Abstract
Maize evolution under domestication is a process that continues today. Case studies suggest that Mexican smallholder family farmers, known as campesinos, contribute importantly to this, but their significance has not been explicitly quantified and analysed as a whole. Here, we examine the evolutionary and food security implications of the scale and scope under which campesinos produce maize. We gathered official municipal-level data on maize production under rainfed conditions and identified campesino agriculture as occurring in municipalities with average yields of less than or equal to 3 t ha−1. Environmental conditions vary widely in those municipalities and are associated with a great diversity of maize races, representing 85.3% of native maize samples collected in the country. We estimate that in those municipalities, around 1.38 × 1011 genetically different individual plants are subjected to evolution under domestication each season. This implies that 5.24 × 108 mother plants contribute to the next generation with their standing genetic diversity and rare alleles. Such a large breeding population size also increases the total number of adaptive mutations that may appear and be selected for. We also estimate that campesino agriculture could potentially feed around 54.7 million people in Mexico. These analyses provide insights about the contributions of smallholder agriculture around the world.
Keywords: genetic diversity, evolutionary services, production environments, food security
1. Introduction
Domestication is not only a fascinating example of evolution, but also a process that changed the course of human history and that continues to influence the fate of humanity. Crop evolution under domestication, which started approximately 10 000 years ago [1], continues today in a range of agricultural systems spanning traditional farming to industrialized large-scale agriculture. For some crops, it is in their centres of origin and/or domestication where the highest diversity is concentrated, and where this process continues to be driven directly by farmers [2]. Mexico is one such centre for many crops, including maize [3–5], one of the most important food crops in the world, providing 30% of food calories to approximately 4.5 billion people in 94 developing countries [6]. It is the most important food crop in Mexico for both urban and rural populations [7].
Maize diversity is represented by a wide array of morphologically distinct crop populations grown across the country, known as native varieties or landraces. These are grouped into 59 maize races [8], containing an impressive level of genetic diversity, even within a single race [5,9,10]. Campesinos are heirs to, and trustees of, the largest genetic diversity of maize in the world [4,5,8–11], which they maintain in their agricultural systems today. Campesinos are smallholder farmers managing family farms producing, at least partially, for self-consumption, relying mostly on family labour and using combinations of animal and mechanical traction, manure and inorganic fertilizers, and planting mostly native varieties. Their farms account for the largest area planted with maize in the country [11,12]. The role campesinos continue to play in maintaining maize landrace diversity has been well documented [13–18]. They maintain traditional knowledge about the performance of their landraces and practices of saving and sharing seed by and among themselves, from one cycle to the next. This allows alleles to pass from one generation to the next, thus continuing the evolutionary processes that sustain and generate crop genetic diversity [3,8,14,19].
In spite of their important role in maintaining maize genetic diversity, there is a common perception that campesinos are unproductive, anachronistic and a hindrance to Mexican agriculture [20,21]. Their demise has been predicted for a long time, particularly after the implementation of the North American Free Trade Agreement (NAFTA), based on the assumption that they are inefficient and at a disadvantage in the face of competition from large-scale commercial farmers in open, globalized markets [22–24]. Current Mexican official agricultural policies are focused on medium-sized and large commercially oriented farmers, considered to be more economically viable, while campesinos are largely ignored [21,23,25]. A reduction in the importance of maize as income for rural households has continued since pre-NAFTA times [26,27], but despite this, plus biased policies and negative predictions about campesino production, they continue to exist and produce maize for Mexico [21,23,25]. A key reason for this is that, for campesinos, maize is a ‘multi-functional’ crop with great cultural importance [13,28], providing a broad scope of benefits, such as food for self-consumption and food security [25,29,30], a diverse gastronomy and multiple products [29,31], and opportunities to participate in local and regional markets to generate income [23,25,32].
There are several regional and case studies characterizing campesino agriculture [23–25,30,33], as well as documenting campesinos’ role in maintaining maize landrace diversity [13–18]. To our knowledge, however, the contributions that campesinos make for the evolution of this crop under domestication and for the maize supply of the country have not been analysed at a national scale; nor has their relevance to national and global scales been acknowledged. To address this knowledge gap, in this paper, we examine the implications of the scale and scope under which campesinos produce maize under rainfed conditions for these two issues. Both issues are complex in their own right, but it is worth addressing them together because they are intrinsically linked and reinforce each other. To accomplish this, we employ a multifaceted approach that integrates (i) maize consumption and human demography, (ii) biophysical variables using geographical information systems, and (iii) population genetics and evolutionary theory.
2. Methods
(a). Identifying campesino maize production at the national level
Our study focuses on the main rainfed agricultural season (May–October) of 2010 (year of the last national Mexican population census). Municipal-level data on area planted and harvested with maize, average yields and total production were obtained from the official government statistics of the Sistema de Información Agroalimentaria y Pesquera (SIAP) annual statistics of agricultural production (http://nube.siap.gob.mx/cierre_agricola/). These data are based on field information collected and validated at the municipality level according to SIAP's Technical Standard for the Generation of Basic Agricultural and Fishery Statistics [34]. Municipalities were grouped into seven classes according to their average maize yield per hectare in increments of 1 t ha−1, from 0 (area was planted with maize but none was harvested) to the maximum observed yield of 8.75 t ha−1. As SIAP's data do not include data on the structure of farm sizes, we estimated it using data from PROCAMPO (a government programme providing a subsidy per hectare of land cultivated to farmers) on the number of beneficiaries that received a payment, and the size of the area they planted with rainfed maize. We cross-classified these data with SIAP's data on municipal average yields and area planted, assuming that a beneficiary corresponded to a farm. We adjusted PROCAMPO's and SIAP's data to estimate the overall distribution of area planted to maize by farm and by yield class (electronic supplementary material, S1.1, Estimating farm size structure). Although PROCAMPO's data are not specifically intended for this purpose, these are the only data available with farm size of maize producers by municipality. Demographic data on total and rural population sizes by municipality were obtained from the national census of 2010. We combined SIAP's and national census’s data using the municipality as a joining term and keeping only those producing maize. The area planted with improved and native varieties was estimated based on the quantity of improved seed reported by different sources (electronic supplementary material, S1.2, Estimating area planted with improved and native varieties and table S1).
(b). Environmental conditions of areas planted with maize
To analyse the variation in environmental conditions, we focused on precipitation, temperature, altitude and slope at 1 km resolution. Precipitation and temperature data from May to October (rainy season) were extracted from Cuervo-Robayo et al. [35]. Altitude and slope data come from Guevara & Arroyo-Cruz [36,37]. Before analyses, data were cropped to agricultural land (without rainfed/irrigation subcategories due to the uncertainty of defining such polygons) according to INEGI [38] for each for the municipalities presenting maize production in 2010 (SIAP's data). These data were then analysed by: (i) visualizing the data with boxplots and violin plots by yield class; (ii) examining differences in the spread of data using Feltz & Miller's test [39]; (iii) analysing the overlap of the conditions by performing a principal component analysis (PCA) and testing for differences among each yield class with a distance-based test of homogeneity of multivariate dispersions [40] (for the PCA, ellipsoids were fitted to the components scores associated with each yield class and altitude was excluded because it is highly correlated with temperature); and (iv) analysing the distance among the environments of each yield class with a dendrogram constructed from a hierarchical clustering analysis. This was done using Ward's method with Euclidean distances and the suggested partition (the one with higher relative loss of inertia). Analyses were performed using ArcMap and the R [41] packages raster [42], maptools [43], cvequality [44], vegan [45] and FactoMineR [46].
(c). Campesinos’ contribution to maize genetic diversity
To estimate the contribution of smallholders to maize genetic diversity, we used classic population genetic formulae using as a proxy of effective population size (Ne) the effective number of breeding individuals (Nb [47]), which we assumed to be the number of ears from where seeds are extracted in order to sow 4.68 million hectares (Mha). This is a good proxy for maize because in maize, mating is random, sex ratio is equal, generations are not overlapping and the number of breeding individuals is relatively constant over generations. Owing to the lack of empirical data at the national level for the number of ears set aside to generate the seed needed to plant 1 ha, and because this probably changes from region to region, we used four estimations that range from 114 to 290 ears ha−1 (electronic supplementary material, S1.3, Estimating the amount of saved seed to plant 1 ha).
The effect of genetic drift on heterozygosity (H) in the absence of mutation is calculated with the formula Hn+1 = (1 – 1/2Ne)Hn [48], where Ne is the effective population size. The decay of heterozygosity can then be estimated as 1/(2Ne). We used this formula to estimate the decay of heterozygosity in the Mexican maize population using Nb instead of Ne. Nb was estimated as the number of mother plants that was used for planting the total rainfed maize area sown in 2010. We then estimated the number of new mutations by the formula μ2Nb, where µ is the mutation rate. Here, we only considered substitutions, using the mutation rate of μ = 1.63 × 10−8 [49]. The former number of mutations gives an estimate at the site level, so we next estimated the total number of mutations for the entire coding region (genes that generate proteins) of the maize genome. For this, we assumed that the amount of DNA bases in coding regions is around 2% of the 2700 Mb of the maize genome [50] and multiplied it for the number of mutations estimated previously. Analyses were performed using R [41].
(d). Assessing the potential contribution of rainfed production to maize demand
The potential population that could be fed from maize production in 2010 was estimated by combining municipal-level data on maize production and rural population, assuming a per capita consumption of 108.5 kg. This number was calculated using a consumption of tortillas of 217.9 and 70.2 g person−1 day−1 of maize grain, employed by the government to develop poverty lines for the country [51] (electronic supplementary material, table S2). We calculated four parameters for each municipality: (i) the size of the total population that could be fed from local maize production, regardless of the population present; (ii) the size of the rural population in the municipality that could be fed from local maize production; (iii) the size of any additional population that could be fed from surpluses if available beyond (ii); and (iv) the size of the net population that could be or not be fed from local maize production (see details in electronic supplementary material, S1.4, Estimating per capita annual maize consumption). We also calculated the size of the rural population that could be fed by year from 2003 to 2015 by following the same procedure (electronic supplementary material, S1.4, Estimating per capita annual maize consumption). This analysis is restricted to municipalities where there were demographic data in both census years to be able to calculate population rates of change (90.4% of those planted with maize in 2010).
3. Results
(a). Identifying campesino rainfed maize production at the national level
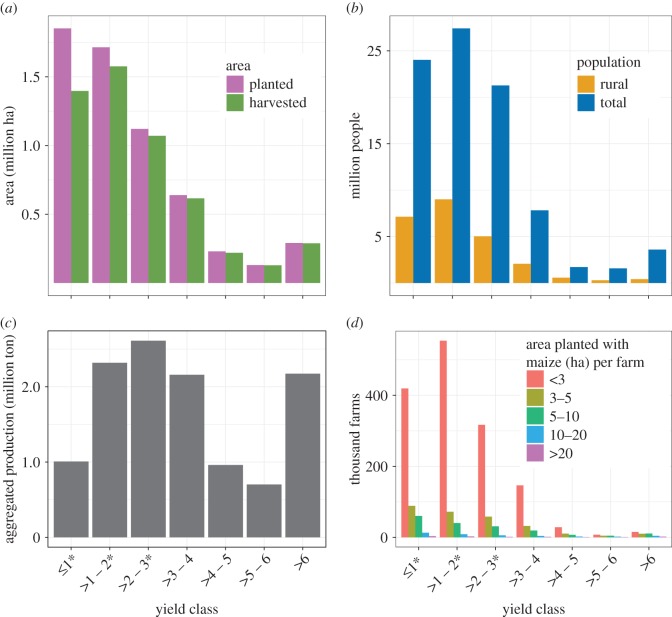
During the rainfed season of 2010, maize production took place in 2271 municipalities (92.4% of the total; electronic supplementary material, table S3). Municipalities in yield classes of less than or equal to 3 t ha−1 accounted for 78.4% of the area planted with maize, generating 49.8% of the total production (figure 1). Most of the country's total and rural population was located there. Municipalities in yield classes of greater than 3 t ha−1 accounted for only 21.6% of the planted area, producing 50.2% of the total production (figure 1). We estimated that 1.99 million farms planted maize across all municipalities, with 88.6% planting up to 5 ha farm−1 (electronic supplementary material, table S4). Most rainfed maize production occurred in municipalities with average yields of less than or equal to 3 t ha−1 and in farms planting up to 5 ha of maize, involving 1.68 million farms. Municipalities in yield classes below and above the 3 t ha−1 threshold produced each about half of the total rainfed maize production. Those below the threshold, however, accounted for 4/5 of the total area planted, while those above it accounted for the rest. This contrast is explained by differences in yields (electronic supplementary material, figure S1). The overall average yield for municipalities in yield classes of less than or equal to 3 t ha−1 was 1.3 (±0.7) t ha−1, while for municipalities above this threshold was 4.4 (±1.4) t ha−1.
Figure 1.
Summary characteristics of each yield class for the year 2010. (a) Area planted and harvested with rainfed maize and (b) rainfed maize production according to SIAP, (c) total and rural population according to INEGI and (d) estimated number of farms by area planted with maize per farm. *Yield classes mostly representing campesino agriculture.
While there are no specific data on the type of germplasm used to plant each yield class, we estimated that improved varieties—mostly hybrids, and thus not subject to evolution under domestication in situ because seed generated elsewhere is purchased every year—were used in about 20% of the total rainfed area planted with maize (i.e. the majority of the area planted in municipalities in the top four yield classes, greater than 3 t ha−1; see supplementary material, S1.2, Estimating area planted with improved and native varieties). The rest of the area, comprising municipalities in the lower three yield classes, was mostly planted with the farmers’ own seed, thus subject to evolution under domestication. The presence of native varieties is widespread in municipalities in yield classes of less than or equal to 3 t ha−1, as demonstrated by the fact that the Global Maize Project [8] collected 85.3% of their nationwide samples of native landraces in these municipalities (electronic supplementary material, table S5).
Based on these data, we consider that campesino maize agriculture corresponds mostly to farms producing this crop in municipalities in yield classes of less than or equal to 3 t ha−1. The bulk of smallholder farmers (planting less than or equal to 5 ha with maize) is located there, mostly relying on native landraces and traditional seed management practices, with relatively low average yields (1.3 ± 0.7 t ha−1). They are more likely to consume a larger proportion of their production and have smaller surpluses to sell. Commercially oriented agriculture corresponds mostly to farmers producing in municipalities in yield classes greater than 3 t ha−1 relying mostly on improved varieties.
(b). Campesino farmers produce maize under a wide variety of environments
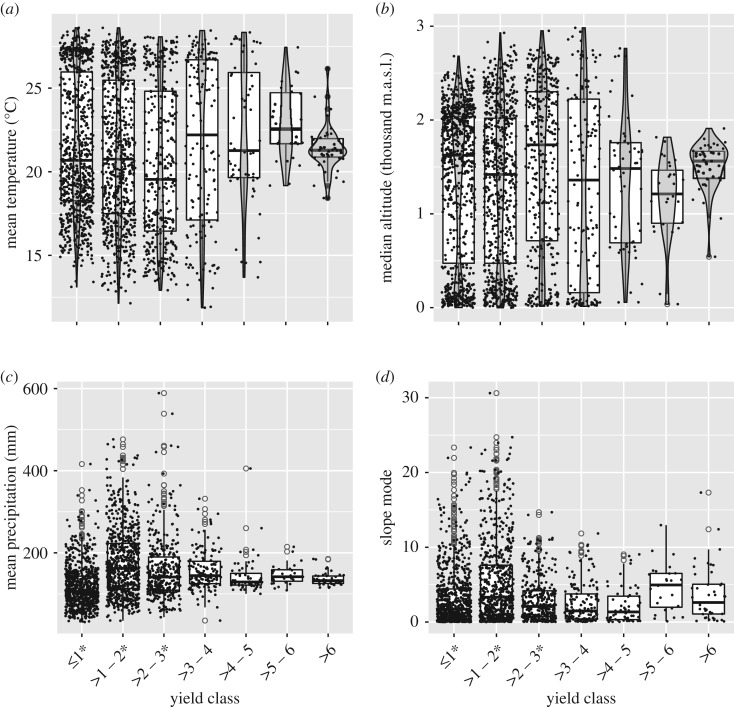
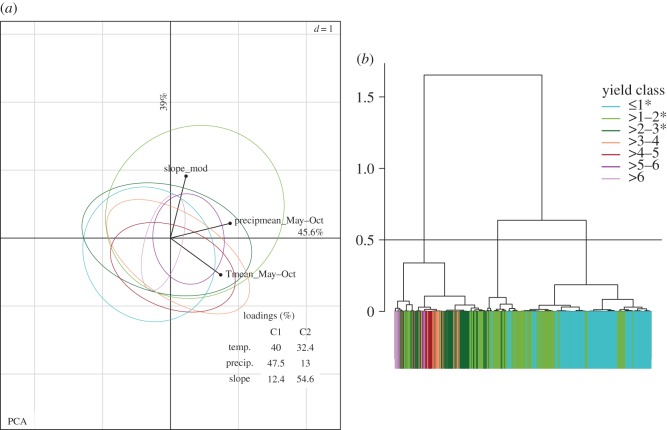
The spread (variance) of the environmental conditions of maize areas differ greatly among yield classes; lower yield classes have a higher spread, that decreases in the higher yield classes (figure 2; electronic supplementary material, table S6). Altitude and temperature have a bimodal distribution that reflects the two main environments where maize is cultivated: temperate and tropical, consistent with genetic differentiation among highland and lowland maize [5,9]. The PCA shows that there is a degree of environmental overlap among the seven yield classes (figure 3a), but significant differences exist among most yield classes (see test for homogeneity in electronic supplementary material, table S7). The widest ellipsoids include the lowest three yield classes (i.e. campesino agriculture), and the narrower ellipsoids contain commercially oriented agriculture. A dendrogram from a hierarchical clustering of the PCA (figure 3b) shows that the municipalities can be grouped in k = 3 clusters. Of these, two include most municipalities in yield classes less than or equal to 2 t ha−1, while municipalities greater than 2 t ha−1 form the other cluster. Within this cluster, there is a substantial mix of municipalities in different yield classes, showing an overlap of environmental conditions among them, except for municipalities in yield classes greater than 5 t ha−1 that are grouped together. Municipalities in higher yield classes are concentrated in relatively few regions of the country, and those in lower yield classes are widely scattered across Mexico, where most native landraces have been collected [8] (electronic supplementary material, figure S2).
Figure 2.
Box plots of the distribution of municipalities planted to rainfed maize in 2010 grouped by yield class for four environmental variables: (a) mean temperature during rainy season (May–Oct), (b) median altitude, (c) mean precipitation during rainy season and (d) slope mode. For (a,b), violin plots are shown on the top of boxplots, showing that for levels 1–3, the data have a bimodal distribution. *Yield classes mostly representing campesino agriculture (see electronic supplementary material, table S6 for equality of coefficients of variation test results). (Online version in colour.)
Figure 3.
Environmental conditions of maize producing municipalities: (a) PCA for the temperature, precipitation and slope environmental variables of municipalities grouped by yield classes (colour codes in (b)). Loadings are shown for the first two components (see electronic supplementary material, table S7 for test for homogeneity). (b) Dendrogram from a hierarchical clustering performed on the PCA. The horizontal line cuts in the suggested number of clusters (k = 3). Colours represent municipalities grouped by yield classes. *Yield classes mostly representing campesino agriculture.
(c). Campesinos contribute to maintain and generate maize genetic diversity
As shown above, 4.68 Mha were planted with rainfed maize in approximately 1.68 million farms in municipalities with average yields of up to 3 t ha−1, representative of campesino agriculture. Assuming that around 30 000 plants ha−1 were grown [52], this results in 1.38 × 1011 genetically different individual plants, especially considering that Mexican maize landraces present a high diversity that is structured mostly according to the interaction of latitude and altitude and not race identity [9,10,53]. On each of those individual plants, open pollination increases diversity through recombination. While most of the resulting seed of the 30 000 plants ha−1 are destined for human consumption or sale, in campesino agriculture, a subset is set aside for planting in the next cycle. This is about 114–290 ears ha−1, depending on how it is calculated. Because seeds from the same ear have the same mother, this reduces the population to that number of effective families (a subestimate, considering that fathers of each kernel would likely be different) passing to the next generation. Assuming our lowest estimate (electronic supplementary material, table S8) of 114 maize families (ears) per hectare in 4.68 Mha, this means an Nb of 5.24 × 108 plants contributing to the next generation with their background genetic diversity along with rare alleles. This Nb is of the same order of magnitude as the Ne = 1.1 × 108 that has been estimated for Mexican maize (considering together lowlands and highlands maize estimated Ne [54]).
Selection of the ears constituting the Nb is done independently by around 2.52 million smallholders (considering about 1.68 million farms with 1.5 people per farm involved in the seed selection process), in different environments (figures 2 and 3) and in a process in which both men and women make selection decisions based on their own multiple criteria. Therefore, there are local and regional differences regarding which types of ears are looked for and which individual plants would survive to produce seed, thus making the pool of Nb = 5.24 × 108 a diverse sample of the entire population. Considering this lower estimate of Nb, we estimate a decay of heterozygosity of 9.53 × 10−10 per generation (electronic supplementary material, table S8). Depending on the number of maize ears per hectare used to plant 4.68 Mha, an upper limit of 9.23 × 108 to 2.35 × 109 new mutations by generation can be estimated to occur in the coding regions of the maize genome (electronic supplementary material, table S8).
(d). Campesino maize farmers contribute importantly to the national maize food supply
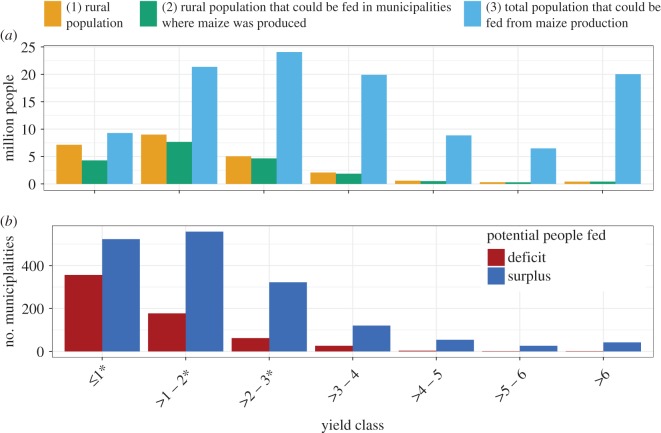
Farmers representing mostly commercially oriented agriculture (yields greater than 3 t ha−1) produced enough to potentially feed 55.3 million people; but farmers representing mostly campesino agriculture (yields less than or equal to 3 t ha−1) could potentially feed an additional 54.7 million people, including all the rural population in municipalities with campesino agriculture (21.1 million people), and with surpluses that could be used to feed an extra 33.6 million people (electronic supplementary material, table S3). However, at a disaggregate level, important differences appear (figure 4). In certain municipalities, local production would be insufficient to feed all the rural population present, while in others, surpluses would be available to feed additional people (figure 4a). Results show that within each yield class, the surplus maize produced, at least in theory, could contribute to cover any deficit, and there still could be enough to feed additional people elsewhere. These results also can be presented as number of municipalities with surpluses and deficits (figure 4b), with their spatial distribution across the country (electronic supplementary material, figure S3). The patterns described above are consistent for more than a decade for which municipal-level data are publicly available (2003–2015; electronic supplementary material, figure S4).
Figure 4.
(a) Comparison by yield class of the rural population present (bar 1) with the estimated rural population that could be fed without (bar 2) and with local surpluses (bar 3). The difference between the size of bars 1 and 2 corresponds to the size of the rural population that could not be fed from local production, but due to municipalities with surpluses that could cover any deficit, the estimated total population that could be fed (bar 3) would be larger than the rural population present (difference between bars 1 and 3). (b) Number of surplus and deficit municipalities based on whether local maize production could or could not feed the resident rural population. *Yield classes mostly representing campesino agriculture.
4. Discussion
(a). Campesino farmers contribute to maintain and generate maize genetic diversity, an ‘evolutionary service’ important as a global public good
Genetic data suggest that maize Ne started to recover from its domestication bottleneck only around 1000 years ago, with a dramatic expansion in the last 200 years [54]. This is not only congruent with the Nb of 5.24 × 108 we estimate for campesino maize, but also shows that the current time may represent the point of highest diversity in maize history [55]. Empirical studies have documented the evolutionary signatures of maize domestication (e.g. [5,10,54,55]). However, those studies have focused on extant diversity, but not on the implications of the scope and extent in which campesinos produce maize in Mexico. Here, we suggest that campesinos not only contribute to maintaining the evolution of maize, but that the scale at which this is done becomes in itself an irreplaceable evolutionary or evosystem service [56,57] (defined as ‘the uses or services to humans that are produced from the evolutionary process' [56]). This evosystem service emerges because maize campesino agriculture combines in a single system three of the main factors known to positively affect adaptive evolution: large effective population size, high standing genetic diversity and environmental change [58–61], as follows:
-
(i)
Our estimated slow decay of heterozygosity (9.53 × 10−10 per generation) causes a small loss of standing genetic diversity and rare alleles (frequency less than 1–5%), a primary source of adaptive genetic variation for crop domestication and breeding [55]. Conserving this diversity in situ thus increases the possibility that beneficial variation may be immediately available to experience new selective pressures [58], including alleles that can suffer selective sweeps [62], as well as several different alleles from which polygenic traits can undergo adaptive change [63].
-
(ii)
Growing a large amount of genetically diverse maize plants is useful not only for preserving the extant genetic diversity, but also for creating new diversity by means of mutation, as exemplified by the large estimated number of new mutations that could appear in the coding regions of the maize genome (9.23 × 108 to 2.35 × 109 per generation). Most new mutations would be neutral or detrimental to the mutant individual, but a small percentage may be beneficial and under the right selection pressure will increase their frequency rapidly [64–66].
-
(iii)
Natural selection depends on local conditions. Given the wide range of environments where maize landraces are grown in Mexico (particularly in municipalities in yield classes less than or equal to 3 t ha−1; figure 3b), it is likely that different alleles are being favoured by different selection pressures, in a process that historically as well as presently has involved seed exchange and farmer's migration. This creates a setting of recurrent adaptation to a changing environment, which is a powerful driver of molecular adaptation [59].
To put these numbers into perspective, in 2010, approximately 41 Mha of maize were harvested in Canada, the USA and Mexico together [67]. However, US and Canadian maize agriculture relies almost exclusively on hybrids, largely genetically modified varieties, whose seeds are bought each cycle and sown in large but genetically homogeneous areas. Additionally, although genetic diversity can be sourced into breeding materials from landraces [68], hybrids are reproduced avoiding gene flow among them and with landraces. Thus, the Ne and diversity of maize for the whole North American region comes mostly from the maize grown in the 4.68 million ha under Mexican campesino agriculture.
The large maize population maintained by campesino agriculture not only preserves genetic diversity, but also provides multiple opportunities for the appearance of potentially beneficial mutations. This is important because we cannot predict the future, and the presence of genetic diversity is fundamental for adaptive evolution in response to changing environmental conditions [68,69]. Therefore, the larger the scale and the more diverse the scope of this process, the more genetic diversity available; and thus, the higher the probability that some alleles, although currently rare or unknown, will become widely adaptive under new, and in many cases unpredictable, conditions [19] (that is, the higher the option value of genetic diversity under evolution). This constitutes not only an evosystem service, but also a positive externality—an uncompensated positive impact of one agent's actions (i.e. campesinos growing maize) over the welfare of another (i.e. current and future maize consumers worldwide). If this diversity were no longer available, breeding programmes would lack an important source of novel genetic variation to tap in order to allow continued genetic gains in a dynamic agricultural environment [68].
The evolutionary service provided by campesino agriculture cannot be replaced by the conservation of seeds in gene banks (ex situ conservation), because those seeds represent just a snapshot of the genetic diversity present at the time of collection and are no longer under evolution [19]. Therefore, although ex situ conservation is a fundamental component of a crop conservation strategy, the capacity to generate and maintain adaptive genetic change in response to dynamic environmental conditions cannot be accomplished by gene banks alone. Similarly, for in situ conservation to be successful, we should consider that (i) large maize populations are needed, as suggested by our population genetics calculations, and (ii) these populations should be spread along different environments, because the loss environmental range may imply the loss of genetic diversity [69]. Considering the extent and scope of Mexican campesino agriculture, this cannot be achieved by targeting just a handful of farmers in a few locations to conserve maize races based in their phenotypes [70].
(b). Campesino maize farmers are crucial for national food security
Campesinos also make a crucial contribution to the maize supply of Mexico. While some individual campesino households may not produce enough for their needs [23,33], as a group, they produce important surpluses, which have the potential to not only feed themselves, but also to make a substantial contribution to feed others in their communities and regions. To put our results in perspective, in 2010, Mexico produced 23.3 million tons (MT) of mostly white maize, making it self-sufficient in maize for human consumption, and imported 7.7 MT of mostly yellow maize intended for animal feed. Campesino agriculture contributed 25.5% of the total national production, with the potential to feed 48.7% of all the Mexican population (electronic supplementary material, tables S3 and S10).
While campesinos produce maize due to a multiplicity of values they ascribe to this crop, particularly to their native landraces, these values tend to be systematically ignored by markets [71]. Therefore, their decisions to produce maize may appear uneconomical and inefficient when judged by standard financial profitability criteria. The discrepancy between financial profitability and the willingness of farmers to produce maize reflects a market that fails to incorporate the multiple values these farmers ascribe to this crop into market prices [71]. Hence, the large number of campesinos that produce maize under highly diverse (and in many instances adverse) environmental conditions can be seen as producing another positive externality of national relevance: more maize produced and people fed than would have occurred if farmers were purely profit-driven. Our results, however, should not be interpreted as implying that rainfed maize production is feeding the entire population of rural areas. Available data simply indicate the potential to achieve this.
(c). Strengthening campesino agriculture and its positive externalities
Our results show a yield gap between campesino and commercially oriented agriculture. This gap is normally attributed, at least partially, to the extensive reliance of the former on native landraces and on improved varieties by the latter. However, many native landraces can provide reasonably high yields with modest amounts of inputs and small changes in agronomic management (electronic supplementary material, figure S5a and table S9) and are competitive or even out-yield hybrids in many environments [70] (electronic supplementary material, figure S5b), so the observed low productivity is not inherently related to the use of native landraces. Moreover, the remarkable genetic diversity and adaptability of native landraces to wide-ranging agro-ecological conditions [52,72] allow campesinos to produce maize at the wide scale and in areas where improved varieties are unlikely to be adapted [14,73].
Mexican campesinos are generating positive externalities of national and global relevance that should be considered public goods and supported as such, particularly because there is no guarantee that they will continue to provide these goods in the future. No effective policies and programmes exist in Mexico to reward and support these farmers’ contributions. Some practices related to local education, development and marketing of products from landraces with added value, and other practices supporting in situ conservation, have been promoted and need to be supported at a larger scale [74]. However, to truly encompass the extent of maize genetic diversity, campesino agriculture needs to be recognized and supported as a production system that generates conservation outcomes. Our results suggest policy-relevant areas for further research such as: (i) exploring new approaches to improve the yields and quality of native varieties under the conditions that campesinos face, so as to improve their productivity while still keeping them under evolution; (ii) improving our understanding of maize trading at the local level and how surpluses reach consumers, either through markets or other exchange mechanisms, as the basis for identifying interventions to improve trading conditions to benefit both producers and consumers; (iii) understanding the conditions that foster farmers to continue the management of this diversity, as well as the circumstances that hinder it, in order to design better interventions to support them; and (iv) refining population genetics models to better quantify campesinos’ contribution for generating and maintaining maize genetic diversity, as the basis for monitoring and safeguarding these processes. As evolution under domestication continues to occur in other crops in other countries—especially in regions of high agrobiodiversity—the results presented here suggest that smallholders’ contributions in those regions merit closer examination.
Supplementary Material
Acknowledgements
Thanks to M. Wilcox and Q. Orozco-Ramírez, G. Tamaríz, A. Gálvez, L. Saad, D. Piñero, J. Lauderdale, M. Cano, G. Gordillo, S. Brush, E. Palacios Moreno, C. Burgeff, B. Blonder, editor L. Galloway and three anonymous reviewers for comments and inputs to the paper.
Data accessibility
Data, spreadsheets and R scripts used for analyses are available at the Dryad repository: http://dx.doi.org/10.5061/dryad.79q870b [75].
Authors' contributions
M.R.B., A.M.-Y. and J.S. designed research; M.R.B., A.M.-Y., H.P., A.P.-M., F.A. and O.O.-G. performed research; H.P. and D.O.-S. contributed new reagents/analytic tools; M.R.B., A.M.-Y., J.S., A.P.-M. and H.P analysed data; M.R.B., A.M.-Y., J.S., A.P.-M., H.P. and F.A. wrote the paper.
Competing interests
We declare we have no competing interests.
Funding
Partial funding was provided by ‘The Global Alliance for the Future of Food’, CONACYT grant no. 247730 and SEMARNAT's Grant 2013. This work is a following up product of the Global Native Maize Project.
References
- 1.Larson G, et al. 2014. Current perspectives and the future of domestication studies. Proc. Natl Acad. Sci. USA 111, 6139–6146. ( 10.1073/pnas.1323964111) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brush SB. 2004. Farmers' bounty: locating crop diversity in the contemporary world. New Haven, CT: Yale University Press. [Google Scholar]
- 3.Doebley J. 2004. The genetics of maize evolution. Annu. Rev. Genet. 38, 37–59. ( 10.1146/annurev.genet.38.072902.092425) [DOI] [PubMed] [Google Scholar]
- 4.Kato TA, Mapes C, Mera ML, Serratos JA, Bye RA (eds). 2009. Origen y diversificación del maíz: una revisión analítica. México, DF: Universidad Nacional Autónoma de México, Comisión Nacional para el Conocimiento y Uso de la Biodiversidad; See http://www.biodiversidad.gob.mx/genes/pdf/origen_div_maiz.pdf. [Google Scholar]
- 5.Hufford MB, et al. 2012. Comparative population genomics of maize domestication and improvement. Nat. Genet. 44, 808–811. ( 10.1038/ng.2309) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shiferaw B, Prasanna BM, Hellin J, Bänziger M. 2011. Crops that feed the world 6. Past successes and future challenges to the role played by maize in global food security. Food Secur. 3, 307–327. ( 10.1007/s12571-011-0140-5) [DOI] [Google Scholar]
- 7.Fernandez-Suarez R, Morales-Chavez LA, Galvez-Mariscal A. 2013. Importancia de los maíces nativos de México en la dieta nacional: una revisión indispensable. Rev. Fitotec. Mex. 36(Suppl. 3-A), 275–283. [Google Scholar]
- 8.CONABIO. 2011. Recopilación, generación, actualización y análisis de información acerca de la diversidad genética de maíces y sus parientes silvestres en México. México, DF: Comisión Nacional para el Conocimiento y Uso de la Biodiversidad; See http://www.biodiversidad.gob.mx/genes/pdf/proyecto/InformedeGestion_V1.pdf. [Google Scholar]
- 9.Arteaga MC, Moreno-Letelier A, Mastretta-Yanes A, Vazquez-Lobo A, Breña-Ochoa A, Moreno-Estrada A, Eguiarte LE, Piñero D. 2016. Genomic variation in recently collected maize landraces from Mexico. Genom. Data 7, 38–45. ( 10.1016/j.gdata.2015.11.002) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Romero-Navarro JA, et al. 2017. A study of allelic diversity underlying flowering-time adaptation in maize landraces. Nat. Genet. 49, 479–480. ( 10.1038/ng.3784) [DOI] [PubMed] [Google Scholar]
- 11.Mera-Ovando LM, Mapes-Sánchez C. 2009. El maíz. Aspecto biológicos. In Origen y diversificación del maíz: una revisión analítica (eds Kato TA, Mapes C, Mera LM, Serratos JA, Bye RA), pp. 19–32. México, DF: Universidad Nacional Autónoma de México, Comisión Nacional para el Conocimiento y Uso de la Biodiversidad, Editorial Impresora Apolo. [Google Scholar]
- 12.SIAP. 2007. Situación actual y perspectivas del maíz en México 1996–2012. México, DF: Sistema de Información Agroalimentaria y Pesquera; See http://www.campomexicano.gob.mx/portal_siap/Integracion/EstadisticaDerivada/ComercioExterior/Estudios/Perspectivas/maiz96-12.pdf. [Google Scholar]
- 13.Bellon MR, Brush SB. 1994. Keepers of maize in Chiapas, Mexico. Econ. Bot. 48, 196–209. ( 10.1007/BF02908218) [DOI] [Google Scholar]
- 14.Perales H, Brush SB, Qualset CO. 2003. Landraces of maize in central Mexico: an altitudinal transect. Econ. Bot. 57, 7–20. [Google Scholar]
- 15.Pressoir G, Berthaud J. 2004. Patterns of population structure in maize landraces from the Central Valleys of Oaxaca in Mexico. Heredity 92, 88–94. ( 10.1038/sj.hdy.6800387) [DOI] [PubMed] [Google Scholar]
- 16.Pressoir G, Berthaud J. 2004. Population structure and strong divergent selection shape phenotypic diversification in maize landraces. Heredity 92, 95–101. ( 10.1038/sj.hdy.6800388) [DOI] [PubMed] [Google Scholar]
- 17.Perales HR, Benz BF, Brush SB. 2005. Maize diversity and ethnolinguistic diversity in Chiapas, Mexico. Proc. Natl Acad. Sci. USA 102, 949–954. ( 10.1073/pnas.0408701102) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Orozco-Ramírez Q, Ross-Ibarra J, Santacruz-Varela A, Brush S. 2016. Maize diversity associated with social origin and environmental variation in southern Mexico. Heredity 116, 477–484. ( 10.1038/hdy.2016.10) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bellon MR, Dulloo E, Thormann I, Sardos J, Burdon J. 2017. In-situ conservation—harnessing natural and human derived evolutionary forces to ensure future crop adaptation. Evol. Appl. 10, 965–977. ( 10.1111/eva.12521) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Diario Oficial de la Federación. 2013. DECRETO por el que se aprueba el Programa Sectorial de Desarrollo Agropecuario, Pesquero y Alimentario 2013–2018. See http://www.sagarpa.gob.mx/ganaderia/Documents/2015/MANUALES%20Y%20PLANES/Programa_Sectorial_SAGARPA_2013-2018%20(1).pdf.
- 21.Fox J, Haight L. 2010. Mexican agricultural policy: multiple goals and conflicting interests. In Subsidizing inequality: Mexican corn policy since NAFTA (eds Fox J, Haight L), pp. 9–50. Santa Cruz, CA: Woodrow Wilson International Center for Scholars. [Google Scholar]
- 22.Levy S, van Wijnbergen S. 1992. Maize and the Free Trade Agreement between Mexico and the United States. World Bank Econ. Rev. 6, 481–502. ( 10.1093/wber/6.3.481) [DOI] [Google Scholar]
- 23.Eakin H, Perales H, Appendini K, Sweeney S. 2014. Selling maize in Mexico: the persistence of peasant farming in an era of global markets. Dev. Change 45, 133–155. ( 10.1111/dech.12074) [DOI] [Google Scholar]
- 24.Yuñez NA. 2010. Las políticas públicas dirigidas al sector rural: el cáracter de las reformas para el cambio estructural. In Los grandes problemas de México. XI economía rural (ed. Yuñez-Naude A.), pp. 23–62. México, DF: El Colegio de México. [Google Scholar]
- 25.Eakin H, Appendini K, Sweeney S, Perales H. 2015. Correlates of maize land and livelihood change among maize farming households in Mexico. World Dev. 70, 78–91. ( 10.1016/j.worlddev.2014.12.012) [DOI] [Google Scholar]
- 26.de Janvry A, Sadoulet E. 2001. Income strategies among rural households in Mexico: the role of off-farm activities. World Dev. 29, 467–480. ( 10.1016/S0305-750X(00)00113-3) [DOI] [Google Scholar]
- 27.Yúnez-Naude A, Rivera-Ramírez F, Chávez-Alvarado MA, Mora-Rivera JJ, Taylor JE (eds). 2015. La Economía del Campo Mexicano: Tendencias y Retos para su Desarrollo. México, DF: El Colegio de México. [Google Scholar]
- 28.Bellon MR. 1996. The dynamics of crop infraspecific diversity: conceptual framework at the farmer level. Econ. Bot. 50, 26–39. ( 10.1007/BF02862110) [DOI] [Google Scholar]
- 29.Appendini K, Quijada MG. 2016. Consumption strategies in Mexican rural households: pursuing food security with quality. Agric. Hum. Values 33, 439–454. ( 10.1007/s10460-015-9614-y) [DOI] [Google Scholar]
- 30.Dyer G, Taylor JE, Boucher S. 2006. Subsistence response to market shocks. Am. J. Agric. Econ. 88, 279–291. ( 10.1111/j.1467-8276.2006.00858.x) [DOI] [Google Scholar]
- 31.Hellin J, Orenstein O, Beuchelt T, Camacho C, Flores D. 2013. Maize stover use and sustainable crop production in mixed crop-livestock systems in Mexico. Field Crop Res. 153, 12–21. ( 10.1016/j.fcr.2013.05.014) [DOI] [Google Scholar]
- 32.Keleman A, Hellin J. 2009. Specialty maize varieties in Mexico: a case study in market-driven agro-biodiversity conservation. J. Lat. Am. Geogr. 8, 147–174. [Google Scholar]
- 33.Gordillo de Anda G, de Janvry A, Sadoulet E. 1999. La Segunda Reforma Agraria de México: Respuestas de Familias y Comunidades, 1990–1994. México, DF: Fondo de Cultura Económica. [Google Scholar]
- 34.SIAP. 2016. Norma técnica para la generación de estadística básica agropecuaria y pesquera, pp. 348–355. México, DF: Secretaria de Agricultura, Ganadería, Desarrollo Rural, Pesca y Alimentación; See http://www.gob.mx/siap/normatividad-estadistica. [Google Scholar]
- 35.Cuervo-Robayo AP, Tellez-Valdez O, Gomez-Albores MA, Venegas-Barrera CS, Manjarrez J, Martinez M. 2014. An update of high-resolution monthly climate surfaces for Mexico. Int. J. Climatol. 34, 2427–2437. ( 10.1002/joc.3848) [DOI] [Google Scholar]
- 36.Guevara M, Arroyo-Cruz CE. 2016. Modelo digital de elevaciones corregido resolución 1 km, escala: 1 : 4 000 000, edición: 1. Cd de México, Mexico: Comisión Nacional para el Conocimiento y Uso de la Biodiversidad. [Google Scholar]
- 37.Guevara M, Arroyo-Cruz CE. 2016. ‘Pendiente’, escala: 1 : 4 000 000, edición: 1. Cd de México, Mexico: Comisión Nacional para el Conocimiento y Uso de la Biodiversidad. [Google Scholar]
- 38.INEGI. 2015. Conjunto de datos vectoriales de uso de suelo y vegetación escala 1 : 250 000, serie V (capa unión). México, DF: Instituto Nacional de Estadística y Geografía. [Google Scholar]
- 39.Feltz CJ, Miller GE. 1996. An asymptotic test for the equality of coefficients of variation from k populations. Stat. Med. 15, 647–658. ( 10.1002/(SICI)1097-0258(19960330)15:6%3C647::AID-SIM184%3E3.0.CO;2-P) [DOI] [PubMed] [Google Scholar]
- 40.Anderson MJ. 2006. Distance-based tests for homogeneity of multivariate dispersions. Biometrics 62, 245–253. ( 10.1111/j.1541-0420.2005.00440.x) [DOI] [PubMed] [Google Scholar]
- 41.R Core Team. 2012. R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; See http://www.R-project.org. [Google Scholar]
- 42.Hijmans RJ, et al. 2014. raster: geographic data analysis and modeling. See http://cran.at.r-project.org/web/packages/raster/index.html.
- 43.Bivand R, et al. 2015. maptools: tools for reading and handling spatial objects. See http://cran.r-project.org/web/packages/maptools/index.html.
- 44.Marwick B, Krishnamoorthy K.. 2016. cvequality: tests for the equality of coefficients of variation from multiple groups. See https://cran.r-project.org/web/packages/cvequality/index.html.
- 45.Oksanen J, et al. 2016. vegan: community ecology package. See https://cran.r-project.org/web/packages/vegan/index.html.
- 46.Husson F, Josse J, Le S, Mazet J.. 2017. FactoMineR: multivariate exploratory data analysis and data mining. See https://CRAN.R-project.org/package=FactoMineR.
- 47.Caballero A. 1994. Developments in the prediction of effective population size. Heredity 73, 657–679. ( 10.1038/hdy.1994.174) [DOI] [PubMed] [Google Scholar]
- 48.Gillespie JH. 1998. Population genetics: a concise guide. Baltimore, MD: Johns Hopkins University Press. [Google Scholar]
- 49.Jiao Y, et al. 2012. Genome-wide genetic changes during modern breeding of maize. Nat. Genet. 44, 812–815. ( 10.1038/ng0914-1039) [DOI] [PubMed] [Google Scholar]
- 50.Schnable PS, et al. 2009. The B73 maize genome: complexity, diversity, and dynamics. Science 326, 1112–1115. ( 10.1126/science.1178534) [DOI] [PubMed] [Google Scholar]
- 51.CONEVAL. 2012. Construcción de las líneas de bienestar: documento metodológico. México, DF: Consejo Nacional de Evaluación de la Política de Desarrollo Social. [Google Scholar]
- 52.Mercer KL, Martínez-Vásquez A, Perales HR. 2008. Asymmetrical local adaptation of maize landraces along an altitudinal gradient. Evol. Appl. 1, 489–500. ( 10.1111/j.1752-4571.2008.00038.x) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ruiz CJA, et al. 2008. Climatic adaptation and ecological descriptors of 42 Mexican maize races. Crop Sci. 48, 1502–1512. ( 10.2135/cropsci2007.09.0518) [DOI] [Google Scholar]
- 54.Wang L, Beissinger TM, Lorant A, Rossi-Ibarra C, Ross-Ibarra J, Hufford MB. 2017. The interplay of demography and selection during maize domestication and expansion. Genome Biol. 18, 215 ( 10.1186/s13059-017-1346-4) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Beissinger TM, Wang L, Crosby K, Durvasula A, Hufford MB, Ross-Ibarra J. 2016. Recent demography drives changes in linked selection across the maize genome. Nat. Plants 2, 16084 ( 10.1038/nplants.2016.84) [DOI] [PubMed] [Google Scholar]
- 56.Faith DP, Magallon S, Hendry AP, Conti E, Yahara T, Donoghue MJ. 2010. Evosystem services: an evolutionary perspective on the links between biodiversity and human well-being. Curr. Opin. Environ. Sustain. 2, 1–9. ( 10.1016/j.cosust.2010.04.002) [DOI] [Google Scholar]
- 57.Bellon MR, Gotor E, Caracciolo F. 2015. Conserving landraces and improving livelihoods: how to assess the success of on-farm conservation projects? Int. J. Agr. Sustain. 13, 167–182. ( 10.1080/14735903.2014.986363) [DOI] [Google Scholar]
- 58.Barrett R, Schluter D. 2008. Adaptation from standing genetic variation. Trends Ecol. Evol. 23, 38–44. ( 10.1016/j.tree.2007.09.008) [DOI] [PubMed] [Google Scholar]
- 59.Lourenço JM, Glémin S, Galtier N. 2013. The rate of molecular adaptation in a changing environment. Mol. Biol. Evol. 30, 1292–1301. ( 10.1093/molbev/mst026) [DOI] [PubMed] [Google Scholar]
- 60.Gossmann TI, Keightley PD, Eyre-Walker A. 2012. The effect of variation in the effective population size on the rate of adaptive molecular evolution in Eukaryotes. Genome Biol. Evol. 4, 658–667. ( 10.1093/gbe/evs027) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lanfear R, Kokko H, Eyre-Walker A. 2014. Population size and the rate of evolution. Trends Ecol. Evol. 29, 33–41. ( 10.1016/j.tree.2013.09.009) [DOI] [PubMed] [Google Scholar]
- 62.Dwivedi SL, Ceccarelli S, Blair MW, Upadhyaya HD, Are AK, Ortiz R. 2016. Landrace germplasm for improving yield and abiotic stress adaptation. Trends Plant Sci. 21, 31–42. ( 10.1016/j.tplants.2015.10.012) [DOI] [PubMed] [Google Scholar]
- 63.Pritchard JK, Di Rienzo A. 2010. Adaptation—not by sweeps alone. Nat. Rev. Genet. 11, 665–667. ( 10.1038/nrg2880) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Gillespie JH. 1984. Molecular evolution over the mutational landscape. Evolution 38, 1116–1129. ( 10.2307/2408444) [DOI] [PubMed] [Google Scholar]
- 65.Orr HA. 2003. The distribution of fitness effects among beneficial mutations. Genetics 163, 1519–1526. ( 10.1016/j.jtbi.2005.05.001) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Eyre-Walker A, Keightley PD. 2007. The distribution of fitness effects of new mutations. Nat. Rev. Genet. 8, 610–618. ( 10.1038/nrg2146) [DOI] [PubMed] [Google Scholar]
- 67.FAOSTAT. 2018. Statistics division. See http://faostat.fao.org. Accessed July 2018.
- 68.Smith JSC, Gardner CAC, Costich DE. 2017. Ensuring the genetic diversity of maize and its wild relatives. In Achieving sustainable cultivation of maize. Volume 1: from improved varieties to local applications (ed. Watson D.), pp. 3–50. Cambridge, UK: Burleigh Dodds Science Publishing. [Google Scholar]
- 69.Bruford MW, Davies N, Dulloo ME, Faith DP, Walters M. 2017. Monitoring changes in genetic diversity. In The Geo handbook on biodiversity observation networks (eds Walters M, Scholes RJ), pp. 107–128. Cham, Germany: Springer. [Google Scholar]
- 70.Perales H. 2016. Landrace conservation of maize in Mexico and evolutionary breeding. In Enhancing crop genepool use: capturing wild relative and landrace diversity for crop improvement (eds Maxted N, Dulloo ME, Ford-Lloyd BV), pp. 271–281. Wallingford, UK: CAB Intl. [Google Scholar]
- 71.Arslan A, Taylor JE. 2009. Farmers' subjective valuation of subsistence crops: the case of traditional maize in Mexico. Am. J. Agric. Econ. 91, 956–972. ( 10.1111/j.1467-8276.2009.01323.x) [DOI] [Google Scholar]
- 72.Perales H, Golicher D. 2014. Mapping the diversity of maize races in Mexico. PLoS ONE 9, 1–20. ( 10.1371/journal.pone.0114657) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Bellon MR, Hodson D, Bergvinson D, Beck D, Martinez-Romero E, Montoya Y. 2005. Targeting agricultural research to benefit poor farmers: relating poverty mapping to maize environments in Mexico. Food Policy 30, 476–492. ( 10.1016/j.foodpol.2005.09.003) [DOI] [Google Scholar]
- 74.CONABIO. 2017. Ecosystems and agro-biodiversity across small and large-scale maize production systems. Geneva, Switzerland: TEEB Agriculture & Food, UNEP; See http://teebweb.org/agrifood/home/maize. [Google Scholar]
- 75.Bellon MR, Mastretta-Yanes A, Ponce-Mendoza A, Ortiz-Santamaría D, Oliveros-Galindo O, Perales H, Acevedo F, Sarvkhán J. 2018. Data from: Evolutionary and food supply implications of ongoing maize domestication by Mexican campesinos Dryad Digital Repository. ( 10.5061/dryad.79q870b) [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Citations
- Bellon MR, Mastretta-Yanes A, Ponce-Mendoza A, Ortiz-Santamaría D, Oliveros-Galindo O, Perales H, Acevedo F, Sarvkhán J. 2018. Data from: Evolutionary and food supply implications of ongoing maize domestication by Mexican campesinos Dryad Digital Repository. ( 10.5061/dryad.79q870b) [DOI] [PMC free article] [PubMed]
Supplementary Materials
Data Availability Statement
Data, spreadsheets and R scripts used for analyses are available at the Dryad repository: http://dx.doi.org/10.5061/dryad.79q870b [75].