Abstract
Purpose
The main aim of our study was to evaluate the benefit of the use of non-apoptotic spermatozoa selected by magnetic-activated cell sorting (MACS) for ICSI procedures for couples in which the women had good prognoses and the male factor of infertility was teratozoospermia.
Methods
Twenty-six couples were treated with ICSI after MACS selection of non-apoptotic spermatozoa following a sibling oocyte approach. Half of the oocytes were microinjected with conventionally prepared spermatozoa, and the other half were microinjected with non-apoptotic, MACS-selected spermatozoa. To assess the influence of MACS selection of spermatozoa on the outcomes of the ICSI cycles, the fertilization, embryo quality, pregnancy, and delivery rates were evaluated and compared between the sibling oocyte groups.
Results
When subpopulations of couples according to female age were analyzed, a significant difference in quality of blastocyst was observed. More precisely, in a group that was treated with MACS-ICSI, a higher percentage of good quality blastocysts was found among women older than 30 years (75.0 vs. 33.3%; P = 0.028), while there was no difference among younger women. If all included couples were compared regardless of age, no significant difference was observed in the outcome of the ICSI/MACS-ICSI cycles in terms of oocytes and embryos. Additionally, after the ICSI and MACS-ICSI procedures, the morphologies of the prepared spermatozoa were compared. Results showed that the overall percentage of morphologically normal spermatozoa did not differ significantly between the ICSI and MACS-ICSI procedures. However, detailed analyses of the morphologically abnormal spermatozoa revealed significantly more spermatozoa with abnormal tails after MACS-ICSI procedure, which may be potential consequence of the selection procedure. Moreover, the trends towards less spermatozoa with abnormal heads and towards more spermatozoa with abnormal necks and midpieces after MACS-ICSI procedure were revealed, although the differences were not significant.
Conclusions
Couples dealing with male infertility due to teratozoospermia can benefit from MACS selection of spermatozoa with higher percentage of good quality blastocysts but only when the woman is older than 30 years.
Keywords: MACS, Spermatozoa selection, Blastocyst, Spermatozoa morphology
Introduction
In IVF/ICSI, it is of exceptional importance to select the optimal spermatozoon for fertilization. However, in clinical practice, it is occasionally difficult to decide which spermatozoon is optimal because the final selection of the spermatozoon used for ICSI is usually based on the clinical embryologist’s assessment of sperm morphology. Although the assessment of sperm morphology is well described [1], this procedure remains somehow subjective and depends heavily on the experience of the clinical embryologist. Morphology-based sperm selection was recently elevated to its upper limits with the introduction of motile sperm organelle morphology examination into clinical practice. With this approach, it is possible to examine sperm heads under special contrast at high magnification and subsequently microinject the spermatozoa into oocytes (i.e., intracytoplasmic morphologically selected sperm injection (IMSI)) [2]. Substantial amounts of available data demonstrate that IMSI can significantly improve the outcomes of ICSI treatment [3–7]; however, some lack of clarity in this method remains because some literature opposes the benefits of such treatment [8–10]. The drawback of sperm selection techniques that are based on morphology assessment mainly results from the inability of these techniques to detect spermatozoa with impaired DNA integrity in terms of DNA fragmentation and denaturation and epigenetic changes. Some studies showed that IMSI/MSOME approach can improve the selection of spermatozoa with better genetic material in terms of DNA fragmentation [11–13] or molecular karyotype [14] to some extent, although other studies indicated that there is no correlation of IMSI/MSOME with improved DNA fragmentation of selected spermatozoa [9, 15]. Therefore, some approaches from some other perspectives that would also enable the selection of spermatozoa need to be introduced into clinical practice. One such approach is the selection of non-apoptotic spermatozoa with the aid of the magnetic labeling of apoptotic spermatozoa. When spermatozoa undergo the terminal phase of apoptosis, the phosphatidylserine is externalized on the outer side of plasma membrane and can be used as a biomarker or a target molecule for selection [16–18]. For selection purposes, a phospholipid binding protein annexin V can be utilized, since it covalently binds to phosphatidylserine, but it does not pass the plasma membrane of non-apoptotic cells [18]. Furthermore, annexin V is bound to microbeads, and this enables that spermatozoa, which bind to annexin V-microbead complexes, can be retained in separation column placed in magnetic field. The spermatozoa that pass this column do not express phosphatidylserine and are therefore characterized as non-apoptotic. Because the spermatozoa that are already programmed to undergo apoptosis or already in this process can still swim and appear normal, the only manner in which to avoid the selection of these spermatozoa for ICSI is their exclusion using the MACS approach. Some studies have demonstrated that, using this approach, it is possible to reduce the number of spermatozoa with fragmented DNA [19, 20], improve the spermatozoa acrosome reaction [21], improve the mitochondrial membrane potential [22], and increase the embryo implantation and pregnancy rates [23]. Additionally, just recently, Sanchez-Martin et al. [24] showed that MACS can lower miscarriage rate; however, as critically evaluated in a meta-analysis from a few years ago, despite the improved pregnancy rate, there are no significant improvements in the implantation or miscarriage rates [25]. No improvement in reproductive outcome after MACS was reported also by Romany et al. [26], although this study included unselected male population. Therefore, it is possible that these inconsistent results are a consequence of the different designs and populations of patients included in these studies. Because there are currently no sibling oocyte studies in which non-apoptotic MACS-selected spermatozoa have been used for fertilization and the results compared with those from non-selected spermatozoa, and because the populations of patients for whom this approach would be beneficial remain unclear, we performed a prospective study with a defined and selected population of patients. Thus, the main aim of our study was to evaluate the benefit of the use of MACS-selected non-apoptotic spermatozoa for ICSI procedures in couples in which the female had a good prognosis, and male infertility was due to teratozoospermia as defined by the strict Kruger criteria. To assess the influence of this sperm selection technique on the outcomes of ICSI cycles, fertilization, embryo quality, and the pregnancy and delivery rates were compared between sibling oocyte groups.
Materials and methods
Experimental design
This prospective sibling oocyte study was performed at our IVF center between April and December of 2016, and it was approved by the Slovenian National Medical Ethics Committee (no. 95/08/15). In this study, we included 26 couples who were treated with ICSI due to male infertility due to teratozoospermia (as defined by the strict Kruger criteria; i.e., below 15% of spermatozoa were assessed with normal morphology). The women in the couples included in this study were not older than 36 years and were required to have a normal ovarian response to controlled ovarian hyperstimulation (as defined by the presence of least six mature oocytes after ultrasound retrieval). For almost all of the couples, this was their first or second ICSI cycle; for only three couples, this was their fourth ICSI cycle. None of the included couples had a record of previous births or pregnancies. In each couple, one half of the mature oocytes was fertilized with ICSI using spermatozoa that were prepared with conventional sperm preparation method, and the second half was fertilized with ICSI using spermatozoa that were selected with the MACS procedure (MACS-ICSI).
Controlled ovarian hyperstimulation protocols
The women included in this study were treated with two different controlled ovarian hyperstimulation (COH) protocols, i.e., GnRH antagonist (23 women in their first or second ICSI cycle) and GnRH agonist protocols (3 women for whom this was their fourth ICSI cycle). The GnRH antagonist protocol began on day 2 of the menstrual cycle with an initial daily dose of 200–225-IU recombinant FSH (follitropin alfa, Gonal F, Merck Serono or follitropin beta, Puregon MSD). The GnRH antagonist cetrorelix acetate (Cetrotide; Asta Medica AG, Frankfurt, Germany) was administered at a dose of 0.25 mg per day to the women from the day on which the dominant follicle reached a mean diameter ≤ 14 mm until the day of HCG administration. HCG (Pregnyl; N.V. Organon, Oss, the Netherlands) was administered at a dose of 10,000 IU when the leading follicle reached a diameter of 18 mm. The GnRH agonist protocol began on day 22 of the menstrual cycle with the administration of the GnRH analogue (Suprefact; Hoechst AG, Frankfurt/Main, Germany) at a daily dose of 0.6 ml (600 pg) s.c. After 14 days, pituitary desensitization was examined by E2 determination and a B-mode ultrasound scan. Once the criteria for desensitization were fulfilled (i.e., E2 ≤ 0.05 nmol/L, follicles ≤ 5 mm in diameter, and an endometrial thickness ≤ 5 mm), ovarian stimulation with a daily dose of 200–225-IU rFSH was initiated (follitropin alfa, Gonal F Merck Serono or follitropin beta, Puregon MSD). The GnRH analogue administration was continued until the HCG administration. HCG was administered at a dose of 10,000 IU when the leading follicle reached a diameter of 20 mm. Oocyte retrieval was performed 36 h after HCG administration.
Semen quality evaluation (concentration, motility, and morphology) and criteria
The semen quality was evaluated as previously described by Golob et al. [27]. The same protocols were used to evaluate native semen, conventionally prepared spermatozoa, and MACS-selected spermatozoa. Briefly, motility was evaluated by placing a 10 μL sample on a glass slide, covering it with a coverslip, and observing it under a microscope on several fields to determine the representative average sperm motility. To determine the spermatozoa concentration, the sample was diluted in sperm counting solution, placed on improved Neubauer counting chamber, and assessed under a microscope. To determine the sperm morphology, air-dried smears of the spermatozoa samples were fixed in ethanol and stained with the Papanicolaou method. The stained samples were assessed with oil immersion at 1000 magnification, and at least 100 spermatozoa were examined to determine the proportions with normal and abnormal morphology. For the sperm morphology evaluation, the strict Kruger criteria were used [28]. The morphology of a spermatozoon was initially examined at the head, and if the head was abnormal, the spermatozoon was assessed as abnormal, and the neck/midpiece and tail morphologies were not further examined. The men were considered teratozoospermic if the sperm concentration was ≥ 15 million spermatozoa/ml, the total motility was ≥ 40%, the progressive motility was ≥ 32%, and the percentage of spermatozoa with normal morphology was ≤ 14%.
Preparation of semen sample for ICSI and MACS-ICSI
After obtaining the sperm sample on the day of oocyte retrieval, the sample was assessed for volume, concentration, motility, and the presence of round cells. After assessment, 2 ml of the sample was placed on top of a two-layer density gradient. If the volume of a sperm sample was lower than 2 ml, the required volume of medium used for sperm preparation (Sydney IVF Sperm Medium (Cook) or Sperm Preparation Medium (Origio) was used depending on availability) was added to obtain a 2 ml sample. The two-layer density gradient consisted of a 2-ml fraction of 100% Pure Sperm (Nidacon) and a 2-ml fraction of 40% Pure Sperm. The gradient with the sperm was centrifuged for 30 min at 1200 rpm, and the upper two layers (i.e., the seminal plasma and 40% fraction of Pure Sperm) were then discarded. The 100% fraction was then divided into half, 4 ml of sperm preparation medium was added to each half, and the samples were centrifuged for 10 min at 1400 rpm. After centrifugation, all of the supernatant was removed from the first half of washed sperm, and the washed spermatozoa were used for the MACS of the non-apoptotic spermatozoa (the protocol is described in the next paragraph). In the second half of the washed sperm, most of the supernatant was removed, and 0.5–0.8-ml sperm preparation medium was added to the remaining sperm pellet and medium to allow swim-up. After the swim-up technique, the sperm fraction was used to make droplets under paraffin oil, and the droplets were used to perform ICSI. After ICSI, the tube with the prepared sperm was transported to our andrology department for sperm morphology evaluation.
MACS of the prepared semen samples
The selection of the non-apoptotic spermatozoa was performed using a MACS ART Annexin V System (Miltenyi) as described by the manufacturer. Briefly, after density gradient centrifugation and washing, 0.1 ml MACS ART Annexin V Reagent was used to resuspend the pellet of spermatozoa, and MACS ART Binding Buffer was added to a final volume of 0.5 ml. After mixing, the suspension was incubated for 15 min at room temperature to allow the binding of annexin V-conjugated magnetic microbead complexes to apoptotic spermatozoa, which externalize phosphatidylserine. For magnetic separation, MACS ART MS Columns were placed into a MiniMACS Separator (Fig. 1), rinsed once with 1 ml of MACS ART Binding Buffer (which was discarded after flow-through), and the spermatozoa suspension was then placed in the column. The apoptotic spermatozoa, which were bound to annexin V-conjugated magnetic microbeads, were then retained in the column. That was because the column was placed in MiniMACS Separator which is basically a magnet forming magnetic field, which causes the retention of magnetically labeled cells. Therefore, the spermatozoa, which were not magnetically labeled with annexin V-conjugated magnetic microbeads, did pass the column and were collected. Because these spermatozoa were not labeled, they represent the non-apoptotic population. After this first fraction of spermatozoa was collected, the column was washed once with 0.5 ml MACS ART Binding Buffer to collect any remaining unlabeled spermatozoa from the column. This after flow-through fraction was combined with the fraction from the previous step. This mixture of enriched viable spermatozoa was then washed by adding 4 ml of sperm preparation medium and centrifuged 10 min at 1400 rpm. Next, most of the supernatant was removed, and 0.5–0.8 ml of sperm preparation medium was added to the remaining sperm pellet and medium to allow swim-up. The fraction of sperm obtained after the swim-up technique was used to make droplets under paraffin oil, and these droplets were used to perform ICSI. After ICSI, the tube with the prepared sperm was transported to our andrology department for evaluation of sperm morphology.
Fig. 1.
A photo taken during MACS sorting of non-apoptotic spermatozoa. As it can be seen on the photo, MACS ART MS Column is placed into MiniMACS Separator, which is attached to MACS MultiStand. After washing of the column with MACS ART Binding Buffer (left bigger bottle standing on the MultiStand), the sample of spermatozoa mixed with MACS ART Annexin V Reagent (right smaller bottle standing on the MultiStand) is applied into the column and unlabeled (non-apoptotic) spermatozoa that pass through the column are collected in a centrifuge tube standing under the column. The column is washed once with MACS ART Binding Buffer to collect all unlabeled spermatozoa. The unlabeled spermatozoa are then washed with sperm preparation medium and prepared for ICSI
In vitro fertilization
Fertilized oocytes were cultured until the day after ICSI in fertilization medium (Cook, Australia) and examined for the presence of pronuclei 16–18 h after fertilization. Normally fertilized zygotes (2PN) were cultured in the continuous culture medium SAGE 1-Step (Origio) until transferred into the uterus on day 3 or on day 5. If embryo transfer was performed on day 3 and there were supernumerary embryos, they were cultured until the fifth or sixth day to be cryopreserved (if they developed to blastocyst stage). On day 3, the embryos were defined as good quality when having 6–10 equally sized blastomeres and less than 15% fragmentation, as fair quality if there were unequally sized blastomeres and less than 15% fragmentation or equally and unequally sized blastomeres with 15–50% fragmentation, and as poor quality if fragmentation represented more than 50% of the embryo volume. On day 5, the embryos were graded according to Gardner et al. [29]. Briefly, the blastocysts were graded as good quality if they developed, expanded, or began to hatch and expressed good inner cell mass (ICM) and trophoectoderm (TE) morphologies; as fair quality if they developed, expanded, or began to hatch and exhibited good morphology of only either the ICM or TE; and as poor quality if they exhibited abnormal morphologies of both structures. In the last group, early blastocysts (grade 1) were also included. In each patient, at most, two embryos were transferred. If the embryo transfer was performed on day 3, the remaining embryos were cultured until day 5/6 to achieve the blastocyst stage. These surplus blastocysts were then cryopreserved.
Statistical analysis
To determine the differences between the morphologies of conventionally prepared spermatozoa and MACS-selected spermatozoa, a Mann-Whitney test was used. To determine other differences between the tested groups, a multivariate analysis was performed. P values under 0.05 were recognized as statistically significant.
Results
Twenty-six couples who were treated with ICSI in 2016 due to male infertility (more precisely, due to teratozoospermia as defined by the strict Kruger criteria; i.e., less than 15% of the spermatozoa exhibited a normal morphology) were included in this prospective study. The overall results are presented in Table 1. Briefly, in both groups (i.e., the standard ICSI and MACS-ICSI groups), similar numbers of oocytes were microinjected, and this resulted in similar proportions of fertilized oocytes (ICSI vs. MACS-ICSI, 77.4 vs. 68.5%, respectively) and cleaved embryos (98.1 vs. 97.7%, respectively). Additionally, the quality of the day 3 and day 5 embryos did not significantly differ, and the proportions of frozen blastocysts and cycles with freezing were similar between the groups. The transfers of the embryos into the uterus (ET) were performed in all cycles (Table 2); specifically, there were nine transfers with embryos obtained using standard ICSI, 16 with embryos obtained using MACS-ICSI, and one with 2 embryos obtained from both approaches. These ETs resulted in seven deliveries (three following standard ICSI and four following MACS-ICSI). Altogether, the supernumerary embryos were frozen in 21 cycles (80.8%); some of these (from 13 cycles) were already thawed, and this resulted in five additional ongoing pregnancies and one miscarriage.
Table 1.
The outcome of the ICSI cycles in terms of oocytes and embryos according to the method of sperm selection
| Standard ICSI | MACS-ICSI | |
|---|---|---|
| Number of patients | 26 | |
| Mean female age (± SD) | 31.3 ± 2.4 | |
| Average oocyte number (together) | 13.1 ± 6.1 | |
| Injected oocytes | 133 | 127 |
| Fertilized oocytes | 103 (77.4%) | 87 (68.5%) |
| Cleaved embryos | 101 (98.1%) | 85 (97.7%) |
| Good quality day 3 embryos | 59 (58.4%) | 53 (62.4%) |
| Fair quality day 3 embryos | 25 (24.8%) | 16 (18.8%) |
| Poor quality day 3 embryos | 17 (17.0%) | 16 (18.8%) |
| Number of embryos cultured until the day 5/6 | 96 | 75 |
| Number of blastocyst | 43 (44.8%) | 31 (41.3%) |
| Good quality blastocyst | 19 (44.2%) | 21 (67.7%) |
| Fair quality blastocyst | 8 (18.6%) | 7 (22.5%) |
| Poor quality blastocyst | 16 (37.2%) | 3 (9.7%) |
| Number of frozen blastocysts | 36 (35.6%) | 23 (27.1%) |
| Number of cycles with blastocyst freezing | 18 (69.2%) | 12 (46.2%) |
No statistically significant differences were observed (P value < 0.05)
Table 2.
The outcome of ICSI cycles in terms of pregnancies and deliveries according to the sperm selection method
| Standard ICSI | MACS-ICSI | |
|---|---|---|
| Number of ET together | 26 | |
| Number of pregnancies together | 8 (30.8%) | |
| Number of ET (standard ICSI or MACS-ICSI embryos only) | 9 | 16 |
| Number of pregnancies (standard ICSI or MACS-ICSI embryos only) | 3 (33.3%) | 5 (31.3%) |
| Number of ET with embryos from both treatments | 1 | |
| Mean number of transferred embryos (together) | 1.2 ± 0.4 | |
| Mean number of transferred embryos (standard ICSI embryos or MACS-ICSI embryos only) | 1.2 ± 0.6 | 1.1 ± 0.3 |
| Implantation rate (standard ICSI embryos or MACS-ICSI embryos only) | 27.3% | 27.7% |
| Number of deliveries together | 7 | |
| Number of deliveries regarding to the method | 3 | 4 |
No statistically significant differences were observed (P value < 0.05)
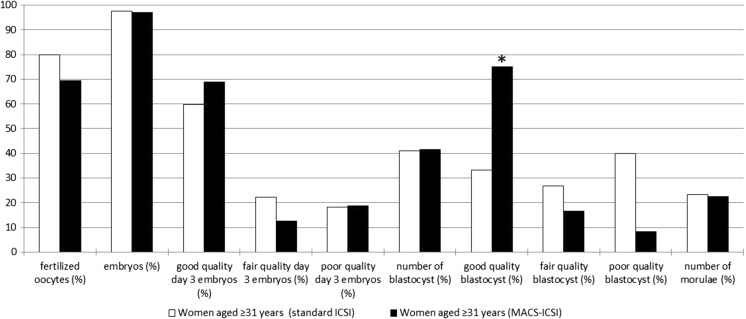
As mentioned above, comparisons of the embryos from all patients revealed no difference in the quality of the day 5/6 blastocysts, although the statistical analysis revealed that the ages of the patients might have influenced this result. Therefore, the patients were divided into two groups, i.e., patients aged 30 years or less and patients aged 31 years or more. The detailed results are presented in Table 3. In the group of patients under 31 years (eight patients), no differences were observed in the numbers (ICSI vs. MACS-ICSI; 56.5 vs. 41.2%, respectively) or quality of the day 5/6 blastocysts (good quality 69.2 vs. 42.9%, fair quality 0 vs. 42.9%, poor quality 30.8 vs. 14.3%) or in the numbers (100 vs. 95.2%) or quality of day 3 embryos (good quality 54.2 vs. 45.0%, fair quality 33.3 vs. 40.0%, poor quality 12.5 vs. 15.0%). Moreover, there was no significant difference in the proportions of fertilized oocytes and embryos. ETs were performed four times with embryos that were obtained using standard ICSI and four times with embryos that were obtained using MACS-ICSI. Altogether in this group, five pregnancies (two from standard ICSI and three from MACS-ICSI) and four deliveries (two from standard ICSI and two from MACS-ICSI) were achieved. In contrast, in the group of patients aged 31 years or more, the analyses revealed that the proportion of good quality day 5/6 blastocysts was higher after MACS-ICSI treatment (33.3 and 75.0% in the ICSI and MACS-ICSI groups, respectively; P = 0.028), although the proportions of embryos that reached the blastocyst stage were similar (41.1 vs. 41.4%; Fig. 2). There were also no differences in the proportions of day 3 embryos (97.5 and 97.0% in the ICSI and MACS-ICSI groups, respectively) or their quality (good quality 59.7 vs. 68.8%, fair quality 22.1 vs. 12.5%, poor quality 18.2 vs. 18.8%). ETs were performed five times with embryos that were obtained using standard ICSI, 12 times with embryos obtained using MACS-ICSI, and once with 2 embryos obtained from both approaches. Altogether in this group, three pregnancies (one from standard ICSI and two from MACS-ICSI) and three deliveries (one from standard ICSI and two from MACS-ICSI) were achieved.
Table 3.
The outcome of the ICSI cycles regarding to the age of patients
| Women aged ≤ 30 years | Women aged ≥ 31 years | |||
|---|---|---|---|---|
| Standard ICSI | MACS-ICSI | Standard ICSI | MACS-ICSI | |
| Number of patients | 8 | 18 | ||
| Mean female age (± SD) | 28.6 ± 1.3 | 32.4 ± 1.8 | ||
| Average oocyte number (together) | 11.3 ± 6.0 | 13.9 ± 6.3 | ||
| Injected oocytes | 34 | 32 | 99 | 95 |
| Fertilized oocytes | 24 (70.6%) | 21 (65.6%) | 79 (79.8%) | 66 (69.5%) |
| Cleaved embryos | 24 (100%) | 20 (95.2%) | 77 (97.5%) | 64 (97.0%) |
| Good quality day 3 embryos | 13 (54.2%) | 9 (45.0%) | 46 (59.7%) | 44 (68.8%) |
| Fair quality day 3 embryos | 8 (33.3%) | 8 (40.0%) | 17 (22.1%) | 8 (12.5%) |
| Poor quality day 3 embryos | 3 (12.5%) | 3 (15.0%) | 14 (18.2%) | 12 (18.8%) |
| Number of embryos cultured until the day 5/6 | 23 | 17 | 73 | 58 |
| Number of blastocyst | 13 (56.5%) | 7 (41.2%) | 30 (41.1%) | 24 (41.4%) |
| Good quality blastocyst | 9 (69.2%) | 3 (42.9%) | 10 (33.3%)* | 18 (75.0%)* |
| Fair quality blastocyst | 0 | 3 (42.9%) | 8 (26.7%) | 4 (16.7%) |
| Poor quality blastocyst | 4 (30.8%) | 1 (14.3%) | 12 (40.0%) | 2 (8.3%) |
| Number of frozen blastocysts | 9 (37.5%) | 6 (30.0%) | 27 (35.1%) | 17 (26.6%) |
| Number of cycles with blastocyst freezing | 5 (62.5%) | 5 (62.5%) | 13 (72.2%) | 7 (38.9%) |
| Number of ET together | 8 | 18 | ||
| Number of pregnancies together | 5 (62.5%) | 3 (16.7%) | ||
| Number of ET (standard ICSI or MACS-ICSI embryos only) | 4 | 4 | 5 | 12 |
| Number of pregnancies (standard ICSI or MACS-ICSI embryos only) | 2 (50.0%) | 3 (75.0%) | 1 (20.0%) | 2 (16.7%) |
| Number of ET with embryos from both treatments | 0 | 1 | ||
| Mean number of transferred embryos (together) | 1.1 ± 0.4 | 1.2 ± 0.4 | ||
| Mean number of transferred embryos (standard ICSI embryos or MACS-ICSI embryos only) | 1.3 ± 0.5 | 1.0 ± 0 | 1.2 ± 0.4 | 1.2 ± 0.4 |
| Number of deliveries together | 4 | 3 | ||
| Number of deliveries regarding to the method | 2 | 2 | 1 | 2 |
The observed statistically significant difference is marked with asterisk (P value < 0.05)
Fig. 2.
The outcome of the ICSI cycles in terms of oocytes and embryo development according to the sperm selection method among women aged 31 years or more. The only observed statistically significant difference (marked with asterisk) was a higher proportion of good quality day 5/6 blastocysts following MACS-ICSI treatment (33.3 and 75.0% for the ICSI and MACS-ICSI procedures, respectively; P = 0.028)
The morphology of conventionally prepared semen and MACS-selected spermatozoa
After the ICSI and MACS-ICSI procedures, the morphology of prepared spermatozoa was evaluated to determine the influence of the sperm selection technique on sperm quality parameters. The percentage of morphologically normal spermatozoa varied widely between samples from 3 to 40% in the conventionally prepared spermatozoa and from 2 to 30% in the MACS-selected population. This variation indicates that density gradient centrifugation itself highly enriched the population of morphologically normal spermatozoa because the percentages of morphologically normal spermatozoa in the native semen samples from males with teratozoospermia ranged from 1 to 14%. The analysis revealed that the percentage of morphologically normal spermatozoa did not differ significantly between the MACS-selected non-apoptotic group and the conventionally prepared group (conventionally prepared vs. MACS-selected; 12.6 ± 9.1% (median, 12%) vs. 11.6 ± 8.4% (median, 10%), respectively; P = 0.417). Detailed analyses of the morphologically abnormal spermatozoa revealed that, compared with the conventionally prepared group, among MACS-selected non-apoptotic spermatozoa, there were significantly more spermatozoa with abnormal tails (11.7 ± 11.1% (median, 10%) vs. 18.9 ± 13.2% (median, 14%), respectively; P = 0.046) and a trend towards more spermatozoa with abnormal necks and midpieces (6.7 ± 6.1% (median, 5%) vs. 8.5 ± 6.4% (median, 9%), respectively; P = 0.221). In contrast, there was a trend for more spermatozoa with abnormal heads in the conventionally prepared sample of spermatozoa (69.0 ± 17.8% (median, 75%) vs. 61.0 ± 19.5% (median, 67%) in the conventionally prepared and MACS-ICSI groups, respectively; P = 0.092), although the difference was not significant.
Discussion
In many studies that have been conducted on the topic of the MACS selection of non-apoptotic spermatozoa, the selected spermatozoa were only analyzed for their characteristics and were not functionally used in IVF/ICSI procedures. Additionally, to our knowledge, this is the first study in which MACS-selected spermatozoa were used to fertilize sibling oocytes because all other studies have only used similar populations of patients for comparison [23, 24, 26, 30–32]. Consequently, the data regarding whether the use of this selection technique is of any advantage in IVF/ICSI procedures remain inconclusive. For this reason, we performed this prospective study on sibling oocytes where MACS-selected non-apoptotic spermatozoa were used to fertilize oocytes and obtained embryos were cultured up to fifth/sixth day and transferred into uterus when of better quality as embryos obtained from sibling oocytes fertilized with conventionally prepared spermatozoa. To determine the effects of the MACS procedure, the outcome of ICSI/MACS-ICSI cycles was evaluated on several levels that ranged from the basic characteristics of the obtained spermatozoa and embryos to pregnancies and deliveries. Because we included couples dealing with male infertility due to teratozoospermia as defined by the strict Kruger criteria in our study, the morphology of conventionally prepared and MACS-selected spermatozoa were evaluated after ICSI to determine the effects of MACS selection. The results revealed that the overall percentage of morphologically normal spermatozoa did not differ significantly between the ICSI and MACS-ICSI procedures. However, detailed analyses of the morphologically abnormal spermatozoa revealed significantly more spermatozoa with abnormal tails after MACS-ICSI procedure, which may be potential consequence of the selection procedure. Moreover, the trends towards less spermatozoa with abnormal heads and towards more spermatozoa with abnormal necks and midpieces after MACS-ICSI procedure were revealed, although the differences were not significant. This observation is partly in concordance with study by Cakar et al. [33], who showed that the motility of spermatozoa was significantly impaired after MACS when compared to fresh samples. This could be due to more tail abnormalities caused by MACS procedure as we noticed in our study, although we did not evaluate the motility specifically. Furthermore, they observed that the percentage of morphologically normal spermatozoa was significantly lower after swim-up followed by MACS then when only swim-up spermatozoa were analyzed. Additionally, there was also one study on an animal model (boar), which revealed the negative effect of MACS on motility and viability of boar spermatozoa [34]. This study also showed that MACS selected non-apoptotic fraction contains significantly lower proportions of normal spermatozoa as a control [34]. The authors of this study suggest that boar spermatozoa are more sensitive than humans, or maybe the magnetic field could have some negative influence. These results are somewhat contradictory to previously published studies because they have demonstrated that MACS selection can improve the percentage of spermatozoa with normal morphology [23, 35, 36]. However, other studies have demonstrated that MACS does not improve this percentage [37, 38]. These differences may be consequences of different patient populations and different sperm preparation approaches before MACS selection. For example, Dirican et al. [23] studied oligoasthenozoospermic patients and used MACS before DGC, Said et al. [35] and Aziz et al. [36] studied normozoospermic patients and used MACS after DGC, Nadalini et al. [37] also studied normozoospermic patients and used MACS after DGC but compared the non-apoptotic spermatozoa fraction to that resulting from a DGC procedure in combination with swim-up, and Curti et al. [38] who studied teratozoospermic patients with sperm chromatin dispersion test above 16% and used MACS after swim-up.
Our observed differences in morphology did not seem to influence spermatozoa functionality because the ICSI/MACS-ICSI cycle outcomes were very comparable between the approaches, and we did not find any significant differences in any stages of the outcome when all couples were compared. However, because the results indicated a trend that suggested that the percentage of good quality blastocysts might have been influenced by the age of the female, additional analysis was performed. These analyses revealed that the MACS procedure improved the percentage of good quality blastocysts in the female patients aged over 30 years old. This difference may be explained by capability of oocytes to repair damaged paternal DNA [39]. Damaged paternal DNA can lead to impaired fertilization, impaired embryo development, impaired implantation, pregnancy loss, or even developmental abnormalities [40–43]. The DNA repair capabilities of oocytes are better in younger women and decrease with age [44], and our results suggest that the need for such repair mechanisms can be reduced by improving the quality of the spermatozoa with the MACS procedure. This observation accords with reports that have demonstrated that spermatozoa DNA integrity can be significantly improved via the use of the MACS selection of non-apoptotic spermatozoa. Degheidy et al. [45] demonstrated that MACS can significantly reduce DNA fragmentation in infertile varicocele patients, and Troya and Zorrilla [32] additionally demonstrated that couples who become pregnant following the use of MACS-selected spermatozoa for ICSI had DNA fragmentation index under 14%, which is normal. In addition to improving the DNA fragmentation index, MACS has been demonstrated to improve the percentage of euploid spermatozoa. This benefit has previously been revealed in normozoospermic patients with implantation failure in whom it has been reported that MACS can lower the percentage of aneuploidies (tested for chromosomes 18, X, and Y) [46]. Furthermore, Esbert et al. [47] studied patients with abnormal spermatozoa based on FISH and revealed that spermatozoa with disomy and nullisomy 13, 18, and 21, X, Y, and diploidy are more likely to be recognized by annexin microbeads and retained in MACS columns.
As explained above, the percentage of good quality blastocysts was the only difference observed between the studied groups, and this difference was only observed in the subpopulation of couples in which the female was aged over 30 years. Other differences were not found. Similarly, no differences were observed by Romany et al. [26] when a MACS-ICSI procedure was applied in oocyte donation cycles because the donating women were young. In this study, very similar fertilization rates, embryo quality rates, pregnancy rates, and live-birth rates were achieved, although the absence of any selection of the male patients was a shortcoming of this study. In other studies, some benefits of the MACS selection of non-apoptotic spermatozoa have been reported. Sánchez-Martín et al. [24] found that the MACS procedure lowers the miscarriage rate, although it does not improve the live-birth rate in autologous or donor oocyte ICSI cycles. In contrast, Troya and Zorrilla [32] reported no improvement in the miscarriage rate due to MACS, although they did observe an improvement in the clinical pregnancy rate. There are a few other reports of improved pregnancy rates, including the following: a report by San Celestino et al. [31] in which oocyte donation cycles were examined, and only frozen sperm samples were used; a report by Khalid and Qureshi [48] in which couples with unexplained infertility were examined following intrauterine insemination; and a report by Buzzi et al. [30] in which oocyte donation associated with severe male factor infertility was examined. An improved pregnancy rate combined with an improved embryo cleavage rate among oligoasthenozoospermic patients was reported by Dirican et al. [23]. Moreover, a meta-analysis revealed that sperm selection using MACS significantly improves the pregnancy rate, although the miscarriage rate is not improved [25].
In conclusion, the selection of non-apoptotic spermatozoa using MACS can be a useful approach but only in a clearly defined population of couples in which the female is aged over 30 years. The results of our study demonstrated that such couples dealing with male infertility due to teratozoospermia can benefit from this procedure by obtaining a higher percentage of good quality blastocysts, although a study with higher number of patients should be performed to confirm our conclusions.
Acknowledgements
The authors would like to thank all gynecologists, clinical embryologists, medical nurses, and other staff of the Reproductive Unit, Department of Obstetrics and Gynecology, University Medical Centre Ljubljana, for all the support. We would like to thank also our Andrology Laboratory for the semen analysis.
Funding information
The study was funded with tertiary project of University Medical Centre Ljubljana, Slovenia, granted to Dr. Martin Stimpfel.
Compliance with ethical standards
Conflict of interests
The authors declare that they have no conflict of interests.
References
- 1.World Health Organization . WHO Laboratory manual for the examination and processing of human semen. 5. Geneva: WHO Press; 2010. [Google Scholar]
- 2.Bartoov B, Berkovitz A, Eltes F, Kogosowski A, Menezo Y, Barak Y. Real-time fine morphology of motile human sperm cells is associated with IVF-ICSI outcome. J Androl. 2002;23:1–8. doi: 10.1002/j.1939-4640.2002.tb02595.x. [DOI] [PubMed] [Google Scholar]
- 3.Knez K, Zorn B, Tomazevic T, Vrtacnik-Bokal E, Virant-Klun I. The IMSI procedure improves poor embryo development in the same infertile couples with poor semen quality: a comparative prospective randomized study. Reprod Biol Endocrinol. 2011;9:123. doi: 10.1186/1477-7827-9-123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Knez K, Tomazevic T, Zorn B, Vrtacnik-Bokal E, Virant-Klun I. Intracytoplasmic morphologically selected sperm injection improves development and quality of preimplantation embryos in teratozoospermia patients. Reprod BioMed Online. 2012;25:168–179. doi: 10.1016/j.rbmo.2012.03.011. [DOI] [PubMed] [Google Scholar]
- 5.Knez K, Tomazevic T, Vrtacnik-Bokal E, Virant-Klun I. Developmental dynamics of IMSI-derived embryos: a time-lapse prospective study. Reprod BioMed Online. 2013;27:161–171. doi: 10.1016/j.rbmo.2013.04.002. [DOI] [PubMed] [Google Scholar]
- 6.Shalom-Paz E, Anabusi S, Michaeli M, Karchovsky-Shoshan E, Rothfarb N, Shavit T, Ellenbogen A. Can intra cytoplasmatic morphologically selected sperm injection (IMSI) technique improve outcome in patients with repeated IVF-ICSI failure? a comparative study. Gynecol Endocrinol. 2015;31:247–251. doi: 10.3109/09513590.2014.982085. [DOI] [PubMed] [Google Scholar]
- 7.Setti AS, Braga DP, Figueira RC, Iaconelli A, Jr, Borges E. Intracytoplasmic morphologically selected sperm injection results in improved clinical outcomes in couples with previous ICSI failures or male factor infertility: a meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2014;183:96–103. doi: 10.1016/j.ejogrb.2014.10.008. [DOI] [PubMed] [Google Scholar]
- 8.Gatimel N, Parinaud J, Leandri RD. Intracytoplasmic morphologically selected sperm injection (IMSI) does not improve outcome in patients with two successive IVF-ICSI failures. J Assist Reprod Genet. 2016;33:349–355. doi: 10.1007/s10815-015-0645-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fortunato A, Boni R, Leo R, Nacchia G, Liguori F, Casale S, Bonassisa P, Tosti E. Vacuoles in sperm head are not associated with head morphology, DNA damage and reproductive success. Reprod BioMed Online. 2016;32:154–161. doi: 10.1016/j.rbmo.2015.10.009. [DOI] [PubMed] [Google Scholar]
- 10.Teixeira DM, Barbosa MA, Ferriani RA, Navarro PA, Raine-Fenning N, Nastri CO, Martins WP. Regular (ICSI) versus ultra-high magnification (IMSI) sperm selection for assisted reproduction. Cochrane Database Syst Rev. 2013;7:CD010167. doi: 10.1002/14651858.CD010167.pub2. [DOI] [PubMed] [Google Scholar]
- 11.Maettner R, Sterzik K, Isachenko V, Strehler E, Rahimi G, Alabart JL, Sánchez R, Mallmann P, Isachenko E. Quality of human spermatozoa: relationship between high-magnification sperm morphology and DNA integrity. Andrologia. 2014;46:547–555. doi: 10.1111/and.12114. [DOI] [PubMed] [Google Scholar]
- 12.Hammoud I, Boitrelle F, Ferfouri F, Vialard F, Bergere M, Wainer B, Bailly M, Albert M, Selva J. Selection of normal spermatozoa with a vacuole-free head (x6300) improves selection of spermatozoa with intact DNA in patients with high sperm DNA fragmentation rates. Andrologia. 2013;45:163–170. doi: 10.1111/j.1439-0272.2012.01328.x. [DOI] [PubMed] [Google Scholar]
- 13.Wilding M, Coppola G, di Matteo L, Palagiano A, Fusco E, Dale B. Intracytoplasmic injection of morphologically selected spermatozoa (IMSI) improves outcome after assisted reproduction by deselecting physiologically poor quality spermatozoa. J Assist Reprod Genet. 2011;28:253–262. doi: 10.1007/s10815-010-9505-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Garolla A, Sartini B, Cosci I, Pizzol D, Ghezzi M, Bertoldo A, Menegazzo M, Speltra E, Ferlin A, Foresta C. Molecular karyotyping of single sperm with nuclear vacuoles identifies more chromosomal abnormalities in patients with testiculopathy than fertile controls: implications for ICSI. Hum Reprod. 2015;30:2493–2500. doi: 10.1093/humrep/dev202. [DOI] [PubMed] [Google Scholar]
- 15.Lavolpe M, Lorenzi D, Greco E, Nodar F, Alvarez SC. Relationship between sperm DNA fragmentation and nuclear vacuoles. JBRA Assist Reprod. 2015;19:70–74. doi: 10.5935/1518-0557.20150016. [DOI] [PubMed] [Google Scholar]
- 16.Oosterhuis GJ, Mulder AB, Kalsbeek-Batenburg E, Lambalk CB, Schoemaker J, Vermes I. Measuring apoptosis in human spermatozoa: a biological assay for semen quality? Fertil Steril. 2000;74:245–250. doi: 10.1016/S0015-0282(00)00623-3. [DOI] [PubMed] [Google Scholar]
- 17.Vermes I, Haanen C, Steffens-Nakken H, Reutelingsperger C. A novel assay for apoptosis. Flow cytometric detection of phosphatidylserine expression on early apoptotic cells using fluorescein labelled Annexin V. J Immunol Methods. 1995;184:39–51. doi: 10.1016/0022-1759(95)00072-I. [DOI] [PubMed] [Google Scholar]
- 18.Grunewald S, Paasch U. Sperm selection for ICSI using annexin V. Methods Mol Biol. 2013;927:257–262. doi: 10.1007/978-1-62703-038-0_23. [DOI] [PubMed] [Google Scholar]
- 19.Rawe VY, Boudri HU, Alvarez Sedó C, Carro M, Papier S, Nodar F. Healthy baby born after reduction of sperm DNA fragmentation using cell sorting before ICSI. Reprod BioMed Online. 2010;20:320–323. doi: 10.1016/j.rbmo.2009.12.004. [DOI] [PubMed] [Google Scholar]
- 20.Bucar S, Gonçalves A, Rocha E, Barros A, Sousa M. Sá R. DNA fragmentation in human sperm after magnetic-activated cell sorting. J Assist Reprod Genet. 2015;32:147–154. doi: 10.1007/s10815-014-0370-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Grunewald S, Baumann T, Paasch U, Glander HJ. Capacitation and acrosome reaction in nonapoptotic human spermatozoa. Ann N Y Acad Sci. 2006;1090:138–146. doi: 10.1196/annals.1378.015. [DOI] [PubMed] [Google Scholar]
- 22.de Vantéry Arrighi C, Lucas H, Chardonnens D, de Agostini A. Removal of spermatozoa with externalized phosphatidylserine from sperm preparation in human assisted medical procreation: effects on viability, motility and mitochondrial membrane potential. Reprod Biol Endocrinol. 2009;7:1. doi: 10.1186/1477-7827-7-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dirican EK, Ozgün OD, Akarsu S, Akin KO, Ercan O, Uğurlu M, Camsari C, Kanyilmaz O, Kaya A, Unsal A. Clinical outcome of magnetic activated cell sorting of non-apoptotic spermatozoa before density gradient centrifugation for assisted reproduction. J Assist Reprod Genet. 2008;25:375–381. doi: 10.1007/s10815-008-9250-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sánchez-Martín P, Dorado-Silva M, Sánchez-Martín F, González Martínez M, Johnston SD, Gosálvez J. Magnetic cell sorting of semen containing spermatozoa with high DNA fragmentation in ICSI cycles decreases miscarriage rate. Reprod BioMed Online. 2017;34:506–512. doi: 10.1016/j.rbmo.2017.01.015. [DOI] [PubMed] [Google Scholar]
- 25.Gil M, Sar-Shalom V, Melendez Sivira Y, Carreras R, Checa MA. Sperm selection using magnetic activated cell sorting (MACS) in assisted reproduction: a systematic review and meta-analysis. J Assist Reprod Genet. 2013;30:479–485. doi: 10.1007/s10815-013-9962-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Romany L, Garrido N, Motato Y, Aparicio B, Remohí J, Meseguer M. Removal of annexin V-positive sperm cells for intracytoplasmic sperm injection in ovum donation cycles does not improve reproductive outcome: a controlled and randomized trial in unselected males. Fertil Steril. 2014;102:1567–75.e1. doi: 10.1016/j.fertnstert.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 27.Golob B, Poljak M, Verdenik I, Kolbezen Simoniti M, Vrtacnik Bokal E, Zorn B. High HPV infection prevalence in men from infertile couples and lack of relationship between seminal HPV infection and sperm quality. Biomed Res Int. 2014;2014:956901. doi: 10.1155/2014/956901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kruger TF, Menkveld R, Stander FS, Lombard CJ, Van der Merwe JP, van Zyl JA, Smith K. Sperm morphologic features as a prognostic factor in in vitro fertilization. Fertil Steril. 1986;46:1118–1123. doi: 10.1016/S0015-0282(16)49891-2. [DOI] [PubMed] [Google Scholar]
- 29.Gardner DK, Lane M, Stevens J, Schlenker T, Schoolcraft WB. Blastocyst score affects implantation and pregnancy outcome: towards a single blastocyst transfer. Fertil Steril. 2000;73:1155–1158. doi: 10.1016/S0015-0282(00)00518-5. [DOI] [PubMed] [Google Scholar]
- 30.Buzzi J, Valcarcel A, Lombardi E, Oses R, Rawe V, Young E. Magnetic activated cell sorting (MACS) improves oocyte donation results associated to severe male factor infertility. Hum Reprod. 2010;25(suppl 1):i118–i152. [Google Scholar]
- 31.San Celestino M, Agudo D, Alonso M, Sanjurjo P, Becerra D, Bronet F, Garcia-Velasco JA, Pacheco A. Improved pregnancy rate after sperm magnetic separation technique in egg donation cycles using frozen sperm samples. Hum Reprod. 2011;26(suppl 1):i123–i148. [Google Scholar]
- 32.Troya J, Zorrilla I. Annexin V-MACS in infertile couples as method for separation of sperm without DNA fragmentation. JBRA Assist Reprod. 2015;19:66–69. doi: 10.5935/1518-0557.20150015. [DOI] [PubMed] [Google Scholar]
- 33.Cakar Z, Cetinkaya B, Aras D, Koca B, Ozkavukcu S, Kaplanoglu İ, Can A, Cinar O. Does combining magnetic-activated cell sorting with density gradient or swim-up improve sperm selection? J Assist Reprod Genet. 2016;33:1059–1065. doi: 10.1007/s10815-016-0742-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mrkun J, Dolensek T, Knific T, Pislar A, Kosec M, Kos J, Zrimsek P. Elimination of apoptotic boar spermatozoa using magnetic activated cell sorting. Acta Vet Brno. 2014;83:13–18. doi: 10.2754/avb201483010013. [DOI] [Google Scholar]
- 35.Said TM, Grunewald S, Paasch U, Glander HJ, Baumann T, Kriegel C, Li L, Agarwal A. Advantage of combining magnetic cell separation with sperm preparation techniques. Reprod BioMed Online. 2005;10:740–746. doi: 10.1016/S1472-6483(10)61118-2. [DOI] [PubMed] [Google Scholar]
- 36.Aziz N, Said T, Paasch U, Agarwal A. The relationship between human sperm apoptosis, morphology and the sperm deformity index. Hum Reprod. 2007;22:1413–1419. doi: 10.1093/humrep/dem016. [DOI] [PubMed] [Google Scholar]
- 37.Nadalini M, Tarozzi N, Di Santo M, Borini A. Annexin V magnetic-activated cell sorting versus swim-up for the selection of human sperm in ART: is the new approach better then the traditional one? J Assist Reprod Genet. 2014;31:1045–1051. doi: 10.1007/s10815-014-0267-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Curti G, Skowronek F, Vernochi R, Rodriguez-Buzzi AL, Rodriguez-Buzzi JC, Casanova G, Sapiro R. Morphological evaluation of sperm from infertile men selected by magnetic activated cell sorting (MACS) Reprod Biol. 2014;14:289–292. doi: 10.1016/j.repbio.2014.07.002. [DOI] [PubMed] [Google Scholar]
- 39.Ménézo Y, Dale B, Cohen M. DNA damage and repair in human oocytes and embryos: a review. Zygote. 2010;18:357–365. doi: 10.1017/S0967199410000286. [DOI] [PubMed] [Google Scholar]
- 40.Barroso G, Valdespin C, Vega E, Kershenovich R, Avila R, Avendaño C, Oehninger S. Developmental sperm contributions: fertilization and beyond. Fertil Steril. 2009;92:835–848. doi: 10.1016/j.fertnstert.2009.06.030. [DOI] [PubMed] [Google Scholar]
- 41.Schulte RT, Ohl DA, Sigman M, Smith GD. Sperm DNA damage in male infertility: etiologies, assays, and outcomes. J Assist Reprod Genet. 2010;27:3–12. doi: 10.1007/s10815-009-9359-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Speyer BE, Pizzey AR, Ranieri M, Joshi R, Delhanty JD, Serhal P. Fall in implantation rates following ICSI with sperm with high DNA fragmentation. Hum Reprod. 2010;25:1609–1618. doi: 10.1093/humrep/deq116. [DOI] [PubMed] [Google Scholar]
- 43.Zini A, Boman JM, Belzile E, Ciampi A. Sperm DNA damage is associated with an increased risk of pregnancy loss after IVF and ICSI: systematic review and meta-analysis. Hum Reprod. 2008;23:2663–2668. doi: 10.1093/humrep/den321. [DOI] [PubMed] [Google Scholar]
- 44.Titus S, Li F, Stobezki R, Akula K, Unsal E, Jeong K, Dickler M, Robson M, Moy F, Goswami S, Oktay K. Impairment of BRCA1-related DNA double-strand break repair leads to ovarian aging in mice and humans. Sci Transl Med. 2013;5:172ra21. doi: 10.1126/scitranslmed.3004925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Degheidy T, Abdelfattah H, Seif A, Albuz FK, Gazi S, Abbas S. Magnetic activated cell sorting: an effective method for reduction of sperm DNA fragmentation in varicocele men prior to assisted reproductive techniques. Andrologia. 2015;47:892–896. doi: 10.1111/and.12343. [DOI] [PubMed] [Google Scholar]
- 46.Vendrell X, Ferrer M, García-Mengual E, Muñoz P, Triviño JC, Calatayud C, Rawe VY, Ruiz-Jorro M. Correlation between aneuploidy, apoptotic markers and DNA fragmentation in spermatozoa from normozoospermic patients. Reprod BioMed Online. 2014;28:492–502. doi: 10.1016/j.rbmo.2013.12.001. [DOI] [PubMed] [Google Scholar]
- 47.Esbert M, Godo A, Soares SR, Florensa M, Amorós D, Ballesteros A, Vidal F. Spermatozoa with numerical chromosomal abnormalities are more prone to be retained by Annexin V-MACS columns. Andrology. 2017;5:807–813. doi: 10.1111/andr.12376. [DOI] [PubMed] [Google Scholar]
- 48.Khalid SN, Qureshi IZ. Pregnancy rate improves in couples with unexplained infertility following intrauterine insemination (IUI) with magnetically selected non-apoptotic sperms. Fertil Steril. 2011;96:S25. doi: 10.1016/j.fertnstert.2011.07.104. [DOI] [Google Scholar]