Abstract
Purpose
To examine the effect of low and very low estradiol responses in oocyte donors receiving gonadotropins on clinical outcomes of donor in vitro fertilization (IVF) cycles and to identify possible mechanisms responsible for low estradiol response.
Methods
This is a retrospective cohort study of oocyte donors undergoing antagonist IVF cycles with progression to oocyte retrieval between January 2010 and December 2016 at a single urban academic fertility center. Oocyte yield, fertilization rate, blastocyst rate, percentage of normal embryos on preimplantation genetic screening (PGS), pregnancy outcomes, and follicular fluid steroid profiles were compared between donors with normal estradiol response and those with low estradiol response.
Results
Three hundred sixty-six antagonist oocyte donor IVF cycles were identified: 42 cycles had a normal estradiol response (NE2), defined as peak serum estradiol (E2) of over 200 pg/mL per retrieved oocyte; 140 cycles had an intermediate estradiol response (iE2), defined as peak serum E2 between 100 and 200 pg/mL per retrieved oocyte; 110 cycles had a low estradiol response (LE2), defined as peak serum E2 between 50 and 100 pg/mL per retrieved oocyte; and 74 cycles had a very low estradiol response (vLE2), defined as peak serum E2 less than 50 pg/mL per retrieved oocyte. LE2 cycles resulted in a greater number of mature oocytes (22.4 vs. 13.6, p < 0.017), and fertilizations versus NE2 donors (18.5 vs. 10.7, p < 0.017), although the number of transferred or cryopreserved blastocysts were similar between groups (8.6, 6.9 vs. 4.8, p = 0.095, p = 1). The percentage of chromosomally normal embryos after PGS was similar between LE2, vLE2, and NE2 cycles (66.4, 71.8 vs. 63.1%, p = 0.99, p = 1). Pregnancy outcomes were similar between LE2, vLE2, and NE2 cycles. Serum AMH obtained on the day of peak E2 was similar to baseline serum AMH and did not differ between LE2 versus NE2 cycles. Follicular fluid E2 levels paralleled serum E2 levels and were lower in LE2 cycles versus NE2 cycles.
Conclusion
The prevalence of very low E2 responses in donors appears to be high (20.2%). In contrast to autologous IVF cycles, LE2 does not portend poor outcomes in oocyte donors.
Electronic supplementary material
The online version of this article (10.1007/s10815-018-1192-7) contains supplementary material, which is available to authorized users.
Keywords: In vitro fertilization, Oocyte donor, Estradiol, Follicular fluid, COS response
Introduction
Serial estradiol levels during IVF stimulation cycles allow providers to assess response and titrate gonadotropin dose. At peak maturation, mature oocytes are estimated to secrete 200 to 300 pg/mL estradiol (E2) [1]. In autologous IVF cycles, low serum E2 is thought to portend poor outcome [2, 3] due to few retrieved oocytes with subsequently reduced fertilization and embryo progression. According to the Center for Disease Control data, 20,481 donor oocyte or donor embryo cycles were conducted in the USA in 2014, an increase from 16,161 cycles in 2005 [4]. Expected hormone profiles in oocyte donors are assumed to follow similar principles to their infertile counterparts, although stimulation responses in this population have not been extensively studied. While the majority of donor oocyte cycles have excellent recipient outcomes, understanding the nuances of this population’s response is critical to ensuring the safety of donor patients during IVF stimulation and optimizing pregnancy outcomes for intended parents.
The Columbia University Fertility Center (CUFC) donor program includes both directed anonymous and known donor cycles as well as stem cell donation in collaboration with the New York Stem Cell Foundation. Since the CUFC donor program’s origins in 1995, we have noted a minority of donor patients in which serum E2 drastically differs from oocyte yield. For a handful of donor patients, peak E2 levels remained below 500 pg/mL despite excellent oocyte yield and embryo progression. These findings are in contrast to expected steroid profiles and IVF response during gonadotropin stimulation.
The purpose of this study was to further investigate the outcomes of donors with low E2 responses. Furthermore, we aimed to investigate potential mechanisms of the observed lower E2 responses through analysis of serum and follicular fluid hormone profiles in donors with low E2 versus typical E2 response. We also assessed anti-Müllerian Hormone (AMH) levels as another measure of granulosa cell function.
Materials and methods
Study population
This retrospective study included all oocyte donor cycles that were conducted at the CUFC between January 2010 and December 2016. This time period was selected due to availability of patient charts for review—patient charts seen prior to January 2010 had been archived at the time of data collection. Women in the CUFC donor oocyte and donor stem cell programs are voluntary participants and have been previously screened for acceptance based on age, medical health, ovarian reserve testing, and genetic screening criteria. Eligible oocyte donors were healthy women, aged 21 to 32 years, with a body mass index (BMI) between 18.5 and 29.9 kg/m2, with no medical contraindications to ovarian stimulation and oocyte retrieval, and with adequate ovarian reserve testing, defined as an antral follicle count (AFC) over 12 and an AMH level over 1 ng/mL. Oocyte donors found to be a carrier for genetic disease as detected by Counsyl (Counsyl, Inc.) genetic carrier panel were able to participate in anonymous oocyte donation if acceptable to prospective recipients, while donors identified to be affected by genetic diseases were only eligible for stem cell donation. Following oocyte retrieval, all oocyte donors are provided an equivalent monetary compensation, regardless of number of oocytes collected. Both anonymous directed oocyte donor and stem cell donor cycles were included in the study. Known donor cycles were excluded, as these women were not held to age or ovarian reserve criteria for enrollment in the donor program. Only cycles using follicle-stimulating hormone (FSH) recombinant preparations and gonadotropin receptor hormone (GnRH) antagonist in vitro fertilization (IVF) protocols were included. We divided the donor cycles into groups based on serum E2 responses per oocyte retrieved. Four groups were identified: cycles with normal E2 response (NE2), defined as a peak serum E2 of over 200 pg/mL per retrieved oocyte; cycles with an intermediary E2 response (iE2), defined as a peak serum E2 between 100 and 200 pg/mL per retrieved oocyte; cycles with low estradiol response (LE2), defined as peak serum E2 between 50 and 100 pg/mL per retrieved oocyte; and cycles with very low E2 response (vLE2), defined as a peak serum E2 less than 50 pg/mL per retrieved oocyte. This division of estradiol response and our definition of low estradiol response were arbitrary because there is no prior literature defining a normal versus abnormal estradiol response following gonadotropin stimulation. Therefore, we aimed to present and study a spectrum of estradiol responses, with particular focus on the lower end of this spectrum. The definition of our lowest-defined estradiol group, with peak serum E2 less than 50 pg/mL per oocyte retrieved, is based on expected baseline follicular phase serum estradiol concentration and the expected level of estradiol during leuprolide acetate suppression prior to initiation of gonadotropins for IVF.
Demographic information and baseline infertility testing information were collected, including age, body mass index (BMI, kg/m2), race, ethnicity, AMH level (ng/mL), baseline follicle-stimulating hormone (FSH) level (mIU/mL), and antral follicle count (AFC). IVF stimulation cycle data were collected, including number of days of oral contraceptive pills taken prior to initiating injectable gonadotropins, number of days of gonadotropin stimulation, total number of gonadotropins (IU), peak serum E2 (pg/mL), and number of oocytes retrieved. The number of mature oocytes, defined as metaphase II oocytes, number of fertilized embryos, number of blastocysts, and number of embryos transferred were noted. For donor cycles resulting in a recipient embryo transfer, pregnancy outcomes, including conception rate, biochemical pregnancy rate, miscarriage rate, and ongoing pregnancy rate, were noted. For donor cycles resulting in subsequent preimplantation screening (PGS), the percentage of normal embryos was noted. Data points were obtained through abstraction from existing patient records. The Columbia University Institutional Review Board approved this study, IRB #AAAR1547.
Donor IVF stimulation and oocyte retrieval
Once accepted into the CUFC oocyte donor program and matched to a recipient patient (if applicable), oocyte donors were placed on oral contraceptive pills (OCPs) for a brief period with subsequent initiation of gonadotropins 3 days after the last OCP dose. Starting gonadotropin dosing for donor patients ranged from 150 to 300 IU Gonal-F® (EMD Serono, Inc.) or Follistim® (Merck) with or without 75 to 150 IU Menopur® (Ferring Pharmaceuticals) based on baseline ovarian reserve testing and donor age. Gonadotropins were injected nightly, and ovarian stimulation was monitored with regular ultrasound and serum hormonal assays, occurring after four nights of medications, then every 1–2 days as dictated by ovarian response. Gonadotropin-releasing hormone antagonist was initiated when lead follicle diameter reached 13 mm or with an E2 rise above 300 pg/mL. Oocyte trigger with 10,000 U human chorionic gonadotropin or 4 mg leuprolide acetate and 1000 U human chorionic gonadotropin was administered when 2–3 follicles achieved a maximum follicle diameter of 18–21 mm. Oocyte retrieval was scheduled 35 h following the trigger dose. Oocyte retrieval was performed by transvaginal ultrasound-guided needle aspiration under deep sedation anesthesia.
Serum hormone assays
Serum AMH is measured and recorded for all oocyte donors during the screening and selection process. AMH was quantified using the AMH Gen II ESLISA immunoassay (Beckman Coulter, Inc.) according to routine protocols. Throughout the oocyte stimulation cycle, serial serum E2 levels are collected and quantified using IMMULITE® immunoassay (Siemens) according to routine protocols. Serum blood samples from day of oocyte trigger are preserved for 1 month according to CWRC standard operating procedures for the endocrinology laboratory. Peak E2 and AMH levels were repeated from the preserved day of oocyte trigger serum samples for a small number of oocyte donors (n = 9). Additionally, low E2 serum samples were confirmed through an additional assay system, the Beckman Access 2 Immunoassay System. Differences between assay systems were found to be within the coefficient of variation. The intra and interassay coefficients of variation for E2 and AMH did not exceed 6 and 12%, respectively.
Follicular fluid steroid hormone assays
Follicular fluid from a small sample of oocyte donors (n = 9) was collected for analysis of steroid hormone profile assays. Following identification of oocytes, the discarded follicular fluid was centrifuged, and supernatant was frozen at − 20 °C for subsequent analysis. Follicular fluid AMH levels were measured using the AMH Gen II ESLISA immunoassay (Beckman Coulter, Inc.), and follicular fluid E2 and progesterone levels were measured using IMMULITE® immunoassay (Siemens) according to routine protocols.
Outcome measures
The division of women into groups having normal or low E2 responses to gonadotropin stimulation was based on the serum E2 level per oocyte retrieved. We analyzed oocyte yield, the fertilization rate, number of high-quality blastocysts, serum AMH on day of oocyte trigger, follicular fluid estradiol and AMH levels, rate of conception, biochemical pregnancy, miscarriage, ongoing pregnancy, and percentage of normal embryos following PGS. Conception was defined as a positive serum human chorionic gonadotropin level following embryo transfer. Biochemical pregnancy was defined as conception with subsequent decline in human chorionic gonadotropin without ultrasound evidence of pregnancy (gestational sac with yolk sac). Miscarriage was defined as conception with ultrasound evidence of pregnancy and subsequent pregnancy loss. Ongoing pregnancy was defined as conception with ultrasound evidence of pregnancy with a fetal heart rate and subsequent discharge from fertility practice, typically at 8 weeks gestation.
Statistical analysis
NE2, iE2, LE2, and vLE2 oocyte donor continuous variables were compared using ANOVA with Bonferroni correction for multiple comparisons. NE2, iE2, LE2, and vLE2 oocyte donor categorical variables were compared using chi-square analysis. Statistical analyses were performed using GraphPad Prism Software for Mac, version 6 (GraphPad, Inc., San Diego, CA), and Stata/IC 15 (StatCorp LLC, College Station, TX). Given multiple comparisons between NE2, iE2, LE2, and vLE2 oocyte donor groups; a p value of less than 0.017 was considered statistically significant.
Results
Demographics and baseline characteristics
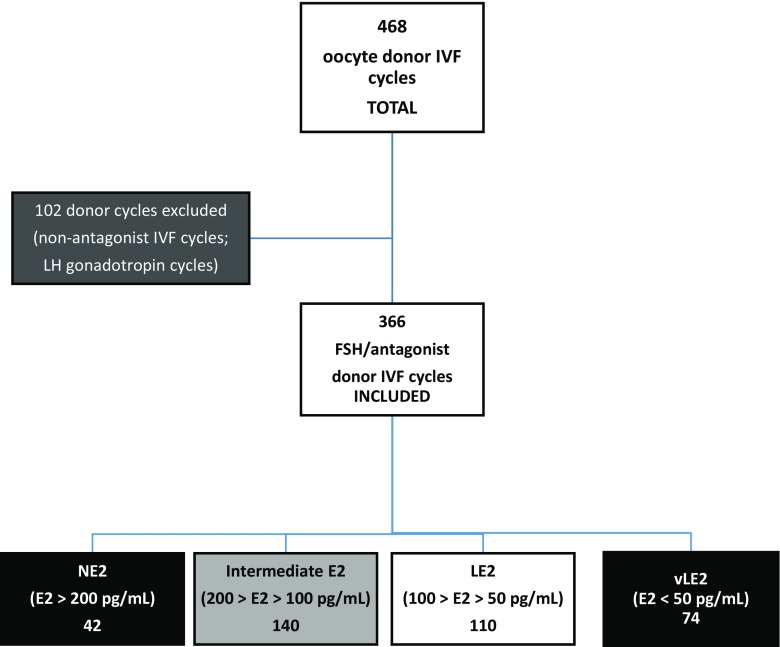
Between January 2010 and December 2016, a total of 468 donor cycles were identified. Of these, 366 cycles followed a follicle-stimulating hormone (FSH) and gonadotropin receptor hormone (GnRH) antagonist in vitro fertilization (IVF) protocol. The responses of E2 were as follows: 42 cycles with NE2, 140 cycles with iE2, 110 cycles with LE2, and 74 cycles with vLE2. These groups are depicted in Fig. 1.
Fig. 1.
Donor in vitro fertilization (IVF) cycles conducted at CWRC 2010–2016
The demographics and baseline characteristics of donors in NE2, iE2, LE2, and vLE2 groups are presented in Table 1. Compared to donors with NE2 cycles, iE2, LE2, and vLE2 donors were younger. Compared to donors with NE2 cycles, there were fewer patients identifying as Asian, and a higher percentage of patients identifying as White in LE2 and vLE2 cycles. No other differences in BMI, ethnicity, baseline AMH, or baseline AFC were detected. Additionally, there were no differences in the number of days of oral contraceptive pills used prior to gonadotropin cycle start. When adjusted to account for donors undergoing multiple cycles, results remained similar.
Table 1.
Demographics and baseline characteristics
| NE2 (n = 42) | iE2 (n = 140) | p value | LE2 (n = 110) | p-value | vLE2 (n = 74) | p value | ||
|---|---|---|---|---|---|---|---|---|
| Age (years) | 27.4 ± 3.0 | 26.0 ± 2.9 | 0.013a | 26 ± 2.6 | 0.008a | 25.2 ± 2.2 | < 0.0001a | |
| BMI (kg/m2) | 22.3 ± 2.8 | 22.7 ± 2.9 | 1a | 22.4 ± 2.5 | 1a | 22.5 ± 2.6 | 1a | |
| Race and ethnicity | American Indian | 0 | 0 | 1b | 0 | 1b | 1 (1.4) | 1b |
| Asian | 14 (33.3) | 29 (20.7) | 0.3b | 11 (10) | 0.003b | 2 (2.7) | < 0.001b | |
| Black | 3 (7.1) | 12 (8.6) | 1b | 4 (3.6) | 1b | 6 (8.1) | 1b | |
| White | 24 (57) | 89 (63.6) | 1b | 85 (77.3) | 0.06b | 63 (85.1) | 0.006b | |
| Other/mixed races | 1 (2.4) | 8 (5.7) | 1b | 10 (9.1) | 0.87b | 1 (1.4) | 1b | |
| Unknown | 0 | 2 (1.4) | 1b | 0 | 1b | 1 (1.4) | 1b | |
| Hispanic | 5 (11.9) | 21 (15) | 1b | 16 (14.5) | 1b | 8 (10.8) | 1b | |
| AMH (ng/mL) | 4.9 ± 3.5 | 4.1 ± 2.93 | 0.88a | 4.1 ± 2.9 | 0.50a | 4.0 ± 2.5 | 0.48a | |
| AFC | 21 ± 5.2 | 20.6 ± 6.0 | 1a | 20.8 ± 5.9 | 1a | 19.8 ± 5.7 | 1a | |
NE2 normal estradiol response (E2 > 200 pg/mL per oocyte); iE2 intermediate E2 response (200 > E2 > 100 pg/mL per oocyte); LE2 low E2 response (100 > E2 > 50 pg/mL per oocyte); vLE2 very low E2 response (E2 < 50 pg/mL per oocyte); BMI body mass index; AMH anti-Müllerian hormone; AFC antral follicle count. Data are represented as mean ± standard deviation or n (% total). Statistical significance defined by p < 0.017
aANOVA with Bonferroni correction for multiple comparisons
bChi-square test
NE2 versus LE2 donor IVF cycle outcomes
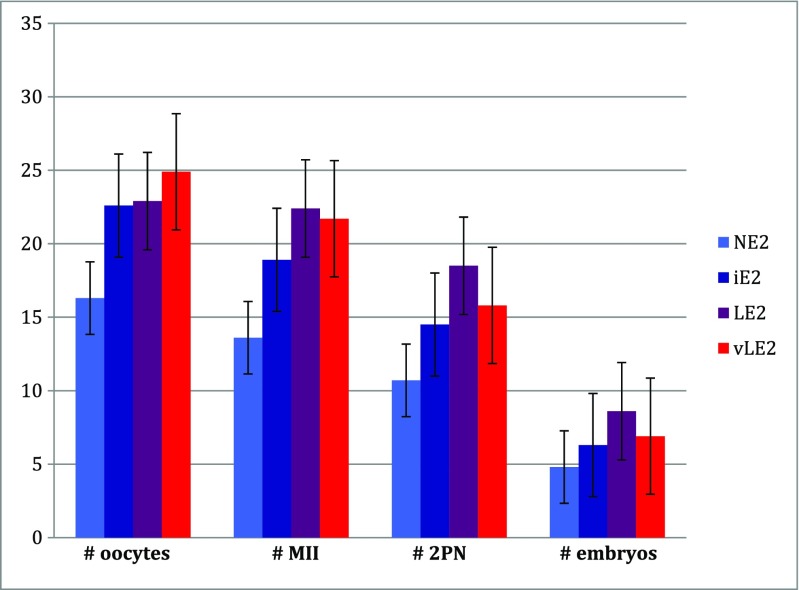
The IVF cycle outcomes of NE2, iE2, LE2, and vLE2 are presented in Table 2 and Fig. 2. Compared to NE2 cycles, LE2 and vLE2 cycles used higher total gonadotropins (2037.5 IU, 2188.7 IU versus 1764.6 IU, p = 0.011, p < 0.0001), underwent longer gonadotropin stimulation (10.5 days, 11.3 days versus 9.8 days, p < 0.0001, p < 0.0001), and had a higher number of oocytes obtained on day of retrieval (22.9, 25 versus 16.3, p < 0.0001). Compared to NE2 cycles, LE2 cycles, but not vLE2 cycles had a higher number of mature oocytes identified (22.4 versus 13.6, p = 0.012) and higher number of fertilized embryos (18.5 versus 10.7, p = 0.004). LE2 and vLE2 cycles had higher total number of embryos available for embryo transfer, embryo cryopreservation, or embryo biopsy compared to NE2 cycles, though this difference was not statistically significant after adjustment for multiple comparisons (8.6, 6.9 versus 4.8, p = 0.095, p = 1).
Table 2.
IVF stimulation cycle outcomes
| NE2 | iE2 | p valuea | LE2 | p valuea | vLE2 | p valuea | |
|---|---|---|---|---|---|---|---|
| Total gonadotropins (IU) | 1764.6 ± 470.6 | 1803.3 ± 439.2 | 1 | 2034.9 ± 476.7 | 0.011 | 2190.6 ± 645.7 | < 0.0001 |
| No. of days of stimulation | 9.8 ± 0.8 | 10.2 ± 0.9 | 0.257 | 10.5 ± 1.0 | < 0.0001 | 11.3 ± 1.2 | < 0.0001 |
| Max E2 (pg/mL) | 4177.3 ± 2180.5 | 3068.4 ± 1114.8 | < 0.0001 | 1736.5 ± 773.1 | < 0.0001 | 840.6 ± 390.1 | < 0.0001 |
| Gonadotropin:E2 | 0.55 ± 0.4 | 0.69 ± 0.4 | 1 | 1.43 ± 0.7 | 0.003 | 3.59 ± 2.8 | <0.0001 |
| No. of oocytes | 16.3 ± 8.2 | 22.6 ± 8.1 | < 0.0001 | 22.9 ± 8.8 | 0.0001 | 24.9 ± 8.8 | < 0.0001 |
| No. of MII | 13.6 ± 6.5 | 18.9 ± 5.9 | 0.457 | 22.4 ± 9.1 | 0.012 | 21.7 ± 9.7 | 0.034 |
| No. of 2PN | 10.7 ± 6.1 | 14.5 ± 5.7 | 0.651 | 18.5 ± 8.2 | 0.004 | 15.8 ± 5.2 | 0.186 |
| No. of embryos | 4.8 ± 3.2 | 6.3 ± 3.9 | 1 | 8.6 ± 6.0 | 0.095 | 6.9 ± 3.1 | 1 |
NE2 normal estradiol response (E2 > 200 pg/mL per oocyte); iE2 intermediate E2 response (200 > E2 > 100 pg/mL per oocyte); LE2 low E2 response (100 > E2 > 50 pg/mL per oocyte); vLE2 very low E2 response (E2 < 50 pg/mL per oocyte). Data are represented as mean ± standard deviation. Values in italics denote statistical significance, defined by p < 0.017
aANOVA with Bonferroni correction for multiple comparisons
Fig. 2.
Stimulation cycle outcomes. NE2 normal estradiol response (E2 > 200 pg/mL per oocyte); iE2 intermediate E2 response (200 > E2 > 100 pg/mL per oocyte); LE2 low E2 response (100 > E2 > 50 pg/mL per oocyte); vLE2 very low E2 response (E2 < 50 pg/mL per oocyte). Error bars indicate standard error
The 366 donor IVF cycles resulted in 340 embryo transfers (ETs) during the study period: 31 ETs from NE2 cycles, 125 ETs from iE2 cycles, 116 ETs from LE2 cycles, and 68 ETs from vLE2 cycles. The pregnancy outcomes of these ETs were comparable between all E2 cohorts, with an overall pregnancy rate of 62.4% per embryo transferred (Supplemental Table 1). Out of 366 donor IVF cycles, 24 cycles involved preimplantation genetic screening (PGS) of the embryos: five from NE2 cycles, six from iE2 cycles, nine from LE2 cycles, and four from vLE2 cycles. Although the numbers of PGS cycles was not high, there was no numerical difference in pregnancy rates between donors with normal or low estradiol responses. Percentage of euploid embryos following PGS testing did not differ significantly between E2 cohorts, with an overall euploid rate of 65.6%.
Oocyte donors may complete a maximum of six lifetime donor cycles through the CUFC donor program. During the study period, 87 donors underwent more than one stimulation cycle. Of donors with repeat cycles, 62 donors were identified with at least one LE2 or vLE2 cycle, and 33 donors exhibited multiple LE2 or vLE2 cycles. Fourteen donors had LE2 or vLE2 with every IVF stimulation cycle.
Additional hormone assays
Serum hormone assays were conducted for nine donors who underwent IVF stimulation and retrieval during the study data collection period: six with LE2 response, two with iE2 response, and one with NE2 response. Serum E2 from date of trigger was repeated on a second assay system and found to be similar to prior measurement. Serum AMH from date of trigger was measured and compared to baseline AMH. While our practice does not routinely measure serum AMH on date of trigger, these data were collected for the purposes of this study in order to further assess granulosa cell function following gonadotropin stimulation. Serum AMH normally shows a small decline during gonadotropin stimulation, and here, values at trigger showed a similar pattern between women with different E2 responses.
Follicular fluid hormone assays
Follicular fluid hormone assays were conducted for these same nine donors who underwent IVF stimulation and oocyte retrieval during the study data collection period. Follicular fluid from LE2 donors paralleled the serum findings. Follicular fluid progesterone was high in all subjects (mean 8210 ng/mL). Follicular fluid AMH closely approximated serum AMH on date of oocyte trigger and was not lower in women with low E2 responses.
Discussion
While IVF techniques and technology have evolved through the decades, E2 has remained a constant in cycle management, enabling dose adjustments and timing of trigger to optimize patient outcomes. In autologous cycles, low E2 most often portends poor outcome [2]. Similarly, very high estradiol levels can negatively affect cycle and pregnancy outcomes, such as E2 to oocyte ratios greater than 350 pg/mL [5–7] or in patients with ovarian hyperstimulation syndrome [8, 9]. Prior studies have indicated an optimal E2 to oocyte ratio of at least 100–200 pg/mL which correlates with best embryo development and pregnancy outcomes [5]. Beyond predicting cycle outcome, E2 levels allow providers to appropriately counsel patients, set expectations, and cancel IVF cycles when faced with poor outlook.
Our study suggests that healthy, non-infertile oocyte donors do not conform to this trend. In fact, just over half (n = 184, 50.2%) of our donor cycles studied demonstrated an E2 to oocyte ratio less than 100 pg/mL, considered suboptimal in prior studies, and 74 cycles (20.2%) demonstrated an E2 to oocyte ratio less than 50 pg/mL. Despite no differences in age or baseline ovarian reserve characteristics, donor cycles with low E2 response received significantly greater total gonadotropin dose and longer stimulation, resulting in higher oocyte yield and subsequently improved embryo development compared to donor cycles with typical E2 response. Pregnancy outcomes and percentage of euploid embryos following PGS were similar between typical and low estradiol response cycles.
In the past, altered gonadotropin responses were due, at least in part, to less pure urinary-based gonadotropin preparations with batch to batch variability [10]. This is not considered to be relevant here in that only FSH recombinant preparations were used.
Serum and follicular fluid hormonal assays were performed to further investigate granulosa cell function in cycles with low E2 response. In human females, AMH is produced exclusively by the preantral and antral follicle granulosa cells [11]. Baseline serum AMH is a well-known marker of ovarian reserve in infertile patients and a useful predictor of response to ovarian stimulation [12–16]. At the CUFC, AMH is also used as a screening criterion for women applying to the oocyte donation program. Further studies of AMH during IVF ovarian stimulation cycles have shown a positive correlation between AMH level, oocyte quality, and embryo quality [17]. Similarly, analysis of follicular fluid AMH has shown a positive correlation with oocyte fertilization [11, 18], embryo quality [11], and embryo implantation [19].
In our study, baseline serum AMH of oocyte donors with low E2 response did not differ from those with normal E2 response. Day of trigger AMH between low E2 and normal E2 are likely similar, although our analysis is limited by low sample size. Values are similar to previously reported serum AMH levels on day of trigger [17]. In contrast, follicular fluid AMH in our cohort demonstrated wider variability, with a mean level of 7.0 ng/mL in low E2 cycles versus 3.9 ng/mL in cycles with E2 to oocyte ratio greater than 100 pg/mL. These levels are congruent with previously reported follicular fluid AMH levels [11] and interestingly would predict that our donor cycles with low E2 response may have higher quality embryos. However, our small sample size limits these conclusions.
Repeat E2 on day of trigger was similar to the original values for all tested donors, with an expected minimal decrease following the freeze-thaw process. Follicular fluid E2 paralleled serum E2, demonstrating lower values in LE2 donor cycles. The follicular fluid E2 from our LE2 cycles is lower than previously reported normal ranges expected from preovulatory follicles: 530.05 ± 211.56 ng/mL [20] or 301.00–577.00 ng/mL [21]. However, decreased follicular fluid E2 did not correlate with poor fertilization, embryo quality, or pregnancy outcomes, consistent with prior findings by Foong et al. [22]
Follicular fluid progesterone levels were uniformly high and consistent with previously reported ranges from preovulatory follicles in oocyte donors: 7652 ± 3479 ng/mL [23].
It is known that normal fertilization and embryo development may occur with low serum E2 levels in other clinical scenarios. This can occur with letrozole use in women with hormone-sensitive cancer, without significantly reducing oocyte yield [24]. Similarly, case reports of patients with congenital adrenal hyperplasia due to 17-alpha-hydroxylase deficiency have described successful in vitro fertilization cycles with subsequent conception and live births despite markedly low serum estradiol during controlled ovarian stimulation [25, 26]. These examples, in addition to our cohort of oocyte donors with low estradiol response, suggest that in select populations, a low estradiol response may not portend a poor outcome. When compared to autologous cycles with low estradiol response, these differences in outcomes may have several explanations, including reduced ovarian reserve, oocyte quality, and the effects of aging in women undergoing autologous cycles. Additionally, while prior studies have demonstrated a correlation between low estradiol and poor outcomes, the findings of our study indicate that this relationship is not causative, indicating likely numerous additional influences on final cycle outcomes.
The authors acknowledge there are limitations to this study. First, the inherent limitations as a retrospective review, and the small sample size may limit the wider application of these study findings. Furthermore, the small number of samples available for follicular fluid analysis limits our ability to draw definitive conclusions, though trends in the data were identified. Additionally, our study defined estradiol response according to oocyte retrieved. However, particularly in this population studied, there is an excellent correlation between oocyte yield at oocyte retrieval and the number determined by ultrasound follicle measurements.
Conclusion
Our study indicates that a LE2 response is common in the donor oocyte population, but that this finding does not correlate with prognosis. In these patients, ultrasound findings are more predictive of response to ovarian stimulation. Given the significantly increased doses of gonadotropins received by these donors, we suggest that ultrasound over serum E2 should be considered when adjusting dose in order to limit unnecessary gonadotropin use and limit cycle expense. Our follicular fluid analysis suggests that granulosa cells of donors with LE2 response have intact AMH and progesterone secretion, but may have altered E2 synthesis or secretion.
Electronic supplementary material
(DOCX 15 kb)
Acknowledgements
The authors would like to acknowledge Michel Ferin, Nancy Cotui, and Kimberly Hamroff for their expertise and assistance with conducting all serum and follicular fluid hormone assays. The authors also acknowledge the CUFC Embryology Division for their assistance in facilitating follicular fluid collection.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
References
- 1.Fritz M, Speroff L. Clinical gynecologic endocrinology and infertility. 8. Philadelphia: Lippincott Williams & Wilkins; 2011. [Google Scholar]
- 2.Mittal S, Gupta P, Malhotra N, Singh N. Serum estradiol as a predictor of success of in vitro fertilization. J Obstet Gynaecol India. 2014;64:124–129. doi: 10.1007/s13224-013-0470-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hu X, Luo Y, Huang K, Li Y, Xu Y, Zhou C, Mai Q. New perspectives on criteria for the determination of HCG trigger timing in GnRH antagonist cycles. Medicine. 2016;95:e3691. doi: 10.1097/MD.0000000000003691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Centers for Disease Control and Prevention, American Society for Reproductive Medicine, Society for Assisted Reproductive Technology. 2014 assisted reproductive technology national summary report: Atlanta; 2016.
- 5.Orvieto R, Zohav E, Scharf S, et al. The influence of estradiol/follicle and estradiol/oocyte ratios on the outcome of controlled ovarian stimulation for in vitro fertilization. Gynecol Endocrinol. 2007;23:72–75. doi: 10.1080/09513590601137137. [DOI] [PubMed] [Google Scholar]
- 6.Yang J, Chen H, Lien Y, Chen S, Ho H, Yang Y. Elevated E2: oocyte ratio in women undergoing IVF and tubal ET. Correlation with a decrease in the implantation rate. J Reprod Med. 2001;46:434–438. [PubMed] [Google Scholar]
- 7.Vaughan DA, Harrity C, Sills ES, Mocanu EV. Serum estradiol:oocyte ratio as a predictor of reproductive outcome: an analysis of data from >9000 IVF cycles in the Republic of Ireland. J Assist Reprod Genet. 2016;33:481–488. doi: 10.1007/s10815-016-0664-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Haas J, Baum M, Meridor K, Hershko-Klement A, Elizur S, Hourvitz A, Orvieto R, Yinon Y. Is severe OHSS associated with adverse pregnancy outcomes? Evidence from a case-control study. Reprod BioMed Online. 2014;29:216–221. doi: 10.1016/j.rbmo.2014.04.015. [DOI] [PubMed] [Google Scholar]
- 9.Raziel A, Friedler S, Schachter M, Strassburger D, Mordechai E, Ron-El R. Increased early pregnancy loss in IVF patients with severe ovarian hyperstimulation syndrome. Hum Reprod. 2002;17:107–110. doi: 10.1093/humrep/17.1.107. [DOI] [PubMed] [Google Scholar]
- 10.Karlsson MO, Wade JR, Loumaye E, Munafo A. The population pharmacokinetics of recombinant- and urinary-human follicle stimulating hormone in women. Br J Clin Pharmacol. 1998;45:13–20. doi: 10.1046/j.1365-2125.1998.00644.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kim J, Lee J, Chang H, Jee B, Suh C, Kim S. Anti-Mullerian hormone levels in the follicular fluid of the preovulatory follicle: a predictor for oocyte fertilization and quality of embryo. J Korean Med Sci. 2014;29:1266–1270. doi: 10.3346/jkms.2014.29.9.1266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Seifer D, Maclaughlin D. Mullerian inhibiting substance is an ovarian growth factor of emerging clinical significance. Fertil Steril. 2007;88:539–546. doi: 10.1016/j.fertnstert.2007.02.014. [DOI] [PubMed] [Google Scholar]
- 13.Vet AD, Laven J, Jong FD, Themmen A, Fauser B. Antimullerian hormone serum levels: a putative marker for ovarian aging. Fertil Steril. 2002;77:357–362. doi: 10.1016/S0015-0282(01)02993-4. [DOI] [PubMed] [Google Scholar]
- 14.Seifer D, MacLaughlin D, Christian B, Feng B, Shelder R. Early follicular serum mullerian-inhibiting substance levels are associated with ovarian response during assisted reproductive technology cycles. Fertil Steril. 2002;77:468–471. doi: 10.1016/S0015-0282(01)03201-0. [DOI] [PubMed] [Google Scholar]
- 15.van Rooij IA, Broekmans FJ, te Velde ER, et al. Serum anti-Mullerian hormone levels: a novel measure of ovarian reserve. Hum Reprod. 2002;17:3065–3071. doi: 10.1093/humrep/17.12.3065. [DOI] [PubMed] [Google Scholar]
- 16.Lee J, Kim S, Kim S, et al. Anti-Mullerian hormone dynamics during controlled ovarian hyperstimulation and optimal timing of measurement for outcome prediction. Hum Reprod. 2010;25:2597–2604. doi: 10.1093/humrep/deq204. [DOI] [PubMed] [Google Scholar]
- 17.Silberstein T, MacLaughlin D, Shai I, et al. Mullerian inhibiting substance levels at the time of HCG administration in IVF cycles predict both ovarian reserve and embryo morphology. Hum Reprod. 2006;21:159–163. doi: 10.1093/humrep/dei270. [DOI] [PubMed] [Google Scholar]
- 18.Takahashi C, Fujito A, Kazuka M, Sugiyama R, Ito H, Isaka K. Anti-Mullerian hormone substance from follicular fluid is positively associated with success in oocyte fertilization during in vitro fertilization. Fertil Steril. 2008;89:586–591. doi: 10.1016/j.fertnstert.2007.03.080. [DOI] [PubMed] [Google Scholar]
- 19.Fanchin R, Lozano DM, Frydman N, et al. Anti-Mullerian hormone concentrations in the follicular fluid of the preovulatory follicle are predictive of the implantation potential of the ensuing embryo obtained by in vitro fertilization. J Clin Endocrinol Metab. 2007;92:1796–1802. doi: 10.1210/jc.2006-1053. [DOI] [PubMed] [Google Scholar]
- 20.Li L, Ferin M, Sauer M, Lobo R. Dehydroepiandrosterone in follicular fluid is produced locally, and levels correlate negatively with in vitro fertilization outcomes. Fertil Steril. 2011;95:1830–1832. doi: 10.1016/j.fertnstert.2010.11.007. [DOI] [PubMed] [Google Scholar]
- 21.Li L, Ferin M, Sauer M, Lobo R. Serum and follicular fluid ghrelin levels negatively reflect human oocyte quality and in vitro embryo development. Fertil Steril. 2011;96:1116–1120. doi: 10.1016/j.fertnstert.2011.08.017. [DOI] [PubMed] [Google Scholar]
- 22.Foong S, Abbott D, Lesnick T, Session D, Walker D, Dumesic D. Diminished intrafollicular estradiol levels in in vitro fertilization cycles from women with reduced ovarian response to recombinant human follicle-stimulating hormone. Fertil Steril. 2005;83:1377–1383. doi: 10.1016/j.fertnstert.2004.11.041. [DOI] [PubMed] [Google Scholar]
- 23.de los Santos MJ, Garcia-Laez V, Beltran-Torregrosa D, et al. Hormonal and molecular characterization of follicular fluid, cumulus cells and oocytes from pre-ovulatory follicles in stimulated and unstimulated cycles. Hum Reprod. 2012;27:1596–1605. doi: 10.1093/humrep/des082. [DOI] [PubMed] [Google Scholar]
- 24.Rodgers RJ, Reid GD, Koch J, Deans R, Ledger WL, Friedlander M, Gilchrist RB, Walters KA, Abbott JA. The safety and efficacy of controlled ovarian hyperstimulation for fertility preservation in women with early breast cancer: a systematic review. Hum Reprod. 2017;32:1033–1045. doi: 10.1093/humrep/dex027. [DOI] [PubMed] [Google Scholar]
- 25.Bianchi PH, Gouveia GR, Costa EM, et al. Successful live birth in a woman with 17alpha-hydroxylase deficiency through IVF frozen-thawed embryo transfer. J Clin Endocrinol Metab. 2016;101:345–348. doi: 10.1210/jc.2015-3201. [DOI] [PubMed] [Google Scholar]
- 26.Kitajima M, Miura K, Inoue T, et al. Two consecutive successful live birth in woman with 17alpha hydroxylase deficiency by frozen-thaw embryo transfer under hormone replacement endometrium preparation. Gynecol Endocrinol. 2017;34:1–4. doi: 10.1080/09513590.2017.1393512. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX 15 kb)