Abstract
The management of meniscal injuries remains difficult and challenging. Although several clinical options exist for the treatment of such injuries, complete regeneration of the damaged meniscus has proved difficult due to the limited healing capacity of the tissue. With the advancements in tissue engineering and cell-based technologies, new therapeutic options for patients with currently incurable meniscal lesions now potentially exist. This review will discuss basic anatomy, current repair techniques and treatment options for loss of meniscal integrity. Specifically, we focus on the possibility and feasibility of the latest tissue engineering approaches, including 3D printing technologies. Therefore, this discussion will facilitate a better understanding of the latest trends in meniscal repair and regeneration, and contribute to the future application of such clinical therapies for patients with meniscal injuries.
1. Introduction
The meniscus is a crescent-shaped fibrocartilaginous tissue, comprised of both a medial and a lateral component positioned between the corresponding femoral condyle and tibial plateau, and plays important roles in the knee joint, including force transmission, shock absorption, joint lubrication, and the provision of joint stability.1, 2, 3 For many in the young and active population who injure their knee, this commonly involves injury to the menisci.4,5 The previously reported mean annual incidence of meniscal lesions per 10,000 populations was 9.0 for males and 4.2 for females.6 However, it has been widely accepted that a meniscus tear does not heal spontaneously, owing to its hypovascularity and hypocellularity.1,3,7 In the absence of effective long-term repair of these meniscal injuries, damage to the knee may compromise athletic careers and lead to development of osteoarthritis at an early age.8,9 Therefore, the development of novel therapeutic methods for meniscal repair is both timely and necessary.
Historically, the menisci were considered to be a functionless vestigial structure, one that should be entirely removed once damaged.10 Since King reported “the function of the semilunar cartilages” in 1936 11, the importance of meniscal functions in the knee joint has been gradually recognized. In 1948, Fairbank reported joint degenerative changes were observed after meniscectomy, suggesting such changes were due to loss of the weight-bearing function of the meniscus.12 In the 1980s, several biomechanical studies addressed the importance of the meniscus as a joint stabilizer and shock absorber in the knee joint,13,14 and meniscal preservation has been recognized to be essential to retention of normal knee biomechanics. However, meniscectomy to remove the damaged, unstable portion of the meniscus has still been the gold standard of surgical treatment for meniscal tears, since there have not been effective therapeutic methods developed for such tears.
In this review, we focus on the current strategies and therapeutic methods for meniscal repair and regeneration following such previously incurable meniscal tears, and highlight recent advances in meniscal tissue engineering approaches. This will facilitate an understanding of the latest trends for meniscal repair and regeneration, and contribute to the future application of such clinical therapies in patients with meniscal injuries that previously were progressive and led to an increased risk for development of osteoarthritis.
2. Anatomy, biochemistry and biomechanics
The meniscus is a crescent-shaped highly complex structure with low cellularity and dense extracellular matrix (ECM), which is stabilized by the medial collateral ligament, the transverse ligament, the meniscofemoral ligaments, and attachments at the anterior and posterior horns.1,15 Vascularization in the adult meniscus exists only in the peripheral 10–25% of the tissue, and the extent of the vascular zone has implications for the healing potential of meniscal tears.7 Biochemically, normal menisci compose of 72% water, 22% collagen, 0.8% glycosaminoglycans and 0.12% DNA.16 Collagen is the main ECM component of the meniscus, and different collagen types exist in each region of the tissue. In the red-red zone (vascular zone), collagen type I is predominant at approximately 80% composition by dry weight. In the white-white zone (avascular zone), 40% of the tissue by dry weight is collagen type I and 60% is collagen type II.17 Regarding meniscal cells, cells in the red-red zone are more fibroblast-like in appearance and connected via cellular networks, while cells in the white-red zone and white-white zone are more chondrocyte-like and exist as single cells.1,18 Cells of the menisci require loading to maintain the integrity of the menisci, as loss of loading can lead to derepression of catabolic genes and potential induction of tissue atrophy.19 Loss of appropriate loading after an injury could thus contribute to the progressive deterioration of the injured tissue. Collagen fiber arrangement is highly specialized, with the majority being circumferentially aligned.20,21 This circumferential orientation creates biomechanically optimal resistance to hoop stresses, resulting from displacement of the meniscus from the tibial plateau during normal weight-bearing.3 The organization of the collagen fibers, as well as the proteoglycan in the meniscus is quite complex, with different organization near the surface, the periphery, and in the central, more cartilaginous zones.22,23
3. Current surgical repair techniques
Based on meniscal anatomy and vascularity, the meniscus has limited healing capacities especially in central two-thirds avascular zone, while meniscal tears in peripheral vascular zone should be reparable.24 To preserve important meniscal functions, surgeons should always consider repairing the meniscal injury as extensively as possible. As many studies have addressed, meniscal tears in the vascular zone of peripheral area, such as vertical longitudinal tears, have good indications for effective meniscal repair.25,26 As horizontal tears were previously considered to be rarely healed due to the involvement of inner avascular zone, either non-operative treatment or meniscectomy were chosen.27 Recently, meniscal preservation techniques suturing the tear have been attempted to preserve meniscal function, and successful outcomes can be obtained especially in younger patients without degenerative meniscal changes.28,29 On the other hand, meniscal tears that include lesions of the avascular zone, such as radial tears, are not expected to spontaneously heal, and thus such tears have been mostly treated by partial meniscectomy.30 To overcome this problem following injuries to the avascular zone tissue, several suture techniques have been recently developed to enhance healing in this zone, and some case series reported the improvement of clinical outcomes in short-term results.31, 32, 33, 34, 35 Regarding other types of meniscal tears, meniscal root tears36,37 and discoid lateral meniscal tears38, 39, 40 have been paid attention to as recent topics, and several suture and/or preservation techniques have been developed and tested in clinical practice. To further enhance the repair potential, some biologic augmentation have been attempted to promote the healing of torn meniscal sites in clinical practice, and these include mechanical stimulation of the adjacent synovium or the meniscus by rasping,41 augmentation with fibrin clots42 or platelet-rich plasma,43 as well as introduction of stem cells from the bone marrow by a marrow venting technique,44 which has been proven to improve the meniscal healing rate. In experimental studies using large preclinical porcine models, mesenchymal stem cell (MSC)-based therapies were demonstrated to be feasible for meniscal repair.45,46 Also, biomaterial augmentation via wrapping the meniscal tear (e.g. collagen membrane or nanofibrous scaffold) has been investigated to enhance the healing of the torn meniscal sites in either experimental47 or clinical studies,48 and such a technique will be expected as a potential therapeutic method.
4. Current therapeutic options for meniscal deficiency
Once the injured part of the meniscus is excised, the natural healing response will not occur at the site of the injury due to its limited healing capacity, and thus the meniscal deficiency will remain. Several therapeutic options have been proposed to reconstruct the resected meniscus and/or substitute an autologous tissue, allograft or meniscal substitute in the case where the original meniscus has been removed.49
4.1. Autologous tissues
Autologous tissues such as fat pad,50 tendon,51, 52, 53 periosteum,54 synovial flap55 and perichondrium56 have been used as an autograft in preclinical animal or clinical studies. However, satisfactory results were rarely obtained owing to compromised mechanical properties, inferior vascularization, and differences in the shape and internal structure of the repair tissue.49 Therefore, it can be concluded that these tissues are not a good option for effective replacement of the meniscus.
4.2. Allografts
Meniscal allograft transplantations have been widely performed for meniscal deficiency after total or nearly total meniscectomy. The meniscal transplantation emerges as a good indication for patients with a stable joint, appropriate alignment, and with early osteoarthritis of the knee, while these procedures are contraindicated for patients with severe osteoarthritis.57 A recent systematic review concluded that meniscal allograft transplantation appears to provide good clinical results over short-term and midterm follow-up, with improvement in knee function.58 In a long-term follow-up study (mean follow-up time of 152 months), meniscal transplantation resulted in significant improvements in pain and functional outcomes over the study period, despite an increase in joint space narrowing.59 Also, this study reported 34.7% of the patients underwent some type of revision surgery including total knee arthroplasty at the final follow-up. On the other hand, some drawbacks of allografts were reported to include immunological reaction to the implanted tissue, potential disease transmission, and limited donor availability.58,60 Also, graft size matching, appropriate preservation techniques, and surgical transplantation technique are important issues for the success of such transplantation procedures. Future studies will be necessary to optimize these parameters to improve surgical outcomes.49
4.3. Meniscal substitutes
Several materials have been developed and assessed for efficacy in addressing meniscal deficiency, either in vitro or in vivo. There have been two implants that have been made available for clinical practice, one is a collagen meniscus implant (CMI®) and the other, a polyurethane polymeric implant (Actifit®).1,61 These two implants can be offered to patients with an intact peripheral meniscal rim and limited damaged cartilage after meniscectomy. Recent long-term follow-up studies have reported that CMI® provided significant pain relief and functional improvement with safety and a low rate of implant failure.62,63 Additionally, some studies reported that CMI® showed better clinical outcomes than did partial meniscectomy, but with limited evidence provided,64,65 and thus further studies are necessary to allow drawing stronger conclusions regarding this approach. Similarly, Actifit® showed safety and effectiveness with improved clinical outcomes with short- and middle-term follow-up.66, 67, 68, 69 On the other hand, several studies reported negative outcomes based on MRI results with these two implants, despite the general observation of improved clinical scores.66,67,70 These implants were partially or total resorbed during follow-up. Also, the implants mostly showed hyperintensity and/or extrusion, accompanied by subchondral bone edema. Thus, there is likely still room for improvement, regarding the development of such implants serving as meniscal substitutes.
Surgeons agree that meniscal allograft transplantation is currently the best treatment for symptomatic meniscectomied patients, but a number of issue still remain such as graft availability, size matching, high costs, possible disease transmission, and limited widespread practice of this procedure, as mentioned above.61 As an alternative to this procedure, total meniscus replacement using a synthetic meniscus has been assessed for such patients, although the shape of implant is anatomically different. Recently, a synthetic polyethylene reinforced polycarbonate urethane (PCU) meniscus implant (NUsurface®) has been developed to the stage of clinical trials.61,71 Some preliminary data based on MRI results showed restoration of the joint space and maintenance of the cartilage signal intensity at 12 months post-surgery.61
5. New trends for meniscal regeneration – possibilities and feasibilities
Although several clinical options exist for addressing an incurable meniscal deficiency as discussed above, effective long-term repair methods are not available for these injuries. Therefore, the development of novel therapeutic methods for meniscus repair is both timely and necessary. Recently, tissue engineering approaches that involve the use of cells and biomaterial scaffolds have gained increasing attention as potential regenerative therapies in the field of musculoskeletal medicine.1,72,73 These approaches are still primarily in the preclinical phases of development, but likely will progress to the clinical application stage in near future. Thus, the likelihood and feasibility of tissue engineering approaches to become effective interventions will be discussed in the following section.
5.1. Tissue engineering approaches
Tissue engineering is defined as the application of the principles of biology and engineering to the development of functional substitutes for damaged tissue, and usually utilize a combination of cells, scaffolds, and growth factors.74 Regarding the selection of cell sources, meniscal cells,75 chondrocytes,76 and mesenchymal stem cells (MSCs) derived from bone marrow,77 fat tissue78 or synovium45 have been used. However, there is still no consensus regarding the best cell resource for meniscal regeneration due to the lack of comparative studies being performed. Scaffolds for tissue engineering the meniscus may be categorized into four broad classes: synthetic polymers (e.g. polyurethane (PU), polycaprolactone (PCL), polylactic acid (PLA), polyglycolic acid (PGA), polylactic co-glycolic acid (PLGA)), hydrogels, natural matrix (e.g. collagen, hyaluronan), or tissue-derived materials (e.g. decellularized ECM).1 For the ideal selection of a scaffold for meniscal regeneration, it is important to know the material properties of each of the potential scaffolds in order to pick the most appropriate for the environment for which they will be used. In general, synthetic polymers are easy to handle and have high mechanical properties, while natural materials retain higher bioactivity, which might be advantageous for tissue healing and remodeling.1 Similar to cell sources, there is still no consensus regarding the ideal materials for a scaffold to be used for meniscal regeneration.
As listed in Table 1, we have outlined the recent pre-clinical animal studies of cell-based meniscal tissue engineering. There have been some promising studies reported, and thus, some of these approaches may be expected to lead to the initiation of clinical trials in the near future.
Table 1.
Summary of cell-based meniscal tissue engineering in preclinical large animal study.
| Authors | Reference | Animal | Experimental model | Cell source | Scaffold | Follow-up period | Outcome measure | Outcomes |
|---|---|---|---|---|---|---|---|---|
| Weinand C et al. | Am J Sports Med 2006 | Pig | 1 cm buckethandle lesion | Chondrocyte | Woven Vicryl mesh | 12 weeks | Gross, histology | Macroscopic & histological healing |
| Weinand C et al. | Arch Orthop Trauma Surg 2006 | Pig | 1 cm buckethandle lesion in avascular zone | Chondrocyte | Woven Vicryl mesh PLGA | 12 weeks | Gross, histology, gross mechanical test | Bonding lesion, healing by new tissue |
| Martinek V et al. | Arch Orthop Trauma Surg 2006 | Sheep | Subtotal meniscectomy | Meniscal fibrochondrocyte | CMI | 3 months | Gross, histology | Enhanced vascularization & re-modelling, higher content of ECM |
| Kon E et al. | Tissue Eng Part A 2008 | Sheep | Total meniscectomy | Chondrocyte | Hyaluronic acid/polycaprolactone | 4 months | Gross, histology | Better implant appearance & integrity, lower joint degeneration, cellular infiltration & vascularization |
| Zhang H et al. | Clin Orthop Relat Res 2009 | Goat | φ3 mm defect | Bone marrow MSC transfected w/hIGF-1 | Calcium alginate gel | 16 weeks | Histology, electron microscopy, proteoglycan determination, MRI | Fibrocartilaginous repair, higher proteoglycan content |
| Kon E et al. | Tissue Eng Part A 2012 | Sheep | Total meniscectomy | Chondrocyte | Hyaluronic acid/polycaprolactone | 12 months | Gross, histology | No extrusion, fibrocartilaginous repair |
| Gu Y et al. | Exp Ther Med 2012 | Dog | 2.5 mm width defect | Myoblast | PLGA | 12 weeks | Gross, histology, immunohistochemistry | Good defect filling & integration, fibrocartilaginous repair |
| Moriguchi Y et al. | Biomaterials 2013 | Pig | φ4 mm defect | Synovial MSC | Scaffold-free | 6 months | Gross, histology | Fibrocartilaginous repair, chondroprotective effect |
| Zhu WH et al. | Mol Med Rep 2014 | Dog | 2 mm width defect | Myoblast transfected w/hCDMP-2 | PLA/PGA | 12 weeks | Gross, histology, immunohistochemistry, collagen I & II, GAG quantification | Fibrocartilage‑like tissue regeneration |
CMI: collagen meniscal implant, ECM: extracellular matrix, hIGF-1: human insulin-like growth factor-1, PLGA: poly(lactic-co-glycolide acid), MSC: mesenchymal stem cell, hCDMP-2: human cartilage-derived morphogenetic protein-2, PLA: polylactic acid, PGA: polyglycolic acid, GAG: glycosaminoglycan.
5.2. Anatomy-based meniscal regeneration
While a number of meniscal biomaterial scaffolds and tissue engineering approaches have been developed and show promise, complete meniscus regeneration remains challenging because of the difficulty in reproducing the complex meniscal collagen fiber arrangements and the anatomically complex meniscus structure composed of a region-specific matrix organization and biochemical composition, as potential limitations of previous studies and currently available treatments.22,23 Specifically, many biomaterials resemble the shape of native meniscus, but with a porous structure inside,1,61 although the meniscus has unique collagen fiber arrangements.20, 21, 22, 23, Recently, a fiber-reinforced scaffold, composed of a type I collagen and hyaluronic acid sponge reinforced with a unique pattern of continuous tyrosine-derived biodegradable polymer fiber, has been developed and tested in large animal studies.79,80 The fiber-reinforced scaffolds were implanted into sheep knee joints after a total meniscectomy, and it exhibited formation of a functional neo-meniscus tissue, with the potential to prevent joint degeneration at one year postoperatively. Thus, such fiber-reinforced scaffolds might provide the appropriate structural properties to take over the load-bearing role that is required of the meniscus, and provide long-term chondro-protective effects. As another anatomy-based meniscal tissue engineering approach, the applicability of decellularized meniscal ECM (dmECM) has been investigated by specifically comparing region-dependent effects of the dmECMs on 3-dimensional constructs seeded with human bone marrow MSCs in an experimental study.81,82 Such studies have shown that the inner dmECM (avascular zone) enhances the fibrocartilaginous differentiation of MSCs, while the outer dmECM (vascular zone) promotes a more fibroblastic phenotype, supporting the feasibility to engineer a meniscus-like tissue that mimics the anatomy and biochemistry of the native meniscus by using region-specific dmECM preparations.
5.3. 3D bioprinting meniscal regeneration
Since 3D printing was first conceptualized and invented by Charles Hull in the early 1980s, these technologies have garnered attention in the field of regenerative medicine, especially in the last decade.83,84 The recent advancements in medical imaging such as CT and MRI can provide the exact 3D reconstructed images for creating 3D objects via printing. Going forward, 3D printed objects will contribute significantly to the visualization of complex pathologies, surgical planning, manufacture of patient-specific instruments and implants, as well as making patient-specific scaffolds for tissue engineering approaches for meniscal repair.85
As the meniscus has highly complex shape and structure, the application of 3D printing technology will be very appropriate for meniscal tissue engineering. As listed in Table 2, there have been several studies recently reported regarding 3D printed menisci, but the long-term evidence for their effectiveness in meniscal repair is still limited. Mostly, these studies trend to reproduce the meniscal fiber arrangement using a 3D printer,86, 87, 88 and such design of the scaffold should potentially improve the clinical outcomes. For that approach to be accepted, further comparative studies with the more conventional methods discussed earlier will be necessary.
Table 2.
Summary of 3D printed meniscus.
| Authors | Reference | Experimental model | Cell source | Scaffold | Growth factor | Shape | internal structure | Outcome measure |
|---|---|---|---|---|---|---|---|---|
| Ballyns JJ et al. | Tissue Eng Part C Methods 2010 | in vitro | – | Alginate hydrogel | – | Meniscus | unknown | Geometric analysis |
| Grogan SP et al. | Acta Biomater 2013 | in vitro | Meniscal cell | Methacrylated gelatin | – | Parallelepiped | Fiber | Cell viablity, histology, immunohistochemistry, PCR, SEM, mechanical testing |
| Lee CH et al. | Sci Transl Med. 2014 | Sheep | – | PCL | CTGF & TGFβ3 | Meniscus | Fiber | Gross, histology, mechanical testing |
| van Bochove B et al. | Macromol Biosci 2016 | in vitro | – | PTMC | – | Meniscus | pore | micro CT, mechanical testing |
| Warren PB et al. | Connect Tissue Res 2017 | Rat | – | PCL | – | Cylinder | Strand-pore | Histology, immunohistochemistry |
| Zhang ZZ et al. | Am J Sports Med 2017 | Rabbit | Bone marrow MSC | PCL | – | Meniscus | Fiber | Gross, histology, SEM, mechanical testing |
PCR: polymerase chain reaction, SEM: Scanning electron microscopy, PCL: poly-ε-caprolactone, CTGF: connective tissue growth factor, TGFβ3: transforming growth factor–β3, PTMC: poly(trimethylene carbonate), MSC: mesenchymal stem cell.
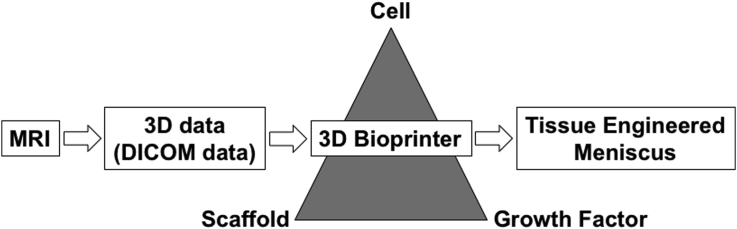
3D printing is an innovative and promising technique for regenerative medicine, and it will enable provision of “made-to-order” medicine according to the needs of the individual condition by the fabrication of size-matching implant (Fig. 1). However, the approach currently still has some limitations. First, 3D printing costs are currently high because such technologies require hardware, software, manpower for maintenance and the cost of printing materials.85 Secondly, the safety of 3D printing technology is another concern, and potential risks have not been fully elucidated as this technology continues to integrate and gain popularity into medical practice.85 Thirdly, the accuracy of 3D printer may be considered as another limitation. The higher resolution of initial 3D imaging and more accurate printing techniques will be needed to make for more reliable procedures.83 However, in the future when many of the current obstacles are overcome, 3D printing of stem cell-biological ECM composites may lead to engineered meniscal constructs that can then be exposed to biomechanical loading during development to make them more functional prior to implantation,89 and enhance the chances for success after implantation to repair meniscal defects.
Fig. 1.
Schematic representation of the fabrication of an order-made bioactive meniscal implant in 3D bioprinter.
6. Conclusions and future perspectives
With recent advancements in surgical techniques, biomaterials and cell-based technologies in tissue engineering, we may have new therapeutic options on the horizon for addressing meniscal injuries in clinical practice. On the other hand, there are still several potential problems to be solved, considering complete meniscus regeneration remains challenging for managing meniscal deficiencies with currently available techniques. First, the selection and design of biomaterials with sufficient mechanical strength and long-term durability for the optimal repair and remodeling of menisci have not been fully elucidated. Additionally, such biomaterials are essential to also prevent the progression of cartilage degeneration in a long-term. Due to the lack of evidences regarding these issues, further studies (e.g. high quality comparative studies) will be needed and should be conducted in a methodologically rigorous fashion. Secondly, current approaches indicate that meniscal function could not be fully restored effectively, based on observing meniscal extrusion and the progression of joint space narrowing after biomaterial implantation in many cases.66,67,70 It is likely that newly developed technologies such as 3D printing may be a key technology to assist in solving several of these problems, getting closer to the native meniscus regarding anatomical and biomechanical aspects. Also, surgical techniques will need to be refined, especially in the prevention of meniscal extrusion and the restoration of meniscal functions. Furthermore, surgeons should consider the cost-effectiveness to apply these new techniques into clinical practice. A last consideration is that an ideal cell-seeded, size-matching meniscal implant mimicking the native meniscus may not be absolutely needed to satisfy the needs of all patients (i.e. precision medicine). Therefore, further studies (e.g. cellular versus acellular scaffolds) are needed to determine whether the increased intervention costs can be balanced with the observable advantages of these new technologies (cost/benefit analysis). However, as the technologies become perfected and implemented, the costs will likely come down, making the optimal choices for individual patients more feasible.
Disclosure statement
No competing financial interests exist.
Acknowledgement
This study was supported by a Grant-in-Aid for Young Scientists (A), the Japan Society for the Promotion of Science (grant number JP16H06264) (to K.S.), Alcare award of Japan Orthopaedics and Traumatology Foundation 2016 (grant number 343) (to K. S.), and a grant from the Japan Agency for Medical Research and Development (grant number JP18he0702224) (to N.N.).
Contributor Information
Kazunori Shimomura, Email: kazunori-shimomura@umin.net.
Shuichi Hamamoto, Email: tcjck805@yahoo.co.jp.
David A. Hart, Email: hartd@ucalgary.ca.
Hideki Yoshikawa, Email: yhideki@ort.med.osaka-u.ac.jp.
Norimasa Nakamura, Email: norimasa.nakamura@ohsu.ac.jp.
References
- 1.Makris E.A., Hadidi P., Athanasiou K.A. The knee meniscus: structure-function, pathophysiology, current repair techniques, and prospects for regeneration. Biomaterials. 2011;32(30):7411–7431. doi: 10.1016/j.biomaterials.2011.06.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.McDermott I.D., Amis A.A. The consequences of meniscectomy. J Bone Joint Surg Br. 2006;88(12):1549–1556. doi: 10.1302/0301-620X.88B12.18140. [DOI] [PubMed] [Google Scholar]
- 3.Fox A.J., Bedi A., Rodeo S.A. The basic science of human knee menisci: structure, composition, and function. Sports health. 2012;4(4):340–351. doi: 10.1177/1941738111429419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mitchell J., Graham W., Best T.M. Epidemiology of meniscal injuries in US high school athletes between 2007 and 2013. Knee Surg Sports Traumatol Arthrosc. 2016;24(3):715–722. doi: 10.1007/s00167-015-3814-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Takeda H., Nakagawa T., Nakamura K., Engebretsen L. Prevention and management of knee osteoarthritis and knee cartilage injury in sports. Br J Sports Med. 2011;45(4):304–309. doi: 10.1136/bjsm.2010.082321. [DOI] [PubMed] [Google Scholar]
- 6.Hede A., Jensen D.B., Blyme P., Sonne-Holm S. Epidemiology of meniscal lesions in the knee. 1,215 open operations in Copenhagen 1982-84. Acta Orthop Scand. 1990;61(5):435–437. doi: 10.3109/17453679008993557. [DOI] [PubMed] [Google Scholar]
- 7.Arnoczky S.P., Warren R.F. Microvasculature of the human meniscus. Am J Sports Med. 1982;10(2):90–95. doi: 10.1177/036354658201000205. [DOI] [PubMed] [Google Scholar]
- 8.Nawabi D.H., Cro S., Hamid I.P., Williams A. Return to play after lateral meniscectomy compared with medial meniscectomy in elite professional soccer players. Am J Sports Med. 2014;42(9):2193–2198. doi: 10.1177/0363546514540271. [DOI] [PubMed] [Google Scholar]
- 9.Stein T., Mehling A.P., Welsch F., von Eisenhart-Rothe R., Jager A. Long-term outcome after arthroscopic meniscal repair versus arthroscopic partial meniscectomy for traumatic meniscal tears. Am J Sports Med. 2010;38(8):1542–1548. doi: 10.1177/0363546510364052. [DOI] [PubMed] [Google Scholar]
- 10.Sutton J.B. MK Lewis; London: 1897. Ligaments: Their Nature and Morphology. [Google Scholar]
- 11.King D. The function of the semilunar cartilages. J Bone Joint Surg. 1936;18-B:1069–1076. [Google Scholar]
- 12.Fairbank T.J. Knee joint changes after meniscectomy. J Bone Joint Surg Br. 1948;30B(4):664–670. [PubMed] [Google Scholar]
- 13.Baratz M.E., Fu F.H., Mengato R. Meniscal tears: the effect of meniscectomy and of repair on intraarticular contact areas and stress in the human knee. A preliminary report. Am J Sports Med. 1986;14(4):270–275. doi: 10.1177/036354658601400405. [DOI] [PubMed] [Google Scholar]
- 14.Jaspers P., de Lange A., Huiskes R., van Rens T.J. The mechanical function of the meniscus, experiments on cadaveric pig knee-joints. Acta Orthop Belg. 1980;46(6):663–668. [PubMed] [Google Scholar]
- 15.McDermott I.D., Masouros S.D., Bull A.M.J., Amis A.A. Anatomy. In: Beaufils P., Verdonk R., editors. The Meniscus. Springer Berlin Heidelberg; Berlin, Heidelberg: 2010. pp. 11–18. [Google Scholar]
- 16.Herwig J., Egner E., Buddecke E. Chemical changes of human knee joint menisci in various stages of degeneration. Ann Rheum Dis. 1984;43(4):635–640. doi: 10.1136/ard.43.4.635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cheung H.S. Distribution of type I, II, III and V in the pepsin solubilized collagens in bovine menisci. Connect Tissue Res. 1987;16(4):343–356. doi: 10.3109/03008208709005619. [DOI] [PubMed] [Google Scholar]
- 18.Hellio Le Graverand M.P., Ou Y., Schield-Yee T. The cells of the rabbit meniscus: their arrangement, interrelationship, morphological variations and cytoarchitecture. J Anat. 2001;198(Pt 5):525–535. doi: 10.1046/j.1469-7580.2000.19850525.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Natsu-Ume T., Majima T., Reno C., Shrive N.G., Frank C.B., Hart D.A. Menisci of the rabbit knee require mechanical loading to maintain homeostasis: cyclic hydrostatic compression in vitro prevents derepression of catabolic genes. J Orthop Sci. 2005;10(4):396–405. doi: 10.1007/s00776-005-0912-x. [DOI] [PubMed] [Google Scholar]
- 20.Bullough P.G., Munuera L., Murphy J., Weinstein A.M. The strength of the menisci of the knee as it relates to their fine structure. J Bone Joint Surg Br. 1970;52(3):564–567. [PubMed] [Google Scholar]
- 21.Petersen W., Tillmann B. Collagenous fibril texture of the human knee joint menisci. Anat Embryol. 1998;197(4):317–324. doi: 10.1007/s004290050141. [DOI] [PubMed] [Google Scholar]
- 22.Andrews S.H.J., Adesida A.B., Abusara Z., Shrive N.G. Current concepts on structure-function relationships in the menisci. Connect Tissue Res. 2017;58(3–4):271–281. doi: 10.1080/03008207.2017.1303489. [DOI] [PubMed] [Google Scholar]
- 23.Andrews S.H., Rattner J.B., Abusara Z., Adesida A., Shrive N.G., Ronsky J.L. Tie-fibre structure and organization in the knee menisci. J Anat. 2014;224(5):531–537. doi: 10.1111/joa.12170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Woodmass J.M., LaPrade R.F., Sgaglione N.A., Nakamura N., Krych A.J. Meniscal repair: reconsidering indications, techniques, and biologic augmentation. J Bone Joint Surg Am. 2017;99(14):1222–1231. doi: 10.2106/JBJS.17.00297. [DOI] [PubMed] [Google Scholar]
- 25.Grant J.A., Wilde J., Miller B.S., Bedi A. Comparison of inside-out and all-inside techniques for the repair of isolated meniscal tears: a systematic review. Am J Sports Med. 2012;40(2):459–468. doi: 10.1177/0363546511411701. [DOI] [PubMed] [Google Scholar]
- 26.Fillingham Y.A., Riboh J.C., Erickson B.J., Bach B.R., Jr., Yanke A.B. Inside-Out versus all-inside repair of isolated meniscal tears: an updated systematic review. Am J Sports Med. 2017;45(1):234–242. doi: 10.1177/0363546516632504. [DOI] [PubMed] [Google Scholar]
- 27.Yim J.H., Seon J.K., Song E.K. A comparative study of meniscectomy and nonoperative treatment for degenerative horizontal tears of the medial meniscus. Am J Sports Med. 2013;41(7):1565–1570. doi: 10.1177/0363546513488518. [DOI] [PubMed] [Google Scholar]
- 28.Kurzweil P.R., Lynch N.M., Coleman S., Kearney B. Repair of horizontal meniscus tears: a systematic review. Arthroscopy. 2014;30(11):1513–1519. doi: 10.1016/j.arthro.2014.05.038. [DOI] [PubMed] [Google Scholar]
- 29.Pujol N., Bohu Y., Boisrenoult P., Macdes A., Beaufils P. Clinical outcomes of open meniscal repair of horizontal meniscal tears in young patients. Knee Surg Sports Traumatol Arthrosc. 2013;21(7):1530–1533. doi: 10.1007/s00167-012-2099-y. [DOI] [PubMed] [Google Scholar]
- 30.Foad A. Self-limited healing of a radial tear of the lateral meniscus. Knee Surg Sports Traumatol Arthrosc. 2012;20(5):933–936. doi: 10.1007/s00167-011-1660-4. [DOI] [PubMed] [Google Scholar]
- 31.Bhatia S., Civitarese D.M., Turnbull T.L. A novel repair method for radial tears of the medial meniscus: biomechanical comparison of transtibial 2-tunnel and double horizontal mattress suture techniques under cyclic loading. Am J Sports Med. 2016;44(3):639–645. doi: 10.1177/0363546515615565. [DOI] [PubMed] [Google Scholar]
- 32.Matsubara H., Okazaki K., Izawa T. New suture method for radial tears of the meniscus: biomechanical analysis of cross-suture and double horizontal suture techniques using cyclic load testing. Am J Sports Med. 2012;40(2):414–418. doi: 10.1177/0363546511424395. [DOI] [PubMed] [Google Scholar]
- 33.Choi N.H., Kim T.H., Son K.M., Victoroff B.N. Meniscal repair for radial tears of the midbody of the lateral meniscus. Am J Sports Med. 2010;38(12):2472–2476. doi: 10.1177/0363546510376736. [DOI] [PubMed] [Google Scholar]
- 34.Cinque M.E., Geeslin A.G., Chahla J., Dornan G.J., LaPrade R.F. Two-tunnel transtibial repair of radial meniscus tears produces comparable results to inside-out repair of vertical meniscus tears. Am J Sports Med. 2017;45(10):2253–2259. doi: 10.1177/0363546517704425. [DOI] [PubMed] [Google Scholar]
- 35.Tsujii A., Amano H., Tanaka Y. Second look arthroscopic evaluation of repaired radial/oblique tears of the midbody of the lateral meniscus in stable knees. J Orthop Sci. 2018;23(1):122–126. doi: 10.1016/j.jos.2017.09.023. [DOI] [PubMed] [Google Scholar]
- 36.Bhatia S., LaPrade C.M., Ellman M.B., LaPrade R.F. Meniscal root tears: significance, diagnosis, and treatment. Am J Sports Med. 2014;42(12):3016–3030. doi: 10.1177/0363546514524162. [DOI] [PubMed] [Google Scholar]
- 37.LaPrade C.M., James E.W., Cram T.R., Feagin J.A., Engebretsen L., LaPrade R.F. Meniscal root tears: a classification system based on tear morphology. Am J Sports Med. 2015;43(2):363–369. doi: 10.1177/0363546514559684. [DOI] [PubMed] [Google Scholar]
- 38.Smuin D.M., Swenson R.D., Dhawan A. Saucerization versus complete resection of a symptomatic discoid lateral meniscus at short- and long-term follow-up: a systematic review. Arthroscopy. 2017;33(9):1733–1742. doi: 10.1016/j.arthro.2017.03.028. [DOI] [PubMed] [Google Scholar]
- 39.Lee Y.S., Teo S.H., Ahn J.H., Lee O.S., Lee S.H., Lee J.H. Systematic review of the long-term surgical outcomes of discoid lateral meniscus. Arthroscopy. 2017;33(10):1884–1895. doi: 10.1016/j.arthro.2017.04.006. [DOI] [PubMed] [Google Scholar]
- 40.Matsuo T., Kinugasa K., Sakata K., Ohori T., Mae T., Hamada M. Post-operative deformation and extrusion of the discoid lateral meniscus following a partial meniscectomy with repair. Knee Surg Sports Traumatol Arthrosc. 2017;25(2):390–396. doi: 10.1007/s00167-016-4393-6. [DOI] [PubMed] [Google Scholar]
- 41.Taylor S.A., Rodeo S.A. Augmentation techniques for isolated meniscal tears. Current Rev Musculoskel Med. 2013;6(2):95–101. doi: 10.1007/s12178-013-9165-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Henning C.E., Lynch M.A., Yearout K.M., Vequist S.W., Stallbaumer R.J., Decker K.A. Arthroscopic meniscal repair using an exogenous fibrin clot. Clin Orthop Relat Res. 1990;(252):64–72. [PubMed] [Google Scholar]
- 43.Griffin J.W., Hadeed M.M., Werner B.C., Diduch D.R., Carson E.W., Miller M.D. Platelet-rich plasma in meniscal repair: does augmentation improve surgical outcomes? Clin Orthop Relat Res. 2015;473(5):1665–1672. doi: 10.1007/s11999-015-4170-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dean C.S., Chahla J., Matheny L.M., Mitchell J.J., LaPrade R.F. Outcomes after biologically augmented isolated meniscal repair with marrow venting are comparable with those after meniscal repair with concomitant anterior cruciate ligament reconstruction. Am J Sports Med. 2017;45(6):1341–1348. doi: 10.1177/0363546516686968. [DOI] [PubMed] [Google Scholar]
- 45.Moriguchi Y., Tateishi K., Ando W. Repair of meniscal lesions using a scaffold-free tissue-engineered construct derived from allogenic synovial MSCs in a miniature swine model. Biomaterials. 2013;34(9):2185–2193. doi: 10.1016/j.biomaterials.2012.11.039. [DOI] [PubMed] [Google Scholar]
- 46.Hatsushika D., Muneta T., Nakamura T. Repetitive allogeneic intraarticular injections of synovial mesenchymal stem cells promote meniscus regeneration in a porcine massive meniscus defect model. Osteoarthritis Cartilage. 2014;22(7):941–950. doi: 10.1016/j.joca.2014.04.028. [DOI] [PubMed] [Google Scholar]
- 47.Shimomura K., Bean A.C., Lin H., Nakamura N., Tuan R.S. In vitro repair of meniscal radial tear using aligned electrospun nanofibrous scaffold. Tissue Eng. 2015;21(13–14):2066–2075. doi: 10.1089/ten.tea.2014.0549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Piontek T., Ciemniewska-Gorzela K., Naczk J. Complex meniscus tears treated with collagen matrix wrapping and bone marrow blood injection: a 2-year clinical follow-up. Cartilage. 2016;7(2):123–139. doi: 10.1177/1947603515608988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Liu C., Toma I.C., Mastrogiacomo M., Krettek C., von Lewinski G., Jagodzinski M. Meniscus reconstruction: today's achievements and premises for the future. Arch Orthop Trauma Surg. 2013;133(1):95–109. doi: 10.1007/s00402-012-1624-2. [DOI] [PubMed] [Google Scholar]
- 50.Milachowski K.A., Kohn D., Wirth C.J. Meniscus replacement using Hoffa's infrapatellar fat bodies–initial clinical results. Unfallchirurgie. 1990;16(4):190–195. doi: 10.1007/BF02588774. [DOI] [PubMed] [Google Scholar]
- 51.Kohn D. Autograft meniscus replacement: experimental and clinical results. Knee Surg Sports Traumatol Arthrosc. 1993;1(2):123–125. doi: 10.1007/BF01565466. [DOI] [PubMed] [Google Scholar]
- 52.Pressel T., VL G., Kohn D., Wirth C.J. Meniscus replacement with quadriceps tendon–a long-term analysis. Zeitschrift fur Orthopadie und ihre Grenzgebiete. 2005;143(1):42–47. doi: 10.1055/s-2005-836359. [DOI] [PubMed] [Google Scholar]
- 53.Johnson L.L., Feagin J.A., Jr. Autogenous tendon graft substitution for absent knee joint meniscus: a pilot study. Arthroscopy. 2000;16(2):191–196. doi: 10.1016/s0749-8063(00)90035-5. [DOI] [PubMed] [Google Scholar]
- 54.Walsh C.J., Goodman D., Caplan A.I., Goldberg V.M. Meniscus regeneration in a rabbit partial meniscectomy model. Tissue Eng. 1999;5(4):327–337. doi: 10.1089/ten.1999.5.327. [DOI] [PubMed] [Google Scholar]
- 55.Cisa J., Basora J., Madarnas P., Ghibely A., Navarro-Quilis A. Meniscal repair by synovial flap transfer. Healing of the avascular zone in rabbits. Acta Orthop Scand. 1995;66(1):38–40. doi: 10.3109/17453679508994636. [DOI] [PubMed] [Google Scholar]
- 56.Bruns J., Kampen J., Kahrs J., Plitz W. Autologous meniscus replacement with rib perichondrium. Experimental results. Orthopä. 2000;29(2):145–150. doi: 10.1007/s001320050023. [DOI] [PubMed] [Google Scholar]
- 57.Peters G., Wirth C.J. The current state of meniscal allograft transplantation and replacement. Knee. 2003;10(1):19–31. doi: 10.1016/s0968-0160(02)00139-4. [DOI] [PubMed] [Google Scholar]
- 58.Rosso F., Bisicchia S., Bonasia D.E., Amendola A. Meniscal allograft transplantation: a systematic review. Am J Sports Med. 2015;43(4):998–1007. doi: 10.1177/0363546514536021. [DOI] [PubMed] [Google Scholar]
- 59.Vundelinckx B., Vanlauwe J., Bellemans J. Long-term subjective, clinical, and radiographic outcome evaluation of meniscal allograft transplantation in the knee. Am J Sports Med. 2014;42(7):1592–1599. doi: 10.1177/0363546514530092. [DOI] [PubMed] [Google Scholar]
- 60.Rodeo S.A., Seneviratne A., Suzuki K., Felker K., Wickiewicz T.L., Warren R.F. Histological analysis of human meniscal allografts. A preliminary report. J Bone Joint Surg Am. 2000;82-a(8):1071–1082. doi: 10.2106/00004623-200008000-00002. [DOI] [PubMed] [Google Scholar]
- 61.Vrancken A.C., Buma P., van Tienen T.G. Synthetic meniscus replacement: a review. Int Orthop. 2013;37(2):291–299. doi: 10.1007/s00264-012-1682-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Monllau J.C., Gelber P.E., Abat F. Outcome after partial medial meniscus substitution with the collagen meniscal implant at a minimum of 10 years' follow-up. Arthroscopy. 2011;27(7):933–943. doi: 10.1016/j.arthro.2011.02.018. [DOI] [PubMed] [Google Scholar]
- 63.Bulgheroni E., Grassi A., Bulgheroni P., Marcheggiani Muccioli G.M., Zaffagnini S., Marcacci M. Long-term outcomes of medial CMI implant versus partial medial meniscectomy in patients with concomitant ACL reconstruction. Knee Surg Sports Traumatol Arthrosc. 2015;23(11):3221–3227. doi: 10.1007/s00167-014-3136-9. [DOI] [PubMed] [Google Scholar]
- 64.Rodkey W.G., DeHaven K.E., Montgomery W.H., 3rd Comparison of the collagen meniscus implant with partial meniscectomy. A prospective randomized trial. J Bone Joint Surg Am. 2008;90(7):1413–1426. doi: 10.2106/JBJS.G.00656. [DOI] [PubMed] [Google Scholar]
- 65.Zaffagnini S., Marcheggiani Muccioli G.M., Lopomo N. Prospective long-term outcomes of the medial collagen meniscus implant versus partial medial meniscectomy: a minimum 10-year follow-up study. Am J Sports Med. 2011;39(5):977–985. doi: 10.1177/0363546510391179. [DOI] [PubMed] [Google Scholar]
- 66.Leroy A., Beaufils P., Faivre B., Steltzlen C., Boisrenoult P., Pujol N. Actifit(R) polyurethane meniscal scaffold: MRI and functional outcomes after a minimum follow-up of 5 years. Orthop Traumatol Surg Res: OTSR. 2017;103(4):609–614. doi: 10.1016/j.otsr.2017.02.012. [DOI] [PubMed] [Google Scholar]
- 67.Schuttler K.F., Haberhauer F., Gesslein M. Midterm follow-up after implantation of a polyurethane meniscal scaffold for segmental medial meniscus loss: maintenance of good clinical and MRI outcome. Knee Surg Sports Traumatol Arthrosc. 2016;24(5):1478–1484. doi: 10.1007/s00167-015-3759-5. [DOI] [PubMed] [Google Scholar]
- 68.Bulgheroni E., Grassi A., Campagnolo M., Bulgheroni P., Mudhigere A., Gobbi A. Comparative study of collagen versus synthetic-based meniscal scaffolds in treating meniscal deficiency in young active population. Cartilage. 2016;7(1):29–38. doi: 10.1177/1947603515600219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bouyarmane H., Beaufils P., Pujol N. Polyurethane scaffold in lateral meniscus segmental defects: clinical outcomes at 24 months follow-up. Orthop Traumatol Surg Res: OTSR. 2014;100(1):153–157. doi: 10.1016/j.otsr.2013.10.011. [DOI] [PubMed] [Google Scholar]
- 70.Hirschmann M.T., Keller L., Hirschmann A. One-year clinical and MR imaging outcome after partial meniscal replacement in stabilized knees using a collagen meniscus implant. Knee Surg Sports Traumatol Arthrosc. 2013;21(3):740–747. doi: 10.1007/s00167-012-2259-0. [DOI] [PubMed] [Google Scholar]
- 71.Linder-Ganz E., Elsner J.J., Danino A., Guilak F., Shterling A. A novel quantitative approach for evaluating contact mechanics of meniscal replacements. J Biomech Eng. 2010;132(2):024501. doi: 10.1115/1.4000407. [DOI] [PubMed] [Google Scholar]
- 72.Pereira H., Frias A.M., Oliveira J.M., Espregueira-Mendes J., Reis R.L. Tissue engineering and regenerative medicine strategies in meniscus lesions. Arthroscopy. 2011;27(12):1706–1719. doi: 10.1016/j.arthro.2011.08.283. [DOI] [PubMed] [Google Scholar]
- 73.Moran C.J., Busilacchi A., Lee C.A., Athanasiou K.A., Verdonk P.C. Biological augmentation and tissue engineering approaches in meniscus surgery. Arthroscopy. 2015;31(5):944–955. doi: 10.1016/j.arthro.2014.11.044. [DOI] [PubMed] [Google Scholar]
- 74.Langer R., Vacanti J.P. Tissue engineering. Science. 1993;260(5110):920–926. doi: 10.1126/science.8493529. [DOI] [PubMed] [Google Scholar]
- 75.Kang S.W., Son S.M., Lee J.S. Regeneration of whole meniscus using meniscal cells and polymer scaffolds in a rabbit total meniscectomy model. J Biomed Mater Res A. 2006;77(4):659–671. doi: 10.1002/jbm.a.30579. [DOI] [PubMed] [Google Scholar]
- 76.Kon E., Chiari C., Marcacci M. Tissue engineering for total meniscal substitution: animal study in sheep model. Tissue Eng. 2008;14(6):1067–1080. doi: 10.1089/ten.tea.2007.0193. [DOI] [PubMed] [Google Scholar]
- 77.Yamasaki T., Deie M., Shinomiya R., Yasunaga Y., Yanada S., Ochi M. Transplantation of meniscus regenerated by tissue engineering with a scaffold derived from a rat meniscus and mesenchymal stromal cells derived from rat bone marrow. Artif Organs. 2008;32(7):519–524. doi: 10.1111/j.1525-1594.2008.00580.x. [DOI] [PubMed] [Google Scholar]
- 78.Moutos F.T., Guilak F. Functional properties of cell-seeded three-dimensionally woven poly(epsilon-caprolactone) scaffolds for cartilage tissue engineering. Tissue Eng. 2010;16(4):1291–1301. doi: 10.1089/ten.tea.2009.0480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Merriam A.R., Patel J.M., Culp B.M., Gatt C.J., Jr., Dunn M.G. Successful total meniscus reconstruction using a novel fiber-reinforced scaffold: a 16- and 32-week study in an ovine model. Am J Sports Med. 2015;43(10):2528–2537. doi: 10.1177/0363546515595065. [DOI] [PubMed] [Google Scholar]
- 80.Patel J.M., Merriam A.R., Culp B.M., Gatt C.J., Jr., Dunn M.G. One-year outcomes of total meniscus reconstruction using a novel fiber-reinforced scaffold in an ovine model. Am J Sports Med. 2016;44(4):898–907. doi: 10.1177/0363546515624913. [DOI] [PubMed] [Google Scholar]
- 81.Shimomura K., Rothrauff B.B., Tuan R.S. Region-specific effect of the decellularized meniscus extracellular matrix on mesenchymal stem cell-based meniscus tissue engineering. Am J Sports Med. 2017;45(3):604–611. doi: 10.1177/0363546516674184. [DOI] [PubMed] [Google Scholar]
- 82.Rothrauff B.B., Shimomura K., Gottardi R., Alexander P.G., Tuan R.S. Anatomical region-dependent enhancement of 3-dimensional chondrogenic differentiation of human mesenchymal stem cells by soluble meniscus extracellular matrix. Acta Biomater. 2017;49:140–151. doi: 10.1016/j.actbio.2016.11.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Li C., Cheung T.F., Fan V.C., Sin K.M., Wong C.W., Leung G.K. Applications of three-dimensional printing in surgery. Surg Innovat. 2017;24(1):82–88. doi: 10.1177/1553350616681889. [DOI] [PubMed] [Google Scholar]
- 84.Tack P., Victor J., Gemmel P., Annemans L. 3D-printing techniques in a medical setting: a systematic literature review. Biomed Eng Online. 2016;15(1):115. doi: 10.1186/s12938-016-0236-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wong T.M., Jin J., Lau T.W. The use of three-dimensional printing technology in orthopaedic surgery. J Orthop Surg. 2017;25(1) doi: 10.1177/2309499016684077. 2309499016684077. [DOI] [PubMed] [Google Scholar]
- 86.Grogan S.P., Chung P.H., Soman P. Digital micromirror device projection printing system for meniscus tissue engineering. Acta Biomater. 2013;9(7):7218–7226. doi: 10.1016/j.actbio.2013.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lee C.H., Rodeo S.A., Fortier L.A., Lu C., Erisken C., Mao J.J. Protein-releasing polymeric scaffolds induce fibrochondrocytic differentiation of endogenous cells for knee meniscus regeneration in sheep. Sci Transl Med. 2014;6(266) doi: 10.1126/scitranslmed.3009696. 266ra171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zhang Z.Z., Wang S.J., Zhang J.Y. 3D-Printed poly(epsilon-caprolactone) scaffold augmented with mesenchymal stem cells for total meniscal substitution: a 12- and 24-week animal study in a rabbit model. Am J Sports Med. 2017;45(7):1497–1511. doi: 10.1177/0363546517691513. [DOI] [PubMed] [Google Scholar]
- 89.Nordberg R.C., Bodle J.C., Loboa E.G. Mechanical stimulation of adipose-derived stem cells for functional tissue engineering of the musculoskeletal system via cyclic hydrostatic pressure, simulated microgravity, and cyclic tensile strain. Meth Mol Biol. 2018;1773:215–230. doi: 10.1007/978-1-4939-7799-4_18. [DOI] [PubMed] [Google Scholar]