Abstract
Context: Inflammation and cell differentiation lead to a number of severe diseases. In the recent years, various studies focused on the anti-inflammatory and anticancer activity of essential oils (EOs) of numerous plants, including different Pinus species.
Objective: The phytochemical composition, anti-inflammatory and cytotoxic activity of EOs from needles and twigs of Pinus heldreichii Christ (Pinaceae) and P. peuce Griseb., and from needles, twigs and cones of P. mugo Turra were determined.
Materials and methods: For separation and identification of the EOs, gas chromatography/flame ion detector (GC/FID) and GC/mass spectrometry were performed. The amount of secreted IL-6 in a lipopolysaccharide (LPS)-stimulated macrophage model was quantified (concentration of oils: 0.0001–0.2%, 3 h incubation). Cytotoxicity on the cancer cell lines HeLa, CaCo-2 and MCF-7 were determined using a MTT (Thiazolyl Blue Tetrazolium Bromide) assay (concentration of oils: 0.001–0.1%, 24 h incubation).
Results: The most prominent members in the oils include: δ-3-carene, α-pinene and linalool-acetate (P. mugo); α-pinene, β-phellandrene and β-pinene (P. peuce); limonene, α-pinene and (E)-caryophyllene (P. heldreichii). EOs showed significant cytotoxic effects on cancer cell lines (IC50 0.007 to >0.1%), with a reduction in cell viability with up to 90% at a concentration of 0.1%, and anti-inflammatory activity (IC50 0.0008–0.02%) with a reduction of IL-6 secretion with up to 60% at a concentration of 0.01%.
Discussion and conclusion: The EOs of needles and twigs from P. peuce and P. heldreichii as well as of needles, twigs and cones of P. mugo can be considered as promising agents for anticancer and anti-inflammatory drugs.
Keywords: Pinus mugo, Pinus peuce, Pinus heldreichii, inflammation, cancer
Introduction
Essential oils (EOs) are plant volatile oils that have been widely used in traditional medicine. Recently, EOs biological activity (antimicrobial, anti-inflammatory, antifungal, antiviral, antioxidant and anticancer) was shown in scientific studies (Bakkali et al. 2008; Adorjan & Buchbauer 2010; Bayala et al. 2014). The chemical composition of the EOs is affected by many factors, such as genetic variation, geographic location, climate, season, stress during growth, maturity, drying and storage, thus influencing their biological properties (Croteau 1986; Hussain et al. 2008; Raut & Karuppayil 2014).
Inflammatory and oxidative disorders lead to a number of diseases including cancer (Lahlou 2004; Bayala et al. 2014). Cancer belongs to the second largest diseases, causing more than 10% of deaths worldwide (Loizzo et al. 2008). Several drugs are available as anti-inflammatory (i.e., non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids) and anticancer agents. Treatment of cancer with chemotherapy and/or radiation is associated with high systemic toxicity and development of drug resistance, resulting in unsuccessful treatment (Buchholz & Gress 2009). Medical treatment of inflammatory disorders provides an effective curative response, but it is frequently connected to several side effects due to its regular use (Perrone et al. 2003; Hippisley-Cox & Coupland 2005). This led to an increased interest of research in considering plant extracts as potential source for the development of novel alternative anticancer or anti-inflammatory agents (Efferth et al. 2007).
Recent studies reported that EOs from some plants reduce and inhibit the production of inflammatory mediators by different mechanisms and exert anti-proliferative and cytotoxic effects against cancer cells (Adorjan & Buchbauer 2010). For example, the EOs of Abies koreana E.H.Wilson (Pinaceae) showed a significant inhibition of the pro-inflammatory mediators IL-1β, IL-6, TNF-α, NO and PGE2 in LPS-stimulated macrophages (Yoon et al. 2009). Illicium anisatum L. (Illiciaceae) EO decreased the expression of iNOS and COX-2 and is additionally an effective inhibitor of LPS-induced PGE2 and NO production in macrophages (Kim et al. 2009). The EOs of Cymbopogon flexuosus Stapf. (Poaceae) showed anticancer activity against 502713 (colon) and IMR-32 (neuroblastoma) cell lines and led to apoptosis in HL-60 (promyeloblast) cell lines (Sharma et al. 2009). Anemopsis californica (Nutt.) Hook. & Arn. (Saururaceae) EOs showed anti-proliferative activity of AN3CA (uterine) and HeLa (cervical) cell lines; EOs such as β-caryophyllene and α-humulene exerted significant anticancer activity in MCF-7 (breast) and DLD-1 (colon) cell lines (Legault & Pichette 2007; Medina-Holguín et al. 2008).
There are several studies investigating the chemical composition of the EOs of selected parts of Pinus mugo Turra (Pinaceae), P. peuce Griseb. and P. heldreichii Christ obtained from Kosovo flora, but to the best of our knowledge there are, as yet, no reports about the anti-inflammatory activity and cytotoxic effects of the EOs from these Pinus spp. (Stevanović-Janežić & Vilotić 1998; Hajdari et al. 2015). In this study, we assess the anti-inflammatory and cytotoxic potential of the EOs of P. mugo (needles, twigs and cones), P. peuce (needles and twigs) and P. heldreichii (needles and twigs), identify the main components and compare the chemical composition of these Pinus spp. with previous studies.
Materials and methods
Chemicals and reagents
Dulbecco’s minimum essential medium (DMEM), foetal bovine serum (FBS), l-glutamine and a penicillin/streptomycin mixture were obtained from Life Technologies (Carlsbad, CA). Raw 264.7, HeLa, CaCo-2 and MCF-7 cell lines were purchased from American Type Culture Collection (ATCC-TIB-71, ATCC-CCL-2, ATCC-HTB-37, ATCC-HTB-22). Thiazolyl Blue Tetrazolium Bromide (MTT) was obtained from Sigma-Aldrich. Hydrochloric acid, disodium hydrogen phosphate, potassium chloride, sodium chloride and sodium dodecyl sulphate (SDS) were purchased from Merck (Darmstadt, Germany). Potassium dihydrogen phosphate was obtained from Sigma-Aldrich (St. Louis, MO).
Plant material
Needles, twigs and cones of P. mugo, P. peuce and P. heldreichii collected from July to September 2013 from wild populations in Kosovo. The populations were originated from ‘Sharri’ National Park, Mountain (Mt.) Ošljak (P. mugo – 42°14′41E″N, 20°55′06″E, 1740 m a.s.l; P. peuce – 42°15′11″N, 20°54′56″E, 1627 m a.s.l.; P. heldreichii – 42°11′02″N, 20°54′56″E, 1555 m a.s.l). The collection sites were recorded using a Global Positioning System (GPS) receiver (GARMIN, eTrex® 30). Two to four replicate samples of needles, twigs and cones were analyzed, each sample was gathered from 2–3 individual plants from each population. Samples were distilled and analyzed separately. Voucher specimens of each population were deposited at the Herbarium of the Department of Biology, University of Prishtina and authenticated by the University of Prishtina staff botanist Avni Hajdari.
Essential oil extraction
The plant material was air-dried in shade at room temperature and cut into small pieces (<0.5 cm). Separated needles, twigs (only wooden parts) and cones were subjected to essential oil distillation. For distillation, 50 g of dry tissue was placed into 0.5 L of water in a 1 L flask and distilled in a Clevenger apparatus for 3 h at a rate of 3 mL/min. The samples were stored in the dark at −18 °C in the freezer pending further analysis. The yield of essential oil is expressed as a percentage of the mass of the essential oil with respect to the air-dried material (% v/w of dried material).
Gas chromatography/flame ionization detector (GC/FID) and gas chromatography/mass spectrometry (GC/MS) analysis
GC/FID analyses were performed using an Agilent 7890 A GC System equipped with an FID detector (Agilent Technologies, Santa Clara, CA). The separation was conducted on an HP-5MS column 30 m × 0.25 mm with 0.25 μm film thickness. Helium was used as carrier gas with an initial flow rate of 0.6 mL/min and subsequently at a constant pressure of 50 psi. The front inlet was maintained at 250 °C in a split ratio of 50:1. The GC oven temperature was increased from 60 °C to 260 °C at a rate of 5 °C/min and the FID operated at 280 °C with an air flow of 350 mL/min and a hydrogen flow of 35 mL/min. The injection volume was 1 μL.
GC/MS analyses were performed using an Agilent 7890 A GC system coupled to a 5975 C MSD (Agilent Technologies). The ionization energy was 70 eV with a mass range of 40–400 m/z. The separation was conducted with the same column and temperature program as for the analytical GC.
Identification of the components of the essential oil was made by comparing their Kovats retention indexes with those in the literature (Adams 1997). The calculation of the Kovats index was made based on a linear interpolation of the retention time of the homologous series of n-alkanes (C9-C28) under the same operating conditions. Furthermore, the components were identified by comparing the mass spectra of each constituent with those stored in the NIST 08.L and WILEY MS 9th database and with mass spectra from the literature (Adams 1997). The percentage composition of the oils was computed by the normalization method from the GC peak areas, calculated as the mean of three samples, without correction factors.
Assay of anti-inflammatory activity
To examine the anti-inflammatory effect of the EOs, an assay with murine macrophages was performed as described in Mueller et al. (2010). RAW 264.7 cells were seeded at a density of 2 × 106 cells/mL into 24 well plates and incubated for 24 h at 37 °C, using DMEM (+10% FBS, 100 U/mL penicillin/streptomycin, 4 mM l-glutamine) as culture medium. On the following day, the oils were added in concentrations ranging from 0.0001–0.2% and the cells were further incubated for 3 h. The macrophages were then treated with LPS at a final concentration of 1 μg/mL to stimulate cytokine production. After 24 h of incubation, the supernatant was removed and centrifuged to remove any cell residues. The IL-6 secretion was quantified using an ELISA, according to the manufacturer’s protocol (eBioscience).
In parallel, the viability of the LPS-stimulated cells was assessed using an MTT assay, based on the reduction of MTT to formazan by living cells. 50 μL of MTT solution (5 mg/mL in 1 × phosphate-buffered saline, PBS) was added to the cells and the plate was incubated for 2 h at normal culture conditions. The supernatant was then removed and the cells were lysed with a buffer, containing 10% SDS in 0.01 N HCl. Using a microplate reader (Infinite M200, Tecan, Austria), the optical density was measured at 570 nm, corrected by a reference wavelength of 690 nm.
To reduce any variation from differences in cell density, the ELISA results were normalized to the MTT values. The concentration of cytokines of the positive control (cells only treated with LPS) was defined as 100%. All results from the tested oils were then calculated as a percentage of the positive control. The entire inflammation assay was performed in triplicate on independent days.
Assay of cytotoxicity to cancer cell lines
To determine cell viability and thus potential cytotoxicity of substances to three different cancer cell lines (HeLa, CaCo-2, MCF-7), an MTT-assay was performed as described above, but in 96-well plates. Cells were seeded at a density of 2 × 106 cells/mL and incubated for 24 h before addition of the test substances at concentrations ranging from 0.001–0.1% and further incubation of 24 h. Then, the wells were loaded with 10 μL MTT solution. After another 2 h of incubation, the supernatant of each well was removed and the cells were lysed with 100 μL of lysis buffer. The absorption was measured at 570 nm and corrected by a reference wavelength of 690 nm. The amount of cells of the positive control (cells only incubated with DMEM) was defined as 100%. The results from the test substances were calculated as a % of the positive control. The entire assay of cytotoxicity was performed in triplicate on independent days.
Statistical calculations
The results of a triplicate are expressed as means ± standard deviation. IC50 was determined using a logistic dose-response model of the Table Curve 2 D software (Systat Software, San Jose, CA).
Results
Essential oil composition
The yield and chemical composition of oils from three Pinus spp. are presented in Table 1. In total, 58 compounds of the EOs were characterized from P. mugo needles, twigs and cones, representing 97.2, 96.1 and 72.1% of the total identified components, respectively. The main components detected in needles, twigs and cones oils are: δ-3-carene (19.3, 28.1 and 15.8%); α-pinene (21.3, 8.2 and 4.1%); (E)-caryophyllene (6.7, 4.1 and 20.2%); limonene (3.7, 19.5 and 2.2%); β-pinene (3.7, 11 and 1.6%), linalool-acetate (0.1, 7.2 and 1.2%), germacrene D (6.1, 0.4 and 1.2%), linalool (0.2, 0.1 and 4.6%).
Table 1.
Composition [%] of the EOs of P. mugo, P. peuce and P. heldreichii.
|
P. mugo |
P. peuce |
P. heldreichii |
||||||
|---|---|---|---|---|---|---|---|---|
| Constituentsa | KIb | Needles | Twigs | Cones | Needles | Twigs | Needles | Twigs |
| Tricyclene | 926 | 0.80 | 0.17 | tr | 0.53 | 0.13 | 0.16 | tr |
| α-Thujene | 930 | 1.44 | 0.26 | 0.20 | – | – | – | – |
| α-Pinene | 940 | 21.34 | 8.20 | 4.10 | 36.79 | 15.96 | 10.57 | 14.32 |
| Camphene | 948 | 2.82 | 0.65 | 0.30 | 8.04 | 2.00 | 0.64 | 0.43 |
| Thuja-2,4(10)-diene | 960 | 0.11 | 0.54 | 0.70 | – | – | – | – |
| Sabinene | 975 | 0.95 | 1.12 | 0.40 | 0.06 | 0.35 | tr | 0.03 |
| β-Pinene | 979 | 3.66 | 10.99 | 1.60 | 13.00 | 21.48 | 1.66 | 0.89 |
| Myrcene | 990 | 1.94 | 2.36 | 1.10 | 0.79 | 1.98 | 1.56 | 1.86 |
| α-Phellandrene | 1005 | 0.26 | 0.38 | tr | 0.21 | tr | tr | tr |
| δ-3-Carene | 1011 | 19.29 | 28.05 | 15.80 | 0.20 | 0.86 | 0.11 | tr |
| α-Terpinene | 1018 | 0.37 | 0.23 | 0.20 | tr | tr | – | – |
| p-Cymene | 1024 | 0.32 | 0.38 | 0.30 | – | – | – | – |
| β-Phellandrene | 1029 | – | – | – | 6.07 | 35.82 | – | – |
| Limonene | 1031 | 3.67 | 19.50 | 2.20 | – | – | 43.93 | 64.22 |
| β-E-Ocimene | 1050 | 0.60 | tr | tr | 0.44 | 0.27 | – | – |
| γ-Terpinene | 1062 | 0.72 | 0.42 | 0.40 | 0.27 | 0.17 | – | – |
| γ-Terpinolene | 1088 | 3.60 | 2.62 | 1.10 | 0.29 | 0.35 | 0.20 | 0.05 |
| Linalool | 1098 | 0.24 | 0.12 | 4.60 | 0.69 | 0.21 | – | – |
| α-Campholenal | 1127 | tr | 0.31 | 0.20 | tr | 0.11 | – | – |
| E-Pinocarveol | 1139 | 0.16 | 0.20 | 0.20 | 0.44 | 0.24 | 0.43 | 0.04 |
| Z-Verbenol | 1141 | – | – | – | 0.32 | 0.15 | – | – |
| Camphor | 1146 | 0.42 | 0.29 | 0.30 | – | – | – | – |
| Borneol | 1165 | 0.60 | 0.57 | 1.00 | 0.11 | 0.22 | 0.16 | tr |
| Terpinene-4-ol | 1177 | 0.10 | 0.21 | tr | tr | 0.25 | 0.20 | tr |
| p-Cymen-8-ol | 1183 | 0.23 | 0.23 | tr | – | – | – | – |
| α-Terpinol | 1188 | 0.25 | 0.24 | 0.80 | 9.27 | 2.37 | 0.31 | 0.11 |
| Methyl salicylate | 1191 | 0.23 | 0.88 | 0.20 | – | – | – | – |
| Myrtenol | 1195 | 0.50 | 0.20 | 0.20 | 0.20 | 0.24 | 0.61 | 0.06 |
| E-Piperitol | 1208 | – | – | – | 1.20 | 0.29 | – | – |
| Thymol methyl ether | 1235 | 0.16 | tr | 1.10 | – | – | – | – |
| Carvone | 1243 | – | – | – | – | – | 0.22 | 0.14 |
| Linalool acetate | 1256 | 0.11 | 7.23 | 1.20 | 0.14 | tr | – | – |
| Bornyl acetate | 1285 | 3.03 | tr | 0.50 | 4.23 | 3.30 | 0.35 | 0.60 |
| α-Copane | 1376 | 0.14 | 0.19 | 0.20 | 0.23 | 0.36 | 0.16 | tr |
| β-Bourbonene | 1384 | 0.25 | tr | tr | 0.13 | 0.61 | 0.13 | tr |
| β-Elemene | 1391 | 1.46 | 0.86 | 0.30 | tr | tr | 0.18 | tr |
| Longifolene | 1407 | 0.33 | tr | 1.40 | – | – | tr | 1.31 |
| Unknown 1 | 1420 | tr | 0.23 | 0.90 | – | – | – | – |
| E-Caryophyllene | 1418 | 6.68 | 4.05 | 20.20 | tr | tr | 4.40 | 0.33 |
| β-Copaene | 1432 | 0.33 | 0.23 | 0.50 | tr | 0.11 | 0.11 | 0.11 |
| α-Humulene | 1454 | 0.43 | 0.17 | tr | tr | 0.13 | 2.11 | 0.95 |
| Aromadendrene | 1458 | 1.07 | 0.88 | 3.20 | – | – | – | – |
| Z-Muurola-4(14),5-diene | 1466 | 0.37 | 0.05 | 0.20 | 0.23 | 1.01 | tr | 0.41 |
| β-Chamigrene | 1477 | tr | 0.27 | 0.30 | – | – | – | – |
| γ-Muurolene | 1479 | 0.87 | tr | 0.60 | – | – | 0.93 | 1.39 |
| Germacrene D | 1481 | 6.14 | 0.39 | 1.20 | 10.00 | 4.69 | 17.17 | 2.04 |
| Bicyclogermacrene | 1500 | 2.11 | 0.61 | 0.30 | 0.86 | 0.32 | – | – |
| α-Muurolene | 1500 | 0.71 | 0.20 | 0.30 | – | – | 0.22 | 0.14 |
| γ-Cadinene | 1519 | 0.90 | 0.19 | 0.30 | 0.29 | 0.81 | 0.40 | 0.19 |
| β-Cadinene | 1519 | – | – | – | – | – | 0.92 | 0.18 |
| δ-Cadinene | 1523 | 2.33 | 0.43 | 0.70 | 0.86 | 0.90 | 1.08 | 0.18 |
| trans-Cadina-1,4-diene | 1534 | 0.52 | 0.10 | 0.40 | – | – | 0.22 | 0.12 |
| α-Cadinene | 1538 | 0.41 | 0.31 | 0.70 | 0.31 | tr | – | – |
| E-Nerolidol | 1561 | 0.43 | 0.20 | 0.20 | 0.24 | tr | – | – |
| Germacrene-4-ol | 1582 | tr | tr | tr | 0.27 | 0.13 | 0.14 | 0.07 |
| Spathulenol | 1578 | 1.09 | 0.21 | 0.20 | 0.33 | 0.40 | 0.97 | tr |
| Caryophyllene oxide | 1583 | 1.49 | 0.29 | 0.40 | 0.14 | 0.22 | 3.11 | 2.09 |
| Humolene epoxide II | 1636 | – | – | – | 0.35 | 0.16 | 0.12 | 0.15 |
| Z-Cadin-4-en-7ol | 1636 | – | – | – | 0.51 | 0.42 | 0.65 | 0.07 |
| epi-α-Cadinol | 1640 | 0.99 | tr | 0.30 | – | – | 0.11 | 0.58 |
| α-Muurolol | 1645 | 0.18 | 0.14 | 1.30 | 0.19 | 0.00 | 0.29 | 0.09 |
| α-Cadinol | 1653 | – | – | – | 0.14 | 0.13 | 0.17 | 0.08 |
| Khusinol | 1680 | – | – | – | – | – | 0.20 | 0.67 |
| Eudesma-4(15),7-diene-1-β-ol | 1688 | – | – | – | 0.19 | 0.18 | 0.17 | 0.12 |
| E-Nerolidylacetate | 1717 | – | – | – | – | – | tr | 0.72 |
| Oplopanone | 1733 | – | – | – | – | – | tr | 0.65 |
| Benzyl benzoate | 1762 | – | – | – | – | – | tr | 0.44 |
| Phenyl ethyl octanoate | 1847 | – | – | – | – | – | tr | 0.59 |
| 3Z-Hexenyl cinnamate | 1881 | – | – | – | – | – | tr | 0.43 |
| Abietadiene | 2087 | tr | tr | 0.20 | – | – | – | – |
| Unknown 2 | 2116 | tr | 0.71 | 20.20 | – | – | – | – |
| Unknown 3 | 2159 | 0.91 | 0.69 | tr | – | – | – | – |
| Unknown 4 | 0.06 | 0.60 | 4.80 | – | – | – | – | |
| Total identified | 97.15 | 96.12 | 72.1 | 98.55 | 97.32 | 95.09 | 96.83 | |
| Yield %v/w (min and max values) | 0.5–0.6 | 1.1–1.2 | 0.4–0.5 | 0.7–1.0 | 3.0–3.3 | 0.2–0.3 | 0.8–1.2 | |
Compounds are listed in order of elution from an HP-5MS column and their percentages were obtained by FID peak-area normalization.
Kovats indices calculated against a C9-C28 n-alkanes mixture on the HP5 MS column. tr: traces.
Forty-five compounds were characterized in the oils of needles and twigs of P. peuce, representing 98.6% and 97.3% of the total identified components, respectively. The major constituents identified on the needle and twig oils, respectively are: α-pinene (36.8% and 16%); β-phellandrene (6.1% and 35.8%); β-pinene (13% and 21.5%), germacrene D (10% and 4.7%); α-terpinol (9.3% and 2.4%); camphene (8% and 2%); bornyl acetate (4.2% and 3.3%).
The major constituents from 47 ones, identified from the EOs of P. heldreichii in needles and twigs, respectively are: limonene (43.9% and 64.2%); germacrene D (17.2% and 2%); α-pinene (10.6, and 14.3%); (E)-caryophyllene (4.4% and 0.3%); caryophyllene oxide (3.1% and 2.1%); α-humulene (2.1% and 1%). The constituents identified represent 95.1% and 96.8% of the total oils in needles and twigs.
Anti-inflammatory effect
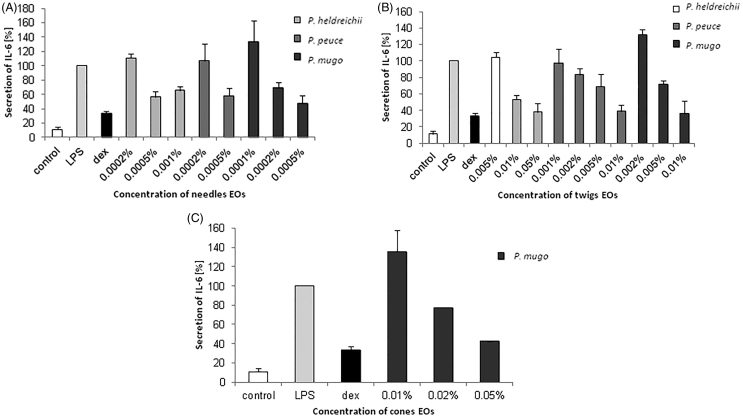
EOs obtained from needles of P. heldreichii, P. peuce and P. mugo reduced the secretion of the pro-inflammatory cytokine IL-6 by at least 35% at a concentration of 0.001%, while the oils obtained from twigs of these three Pinus spp. and the cones of P. mugo decreased the secretion of IL-6 by at least 60% at a concentration of 0.01% for the oils obtained from twigs and 0.05% for the cones oils (Figure 1). IC50 values were determined with the needles EOs of P. heldreichii and P. mugo showing the lowest IC50 (0.0002), followed by P. peuce needles and twigs oils (>0.0005). EOs obtained from twigs of P. heldreichii and P. mugo showed an IC50 of >0.004 and the oils of cones of P. mugo had an IC50 of 0.02 (Table 2).
Figure 1.
Concentration-depended reduction of IL-6 secretion of LPS-stimulated macrophages, as determined by ELISA, after treatment with (A) the needle and (B) twig oils of P. heldreichii, P. peuce and P. mugo, (C) the cone oils of P. mugo and 5 μM dexamethasone (dex).
Table 2.
IC50 values [%] of P. heldreichii, P. peuce and P. mugo.
| IC50 [%] |
||||
|---|---|---|---|---|
| For cytotoxicity |
||||
| Substances | For IL-6 reduction | To HeLa | To CaCo-2 | To MCF-7 |
| P. heldreichii twigs | 6 × 10−3 | 6 × 10−2 | >1 × 10−1 | >1 × 10−1 |
| P. heldreichii needles | 2 × 10−4 | 2 × 10−2 | 2 × 10−2 | >1 × 10−1 |
| P. peuce twigs | 8 × 10−4 | 2 × 10−2 | 4 × 10−2 | 6 × 10−2 |
| P. peuce needles | >5 × 10−4 | 7 × 10−3 | 2 × 10−2 | 6 × 10−2 |
| P. mugo twigs | 4 × 10−3 | 6 × 10−2 | 6 × 10−2 | >1 × 10−1 |
| P. mugo needles | 2 × 10−4 | 3 × 10−1 | 2 × 10−2 | 3 × 10−1 |
| P. mugo cones | 2 × 10−2 | >1 × 10−1 | >1 × 10−1 | >1 × 10−1 |
Cytotoxicity to cancer cell lines
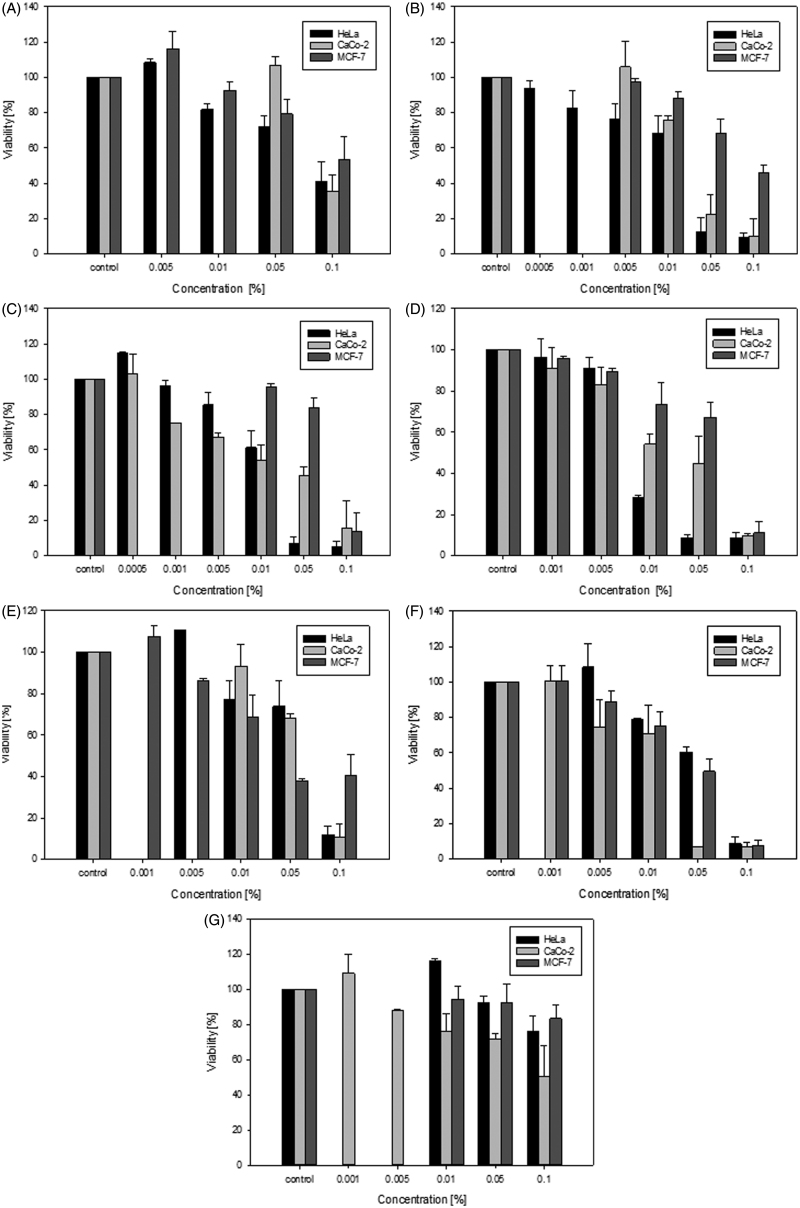
All EOs showed a significant cytotoxic effect on the cancer cell lines HeLa, CaCo-2 and MCF-7 (Figure 2). EOs obtained from P. heldreichii needles, P. peuce needles and twigs, and P. mugo needles and twigs at 0.1% concentration decreased the cell viability of HeLa by at least 90%, followed by oils obtained from P. heldreichii twigs with almost 60%. EOs of P. mugo cones had little cytotoxic effect with about 25%. IC50 values were determined with oils of the needles of P. peuce having the lowest value (0.007), followed by oils of the needles of P. heldreichii and oils of the twigs of P. peuce (0.02). Oils obtained from twigs of P. heldreichii and P. mugo showed an IC50 of 0.06, followed by P. mugo cones (>0.1) and P. mugo needles (0.3) (Table 2).
Figure 2.
Concentration-depended cytotoxic effects of the oils of (A) P. heldreichii twigs, (B) P. heldreichii needles, (C) P. peuce twigs, (D) P. peuce needles, (E) P. mugo twigs, (F) P. mugo needles and (G) P. mugo cones on the three cancer cell lines HeLa, CaCo-2 and MCF-7, using an MTT-assay.
EOs obtained from P. peuce needles and from P. mugo needles and twigs led to a reduction of metabolic activity of CaCo-2 by 80–90%, followed by the oils of P. heldreichii twigs and oils of P. mugo cones with about 50–65%. IC50 values ranged from 0.02 for the oils of the needles of P. heldreichii, P. peuce and P. mugo to 0.04 for P. peuce twigs oils and 0.06 for P. mugo twigs oils. EOs of P. heldreichii twigs and P. mugo cones showed the highest IC50 with >0.1 (Table 2).
EOs of P. peuce twigs, P. peuce needles and P. mugo needles decreased cell viability of MCF-7 by at least 85%. EOs of P. heldreichii twigs, P. heldreichii needles and P. mugo twigs reduced metabolic activity of MCF-7 by 45–60%, while the EOs of P. mugo cones showed least cytotoxic effect to MCF-7 with only 15%. IC50 values ranged from 0.06 for P. peuce (needles and twigs) oils to >0.1 for P. heldreichii (needles and twigs) and P. mugo (twigs and cones) oils. Essential oil of P. mugo needles showed the highest IC50 with 0.3 (Table 2).
Discussion
The EOs of the needles and twigs of P. mugo, P. peuce and P. heldreichii exert significant anti-inflammatory activity. In fact, the EOs from the twigs of all Pinus spp. showed a higher anti-inflammatory activity than their needles or cones.
α-Pinene, a major compound in all Pinus spp. may have an impact on the secretion profile of cytokines and thus, may play an important role in the anti-inflammatory activity. According to previous studies, the EOs of Abies koreana, which is rich in bornyl acetate, limonene and α-pinene, inhibited the IL-1β, IL-6 and TNF-α production in LPS-stimulated macrophages (Yoon et al. 2009). Furthermore, the EOs of Olea europaea L. (Oleaceae), with α-pinene as main constituent, exhibited significant anti-inflammatory activity, assessed by the inhibition of paw oedema (Haloui et al. 2010). Hippeli et al. (2004) hypotize, that the, up to now, unknown anti-inflammatory potential of P. mugo EOs and its main compounds α-pinene, limonene and δ-3-carene, is generated by the down-regulation of increased activity of neutrophils, thus reducing the inflammatory reaction in the body. They also assume that hydrogen peroxide, which was found in aged P. mugo oils, maintains the accumulation of microbicide agents. Further in vitro studies might confirm this hypothesis.
To date, the cytotoxic activity of the EOs from P. heldreichii, P. mugo and P. peuce on the cancer cell lines HeLa, MCF-7 and CaCo-2 has not been studied. EOs of P. mugo needles exerted the highest activity on the three cancer cell lines, while EOs of P. mugo cones exerted the least effect. According to the American National Center Institute, plant extracts that show cytotoxicity with an IC50 < 30 μg/mL can be considered as potential agents for the development of anticancer drugs (Suffness & Pezzuto 1990). Thus, on HeLa cell lines, EOs of P. peuce needles, P. peuce twigs and P. heldreichii needles with IC50 lower than 0.03% can be considered as promising agents for anticancer drug development while on CaCo-2 cell lines, the EOs from P. mugo, P. peuce and P. heldreichii needles can be considered as promising agents. The cytotoxicity of these EOs may be attributed to the major components, such as phellandrene, α-pinene, β-pinene, germacrene D and α-terpinol. These constituents are similar to the main compounds of Myristica fragrans Houtt. (Myristicaceae), which showed cytotoxic activities on CaCo-2 cell lines in a previous study (Piras et al. 2012). On MCF-7 cell lines, the highest activity was exerted by the EOs of P. mugo needles, followed by P. peuce needles and twigs. This effect can be attributed to the high amount of α-pinene in the oils, which showed anti-proliferative activities on MCF-7 cell lines in a previous study (Cole et al. 2007).
Even though the EOs obtained from twigs and needles of P. heldreichii were rich in limonene, with higher concentration in P. heldreichii twigs, the better results were achieved from P. heldreichii needles. Thus, limonene was not the main responsible compound for the activity towards HeLa, MCF-7 and Caco-2 cell lines. These results indicate that the higher activity of P. heldreichii needles are related to other main components such as germacrene D and β-pinene. Similar results were earlier reported, showing little cytotoxic activity of EOs rich in limonene (Bakarnga-Via et al. 2014). This is in contrary to other studies, showing good cytotoxic activity of limonene (Miller et al. 2013). These finding suggest that the cytotoxic activity of the EOs are not always related to the main components only, indicating that the activity is related to the synergism or cumulative effect of different EO components, including the components in trace (Bakkali et al. 2008).
Moreover, the chemical composition of the EOs from P. mugo needles, twigs and cones showed differences to each other. The major compounds of the essential oil in the needles are α-pinene and δ-3-carene (21.3% and 19.3%), which is similar to the P. mugo essential oil compositions reported by Hajdari et al. (2015) [α-pinene (17.0-24.5%), followed by δ-3-carene (15.5-27.9%), germacrene D (4.0-9.9%) and (E)-caryophyllene (4.3-9.0%)] and Stevanovic et al. (2005) [δ-3-carene (23.9%), α-pinene (17.9%)]. The major compounds on twigs EOs were δ-3-carene (28.1%), limonene (19.5%) and β-pinene (11.0%), similar to essential oil compositions reported by Hajdari et al. (2015) [3-δ-carene (24.0-51.7%) followed by limonene + β-phellandrene (12.7-24.3%), (E)-caryophyllene (4.0-10.9%), β-pinene (2.2-15.4%) and α-pinene (4.5-8.8%)], while in cone EOs the major compounds were (E)-caryophyllene (20.2%) and δ-3-carene (15.8%). Similar chemical composition was previously reported by Hajdari et al. (2015) [δ-3-carene (10.5-31.5%), followed by (E)-caryophyllene (10.4-27.0%)]. These essential oil compositions are very different to previous studies, which reported α-pinene (33% from P. mugo in Scotland) and bornyl acetate (11.5% from P. mugo in Italy) as main components, (Tsitsimpikou et al. 2001; Venditti et al. 2013). The different origins of the collected plants might explain these disagreements.
Limonene is the major compound in both, needle and twig EOs from P. heldreichii, with higher amount present in the twigs, followed by α-pinene and germacrene D, which is confirming to the results by Nikolić et al. (2007) who identified the same most abundant compounds in needle EOs in populations of Mts. Lovcen, Zeletin and Bjelasica; Montenegro, and Mt. Zlatibor to Mt. Pester; Serbia. There is a partial divergence to the dominant constituents (germacrene D > limonene > α-pinene) found by Nikolić et al. (2015) in needle EOs in populations of Mts. Ošljak and Galičica; Macedonia. These results are also similar to the investigation of EOs of needles, twigs and twigs with needles of P. heldreichii populations from Mt. Prenj (Bosnia and Herzigovina) by Chalchat et al. (1994) and to the research about the EOs of needles of P. heldreichii populations from two different locations in Serbia by Simić et al. (1996), which also show the highest amounts in limonene and germacrene D.
The dominant compounds in the EOs of the needles from P. peuce was found to be α-pinene, followed by β-pinene, germacrene D and α-terpinol, which is similar to the study of Nikolić et al. (2008) [α-pinene > germacrene D > camphene > β-pinene in populations of Mts. Zeletin and Sjekirica; Montenegro, and Mt. Mokra Gora; Serbia], Nikolić et al. (2014) [α-pinene > germacrene D > β-pinene > camphene in populations of Mts. Ošljak and Pelister; Macedonia], Karapandzova et al. 2010, 2011) [α-pinene > germacrene D > bornyl acetate > β-pinene in populations of Mt. Pelister; Macedonia; α-pinene > germacrene D > β-pinene > bornyl acetate in populations of Mts. Nidze and Shara; Macedonia; α-pinene > germacrene D > β-pinene > (E)-caryophyllene in populations of Mt. Karadzica; Macedonia] and Koukos et al. (2000) [α-pinene > β-pinene > citronellol in populations of Rhodope Mountains; northern Greece]. α-Pinene was identified as the main compound in the needles EOs of P. peuce collected from the Sar Planina mountain in Serbia, followed by γ-muurlene, δ-3-carene, sabinene, bornyl acetate and limonene (Simić et al. 1995). In fact, Simić et al. (1995) used a SE-30 capillary column, which could partially explain the disagreements in the main compounds (high amounts of γ-muurlene, δ-3-carene and sabinene) compared to the presented results and cited findings. In the twigs EOs of P. peuce the major compounds were found to be β-phellandrene followed by β-pinene and α-pinene which is similar to the study by Koukos et al. (2000) [β-phellandrene > citronellol > β-pinene > α-pinene in populations of Rhodope Mountains; northern Greece].
All these differences in the cytotoxic effects and anti-inflammatory activity are related to the factors influencing the chemical composition of the EOs, reported by Hussain et al. (2008) and Raut and Karuppayil (2014). Thus, these factors should be considered for characterization of the biological properties of plants harvested in different countries.
Conclusions
The major compounds of the EOs in the needles were α-pinene and δ-3-carene, in the twigs the main compounds were δ-3-carene, limonene and β-pinene, while in cone EOs the major components were (E)-caryophyllene and δ-3-carene. The EOs of the twigs of all three Pinus spp. and the cones of P. mugo showed the most significant anti-inflammatory activity in LPS-stimulated macrophages. The EOs of the needles and twigs from P. peuce and P. mugo, as well as the needles of P. heldreichii, exerted the strongest cytotoxic activity on HeLa and CaCo-2 cell lines. On MCF-7 cell lines, the highest activity was shown by P. peuce (twigs and needles) and P. mugo needles. These EOs should be considered as potential agents for the development of new phytotherapeutic anticancer medicine.
Disclosure statement
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of this article.
References
- Adams RP.1997. Identification of essential oil components by gas chromatography/mass spectroscopy. J Am Soc Mass Spectrom. 8:671–672. [Google Scholar]
- Adorjan B, Buchbauer G.. 2010. Biological properties of essential oils: an updated review. Flavour Frag J. 25:407–426. [Google Scholar]
- Bakarnga-Via I, Hzounda JB, Fokou PV, Tchokouaha LR, Gary-Bobo M, Gallud A, Garcia M, Walbadet L, Secka Y, Dongmo PM.. 2014. Composition and cytotoxic activity of essential oils from Xylopia aethiopica (Dunal) A. Rich, Xylopia parviflora (A. Rich) Benth.) and Monodora myristica (Gaertn) growing in Chad and Cameroon. BMC Complement Altern Med. 14:125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bakkali F, Averbeck S, Averbeck D, Idaomar M.. 2008. Biological effects of essential oils-a review. Food Chem Toxicol. 46:446–475. [DOI] [PubMed] [Google Scholar]
- Bayala B, Bassole I, Scifo R, Gnoula C, Morel L, Lobaccaro JM, Simpore J.. 2014. Anticancer activity of essential oils and their chemical components – a review. Am J Cancer Res. 4:591. [PMC free article] [PubMed] [Google Scholar]
- Buchholz M, Gress TM.. 2009. Molecular changes in pancreatic cancer. Expert Rev Anticancer Ther. 9:1487–1497. [DOI] [PubMed] [Google Scholar]
- Chalchat J, Garry R, Gorunović MS.. 1994. Contributions à l'étude chimiotaxonomique des pins endémiques des Balkans: variation de la composition chimique de l'huile essentielle de Pinus heldreichii Christ., Pinacées différentes selon le site, la partie et l'âge des spécimens étudiés. Pharmazie. 49:852–854. [Google Scholar]
- Cole RA, Bansal A, Moriarity DM, Haber WA, Setzer WN.. 2007. Chemical composition and cytotoxic activity of the leaf essential oil of Eugenia zuchowskiae from Monteverde, Costa Rica. J Nat Med. 61:414–417. [Google Scholar]
- Croteau R.1986. Biochemistry of monoterpenes and sesquiterpenes of essential oils. J Herbs Spices Med Plants. 1:81–135. [Google Scholar]
- Efferth T, Fu YJ, Zu YG, Schwarz G, Konkimalla VS, Wink M.. 2007. Molecular target-guided tumor therapy with natural products derived from traditional Chinese medicine. Curr Med Chem. 14:2024–2032. [DOI] [PubMed] [Google Scholar]
- Hajdari A, Mustafa B, Ahmeti G, Pulaj B, Lukas B, Ibraliu A, Stefkov G, Quave CL, Novak J.. 2015. Essential oil composition variability among natural populations of Pinus mugo Turra in Kosovo. SpringerPlus. 4:1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haloui E, Marzouk Z, Marzouk B, Bouftira I, Bouraoui A, Fenina N.. 2010. Pharmacological activities and chemical composition of the Olea europaea L. leaf essential oils from Tunisia. J Food Agric Environ. 8:204–208. [Google Scholar]
- Hippeli S, Graßmann J, Vollmann R, Elstner EF.. 2004. Latschenkieferöl: Möglicher Wirkmechanismus für antientzündliche Wirkung. Pharm Zeitung. 149:35–36. [Google Scholar]
- Hippisley-Cox J, Coupland C.. 2005. Risk of myocardial infarction in patients taking cyclo-oxygenase-2 inhibitors or conventional non-steroidal anti-inflammatory drugs: population based nested case-control analysis. BMJ. 330:1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hussain AI, Anwar F, Sherazi ST, Przybylski R.. 2008. Chemical composition, antioxidant and antimicrobial activities of basil (Ocimum basilicum) essential oils depends on seasonal variations. Food Chem. 108:986–995. [DOI] [PubMed] [Google Scholar]
- Karapandzova M, Stefkov G, Kulevanova S.. 2010. Essential oils composition of Pinus peuce Griseb. (Pinaceae) growing on Pelister Mtn., Republic of Macedonia. Maced Pharm Bull. 56:13–22. [Google Scholar]
- Karapandzova M, Stefkov G, Trajkovska-Dokic E, Kaftandzieva A, Kulevanova S.. 2011. Antimicrobial activity of needle essential oil of Pinus puece Griseb. (Pinaceae) from Macedonian flora. Maced Pharm Bull. 57:25–36. [Google Scholar]
- Kim JY, Kim SS, Oh TH, Baik J, Song G, Lee N, Hyun CG.. 2009. Chemical composition, antioxidant, anti-elastase, and anti-inflammatory activities of Illicium anisatum essential oil. Acta Pharmaceut. 59:289–300. [DOI] [PubMed] [Google Scholar]
- Koukos PK, Papadopoulou KI, Patiaka DT, Papagiannopoulos AD.. 2000. Chemical composition of essential oils from needles and twigs of Balkan pine (Pinus peuce Grisebach) grown in Northern Greece. J Agric Food Chem. 48:1266–1268. [DOI] [PubMed] [Google Scholar]
- Lahlou M.2004. Essential oils and fragrance compounds: bioactivity and mechanisms of action. Flavour Frag J. 19:159–165. [Google Scholar]
- Legault J, Pichette A.. 2007. Potentiating effect of beta-caryophyllene on anticancer activity of alpha-humulene, isocaryophyllene and paclitaxel. J Pharm Pharmacol. 59:1643–1647. [DOI] [PubMed] [Google Scholar]
- Loizzo MR, Tundis R, Menichini F, Saab AM, Statti GA.. 2008. Antiproliferative effects of essential oils and their major constituents in human renal adenocarcinoma and amelanotic melanoma cells. Cell Prolif. 41:1002–1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medina-Holguín AL, Holguín FO, Micheletto S, Goehle S, Simon JA, O´Connell MA.. 2008. Chemotypic variation of essential oils in the medicinal plant, Anemopsis californica. Phytochemistry. 69:919–927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller JA, Lang JE, Ley M, Nagle R, Hsu CH, Thompson PA, Cordova C, Waer A, Chow HH.. 2013. Human breast tissue disposition and bioactivity of limonene in women with early-stage breast cancer. Cancer Prev Res (Phila). 6:577–584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mueller M, Hobiger S, Jungbauer A.. 2010. Anti-inflammatory activity of extracts from fruits, herbs and spices. Food Chem. 117:660–667. [Google Scholar]
- Nikolić B, Ristić M, Bojović S, Marin PD.. 2007. Variability of the needle essential oils of Pinus heldreichii from different populations in Montenegro and Serbia. Chem Biodivers. 4:905–916. [DOI] [PubMed] [Google Scholar]
- Nikolić B, Ristić M, Bojović S, Marin PD.. 2008. Variability of the needle essential oils of Pinus peuce from different populations in Montenegro and Serbia. Chem Biodivers. 5:1377–1388. [DOI] [PubMed] [Google Scholar]
- Nikolić B, Ristić M, Bojović S, Matevski V, Krivošej Z, Marin PD.. 2014. Essential oil composition of the needles collected from natural populations of Macedonian pine (Pinus peuce Griesb.) from the Scardo-Pindic mountain system. Chem Biodivers. 11:934–948. [DOI] [PubMed] [Google Scholar]
- Nikolić B, Ristić M, Bojović S, Krivošej Z, Matevski V, Marin PD.. 2015. Population variability of essential oils of Pinus heldreichii from the Scardo-Pindic mountains Ošljak and Galičica. Chem Biodivers. 12:295–308. [DOI] [PubMed] [Google Scholar]
- Perrone MR, Artesani MC, Viola M, Gaeta F, Caringi M, Quaratino D, Romano A.. 2003. Tolerability of rofecoxib in patients with adverse reactions to nonsteroidal anti-inflammatory drugs: a study of 216 patients and literature review. Int Arch Allergy Immunol. 132:82–86. [DOI] [PubMed] [Google Scholar]
- Piras A, Rosa A, Marongiu B, Atzeri A, Dessi MA, Falconieri D, Porcedda S.. 2012. Extraction and separation of volatile and fixed oils from seeds of Myristica fragrans by supercritical CO2: chemical composition and cytotoxic activity on Caco-2 cancer cells. J Food Sci. 77:C448–C453. [DOI] [PubMed] [Google Scholar]
- Raut JS, Karuppayil SM.. 2014. A status review on the medicinal properties of essential oils. Ind Crops Prod. 62:250–264. [Google Scholar]
- Sharma PR, Mondhe DM, Muthiah S, Pal HC, Shahi AK, Saxena AK, Qazi GN.. 2009. Anticancer activity of an essential oil from Cymbopogon flexuosus. Chem Biol Interact. 179:160–168. [DOI] [PubMed] [Google Scholar]
- Simić N, Palić R, Andjelković S, Kitić D, Gasić MJ.. 1995. Essential oil of Pinus peuce needles. FU Phys Chem Tech. 1:188–192. [Google Scholar]
- Simić N, Palić R, Andjelković S, Vajs V, Milosavljević S.. 1996. Essential oil of Pinus heldreichii needles. J Essent Oil Res. 8:1–5. [Google Scholar]
- Stevanović-Janežić TM, Vilotić DM.. 1998. Etarska ulja iz endemskih borova munike (Pinus heldreichii Christ.) i molike (Pinus peuce Griseb.). Lekovite Sirovine XLVII, Beograd. 47:109–114. [Google Scholar]
- Stevanovic T, Garneau FX, Jean FI, Gagnon HI, Vilotic D, Petrovic S, Ruzic N, Pichette A.. 2005. The essential oil composition of Pinus mugo Turra from Serbia. Flavour Frag J. 20:96–97. [Google Scholar]
- Suffness M, Pezzuto JM.. 1990. Assays related to cancer drug discovery. Meth Plant Biochem. 6:71–133. [Google Scholar]
- Tsitsimpikou C, Petrakis PV, Ortiz A, Harvala C, Roussis V.. 2001. Volatile needle terpenoids of six Pinus species. J Essent Oil Res. 13:174–178. [Google Scholar]
- Venditti A, Serrilli AM, Vittori S, Papa F, Maggi F, Di Cecco M, Ciaschetti G, Bruno M, Rosselli S, Bianco A.. 2013. Secondary metabolites from Pinus mugo Turra subsp. mugo growing in the Majella National Park (Central Apennines, Italy). Chem Biodiv. 10:2091–2100. [DOI] [PubMed] [Google Scholar]
- Yoon WJ, Kim SS, Oh TH, Lee NH, Hyun CG.. 2009. Abies koreana essential oil inhibits drug-resistant skin pathogen growth and LPS-induced inflammatory effects of murine macrophage. Lipids. 44:471–476. [DOI] [PubMed] [Google Scholar]