Highlights
-
•
Static magnetic field stimulation was delivered on cervical spinal cord.
-
•
Trans-spinal static magnetic field stimulation (tsSMS) decreased MEP amplitudes.
-
•
The suppressive effect of tsSMS was not maintained after the intervention ceased.
Abbreviations: tsSMS, transspinal static magnetic stimulation; tSMS, transcranial static magnetic stimulation; MEP, motor evoked potential; FDI, first dorsal interosseous; TMS, transcranial magnetic stimulation; M1, primary motor cortex; EMG, electromyography
Keywords: Static magnetic field, Spinal cord, Motor evoked potential, Neuromodulation
Abstract
Objective
Transcranial static magnetic field stimulation has recently been demonstrated to modulate cortical excitability. In the present study, we investigated the effect of transspinal static magnetic field stimulation (tsSMS) on excitability of the corticospinal tract.
Methods
A compact magnet for tsSMS (0.45 Tesla) or a stainless steel cylinder for sham stimulation was positioned over the neck (C8 level) of 24 able-bodied subjects for 15 min. Using 120% of the resting motor threshold transcranial magnetic stimulation intensity, motor evoked potentials (MEPs) were measured from the first digital interosseous muscle before, during, and after the tsSMS or sham intervention.
Results
Compared with baseline MEP amplitudes were decreased during tsSMS, but not during sham stimulation. Additionally, during the intervention, MEP amplitudes were lower with tsSMS than sham stimulation, although these effects did not last after the intervention ceased.
Conclusions
The results suggest that static magnetic field stimulation of the spinal cord by a compact magnet can reduce the excitability of the corticospinal tract.
Significance
Transspinal static magnetic field stimulation may be a new non-invasive neuromodulatory tool for spinal cord stimulation. Its suppressive effect may be applied to patients who have pathological hyperexcitability of the spinal neural network.
1. Introduction
It has recently been proposed that spinal cord stimulation can be an effective tool for the neurorehabilitation of spinal cord injury patients. For example, invasive methods such as epidural spinal cord stimulation have been demonstrated to facilitate voluntary limb movement as well as standing and walking in spinal cord injury patients (Herman et al., 2002, Harkema et al., 2011, Angeli et al., 2014). However, since epidural stimulation is an invasive method, there is considerable need to develop more non-invasive spinal cord stimulation methods. In this study, we developed and examined a new non-invasive spinal cord stimulation tool that utilizes static magnetic fields.
Local applications of static magnetic fields to the human cortex modulates cortical excitability (Oliviero et al., 2011, Kirimoto et al., 2014, Gonzalez-Rosa et al., 2015, Nojima et al., 2015, Kirimoto et al., 2016a, Kirimoto et al., 2016b, Nojima et al., 2016). Oliviero et al. (2011) were the first to report that transcranial static magnetic field stimulation (tSMS) using small, strong magnets over the human motor cortex decreased excitability of the motor cortex. Following their report, other studies have also shown similar suppressive effects on the somatosensory cortex (Kirimoto et al., 2014) and visual cortex (Gonzalez-Rosa et al., 2015). For instance, Gonzalez-Rosa et al. (2015) demonstrated that tSMS applied to the visual cortex impaired the performance of subjects in a visual searching task. Conversely, Carrasco-Lopez et al. (2017) showed that tSMS over the parietal cortex improved the detection of somatosensory stimuli. These two studies suggested that tSMS can alter not only cortical excitability, but also human behavior. The safety of tSMS has been assessed, and prolonged tSMS (2 h) does not appear to produce neural damage (Oliviero et al., 2015). However, the above studies suggested that tSMS cannot increase neural excitability; instead, it only has suppressive effects. Indeed, stimuli from both the north and south poles of magnets suppress neural excitability (Oliviero et al., 2011). Conversely, a recent study reported no modulatory effect of tSMS over the motor and somatosensory cortices (Kufner et al., 2017). As tSMS is relatively new and has only been applied to humans for 6–7 years, more evidence regarding the effects and mechanisms of static magnetic field stimulation is necessary. In particular, previous studies have applied tSMS to the cortices, but not the spinal cord.
A recent study utilizing direct current stimulation (DCS) indicated that not only transcranial tDCS but also transspinal DCS modulates corticospinal excitability (Bocci et al., 2015c). Therefore, it is possible that static magnetic field stimulation over the spinal cord may modulate corticospinal excitability. In the present study, we investigated the effects of transspinal static magnetic field stimulation (tsSMS) applied over the cervical spinal cord on corticospinal excitability. As most previous tSMS studies have shown only suppressive effects of tSMS, we hypothesized that tsSMS would decrease corticospinal excitability. As a preliminary step toward clinical applications for patients, we assessed able-bodied individuals in the present study.
2. Methods
This experiment was a sham-controlled study with a crossover design.
2.1. Subjects
Twenty-four able-bodied adults participated in this experiment (23 men and 1 woman, mean age 26 ± 3 years). The study was approved by the Ethics Committee of the Graduate School of Arts and Sciences, the University of Tokyo, and informed consent was obtained from all subjects. None of the subjects had prior knowledge of the effects of static magnetic fields on neural circuits or had previously received static magnetic field stimulation.
2.2. Transcranial magnetic stimulation (TMS) measurement
During the experiment, subjects sat in a chair in a fixed prone position with their forearm on an armrest. The effects of tsSMS on corticospinal excitability were evaluated by measuring the amplitude of motor evoked potentials (MEPs) recorded from the right first dorsal interosseous (FDI) muscle. MEPs were induced by TMS delivered to the primary motor cortex (M1). Single-pulse TMS was applied using a figure-of-eight coil (70-mm diameter for each coil) connected to a magnetic stimulator (Rapid2, Magstim, U.K.). The coil was positioned on the left M1 of the finger muscle area. The coil position was determined by searching for the point where MEPs were obtained from the FDI muscle. The resting motor threshold was defined as the lowest TMS intensity that elicited more than five MEPs greater than 50 μV in the FDI muscle in ten stimuli. FDI MEPs were recorded at 120% of this resting motor threshold for each participant. TMS measurements were performed before (pre), 5 min, and 10 min after the start of the tsSMS and sham intervention (during-5 and during-10 respectively), and immediately after (post-0) and 5 min after (post-5) the end of the intervention. Twenty MEPs were recorded in each measurement session, with subjects remaining relaxed between measurement sessions.
Electromyographic (EMG) signals were recorded with bipolar Ag/AgCl surface electrodes (Vitrode F-150S, 18 × 36 mm, Nihon Kohden, Tokyo, Japan), amplified (MEB-6108 amplifier, Nihon Kohden, Tokyo, Japan), and filtered with a band pass filter of 5–1500 Hz. The signals were transferred to an A/D converter (Power lab, AD Instruments, Australia) at 4000 Hz and stored on a computer.
2.3. TsSMS intervention
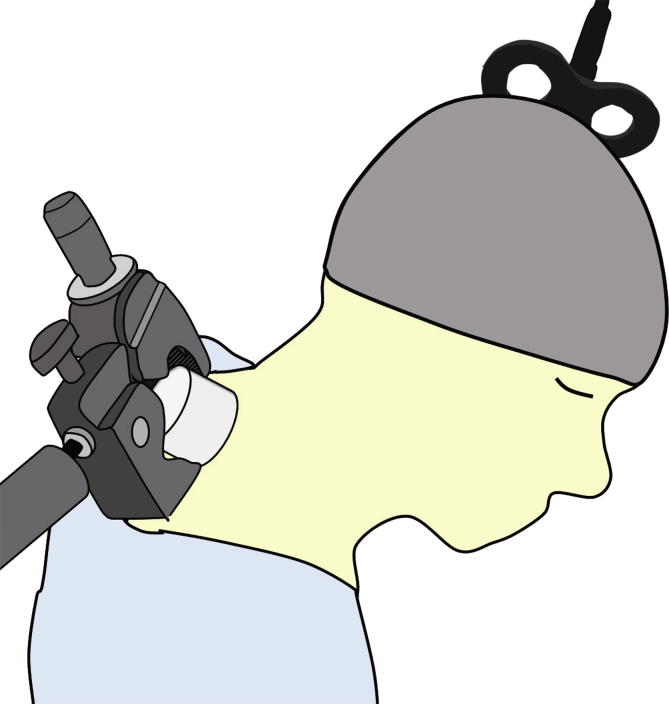
To obtain static magnetic fields, we utilized a cylindrical neodymium magnet (NdFeb; diameter 50 mm, length 30 mm) with a maximum magnetic flux density of 0.459 Tesla (NeoMag, Ichikawa, Japan), which is the same type used in some previous studies (Kirimoto et al., 2014, Nojima et al., 2015, Kirimoto et al., 2016a, Nojima et al., 2016). For sham stimulation, we used a non-magnetic stainless steel cylinder of the same size and weight. The magnet and stainless steel cylinder were set on the skin of the participant’s neck (over the C8 vertebra) using a stand with a movable arm (C-stand, Avenger, Cassola, Italy). As it has been reported that the suppressive effects of static magnetic field stimulation are not polarity-dependent (Oliviero et al., 2011), we selected only the north polarity for stimulation. During the entire experimental session, subjects maintained a slight neck flexion (Fig. 1). The duration of the magnet or sham stimulation was 15 min, and the subjects were asked to relax during the intervention and TMS measurements. The 15 min duration was chosen as the median value of the durations used in previous tSMS studies, which ranged from 10 to 20 min (10 min: Gonzalez-Rosa et al., 2015, 15 min: Kirimoto et al., 2014, Kirimoto et al., 2016a, Kirimoto et al., 2016b, 20 min: Oliviero et al., 2011, Nojima et al., 2015).
Fig. 1.
Experimental setup. A cylindrical neodymium magnet for tsSMS or a stainless cylinder of the same size and appearance for sham stimulation, was positioned on the subject’s neck over C8 during the intervention step. They were fixed in place by a movable arm on a stand. MEPs from the FDI muscle were recorded by TMS applied over the left M1 cortex.
The experiment was performed using a double-blind design as follows. Experimenter 1 decided whether the real magnet or sham stainless steel cylinder would be utilized, and fixed it to the stand. Experimenter 2, who was blinded as to whether the intervention was real or sham, recorded the MEPs and analyzed the data. All subjects participated in two experimental sessions (tsSMS and sham stimulation) on separate days. The experimental days were at least three days apart.
To estimate the affected intensity of the static magnetic field applied in the present study, we calculated the distance between the neck surface and spinal cord using a 3 Tesla magnetic resonance imaging system (MRI: MAGNETOM Prisma 3 T, Siemens) in three subjects. Participants flexed their neck to the same extent as for the MEP measurements.
2.4. Data analysis
The amplitudes of MEPs (peak-to-peak) were calculated and normalized with respect to MEPs measured before the intervention. We also measured background EMG signals in a 50 ms window just before the TMS was delivered. If a single data point accounted for more than 20 μV of the amplitude in the window, the trials were rejected from the data analysis. This rejection criteria was similar to that used in some previous studies that measured MEPs from resting muscles (Uematsu et al., 2010, Mizuguchi et al., 2011). When the rejection ratio was greater than 10% for the whole trial, we rejected the entire dataset for the subject. In addition, we calculated the root mean square in the window as the magnitude of background EMG.
To assess the differences between the MEP amplitudes and background EMG between conditions, we performed a two-way repeated measures ANOVA (two conditions [tsSMS and sham stimulation] × five times [pre, during-5, during-10, post-0, and post-5]). When a significant main effect or interaction was found, post hoc analysis with Bonferroni’s correction was performed. The MEP amplitude was expressed as mean ± standard error. Threshold for statistical significance set to 0.05.
3. Results
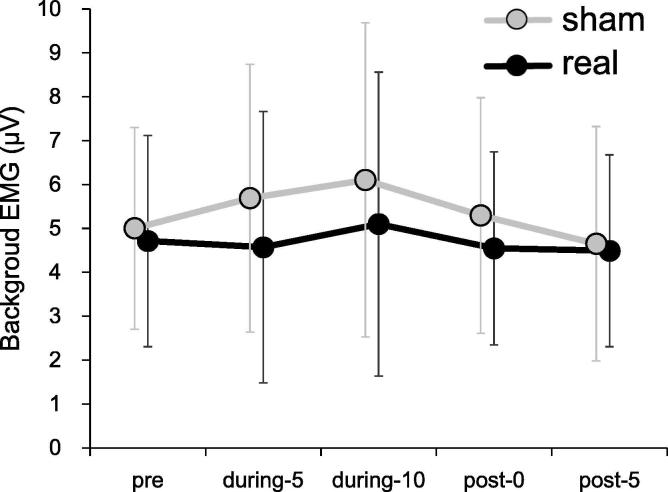
The resting motor threshold in FDI muscle was 69.4 ± 10.2% of the maximal output of the magnetic stimulator with tsSMS and 68.4 ± 11.2% in the sham condition, with no significant difference between the conditions (p = 0.42). In four subjects, many trials had to be rejected due to high background EMG level (range: 17.5–25.5% rejection in the entire trials). We judged that these four subjects were not able to maintain the resting condition and so all their trials were rejected from the statistical analysis. Of the remaining 20 subjects, data in 1.3% of all individual trials (51 trials) were excluded from data analysis due to the appearance of background EMG activity just before TMS application. After removal of trials with high EMG activity, assessment of the magnitude of the background EMG using a two-way repeated measures ANOVA did not detect significant main effect of condition (F(1, 19) = 1.78, p = 0.20) and time (F(1.71, 32.41) = 2.85, p = 0.08) or interaction (F(4, 76) = 0.94, p = 0.45) (Fig. 2).
Fig. 2.
Time-course of the background EMG. No significant differences were observed. Error bars represent standard deviation.
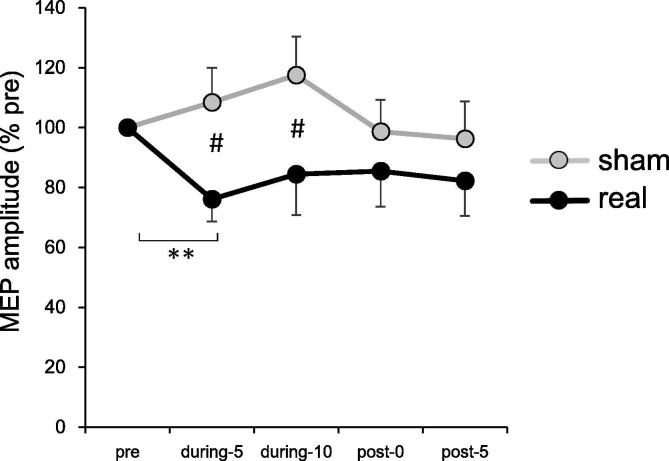
Fig. 3 shows the normalized MEP amplitude of FDI muscle with tsSMS and in the sham conditions. A two-way repeated measures ANOVA showed a significant main effect of condition (F(1, 19) = 6.79, p = 0.02) and interaction (F(4, 76) = 3.05, p = 0.02), but no significant main effect of time (F(4, 76) = 1.79, p = 0.14). Post-hoc Bonferroni tests indicated that the MEP amplitude in the tsSMS condition was significantly smaller than that in the sham condition at the during-5 (p = 0.02, t (19) = 3.14) and during-10 (p = 0.01, t (19) = 3.44) timepoints. Furthermore, we found a significant decrease in the MEP amplitude at the during-5 timepoint compared with the pre-condition (p = 0.0014, t (19) = 4.35).
Fig. 3.
Changes in normalized MEP amplitude before (pre), 5 min (during-5), and 10 min (during-10) after the start of the intervention, and immediately after (post-0) and 5 min after the end of the intervention (post-5). The asterisks (**) represent a significant difference between the pre and during-5 timepoints in the tsSMS condition (p < 0.01). The sharp symbol (#) represents significant differences between tsSMS and the sham condition at during-5 and during-10 timepoints (p < 0.05). Error bars are standard error.
The average distance between the skin and spinal cord was 4.5 cm (range: 4.2–4.7 cm).
4. Discussion
The aim of the present study was to investigate the effect of tsSMS applied over the cervical spinal cord (C8) on corticospinal excitability measured from a finger muscle. Our main findings were: 1) MEP amplitudes in the FDI muscle decreased during tsSMS compared with the pre-intervention and sham conditions, and 2) the effects of tsSMS were not prominently observed after stimulation ceased. Additionally, the MEP amplitude was not significantly modulated in the sham condition, indicating that application of the non-magnetic stainless steel cylinder did not affect neural excitability, unlike tsSMS.
We observed a significant reduction in MEPs during but not after tsSMS. This was in contrast to previous reports showing that the suppressive effects of static magnetic field stimulation of the cortex persist for a few minutes (Oliviero et al., 2011, Kirimoto et al., 2014). It may be that the static magnetic field applied over the spinal cord in the present condition was not strong enough to produce sustained suppression. However, a previous study utilizing 1.5 and 7 Tesla MRI scanners suggested that the degree of the inhibitory effect on the motor cortex does not depend on the strength of the static magnetic field (Schlamann et al., 2010). Thus, stronger stimulation may not have elicited different effects.
It is well known that the intensity of a static magnetic field dramatically decreases with increasing distance from the magnet (Paulus, 2011, Rivadulla et al., 2014, Kirimoto et al., 2016b). For example, the intensity of the magnet axis was approximately 0.48 Tesla at the magnet surface (distance 0 cm), whereas it would drop to 0.15 Tesla at a distance of 2 cm (Paulus, 2011). As the distance between the scalp and the primary motor cortex is about 2 cm (Stokes et al., 2005, Oliviero et al., 2011), this distance should be enough to allow modulation of neural excitability (Oliviero et al., 2011). Indeed, alteration of EMG in rabbits was seen with a magnetic field of only 20 mTesla (Kholodov, 1974). Moreover, in an in vitro study, a 125 mTesla static magnetic field reduced the voltage of activated sodium channels (Rosen, 2003a). As a supplementary measurement, we used MRI of three subjects to determine that the average distance between the neck surface and spinal cord was 4.5 cm. The distance to the cortex, which was the target of previous human studies, is about 2 cm (Stokes et al., 2005, Oliviero et al., 2011). Thus, the present study may be the first human study to show that neural excitability can be modulated even at a distance of 4–5 cm. At this distance, a static magnetic field of around 50 mTesla would likely act on the spinal cord (Kirimoto et al., 2016a, Kirimoto et al., 2016b). Although that is much weaker than normally used in human cortical studies, many animal or in vitro studies have confirmed that such weak static magnetic fields can alter neural excitability or function (Kholodov, 1974, Ben Yakir-Blumkin et al., 2014). Thus, we expect that the magnetic intensity in the present study was sufficient to transiently modulate corticospinal excitability. Methodologically, if a stimulation distance of 4–5 cm between the magnet and target neural circuits can elicit neuromodulation, it may be possible to apply static magnetic field stimulation for deeper brain regions such as the lower limb area of M1 or the cingulate cortex.
In the present study, we measured only MEP data, which reflects both cortical and spinal neural excitability. Previous studies have shown that trans-spinal direct current stimulation can also modulate cortical neural circuits (Bocci et al., 2015a, Bocci et al., 2015b), in addition to spinal cord excitability (Lamy et al., 2012, Hubli et al., 2013). Thus, it is reasonable to assume that tsSMS would affect the motor cortex in addition to the spinal cord. However, as it has been proposed that static magnetic fields can suppress membrane excitability (Rosen, 2003b), the suppressive effect of tsSMS could also have been caused by non-synaptic changes. Consequently, any effect produced in the spinal neurons would not ascend to the cortices through the synapses. If this is the case, then the question is how tsSMS affects spinal cord excitability. Previous studies utilizing animal and cellular models have proposed that static magnetic fields can alter membrane ion channel function (Rosen, 2003a, Coots et al., 2004). It is therefore possible that in the present study, ion channel function in the motor neuron membranes may have been modulated, resulting in depression of the MEPs. On the other hand, Coots et al. (2004) showed that non-synaptic axonal excitability in mammalian spinal cord decreased during exposure to a static magnetic field, with probable conduction blocks in some small nerve fibers. This may mean that in addition to modulation of motor neuron excitability, excitability in corticospinal tract fibers just before their connection to motor neurons may have also been depressed. Such mechanisms would result in a reduction in the total descending impulses reaching the muscle.
This study has several limitations. First, we did not assess the gain function of corticospinal excitability. The aftereffect of static magnetic field stimulation has been estimated to be short (Oliviero et al., 2011, Kirimoto et al., 2014, Nojima et al., 2015). Indeed, we found the aftereffect to be very short or absent, and so we did not have sufficient time to record a large number of MEPs. Thus, we measured MEPs at only one stimulus intensity level. Second, we did not perform functional motor or sensory tests. The effects of the static magnetic field intervention on behavioral indices have been confirmed in animal studies (Saunders, 2005). Even in humans, the static magnetic field stimulation can alter sensory function (Gonzalez-Rosa et al., 2015, Carrasco-Lopez et al., 2017). Thus, it is possible that tsSMS can also modify human motor or sensory function. This should be examined in future work. Furthermore, we asked the subjects to hold their necks in a flexion position during the trials. A previous study indicated that spinal cord excitability decreases during neck flexion (Bulbulian et al., 2002). Therefore, data in the present study might have been biased by the effect of neck flexion. However, if this were the case, the effect would be the same between the different conditions and time-points, because the position was held constant. Therefore, the observed differences elicited by the intervention do not appear to substantially depend on neck flexion.
We demonstrated that tsSMS can suppress corticospinal excitability and thus has potential as a new non-invasive tool for spinal cord stimulation. The observed suppressive effect could possibly be applied to spinal cord injury, stroke, or multiple sclerosis patients who have pathological hyperexcitability of the spinal neural network. For example, it is possible that involuntary spastic muscle contraction due to spinal cord injury could be relieved by tsSMS. However, since static magnetic field stimulation induces only suppression of the motor system (Oliviero et al., 2011, Nojima et al., 2015), tSMS might not be useful for able-bodied people who want to improve motor behaviors, such as building muscle strength. A recent study reported that tSMS over the parietal cortex facilitated somatosensory detection (Carrasco-Lopez et al., 2017). Thus, tSMS may be able to facilitate systems other than the motor system in both patients and able-bodied people. Unlike epidural spinal cord stimulation, which has been utilized as a strong neuromodulatory tool (Gerasimenko et al., 2008), tsSMS is not likely to be effective for increasing neural activity. Previous studies have shown the efficacy of epidural stimulation in facilitating voluntary movements, such as standing and walking in patients with spinal cord injury (Dimitrijevic et al., 1998, Herman et al., 2002, Minassian et al., 2004, Gerasimenko et al., 2008, Harkema et al., 2011). Thus, clinical interventions involving tsSMS appear to have different mechanisms from those implicated in epidural stimulation. However, although tsSMS itself is not be expected to induce long-term plasticity, simultaneous use of tsSMS and motor training or other neuromodulatory techniques might induce long-term plastic changes in motor system neural networks. This could be a promising direction for neurorehabilitation research. Moreover, since the neodymium magnet is inexpensive and does not require specific operational skills, it may be easy to utilize tsSMS for neurorehabilitation of corticospinal excitability within the home.
5. Conclusion
Static magnetic field stimulation applied over the cervical spinal cord decreased corticospinal excitability in the finger muscle of able-bodied subjects. Trans-spinal static magnetic field stimulation could be a new non-invasive neuromodulatory tool for the corticospinal tract.
Acknowledgments
This study was supported by Grants from the Magnetic Health and Science Foundation, JSPS KAKENHI Grant Number 15J09559 and 26242056.
Conflicts of interest
None.
References
- Angeli C.A., Edgerton V.R., Gerasimenko Y.P., Harkema S.J. Altering spinal cord excitability enables voluntary movements after chronic complete paralysis in humans. Brain. 2014;137:1394–1409. doi: 10.1093/brain/awu038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben Yakir-Blumkin M., Loboda Y., Schachter L., Finberg J.P. Neuroprotective effect of weak static magnetic fields in primary neuronal cultures. Neuroscience. 2014;278:313–326. doi: 10.1016/j.neuroscience.2014.08.029. [DOI] [PubMed] [Google Scholar]
- Bocci T., Barloscio D., Vergari M., Di Rollo A., Rossi S., Priori A. Spinal direct current stimulation modulates short intracortical inhibition. Neuromodulation. 2015;18:686–693. doi: 10.1111/ner.12298. [DOI] [PubMed] [Google Scholar]
- Bocci T., Caleo M., Vannini B., Vergari M., Cogiamanian F., Rossi S. An unexpected target of spinal direct current stimulation: Interhemispheric connectivity in humans. J. Neurosci. Methods. 2015;254:18–26. doi: 10.1016/j.jneumeth.2015.07.012. [DOI] [PubMed] [Google Scholar]
- Bocci T., Marceglia S., Vergari M., Cognetto V., Cogiamanian F., Sartucci F. Transcutaneous spinal direct current stimulation modulates human corticospinal system excitability. J. Neurophysiol. 2015;114:440–446. doi: 10.1152/jn.00490.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bulbulian R., Burke J., Dishman J.D. Spinal reflex excitability changes after lumbar spine passive flexion mobilization. J. Manipulative Physiol. Ther. 2002;25:526–532. doi: 10.1067/mmt.2002.127073. [DOI] [PubMed] [Google Scholar]
- Carrasco-Lopez C., Soto-Leon V., Cespedes V., Profice P., Strange B.A., Foffani G. Static magnetic field stimulation over parietal cortex enhances somatosensory detection in humans. J. Neurosci. 2017;37:3840–3847. doi: 10.1523/JNEUROSCI.2123-16.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coots A., Shi R., Rosen A.D. Effect of a 0.5-T static magnetic field on conduction in guinea pig spinal cord. J. Neurol. Sci. 2004;222:55–57. doi: 10.1016/j.jns.2004.04.010. [DOI] [PubMed] [Google Scholar]
- Dimitrijevic M.R., Gerasimenko Y., Pinter M.M. Evidence for a spinal central pattern generator in humans. Ann. N. Y. Acad. Sci. 1998;860:360–376. doi: 10.1111/j.1749-6632.1998.tb09062.x. [DOI] [PubMed] [Google Scholar]
- Gerasimenko Y., Roy R.R., Edgerton V.R. Epidural stimulation: comparison of the spinal circuits that generate and control locomotion in rats, cats and humans. Exp. Neurol. 2008;209:417–425. doi: 10.1016/j.expneurol.2007.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Rosa J.J., Soto-Leon V., Real P., Carrasco-Lopez C., Foffani G., Strange B.A. Static magnetic field stimulation over the visual cortex increases alpha oscillations and slows visual search in humans. J. Neurosci. 2015;35:9182–9193. doi: 10.1523/JNEUROSCI.4232-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harkema S., Gerasimenko Y., Hodes J., Burdick J., Angeli C., Chen Y. Effect of epidural stimulation of the lumbosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: a case study. Lancet. 2011;377:1938–1947. doi: 10.1016/S0140-6736(11)60547-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herman R., He J., D'Luzansky S., Willis W., Dilli S. Spinal cord stimulation facilitates functional walking in a chronic, incomplete spinal cord injured. Spinal Cord. 2002;40:65–68. doi: 10.1038/sj.sc.3101263. [DOI] [PubMed] [Google Scholar]
- Hubli M., Dietz V., Schrafl-Altermatt M., Bolliger M. Modulation of spinal neuronal excitability by spinal direct currents and locomotion after spinal cord injury. Clin. Neurophysiol. 2013;124:1187–1195. doi: 10.1016/j.clinph.2012.11.021. [DOI] [PubMed] [Google Scholar]
- Kholodov YA. Influence of magnetic fields on biological objects. NTIS Rep 1974; JPRS 63038.
- Kirimoto H., Asao A., Tamaki H., Onishi H. Non-invasive modulation of somatosensory evoked potentials by the application of static magnetic fields over the primary and supplementary motor cortices. Sci. Rep. 2016;6:34509. doi: 10.1038/srep34509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirimoto H., Tamaki H., Matsumoto T., Sugawara K., Suzuki M., Oyama M. Effect of transcranial static magnetic field stimulation over the sensorimotor cortex on somatosensory evoked potentials in humans. Brain Stimul. 2014;7:836–840. doi: 10.1016/j.brs.2014.09.016. [DOI] [PubMed] [Google Scholar]
- Kirimoto H., Tamaki H., Onishi H. Transcranial static magnetic field stimulation – new non-invasive brain stimulation tool. J. Phys. Fitness Sports Med. 2016;5:205–211. [Google Scholar]
- Kufner M., Bruckner S., Kammer T. No modulatory effects by transcranial static magnetic field stimulation of human motor and somatosensory cortex. Brain Stimul. 2017;10:703–710. doi: 10.1016/j.brs.2017.03.001. [DOI] [PubMed] [Google Scholar]
- Lamy J.C., Ho C., Badel A., Arrigo R.T., Boakye M. Modulation of soleus H reflex by spinal DC stimulation in humans. J. Neurophysiol. 2012;108:906–914. doi: 10.1152/jn.10898.2011. [DOI] [PubMed] [Google Scholar]
- Minassian K., Jilge B., Rattay F., Pinter M.M., Binder H., Gerstenbrand F. Stepping-like movements in humans with complete spinal cord injury induced by epidural stimulation of the lumbar cord: electromyographic study of compound muscle action potentials. Spinal Cord. 2004;42:401–416. doi: 10.1038/sj.sc.3101615. [DOI] [PubMed] [Google Scholar]
- Mizuguchi N., Sakamoto M., Muraoka T., Nakagawa K., Kanazawa S., Nakata H. The modulation of corticospinal excitability during motor imagery of actions with objects. PLoS One. 2011;6:e26006. doi: 10.1371/journal.pone.0026006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nojima I., Koganemaru S., Fukuyama H., Mima T. Static magnetic field can transiently alter the human intracortical inhibitory system. Clin. Neurophysiol. 2015;126:2314–2319. doi: 10.1016/j.clinph.2015.01.030. [DOI] [PubMed] [Google Scholar]
- Nojima I., Koganemaru S., Mima T. Combination of static magnetic fields and peripheral nerve stimulation can alter focal cortical excitability. Front Hum Neurosci. 2016;10:598. doi: 10.3389/fnhum.2016.00598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliviero A., Carrasco-Lopez M.C., Campolo M., Perez-Borrego Y.A., Soto-Leon V., Gonzalez-Rosa J.J. Safety study of transcranial static magnetic field stimulation (tSMS) of the human cortex. Brain Stimul. 2015;8:481–485. doi: 10.1016/j.brs.2014.12.002. [DOI] [PubMed] [Google Scholar]
- Oliviero A., Mordillo-Mateos L., Arias P., Panyavin I., Foffani G., Aguilar J. Transcranial static magnetic field stimulation of the human motor cortex. J. Physiol. 2011;589:4949–4958. doi: 10.1113/jphysiol.2011.211953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulus W. Transcranial static magnetic field stimulation in man: making things as simple as possible? J. Physiol. 2011;589:5917–5918. doi: 10.1113/jphysiol.2011.221655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rivadulla C., Foffani G., Oliviero A. Magnetic field strength and reproducibility of neodymium magnets useful for transcranial static magnetic field stimulation of the human cortex. Neuromodulation. 2014;17:438–441. doi: 10.1111/ner.12125. discussion 41-2. [DOI] [PubMed] [Google Scholar]
- Rosen A.D. Effect of a 125 mT static magnetic field on the kinetics of voltage activated Na+ channels in GH3 cells. Bioelectromagnetics. 2003;24:517–523. doi: 10.1002/bem.10124. [DOI] [PubMed] [Google Scholar]
- Rosen A.D. Mechanism of action of moderate-intensity static magnetic fields on biological systems. Cell Biochem. Biophys. 2003;39:163–173. doi: 10.1385/CBB:39:2:163. [DOI] [PubMed] [Google Scholar]
- Saunders R. Static magnetic fields: animal studies. Prog. Biophys. Mol. Biol. 2005;87:225–239. doi: 10.1016/j.pbiomolbio.2004.09.001. [DOI] [PubMed] [Google Scholar]
- Schlamann M., Yoon M.S., Maderwald S., Pietrzyk T., Bitz A.K., Gerwig M. Short term effects of magnetic resonance imaging on excitability of the motor cortex at 1.5T and 7T. Acad Radiol. 2010;17:277–281. doi: 10.1016/j.acra.2009.10.004. [DOI] [PubMed] [Google Scholar]
- Stokes M.G., Chambers C.D., Gould I.C., Henderson T.R., Janko N.E., Allen N.B. Simple metric for scaling motor threshold based on scalp-cortex distance: application to studies using transcranial magnetic stimulation. J. Neurophysiol. 2005;94:4520–4527. doi: 10.1152/jn.00067.2005. [DOI] [PubMed] [Google Scholar]
- Uematsu A., Obata H., Endoh T., Kitamura T., Hortobagyi T., Nakazawa K. Asymmetrical modulation of corticospinal excitability in the contracting and resting contralateral wrist flexors during unilateral shortening, lengthening and isometric contractions. Exp. Brain Res. 2010;206:59–69. doi: 10.1007/s00221-010-2397-x. [DOI] [PubMed] [Google Scholar]