, alanine transaminase, creatinine, and uric acid.
Table 3. Multivariable Cox proportional hazards model for the composite endpoint of technique failure or all-cause death.
| Hazard | 95% Confidence | |||||
|---|---|---|---|---|---|---|
| Covariate | Ratio | Interval | P Value | |||
| Visual analogue scale score of pruritus intensity | 1.64 | 1.18 | - | 2.28 | 0.003 | |
| Daily urine amount < 0.68 L/day | 659.10 | 30.14 | - | 14414.90 | < | 0.001 |
| Peritoneal creatinine clearance < 41.4 L/week/1.73 m2 | 43.70 | 6.76 | - | 282.67 | < | 0.001 |
| 4-hour dialysate/plasma creatinine > 0.736 | 33.12 | 4.17 | 262.75 | < | 0.001 | |
| Body mass index ≤ 24.4 kg/m2 | 66.88 | 6.56 | - | 681.85 | < | 0.001 |
| Heart Failure | 6.75 | 1.11 | - | 40.89 | 0.038 | |
| Fasting glucose (mg/dL) | 1.06 | 1.04 | - | 1.09 | < | 0.001 |
| Platelets < 210 103/μL | 6.10 | 1.81 | - | 20.58 | 0.004 | |
| Alanine transaminase (U/L) | 1.07 | 1.02 | - | 1.12 | 0.010 | |
| Creatinine < 11.1 mg/dL | 6.01 | 1.18 | - | 30.54 | 0.031 | |
| Uric acid ≥ 6.3 mg/dL | 24.53 | 3.28 | - | 184.07 | 0.002 | |
The Goodness-of-fit assessment, concordance = 0.905, indicates a high discrimination power.
Determinants of uremic pruritus
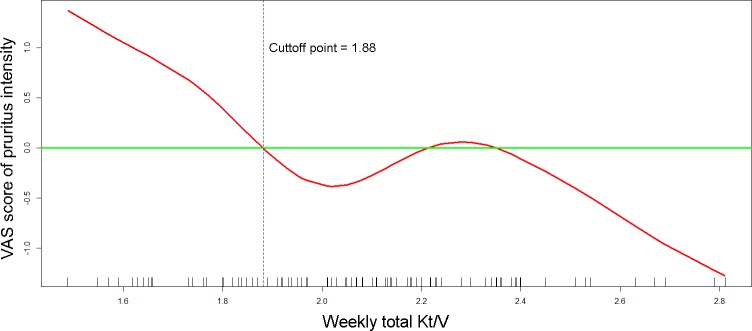
Table 4 shows the results of the multivariable linear regression analyses that were performed to identify the determinants associated with VAS scores of pruritus. The GAMs revealed nonlinear relationships between VAS scores of pruritus intensity and weekly total Kt/V, duration of dialysis, age, high-sensitivity C-reactive protein (hsCRP), and total cholesterol. In Fig 2, the GAM plot reveals that the dose of weekly total Kt/V (in red) intersected the zero VAS score (in green) at a value of 1.88 after adjusted for other covariates. This intersection indicated that a weekly total Kt/V < 1.88 is associated with the aggravation of pruritus intensity. As shown in Table 4, a total Kt/V of less than 1.88, a duration of dialysis longer than 4.1 years, a hsCRP level of > 0.16 mg/L, higher normalized protein nitrogen appearance (nPNA) levels, and higher intact parathyroid hormone (iPTH) levels were independent determinants of higher VAS scores of pruritus intensity. In addition, a patient age between 41.3 and 64.9 years and a cholesterol level between 149.2 and 226.1 mg/dL were associated with lower VAS scores of uremic pruritus.
Table 4. Multivariable linear regression analysis of the determinants of visual analogue scale scores of pruritus intensity.
| Parameter | Standard | |||
|---|---|---|---|---|
| Covariate | Estimate | Error | P value | |
| Intercept | -5.530 | 1.322 | < | 0.001 |
| Weekly total Kt/V < 1.88 | 1.165 | 0.577 | 0.047 | |
| Duration of dialysis > 4.1 (year) | 1.153 | 0.529 | 0.032 | |
| Normalized protein nitrogen appearance (g/kg/day) | 3.415 | 0.989 | 0.001 | |
| 41.3 ≤ age < 64.9 (years) | -1.371 | 0.494 | 0.007 | |
| High-sensitivity C-reactive protein > 0.16 (mg/L) | 1.024 | 0.468 | 0.032 | |
| 149.2 ≤ total cholesterol < 226.1 (mg/dL) | -1.743 | 0.451 | < | 0.001 |
| Intact parathyroid hormone (pg/mL) | 0.002 | 0.001 | 0.002 | |
Goodness-of-fit assessment: R2 = 0.377. The R2 value indicates that the correlation between the observed and predicted visual analogue scale scores is 0.614.
Fig 2. The generalized additive model plot of the relationship between weekly total Kt/V and the visual analogue scale (VAS) score of pruritus intensity.
The model of the plot is adjusted for duration of dialysis, normalized protein nitrogen appearance, age, high-sensitivity C-reactive protein, total cholesterol, and intact parathyroid hormone. The little vertical bars on the horizontal axis display the distribution of individual observations. The plot indicates that a weekly total Kt/V < 1.88 is associated with the aggravation of pruritus intensity.
Discussion
In this prospective cohort study assessing the role of uremic pruritus in patients receiving maintenance PD, we found that patients with a higher intensity of pruritus were significantly associated with more technique failure and worse patient survival. In addition, we found that a weekly total Kt/V of less than 1.88, a longer duration of dialysis, a higher dietary protein intake, and higher blood levels of iPTH and hsCRP were independent determinants of higher VAS scores of pruritus intensity in PD patients.
While evidence has shown an association between uremic pruritus and poor outcomes in HD patients, this is the first study to report the detrimental role of uremic pruritus on technique failure and death in PD patients. In chronic HD patients, Narita et al. showed that severe pruritus (VAS > 7.0) was independently associated with death (HR 1.60, 95% CI 1.16–2.38) after adjusting for diabetes, age, β2-microglobulin, and albumin [9]. Chen et al. also reported that HD patients with moderate/severe uremic pruritus (VAS > 4.0) suffered from higher mortality (HR 3.13, 95% CI 1.60–6.12) after adjusting for age, diabetes, albumin, hemoglobin, phosphate, iPTH, and hsCRP [17]. Among dialysis patients, the unfavorable effects of uremic pruritus may be related to inflammation, which is associated with higher cardiovascular morbidities and mortality [30,31]. The association found between uremic pruritus and hsCRP in our study also supports this hypothesis. Furthermore, uncontrollable itching due to pruritus results in poor skin health and scratch wounds, which may predispose PD patients to higher risks of exit site infection, peritonitis, and technique failure.
In this study, we also noted a higher intensity of uremic pruritus among patients with a weekly total Kt/V < 1.88 compared with those with a higher solute clearance. According to clinical guidelines, the target dose of weekly total Kt/V should be 1.7 or greater to ensure adequate solute clearance in PD [22,23]. This dialysis target is based on randomized clinical trials evaluating the outcomes of mortality or technique failure but not considering pruritus or issues related to quality of life [32,33]. Our previous study suggested a Kt/V value ≥ 1.5 to reduce the intensity of uremic pruritus in HD patients [34], while guidelines on HD suggest a target Kt/V value ≥ 1.4 to reduce mortality [35]. Similarly, the cutoff value of 1.88 for weekly total Kt/V suggested by our study is slightly above the target value of 1.7 that is recommended by PD guidelines, indicating that a solute clearance higher than the current standard may not further improve survival but is able to remove more pruritogenic substances and improve quality of life. Nevertheless, further randomized clinical trials or large cohort studies are necessary for further clarification of the dialyzable pruritogenic substance and the optimal dialysis target for reducing uremic pruritus in PD patients.
In the present study, we used VAS score to measure intensity of uremic pruritus in patients with maintenance PD. To date, no perfect method of objectively measuring pruritus is yet available. As pruritus is a subjective symptom and the VAS score of pruritus intensity is monodimensional, the measured scores are vulnerable to various factors such as emotional change or care satisfaction [36]. However, measuring the VAS score remains a simple and rapid method for assessing pruritus intensity in clinical studies and is therefore widely used.
This study is subject to several limitations. First, as VAS score was measured on a single occasion at baseline, changes of VAS score and time-dependent covariates were not considered in the study. Nevertheless, the VAS score at baseline could serve as one of the useful risk factors for assessing technique failure or death, which reproduce the typical situation of clinical practice. Second, unmeasured confounders are unavoidable due to the observational nature of this cohort study. However, the influence of residual confounders should be small because the concordance in the Cox proportional hazards model and R2 values in the linear regression models were reasonably high. Third, the sample size was relatively small, and the follow-up duration was limited in our study. To increase the number of events and achieve adequate statistical power in survival analysis, we therefore analyzed the composite endpoint. Technique failure and death are related outcomes which both indicate the termination of PD in clinical practice, and have been analyzed as composite events in previous studies[16]. Finally, because the participants were enrolled from a single medical center in Taiwan, the generalizability of the study results to other countries or races may be limited. To clarify the optimal dialysis target considering both survival and quality of life, multicenter randomized clinical trials with a larger sample size and a longer follow-up time, as well as repeated measurement of VAS scores and PD parameters, are required in future studies.
In conclusion, our results show that uremic pruritus is an independent risk factor of technique failure and death in patients receiving maintenance PD. We also found that a weekly total Kt/V < 1.88 is associated with higher intensity of uremic pruritus in PD patients. Randomized clinical trials or larger cohort studies are needed to clarify the optimal dialysis target considering both survival and quality of life in PD patients.
Supporting information
Patients were categorized as those with pruritus (VAS score > 0) or those without pruritus (VAS score = 0). VAS, visual analogue scale.
(TIF)
Acknowledgments
The authors gratefully thank Dr. Fu-Chang Hu for his valuable advice on statistical analysis.
Data Availability
All relevant data are within the paper.
Funding Statement
This study was supported by research grants to Dr. Hon-Yen Wu from the National Health Research Institutes, Taiwan (NHRI-EX106-10510PC, NHRI-EX107-10510PC), and the Far Eastern Memorial Hospital, New Taipei City, Taiwan (FEMH-EX106-10510PC, FEMH-EX107-10510PC), as well as by research grants to Dr. Mei-Ju Ko from the Department of Health, Taipei City Government, Taipei, Taiwan (10301-62-030, 10501-62-004), and Taipei City Hospital, Ren-Ai Branch, Taipei, Taiwan (No.15, 2015; No.14, 2016). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Bencini PL, Montagnino G, Citterio A, Graziani G, Crosti C, Ponticelli C (1985) Cutaneous abnormalities in uremic patients. Nephron 40: 316–321. 10.1159/000183485 [DOI] [PubMed] [Google Scholar]
- 2.Mistik S, Utas S, Ferahbas A, Tokgoz B, Unsal G, Sahan H, et al. (2006) An epidemiology study of patients with uremic pruritus. J Eur Acad Dermatol Venereol 20: 672–678. 10.1111/j.1468-3083.2006.01570.x [DOI] [PubMed] [Google Scholar]
- 3.Ko MJ, Yang JY, Wu HY, Hu FC, Chen SI, Tsai PJ, et al. (2011) Narrowband ultraviolet B phototherapy for patients with refractory uraemic pruritus: a randomized controlled trial. Br J Dermatol 165: 633–639. 10.1111/j.1365-2133.2011.10448.x [DOI] [PubMed] [Google Scholar]
- 4.Ko MJ, Chiu HC, Jee SH, Hu FC, Tseng CH (2013) Postprandial blood glucose is associated with generalized pruritus in patients with type 2 diabetes. Eur J Dermatol 23: 688–693. 10.1684/ejd.2013.2100 [DOI] [PubMed] [Google Scholar]
- 5.Ko MJ, Peng YS, Chen HY, Hsu SP, Pai MF, Yang JY, et al. (2014) Interleukin-31 is associated with uremic pruritus in patients receiving hemodialysis. J Am Acad Dermatol. [DOI] [PubMed] [Google Scholar]
- 6.Morton CA, Lafferty M, Hau C, Henderson I, Jones M, Lowe JG (1996) Pruritus and skin hydration during dialysis. Nephrol Dial Transplant 11: 2031–2036. [DOI] [PubMed] [Google Scholar]
- 7.Massry SG, Popovtzer MM, Coburn JW, Makoff DL, Maxwell MH, Kleeman CR (1968) Intractable pruritus as a manifestation of secondary hyperparathyroidism in uremia. Disappearance of itching after subtotal parathyroidectomy. N Engl J Med 279: 697–700. 10.1056/NEJM196809262791308 [DOI] [PubMed] [Google Scholar]
- 8.Graf H, Kovarik J, Stummvoll HK, Wolf A (1979) Disappearance of uraemic pruritus after lowering dialysate magnesium concentration. Br Med J 2: 1478–1479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Narita I, Alchi B, Omori K, Sato F, Ajiro J, Saga D, et al. (2006) Etiology and prognostic significance of severe uremic pruritus in chronic hemodialysis patients. Kidney Int 69: 1626–1632. 10.1038/sj.ki.5000251 [DOI] [PubMed] [Google Scholar]
- 10.Wu HY, Peng YS, Chen HY, Tsai WC, Yang JY, Hsu SP, et al. (2016) A Comparison of Uremic Pruritus in Patients Receiving Peritoneal Dialysis and Hemodialysis. Medicine (Baltimore) 95: e2935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Blake PG, Flowerdew G, Blake RM, Oreopoulos DG (1993) Serum albumin in patients on continuous ambulatory peritoneal dialysis—predictors and correlations with outcomes. J Am Soc Nephrol 3: 1501–1507. [DOI] [PubMed] [Google Scholar]
- 12.Genestier S, Hedelin G, Schaffer P, Faller B (1995) Prognostic factors in CAPD patients: a retrospective study of a 10-year period. Nephrol Dial Transplant 10: 1905–1911. [PubMed] [Google Scholar]
- 13.Davies SJ, Russell L, Bryan J, Phillips L, Russell GI (1995) Comorbidity, urea kinetics, and appetite in continuous ambulatory peritoneal dialysis patients: their interrelationship and prediction of survival. Am J Kidney Dis 26: 353–361. [DOI] [PubMed] [Google Scholar]
- 14.Jager KJ, Merkus MP, Dekker FW, Boeschoten EW, Tijssen JG, Stevens P, et al. (1999) Mortality and technique failure in patients starting chronic peritoneal dialysis: results of The Netherlands Cooperative Study on the Adequacy of Dialysis. NECOSAD Study Group. Kidney Int 55: 1476–1485. 10.1046/j.1523-1755.1999.00353.x [DOI] [PubMed] [Google Scholar]
- 15.Wang T, Heimburger O, Waniewski J, Bergstrom J, Lindholm B (1998) Increased peritoneal permeability is associated with decreased fluid and small-solute removal and higher mortality in CAPD patients. Nephrol Dial Transplant 13: 1242–1249. [DOI] [PubMed] [Google Scholar]
- 16.Wu HY, Hung KY, Huang JW, Chen YM, Tsai TJ, Wu KD (2008) Initial glucose load predicts technique survival in patients on chronic peritoneal dialysis. Am J Nephrol 28: 765–771. 10.1159/000128608 [DOI] [PubMed] [Google Scholar]
- 17.Chen HY, Chiu YL, Hsu SP, Pai MF, Lai CF, Yang JY, et al. (2010) Elevated C-reactive protein level in hemodialysis patients with moderate/severe uremic pruritus: a potential mediator of high overall mortality. QJM 103: 837–846. 10.1093/qjmed/hcq036 [DOI] [PubMed] [Google Scholar]
- 18.Weber MA, Schiffrin EL, White WB, Mann S, Lindholm LH, Kenerson JG, et al. (2014) Clinical practice guidelines for the management of hypertension in the community: a statement by the American Society of Hypertension and the International Society of Hypertension. J Clin Hypertens (Greenwich) 16: 14–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mancia G, Fagard R, Narkiewicz K, Redon J, Zanchetti A, Bohm M, et al. (2013) 2013 ESH/ESC guidelines for the management of arterial hypertension: the Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 34: 2159–2219. 10.1093/eurheartj/eht151 [DOI] [PubMed] [Google Scholar]
- 20.Yosipovitch G, Zucker I, Boner G, Gafter U, Shapira Y, David M (2001) A questionnaire for the assessment of pruritus: validation in uremic patients. Acta Derm Venereol 81: 108–111. [DOI] [PubMed] [Google Scholar]
- 21.Furue M, Ebata T, Ikoma A, Takeuchi S, Kataoka Y, Takamori K, et al. (2013) Verbalizing Extremes of the Visual Analogue Scale for Pruritus: A Consensus Statement. Acta Derm Venereol 93: 214–215. 10.2340/00015555-1446 [DOI] [PubMed] [Google Scholar]
- 22.Lo WK, Bargman JM, Burkart J, Krediet RT, Pollock C, Kawanishi H, et al. (2006) Guideline on targets for solute and fluid removal in adult patients on chronic peritoneal dialysis. Perit Dial Int 26: 520–522. [PubMed] [Google Scholar]
- 23.Peritoneal Dialysis Adequacy Work Group (2006) Clinical practice guidelines for peritoneal dialysis adequacy. Am J Kidney Dis 48 Suppl 1: S98–129. [DOI] [PubMed] [Google Scholar]
- 24.Twardowski ZJ (1987) Peritoneal equilibration test. Pent Dial Bull 7: 138–147. [Google Scholar]
- 25.Bergstrom J, Heimburger O, Lindholm B (1998) Calculation of the protein equivalent of total nitrogen appearance from urea appearance. Which formulas should be used? Perit Dial Int 18: 467–473. [PubMed] [Google Scholar]
- 26.Yee TW, Wild CJ (1996) Vector Generalized Additive Models. Journal of the Royal Statistical Society Series B (Methodological) 58: 481–493. [Google Scholar]
- 27.Yee TW (2015) Vector generalized linear and additive models: with an implementation in R: Springer. [Google Scholar]
- 28.Yee TW (2009) VGAM: Vector generalized linear and additive models. R package version 1.0–5. URL: https://CRANR-projectorg/package=VGAM
- 29.Moore DF (2016) Model Selection and Interpretation Applied Survival Analysis Using R: Springer; pp. 73–86. [Google Scholar]
- 30.Harris TB, Ferrucci L, Tracy RP, Corti MC, Wacholder S, Ettinger WH Jr., et al. (1999) Associations of elevated interleukin-6 and C-reactive protein levels with mortality in the elderly. Am J Med 106: 506–512. [DOI] [PubMed] [Google Scholar]
- 31.Yeun JY, Levine RA, Mantadilok V, Kaysen GA (2000) C-Reactive protein predicts all-cause and cardiovascular mortality in hemodialysis patients. Am J Kidney Dis 35: 469–476. [DOI] [PubMed] [Google Scholar]
- 32.Paniagua R, Amato D, Vonesh E, Correa-Rotter R, Ramos A, Moran J, et al. (2002) Effects of increased peritoneal clearances on mortality rates in peritoneal dialysis: ADEMEX, a prospective, randomized, controlled trial. J Am Soc Nephrol 13: 1307–1320. [DOI] [PubMed] [Google Scholar]
- 33.Lo WK, Ho YW, Li CS, Wong KS, Chan TM, Yu AW, et al. (2003) Effect of Kt/V on survival and clinical outcome in CAPD patients in a randomized prospective study. Kidney Int 64: 649–656. 10.1046/j.1523-1755.2003.00098.x [DOI] [PubMed] [Google Scholar]
- 34.Ko MJ, Wu HY, Chen HY, Chiu YL, Hsu SP, Pai MF, et al. (2013) Uremic pruritus, dialysis adequacy, and metabolic profiles in hemodialysis patients: a prospective 5-year cohort study. PLoS One 8: e71404 10.1371/journal.pone.0071404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hemodialysis Adequacy Work Group (2006) Clinical practice guidelines for hemodialysis adequacy, update 2006. Am J Kidney Dis 48 Suppl 1: S2–90. [DOI] [PubMed] [Google Scholar]
- 36.Stander S, Augustin M, Reich A, Blome C, Ebata T, Phan NQ, et al. (2013) Pruritus assessment in clinical trials: consensus recommendations from the International Forum for the Study of Itch (IFSI) Special Interest Group Scoring Itch in Clinical Trials. Acta Derm Venereol 93: 509–514. 10.2340/00015555-1620 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Patients were categorized as those with pruritus (VAS score > 0) or those without pruritus (VAS score = 0). VAS, visual analogue scale.
(TIF)
Data Availability Statement
All relevant data are within the paper.