Abstract
Sinomenine (SIN), a pure alkaloid isolated from Sinomenium acutum, has been widely used in arthritis for its anti-inflammatory effect, but little is known about the effect of SIN on human ulcerative colitis (UC). In the present study, the effect and mechanism of SIN was examined in a dextran sulfate sodium (DSS)-induced murine colitis model, which mimics human UC. Oral administration of SIN significantly suppressed the elevated disease activity index and ameliorated colonic histological damage in a DSS-induced colitis model. Tumor necrosis factor-α, interleukin-6 and inducible nitric oxide synthase levels were also reduced as detected by reverse transcription-quantitative polymerase chain reaction. In addition, SIN reversed the decreased colon length and colonic superoxide dismutase activity. Furthermore, western blot analysis revealed that nuclear factor-erythroid 2-related factor 2 (Nrf2) and its downstream genes, heme oxygenase-1 and NADP(H) quinone oxidoreductase 1 (NQO-1), were markedly activated by SIN. The current results indicated that SIN alleviated DSS-induced colitis in mice, which may be due to its antioxidant properties and was at least in part dependent on the Nrf2/NQO-1 signaling pathway. Therefore, SIN may have potential applications as a protective drug for patients with UC.
Keywords: ulcerative colitis, sinomenine, inflammation, oxidative stress, nuclear factor erythroid 2-related factor 2
Introduction
Ulcerative colitis (UC) is a chronic, relapsing and non-specific immunological-mediated disorder of the gastrointestinal tract. Together with Crohn's disease (CD), it is a type of inflammatory bowel disease (IBD) (1). Individuals suffering from UC are at a high risk of developing colitis-associated cancer, which causes up to 15% of IBD-associated cases of mortality (2). The pathogenesis of UC is complex and has not been clearly elucidated at present. However, genetic, environmental and immunological factors are all thought to be contributors (3). It has been identified that unbalanced cytokine secretion leads to intestinal tissue damage and epithelial barrier disruption in patients with UC and experimental models of colitis (4). Oxidative stress is thought to be a key factor in the development of UC as a regulator of oxidant/antioxidant balance (5).
Nuclear erythroid 2-related factor 2 (Nrf2) plays an important role in antioxidant reactions. It can regulate the transcription of several enzymes in detoxification and antioxidant responses (6). In general, Nrf2 is located in the cytosol, bound to Kelch-like ECH-associated protein 1. When exposed to oxidative stress, Nrf2 enters the nucleus and combines with antioxidant responsive element to regulate the expression of Phase II enzymes, including heme oxygenase 1 (HO-1) and NAD(P)H quinine oxidoreductases (NQOs). These enzymes are critical for maintaining optimal cellular functions (7,8).
Sinomenine hydrochloride (SIN, purity >97%) is an active alkaloid originally extracted from the medicinal herb Sinomenium acutum. SIN exhibits anti-inflammatory and immune-regulatory effects (9,10), and possesses notable therapeutic capacity in treating arthritis (11,12). Studies in vivo have indicated that SIN protects against several autoimmune and inflammation-associated diseases (13,14). In addition, SIN is able to attenuate 2,4,6-trinitrobenzene sulphonic acid (TNBS)-induced colitis, an animal model that mimics human CD (15,16). However, the therapeutic mechanism remains unclear. In the current study, the therapeutic effects of SIN were investigated in a dextran sulfate sodium (DSS)-induced colitis model, which possesses similar fundamental clinical and histological features to human UC (17).
In the current study, it was identified that SIN alleviated DSS-induced colitis by producing antioxidant and anti-inflammatory effects, partly via activating the Nrf2/NQO-1 pathway.
Materials and methods
Animals
A total of 40 6–8-week-old female C57BL/6 mice (weight, 18–20 g) were purchased from Cavens Laboratory Animal Co., Ltd. (Changzhou, China). The certification number is SCXK (SU) 2011-0003. The mice were fed a standard laboratory diet, had access to sterile water and were housed under controlled conditions (23±2°C, 50±5% humidity and 12 h light/dark cycle). All animal experiments were approved by the Ethics Committee of Changzhou No. 2 People's Hospital (Changzhou, China), following the ARRIVE guidelines (18).
Reagents
SIN (purity >97%) was purchased from Aladdin (Shanghai, China) and salicylazosulfapyridine (SASP) was obtained from Shanghai Sine Tianping Pharmaceutical Co., Ltd. (Shanghai, China). They were both dissolved in 0.9% NaCl solution. DSS (molecular weight, 36,000–50,000 kDa) was purchased from MP Biomedicals (Thermo Fisher Scientific, Inc., Waltham, MA, USA). The reagent for superoxide dismutase (SOD) examination was obtained from Nanjing Jiancheng Bioengineering Institute (Nanjing, China).
Induction and assessment of DSS-induced murine colitis model
A total of 40 mice were randomized into four equal groups: Control group, DSS model group, DSS + SASP group and DSS + SIN group. In the control group, colitis was not induced. The DSS group mice were treated with 3% DSS in their drinking water for 7 days and then were given normal drinking water for 3 days for recovery (19,20). SASP and SIN were administered by gavage from day 1 to day 10 at doses of 400 and 100 mg/kg per day, respectively. All mice were sacrificed on day 11. During the course of the experiment, body weight, stool consistency and bleeding scores of every mouse were observed to measure the disease activity index (DAI) (21,22). The scoring system for the DAI is presented in Table I. The rate of body weight gain or loss in each mouse was calculated using the following formula: Body weight change (%) = [(Weight change at day X)-(Weight at Day 1)]/(Weight at Day 1) ×100.
Table I.
DAI scoring system.
| Score | Weight loss (%) | Stool consistency | Blood in stool |
|---|---|---|---|
| 0 | None | Normal | Negative |
| 1 | 1–5% | Normal | Negative |
| 2 | 6–10% | Loose stool | Hemoccult positive |
| 3 | 11–15% | Loose stool | Hemoccult positive |
| 4 | >15% | Diarrhea | Gross bleeding |
The DAI value is calculated as the scores of weight loss, stool consistency and blood in stool divided by three. Normal stools = well-formed stools; loose stools = pasty stools that did not adhere to the anus; diarrhea = liquid stools that adhered to the anus. DAI, disease activity index.
Histological analysis
After the mice were sacrificed, the colons were removed and the colon length of each animal was measured. The dissected colon tissue was washed with cold phosphate-buffered saline (PBS). Two-thirds of the distal colon was stored at −80°C for biochemical examination, and the rest was fixed in 4% paraformaldehyde for 2 h at room temperature for histopathological analysis. The colon sections were then embedded in paraffin, and stained with hematoxylin and eosin for 5 min at room temperature, according to standard protocols (23). Histological scoring was performed as follows: 0, no signs of inflammation; 1, low leukocyte infiltration; 2, moderate leukocyte infiltration; 3, high leukocyte infiltration, moderate fibrosis, high vascular density, thickening of the colon wall, moderate goblet cell loss and focal loss of crypts; 4, transmural infiltrations, massive loss of goblet cell, extensive fibrosis and diffuse loss of crypts.
SOD activity assessment
Colon tissue was homogenized in cold PBS. The activity level of total superoxide dismutase was detected with total superoxide dismutase assay kit (cat. no. A001-1-1) according to the manufacturer's protocol (Nanjing Jiancheng Bioengineering Institute). The values are expressed as U/mg protein.
RNA isolation and reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted from the colon samples using TRIzol® reagent (Invitrogen; Thermo Fisher Scientific, Inc.). cDNA was synthesized from RNA using a reverse transcriptase kit (Takara Bio, Inc., Otsu, Japan). The reverse transcription conditions were as follows: 37°C for 15 min, 85°C for 5 sec and hold at 4°C. SYBR Green Master mix (Vazyme, Nanjing, China) and an ABI 7500 system (Applied Biosystems; Thermo Fisher Scientific, Inc.) were used to conduct qPCR, in order to analyze gene expression. The cycling conditions were as follows: Initial step at 95°C for 10 min, followed by 40 cycles at 95°C for 15 sec and 60°C for 1 min. The primer sequences used in the present study are presented in Table II. The mRNA expression level for each target gene was normalized to the level of GAPDH. Expression of target genes was analyzed by the 2−ΔΔCq method (24).
Table II.
Primer sequences for reverse transcription-quantitative polymerase chain reaction.
| Gene | Sequences (5′-3′) |
|---|---|
| TNF-α | |
| F | CATCTTCTCAAAATTCGAGTGAC |
| R | TGGGAGTAGACAAGGTACAACCC |
| IL-6 | |
| F | GCTGGTGACAACCACGGCCT |
| R | AGCCTCGACTTGTGAAGTGGT |
| iNOS | |
| F | CCAACCTGCAGGTCTTCGATG |
| R | GTCGATGCACAACTGGGTGAAC |
| HO-1 | |
| F | ATGTGGCCCTGGAGGAGGAGA |
| R | CGCTGCATGGCTGGTGTGTAG |
| NQO-1 | |
| F | GGATTGGACCGAGCTGGAA |
| R | AATTGCAGTGAAGATGAAGGCAAC |
| GAPDH | |
| F | AAGGTCGGAGTCAACGGATTT |
| R | AGATGATGACCCTTTTGGCTC |
TNF-α, tumor necrosis factor-α; IL-6, interleukin-6; iNOS, inducible nitric oxide synthase; HO-1, heme oxygenase-1; NQO-1, NADP (H) quinone oxidoreductase 1; GAPDH, glyceraldehyde-phosphate dehydrogenase; F, forward; R, reverse.
Protein extraction and western blot analysis
Following the treatments, cytosolic and nuclear proteins were isolated from colon samples using a Nuclear and Cytoplasmic Protein Extraction kit (Sangon Biotech Co., Ltd., Shanghai, China) according to the manufacturer's protocol. The protein concentration in the supernatants was determined using an Enhanced BCA Protein Assay kit (Beyotime Institute of Biotechnology, Nanjing, China). Extracts were stored at −70°C until further experimentation.
An equal amount of protein (40 µg) was separated with 12% SDS-PAGE. The proteins were then transferred to polyvinylidene difluoride (PVDF) membranes (EMD Millipore, Billerica, MA, USA) using a wet transfer system (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Non-fat milk (5%) was used to block the PVDF membranes for 2 h at 37°C. Proteins were detected using specific primary antibodies against Nrf2 (cat. no. 12721T; rabbit monoclonal; Cell Signaling Technology, Inc., Danvers, MA, USA), NQO-1 (cat. no. ab80588; rabbit monoclonal; Abcam, Cambridge, MA, USA), β-actin (cat. no. 479T; rabbit monoclonal; Cell Signaling Technology, Inc.) and Histone H3 (cat. no. 4499T; rabbit monoclonal; Cell Signaling Technology, Inc.) diluted 1:1,000 overnight at 4°C. Then, the membranes were incubated with goat anti-rabbit IgG-horseradish peroxidase conjugated secondary antibodies (cat. no. sc-2004; Santa Cruz Biotechnology, Inc., Dallas, TX, USA) diluted 1:5,000 for 1.5 h at room temperature. All antibodies were diluted in Primary Antibody Dilution Buffer (Beyotime Institute of Biotechnology). Proteins were visualized using enhanced chemiluminescent reagents (Thermo Fisher Scientific, Inc.). The immunoblots were quantified using ImageJ software version 1.8 (National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
All experimental results were expressed as the mean ± standard error of the mean of three independent experiments. Statistical analysis was performed using Student's t-test or one-way analysis of variance with Tukey's post-hoc test with GraphPad Prism 5 (GraphPad Software, Inc., La Jolla, CA, USA). P<0.05 was considered to indicate a statistically significant difference.
Results
SIN restores DSS-induced colitis
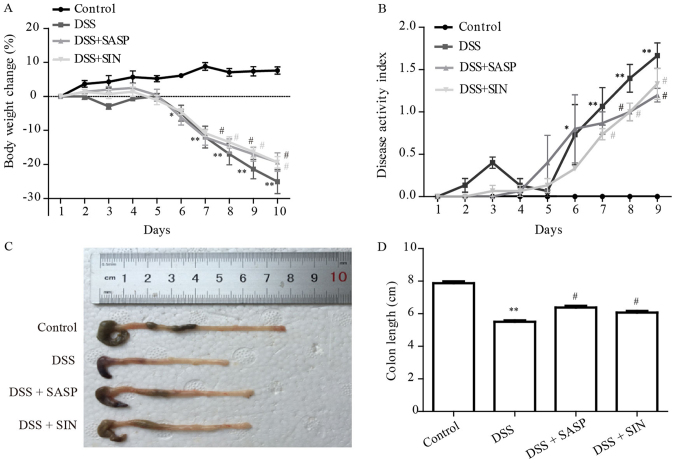
Mice treated with 3% DSS developed severe colitis, which led to body weight loss and high DAI score (body weight change, stool consistency and the presence of blood in the stool) in comparison with the control mice. SASP, an aminosalicylate, was used as a positive control. SASP and SIN administration prevented body weight loss and reduced the DAI score compared with the DSS model group mice (Fig. 1A and B). DSS-induced colonic shortening was also improved by SASP and SIN treatment (Fig. 1C and D).
Figure 1.
SIN ameliorates DSS-induced colitis. The (A) percentage change (%) of mouse body weight, (B) disease activity index and (C and D) colon length of mice treated with DSS alone, DSS + SASP or DSS + SIN. The data are presented as mean ± standard error of the mean. *P<0.05, **P<0.01 vs. control group; #P<0.05 vs. DSS group. DSS, dextran sulfate sodium; SIN, sinomenine; SASP, salicylazosulfapyridine.
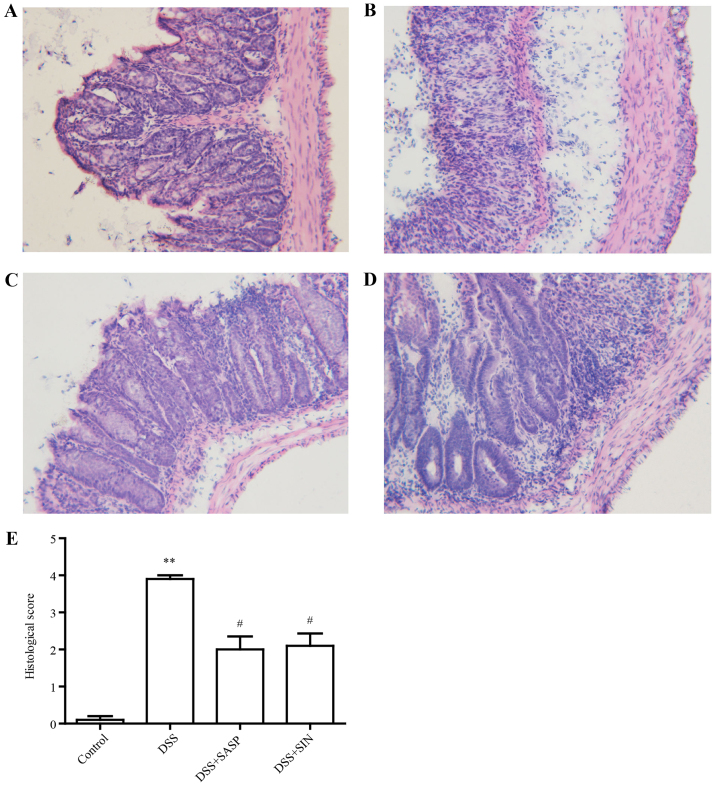
SIN attenuates colitis histological damage
In the control group, the colons of mice exhibited full structure without any obvious damage (Fig. 2A). However, infiltration of inflammatory cells, defection of crypt structure and mucosal ulceration were observed in the colons of DSS model group mice (Fig. 2B). SASP and SIN treatment attenuated this histological damage (Fig. 2C and D). Histological scores indicated that DSS caused histological defects, while SASP and SIN significantly attenuated these pathological changes (Fig. 2E).
Figure 2.
SIN attenuates histological damage in DSS-induced colitis. Hematoxylin and eosin-stained colon sections from (A) control, (B) DSS, (C) DSS + SASP and (D) DSS + SIN treated mice. Magnification, ×20. (E) Summary of the histological scores. Data are presented as mean ± standard error of the mean. **P<0.01 vs. control group; #P<0.05 vs. DSS group. DSS, dextran sulfate sodium; SIN, sinomenine; SASP, salicylazosulfapyridine.
SIN increases SOD activity
To evaluate the antioxidant effects of SIN in a DSS-induced colitis model, the activity of SOD was analyzed using the xanthine/xanthine oxidase method. As presented in Fig. 3, the SOD activity of the DSS-induced colitis group was significantly decreased compared with the control group (P<0.05). Conversely, mice treated with SASP and SIN exhibited increased SOD activity compared with the DSS model group.
Figure 3.
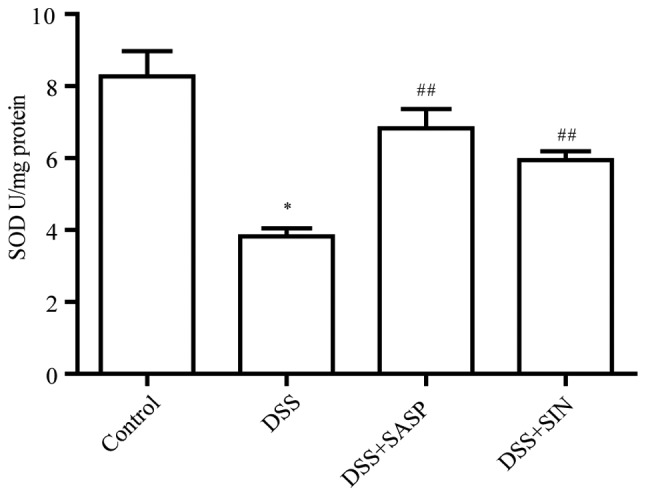
SIN reduces colonic SOD activity. The activity of SOD was analyzed using the xanthine/xanthine oxidase method in the colon tissues of control, DSS, DSS + SASP and DSS + SIN treated mice. Data are represented as mean ± SEM. *P<0.05 vs. control group; ##P<0.01 vs. DSS group. DSS, dextran sulfate sodium; SIN, sinomenine; SASP, salicylazosulfapyridine; SOD, superoxide dismutase.
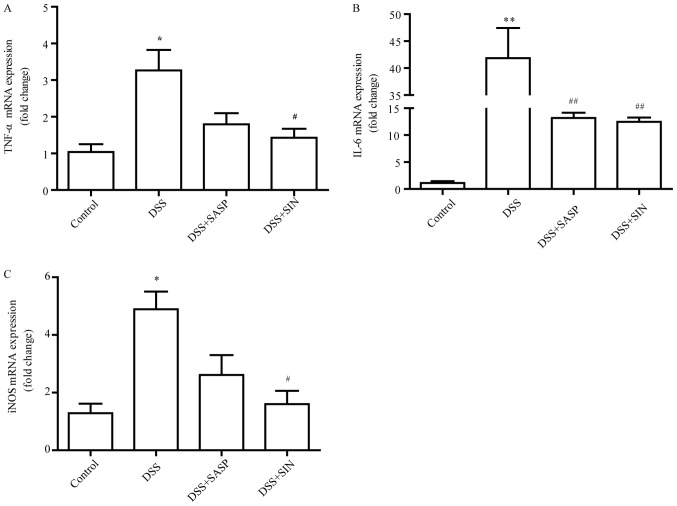
SIN decreases mRNA levels of tumor necrosis factor (TNF)-α, interleukin (IL)-6 and inducible nitric oxide synthase (iNOS)
As presented in Fig. 4A and B, the mRNA levels of TNF-α and IL-6 were greatly increased in colonic tissues following treatment with DSS, whereas SIN significantly suppressed this enhanced expression. DSS treatment enhanced iNOS mRNA expression in the colon compared with the control group (P<0.05), while SASP and SIN treatment significantly inhibited the expression of iNOS induced by DSS (Fig. 4C).
Figure 4.
SIN decreases the mRNA expression levels of TNF-α, IL-6 and iNOS. Reverse transcription-quantitative polymerase chain reaction was performed on gut homogenates to detect (A) TNF-α, (B) IL-6 and (C) iNOS. Data are represented as mean ± standard error of the mean. *P<0.05, **P<0.01 vs. control group; #P<0.05, ##P<0.01 vs. DSS group. DSS, dextran sulfate sodium; IL-6, interleukin-6; iNOS, inducible nitric oxide synthase; SIN, sinomenine; SASP, salicylazosulfapyridine; SOD, superoxide dismutase; TNF-α, tumor necrosis factor-α.
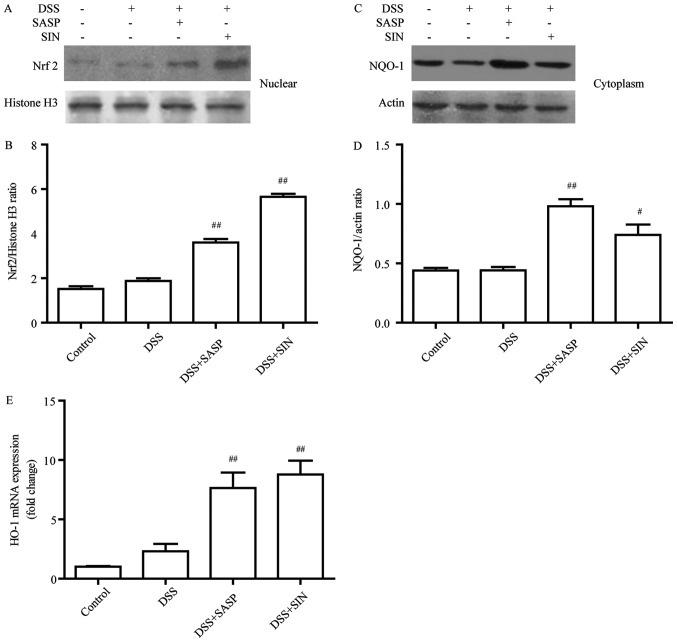
SIN induces the Nrf2/NQO-1 pathway in DSS-induced colitis
Nrf2 protein level was evaluated in the DSS-induced colitis model following treatment with SASP or SIN. As presented in Fig. 5A and B, the nuclear translocation of Nrf2 was significantly improved in the SASP and SIN treatment groups compared with the DSS group. Furthermore, two downstream targets of Nrf2 were examined: HO-1 and NQO-1. The results indicated that the protein level of NQO-1 (Fig. 5C and D) and the mRNA level of HO-1 (Fig. 5E) were significantly increased in SIN-treated mice compared with the DSS group.
Figure 5.
SIN induces the Nrf2/NQO-1 pathway in DSS-induced colitis. Western blot analysis of (A and B) nuclear Nrf2 and (C and D) cytoplasmic NQO-1 protein expression. (E) Reverse transcription-quantitative polymerase chain reaction analysis of HO-1 mRNA expression levels. Data are represented as mean ± standard error of the mean. #P<0.05, ##P<0.01 vs. DSS group. DSS, dextran sulfate sodium; HO-1, heme oxygenase-1; NQO-1, NADP (H) quinone oxidoreductase 1; Nrf2, nuclear factor erythroid 2-related factor 2; SIN, sinomenine; SASP, salicylazosulfapyridine.
Discussion
Ulcerative colitis has a serious negative impact on human health, and is associated with colorectal cancer (25). In clinical practice, aminosalicylates, corticosteroids, immunosuppressors and biological agents are used to treat UC symptoms. These drugs are able to regulate the immune and inflammatory response via specific targets; however, they still have certain adverse effects (26). Therefore, it is important to develop an effective drug for UC with few side effects. In the current study, SASP, an aminosalicylate, was used as a positive control. The effect and mechanism of SIN was investigated in the treatment of DSS-induced colitis.
A previous study demonstrated that TNBS-induced colitis is attenuated by SIN at doses of 30, 100 and 200 mg/kg by gastric gavage (15). Yu et al (16) reported that SIN (100 or 200 mg/kg) administered orally in mice with TNBS-induced colitis results in significant improvements. These findings suggest that the dose and treatment of SIN used in the current study were appropriate.
The current results indicated that DSS-induced colon damage could be ameliorated by SIN treatment, according to the evaluation of DAI, colon length and histological analysis. The DAI in the DSS model group mice increased significantly, whereas SIN administration decreased the DAI. Changes in colon length and histological structure were also detected in the DSS model group and the control group, and SIN attenuated these changes.
Oxidative stress is thought to be a key factor in the development of UC (27). The enhancement of reactive oxygen species and free radicals could play a key role in the pathophysiology of UC. It could lead to destruction of cell membrane integrity resulting from DNA damage, protein oxidation and lipid peroxidation, and subsequent mucosal inflammatory infiltration and ulceration formation (28,29). A decrease of SOD activity may lead to redundant superoxide anions, which usually generates other forms of carbon-, nitrogen- and oxygen-centered radicals, and may aggravate the oxidative damage induced by DSS (30). In the current study, SOD activity was reduced in DSS group mice, while SIN treatment markedly improved the SOD activity. This indicated that SIN administration may counteract DSS-induced colon injury via its antioxidant effect.
It was previously reported that activation of excessive iNOS is correlated with gastrointestinal inflammation, which could accelerate the development of IBD (31). Mouzaoui et al (32) identified that aminoguanidine and curcumin could attenuate the colon damage in IBD by inhibiting iNOS formation. The current results demonstrated that iNOS mRNA expression in DSS group mice was increased, while administration of SIN suppressed these levels.
Pro-inflammatory cytokines play a major role in UC development and progression, particularly TNF-α. TNF-α exhibits a pleiotropic effect in colonic mucosa by activating intracellular signaling (33,34). IL-6 is also one of the crucial cytokines in the pathogenesis of IBD (35). Reduced IL-6 could slow down the development of UC and colitis-associated colon cancer (36). In the current study, RT-qPCR was performed to evaluate the mRNA expression of TNF-α and IL-6 in the colons of mice. DSS-induced colitis mice expressed increased levels of TNF-α and IL-6, while SIN treatment decreased the mRNA levels. This indicated that SIN may alleviate DSS-induced colitis by suppressing the expression of TNF-α and IL-6 at the mRNA level.
Nrf2 signaling plays a crucial role in defending against oxidative stress and inflammation reactions, which are both associated with the occurrence of UC (37). HO-1, an Nrf2 target gene, is activated by stimuli that induce cellular stress, and reduces inflammatory cytokine secretion in numerous diseases, including sepsis and LPS-stimulated macrophages (38,39). Furthermore, HO-1 exerts cytoprotective effects by increasing anti-oxidative capacity and inhibiting oxidative stress (40,41). Numerous natural compounds isolated from plants could ameliorate experimental colitis by activating the Nrf2 signaling pathway. Wagner et al (42) identified that pre-treatment of sulforaphane could reduce DSS-induced colitis in mice by activating Nrf2 signaling and subsequently inhibiting inflammatory mediators. The current results indicated that SIN alleviated DSS-induced colitis via Nrf2 signaling. The mRNA levels of Nrf2 target gene HO-1 were elevated in mice treated with SIN compared with colitis mice. Western blotting indicated that SIN administration activated Nrf2 to induce NQO-1 expression.
In conclusion, the present study demonstrated that SIN treatment alleviated DSS-induced colitis by inhibiting proinflammatory mediators in mice. In addition, SIN may activate the Nrf2/NQO-1 signaling pathway to exert its antioxidant effect. The current study provides evidence that maintaining a balance of oxidative status is important for regulating inflammation in colitis, and that SIN is a potential novel drug for treating UC in patients.
Acknowledgements
Not applicable.
Glossary
Abbreviations
- UC
ulcerative colitis
- DSS
dextran sulfate sodium
- SASP
salicylazosulfapyridine
- SIN
sinomenine
- DAI
disease activity index
- SOD
superoxide dismutase
- iNOS
inducible nitric oxide synthase
- Nrf2
nuclear factor erythroid 2-related factor 2
- HO-1
heme oxygenase-1
- NQO-1
NADP (H) quinone oxidoreductase 1
Funding
The present study was supported by grants from the National Natural Science Foundation of China (grant no. 81672799), Changzhou Health and Family Planning Commission Project (grant nos. ZD201606, QN201711) and Nanjing Medical University School Fund (grant nos. 2016NJMUZD081, 2017NJMU043).
Availability of data and materials
All data generated and analyzed during the study are available from the corresponding author on reasonable request.
Authors' contributions
YZ and LT designed the study and wrote the manuscript. YZ, HL, JS and LC conducted the experiments and performed the statistical analysis. YZ and CQ supervised the experiments, analyzed the data and revised the manuscript. All authors have read and approved the final manuscript.
Ethical approval and consent for participation
Research involving animals was approved by the Ethics Committee of the Affiliated Changzhou No. 2 People's Hospital of Nanjing Medical University (Nanjing, China). All animal protocols performed in this study were conducted strictly based on the guidelines of the Jiangsu Committee on Animal Care.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Abraham C, Cho JH. Inflammatory bowel disease. N Engl J Med. 2009;361:2066–2078. doi: 10.1056/NEJMra0804647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Munkholm P. Review article: The incidence and prevalence of colorectal cancer in inflammatory bowel disease. Aliment Pharmacol Ther. 2003;2(Suppl 18):1–5. doi: 10.1046/j.1365-2036.18.s2.2.x. [DOI] [PubMed] [Google Scholar]
- 3.Kaistha A, Levine J. Inflammatory bowel disease: The classic gastrointestinal autoimmune disease. Curr Probl Pediatr Adolesc Health Care. 2014;44:328–334. doi: 10.1016/j.cppeds.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 4.Danese S, Malesci A, Vetrano S. Colitis-associated cancer: The dark side of inflammatory bowel disease. Gut. 2011;60:1609–1610. doi: 10.1136/gutjnl-2011-300953. [DOI] [PubMed] [Google Scholar]
- 5.Pandurangan AK, Mohebali N, Norhaizan ME, Looi CY. Gallic acid attenuates dextran sulfate sodium-induced experimental colitis in BALB/c mice. Drug Des Devel Ther. 2015;9:3923–3934. doi: 10.2147/DDDT.S86345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Stachel I, Geismann C, Aden K, Deisinger F, Rosenstiel P, Schreiber S, Sebens S, Arlt A, Schafer H. Modulation of nuclear factor E2-related factor-2 (Nrf2) activation by the stress response gene immediate early response-3 (IER3) in colonic epithelial cells: A novel mechanism of cellular adaption to inflammatory stress. J Biol Chem. 2014;289:1917–1929. doi: 10.1074/jbc.M113.490920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Uruno A, Motohashi H. The Keap1-Nrf2 system as an in vivo sensor for electrophiles. Nitric Oxide. 2011;25:153–160. doi: 10.1016/j.niox.2011.02.007. [DOI] [PubMed] [Google Scholar]
- 8.Kim J, Cha YN, Surh YJ. A protective role of nuclear factor-erythroid 2-related factor-2 (Nrf2) in inflammatory disorders. Mutat Res. 2010;690:12–23. doi: 10.1016/j.mrfmmm.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 9.Cheng Y, Zhang J, Hou W, Wang D, Li F, Zhang Y, Yuan F. Immunoregulatory effects of sinomenine on the T-bet/GATA-3 ratio and Th1/Th2 cytokine balance in the treatment of mesangial proliferative nephritis. Int Immunopharmacol. 2009;9:894–899. doi: 10.1016/j.intimp.2009.03.014. [DOI] [PubMed] [Google Scholar]
- 10.Wang Q, Li XK. Immunosuppressive and anti-inflammatory activities of sinomenine. Int Immunopharmacol. 2011;11:373–376. doi: 10.1016/j.intimp.2010.11.018. [DOI] [PubMed] [Google Scholar]
- 11.Tong B, Yu J, Wang T, Dou Y, Wu X, Kong L, Dai Y, Xia Y. Sinomenine suppresses collagen-induced arthritis by reciprocal modulation of regulatory T cells and Th17 cells in gut-associated lymphoid tissues. Mol Immunol. 2015;65:94–103. doi: 10.1016/j.molimm.2015.01.014. [DOI] [PubMed] [Google Scholar]
- 12.Mu H, Yao RB, Zhao LJ, Shen SY, Zhao ZM, Cai H. Sinomenine decreases MyD88 expression and improves inflammation-induced joint damage progression and symptoms in rat adjuvant-induced arthritis. Inflammation. 2013;36:1136–1144. doi: 10.1007/s10753-013-9648-5. [DOI] [PubMed] [Google Scholar]
- 13.Xiong L, Yang L. Effects of alkaloid sinomenine on levels of IFN-γ, IL-1β, TNF-α and IL-6 in a rat renal allograft model. Immunotherapy. 2012;4:785–791. doi: 10.2217/imt.12.80. [DOI] [PubMed] [Google Scholar]
- 14.Li Y, Duan Z, Tian Y, Liu Z, Wang Q. A novel perspective and approach to intestinal octreotide absorption: Sinomenine-mediated reversible tight junction opening and its molecular mechanism. Int J Mol Sci. 2013;14:12873–12892. doi: 10.3390/ijms140612873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cheng H, Xia B, Guo Q, Zhang L, Wang F, Jiang L, Wang Z, Zhang Y, Li C. Sinomenine attenuates 2,4,6-trinitrobenzene sulfonic acid-induced colitis in mice. Int Immunopharmacol. 2007;7:604–611. doi: 10.1016/j.intimp.2007.01.003. [DOI] [PubMed] [Google Scholar]
- 16.Yu Q, Zhu S, Zhou R, Yi F, Bing Y, Huang S, Wang Z, Wang C, Xia B. Effects of sinomenine on the expression of microRNA-155 in 2,4,6-trinitrobenzenesulfonic acid-induced colitis in mice. PloS One. 2013;8:e73757. doi: 10.1371/journal.pone.0073757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Leonardi I, Nicholls F, Atrott K, Cee A, Tewes B, Greinwald R, Rogler G, Frey-Wagner I. Oral administration of dextran sodium sulphate induces a caecum-localized colitis in rabbits. Int J Exp Pathol. 2015;96:151–162. doi: 10.1111/iep.12117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research. J Pharmacol Pharmacother. 2010;1:94–99. doi: 10.4103/0976-500X.72351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bang B, Lichtenberger LM. Methods of inducing inflammatory bowel disease in mice. Curr Protoc Pharmacol. 2016;72:5.58.1–5.58.42. doi: 10.1002/0471141755.ph0558s72. [DOI] [PubMed] [Google Scholar]
- 20.Koboziev I, Karlsson F, Zhang S, Grisham MB. Pharmacological intervention studies using mouse models of the inflammatory bowel diseases: Translating preclinical data into new drug therapies. Inflamm Bowel Dis. 2011;17:1229–1245. doi: 10.1002/ibd.21557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vong LB, Tomita T, Yoshitomi T, Matsui H, Nagasaki Y. An orally administered redox nanoparticle that accumulates in the colonic mucosa and reduces colitis in mice. Gastroenterology. 2012;143:1027–1036.e1023. doi: 10.1053/j.gastro.2012.06.043. [DOI] [PubMed] [Google Scholar]
- 22.Sann H, Erichsen Jv, Hessmann M, Pahl A, Hoffmeyer A. Efficacy of drugs used in the treatment of IBD and combinations thereof in acute DSS-induced colitis in mice. Life Sci. 2013;92:708–718. doi: 10.1016/j.lfs.2013.01.028. [DOI] [PubMed] [Google Scholar]
- 23.Feldman AT, Wolfe D. Tissue processing and hematoxylin and eosin staining. Methods Mol Biol. 2014;1180:31–43. doi: 10.1007/978-1-4939-1050-2_3. [DOI] [PubMed] [Google Scholar]
- 24.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 25.Pandurangan AK, Esa NM. Signal transducer and activator of transcription 3-a promising target in colitis-associated cancer. Asian Pac J Cancer Prev. 2014;15:551–560. doi: 10.7314/APJCP.2014.15.2.551. [DOI] [PubMed] [Google Scholar]
- 26.Lowenberg M, D'Haens G. Novel targets for inflammatory bowel disease therapeutics. Curr Gastroenterol Rep. 2013;15:311. doi: 10.1007/s11894-012-0311-3. [DOI] [PubMed] [Google Scholar]
- 27.Zhu H, Li YR. Oxidative stress and redox signaling mechanisms of inflammatory bowel disease: Updated experimental and clinical evidence. Exp Biol Med (Maywood) 2012;237:474–480. doi: 10.1258/ebm.2011.011358. [DOI] [PubMed] [Google Scholar]
- 28.Wang A, Keita AV, Phan V, McKay CM, Schoultz I, Lee J, Murphy MP, Fernando M, Ronaghan N, Balce D, et al. Targeting mitochondria-derived reactive oxygen species to reduce epithelial barrier dysfunction and colitis. Am J Pathol. 2014;184:2516–2527. doi: 10.1016/j.ajpath.2014.05.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mouzaoui S, Djerdjouri B, Makhezer N, Kroviarski Y, El-Benna J, Dang PM. Tumor necrosis factor-α-induced colitis increases NADPH oxidase 1 expression, oxidative stress, and neutrophil recruitment in the colon: Preventive effect of apocynin. Mediators Inflamm. 2014;2014:312484. doi: 10.1155/2014/312484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fernandes CG, da Rosa MS, Seminotti B, Pierozan P, Martell RW, Lagranha VL, Busanello EN, Leipnitz G, Wajner M. In vivo experimental evidence that the major metabolites accumulating in 3-hydroxy-3-methylglutaryl-CoA lyase deficiency induce oxidative stress in striatum of developing rats: A potential pathophysiological mechanism of striatal damage in this disorder. Mol Genet Metab. 2013;109:144–153. doi: 10.1016/j.ymgme.2013.03.017. [DOI] [PubMed] [Google Scholar]
- 31.Goes AC, Pinto FM, Fernandes GC, Barbosa JS, Correia ES, Ribeiro RA, Guimaraes SB, Lima Junior RC, Brito GA, Rodrigues LV. Electroacupuncture ameliorates experimental colitis induced by TNBS through activation of interleukin-10 and inhibition of iNOS in mice. Acta Cir Bras. 2014;29:787–793. doi: 10.1590/S0102-86502014001900004. [DOI] [PubMed] [Google Scholar]
- 32.Mouzaoui S, Rahim I, Djerdjouri B. Aminoguanidine and curcumin attenuated tumor necrosis factor (TNF)-α-induced oxidative stress, colitis and hepatotoxicity in mice. Int Immunopharmacol. 2012;12:302–311. doi: 10.1016/j.intimp.2011.10.010. [DOI] [PubMed] [Google Scholar]
- 33.Park SY, Neupane GP, Lee SO, Lee JS, Kim MY, Kim SY, Park BC, Park YJ, Kim JA. Protective effects of pogostemon cablin bentham water extract on inflammatory cytokine expression in TNBS-induced colitis in rats. Arch Pharm Res. 2014;37:253–262. doi: 10.1007/s12272-013-0260-x. [DOI] [PubMed] [Google Scholar]
- 34.Leppkes M, Roulis M, Neurath MF, Kollias G, Becker C. Pleiotropic functions of TNF-α in the regulation of the intestinal epithelial response to inflammation. Int Immunology. 2014;26:509–515. doi: 10.1093/intimm/dxu051. [DOI] [PubMed] [Google Scholar]
- 35.Chalaris A, Garbers C, Rabe B, Rose-John S, Scheller J. The soluble Interleukin 6 receptor: Generation and role in inflammation and cancer. Eur J Cell Biol. 2011;90:484–494. doi: 10.1016/j.ejcb.2010.10.007. [DOI] [PubMed] [Google Scholar]
- 36.Moriasi C, Subramaniam D, Awasthi S, Ramalingam S, Anant S. Prevention of colitis-associated cancer: Natural compounds that target the IL-6 soluble receptor. Anticancer Agents Med Chem. 2012;12:1221–1238. doi: 10.2174/187152012803833080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bryan HK, Olayanju A, Goldring CE, Park BK. The Nrf2 cell defence pathway: Keap1-dependent and -independent mechanisms of regulation. Biochem Pharmacol. 2013;85:705–717. doi: 10.1016/j.bcp.2012.11.016. [DOI] [PubMed] [Google Scholar]
- 38.Araujo JA, Zhang M, Yin F. Heme oxygenase-1, oxidation, inflammation, and atherosclerosis. Front Pharmacol. 2012;3:119. doi: 10.3389/fphar.2012.00119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Durante W. Protective role of heme oxygenase-1 against inflammation in atherosclerosis. Front Biosci (Landmark Ed) 2011;16:2372–2388. doi: 10.2741/3860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gao Z, Han Y, Hu Y, Wu X, Wang Y, Zhang X, Fu J, Zou X, Zhang J, Chen X, et al. Targeting HO-1 by epigallocatechin-3-gallate reduces contrast-induced renal injury via anti-oxidative stress and anti-inflammation pathways. PloS One. 2016;11:e0149032. doi: 10.1371/journal.pone.0149032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cheng HT, Yen CJ, Chang CC, Huang KT, Chen KH, Zhang RY, Lee PY, Miaw SC, Huang JW, Chiang CK, et al. Ferritin heavy chain mediates the protective effect of heme oxygenase-1 against oxidative stress. Biochim Biophys Acta. 2015;1850:2506–2517. doi: 10.1016/j.bbagen.2015.09.018. [DOI] [PubMed] [Google Scholar]
- 42.Wagner AE, Will O, Sturm C, Lipinski S, Rosenstiel P, Rimbach G. DSS-induced acute colitis in C57BL/6 mice is mitigated by sulforaphane pre-treatment. J Nutr Biochem. 2013;24:2085–2091. doi: 10.1016/j.jnutbio.2013.07.009. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated and analyzed during the study are available from the corresponding author on reasonable request.