This article reviews scientific advances in the prevention and treatment of substance-use disorder and related developments in public policy. In the past two decades, research has increasingly supported the view that addiction is a disease of the brain. Although the brain disease model of addiction has yielded effective preventive measures, treatment interventions, and public health policies to address substance-use disorders, the underlying concept of substance abuse as a brain disease continues to be questioned, perhaps because the aberrant, impulsive, and compulsive behaviors that are characteristic of addiction have not been clearly tied to neurobiology. Here we review recent advances in the neurobiology of addiction to clarify the link between addiction and brain function and to broaden the understanding of addiction as a brain disease. We review findings on the desensitization of reward circuits, which dampens the ability to feel pleasure and the motivation to pursue everyday activities; the increasing strength of conditioned responses and stress reactivity, which results in increased cravings for alcohol and other drugs and negative emotions when these cravings are not sated; and the weakening of the brain regions involved in executive functions such as decision making, inhibitory control, and self-regulation that leads to repeated relapse. We also review the ways in which social environments, developmental stages, and genetics are intimately linked to and influence vulnerability and recovery. We conclude that neuroscience continues to support the brain disease model of addiction. Neuroscience research in this area not only offers new opportunities for the prevention and treatment of substance addictions and related behavioral addictions (e.g., to food, sex, and gambling) but may also improve our understanding of the fundamental biologic processes involved in voluntary behavioral control.
In the United States, 8 to 10% of people 12 years of age or older, or 20 to 22 million people, are addicted to alcohol or other drugs.1 The abuse of tobacco, alcohol, and illicit drugs in the United States exacts more than $700 billion annually in costs related to crime, lost work productivity, and health care.2–4 After centuries of efforts to reduce addiction and its related costs by punishing addictive behaviors failed to produce adequate results, recent basic and clinical research has provided clear evidence that addiction might be better considered and treated as an acquired disease of the brain (see Box 1 for definitions of substance-use disorder and addiction). Research guided by the brain disease model of addiction has led to the development of more effective methods of prevention and treatment and to more informed public health policies. Notable examples include the Mental Health Parity and Addiction Equity Act of 2008, which requires medical insurance plans to provide the same coverage for substance-use disorders and other mental illnesses that is provided for other illnesses,5 and the proposed bipartisan Senate legislation that would reduce prison sentences for some nonvio-lent drug offenders,6 which is a substantial shift in policy fueled in part by the growing realization among law-enforcement leaders that “reducing incarceration will improve public safety because people who need treatment for drug and alcohol problems or mental health issues will be more likely to improve and reintegrate into society if they receive consistent care.”7
Box 1. Definitions.
In this article, the terms apply to the use of alcohol, tobacco and nicotine, prescription drugs, and illegal drugs.
Substance-use disorder: A diagnostic term in the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) referring to recurrent use of alcohol or other drugs that causes clinically and functionally significant impairment, such as health problems, disability, and failure to meet major responsibilities at work, school, or home. Depending on the level of severity, this disorder is classified as mild, moderate, or severe.
Addiction: A term used to indicate the most severe, chronic stage of substance-use disorder, in which there is a substantial loss of self-control, as indicated by compulsive drug taking despite the desire to stop taking the drug. In the DSM-5, the term addiction is synonymous with the classification of severe substance-use disorder.
Nonetheless, despite the scientific evidence and the resulting advances in treatment and changes in policy, the concept of addiction as a disease of the brain is still being questioned. The concept of addiction as a disease of the brain challenges deeply ingrained values about self-determination and personal responsibility that frame drug use as a voluntary, hedonistic act. In this view, addiction results from the repetition of voluntary behaviors. How, then, can it be the result of a disease process? The concept of addiction as a brain disease has even more disconcerting implications for public attitudes and policies toward the addict. This concept of addiction appears to some to excuse personal irresponsibility and criminal acts instead of punishing harmful and often illegal behaviors. Additional criticisms of the concept of addiction as a brain disease include the failure of this model to identify genetic aberrations or brain abnormalities that consistently apply to persons with addiction and the failure to explain the many instances in which recovery occurs without treatment. (Arguments against the disease model of addiction and counterarguments in favor of it8 are presented in Box S1 in the Supplementary Appendix, available with the full text of this article at NEJM.org.)
Advances in neurobiology have begun to clarify the mechanisms underlying the profound disruptions in decision-making ability and emotional balance displayed by persons with drug addiction. These advances also provide insight into the ways in which fundamental biologic processes, when disrupted, can alter voluntary behavioral control, not just in drug addiction but also in other, related disorders of self-regulation, such as obesity and pathologic gambling and video-gaming — the so-called behavioral addictions. Although these disorders also manifest as compulsive behaviors, with impaired self-regulation, the concept of behavioral addiction is still controversial, particularly as it relates to obesity. (Behavioral addictions are described in Box S2 in the Supplementary Appendix.9) This research has also begun to show how and why early, voluntary drug use can interact with environmental and genetic factors to result in addiction in some persons but not in others.
STAGES OF ADDICTION
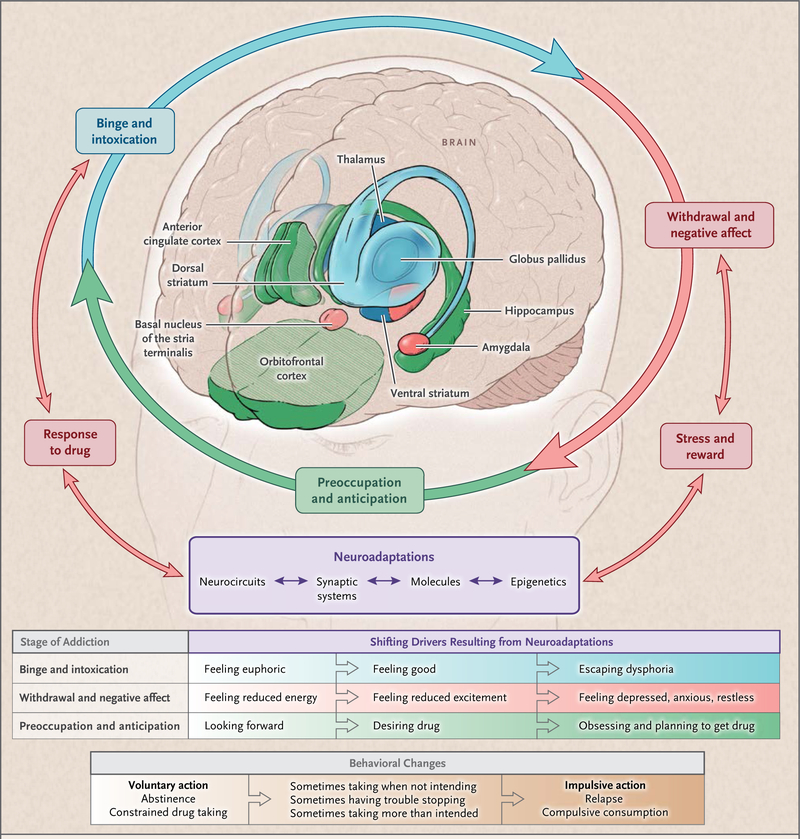
For heuristic purposes, we have divided addiction into three recurring stages: binge and intoxication, withdrawal and negative affect, and preoccupation and anticipation (or craving).10 Each stage is associated with the activation of specific neurobiologic circuits and the consequential clinical and behavioral characteristics (Fig. 1).
Figure 1. Stages of the Addiction Cycle.
During intoxication, drug-induced activation of the brain’s reward regions (in blue) is enhanced by conditioned cues in areas of increased sensitization (in green). During withdrawal, the activation of brain regions involved in emotions (in pink) results in negative mood and enhanced sensitivity to stress. During preoccupation, the decreased function of the prefrontal cortex leads to an inability to balance the strong desire for the drug with the will to abstain, which triggers relapse and reinitiates the cycle of addiction. The compromised neuro-circuitry reflects the disruption of the dopamine and glutamate systems and the stress-control systems of the brain, which are affected by corticotropin-releasing factor and dynorphin. The behaviors during the three stages of addiction change as a person transitions from drug experimentation to addiction as a function of the progressive neuroadaptations that occur in the brain.
BINGE AND INTOXICATION
All known addictive drugs activate reward regions in the brain by causing sharp increases in the release of dopamine.11–13 At the receptor level, these increases elicit a reward signal that triggers associative learning or conditioning. In this type of Pavlovian learning, repeated experiences of reward become associated with the environmental stimuli that precede them. With repeated exposure to the same reward, dopamine cells stop firing in response to the reward itself and instead fire in an anticipatory response to the conditioned stimuli (referred to as “cues”) that in a sense predict the delivery of the reward.14 This process involves the same molecular mechanisms that strengthen synaptic connections during learning and memory formation (Box 2). In this way, environmental stimuli that are repeatedly paired with drug use — including environments in which a drug has been taken, persons with whom it has been taken, and the mental state of a person before it was taken — may all come to elicit conditioned, fast surges of dopamine release that trigger craving for the drug20 (see Box 2 for the mechanisms involved), motivate drug-seeking behaviors, and lead to heavy “binge” use of the drug.21–23 These conditioned responses become deeply ingrained and can trigger strong cravings for a drug long after use has stopped (e.g., owing to incarceration or treatment) and even in the face of sanctions against its use.
Box 2. Drug-Induced Neuroplasticity.
The drug-induced release of dopamine triggers neuroplasticity (systematic changes in the synaptic signaling, or communication, between neurons in various reward regions of the brain).15,16 These neuroplastic changes are fundamental to learning and memory. Experience-dependent learning (such as that which occurs in repeated episodes of drug use) may invoke both long-term potentiation, in which the transmission of signals between neurons increases, and long-term depression, in which signal transmission decreases.
Synaptic strength is controlled by the insertion or removal of receptors that are stimulated by the excitatory neurotransmitter glutamate (which acts largely through α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid [AMPA] and N-methyl-D-aspartate [NMDA] receptors) and by changes in the composition of the subunits of these receptors. Specifically, the insertion of a subunit of the AMPA receptor that is highly permeable to calcium, glutamate receptor 2 (GluR2), enhances the efficiency of transmission and has been shown to contribute to long-term potentiation in animal studies of addiction.17 Changes in long-term potentiation and long-term depression are in turn associated with larger or smaller synapses, respectively, and with differences in the shapes of the dendritic spines in the receptive site of the receiving neuron.18
The up-regulation of AMPA receptors that are highly permeable to calcium increases the responsiveness of the nucleus accumbens to glutamate, which is released by cortical and limbic terminals when exposed to drugs or drug cues.17 Neuroplastic changes triggered by drugs have been uncovered not only in the nucleus accumbens (a crucial brain-reward region) but also in the dorsal striatum (a region implicated in the encoding of habits and routines), the amygdala (a region involved in emotions, stress, and desires), the hippocampus (a region involved in memory), and the prefrontal cortex (a region involved in self-regulation and the attribution of salience [the assignment of relative value]). All these regions of the brain participate in the various stages of addiction, including conditioning and craving (see Fig. 1). These regions also regulate the firing of dopamine cells and the release of dopamine.19
As is true with other types of motivational learning, the greater the motivational attribute associated with a reward (e.g., a drug), the greater the effort a person is willing to exert and the greater the negative consequences he or she will be willing to endure in order to obtain it.24,25 However, whereas dopamine cells stop firing after repeated consumption of a “natural reward” (e.g., food or sex) satiating the drive to further pursue it, addictive drugs circumvent natural satiation and continue to directly increase dopamine levels,11,26 a factor that helps to explain why compulsive behaviors are more likely to emerge when people use drugs than when they pursue a natural reward (Box 2).
WITHDRAWAL AND NEGATIVE AFFECT
An important result of the conditioned physiologic processes involved in drug addiction is that ordinary, healthful rewards lose their former motivational power. In a person with addiction, the reward and motivational systems become reoriented through conditioning to focus on the more potent release of dopamine produced by the drug and its cues. The landscape of the person with addiction becomes restricted to one of cues and triggers for drug use. However, this is only one of the ways in which addiction changes motivation and behavior.
For many years it was believed that over time persons with addiction would become more sensitive to the rewarding effects of drugs and that this increased sensitivity would be reflected in higher levels of dopamine in the circuits of their brains that process reward (including the nucleus accumbens and the dorsal striatum) than the levels in persons who never had a drug addiction. Although this theory seemed to make sense, research has shown that it is incorrect. In fact, clinical and preclinical studies have shown that drug consumption triggers much smaller increases in dopamine levels in the presence of addiction (in both animals and humans) than in its absence (i.e., in persons who have never used drugs).22,23,27,28 This attenuated release of dopa-mine renders the brain’s reward system much less sensitive to stimulation by both drug-related and non–drug-related rewards.29–31 As a result, persons with addiction no longer experience the same degree of euphoria from a drug as they did when they first started using it. It is for this same reason that persons with addiction often become less motivated by everyday stimuli (e.g., relationships and activities) that they had previously found to be motivating and rewarding. Again, it is important to note that these changes become deeply ingrained and cannot be immediately reversed through the simple termination of drug use (e.g., detoxification).
In addition to resetting the brain’s reward system, repeated exposure to the dopamine-enhanc ing effects of most drugs leads to adaptations in the circuitry of the extended amygdala in the basal forebrain; these adaptations result in increases in a person’s reactivity to stress and lead to the emergence of negative emotions.32,33 This “antireward” system is fueled by the neurotransmitters involved in the stress response, such as corticotropin-releasing factor and dynorphin, which ordinarily help to maintain homeostasis. However, in the addicted brain, the antireward system becomes overactive, giving rise to the highly dysphoric phase of drug addiction that ensues when the direct effects of the drug wear off or the drug is withdrawn34 and to the decreased reactivity of dopamine cells in the brain’s reward circuitry.35 Thus, in addition to the direct and conditioned pull toward the “rewards” of drug use, there is a correspondingly intense motivational push to escape the discomfort associated with the aftereffects of use. As a result of these changes, the person with addiction transitions from taking drugs simply to feel pleasure, or to “get high,” to taking them to obtain transient relief from dysphoria (Fig. 1).
Persons with addiction frequently cannot understand why they continue to take the drug when it no longer seems pleasurable. Many state that they continue to take the drug to escape the distress they feel when they are not intoxicated. Unfortunately, although the short-acting effects of increased dopamine levels triggered by drug administration temporarily relieve this distress, the result of repeated bingeing is to deepen the dysphoria during withdrawal, thus producing a vicious cycle.
PREOCCUPATION AND ANTICIPATION
The changes that occur in the reward and emotional circuits of the brain are accompanied by changes in the function of the prefrontal cortical regions, which are involved in executive processes. Specifically, the down-regulation of dopamine signaling that dulls the reward circuits’ sensitivity to pleasure also occurs in pre-frontal brain regions and their associated circuits, seriously impairing executive processes, among which are the capacities for self-regulation, decision making, flexibility in the selection and initiation of action, attribution of salience (the assignment of relative value), and the monitoring of error.36 The modulation of the reward and emotional circuits of prefrontal regions is further disrupted by neuroplastic changes in glutamatergic signaling.37 In persons with addiction, the impaired signaling of dopamine and glutamate in the prefrontal regions of the brain weakens their ability to resist strong urges or to follow through on decisions to stop taking the drug. These effects explain why persons with addiction can be sincere in their desire and intention to stop using a drug and yet simultaneously impulsive and unable to follow through on their resolve. Thus, altered signaling in prefrontal regulatory circuits, paired with changes in the circuitry involved in reward and emotional response, creates an imbalance that is crucial to both the gradual development of compulsive behavior in the addicted disease state and the associated inability to voluntarily reduce drug-taking behavior, despite the potentially catastrophic consequences.
BIOLOGIC AND SOCIAL FACTORS INVOLVED IN ADDICTION
Only a minority of people who use drugs ultimately become addicted — just as not everyone is equally at risk for the development of other chronic diseases. Susceptibility differs because people differ in their vulnerability to various genetic, environmental, and developmental factors. Many genetic, environmental, and social factors contribute to the determination of a person’s unique susceptibility to using drugs initially, sustaining drug use, and undergoing the progressive changes in the brain that characterize addiction.38,39 Factors that increase vulnerability to addiction include family history (presumably through heritability and child-rearing practices), early exposure to drug use (adolescence is among the periods of greatest vulnerability to addiction), exposure to high-risk environments (typically, socially stressful environments with poor familial and social supports and restricted behavioral alternatives and environments in which there is easy access to drugs and permissive normative attitudes toward drug taking), and certain mental illnesses (e.g., mood disorders, attention deficit–hyperactivity disorder, psychoses, and anxiety disorders).40,41
It is estimated that the most severe phenotypic characteristics of addiction will develop in approximately 10% of persons exposed to addictive drugs.42 Thus, although long-term exposure to drugs is a necessary condition for the development of addiction, it is by no means sufficient. Yet for those in whom there is progress to addiction, the neurobiologic changes are distinct and profound.
IMPLICATIONS OF THE BRAIN DISEASE MODEL OF ADDICTION FOR PREVENTION AND TREATMENT
As is the case in other medical conditions in which voluntary, unhealthful behaviors contribute to disease progression (e.g., heart disease, diabetes, chronic pain, and lung cancer), evidence-based interventions aimed at prevention, along with appropriate public health policies, are the most effective ways of changing outcomes. A more comprehensive understanding of the brain disease model of addiction may help to moderate some of the moral judgment attached to addictive behaviors and foster more scientif and public health–oriented approaches to prevention and treatment.
BEHAVIORAL AND MEDICAL INTERVENTIONS
The findings from neurobiologic research show that addiction is a disease that emerges gradually and that has its onset predominantly during a particular risk period: adolescence. Adolescence is a time when the still-developing brain is particularly sensitive to the effects of drugs, a factor that contributes to adolescents’ greater vulnerability to drug experimentation and addiction. Adolescence is also a period of enhanced neuroplasticity during which the underdeveloped neural networks necessary for adult-level judgment (the prefrontal cortical regions) cannot yet properly regulate emotion. Studies have also shown that children and adolescents with evidence of structural or functional changes in frontal cortical regions or with traits of novelty seeking or impulsivity are at greater risk for substance-use disorders.43–45 Awareness of individual and social risk factors and the identification of early signs of substance-use problems make it possible to tailor prevention strategies to the patient. According to research related to the brain disease model of addiction, preventive in-terventions should be designed to enhance social skills and improve self-regulation. Also important are early screening and intervention for the prodromal presentation of mental illness and the provision of social opportunities for personal educational and emotional development.46–49
When prevention has failed and there is need for treatment, research based on the brain disease model of addiction has shown that medical treatment can help to restore healthy function in the affected brain circuitry and lead to improvements in behavior. The health care system already has at its disposal several evidence-based treatment interventions that could improve clinical outcomes in patients with substance-use disorders if properly and comprehensively implemented. During treatment, medication can assist in preventing relapse while the brain is healing and normal emotional and decision-making capacities are being restored. For patients with opioid-use disorder, maintenance therapy with agonists or partial agonists such as methadone or buprenorphine can be essential in helping to control symptoms of withdrawal and cravings.50 Opioid antagonists such as extended-release naltrexone may be used to prevent opioid intoxication.51 Naltrexone and acamprosate have been efficacious in the treatment of alcohol-use disorders, and other medications can help in the recovery from nicotine addiction.27
The brain disease model of addiction has also fostered the development of behavioral interventions to help restore balance in brain circuitry that has been affected by drugs.52 For example, strategies to enhance the salience of natural, healthy rewards such as social contact or exercise could enable those rewards to compete with the direct and acquired motivating properties of drugs. Strategies to mitigate a person’s stress reactivity and negative emotional states could help to manage the strong urges they engender, and strategies to improve executive function and self-regulation could help recovering patients plan ahead in order to avoid situations in which they are particularly vulnerable to taking drugs. Finally, strategies to help patients recovering from addiction to change their circle of friends and to avoid drug-associated environmental cues can reduce the likelihood that conditioned craving will lead to relapse.
PUBLIC HEALTH POLICY
A compelling argument for the translational value of the brain disease model of addiction is the knowledge that the prefrontal and other cortical networks that are so critical for judgment and self-regulation do not fully mature until people reach 21 to 25 years of age.53 As a result, the adolescent brain is much less able to cognitively modulate strong desires and emotions. This observation is particularly relevant to the establishment of 21 years of age as the legal drinking age in the United States, a ruling that is often questioned even though a dramatic reduction in highway deaths followed its institution.54 One could legitimately argue that the study of the neurobiology of addiction provides a compelling argument for leaving the drinking age at 21 years and for increasing the legal smoking age to 21 years, by which time the brain networks that underlie the capacity for self-regulation are more fully formed.
The brain disease model of addiction has also informed policies that take advantage of the infrastructure of primary health care to address substance-use disorders and to provide a model for paying for it through the Mental Health Parity and Addiction Equity Act (MHPAEA) and the Affordable Care Act. Although it is still too early to evaluate the effects of these policies on the nation, an initial examination of the MHPAEA in three states showed increased enrollment and care delivery among patients with substance-use disorders and an overall reduction in spending on emergency department visits and hospital stays.55
The social and financial effects of these laws are also illustrated in the recent legal action taken by the State of New York against Value Options and two other managed-care organiza tions for alleged discrimination against patients who were wrongly denied benefits related to addiction and mental health after patients with diabetes were used as the comparators. The action was taken on the basis of the amount and extent of preauthorization required for the treatment of patients with substance-use disorder versus those with diabetes, the arbitrary and capricious manner in which the insurers stopped treatment, and the lack of treatment alternatives offered or even suggested to patients.56 The settlement has not been contested, and the organizations stopped their discriminatory preauthorization procedures. A similar suit has been filed in California.
Similarly, there are early indications that the integration of primary care and specialty behavioral health care can substantially improve the management of substance-use disorders and the treatment of many addiction-related medical conditions, including the human immunodeficiency virus, hepatitis C virus, cancer, cirrhosis, and trauma.57,58
Despite such reports of benefits to the public from practices and policies generated by research based on the brain disease model of addiction, mobilizing support for further research will require the public to become better educated about the genetic, age-related, and environmental susceptibilities to addiction as they relate to structural and functional changes in the brain. If early voluntary drug use goes undetected and unchecked, the resulting changes in the brain can ultimately erode a person’s ability to control the impulse to take addictive drugs.
Supplementary Material
Footnotes
Dr. McLellan reports receiving fees for serving on the board of directors of Indivior Pharmaceuticals. No other potential conflict of interest relevant to this article was reported.
Disclosure forms are available with the full text of this article at NEJM.org.
REFERENCES
- 1.Results from the 2013 National Sur-vey on Drug Use and Health: summary of national findings. Rockville, MD: Sub-stance Abuse and Mental Health Services Administration, Center for Behavioral Health Statistics and Quality, 2013. [Google Scholar]
- 2.The health consequences of smoking — 50 years of progress. Rockville, MD: Department of Health and Human Services, 2014. [Google Scholar]
- 3.Excessive drinking costs U.S. $223.5 billion. April 17, 2014 (http://www.cdc.gov/features/alcoholconsumption).
- 4.National drug threat assessment, 2011. Washington, DC: Department of Justice, National Drug Intelligence Center, 2011. [Google Scholar]
- 5.Busch SH, Epstein AJ, Harhay MO et al. The effects of federal parity on substance use disorder treatment. Am J Manag Care 2014;20:76–82. [PMC free article] [PubMed] [Google Scholar]
- 6.US Senate working to cut sentences, lower prison population. Voice of America. October 1, 2015 (https://www.voanews.com/a/us-senate-working-to-cut-sentences-to-lower-prison-population/2987683.html).
- 7.Williams T Police leaders join call to cut prison rosters New York Times. October 20, 2015:A1.
- 8.Volkow ND, Koob G. Brain disease model of addiction: why is it so controversial? Lancet Psychiatry 2015;2:677–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Potenza M Perspective: behavioural addictions matter. Nature 2015; 522: S62. [DOI] [PubMed] [Google Scholar]
- 10.Koob GF, Volkow ND. Neurocircuitry of addiction. Neuropsychopharmacology 2010; 35: 217–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Di Chiara G Nucleus accumbens shell and core dopamine: differential role in behavior and addiction. Behav Brain Res 2002; 137: 75–114. [DOI] [PubMed] [Google Scholar]
- 12.Koob GF. Neural mechanisms of drug reinforcement. Ann N Y Acad Sci 1992; 654: 171–91. [DOI] [PubMed] [Google Scholar]
- 13.Wise RA. Dopamine and reward: the anhedonia hypothesis 30 years on. Neurotox Res 2008; 14: 169–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schultz W Getting formal with dopa-mine and reward. Neuron 2002; 36: 241–63. [DOI] [PubMed] [Google Scholar]
- 15.Kauer JA, Malenka RC. Synaptic plasticity and addiction. Nat Rev Neurosci 2007; 8: 844–58. [DOI] [PubMed] [Google Scholar]
- 16.Kourrich S, Calu DJ, Bonci A. Intrinsic plasticity: an emerging player in addiction. Nat Rev Neurosci 2015; 16: 173–84. [DOI] [PubMed] [Google Scholar]
- 17.Wolf ME, Ferrario CR. AMPA receptor plasticity in the nucleus accumbens after repeated exposure to cocaine. Neurosci Biobehav Rev 2010; 35: 185–211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.De Roo M, Klauser P, Garcia PM, Poglia L, Muller D. Spine dynamics and synapse remodeling during LTP and memory processes. Prog Brain Res 2008; 169: 199–207. [DOI] [PubMed] [Google Scholar]
- 19.Volkow ND, Morales M. The brain on drugs: from reward to addiction. Cell 2015; 162: 712–25. [DOI] [PubMed] [Google Scholar]
- 20.Volkow ND, Wang GJ, Telang F, et al. Cocaine cues and dopamine in dorsal striatum: mechanism of craving in cocaine addiction. J Neurosci 2006; 26: 6583–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Weiss F Neurobiology of craving, conditioned reward and relapse. Curr Opin Pharmacol 2005; 5: 9–19. [DOI] [PubMed] [Google Scholar]
- 22.Volkow ND, Wang GJ, Fowler JS, et al. Decreased striatal dopaminergic responsiveness in detoxified cocaine-dependent subjects. Nature 1997; 386: 830–3. [DOI] [PubMed] [Google Scholar]
- 23.Zhang Y, Schlussman SD, Rabkin J, Butelman ER, Ho A, Kreek MJ. Chronic escalating cocaine exposure, abstinence/withdrawal, and chronic re-exposure: effects on striatal dopamine and opioid systems in C57BL/6J mice. Neuropharmacology 2013; 67: 259–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Trifilieff P, Feng B, Urizar E, et al. Increasing dopamine D2 receptor expression in the adult nucleus accumbens enhances motivation. Mol Psychiatry 2013; 18: 1025–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Saddoris MP, Cacciapaglia F, Wightman RM, Carelli RM. Differential dopa-mine release dynamics in the nucleus accumbens core and shell reveal complementary signals for error prediction and incentive motivation. J Neurosci 2015; 35: 11572–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wise RA. Brain reward circuitry: insights from unsensed incentives. Neuron 2002; 36: 229–40. [DOI] [PubMed] [Google Scholar]
- 27.Müller CA, Geisel O, Banas R, Heinz A. Current pharmacological treatment approaches for alcohol dependence. Expert Opin Pharmacother 2014; 15: 471–81. [DOI] [PubMed] [Google Scholar]
- 28.Volkow ND, Tomasi D, Wang GJ, et al. Stimulant-induced dopamine increases are markedly blunted in active cocaine abusers. Mol Psychiatry 2014; 19: 1037–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hägele C, Schlagenhauf F, Rapp M, et al. Dimensional psychiatry: reward dys-function and depressive mood across psychiatric disorders. Psychopharmacology (Berl) 2015; 232: 331–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hyatt CJ, Assaf M, Muska CE, et al. Reward-related dorsal striatal activity differences between former and current cocaine dependent individuals during an interactive competitive game. PLoS One 2012; 7(5): e34917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Konova AB, Moeller SJ, Tomasi D, et al. Structural and behavioral correlates of abnormal encoding of money value in the sensorimotor striatum in cocaine addiction. Eur J Neurosci 2012; 36: 2979–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Davis M, Walker DL, Miles L, Grillon C. Phasic vs sustained fear in rats and humans: role of the extended amygdala in fear vs anxiety. Neuropsychopharmacology 2010; 35: 105–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jennings JH, Sparta DR, Stamatakis AM, et al. Distinct extended amygdala circuits for divergent motivational states. Nature 2013; 496: 224–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Koob GF, Le Moal M. Plasticity of reward neurocircuitry and the ‘dark side’ of drug addiction. Nat Neurosci 2005; 8: 1442–4. [DOI] [PubMed] [Google Scholar]
- 35.Kaufling J, Aston-Jones G. Persistent adaptations in afferents to ventral teg-mental dopamine neurons after opiate withdrawal. J Neurosci 2015; 35: 10290–303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Goldstein RZ, Volkow ND. Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat Rev Neurosci 2011; 12: 652–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Britt JP, Bonci A. Optogenetic interrogations of the neural circuits underlying addiction. Curr Opin Neurobiol 2013; 23: 539–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Demers CH, Bogdan R, Agrawal A. The genetics, neurogenetics and pharmacogenetics of addiction. Curr Behav Neurosci Rep 2014; 1: 33–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Volkow ND, Muenke M. The genetics of addiction. Hum Genet 2012; 131: 773–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Burnett-Zeigler I, Walton MA, Ilgen M, et al. Prevalence and correlates of mental health problems and treatment among adolescents seen in primary care. J Adolesc Health 2012; 50: 559–64. [DOI] [PubMed] [Google Scholar]
- 41.Stanis JJ, Andersen SL. Reducing substance use during adolescence: a translational framework for prevention. Psycho-pharmacology (Berl) 2014; 231: 1437–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Warner LA, Kessler RC, Hughes M, Anthony JC, Nelson CB. Prevalence and correlates of drug use and dependence in the United States: results from the National Comorbidity Survey. Arch Gen Psychiatry 1995; 52: 219–29. [DOI] [PubMed] [Google Scholar]
- 43.Castellanos-Ryan N, Rubia K, Conrod PJ. Response inhibition and reward response bias mediate the predictive relationships between impulsivity and sensation seeking and common and unique variance in conduct disorder and substance misuse. Alcohol Clin Exp Res 2011; 35: 140–55. [DOI] [PubMed] [Google Scholar]
- 44.Nees F, Tzschoppe J, Patrick CJ, et al. Determinants of early alcohol use in healthy adolescents: the differential contribution of neuroimaging and psychological factors. Neuropsychopharmacology 2012; 37: 986–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Quinn PD, Harden KP. Differential changes in impulsivity and sensation seeking and the escalation of substance use from adolescence to early adulthood. Dev Psychopathol 2013; 25: 223–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Durlak JA, Weissberg RP, Dymnicki AB, Taylor RD, Schellinger KB. The impact of enhancing students’ social and emotional learning: a meta-analysis of school-based universal interventions. Child Dev 2011; 82: 405–32. [DOI] [PubMed] [Google Scholar]
- 47.Greenberg MT, Lippold MA. Promoting healthy outcomes among youth with multiple risks: innovative approaches. Annu Rev Public Health 2013; 34: 253–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sandler I, Wolchik SA, Cruden G, et al. Overview of meta-analyses of the prevention of mental health, substance use, and conduct problems. Annu Rev Clin Psychol 2014; 10: 243–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kiluk BD, Carroll KM. New developments in behavioral treatments for substance use disorders. Curr Psychiatry Rep 2013; 15: 420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bell J Pharmacological maintenance treatments of opiate addiction. Br J Clin Pharmacol 2014; 77: 253–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sullivan MA, Bisaga A, Mariani JJ, et al. Naltrexone treatment for opioid dependence: does its effectiveness depend on testing the blockade? Drug Alcohol Depend 2013; 133: 80–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Litten RZ, Ryan ML, Falk DE, Reilly M, Fertig JB, Koob GF. Heterogeneity of alcohol use disorder: understanding mechanisms to advance personalized treatment. Alcohol Clin Exp Res 2015; 39: 579–84. [DOI] [PubMed] [Google Scholar]
- 53.Giedd JN, Blumenthal J, Jeffries NO, et al. Brain development during childhood and adolescence: a longitudinal MRI study. Nat Neurosci 1999; 2: 861–3. [DOI] [PubMed] [Google Scholar]
- 54.DeJong W, Blanchette J. Case closed: research evidence on the positive public health impact of the age 21 minimum legal drinking age in the United States. J Stud Alcohol Drugs Suppl 2014; 75: Suppl 17: 108–15. [DOI] [PubMed] [Google Scholar]
- 55.Report to congressional committees: mental health and substance use — employer’s insurance coverage maintained or enhanced since MHPAEA, but effect of coverage on enrollees varied. Washington, DC: Government Accountability Office, 2011. [Google Scholar]
- 56.Bevilacqua L, Goldman D. Genes and addictions. Clin Pharmacol Ther 2009; 85: 359–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Mertens JR, Weisner C, Ray GT, Fire man B. Walsh K Hazardous drinkers and drug users in HMO primary care: prevalence, medical conditions, and costs. Alochol Clin Exp Res 2005;29:989–98. [DOI] [PubMed] [Google Scholar]
- 58.Weisner C, Mertens J, Parthasarathy S, Moore C, Lu Y. Integrating primary medical care with addiction treatment: a randomized controlled trial. JAMA 2001; 286: 1715–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.