Key Points
Question
Is topical rapamycin an effective and safe for treatment of tuberous sclerosis complex (TSC)-related facial angiofibromas?
Findings
This randomized clinical trial of 179 patients showed clinically meaningful and statistically significant improvement in the appearance of TSC-related facial angiofibromas were observed for both the 1% rapamycin and 0.1% rapamycin treatment groups vs the vehicle-only group, and also for 1% vs 0.1% rapamycin. The topical application of rapamycin was well tolerated and systemic absorption was not observed.
Meaning
Topical rapamycin may be used to treat TSC-related facial angiofibromas; in this trial, the preferred dose was 1% (0.3 g rapamycin per 30 g vehicle).
This randomized clinical trial examines the efficacy and safety of topical rapamycin therapy for treatment of patients with tuberous sclerosis complex–related facial angiofibromas.
Abstract
Importance
Facial angiofibromas occur in approximately 75% of individuals with tuberous sclerosis complex (TSC), causing substantial morbidity and disfigurement. Current therapies are partially effective, uncomfortable, produce scarring, and need repeating to treat recurrence.
Objective
To evaluate the efficacy and safety of topical rapamycin for TSC-related facial angiofibromas.
Design, Setting, and Participants
This prospective, multicenter, randomized, double-blind, vehicle-controlled trial with 6 monthly clinic visits enrolled 179 patients with TSC-related facial angiofibromas not treated within 6 months from May 2012 to March 2014 in 9 clinical sites in the United States and 1 in Australia.
Interventions
Patients were randomized (1:1:1) to topical formulation containing 0.3 g per 30 g (1%) rapamycin, 0.03 g per 30 g (0.1%) rapamycin, or vehicle alone. Participants applied 1.0 mL to designated areas daily at bedtime.
Main Outcomes and Measures
Angiofibroma Grading Scale (AGS) change from baseline scored from photographs by independent masked dermatologists. Safety analyses included adverse events (AEs) and serum rapamycin levels.
Results
All 179 patients randomized (99 [55.3%] female) comprised the primary analysis population (59 in the 1% rapamycin group, 63 in the 0.1% rapamycin group, and 57 in the vehicle-only group). The mean age was 20.5 years (range 3-61 years). Clinically meaningful and statistically significant improvement in facial angiofibromas was observed for both 1% and 0.1% rapamycin relative to the vehicle-only control group, and for 1% vs 0.1% rapamycin, with most of the improvement realized within the first month. At 6 months, AGS mean improvement for 1% rapamycin was 16.7 points compared with 11.0 for 0.1% rapamycin and 2.1 points for vehicle only (P < .001 for 1% and 0.1% vs vehicle only). Compared with baseline, end-of-treatment photos were rated “better” for 81.8% of patients in the 1% rapamycin group, compared with 65.5% for those in the 0.1% rapamycin group and 25.5% for those in the vehicle-only group (P < .001, all 3 pairwise comparisons). Topical rapamycin was generally well-tolerated, with no measurable systemic absorption. Apparent drug-related adverse effects were limited to 10% or less incidence of application site discomfort and/or pain, pruritus, erythema, and irritation. Nearly all AEs were mild, with no drug-related moderate, severe, or serious events.
Conclusions and Relevance
Topical rapamycin appears effective and safe for treatment of TSC-related facial angiofibromas. In this trial, the preferred dose was 1% once daily. Future studies are needed to evaluate prophylactic, early, and long-term use of topical rapamycin, durability of response, and combination therapy with oral mammalian target of rapamycin (mTOR) inhibitors.
Trial Registration
ClinicalTrials.gov Identifier: NCT01526356
Introduction
Tuberous sclerosis complex (TSC) is an autosomal dominant genetic disorder that results in dysregulation of the mammalian target of rapamycin (mTOR) signaling pathway.1,2,3 Symptoms vary among individuals, ranging from mild to severe, and frequently include skin lesions, kidney, heart, and brain tumors, lung disease, seizures, and intellectual disability. Facial angiofibromas occur in approximately 75% of individuals, are associated with substantial morbidity and disfigurement, and negatively impact quality of life (QOL).4,5,6,7
Rapamycin inhibits mTOR activity.3,8 Trials of orally ingested mTOR inhibitors, including rapamycin and its derivatives, have demonstrated positive results for TSC-related subependymal giant cell astrocytomas, renal angiomyolipomas, and lymphangioleiomyomatosis.9,10,11,12 Franz et al10 reported a partial response of TSC-related skin lesions. Oral mTOR inhibitors, however, can be associated with serious adverse effects,9,10 are not approved for dermatologic conditions, and are not indicated in all individuals with TSC.
Tuberous sclerosis complex-related facial angiofibromas have been treated by laser surgery, cryotherapy, dermabrasion, or similar approaches. These treatments can be painful, may lead to scarring, and do not prevent recurrence of lesions.13,14,15 Early reports of topical rapamycin treatment of TSC-related facial angiofibromas suggested efficacy.16,17,18,19,20,21,22,23,24,25,26,27,28 In 2012, Koenig et al21 reported a phase 1 randomized clinical trial in which 73% of patients in the treatment groups reported a perceived improvement in the appearance of their facial angiofibromas compared with 38% of patients in the vehicle-only group. In 2017, Wataya-Kaneda et al27 reported a phase 2 randomized clinical trial showing statistically significant improvements for adults in the high-dose treatment group and children in all treatment groups. No study-related serious adverse events (AEs) occurred in either study but Wataya-Kaneda et al21,27 detected low-level systemic absorption of rapamycin in 10 patients.
The purpose of the Topical Rapamycin to Erase Angiofibromas in TSC—Multicenter Evaluation of Novel Therapy (TREATMENT) Trial was to assess the efficacy and safety of topical rapamycin therapy for treatment of patients with TSC-related facial angiofibromas in a large population.
Methods
Study Design
This was a prospective, multicenter, randomized, double-blind, vehicle-controlled trial (protocol provided in Supplement 1). Patients were recruited from 9 clinical sites in the United States and 1 in Australia. The primary study site was the University of Texas Tuberous Sclerosis Center at the McGovern Medical School, The University of Texas Health Science Center at Houston (UTHealth). A list of collaborators and additional study sites is provided at the end of the text. This study was approved by the UTHealth Committee for the Protection of Human Subjects and site institutional review boards (IRBs), as detailed in Supplement 2. This trial was registered with ClinicalTrials.gov, identifier NCT01526356.
Study Population
Eligible patients had a diagnosis of TSC,5 visible facial angiofibromas, and the ability to comply with trial procedures. Patients were excluded if they were receiving rapamycin or immunosuppressive therapy, had received an oral mTOR inhibitor at any time, were pregnant or nursing, or had immune dysfunction, a dermatologic condition that could interfere with study assessments, known hypersensitivity to the topical formulation or rapamycin, undergone laser surgery, cryotherapy, or other dermatologic treatment for their facial angiofibromas within the preceding 6 months, or participated in any interventional clinical trial within the preceding 30 days. Female patients of child-bearing potential were tested for pregnancy prior to enrollment and required to use appropriate contraceptive methods during the trial. Patients were enrolled by individual sites. Written informed consent was obtained from patients or, in the case of minors or cognitively impaired patients, a legal guardian with assent of the patient.
Randomization and Masking
Enrolled patients were randomized (1:1:1) to receive rapamycin (1% or 0.1%) or vehicle-only using a computerized random number generator. Stratification by site was performed and each site’s randomization was performed in block sizes of 6. Randomization was performed centrally according to study identification (ID) number by personnel not involved with recruitment or analysis. Treatment allocation by study ID number allowed the research pharmacy to prepare bottles of investigational study drug product and ship the product to collaborating sites prior to enrollment. On enrollment, patients were assigned a unique study ID number and given the appropriate bottle of study product. All bottles of experimental product were identical in size, appearance, and texture. Product was contained in amber bottles, stored and shipped at ambient temperature, and labeled identically except for study ID number. The allocation key was kept by the compounding pharmacy until completion of the study. Blinding was maintained throughout the study.
Interventions
At enrollment, each patient’s facial angiofibromas were assessed by investigators and baseline lesion distribution documented. Standardized frontal and 45° oblique digital facial photographs were taken as described in Supplement 2. Patients were provided the experimental product and instructed to apply 1 pump (1.0 mL) of product at bedtime to areas designated at the time of enrollment, for 6 months. Active treatment groups received a topical formulation containing either 0.3 g per 30 g (1%) or 0.03 g per 30 g (0.1%) rapamycin/vehicle. The vehicle-only group received only vehicle (no rapamycin). All bottles of study drug were identical in appearance and produced centrally by the research pharmacy. Patients were instructed to store the experimental product at room temperature in a controlled location.
Depending on age and cognitive ability, patients completed the Dermatology Life Quality Index (DLQI) or the Children’s DLQI (CDLQI), and/or a guardian/representative completed the Family DLQI (FDLQI).28,29,30,31 These QOL questionnaires were completed at baseline, 3 months, and 6 months.
Patients were to return monthly to the study site for digital photographs, physical examination, recording of AEs and concomitant medication, and, if applicable, pregnancy testing. Patients were assessed visually for hypersensitivity to the experimental product and signs of immunosuppression in the adjacent skin or mucous membranes. Unused study drug was collected and a new bottle of study drug dispensed.
Serum rapamycin was assessed in patient blood samples by immunoassay (lower limit of detection <0.5 ng/mL). Initially, blood samples were collected monthly from all patients. Blood draws were performed during clinic visits, which typically occurred in the morning. After rapamycin was not detected in the first several hundred samples, monthly blood sampling was discontinued by recommendation of the Data and Safety Monitoring Board (DSMB), with approval of involved IRBs (data and safety monitoring described in Supplement 2). However, serum rapamycin levels were assayed in all patients at the end of trial.
Outcome Measures
All photographs were evaluated independently by 3 board-certified dermatology readers masked to treatment assignment, visit, and chronology of the photographs, and not involved in any other aspect of the study. The photo readers determined unreadable photos, and therefore patient visits unevaluable for analysis. An algorithm and adjudication rules were used to derive a single consensus score for each patient visit from the 3 readers’ scores.
The primary outcome was change from baseline in the Angiofibroma Grading Scale (AGS) (eTable 1 in Supplement 2). Secondary outcomes consisted of photo readers’ rating (better, same, or worse) of paired baseline and end-of-trial (EOT) photographs for each patient, and QOL questionnaire scores change from baseline.
All patients were included in safety analyses including AEs and assessment of serum rapamycin levels. All AEs were described clinically, evaluated for severity, seriousness, and study relatedness, and coded to MedDRA dictionary terminology. The AEs at the site of application of study product included the phrase “at application site.”
Statistical Analysis
Statistical processing was performed using SAS statistical software (version 9.4 or higher; SAS Institute, Inc). Prestudy sample size estimation determined 50 patients were needed in each treatment group to achieve 80% power to detect an 8-point difference in AGS change assuming a standard deviation (SD) of 9 and 2-sided α of .05. The AGS was analyzed primarily using a restricted maximum likelihood-based repeated measures linear model including fixed effects for treatment group, site, visit, and treatment by visit interaction, with baseline disease severity as a covariate. An analysis of covariance (ANCOVA) model was also used, including fixed effects for treatment group and site, with baseline disease severity score as a covariate, using both last observation carried forward (LOCF) and observed-only values. In addition, a global test for any differences between treatment groups was obtained from the type 3 sums-of-squares table, and the Kruskal-Wallis test used as a global test to test if the mean ranks for disease severity were the same among the 3 treatment groups at each of the study visits.
A secondary outcome was the photo readers’ rating (better, same, or worse) of paired baseline and end of trial photographs for each patient. Readers were shown side-by-side images to compare, but remained unaware of treatment assignment and chronology of images (before vs end of treatment). Overall treatment difference and pairwise comparisons were tested using the Cochran-Mantel-Haenszel (CMH) test stratified by study site with modified ridit scores at the 2-sided α level of significance of .05.
An additional secondary outcome was QOL questionnaire scores change from baseline. The analysis methods for QOL were the same as for AGS, but with baseline QOL score as a covariate.
A 2-sided P = .05 was considered statistically significant for all tests. No adjustments to P values were made to address multiplicity of testing.
Results
A total of 179 patients were enrolled and randomized from May 2012 through March 2014: 59 (33.0%) to 1% rapamycin treatment, 63 (35.2%) to 0.1% rapamycin, and 57 (31.8%) to vehicle only (Figure 1). The treatment groups were generally well balanced for baseline characteristics (eTable 2 in Supplement 2). Of the 179 enrolled patients, 99 (55.3%) were female and 80 (44.7%) male. The mean age was 20.5 years (range 3-61 years). The race/ethnicity distribution was predominantly white (121 [67.6%]), followed by black (14 [7.8%]), mixed white/Hispanic (6 [3.4%]), and then various other combinations. For AGS, with a possible range of 0 to 202, the mean baseline score was 46.4 (range 2.7-132.5).
Figure 1. The TREATMENT Trial CONSORT Diagram.
AGS indicates angiofibroma grading scale; TSC, tuberous sclerosis complex.
aApplication site rash, mild, probably related to study drug.
bPhlebitis superficial, moderate, not related to study drug.
cAll patients excluded on the basis of lacking either baseline or at least 1 postbaseline evaluable photograph.
For the AGS analysis, 159 (84.9%) of 179 patients had evaluable photos at both baseline and at least 1 postbaseline visit. Use of 1% rapamycin (resulting in, at visit 7 for example, a mean 16.7-point improvement) was statistically superior to vehicle only (2.1-point visit 7 mean improvement) overall (P < .001) and at every individual visit (Figure 2). Use of 0.1% rapamycin (11.0-point visit 7 mean improvement) was statistically superior to vehicle only overall (P = .01), and at P ≤ .006 at every individual visit except visit 2 (P = .07). Use of 1% rapamycin was visually superior to 0.1% rapamycin but not statistically significantly superior. These results from the repeated measures linear model are supported by similar findings from the ANCOVA model, both using LOCF and observed-only values, and also the Kruskal-Wallis test.
Figure 2. AGS Change From Baseline by Treatment Group.
Restricted maximum likelihood-based repeated measures linear model. AGS indicates angiofibroma grading scale; LS, least square.
Paired baseline and end-of-trial (EOT) photographs were available for side-by-side rating of same, better, or worse for 164 (91.6%) of 179 patients. The EOT photo was rated better than the baseline photo in the 1% rapamycin group for 45 (81.8%) of 55 patients, compared with 38 (65.5%) of 58 patients in the 0.1% rapamycin group and 13 (25.5%) of 51 in the vehicle-only group (Figure 3). The EOT photo was rated the same as the baseline photo in the 1% rapamycin group for 5 (9.1%) of 55 patients, compared with 11 (19.0%) of 58 in the 0.1% rapamycin group and 15 (29.4%) of 51 in the vehicle-only group. The EOT photo was rated worse than the baseline photo in the 1% rapamycin group for 5 (9.1%) of 55 patients, compared with 9 (15.5%) of 58 in the 0.1% rapamycin group and 23 (45.1%) of 51 in the vehicle-only group. Overall, 1% rapamycin and 0.1% rapamycin were each highly statistically superior to vehicle-only (P < .001) and the 1% rapamycin was statistically superior to the 0.1% rapamycin (P = .03).
Figure 3. Baseline to End-of-Study Paired Photographs Comparison.
Cochran-Mantel-Haenszel test.
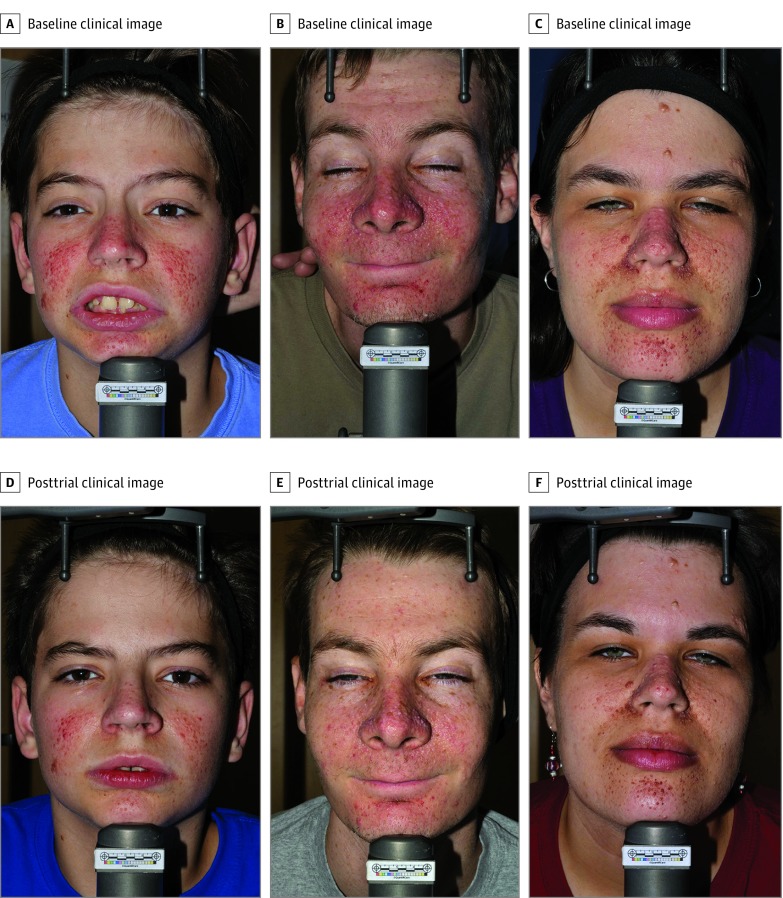
Representative patient photographs are shown in Figure 4 (additional patient photographs are provided in eFigures 1, 2, and 3 in Supplement 2).
Figure 4. Patient Photographs.
Frontal photographs taken at baseline (A-C) and paired with end of trial photographs (D-F) for 3 patients in the 1% rapamycin treatment group.
The QOL scores (possible range 0 to 30) in this study were low at baseline, with mean scores of 3.6 for DLQI, 2.8 for CDLQI, and 4.1 for FDLQI. These scores are in the severity banding range of “small effect” on QOL (2 to 5 points). More than a quarter of patients in the study were rated with a score of 0: 28.6% (26/91) for DLQI, 32.5% (13/40) for CDLQI, and 17.6% (16/91) for FDLQI. Approximately 41% were rated in the severity banding range “no effect” on quality of life (0 to 1 point): 48.4% (44/91) for DLQI, 42.5% (17/40) for CDLQI, and 31.9% (29/91) for FDLQI. This leaves less than 60% of patients in the study who could have experienced any improvement in QOL. In general, QOL scores decreased substantially from baseline to visit 4 and often further to visit 7. The observed change from baseline to visit 4 may represent a regression to the mean phenomenon. There was no apparent rapamycin treatment effect and no statistically significant differences among the treatment groups for DLQI, CDLQI, or FDLQI.
All patients were evaluated for safety. The overall rate of completion of this 6-month study was 83.2%, and highest for 1% rapamycin (86.4%), followed by 0.1% rapamycin (84.1%) and vehicle-only (78.9%) (Figure 1). Two patients were withdrawn owing to an AE, only 1 of which was a suspected drug reaction (mild application site cutaneous eruption). The incidence of individual AEs, patients with at least 1 AE, and patients with at least 1 AE at least possibly related to study drug all increased as function of rapamycin concentration (Table). There were no deaths or drug-related serious AEs. Nearly all AEs were mild, and there were no drug-related moderate or severe AEs. Based on a comparison of incidence, the AEs most likely attributable to the addition of 1% rapamycin to the vehicle formulation were application site pain, application site pruritus, application site erythema, application site irritation, and possibly sunburn. The data suggest that application site acne and acne may be associated with the vehicle itself, and not the addition of rapamycin. No apparent hypersensitivity or allergic-type AEs were reported. The application of study drug did not appear to predispose to cutaneous infection. Systemic absorption of rapamycin was not detected in any of the more than 500 blood samples assayed.
Table. Adverse Events.
| Event | No. (%) | ||
|---|---|---|---|
| 1% Rapamycin (n = 59) | 0.1% Rapamycin (n = 63) | Vehicle Only (n = 57) | |
| Individual AEs | 44/99 (44.4) | 36/99 (36.4) | 19/99 (9.2) |
| Patients with ≥1 AE | 24 (40.7) | 19 (30.2) | 11 (19.3) |
| Patients with ≥1 drug-related AEa | 18 (30.5) | 11 (17.5) | 6 (10.5) |
| Patients with ≥1 AE grade 3 or 4b | 1 (1.7) | 1 (1.6) | 2 (3.5) |
| Patients with ≥1 serious AEb | 2 (3.4) | 1 (1.6) | 1 (1.8) |
| Most common AEsc | |||
| Application site pain | 6 (10.2) | 2 (3.2) | 1 (1.8) |
| Application site pruritus | 5 (8.5) | 0 | 1 (1.8) |
| Application site acne | 3 (5.1) | 1 (1.6) | 1 (1.8) |
| Cutaneous eruption | 1 (1.7) | 2 (3.2) | 1 (1.8) |
| Pruritus | 0 | 2 (3.2) | 2 (3.5) |
| Application site erythema | 2 (3.4) | 1 (1.6) | 0 |
| Sunburn | 2 (3.4) | 1 (1.6) | 0 |
| Application site irritation | 1 (1.7) | 2 (3.2) | 0 |
| Acne | 0 | 2 (3.2) | 1 (1.8) |
| Nasopharyngitis | 2 (3.4) | 0 | 0 |
| Nasal congestion | 2 (3.4) | 0 | 0 |
| Application site papules | 1 (1.7) | 1 (1.6) | 0 |
| Sinusitis | 1 (1.7) | 1 (1.6) | 0 |
| Application site paraesthesia | 1 (1.7) | 0 | 1 (1.8) |
| Nausea | 0 | 2 (3.2) | 0 |
| Mouth ulceration | 0 | 1 (1.6) | 1 (1.8) |
| Cough | 0 | 1 (1.6) | 1 (1.8) |
Abbreviation: AEs, adverse events.
Drug-related AEs are those assessed by the investigator as possibly, probably, or definitely related to study drug, and does not include AEs assessed as not related or unknown.
None assessed as related to study drug.
AEs experienced by 2 patients or more (≥1.1%) in the study, regardless of assessed relatedness to study drug, by MedDRA dictionary preferred terms.
Discussion
To our knowledge, the TREATMENT Trial is the largest randomized clinical trial to date to evaluate efficacy and safety of topical rapamycin treatment of TSC-related facial angiofibromas.16,17,18,19,20,21,22,23,24,25,26,27,28 Clinically meaningful and statistically significant improvement in the appearance of facial angiofibromas was observed for both 1% and 0.1% rapamycin treatment groups relative to the vehicle-only control, and for 1% vs 0.1% rapamycin, with most of the improvement realized within the first month.
The safety findings from this study reinforce the findings from previous clinical trials and published literature of topical rapamycin. Topical rapamycin was generally well-tolerated and demonstrated a favorable AE profile, with no measurable systemic absorption and with apparent drug-related AEs limited to 10% or lower incidence of discomfort/pain, pruritus, erythema, and irritation at the application site. Nearly all AEs were mild severity, and there were no drug-related serious events or drug-related moderate or severe events. Only 1 patient was withdrawn from the study owing to an AE, for a mild facial cutaneous eruption.
The findings of this trial extend the available knowledge of topical rapamycin, attesting to the generalizability of findings.16,17,18,19,20,21,22,23,24,25,26,27,28 Serious AEs occurred in this TREATMENT Trial as well as in prior studies but none were considered related to treatment.16,20,21,27 Treatment-related AEs were generally mild and dermatologic in nature, occurring at or near the site of application of the topical formulations.16,17,18,20,21,22,23,26,27 Rarely did such events prompt patients to cease therapy.21 Hence, topical rapamycin treatment appears to present a satisfactory balance of benefits to harms.16,17,18,19,20,21,22,23,24,25,26,27,28
Franz et al10 demonstrated oral mTOR inhibitors are partially effective for treatment of TSC-related skin lesions. Patients receiving oral mTOR inhibitor therapy were excluded from the TREATMENT Trial; however, because no systemic absorption of rapamycin was observed, it is worthwhile to contemplate whether topical rapamycin might be safe and/or at least additive as adjunctive therapy for patients receiving oral mTOR inhibitors.16,17,18,19,20,21,22,23,24,25,26,27,28 No studies to date have addressed whether concomitant use of topical and oral mTOR inhibitors might lead to high levels of the drug in the skin or an increased risk for or exacerbation of AEs.
A limited number of small studies have evaluated the use of topical rapamycin for treatment of other TSC-related skin lesions.32,33,34,35 The efficacy and safety of topical rapamycin with respect to these lesions remains uncertain.
Systemic use of rapamycin is associated with a variety of adverse reactions.36 Some adverse reactions, such as hypersensitivity and increased susceptibility to infection and skin cancer, may also be relevant with chronic use of topical rapamycin.
Limitations
There are caveats and limitations to this study. Although the study was terminated before the prespecified enrollment of 230 patients, the enrolled patient sample size provided sufficient statistical power for the study. Although some efficacy analyses showed continued small incremental improvement through the last visit on study, the 6-month duration of the trial appears appropriate for the evaluation of most of the effect of the study drug. The DLQI questionnaire may not be established as an appropriate QOL instrument for TSC-related facial angiofibroma, was not sensitive enough to capture the effect of the disease at baseline, and therefore was not an appropriate tool for the assessment of any change from baseline in QOL. Also, the cognitive impairment that is inherent in many patients with TSC presents a challenge with any self-assessment questionnaire that might be used in this patient population. This study was not designed to assess whether the application of topical rapamycin to newly forming angiofibromas may be more effective than for well-established lesions. The study prohibited the concomitant use of topical and oral administration of rapamycin, and therefore did not evaluate the safety or efficacy of such coadministration, although it is conceivable that topical rapamycin might produce additive improvement of facial angiofibromas if used adjunctly in patients receiving an oral mTOR inhibitor. This study did not continue to monitor patients after discontinuation of study treatment, and therefore provides no information on the durability of benefit after treatment is stopped.
Conclusions
Topical rapamycin therapy appears effective and safe for treatment of TSC-related facial angiofibromas. In this trial, the integrated efficacy and safety findings indicate that 1% rapamycin is preferrable to 0.1% rapamycin. Future studies are needed to explore long-term efficacy and safety of topical rapamycin therapy, the durability of response, whether prophylactic or very early use of topical rapamycin in patients with TSC alters the natural history of facial angiofibromas and other skin lesions, and the efficacy and safety of combining oral and topical mTOR inhibitors for treatment of TSC-related facial angiofibromas.
Trial Protocol
eMethods. Institutional Review Board Approval, Photography Protocol, and Data and Safety Monitoring
eTable 1. The Angiofibroma Grading Scale (AGS)
eTable 2. Baseline Characteristics of Patients
eFigure 1. Photographs of Patient in Figure 4, Parts A and D
eFigure 2. Photographs of Patient in Figure 4, Parts B and E
eFigure 3. Photographs of Patient in Figure 4, Parts C and F
References
- 1.European Chromosome 16 Tuberous Sclerosis Consortium Identification and characterization of the tuberous sclerosis gene on chromosome 16. Cell. 1993;75(7):1305-1315. [DOI] [PubMed] [Google Scholar]
- 2.van Slegtenhorst M, de Hoogt R, Hermans C, et al. Identification of the tuberous sclerosis gene TSC1 on chromosome 9q34. Science. 1997;277(5327):805-808. [DOI] [PubMed] [Google Scholar]
- 3.Narayanan V. Tuberous sclerosis complex: genetics to pathogenesis. Pediatr Neurol. 2003;29(5):404-409. [DOI] [PubMed] [Google Scholar]
- 4.Krueger DA, Northrup H; International Tuberous Sclerosis Complex Consensus Group . Tuberous sclerosis complex surveillance and management: recommendations of the 2012 International Tuberous Sclerosis Complex Consensus Conference. Pediatr Neurol. 2013;49(4):255-265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Northrup H, Krueger DA; International Tuberous Sclerosis Complex Consensus Group . Tuberous sclerosis complex diagnostic criteria update: recommendations of the 2012 International Tuberous Sclerosis Complex Consensus Conference. Pediatr Neurol. 2013;49(4):243-254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tuberous Sclerosis Alliance Tuberous sclerosis complex 2014 constituent survey results. 2015. http://fliphtml5.com/tosk/qyby. Accessed February 14, 2017.
- 7.Northrup H, Koenig MK, Pearson DA, Au KS Tuberous sclerosis complex. GeneReviews(R), 1999. Jul 13 [Updated 2015 Sep 3]. https://www.ncbi.nlm.nih.gov/books/NBK1220/. Accessed February 13, 2017.
- 8.Paghdal KV, Schwartz RA. Sirolimus (rapamycin): from the soil of Easter Island to a bright future. J Am Acad Dermatol. 2007;57(6):1046-1050. [DOI] [PubMed] [Google Scholar]
- 9.Bissler JJ, McCormack FX, Young LR, et al. Sirolimus for angiomyolipoma in tuberous sclerosis complex or lymphangioleiomyomatosis. N Engl J Med. 2008;358(2):140-151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Franz DN, Belousova E, Sparagana S, et al. Long-term use of everolimus in patients with tuberous sclerosis complex: final results from the EXIST-1 study. PLoS One. 2016;11(6):e0158476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Koenig MK, Butler IJ, Northrup H. Regression of subependymal giant cell astrocytoma with rapamycin in tuberous sclerosis complex. J Child Neurol. 2008;23(10):1238-1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Govindarajan B, Willoughby L, Band H, et al. Cooperative benefit for the combination of rapamycin and imatinib in tuberous sclerosis complex neoplasia. Vasc Cell. 2012;4(1):11-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bittencourt RC, Huilgol SC, Seed PT, Calonje E, Markey AC, Barlow RJ. Treatment of angiofibromas with a scanning carbon dioxide laser: a clinicopathologic study with long-term follow-up. J Am Acad Dermatol. 2001;45(5):731-735. [DOI] [PubMed] [Google Scholar]
- 14.El-Musa KA, Shehadi RS, Shehadi S. Extensive facial adenoma sebaceum: successful treatment with mechanical dermabrasion: case report. Br J Plast Surg. 2005;58(8):1143-1147. [DOI] [PubMed] [Google Scholar]
- 15.Papadavid E, Markey A, Bellaney G, Walker NP. Carbon dioxide and pulsed dye laser treatment of angiofibromas in 29 patients with tuberous sclerosis. Br J Dermatol. 2002;147(2):337-342. [DOI] [PubMed] [Google Scholar]
- 16.Balestri R, Neri I, Patrizi A, Angileri L, Ricci L, Magnano M. Analysis of current data on the use of topical rapamycin in the treatment of facial angiofibromas in tuberous sclerosis complex. J Eur Acad Dermatol Venereol. 2015;29(1):14-20. [DOI] [PubMed] [Google Scholar]
- 17.DeKlotz CM, Ogram AE, Singh S, Dronavalli S, MacGregor JL. Dramatic improvement of facial angiofibromas in tuberous sclerosis with topical rapamycin: optimizing a treatment protocol. Arch Dermatol. 2011;147(9):1116-1117. [DOI] [PubMed] [Google Scholar]
- 18.Foster RS, Bint LJ, Halbert AR. Topical 0.1% rapamycin for angiofibromas in paediatric patients with tuberous sclerosis: a pilot study of four patients. Australas J Dermatol. 2012;53(1):52-56. [DOI] [PubMed] [Google Scholar]
- 19.Haemel AK, O’Brian AL, Teng JM. Topical rapamycin: a novel approach to facial angiofibromas in tuberous sclerosis. Arch Dermatol. 2010;146(7):715-718. [DOI] [PubMed] [Google Scholar]
- 20.Jóźwiak S, Sadowski K, Kotulska K, Schwartz RA. Topical use of mammalian target of rapamycin (mTOR) inhibitors in tuberous sclerosis complex-a comprehensive review of the literature. Pediatr Neurol. 2016;61:21-27. [DOI] [PubMed] [Google Scholar]
- 21.Koenig MK, Hebert AA, Roberson J, et al. Topical rapamycin therapy to alleviate the cutaneous manifestations of tuberous sclerosis complex: a double-blind, randomized, controlled trial to evaluate the safety and efficacy of topically applied rapamycin. Drugs R D. 2012;12(3):121-126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mutizwa MM, Berk DR, Anadkat MJ. Treatment of facial angiofibromas with topical application of oral rapamycin solution (1mgmL(-1) ) in two patients with tuberous sclerosis. Br J Dermatol. 2011;165(4):922-923. [DOI] [PubMed] [Google Scholar]
- 23.Pynn EV, Collins J, Hunasehally PR, Hughes J. Successful topical rapamycin treatment for facial angiofibromata in two children. Pediatr Dermatol. 2015;32(3):e120-e123. [DOI] [PubMed] [Google Scholar]
- 24.Salido R, Garnacho-Saucedo G, Cuevas-Asencio I, et al. Sustained clinical effectiveness and favorable safety profile of topical sirolimus for tuberous sclerosis - associated facial angiofibroma. J Eur Acad Dermatol Venereol. 2012;26(10):1315-1318. [DOI] [PubMed] [Google Scholar]
- 25.Tanaka M, Wataya-Kaneda M, Nakamura A, Matsumoto S, Katayama I. First left-right comparative study of topical rapamycin vs. vehicle for facial angiofibromas in patients with tuberous sclerosis complex. Br J Dermatol. 2013;169(6):1314-1318. [DOI] [PubMed] [Google Scholar]
- 26.Tu J, Foster RS, Bint LJ, Halbert AR. Topical rapamycin for angiofibromas in paediatric patients with tuberous sclerosis: follow up of a pilot study and promising future directions. Australas J Dermatol. 2014;55(1):63-69. [DOI] [PubMed] [Google Scholar]
- 27.Wataya-Kaneda M, Nakamura A, Tanaka M, et al. Efficacy and safety of topical sirolimus therapy for facial angiofibromas in the tuberous sclerosis complex: a randomized clinical trial. JAMA Dermatol. 2017;153(1):39-48. [DOI] [PubMed] [Google Scholar]
- 28.Wheless JW, Almoazen H. A novel topical rapamycin cream for the treatment of facial angiofibromas in tuberous sclerosis complex. J Child Neurol. 2013;28(7):933-936. [DOI] [PubMed] [Google Scholar]
- 29.Basra MK, Sue-Ho R, Finlay AY. The Family Dermatology Life Quality Index: measuring the secondary impact of skin disease. Br J Dermatol. 2007;156(3):528-538. Erratum in Br J Dermatol. 2007;156(4):791. [DOI] [PubMed] [Google Scholar]
- 30.Finlay AY, Khan GK. Dermatology Life Quality Index (DLQI)--a simple practical measure for routine clinical use. Clin Exp Dermatol. 1994;19(3):210-216. [DOI] [PubMed] [Google Scholar]
- 31.Lewis-Jones MS, Finlay AY. The Children’s Dermatology Life Quality Index (CDLQI): initial validation and practical use. Br J Dermatol. 1995;132(6):942-949. [DOI] [PubMed] [Google Scholar]
- 32.Knöpfel N, Martín-Santiago A, Bauza A, Hervás JA. Topical 0.2% rapamycin to treat facial angiofibromas and hypomelanotic macules in tuberous sclerosis. Actas Dermosifiliogr. 2014;105(8):802-803. [DOI] [PubMed] [Google Scholar]
- 33.Malissen N, Vergely L, Simon M, Roubertie A, Malinge MC, Bessis D. Long-term treatment of cutaneous manifestations of tuberous sclerosis complex with topical 1% sirolimus cream: A prospective study of 25 patients. [published online May 10, 2017]. J Am Acad Dermatol. 2017;77(3):464-472.e3. doi: 10.1016/j.jaad.2017.04.005 [DOI] [PubMed] [Google Scholar]
- 34.Wataya-Kaneda M, Tanaka M, Nakamura A, Matsumoto S, Katayama I. A novel application of topical rapamycin formulation, an inhibitor of mTOR, for patients with hypomelanotic macules in tuberous sclerosis complex. Arch Dermatol. 2012;148(1):138-139. [DOI] [PubMed] [Google Scholar]
- 35.Wataya-Kaneda M, Tanaka M, Yang L, et al. Clinical and histologic analysis of the efficacy of topical rapamycin therapy against hypomelanotic macules in tuberous sclerosis complex. JAMA Dermatol. 2015;151(7):722-730. [DOI] [PubMed] [Google Scholar]
- 36.Drugs@FDA: FDA Approved Drug Products. Prescribing information for Rapamune. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/021083s059,021110s076lbl.pdf. Accessed July 9, 2017.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Trial Protocol
eMethods. Institutional Review Board Approval, Photography Protocol, and Data and Safety Monitoring
eTable 1. The Angiofibroma Grading Scale (AGS)
eTable 2. Baseline Characteristics of Patients
eFigure 1. Photographs of Patient in Figure 4, Parts A and D
eFigure 2. Photographs of Patient in Figure 4, Parts B and E
eFigure 3. Photographs of Patient in Figure 4, Parts C and F