Abstract
Aloperine (Alo), as a quinolizidine alkaloid extracted from S. alopecuroide, has the positive activities of anti-inflammatory, anti-allergenic, antitumor and anti-viral. However, the role and mechanism of Alo in breast cancer have not been studied yet. In the present study, Alo markedly inhibited the proliferation and suppressed the colony formation ability of the breast cancer cell lines MCF-7 and MDA-MB-231 in a dose-dependent manner by Cell Counting kit-8 and colony formation assays, respectively. In addition, the results of confocal microscopy analysis and flow cytometry detection revealed that Alo induced the apoptosis of MCF-7 and MDA-MB-231 cells, and western blotting indicated that Alo upregulated the protein levels of Bax, caspase-3 and caspase-9, and downregulated the expression of Bcl-2. Furthermore, the results of wound healing, Transwell migration and invasion assays demonstrated that Alo inhibited the migration and invasion of MCF-7 and MDA-MB-231 cells, and reduced the protein levels of matrix metalloproteinase (MMP)-2 and MMP-9. Alo also downregulated the protein expressions of Ras, phosphorylated (p)-Raf proto-oncogene, serine/threonine kinase 1 and p-extracellular signal-regulated kinase 1/2. Furthermore, ISIS 2503, a Ras inhibitor, inhibited colony formation, induced apoptosis, and suppressed the migration and invasion of MCF-7 and MDA-MB-231 cells. These effects were more marked in the presence of ISIS 2503 and Alo, when compared with those of either agent alone. In conclusion, the present study reported a novel use of Alo in inhibiting the proliferation, migration and invasion, and inducing the apoptosis of human breast cancer cells by blocking the Ras signaling pathway.
Keywords: aloperine, breast cancer, proliferation, apoptosis, migration, invasion, Ras
Introduction
Breast cancer is the most common malignant tumor and the leading cause of cancer mortality in women, resulting in 14% of cancer-related deaths (1,2). Besides, Breast cancer is a major malignant tumor threatening women's health in China (3). According to statistics, the new cases and dead cases of breast cancer account for 12.2 and 9.6% annually, respectively (4). Although the incidence of breast cancer in China is low, it is noteworthy that the incidence of breast cancer in China grows rapidly at 3% per year in recent years, which has become one of the leading causes of death in Chinese cities (5). However, the cause of breast cancer is not clear yet, and even about 50% of the causes with breast cancer can not be explained at all, which seriously threatens the life and health of women (6).
Along with the increasing study of molecular mechanisms of breast cancer, the level of comprehensive treatment for breast cancer has been greatly improved. In recent years, multiple treatments have been developed for breast cancer, such as local surgical treatment, radiotherapy and chemotherapy, endocrine therapy, molecular targeted treatment, auxiliary treatment of traditional Chinese medicine and so on, which significantly improves the quality of life and prolongs the survival period of patients (7–11). A large number of clinical data and experimental studies have shown that the adjuvant treatment of breast cancer patients with traditional Chinese medicine can improve the body's metabolism, reduce the toxic and side effects of radiotherapy and chemotherapy, improve the surgical resection rate and endocrine therapy efficacy, and so on (12,13). The development of traditional Chinese medicine plays an important role in reducing the recurrence and metastasis of tumor, prolonging the survival time, and improving the quality of patients' life (14). Moreover, traditional Chinese medicine still has many positive advantages of rich resources, relatively less cytotoxicity, easy access and low cost. Therefore, the study of the targets and mechanisms of traditional Chinese medicine in the comprehensive treatment for breast cancer has been an important subject in the study of breast cancer prevention and treatment.
Aloperine (Alo) (Fig. 1) is an alkaloid extracted from the traditional Chinese medicine Sophora alopecuroides (S. alopecuroides) (15,16). It has been reported that Alo induced apoptosis and inhibited invasion in MG-63 and U2OS human osteosarcoma cells, and induced G2/M phase cell cycle arrest and apoptosis in HCT116 human colon cancer cells (17,18). However, the impact of Alo on breast cancer has not been reported yet. Therefore, the article was designed to investigate the antitumor potential and the underlying molecular mechanisms of Alo on breast cancer.
Figure 1.
Structure of Aloperine.
Materials and methods
Cell culture
Human breast cancer cells MCF-7 and MDA-MB-231 were purchased from the American Type Culture Collection (Manassas, VA, USA). MCF-7 cells were cultured in Dulbecco's modified Eagle's medium (DMEM) (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal bovine serum (FBS; Tianhang, Hangzhou, China), 100 mg/ml streptomycin and 100 U/ml penicillin in 5% CO2 atmosphere at 37°C. MDA-MB-231 cells were cultured in L-15 medium (both Gibco; Thermo Fisher Scientific, Inc.) medium supplied with 10% FBS, 100 mg/ml streptomycin and 100 U/ml penicillin in normal air atmosphere at 37°C.
Cell counting kit-8 (CCK-8) assay
The viabilities of MCF-7 and MDA-MB-231 cells were measured by CCK-8 assay. Briefly, MCF-7 and MDA-MB-231 cells at a density of 1×104 cells/well in 100-µl of complete culture medium were seeded in 96-well plates. After culturing for 24 h, the medium was replaced with serum-free media or serum-free media containing various concentrations of Alo (0, 0.1, 0.2 and 0.4 mM) according to previous studies (17,18), and incubated in a humidified incubator at 37°C for 12, 24 and 48 h, respectively. After incubation, 10 µl of CCK-8 (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was added to each well for another 2 h at 37°C. The optical density (OD) was recorded at 450 nm using a microplate reader (Dojindo Molecular Technology, Rockville, MD, USA).
Colony formation test
Colony formation assay was conducted to evaluate the role of Alo in the proliferative potential of MCF-7 and MDA-MB-231 cells. MCF-7 and MDA-MB-231 cells (1×103 cells/well) were plated in 6-well plates and cultured at 37°C with 5% CO2. The medium was replaced with fresh culture media or fresh culture media containing Alo (0, 0.1, 0.2 and 0.4 mM) every 2–3 days. After two weeks, the plates were fixed with 4% paraformaldehyde for 20 min and stained using 10% crystal violet for 30 min. Then the number of stained colonies was manually counted.
Hoechst 33342 staining
MCF-7 and MDA-MB-231 cells at a density of 2×104 cells/well were seeded in 24-well plates, and after incubation for 24 h, fresh culture media or fresh culture media containing Alo (0, 0.1, 0.2 and 0.4 mM) were added, and incubated in a humidified incubator at 37°C for 24 h. Then the cells were stained with Hoechst 33342 (10 mg/ml) in culture medium at room temperature in the dark for 20 min. Subsequently, the cells were washed twice with PBS and immediately evaluated by a microscope.
Apoptosis analysis
Cell apoptosis was performed using Annexin V Apoptosis Detection kit I (BD Biosciences, Franklin Lakes, NJ, USA). Briefly, MCF-7 and MDA-MB-231 at a density of 2×104 cells were treated with indicated doses of Alo (0, 0.1, 0.2 and 0.4 mM) for 24 h. Then the treated cells were digested with trypsin and washed in cold 1X PBS (4°C) twice, followed by resuspending the cell pellet with 300 µl of 1X Binding Buffer. Next, 5 µl of Annexin V-PE were added to the cell suspension for 15 min in the dark at room temperature, according to the manufacturer's instructions. 7-AAD solution (5 µl) was added in the cell suspension 5 min before flow cytometry analysis and then 200 µl of 1X Binding Buffer was added for flow cytometry analysis. The percentage of apoptotic cells was evaluated by FACS Calibur (BD Biosciences).
Wound healing assay
MCF-7 and MDA-MB-231 cells were cultured in fresh culture media to full confluence. After that, we created a wound using a plastic scraper. After being washed with PBS, the medium was replaced with fresh culture media or fresh culture media containing Alo (0, 0.1, 0.2 and 0.4 mM) and incubated at 37°C for 48 h. Then, the cells were washed twice with PBS and the wound was observed under a microscope (Nikon, Tokyo, Japan).
Transwell migration and invasion assay
Cell migration and invasion assays were performed using Trans-well chambers (8 µm pore-size, Corning Co., Corning, NY, USA). In migration assay, 5×104 cells in fresh culture media were added into the upper chamber. In invasion assay, Matrigel was purchased from BD Biosciences and stored at −20°C. After thawing at 4°C overnight, the Matrigel was diluted in serum-free medium, and 30 µl of the diluted Matrigel were evenly inoculated into the upper chamber to form a gel at 37°C. Cells (1×105) suspended in 300 µl of fresh culture media were seeded into the upper compartments. For trans-well migration and invasion assay, the lower compartments were filled with 600 µl of medium with 20% FBS. After incubation for 24 h, the non-migrate or non-invasive cells were removed from the upper surface of the membrane by scrubbing. The cells that migrated or invaded to the lower surface of the membrane were fixed with 4% paraformaldehyde, stained in 10% crystal violet, and cells were counted under a microscope (Olympus, Tokyo, Japan).
Western blot analysis
The total protein of cells was extracted according to the manufacturer's recommended protocol (Vazyme, Piscataway, NJ, USA). The protein concentrations were determined using the BCA Protein Assay kit (Vazyme, Piscataway, NJ, USA). Samples with equal amounts of protein (50 µg) were fractionated on 10% SDS polyacrylamide gels, transferred to polyvinylidene difluoride membranes (PVDF), and blocked in 5% skim milk in TBST for 1.5 h at 25±1°C. The membranes were then incubated at 4°C overnight with 1:1,000 dilutions (v/v) of the primary antibodies. After washing the membranes with TBST, incubations with 1:1,000 dilutions (v/v) of the secondary antibodies were conducted for 2 h at 25±1°C. Protein expression was detected using an Enhanced Chemiluminescence Detection System. GAPDH was used as a loading control. Antibodies in western blot were purchased Cell Signaling Technology (Beverly, MA, USA), including matrix metalloproteinase (MMP)-2 (cat. no. 4022), MMP-9 (cat. no. 3852), caspase-3 (cat. no. 9662), caspase-9 (cat. no. 9508), Bax (cat. no. 2774), Bcl-2 (cat. no. 2872), Ras (cat. no. 3965), p-Raf1 (cat. no. 9421), Raf1 (cat. no. 4432), p-Erk1/2 (cat. no. 4370), Erk1/2 (cat. no. 9102), GAPDH (cat. no. 8884).
Statistical analyses
GraphPad Prism 5.0 statistical software (GraphPad Software, Inc., La Jolla, CA, USA) was utilized to analyze the above experimental data. Measurement data were represented as the mean ± standard deviation. Statistical differences between means among multiple groups were analyzed by one-way analysis of variance followed by a Bonferroni post hoc analysis. P<0.05 was considered to indicate a statistically significant difference.
Results
Alo inhibited human breast cancer cells proliferation and colony formation
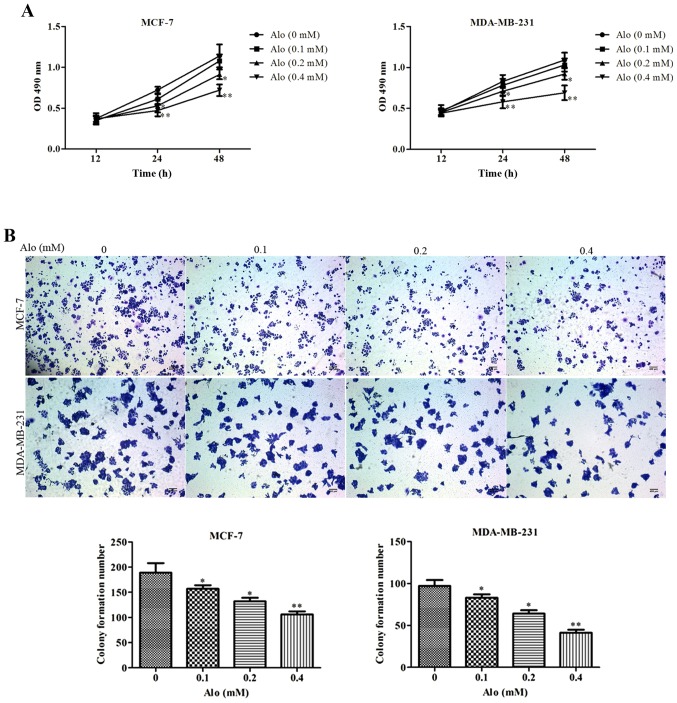
To investigate the impact of Alo on human breast cancer MCF-7 and MDA-MB-231 cells proliferation, MCF-7 and MDA-MB-231 cells were treated with Alo (0.1, 0.2 and 0.4 mM) for 12, 24 and 48 h, respectively. The results of CCK-8 assay showed that Alo suppressed cell proliferation in a dose and time dependent manner (Fig. 2A). Subsequently, to evaluate whether Alo regulated the colony growth of human breast cancer cells, MCF-7 and MDA-MB-231 cells were treated with Alo (0.1, 0.2 and 0.4 mM), and incubated for two weeks. The results revealed that Alo reduced colony formation of MCF-7 and MDA-MB-231 cells compared with the untreated group (Fig. 2B).
Figure 2.
Alo inhibits breast cancer cell proliferation and colony formation. (A) MCF-7 and MDA-MB-231 cell proliferation was evaluated at the indicated time points following treatment with Alo (0.1, 0.2 and 0.4 mM). (B) MCF-7 and MDA-MB-231 cell colony formation was detected following cultured with Alo (0.1, 0.2 and 0.4 mM) for 14 days (scale bars, 400 µm). The results were expressed as the mean ± standard deviation of three independent experiments and each was performed in triplicate. *P<0.05 and **P<0.01 vs. non-Alo treated group (0 mM Alo). Alo, Aloperine; OD, optical density.
Alo induced human breast cancer cells apoptosis
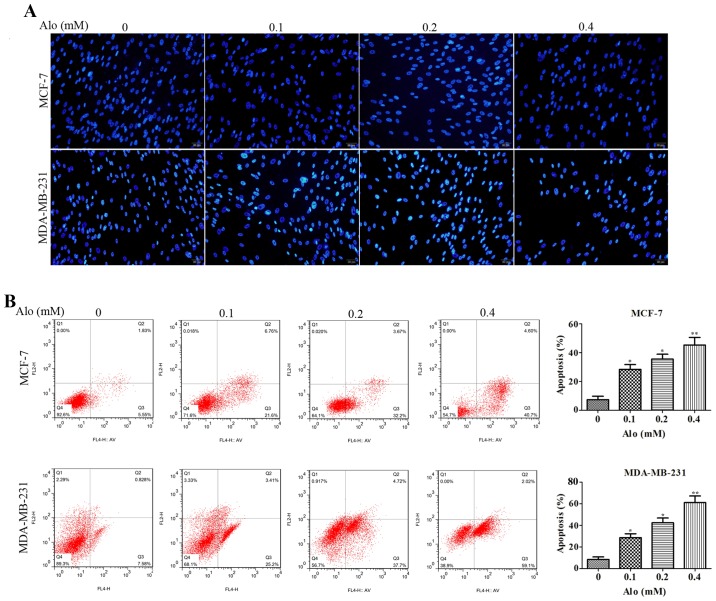
In order to determine whether Alo induced human breast cancer MCF-7 and MDA-MB-231 cells apoptosis, first Hoechst 33342 staining was adapted to evaluate cell apoptosis by morphological examination. The results demonstrated the nuclei of MCF-7 and MDA-MB-231 cells were uniformly stained in the untreated group, indicating these cells had intact cell membrane morphology. However, both MCF-7 and MDA-MB-231 cells treated with Alo (0.1, 0.2 and 0.4 mM) for 24 h clearly exhibited significant morphological changes compared with the untreated group, showing that MCF-7 and MDA-MB-231 cells apoptosis remarkably increased (Fig. 3A). Additionally, the effect of Alo on MCF-7 and MDA-MB-231 cells apoptosis was evaluated by flow cytometry with Annexin V-FITC/PI staining. After treatment of MCF-7 and MDA-MB-231 cells with Alo (0.1, 0.2 and 0.4 mM), we found degree of apoptosis in the Alo group was higher than that in the untreated group (Fig. 3B). These findings suggested that Alo dramatically abated MCF-7 and MDA-MB-231 cells proliferation rate in a dose dependent manner, which was possibly associated with increasing apoptosis.
Figure 3.
Alo induces breast cancer cell apoptosis. (A) Images of apoptotic MCF-7 and MDA-MB-231 cells incubated with vehicle or Alo (0.1, 0.2 and 0.4 mM) for 24 h, as determined by Hoechst 33342 staining (scale bars, 50 µm). (B) MCF-7 and MDA-MB-231 cells exposed to Alo (0.1, 0.2 and 0.4 mM) for 24 h were harvested and analyzed by flow cytometry. The results were expressed as the mean ± standard deviation of three independent experiments and each was performed in triplicate. *P<0.05 and **P<0.01 vs. non-Alo treated group (0 mM Alo). Alo, Aloperine.
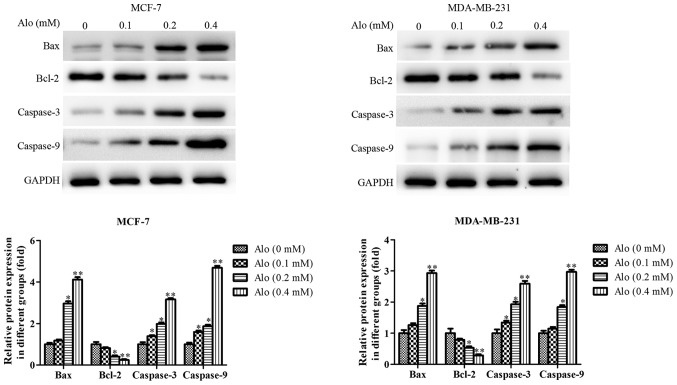
Furthermore, the expressions of apoptosis-related proteins, including caspase-3, caspase-9, Bax and Bcl-2, were detected by western blotting analysis. The results showed that Alo could downregulate the expression of Bcl-2 and upregulate the expressions of caspase-3, caspase-9 and Bax compared with the untreated group in MCF-7 and MDA-MB-231 cells (Fig. 4).
Figure 4.
Alo induces breast cancer cell apoptosis by regulating apoptosis-associated protein expression. Western blotting was performed to analyze the expressions of Bax, Bcl-2, caspase-3 and caspase-9 following Alo (0.1, 0.2 and 0.4 mM) treatment for 24 h. Then the band intensity was quantified by ImageJ software. The results were expressed as the mean ± standard deviation of three independent experiments. *P<0.05 and **P<0.01 vs. non-Alo treated group (0 mM Alo). Alo, Aloperine; Bcl, B-cell lymphoma 2; Bax, Bcl-2-associated X protein.
Alo suppressed human breast cancer cells migration and invasion
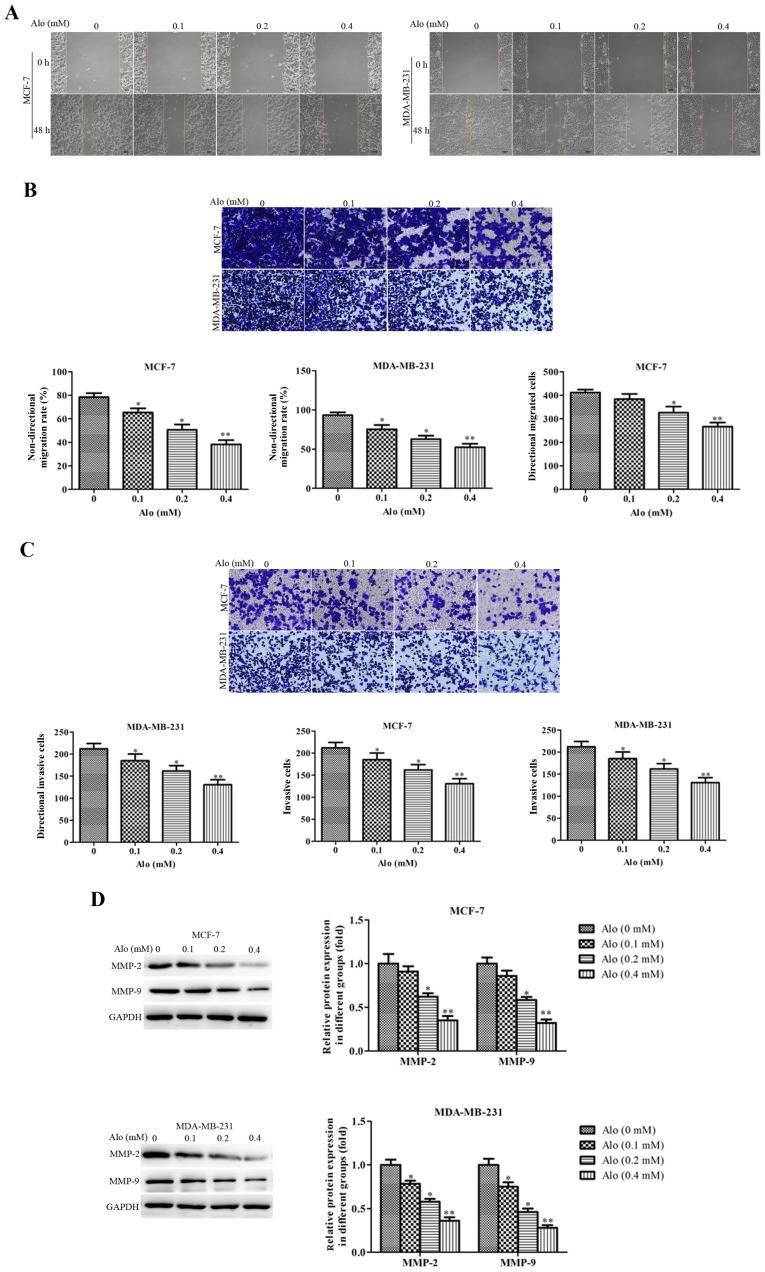
To further confirm the effects of Alo on cell motility, wound healing, trans-well migration and invasion assays were conducted. The results of wound healing and Trans-well migration assays revealed that Alo could significantly suppress MCF-7 cells migration and also remarkably inhibit MDA-MB-231 cells migration in a dose dependent manner compared with the untreated group (Fig. 5A and B). Besides, a similar results in cell invasion was observed. Alo treatment in MCF-7 and MDA-MB-231 cells led to a decreased percent of invasion cells compared with the untreated group (Fig. 5C). These data indicated Alo dramatically depressed cell migration and invasion in MCF-7 and MDA-MB-231 cells.
Figure 5.
Alo suppresses human breast cancer cell migration and invasion. (A) MCF-7 and MDA-MB-231 cell non-directional migration was detected by wound healing assay (scale bars, 200 µm; magnification, ×100). (B) MCF-7 and MDA-MB-231 cell directional migration was evaluated by Transwell assay (scale bars, 200 µm; magnification, ×200). (C) MCF-7 and MDA-MB-231 cell invasion was examined by Transwell invasion assay (scale bars, 200 µm; magnification, ×200). (D) Western blotting analyzed the expression of MMP-2 and MMP-9 following Alo (0.1, 0.2 and 0.4 mM) treatment for 24 h. Then the band intensity was quantified by ImageJ software. The results were expressed as the mean ± standard deviation of three independent experiments and each was performed in triplicate. *P<0.05 and **P<0.01 vs. non-Alo treated group (0 mM Alo). Alo, Aloperine; MMP, matrix metalloproteinase.
MMPs are zinc-dependent proteolytic enzymes of the extracellular matrix (ECM), widely involved in cell migration and invasion (19,20). And as the members of MMPs, MMP-2 and MMP-9 are closely related to the migration and invasion of various types of cancer cells (21,22). Thus, the protein expressions of MMP-2 and MMP-9 were detected to evaluate the effect of Alo on the motility of MCF-7 and MDA-MB-231 cells. It showed that Alo significantly suppressed the protein expressions of MMP-2 and MMP-9 (Fig. 5D).
Alo blocked Ras in human breast cancer cells
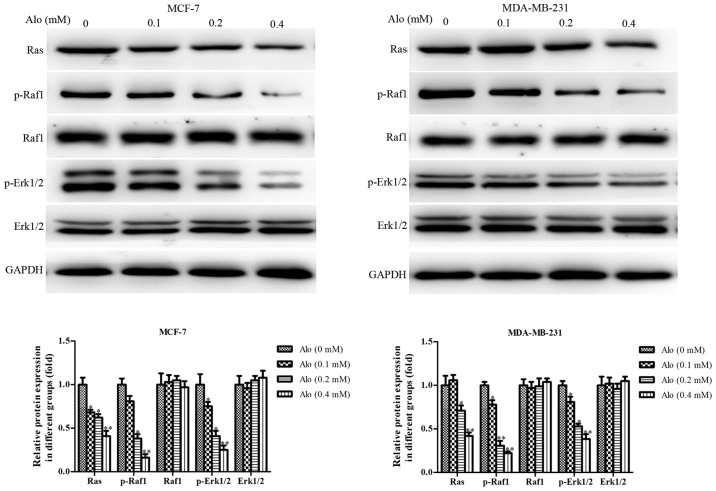
Ras family is usually recognized as an oncogene, including H-Ras, Ha-Ras, K-Ras, Ki-Ras and N-Ras, and Ras/Raf1/ERK1/2 pathway is considered the be the downstream of epidermal growth factor receptor (EGFR), which is closely associated to the incidence and prognosis of various cancers (23). Activated ERK1/2 regulates gene expression and ultimately mediates cell growth, differentiation, migration, invasion and other processes through phosphorylation on transcription factors in the cell cytoplasm and nucleus (24,25). Thus, in this study, we observed that, after treatment of human breast cancer cells MCF-7 and MDA-MB-231 with Alo (0.1, 0.2 and 0.4 mM), the level of Ras was downregulated, and the levels of phosphorylation of Raf1 and Erk1/2 were decreased as well (Fig. 6), which indicated that Alo could block the Ras/Raf1/Erk1/2 signaling pathway.
Figure 6.
Alo blocks the Ras/Raf1/Erk1/2 signaling pathway in breast cancer cells. Western blotting was performed to analyze the expression of Ras, p-Raf1, Raf1, p-Erk1/2 and Erk1/2 following Alo (0.1, 0.2 and 0.4 mM) treatment for 24 h. Then the band intensity was quantified by ImageJ software. The results were expressed as the mean ± standard deviation of three independent experiments. *P<0.05 and **P<0.01 vs. non-Alo treated group (0 mM Alo). Alo, Aloperine; Raf, Raf proto-oncogene, serine/threonine kinase; Erk1/2, extracellular signal-regulated kinase 1/2; p-, phosphorylated.
Alo inhibited proliferation, migration and invasion and induced apoptosis via Ras pathway in human breast cancer cells
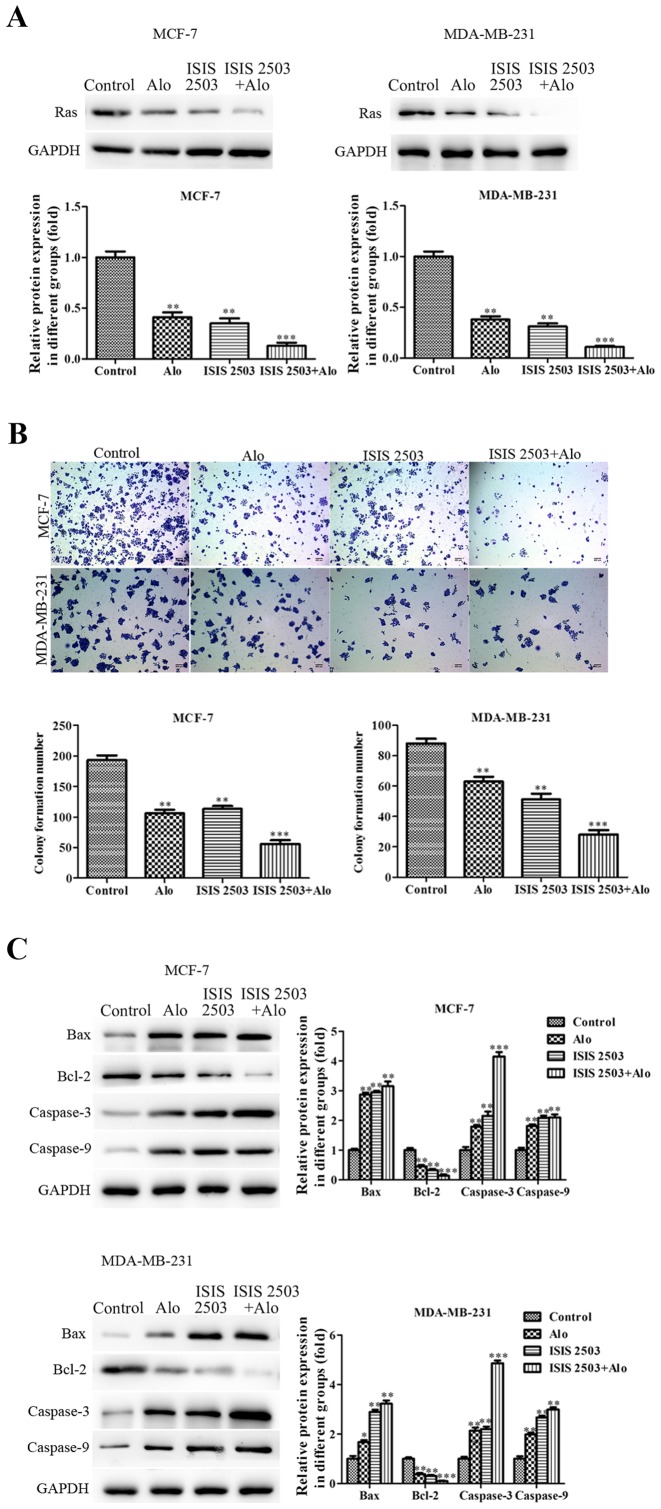
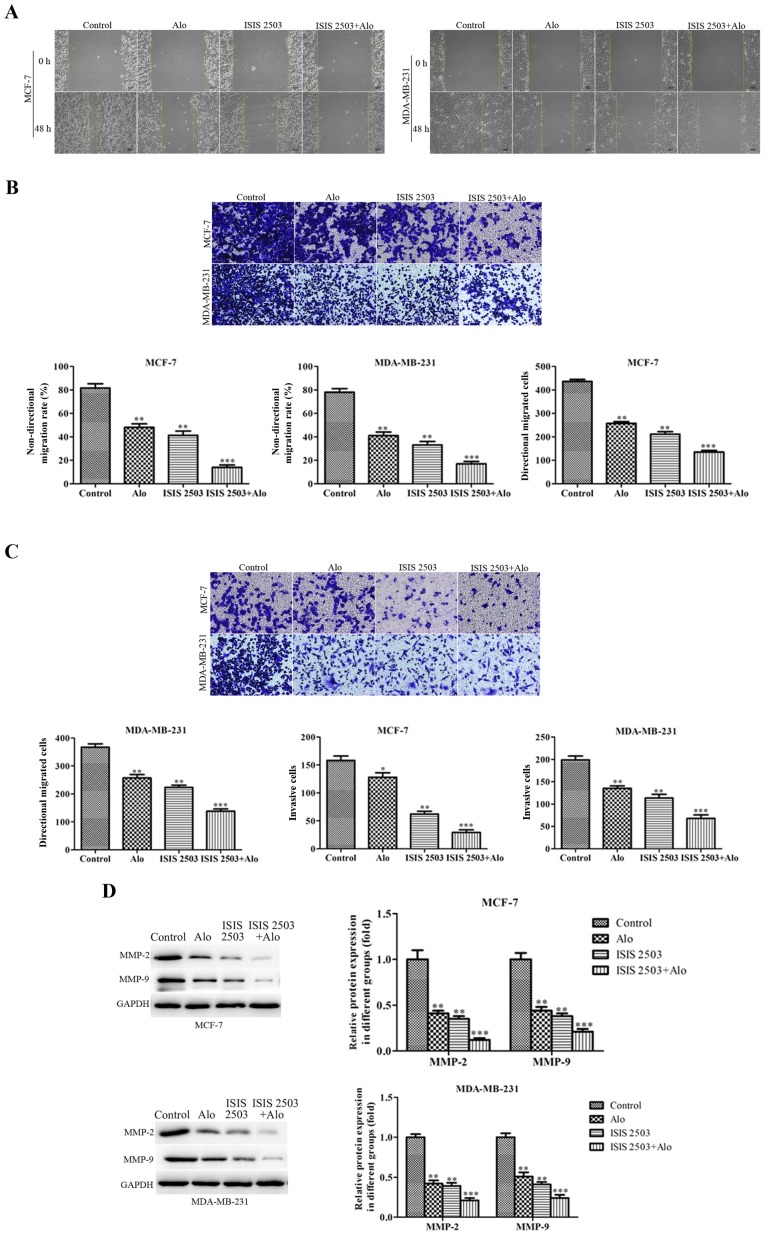
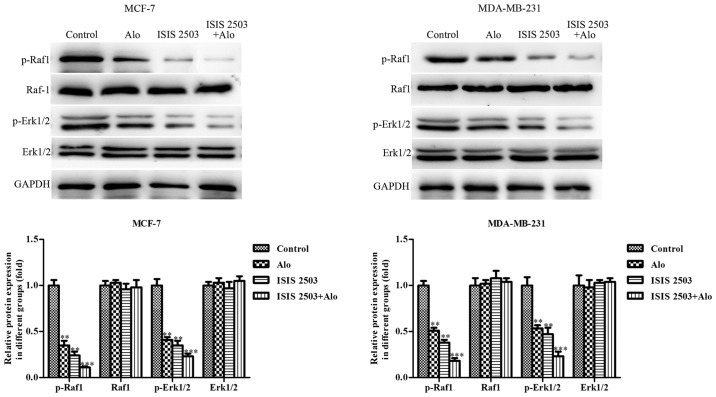
After demonstrating decreased level of Ras following incubation of MCF-7 and MDA-MB-231 cells with Alo (0.1, 0.2 and 0.4 mM), further, we evaluated whether activation of these kinases contributed to the progress of MCF-7 and MDA-MB-231 cells. To further verify the involvement of Ras signaling in inhibiting proliferation, migration and invasion and inducing apoptosis of human breast cancer cells, ISIS 2503, a novel Ras inhibitor with selective activity against H-Ras, was employed to inhibit the Ras expression (26,27). First, we found both Alo (0.4 mM) and ISIS 2503 inhibited the expression of Ras, and co-treatment of Alo and ISIS 2503 inhibited the level of Ras more than either one of them in MCF-7 and MDA-MB-231 cells (Fig. 7A). Then the results demonstrated that Alo (0.4 mM) and ISIS 2503 reduced colony formation (Fig. 7B), and could apparently downregulate the expression of Bcl-2 and upregulate the expressions of caspase-3, caspase-9 and Bax (Fig. 7C). However, co-treatment of Alo (0.4 mM) and ISIS 2503 had better inhibitory effect on the cell colony formation and apoptosis compared with that of either agent alone in MCF-7 and MDA-MB-231 cells. Moreover, we adapted the wound healing (Fig. 8A), trans-well migration (Fig. 8B) and invasion (Fig. 8C) assays to evaluate the impact of ISIS 2503 on cell motility, and the results demonstrated co-treatment of Alo (0.4 mM) and ISIS 2503 remarkably inhibited migration and invasion more than either one of them in MCF-7 and MDA-MB-231 cells. In addition, co-treatment of Alo (0.4 mM) and ISIS 2503 showed more positive activity of inhibition of MMP-2 and MMP-9 levels (Fig. 8D). Furthermore, we found co-treatment of Alo (0.4 mM) and ISIS 2503 had better effects on the decrease of the phosphorylation of Raf1 and Erk1/2 (Fig. 9). These findings suggested that Alo inhibited proliferation, migration and invasion and induced apoptosis maybe through Ras pathway in human breast cancer cells.
Figure 7.
Alo inhibits proliferation and induces apoptosis via the Ras signaling pathway in breast cancer cells. (A) Alo and the Ras inhibitor ISIS 2503 inhibited the expression of Ras, as determined by western blotting analysis. Then the band intensity was quantified by ImageJ software. (B) Images of colony formation of MCF-7 and MDA-MB-231 cells cultured with Alo (0.4 mM) or Ras inhibitor (ISIS 2503) for 14 days (scale bars, 400 µm). (C) Western blotting analyzed the expressions of Bax, Bcl-2, caspase-3 and caspase-9 following Alo (0.4 mM) or Ras inhibitor (ISIS 2503) treatment for 24 h. Then the band intensity was quantified by ImageJ software. The results were expressed as the mean ± standard deviation of three independent experiments and each was performed in triplicate. *P<0.05, **P<0.01 and ***P<0.001 vs. control group. Alo, Aloperine; Bcl, B-cell lymphoma 2; Bax, Bcl-2-associated X protein.
Figure 8.
Alo inhibits migration and invasion via the Ras signaling pathway in breast cancer cells. (A) MCF-7 and MDA-MB-231 cell non-directional migration was detected by wound healing assay (scale bars, 200 µm; magnification, ×100). (B) MCF-7 and MDA-MB-231 cell directional migration was evaluated by Transwell assay (scale bars, 200 µm; magnification, ×200). (C) MCF-7 and MDA-MB-231 cell invasion was examined by Transwell invasion assay (scale bars, 200 µm; magnification, ×200). (D) Western blotting analyzed the expressions of MMP-2 and MMP-9 following Alo (0.4 mM) or Ras inhibitor (ISIS 2503) treatment for 24 h. Then the band intensity was quantified by ImageJ software. The results were expressed as the mean ± standard deviation of three independent experiments and each was performed in triplicate. *P<0.05, **P<0.01 and ***P<0.001 vs. control group. Alo, Aloperine; MMP, matrix metalloproteinase.
Figure 9.
Synergistic effect of Alo and Ras inhibitor (ISIS 2503) on the blockage of the Ras/Raf1/Erk1/2 signaling pathway in breast cancer cells. Western blotting analyzed the expressions of p-Raf1, Raf1, p-Erk1/2 and Erk1/2 following Alo (0.4 mM) or Ras inhibitor (ISIS 2503) treatment for 24 h. Then the band intensity was quantified by ImageJ software. The results were expressed as the mean ± standard deviation of three independent experiments. **P<0.01 and ***P<0.001 vs. control group. Alo, Aloperine; Raf, Raf proto-oncogene, serine/threonine kinase; Erk1/2, extracellular signal-regulated kinase 1/2; p-, phosphorylated.
Discussion
Breast cancer is one of the most common malignant tumors that leads to the death of women, and hematogenous spread occurs easily in the early stage, which threatens the heath of women seriously. At present, the treatments of breast cancer include surgery, chemotherapy, radiotherapy, targeted biotherapy and so on, which is certain to have to cancer cell kill and wound action. However, there are still 25–30% of early breast cancer patients with distant metastasis after 10–15 years of follow-up (28). At the same time, the drug resistance of tumor cells and the side effects of chemotherapeutic drugs are also another difficult problem in the process of cancer treatment. Therefore, the natural compounds from animal and plant have become the main research direction of anticancer drugs (29). Currently, traditional Chinese medicine is still the mainstream of the prevention and treatment of tumor, and the characteristics of multi-component, multi-link and multi-target play a significant role in polygene regulation, complicated pathogenesis, and prevention and treatment of various tumors (30). Moreover, traditional Chinese medicine has been paid more attention because of its unique antitumor effect, sensitivity of radiotherapy and chemotherapy, little toxic and side effects and prolonged the survival time of patients (31). Therefore, this article was designed to explore the effects and mechanisms of Chinese medicine monomer Alo on proliferation, apoptosis, migration and invasion of breast cancer cells. In this study, we found Alo inhibited MCF-7 and MDA-MB-231 cells proliferation and colony formation in a dose dependent manner, changed cell membrane morphology and promoted the apoptosis in MCF-7 and MDA-MB-231 cells. Bcl-2 family includes pro-apoptotic proteins and anti-apoptotic proteins. A pro-apoptotic bcl-2 family protein, Bax, promotes cell apoptosis by activation of caspase and the release of cytochrome c from mitochondria, while Bcl-2, an anti-apoptotic bcl-2 family protein, restrains cell apoptosis via blocking the release of cytochrome c (32,33). Our date showed that Alo could dramatically downregulate the expression of Bcl-2 and upregulate the expression of caspase-3, caspase-9 and Bax in MCF-7 and MDA-MB-231 cells. These results were similar to previous studies (17,18), which further confirmed that Alo had positive inhibitory activity on the progression and development of many types of cancer.
The main cause of death in patients with breast cancer is the invasion and metastasis of the tumor (34). Wound healing, trans-well migration and invasion assays were conducted to further confirm the effects of Alo on cell motility, and the results revealed that Alo could significantly suppress MCF-7 and MDA-MB-231 cells migration and invasion. In the process of tumor invasion and metastasis, the degradation of ECM plays an important role. Moreover, the degradation of ECM mainly depends on proteolytic enzymes, and the most important is MMPs, which are highly conserved zinc dependent endonuclease protease family with the positive activities on degrading most proteins of basement membrane and ECM (35). Furthermore, MMP-2 and MMP-9 have been strongly correlated with the invasiveness of many types of cancer cell (36). Thus, the protein expressions of MMP-2 and MMP-9 were detected to evaluate the effect of Alo on the motility of MCF-7 and MDA-MB-231 cells and we found that Alo significantly suppressed the protein expressions of MMP-2 and MMP-9.
At present, it is believed that the occurrence of human tumor is the result of multiple gene mutations that control the proliferation, differentiation and apoptosis of normal cells, and these mutations include the activation of the oncogenes and the inactivation of the anti-oncogenes. It has been found that mutation or activation of Ras gene and abnormal over-expression of Ras protein exist in about 30% human tumors (37,38). Therefore, research on the regulation of Ras signal transduction pathway plays an important role in the design of antitumor drugs targeting the cellular signal transduction pathway (39). Ras protein is considered as an important element to regulate the signal pathway of cell growth and proliferation. If Ras protein is activated continuously, it can bind downstream effective proteins and transmit signals to downstream signaling elements, which may cause abnormal proliferation of cells and lead to the occurrence of tumors (40). Therefore, we evaluated the role of Alo in Ras and its downstream signaling (41). From the results, we found Alo could significantly downregulate the level of Ras and suppress the levels of phosphorylation of Raf1 and Erk1/2, indicating that Alo could block the Ras/Raf1/Erk1/2 signaling pathway. To further verify role of Ras signaling in occurrence and development of human breast cancer cells (42), we chose a Ras inhibitor, ISIS 2503, to inhibit the Ras signaling pathway, and we found that ISIS 2503 could remarkably inhibit the expression of Ras. Moreover, we also found that co-treatment of Alo and ISIS 2503 had better inhibitory effects on proliferation, migration, invasion and apoptosis of MCF-7 and MDA-MB-231 cells than either one of them. These resulted indicated that Alo had better inhibitory effects on the occurrence and development of breast cancer via blockage of Ras pathway.
The current research is a preliminary study on the antitumor effect of Alo, the other effects of Alo and the mechanisms of Alo in breast cancer are not very clear. Based on the current findings, we will further explore the role of Alo in development of cancer both in vivo and in vitro, and explore the anticancer mechanism of Alo by genomics, proteomics and metabolomics in future. Taken together, the present data suggest that Alo possesses anticancer effects in human breast cancer cells via inhibiting the potential of cell proliferation, migration and invasion, and inducing apoptosis. Furthermore, Alo might act its role in suppressing human breast cancer cells via Ras inactivation. Based on our findings, Alo could be considered as a potential candidate to treat breast cancer.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
DT and YL designed the experiments. DT, YL, XL and ZT performed the experiments. DT and YL wrote the manuscript. All authors reviewed the manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5–29. doi: 10.3322/caac.21254. [DOI] [PubMed] [Google Scholar]
- 3.DeSantis C, Ma J, Bryan L, Jemal A. Breast cancer statics, 2013. CA Cancer J Clin. 2014;64:52–62. doi: 10.3322/caac.21203. [DOI] [PubMed] [Google Scholar]
- 4.DeSantis CE, Bray F, Ferlay J, Lortet-Tieulent J, Anderson BO, Jemal A. International variation in female breast cancer incidence and mortality rates. Cancer Epidemiol Biomarkers Prev. 2015;24:1495–1506. doi: 10.1158/1055-9965.EPI-15-0535. [DOI] [PubMed] [Google Scholar]
- 5.Fan L, Zheng Y, Yu KD, Liu GY, Wu J, Lu JS, Shen KW, Shen ZZ, Shao ZM. Breast cancer in a transitional society over 18 years: Trends and present status in Shanghai, China. Breast Cancer Res Treat. 2009;117:409–416. doi: 10.1007/s10549-008-0303-z. [DOI] [PubMed] [Google Scholar]
- 6.Kamal A, Lakshma Nayak V, Nagesh N, Vishnuvardan MV, Subba Reddy NV. Benzo[b]furan derivatives induces apoptosis by targeting the PI3K/Akt/mTOR signaling pathway in human breast cancer cells. Bioorg Chem. 2016;66:124–131. doi: 10.1016/j.bioorg.2016.04.004. [DOI] [PubMed] [Google Scholar]
- 7.Khpal M, Miller JRC, Petrovic Z, Hassanally D. Local anesthetic delivery via surgical drain provides improved pain control versus direct skin infiltration following axillary node dissection for breast cancer. Breast Cancer. 2018;25:185–190. doi: 10.1007/s12282-017-0810-z. [DOI] [PubMed] [Google Scholar]
- 8.Lauberg T, Tramm T, Nielsen T, Alsner J, Nord S, Myhre S, Sørlie T, Leung S, Fan C, Perou C, et al. Intrinsic subtypes and benefit from postmastectomy radiotherapy in node-positive premenopausal breast cancer patients who received adjuvant chemotherapy-results from two independent randomized trials. Acta Oncol. 2018;25:38–43. doi: 10.1080/0284186X.2017.1401735. [DOI] [PubMed] [Google Scholar]
- 9.Ito K, Park SH, Katsyv I, Zhang W, De Angelis C, Schiff R, Lrie HY. PTK6 regulates growth and survival of endocrine therapy-resistant ER+ breast cancer cells. NPJ Breast Cancer. 2017;3:45. doi: 10.1038/s41523-017-0047-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sui Y, Zhang X, Yang H, Wei W, Wang M. MicroRNA-133a acts as a tumour suppressor in breast cancer through targeting LASP1. Oncol Rep. 2018;39:473–482. doi: 10.3892/or.2017.6114. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 11.Zhang J, Yu K, Han X, Zhen L, Liu M, Zhang X, Ren Y, Shi J. Paeoniflorin influences breast cancer cell proliferation and invasion via inhibition of the Notch-1 signaling pathway. Mol Med Rep. 2018;17:1321–1325. doi: 10.3892/mmr.2017.8002. [DOI] [PubMed] [Google Scholar]
- 12.Wu C, Sun Z, Guo B, Ye Y, Han X, Qin Y, Liu S. Osthole inhibits bone metastasis of breast cancer. Oncotarget. 2017;8:58480–58493. doi: 10.18632/oncotarget.17024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Huang C, Lu CK, Tu MC, Chang JH, Chen YJ, Tu YH, Huang HC. Polyphenol-rich Avicennia marina leaf extracts induce apoptosis in human breast and liver cancer cells and in a nude mouse xenograft model. Oncotarget. 2016;7:35874–35893. doi: 10.18632/oncotarget.8624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhou WJ, Wang S, Hu Z, Zhou ZY, Song CJ. Angelica sinensis polysaccharides promotes apoptosis in human breast cancer cells via CREB-regulated caspase-3 activation. Biochem Biophys Res Commun. 2015;467:562–569. doi: 10.1016/j.bbrc.2015.09.145. [DOI] [PubMed] [Google Scholar]
- 15.Wang H, Guo S, Qian D, Qian Y, Duan JA. Comparative analysis of quinolizidine alkaloids from different parts of Sophora alopecuroides seeds by UPLC-MS/MS. J Pharm Biomed Anal 67–68. 2012:1–21. doi: 10.1016/j.jpba.2011.10.029. [DOI] [PubMed] [Google Scholar]
- 16.Yuan XY, Liu W, Zhang P, Wang RY, Guo JY. Effects and mechanisms of aloperine on 2, 4-dinitrofluorobenzene-induced allergic contact dermatitis in BALB/c mice. Eur J Pharmacol. 2010;629:147–152. doi: 10.1016/j.ejphar.2009.12.007. [DOI] [PubMed] [Google Scholar]
- 17.Chen S, Jin Z, Dai L, Wu H, Wang J, Wang L, Zhou Z, Yang L, Gao W. Aloperine induces apoptosis and inhibits invasion in MG-63 and U2OS human osteosarcoma cells. Biomed Pharmacother. 2018;97:45–52. doi: 10.1016/j.biopha.2018.04.035. [DOI] [PubMed] [Google Scholar]
- 18.Zhang L, Zheng Y, Deng H, Liang L, Peng J. Aloperine induces G2/M phase cell cycle arrest and apoptosis in HCT116 human colon cancer cells. Int J Mol Med. 2014;33:1613–1620. doi: 10.3892/ijmm.2014.1718. [DOI] [PubMed] [Google Scholar]
- 19.Brown GT, Murray GI. Current mechanistic insights into the roles of matrix metalloproteinases in tumour invasion and metastasis. J Pathol. 2015;237:273–281. doi: 10.1002/path.4586. [DOI] [PubMed] [Google Scholar]
- 20.Folgueras AR, Pendás AM, Sánchez LM, López-Otín C. Matrix metalloproteinases in cancer: From new functions to improved inhibition strategies. Int J Dev Biol. 2004;48:411–424. doi: 10.1387/ijdb.041811af. [DOI] [PubMed] [Google Scholar]
- 21.Yang Q, Ji M, Guan H, Shi B, Hou P. Shikonin inhibits thyroid cancer cell growth and invasiveness through targeting major signaling pathways. J Clin Endocrinol Metab. 2013;98:E1909–E1917. doi: 10.1210/jc.2013-2583. [DOI] [PubMed] [Google Scholar]
- 22.Zhang XG, Lu XF, Jiao XM, Chen B, Wu JX. PLK1 gene suppresses cell invasion of undifferentiated thyroid carcinoma through the inhibition of CD44v6, MMP-2 and MMP-9. Exp Ther Med. 2012;4:1005–1009. doi: 10.3892/etm.2012.729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Downward J. Targeting RAS signalling pathways in cancer therapy. Nat Rev Cancer. 2003;3:11–22. doi: 10.1038/nrc969. [DOI] [PubMed] [Google Scholar]
- 24.Hilger RA, Scheulen ME, Strumberg D. The Ras-Raf-MEK-ERK pathway in the treatment of cancer. Onkologie. 2002;25:511–518. doi: 10.1159/000068621. [DOI] [PubMed] [Google Scholar]
- 25.Friday BB, Adjei AA. Advances in targeting the Ras/Raf/MEK/Erk mitogen-activated protein kinase cascade with MEK inhibitors for cancer therapy. Clin Cancer Res. 2008;14:342–346. doi: 10.1158/1078-0432.CCR-07-4790. [DOI] [PubMed] [Google Scholar]
- 26.Dancey JE. Agents targeting ras signaling pathway. Curr Pharm Des. 2002;8:2259–2267. doi: 10.2174/1381612023393071. [DOI] [PubMed] [Google Scholar]
- 27.Adjei AA, Dy GK, Erlichman C, Reid JM, Sloan JA, Pitot HC, Alberts SR, Goldberg RM, Hanson LJ, Atherton PJ, et al. A phase I trial of ISIS 2503, an antisense inhibitor of H-ras, in combination with gemcitabine in patients with advanced cancer. Clin Cancer Res. 2003;9:115–123. [PubMed] [Google Scholar]
- 28.Brackstone M, Townson JL, Chambers AF. Tumor dormancy in breast cancer: An update. Breast Cancer Res. 2007;9:208. doi: 10.1186/bcr1677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Huang KW, Wu HL, Lin HL, Liang PC, Chen PJ, Chen SH, Lee HI, Su PY, Wu WH, Lee PH, et al. Combining antiangiogenic therapy with immunotherapy exerts better therapeutical effects on large tumors in a woodchuck hepatoma model; Proc Natl Acad Sci USA; 2010; pp. 14769–14774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen Y, Hao H, He S, Cai L, Li Y, Hu S, Ye D, Hoidal J, Wu P, Chen X. Lipoxin A4 and its analogue suppress the tumor growth of transplanted H22 in mice: The role of antiangiogenesis. Mol Cancer Ther. 2010;9:2164–2174. doi: 10.1158/1535-7163.MCT-10-0173. [DOI] [PubMed] [Google Scholar]
- 31.Banerjee S, A'Hern R, Detre S, Littlewood-Evans AJ, Evans DB, Dowsett M, Martin LA. Biological evidence for dual antiangiogenic-antiaromatase activity of the VEGFR inhibitor PTK787/ZK222584 in vivo. Clin Cancer Res. 2010;16:4178–4187. doi: 10.1158/1078-0432.CCR-10-0456. [DOI] [PubMed] [Google Scholar]
- 32.Vaux DL, Korsmeyer SJ. Cell death in development. Cell. 1999;96:245–254. doi: 10.1016/S0092-8674(00)80564-4. [DOI] [PubMed] [Google Scholar]
- 33.Ackler S, Mitten MJ, Foster K, Oleksijew A, Refici M, Tahir SK, Xiao Y, Tse C, Frost DJ, Fesik SW, et al. The Bcl-2 inhibitor ABT-263 enhances the response of multiple chemotherapeutic regimens in hematologic tumors in vivo. Cancer Chemother Pharmacol. 2010;66:869–880. doi: 10.1007/s00280-009-1232-1. [DOI] [PubMed] [Google Scholar]
- 34.Edelstein ML, Abedi MR, Wixon J. Gene therapy clinical trials worldwide to 2007-an update. J Gene Med. 2007;9:833–842. doi: 10.1002/jgm.1100. [DOI] [PubMed] [Google Scholar]
- 35.Westermarck J, Kähäri VM. Regulation of matrix metalloproteinase expression in tumor invasion. FASEB J. 1999;13:781–792. doi: 10.1096/fasebj.13.8.781. [DOI] [PubMed] [Google Scholar]
- 36.Liu X, Zhao W, Wang W, Lin S, Yang L. Puerarin suppresses LPS-induced breast cancer cell migration, invasion and adhesion by blockage NF-κB and Erk pathway. Biomed Pharmacother. 2017;92:429–436. doi: 10.1016/j.biopha.2017.05.102. [DOI] [PubMed] [Google Scholar]
- 37.Kim D, Kim SY, Lee JS, Hong YS, Kim JE, Kim KP, Kim J, Jang SJ, Yoon YK, Kim TW. Primary tumor location predicts poor clinical outcome with cetuximab in RAS wild-type metastatic colorectal cancer. BMC Gastroenterol. 2017;17:121. doi: 10.1186/s12876-017-0694-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhou J, Zhang S, Chen X, Zheng X, Yao Y, Lu G, Zhou J. Palbociclib, a selective CDK4/6 inhibitor, enhances the effect of selumetinib in RAS-driven non-small cell lung cancer. Cancer Lett. 2017;408:130–137. doi: 10.1016/j.canlet.2017.08.031. [DOI] [PubMed] [Google Scholar]
- 39.Wang Z, Ma L, Su M, Zhou Y, Mao K, Li C, Peng G, Zhou C, Shen B, Dou J. Baicalin induces cellular senescence in human colon cancer cells via upregulation of DEPP and the activation of Ras/Raf/MEK/ERK signaling. Cell Death Dis. 2018;9:217. doi: 10.1038/s41419-017-0223-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pan Q, Liu R, Banu H, Ma L, Li H. Inhibition of isoprenylcysteine carboxylmethyltransferase sensitizes common chemotherapies in cervical cancer via Ras-dependent pathway. Biomed Pharmacother. 2018;99:169–175. doi: 10.1016/j.biopha.2018.01.048. [DOI] [PubMed] [Google Scholar]
- 41.Yu SH, Wang TH, Au LC. Specific repression of mutant K-Ras by 10–23 DNAzyme: Sensitizing cancer cell to anti-cancer therapies. Biochem Biophys Res Commun. 2009;378:230–234. doi: 10.1016/j.bbrc.2008.11.027. [DOI] [PubMed] [Google Scholar]
- 42.Bolchi C, Pallavicini M, Rusconi C, Diomede L, Ferri N, Corsini A, Fumagalli L, Pedretti A, Vistoli G, Valoti E. Peptidomimetic inhibitors of farnesyltransferase with high in vitro activity and significant cellular potency. Bioorg Med Chem Lett. 2007;17:6192–6196. doi: 10.1016/j.bmcl.2007.09.015. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.