Abstract
Background
Breast radiotherapy is an established adjuvant treatment after breast conserving surgery. One of the important individual factors affecting the final cosmetic outcome after radiation is breast size. The purpose of this review is to summarise the clinical toxicity profile of adjuvant radiotherapy in women with breasts of various sizes, and to evaluate the treatment planning studies comparing target coverage and dose to thoracic organs at risk in relation to breast size.
Conclusions
Inhomogeneity and excessive radiation dose (hot spots) in the planning of target volume as well as large volume of the breast per se, all contribute to a higher rate of acute adverse events and suboptimal final cosmetic outcome in adjuvant breast cancer radiotherapy, regardless of the fractionation schedule. Improved homogeneity leads to a lower rate of ≥ grade 2 toxicity and can be achieved with three-dimensional conformal or modulated radiotherapy techniques. There may be an association between body habitus (higher body mass index, bigger breast size, pendulous breast, and large chest wall separation) and a higher mean dose to the ipsilateral lung and whole heart. A combination of the technical innovations (i.e. the breath-hold technique, prone position with or without holding breath, lateral decubitus position, and thermoplastic bra), dose prescription (i.e. moderate hypofractionation), and irradiated volume (i.e. partial breast irradiation) should be tailored to every single patient in clinical practice to mitigate the risk of radiation adverse effects.
Key words: breast cancer, breast size, radiation side effects, organs at risk, treatment planning
Introduction
With the ageing population and screening programs adopted worldwide, both the incidence and prevalence of breast cancer (BC) are projected to increase over the next decades. Since radiotherapy is one of the key modalities in BC treatment, the absolute number of new BC patients in need of external beam radiotherapy is expected to increase in the immediate future in nearly all European countries.1
Breast conserving surgery in combination with adjuvant radiotherapy has become the standard of care in BC management.2,3 Large retrospective population-based studies nowadays show that breast conserving therapy (BCT) may have an even better outcome in terms of BC-specific and overall survival compared to mastectomy.4 BCT, when compared to more radical surgery, has a positive impact on the patient’s quality of life many years after treatment, especially in terms of body image, sexual activity, and better physical and role functioning.5
Clinicians and researches alike are paying particular attention to reducing acute and late treatment toxicities in a growing number of BC survivors.6 Acute skin toxicity is very common and ranges from mild erythema to moist or dry skin desquamation, with the peak reaction occurring one to two weeks post treatment.7,8 Late skin reactions include skin fibrosis, skin dyspigmentation, and telangiectasia. Acute heart or lung toxicities are rarely seen in BC adjuvant radiation treatment, but late sequelae may be life threatening, with acute coronary event and lung cancer being two possible complications.9
Advances in BC radiotherapy – among them being moderate hypofractionation schedule (HF), intensity modulated radiotherapy (IMRT), and prone or lateral decubitus position – have all the potential to reduce the rates of acute and long-term radiotherapy-related side effects of BCT.6,7,10,11,12,13,14 The observed normal tissue toxicity rates and breast cosmetic outcome depend on treatment and patient-related factors such as the type and number of surgical procedures, systemic treatment, breast size and shape, race, age, comorbidities, smoking, individual sensitivity to ionizing radiation, choice of fractionation and radiation dose, skin bolus, inter-fraction time interval, volume irradiated, and radiotherapy delivery modality.15 It is a widely accepted fact that patient-related factors, such as higher body mass index (BMI) and larger breasts7,10,16 increase the risk of ≥ grade 2 (G2) dermatitis, regardless of the fractionation regimen.11,17
We conducted a review to summarise the clinical toxicity profile of adjuvant radiotherapy in women with breasts of various sizes, and to assess the dosimetric studies of different treatment planning techniques which compared the target coverage (also related to breast size) and dose to thoracic organs at risk with a focus to cardiac subvolumes.
Materials and methods
Literature search and selection criteria
A comprehensive literature search for clinical and dosimetric findings was carried out using PubMed/ Medline from January 1990, with 30 September 2017 being the last search date. Only English literature was considered, using the following key words: “breast cancer” and “radiotherapy”. Subheadings were searched with “organ size”, “3D-conformal radiotherapy (3D-CRT)”, “intensity modulated radiotherapy (IMRT)”, “hybrid-IMRT”, and “volumetric-modulated arc therapy (VMAT)”, “organs at risk”, “treatment planning”, “Heart/radiation effects”, “Coronary Vessels/radiation effects”, and “dosimetric comparison”. Additional relevant references were found in reference lists published with the articles. Clinical studies were selected independently of the number of the patients included. We also searched for treatment planning studies with at least 2 different treatment modalities (i.e. 3D-CRT, IMRT (multi-beam and tangential), hybrid-IMRT, and volumetric modulated arc therapy (VMAT) for left-sided breast cancer).
All selected articles were reviewed in full-text versions and were further divided into clinical or treatment planning articles. In clinical studies, we searched for acute skin toxicity, heart and lung toxicity, secondary malignancy risk, and for possible strategies to modify the toxicity, again taking into account the different breast size categories. Treatment planning studies were reviewed in detail and selected only if the delineated organs at risk included at least one additional heart substructure, namely coronary arteries or cardiac chambers.
Results
The aim of the literature search was to select all clinical and treatment planning studies of adjuvant breast radiotherapy, taking into account the different breast size categories. The search retrieved 6074 articles, 5980 of which were excluded from the review because the content of the article did not match the search criteria, the content was irrelevant to the review topic, or the records were duplicated. Ultimately, 94 articles were found relevant to this study.
Definition of small, medium, and large breast volume
Clinical studies do not define different breast sizes uniformly. Some of the studies differentiate between breast volumes using measures such as clothing and bra size, where a cup size ≥ D categorises woman as having large breasts.7,18 In a study by Pignol et al., breast size was defined as follows: small (USA bra sizes 32A/B, 34A/B, and 36A), medium (USA bra sizes 32C, 34C, 36B/C, and 38A/B/C), or large (all other).7 Some studies use the distance between the edges of the lateral and medial fields, where a breast separation of approximately 18 and 25 cm constitutes medium and larger breast sizes, respectively.19 Modern three-dimensional treatment planning allows for the target volume to be measured, and clinical target volumes (CTV) of ≥ 1.600 cm3, 975–1.600 cm3, and ≤ 500–975 cm3 have been defined as large, medium, and small breasts, respectively.20,21 One study described a standardised and reproducible protocol to measure breast size (the thickness of left and right axillary fat and nipple-to-pectoral muscle distance), finding that anthropometric measurements correlate with the risk of skin toxicity.16
Acute toxicity
Randomised clinical trials and retrospective clinical data from standard tangential two-dimensional radiotherapy with wedges (2D-RT) vs. IMRT show an improvement in planning target volume homogeneity and conformity with IMRT, which may have a clinically significant benefit in reducing the rates of acute dermatitis, moist desquamation, pruritus, palpable breast fibrosis, and acute and chronic oedema in women with all breast sizes.7,10,22,23,24,25 A detailed investigation about the IMRT technique across the studies revealed different planning approaches. IMRT was partly defined as a manual forward-planned technique (F-IMRT)7,10,22,23,25,26,27 and partly as an inverse algorithm7,23, hybrid IMRT (H-IMRT)28, and typically used physical compensators and step-and-shoot multi-leaf collimator (MLC) fields7,10,22,23,25,26 or enhanced dynamic wedges and dynamic MLCs.29 A systematic review and meta-analysis of side effects associated with the use of IMRT in adjuvant BC treatment can be found elsewhere.30
In 2008, Pignol et al. reported a correlation of increased moist desquamation anywhere in the breast with BMI, increasing breast separation, smaller vs. larger breast sizes, and with a higher relative volume of the breast receiving > 105–115 % of the prescribed dose.7 In a multivariate analysis, IMRT was associated with a decreased risk of moist desquamation (odds ratio, OR, 0.418, p = 0.0034) while breast size (per 100 cm3) (OR 1.23, p < 0.0001) was associated with an increased risk.7 Moist desquamation was also correlated with a reduction in the global health status scale (p = 0.0019), ≥ G2 pain score, and with an increase in the breast symptoms scale (p = 0.0028).7 G2–4 acute pain was not statistically different between IMRT or conventional radiotherapy arms at the end of the radiation treatment, nor was the data on chronic pain in 241 patients available after 9.8 years of follow-up (OR = 0.74, range 0.432–1.271).7,31
Six other clinical studies reported a comparison of the clinically adverse events in regard to the three groups of breast sizes. Four compared 2D-RT vs. IMRT7,10,23,25,27 in the supine position, one study compared 3D-CRT vs. IMRT in the prone position28, and one study compared conventional (CF) vs. HF in the prone or supine position (Table 1).32 The percentages of patients experiencing ≥ G2 acute breast toxicity categorised in groups of small, medium, or large-sized are presented separately in Figure 1.
Table 1.
Selected studies evaluating IMRT versus 2D-RT or 3D-CRT. Patients were further stratified by small, medium or large-sized breasts.
| Study | Number of patients | Type of study | Technique | Total dose and Fractionation CF/HF | Breast size (median breast volume)cm3 | Scoring system | G1 or G2 (%) (whole group) | G2 or G3 (%) (whole group) | G3 or G4 (%) (whole group) | General comments |
|---|---|---|---|---|---|---|---|---|---|---|
| Freedman 200627 | 131 | Case-control retrospective study | F-IMRT 2D-RT | 46–50 Gy in 23–25 fractions + boost 10–16 Gy | Breast size was grouped as small (34 A, B; 36 A), medium (34 C; 36 B, C; 38A, B, C), or large (any D or size ≥ 40) | CTCAE v. 3.0 | 30 (IMRT) 28 (2D-RT) | 70 (IMRT) 72 (2D-RT) | 0 (IMRT) 0 (2D-RT) | IMRT is associated with a decrease in severity of acute desquamation compared with a matched control group treated with conventional radiation therapy. |
| Harsolia, 200725 | 172 | Retrospective study | F-IMRT 2D-RT | CF median dose 45 Gy + 16 Gy boost | 1.326 (IMRT) 1.489 (2D-RT) Breast volume divided into groups: 1.000 cm3 (small), 1.000–1.599 cm3 (medium), 1.600 cm3 (large) | NCI CTC v. 2.0 | 41 (IMRT) 85 (2D-RT) | 1 (IMRT) 6 (2D-RT) | Lower rates of ≥ G2 toxicity with IMRT regardless of breast size. ≥ G2 clinical toxicities associated with larger irradiated breast sizes, on average (<1.000 cm3; vs. >1.600 cm3) No G3 acute toxicity with breast volume (<1.000 cm3) and 3% G3 skin reaction in patients with breast volumes 1.600 cm3. |
|
| Freedman 200910 | 804 | Retrospective study | F-IMRT 2D-RT | 46–50 Gy in 23–25 fractions + boost 10–18 Gy | Bra size, (at least 63% with small and medium sizes) Small (32; 34A, B; 36A), Medium (34C; 36B, C;38A, B, C); Large (any D or size 40+) |
CTCAE v. 3.0 | 52 (IMRT) 75 (2D-RT) | More large-breasted patients in IMRT group. IMRT reduces the incidence of ≥ G2 dermatitis in women of all breast sizes. |
||
| Shah 201223 | 335 | Prospective study | IMRT 2D-RT IMRT: HF -inversely planned IMRT CF - forward planned IMRT | Median dose 45 Gy + boost 16 Gy or 42.56 Gy without a boost | 1.378 for the whole group | CTCAE v. 3.0 | 1 (CF-IMRT) 23 (HF-IMRT) 12 (2D-RT) | IMRT is associated with reduced toxicities compared with 2D radiotherapy. | ||
| Breast volume divided into groups: 1.000 cm3 (small), 1.000-1.599 cm3 (medium), 1.600 cm3 (large) | In large-breasted patients, CF-IMRT was associated with reduced acute toxicities, while HF-IMRT was not. | |||||||||
| Hardee 201228 | 97 | Prospective study | H-IMRT 3D-CRT IMRT: hybrid IMRT using a mixture of 3D tangent fields and dynamic multileaf collimator (MLC) IMRT fields in a 2:1 ratio | 46 Gy in 23 fractions + 14-Gy boost or 42.72 Gy in 16 fractions; all in prone position | Breast size was classified as small (A cup, <750 cm3), medium (B-C cups, 750–1.499 cm3), and large (D cup or larger,≥ 1,500 cm3) | RTOG | 5.1% ≥ G2 | Hypofractionated breast radiotherapy is well tolerated when treating patients in the prone position, even among those with large breast volumes. Breast IMRT significantly improves dosimetry but yields only a modest but confirmed benefit in terms of toxicities. | ||
| De Langhe 201432 | 377 | Prospective study | Prone or supine position with INV-IMRT or prone with F-IMRT or prone position with DIBH (n = 22) or supine F-IMRT ± DIBH | 40.05 Gy in 15 fractions + boost 10 Gy in 4 fractions (90–75% of patients) or 50 Gy in 25 fractions for 65% of patients with bra cup size ≥ D | Breast size was classified A, B, C and ≥ D cup | CTCAE v. 3.0 | 57.3 (≥ G2) | CF, supine IMRT, concomitant hormone treatment, high BMI, large breast, smoking during treatment, and genetic variation (in MLH1 rs1800734): all were associated with ≥ G2 toxicity. | ||
2D-RT = standard 2D wedged plan; CTCAE v. 3.0 = common terminology criteria for adverse events for acute radiation dermatitis, version 3.0; CF = conventional fractionation; HF = hypofractionation; DIBH = deep inspiration breath hold; F-IMRT = forward planned intensity modulated radiotherapy; H-IMRT = hybrid intensity modulated radiotherapy; NCI CTC v. 2.0 = National Cancer Institute common toxicity criteria; version 2.0; RTOG = Radiation Therapy Oncology Group criteria
Figure 1.
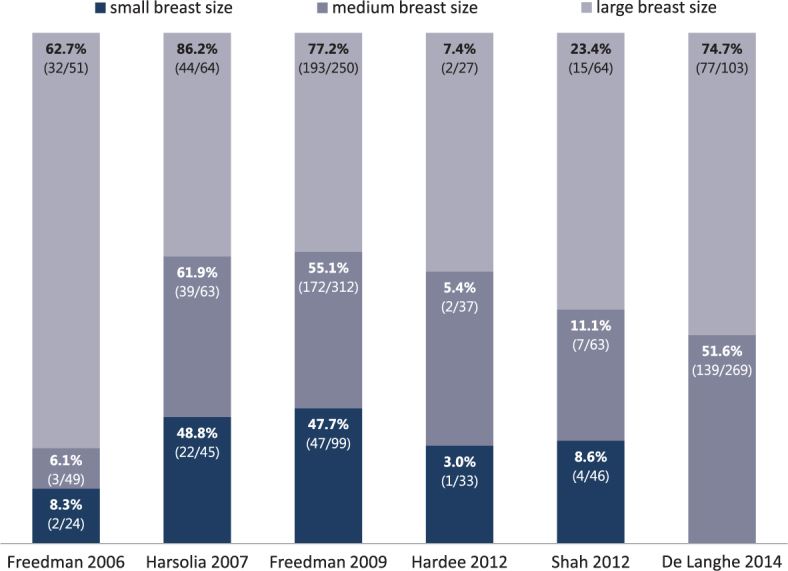
Percentage of patients experiencing ≥ G2 acute breast toxicity, categorised in groups of small, medium, or large-sized breasts as reported in selected studies. The numbers displayed in parentheses are the absolute numbers of patients experiencing toxicity and absolute numbers of patients in a group. De Langhe et al. grouped small and medium-sized breasts in one category.
Harsolia et al. found a correlation between ≥ G2 dermatitis, the development of chronic hyperpigmentation, and breast oedema with a larger than average breast size.25 Interestingly, no ≥ G3 toxicity was reported in smaller breast volumes in either treatment modality (2D-RT vs. IMRT). In comparison with 2D-RT, IMRT improved the rates of ≥ G2 acute oedema (36 % vs. 0 %, p < 0.001), ≥ G2 chronic oedema (30 % vs. 3 %, p = 0.007), and ≥ G2 hyperpigmentation (41 % vs. 3 %, p = 0.001) regardless of the breast volume. No statistically significant difference was observed in acute or chronic rates of ≥ G2 acute dermatitis, pain, chronic hyperpigmentation, or breast induration.25
Increased rates of acute dermatitis, acute and chronic oedema, and chronic hyperpigmentation, irrespective of the treatment technique (prone vs. supine) or fractionation (CF vs. HF), were noted in large-breasted patients (volume > 1.600 cm3) by Shah et al., when compared to patients with a smaller breast size. In large-breasted patients, IMRT was superior to the 2D-RT technique in reducing the rates of ≥ G2 acute dermatitis (0 % vs. 19 %, p = 0.02) and oedema (7 % vs. 24 %, p = 0.06).23
Acute skin toxicity, especially moist desquamation, is associated with late complications of radiotherapy, namely telangiectasia and late subcutaneous fibrosis, as shown in a ten-year update of a Canadian breast IMRT trial and other studies.31,33 Five-year results of simple IMRT (F-IMRT) support the use of BC adjuvant radiotherapy technique that improves homogeneity: the benefit of IMRT was confirmed in a multivariate analysis for both overall cosmesis (p = 0.038) and skin telangiectasia (p = 0.031), although there was no difference in breast shrinkage, breast oedema, tumour bed induration, or pigmentation.24
Hypofractionation
Moderate HF schedule is an established adjuvant treatment in lymph node-negative BC after breast conserving surgery with no differences in disease-related outcomes and with a favourable toxicity profile.34,35,36,37 Moreover, shorter treatment schedules are a cost-effective approach for both the patient and healthcare providers.38 The advantages of a hypofractionated schedule over conventional fractionation, e.g. convenience, a less acute pain, fatigue, and dermatitis, were recently confirmed with prospectively collected physician-assessed data and patient-reported outcome measures in a large comparative analysis by Jagsi et al.37 In this multicentre cohort, the mean breast volume, BMI, and separation distance were slightly smaller in the hypofractionated group: 1270 vs. 980 cm3, 23 vs. 21.9 cm, and 30.8 vs. 28.7, respectively.37
A higher daily fraction size (> 1.8–2.0 Gy) and hot spots (> V105 %) may contribute to so-called ‘triple trouble’ or an unequal distribution of the biological effective dose (BED), although the risk is probably insignificant.39,40 To avoid any of the possible complications (greater fibrosis or late normal tissue effects) with HF, it is generally recommended to limit the volume of hot spots and not to exceed 107 % of the prescribed dose.40 Some authors suggest that patients with a large breast size that precludes achieving the maximum dose of > 107 % should be offered a dose/fractionation that is biologically less intense, for example 45 Gy in 25 daily fractions at 1.8 Gy daily with an addition of a boost dose.41
Similarly as with the CF schedule, high BMI, an increasing PTV volume with a cut off value as small as 500 cm332,35,42, and excessive radiation dose in the target volume (i.e. V107–110 %) contribute to increased acute skin toxicity.17,42 When CF was compared to HF, the CF schedule was a predictive factor for increased ≥ G2 toxicity.17,32,35 One study reported no differences in acute skin toxicity when HF was compared to CF in similar groups of largebreasted women (volume > 1.500 cm3), BMI > 30, or breast separation > 25 cm. Breast volume was the only patient factor significantly associated with moist desquamation in a multivariable analysis (p = 0.01). A very large breast volume (> 2.500 cm3) had a higher rate of focal moist desquamation (40.7 %) compared to breast volume < 2.500 cm3 (11.1 %) (p = 0.002).11 In a randomised clinical trial by Shaitelman et al., where three-quarters of patients were overweight or obese and half of the patients had a Dmax of 107 % or higher, the authors have confirmed the administration of the HF schedule in regard to acute toxic effects to be safe.36
Lung and heart toxicity
The dose-volume predictors for acute and late radiation-induced toxicities are established for the lung and heart as a whole structure.43,44,45 Recent studies have evaluated the dose to the whole heart and the proportional increase in cardiac events after BC radiotherapy. An estimated linear increase of 7.4 % and 4.1 % was found per every Gy mean dose to the whole heart for major coronary events and cardiac mortality, respectively.9,45 A systematic literature review on modern radiation doses to heart and lung in BC radiotherapy showed an estimated absolute 30-year risk for cardiac mortality of 1 % for smokers and 0.3 % for non-smokers.9 Patient-related factors (age and smoking), systemic therapy, the fractionation schedule (total dose and daily fraction), and dose-volume parameters of radiation treatment plan such as mean dose to the whole lung and V20, all constitute risk factors for pneumonitis and lung fibrosis.15,43
In a recent retrospective clinical study of 4688 WBRT-treated BC patients, it was reported that larger breast separation (> 22 cm) was one of the factors significantly increasing the mean heart dose (MHD) for CF by 1.5 % per 1 cm and in HF by 1.7 % per every 1 cm increase, respectively.46 It has been demonstrated that the dose to the heart can be significantly reduced in both CF and HF by means of breathing adaptation and prone positioning.13,46
Hannan et al. found that increasing breast size results in increased mean and maximum point heart doses.13 PTV, BMI, or age were generally unrelated to ipsilateral lung dose. The lung dose decreased markedly in the prone compared to supine position for the whole group of patients. Largebreasted prone-treated patients received a higher ipsilateral lung D5 (greatest dose delivered to 5 cm3) compared to small-breasted prone-treated patients (PTV < 1000 cm3), but without significant differences in V5 or V20.13 Breast shape (i.e. pendulous breasts) can contribute to a higher maximum heart dose as well.47 One small dosimetric study in free-breathing supine-position radiation therapy did not find a correlation of increasing breast separation with higher heart doses, but instead found a correlation with an increased dose to the ipsilateral lung (parameters V5 Gy, V10 Gy and V20 Gy), with the greatest increase noted in breast separation between 25 and 27 cm.48 By contrast, Hardee et al. found an opposite association between breast size and lung dosimetric parameters in the prone position (in-field lung volume, V5, and Dmax). All parameters decreased as breast size increased.28
Secondary malignancy risk
In BC radiotherapy, differences in body habitus may influence doses to organs at risk, but it is not known if small differences in radiation exposure at the time of the first radiation course significantly influence the risk of a secondary non-breast cancer. Compared to the general population, BC patients have an increased risk of secondary non-breast cancers, five or more years after BC diagnosis with and without radiation therapy (RR 1.12; 95 % confidence interval [CI] 1.06–1.19).49 But probably less than 3.5 % of secondary malignancies in BC survivors are attributable to radiation therapy.50 The total dose of radiation, premenopausal age (< 40–45 years) and the irradiated volume of normal tissue all increase the risk for secondary lung, oesophageal, or thyroid cancer, and secondary sarcomas.49,51 The risk of lung cancer increases with the mean dose to the whole lung.52 A systematic review of modern radiation doses to the lung in BC radiotherapy showed an estimated absolute 30-year risk for lung cancer of 4 % for long-term continuing smokers and 0.3 % for non-smokers.9
The radiation dose to the lung increases with lymph node irradiation and the use of IMRT techniques, and decreases with breathing adaptations and prone/lateral decubitus positioning.52 Younger women (< 40 years of age) with an absorbed radiation dose > 1.0 Gy to the contralateral breast have an elevated long-term risk of developing a second primary contralateral BC.52
A study by Bhatnagar et al. suggests that the size of the primary irradiated breast significantly affects the scatter dose to the contralateral breast but not the ipsilateral lung or heart when using IMRT for breast irradiation. The mean volume of the primary irradiated breast in the study was 1167.9 cm3.53 However, Jin et al. found that in the population of women with smaller breasts (360.8 ± 149.1 cm3), the size of the treated breast does not significantly increase the dose to the contra-lateral breast with 2D-RT, 3D-CRT with 3–5 subfields, or tangential IMRT techniques.54
Strategies to modify acute toxicity and dose to organs at risk
Different strategies in BC radiotherapy exist to lower the dose to organs at risk. Approaches could be further divided according to patient or breast positioning modification, breathing adaptation, and treatment volume reduction.
Patient positioning modification
The prone positioning setup demonstrated to be an excellent strategy to spare the ipsilateral lung in 100 % and heart dose in 85–87 % of the patients, independently of their BMI or breast size12,55,56,57,58, but a particular benefit was observed in large-breasted women (CTV > 1000 cm3).12,13,57 Similar findings were confirmed in a study by Formenti et al. but, in this study, the prone treatment position did not necessarily spare the heart in patients with breast volumes smaller than 750 cm3.57 On the other hand, one study reported having achieved similar heart doses for prone and supine 3D-CRT WBRT in women with large breast volumes (the average treated volume was 1804 cm3 in right-sided breasts and 1500 cm3 in left-sided breasts). They also noted a significantly higher incidental dose to the LADCA in the prone position with left-sided BC.59
3D-CRT lateral isocentric decubitus position was recently described as a treatment planning solution. Long-term toxicity results were published by the Institut Curie group.14,60 Women with a median BMI of 26.3 were treated with different types of fractionation. Acute dermatitis of any grade was present in 93 % of the patients and G3 dermatitis in only 2.8 %. In a 1-year follow-up, 94.1 % of cases had no skin reaction, making this technique feasible with excellent toxicity rates, but the results need to be confirmed with a longer follow-up. In a multivariate analysis, the cup size and the fractionation had a significant influence on acute dermatitis.14
Breathing adaptation
The deep inspiration breath-hold (DIBH) technique helps to minimise the “trade-off” between the target and OAR, a compromise often required, and is less resource-intensive than the IMRT technique. It reduces the low-dose irradiation to the heart, left anterior descending coronary artery (LADCA), and lung, ultimately benefiting women of all breast sizes.61 DIBH can be accurately clinically implemented with an acceptable reproducibility and stability in both supine and prone position.61,62 In a group of women with a volume of the treated breast > 750 cm3, supine voluntary DIBH enabled a cardiac sparing and reproducibility superior to that of free-breathing prone position.63
Partial breast irradiation
Another strategy to lower the absorbed radiation dose to the heart is partial breast irradiation. Patients may benefit in terms of lower mean whole heart doses with moderate HF using 3D-CRT and the accelerated partial breast irradiation technique (APBI) with an external beam or interstitial brahcytherapy.64,65,66,67 Meszaros et al. demonstrated the reproducibility of image-guidance intensity modulated APBI and feasibility in terms of acute toxicity and the cosmetic outcome with a median follow up of 3.2 years. In a study, 55 % of the patients had cup size C and 21 % cup size ≥ D.67 Investigators emphasised the necessity of image guidance prior to each radiation fraction to reduce the CTV to PTV margins.67
Breast tissue modification
A thermoplastic bra which helps to raise the lateral breast border is also an option to lower the dose to thoracic organs in BC radiotherapy. Piroth et al. demonstrated an excellent reduction in radiation exposure to the heart (mean dose reduction by ≈ 23 %) and ipsilateral lung (mean dose reduction by ≈ 30 % and V20 by 39.5 %) without additional skin toxicity in women of all breast sizes (clinical target volume ranging from 283.1 to 1581.6 cm3).68
Treatment planning studies
Many different treatment planning options are available in the modern treatment era: 3D-CRT with or without wedged filters, forward-planned IMRT (F-IMRT), inverse-planned IMRT (INV-IMRT), Helical Tomotherapy (HT), VMAT, and hybrid techniques (H-IMRT). The recommended first choice for WBRT varies across numerous treatment planning studies comparing different modalities (i.e. 3D-CRT vs. IMRT vs. VMAT) and usually depends on the available equipment, technical innovations, irradiated volume, treatment planning system with dose calculation algorithm, and skills of the planner.
There are numerous publications comparing the dosimetric parameters of radiotherapy plans, mostly for patients with left-sided early BC. Some of them have been summarised in a review of treatment planning options by Balaji et al. where most authors favoured hybrid planning techniques (3D CRT + IMRT/VMAT) while weighing the target coverage vs. dose to the organs at risk.69 While INV-IMRT is not routinely recommended after breast-only radiation, the use of advanced techniques is increasing in challenging anatomy cases (up to 9.4 % of all BC patients treated with radiotherapy).70 The majority of techniques can be combined with DIBH and/or the prone position. The role of the VMAT technique in clinical practice is still not known precisely and the technique itself is not routinely recommended. The dosimetric data, although promising, need to be validated from a clinical point of view.71 The VMAT technique is sometimes the first choice in complex anatomy cases (including large breasts, bilateral BC adjuvant radiotherapy, pectus excavatum, etc.).71 HT (TomoDirect) can be delivered in a 3D-CRT or IMRT modality in whole-breast adjuvant BC radiotherapy. Both HT modalities have a good PTV coverage and dose homogeneity, but some caution is needed as the dose to ipsilateral lung and heart can be significantly high with the 3D-CRT modality in specific patient anatomic situations. Authors have proposed that simple anatomic measures like maximal heart distance can be helpful in selecting the appropriate treatment strategy.72
For the purpose of this review, we have evaluated the treatment planning studies which included at least coronary arteries or cardiac chambers as organs at risk. Overall, we have found eight treatment planning studies comparing radiotherapy plans in free-breathing CTs.54,73,74,75,76,77,78,79 The studies indicate an improved dose homogeneity with the IMRT54,73,75,78,79 or VMAT73,74 techniques regardless of the PTV volume, but the number of CT study sets compared was relatively low (10–20). The sizes of the target volumes reported by investigators comparing treatment planning approaches were dissimilar, ranging from 296 cm3 (mean value) to 1160 cm3 (median value).74,79 A study by Tan et al. found additional heart subvolumes (left ventricle or LV and anterior myocardial territory or AMT) to be helpful in the IMRT plan optimization process, although there have been no reports available so far for the dose–volume constraints in these two OARs.75
Besides the heart as a whole structure, authors typically delineated LADCA73,74,76,77,78, LV75,76,78,79, right ventricle (RV)76,78, left atrium (LA)76,78, right atrium (RA)76,78, great vessels78, and AMT.54,74,75 The delineation of heart substructures was not uniformly defined or was rarely guided by written instructions, making it difficult to compare the presented studies.54,74,75
Discussion
Growing clinical data on BC adjuvant radiotherapy suggest that a smaller PTV and/or the use of the IMRT technique may be associated with a decreased rates of acute breast toxicity. Most studies evaluated women with small or medium-sized breasts, so maybe all of the results are not directly applicable in large-breasted woman. The clinical studies which reported a comparison of the clinical adverse events in regard to the three categories of breast sizes mostly used the CTCAE v.3.0 scoring system.7,10,23,25,27,28 The rates of ≥ G2 toxicity for the whole group of patients from selected studies used in Figure 1 ranged from 5.1 % to 70 %. The percentage of patients with ≥ G2 toxicity was the highest in the subcategory of large-breasted patients in all the studies, ranging from 7.4 % to 86.2 %.10,23,27,32 The differences in adverse event reporting could at least partly be attributed to different scoring systems, i.e. the subjective scoring by investigators and different planning techniques.
Moderately hypofractionated schedules proved superiority over CF and conformal radiation therapy (3D-CRT or F-IMRT) over 2D-RT in terms of acute and late adverse effects in early BC WBRT.7,12,22,24,26,34,37 Clinical reports are confirming the long-term safety and feasibility of moderately hypofractionated schedules also in women with large breasts.11,17,32,36,38,42,80,81 Most of the investigators attributed higher toxicity rates in BC radiotherapy to dose inhomogeneity and a higher percentage of hot spots, irrespective of breast volume.7,12,17,31 V105–107 % of the prescribed dose (PD) was significantly related to increased desquamation, dermatitis, oedema, and pain12; and V105 % PD82,83 or V110 % PD83 to long-term breast pain. Significant reductions in hot spots can be achieved with 3D-CRT or F-IMRT treatment plans, also in patients with large/pendulous breasts (PTV >1000 cm3).20,26,28,84 An improvement in dose homogeneity was achieved with IMRT, and correlated with less acute toxicity rates in a study by Mulliez et al.85
However, some of the studies show that large breast volume seems to be a risk factor for acute or late adverse events independently of dose inhomogeneity and regardless of the conformal radiotherapy technique (3D-CRT vs. IMRT) or fractionation schedule.11,24,86 A retrospective analysis of selected patients from UK FAST hypofractionation (3D-CRT) trial found that breast size and dosimetric parameters are significantly associated with late effects in a univariate analysis where breast size was the only remaining independent significant risk factor for change in breast appearance when included in a multiple regression model together with other prognostic factors.86 Investigators failed to correlate the breast composition (breast tissue distribution and scar tissue presence) with late adverse effects, with the exception of seroma.87 Combining all data, there is no reason to withhold the hypofractionation schedule in large-breasted women.
The data published support the hypothesis that every Gy of increase in the whole lung mean dose increases the risk for second lung cancer.9 In the long run, as far as the sub-population of continuing smokers is concerned, the second lung cancer risk is even greater and the benefits of adjuvant radiotherapy in early BC may be reduced to the point where long-term risks outweigh the benefits of adjuvant radiotherapy.9 At the same time smokers, portend lifelong cardiac mortality risks and smoking during BC radiotherapy significantly increase the risk of acute ≥ G2 dermatitis.32,52 Smoking cessation counselling may be provided to modify acute and late toxicity risks. Breathing-adapted radiotherapy in the prone or supine position in women with all breast sizes, and prone or lateral setup in medium or large-breasted patients (approximately ≥ 1000 cm3) have been shown to decrease the whole lung and heart dose parameters.12,13,52,57,88
In terms of the heart as an organ at risk, ideally, all treatment planning comparisons of WBRT in patients with left-sided BC should be done in DIBH. Treatment planning or retrospective dose evaluation studies often only include the heart as a whole structure, without separately delineated heart subvolumes, although the dose distribution in the heart itself in BC adjuvant radiotherapy is usually not homogeneous.89 In some patients, the dose to the LADCA can be significantly higher in the prone (without breath-hold) compared to the supine setup, which could also be attributed to the differences in contouring and treatment techniques.59 The routine use of delineation guidelines for thoracic organs at risk and dose reporting with clinical correlation could help us further understand normal tissue complication probability models, especially in the least known dose-response relationships, i.e. for coronary arteries and cardiac chambers. Using individual 3D-CRT planning data, one study independently validated the mean heart dose-based normal tissue complication probability (NTCP) model (published by Darby et al. in 201345) for acute coronary events within 9 years after adjuvant radiotherapy. Investigators found an increase of 16.5 % in the cumulative incidence of acute coronary events per every Gy increase of the mean heart dose. One step further was made in understanding the radiation tolerance for particular cardiac segments, as there are no current models for dose-response relationships. The study found a significant prognostic importance of the left ventricle V5 Gy dose relationship with an acute coronary event.44,90
Treatment planning studies usually compare a limited number of CT study sets and it is not known if small improvement in dosimetric metrics would translate into clinically meaningful lower rates of adverse events for the larger population. For example, one treatment planning study in the modern treatment era reported very little difference in dosimetric parameters between patients of different breast size regardless of the modality (static HT, IMRT, and 3D-CRT).21 Expected absolute differences in the rates of clinical adverse events (3D-CRT vs. other highly conformal techniques) are likely negligible. HT, INV-IMRT, and VMAT may all increase the mean and maximum dose to contralateral breast, and mean dose to the heart and ipsilateral lung compared to F-IMRT or 3D CRT, although doses to organs at risk may also depend on the patient’s anatomy or positioning.52,73,75,91
In order to de-escalate radiotherapy, selected patients will be treated routinely with the moderate HF or accelerated partial breast irradiation techniques (treating only the tumour bed with a safety margin) in the near future, as emerging data confirm a similar 5-year cumulative incidence of loco-regional and distant relapse compared to WBRT.38,92,93 In a 5-year assessment, patients treated with partial radiotherapy approaches self-reported less moderate or marked skin (p = 0.051) or overall breast appearance change (p < 0.0001) compared to the WBRT group.88 APBI using interstitial brachytherapy was able to significantly reduce acute G3 skin toxicity (7 % in WBRT group vs. 0.2 %; p < 0.0001).94 Simple F-IMRT techniques using shortened tangential fields and interstitial brachytherapy APBI were able to minimise the dose to the heart and lung.66,92 A higher APBI-PTV/breast volume ratio most probably contributes to adverse acute and poor final cosmetic results.67 Further clinical trials with longer follow-ups are needed in partial breast radiotherapy, especially in large-breasted woman, to confirm the clinical relevance.
Conclusions
One of the important individual factors affecting the final cosmetic outcome of radiation therapy treatment is the size of the treated PTV. It seems that beside the target volume, inhomogeneity, and a higher percentage of the excessive radiation dose, bigger breast size itself is an independent risk factor for acute adverse effects regardless of the fractionation regimen or dose inhomogeneity, although the lower the excessive radiation dose, the lower the risk of ≥ G2 toxicity. While weighing the risk of BC relapse vs. acute or late treatment toxicity, an appropriate postoperative radiotherapy technique (3D-CRT vs. modulated approaches), patient setup (prone vs. supine with or without a breath hold), and volume irradiated (i.e. candidates for partial breast irradiation) should be optimally selected and tailored to the patient’s anatomy (including BMI, breast separation, or cup size), age, and tumour characteristics. A personalised approach is therefore needed in every single patient, with the patient’s social, economic, or psychological issues to be taken into account.
Disclosure
No potential conflicts of interest were disclosed.
References
- 1.Borras JM, Lievens Y, Barton M, Corral J, Ferlay J, Bray F. et al. How many new cancer patients in Europe will require radiotherapy by 2025? An ESTRO-HERO analysis. Radiother Oncol. 2016;119:5–11. doi: 10.1016/j.radonc.2016.02.016. [DOI] [PubMed] [Google Scholar]
- 2.Fisher B, Anderson S, Bryant J, Margolese RG, Deutsch M, Fisher ER. et al. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med. 2002;347:1233–41. doi: 10.1056/NEJMoa022152. [DOI] [PubMed] [Google Scholar]
- 3.Veronesi U, Cascinelli N, Mariani L, Greco M, Saccozzi R, Luini A. et al. Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med. 2002;347:1227–32. doi: 10.1056/NEJMoa020989. [DOI] [PubMed] [Google Scholar]
- 4.Gentilini OD, Cardoso M-J, Poortmans P.. Less is more. Breast conservation might be even better than mastectomy in early breast cancer patients. Breast. 2017;35:32–3. doi: 10.1016/j.breast.2017.06.004. [DOI] [PubMed] [Google Scholar]
- 5.Arndt V, Stegmaier C, Ziegler H, Brenner H. Quality of life over 5 years in women with breast cancer after breast-conserving therapy versus mastectomy: a population-based study. J Cancer Res Clin Oncol. 2008;134:1311–8. doi: 10.1007/s00432-008-0418-y. [DOI] [PubMed] [Google Scholar]
- 6.Berry S. Advances in breast bancer radiotherapy and the impact on quality of life. J Cancer Biol Res. 2014;2:1041. [Google Scholar]
- 7.Pignol J-P, Olivotto I, Rakovitch E, Gardner S, Sixel K, Beckham W. et al. A multicenter randomized trial of breast intensity-modulated radiation therapy to reduce acute radiation dermatitis. J Clin Oncol. 2008;26:2085–92. doi: 10.1200/JCO.2007.15.2488. [DOI] [PubMed] [Google Scholar]
- 8.Pignol J-P, Vu TTT, Mitera G, Bosnic S, Verkooijen HM, Truong P. Prospective evaluation of severe skin toxicity and pain during postmastectomy radiation therapy. Int J Radiat Oncol. 2015;91:157–64. doi: 10.1016/j.ijrobp.2014.09.022. [DOI] [PubMed] [Google Scholar]
- 9.Taylor C, Correa C, Duane FK, Aznar MC, Anderson SJ, Bergh J. et al. Estimating the risks of breast cancer radiotherapy: evidence from modern radiation doses to the lungs and heart and from previous randomized trials. J Clin Oncol. 2017;35:1641–9. doi: 10.1200/JCO.2016.72.0722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Freedman GM, Li T, Nicolaou N, Chen Y, Ma CC-M, Anderson PR. Breast intensity-modulated radiation therapy reduces time spent with acute dermatitis for women of all breast sizes during radiation. Int J Radiat Oncol. 2009;74:689–94. doi: 10.1016/j.ijrobp.2008.08.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Corbin KS, Dorn PL, Jain SK, Al-Hallaq HA, Hasan Y, Chmura SJ. Hypofractionated radiotherapy does not increase acute toxicity in largebreasted women: results from a prospectively collected series. Am J Clin Oncol. 2014;37:322–6. doi: 10.1097/COC.0b013e31827b45b7. [DOI] [PubMed] [Google Scholar]
- 12.Mulliez T, Veldeman L, van Greveling A, Speleers B, Sadeghi S, Berwouts D. et al. Hypofractionated whole breast irradiation for patients with large breasts: a randomized trial comparing prone and supine positions. Radiother Oncol. 2013;108:203–8. doi: 10.1016/j.radonc.2013.08.040. [DOI] [PubMed] [Google Scholar]
- 13.Hannan R, Thompson RF, Chen Y, Bernstein K, Kabarriti R, Skinner W. et al. Hypofractionated Whole-Breast Radiation Therapy: Does Breast Size Matter? Int J Radiat Oncol. 2012;84:894–901. doi: 10.1016/j.ijrobp.2012.01.093. [DOI] [PubMed] [Google Scholar]
- 14.Bronsart E, Dureau S, Xu HP, Bazire L, Chilles A, Costa E. et al. Whole breast radiotherapy in the lateral isocentric lateral decubitus position: Longterm efficacy and toxicity results. Radiother Oncol. 2017;124:214–9. doi: 10.1016/j.radonc.2017.07.001. [DOI] [PubMed] [Google Scholar]
- 15.Meattini I, Guenzi M, Fozza A, Vidali C, Rovea P, Meacci F. et al. Overview on cardiac, pulmonary and cutaneous toxicity in patients treated with adjuvant radiotherapy for breast cancer. Breast Cancer. 2016;24:52–62. doi: 10.1007/s12282-016-0694-3. [DOI] [PubMed] [Google Scholar]
- 16.Méry B, Vallard A, Trone J-C, Pacaut C, Guy J-B, Espenel S. et al. Correlation between anthropometric parameters and acute skin toxicity in breast cancer radiotherapy patients: a pilot assessment study. Br J Radiol. 2015;88:20150414. doi: 10.1259/bjr.20150414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tortorelli G, Di Murro L, Barbarino R, Cicchetti S, di Cristino D, Falco MD. et al. Standard or hypofractionated radiotherapy in the postoperative treatment of breast cancer: a retrospective analysis of acute skin toxicity and dose inhomogeneities. BMC Cancer. 2013;13:230. doi: 10.1186/1471-2407-13-230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dundas KL, Atyeo J, Cox J. What is a large breast? Measuring and categorizing breast size for tangential breast radiation therapy. Australas Radiol. 2007;51:589–93. doi: 10.1111/j.1440-1673.2007.01898.x. [DOI] [PubMed] [Google Scholar]
- 19.Back M, Guerrieri M, Wratten C, Steigler A. Impact of radiation therapy on acute toxicity in breast conservation therapy for early breast cancer. Clin Oncol (R Coll Radiol) 2004;16:12–6. doi: 10.1016/j.clon.2003.08.005. [DOI] [PubMed] [Google Scholar]
- 20.Vicini FA, Sharpe M, Kestin L, Martinez A, Mitchell CK, Wallace MF. et al. Optimizing breast cancer treatment efficacy with intensity-modulated radiotherapy. Int J Radiat Oncol Biol Phys. 2002;54:1336–44. doi: 10.1016/S0360-3016(02)03746-X. [DOI] [PubMed] [Google Scholar]
- 21.Michalski A, Atyeo J, Cox J, Rinks M, Morgia M, Lamoury G. A dosimetric comparison of 3D-CRT, IMRT, and static tomotherapy with an SIB for large and small breast volumes. Med Dosim. 2014;39:163–8. doi: 10.1016/j.meddos.2013.12.003. [DOI] [PubMed] [Google Scholar]
- 22.Donovan E, Bleakley N, Denholm E, Evans P, Gothard L, Hanson J. et al. Randomised trial of standard 2D radiotherapy (RT) versus intensity modulated radiotherapy (IMRT) in patients prescribed breast radiotherapy. Radiother Oncol. 2007;82:254–64. doi: 10.1016/j.radonc.2006.12.008. [DOI] [PubMed] [Google Scholar]
- 23.Shah C, Wobb J, Grills I, Wallace M, Mitchell C, Vicini FA. Use of intensity modulated radiation therapy to reduce acute and chronic toxicities of breast cancer patients treated with traditional and accelerated whole breast irradiation. Pract Radiat Oncol. 2012;2:e45–51. doi: 10.1016/j.prro.2012.01.008. [DOI] [PubMed] [Google Scholar]
- 24.Mukesh MB, Barnett GC, Wilkinson JS, Moody AM, Wilson C, Dorling L. et al. Randomized controlled trial of intensity-modulated radiotherapy for early breast cancer: 5-year results confirm superior overall cosmesis. J Clin Oncol. 2013;31:4488–95. doi: 10.1200/JCO.2013.49.7842. [DOI] [PubMed] [Google Scholar]
- 25.Harsolia A, Kestin L, Grills I, Wallace M, Jolly S, Jones C. et al. Intensity-modulated radiotherapy results in significant decrease in clinical toxicities compared with conventional wedge-based breast radiotherapy. Int J Radiat Oncol Biol Phys. 2007;68:1375–80. doi: 10.1016/j.ijrobp.2007.02.044. [DOI] [PubMed] [Google Scholar]
- 26.Barnett GC, Wilkinson J, Moody AM, Wilson CB, Sharma R, Klager S. et al. A randomised controlled trial of forward-planned radiotherapy (IMRT) for early breast cancer: baseline characteristics and dosimetry results. Radiother Oncol. 2009;92:34–41. doi: 10.1016/j.radonc.2009.03.003. [DOI] [PubMed] [Google Scholar]
- 27.Freedman GM, Anderson PR, Li J, Eisenberg DF, Hanlon AL, Wang L. et al. Intensity Modulated Radiation Therapy (IMRT) Decreases Acute Skin Toxicity for Women Receiving Radiation for Breast Cancer. Am J Clin Oncol. 2006;29:66–70. doi: 10.1097/01.coc.0000197661.09628.03. [DOI] [PubMed] [Google Scholar]
- 28.Hardee ME, Raza S, Becker SJ, Jozsef G, Lymberis SC, Hochman T. et al. Prone hypofractionated whole-breast radiotherapy without a boost to the tumor bed: comparable toxicity of IMRT versus a 3D conformal technique. Int J Radiat Oncol. 2012;82:e415–23. doi: 10.1016/j.ijrobp.2011.06.1950. [DOI] [PubMed] [Google Scholar]
- 29.McDonald MW, Godette KD, Butker EK, Davis LW, Johnstone PAS. Long-term outcomes of IMRT for breast cancer: a single-institution cohort analysis. Int J Radiat Oncol Biol Phys. 2008;72:1031–40. doi: 10.1016/j.ijrobp.2008.02.053. [DOI] [PubMed] [Google Scholar]
- 30.Jensen KE, Soril LJJ, Stelfox HT, Clement FM, Lin Y, Marshall DA. Side effects associated with the use of intensity-modulated radiation therapy in breast cancer patients undergoing adjuvant radiation therapy: a systematic review and meta-analysis. J Med Imaging Radiat Sci. 2017;48:402–13. doi: 10.1016/j.jmir.2017.09.002. [DOI] [PubMed] [Google Scholar]
- 31.Pignol J-P, Truong P, Rakovitch E, Sattler MG, Whelan TJ, Olivotto IA. Ten years results of the Canadian breast intensity modulated radiation therapy (IMRT) randomized controlled trial. Radiother Oncol. 2016;121:414–9. doi: 10.1016/j.radonc.2016.08.021. [DOI] [PubMed] [Google Scholar]
- 32.De Langhe S, Mulliez T, Veldeman L, Remouchamps V, van Greveling A, Gilsoul M. et al. Factors modifying the risk for developing acute skin toxicity after whole-breast intensity modulated radiotherapy. BMC Cancer. 2014;14:711. doi: 10.1186/1471-2407-14-711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lilla C, Ambrosone CB, Kropp S, Helmbold I, Schmezer P, von Fournier D. et al. Predictive factors for late normal tissue complications following radiotherapy for breast cancer. Breast Cancer Res Treat. 2007;106:143–50. doi: 10.1007/s10549-006-9480-9. [DOI] [PubMed] [Google Scholar]
- 34.Haviland JS, Owen JR, Dewar JA, Agrawal RK, Barrett J, Barrett-Lee PJ. et al. The UK Standardisation of breast radiotherapy (START) trials of radiotherapy hypofractionation for treatment of early breast cancer: 10-year follow-up results of two randomised controlled trials. Lancet Oncol. 2013;14:1086–94. doi: 10.1016/S1470-2045(13)70386-3. [DOI] [PubMed] [Google Scholar]
- 35.De Felice F, Ranalli T, Musio D, Lisi R, Rea F, Caiazzo R. et al. Relation between hypofractionated radiotherapy, toxicity and outcome in early breast cancer. Breast J. 2017;23:563–8. doi: 10.1111/tbj.12792. [DOI] [PubMed] [Google Scholar]
- 36.Shaitelman SF, Schlembach PJ, Arzu I, Ballo M, Bloom ES, Buchholz D. et al. Acute and short-term toxic effects of conventionally fractionated vs hypofractionated whole-breast irradiation: a randomized clinical trial. JAMA Oncol. 2015;1:931–41. doi: 10.1001/jamaoncol.2015.2666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jagsi R, Griffith KA, Boike TP, Walker E, Nurushev T, Grills IS. et al. Differences in the acute toxic effects of breast radiotherapy by fractionation schedule: comparative analysis of physician-assessed and patient-reported outcomes in a large multicenter cohort. JAMA Oncol. 2015;1:918–30. doi: 10.1001/jamaoncol.2015.2590. [DOI] [PubMed] [Google Scholar]
- 38.Franco P, Iorio GC, Bartoncini S, Airoldi M, De Sanctis C, Castellano I. et al. De-escalation of breast radiotherapy after conserving surgery in low-risk early breast cancer patients. Med Oncol. 2018;35:62. doi: 10.1007/s12032-018-1121-8. [DOI] [PubMed] [Google Scholar]
- 39.Jones B, Dale RG, Deehan C, Hopkins KI, Morgan DA. The role of biologically effective dose (BED) in clinical oncology. Clin Oncol (R Coll Radiol) 2001;13:71–81. doi: 10.1053/clon.2001.9221. [DOI] [PubMed] [Google Scholar]
- 40.Yarnold J, Somaiah N, Bliss JM. Hypofractionated radiotherapy in early breast cancer: Clinical, dosimetric and radio-genomic issues. Breast. 2015;24(2):S108–13. doi: 10.1016/j.breast.2015.07.025. [DOI] [PubMed] [Google Scholar]
- 41.Koulis TA, Phan T, Olivotto IA. Hypofractionated whole breast radiotherapy: current perspectives. Breast Cancer (Dove Med Press) 2015;7:363–70. doi: 10.2147/BCTT.S81710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lazzari G, Terlizzi A, Vittoria Scarpati G, Perri F, De Chiara V, Turi B. et al. Predictive parameters in hypofractionated whole-breast 3D conformal radiotherapy according to the Ontario Canadian trial. Onco Targets Ther. 2017;10:1835–42. doi: 10.2147/OTT.S127833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Marks LB, Bentzen SM, Deasy JO, Kong F-MS, Bradley JD, Vogelius IS. et al. Radiation dose-volume effects in the lung. Int J Radiat Oncol Biol Phys. 2010;76:S70–6. doi: 10.1016/j.ijrobp.2009.06.091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gagliardi G, Constine LS, Moiseenko V, Correa C, Pierce LJ, Allen AM. et al. Radiation dose-volume effects in the heart. Int J Radiat Oncol Biol Phys. 2010;76:S77–85. doi: 10.1016/j.ijrobp.2009.04.093. [DOI] [PubMed] [Google Scholar]
- 45.Darby SC, Ewertz M, Hall P. Ischemic heart disease after breast cancer radiotherapy. N Engl J Med. 2013;368:25–27. doi: 10.1056/NEJMc1304601. [DOI] [PubMed] [Google Scholar]
- 46.Pierce LJ, Feng M, Griffith KA, Jagsi R, Boike T, Dryden D. et al. Recent time trends and predictors of heart dose from breast radiation therapy in a large quality consortium of radiation oncology practices. Int J Radiat Oncol. 2017;99:1154–61. doi: 10.1016/j.ijrobp.2017.07.022. [DOI] [PubMed] [Google Scholar]
- 47.Guan H, Dong Y-L, Ding L-J, Zhang Z-C, Huang W, Liu C-X. et al. Morphological factors and cardiac doses in whole breast radiation for left-sided breast cancer. Asian Pac J Cancer Prev. 2015;16:2889–94. doi: 10.7314/apjcp.2015.16.7.2889. [DOI] [PubMed] [Google Scholar]
- 48.Wernicke Ag, Heineman T, Sabbas A, Delamerced M, Chiu Y, Smith M. et al. Impact of a large breast separation on radiation dose delivery to the ipsilateral lung as result of respiratory motion quantified using free breathing and 4D CT-based planning in patients with locally advanced breast cancers: a potential for adverse clinical implications. J Cancer Res Ther. 2013;9:154. doi: 10.4103/0973-1482.110368. [DOI] [PubMed] [Google Scholar]
- 49.Grantzau T, Overgaard J. Risk of second non-breast cancer after radiotherapy for breast cancer: a systematic review and meta-analysis of 762,468 patients. Radiother Oncol. 2015;114:56–65. doi: 10.1016/j.radonc.2014.10.004. [DOI] [PubMed] [Google Scholar]
- 50.Burt LM, Ying J, Poppe MM, Suneja G, Gaffney DK. Risk of secondary malignancies after radiation therapy for breast cancer: comprehensive results. Breast. 2017;35:122–9. doi: 10.1016/j.breast.2017.07.004. [DOI] [PubMed] [Google Scholar]
- 51.Stovall M, Smith SA, Langholz BM, Boice JD, Shore RE, Andersson M. et al. Dose to the contralateral breast from radiotherapy and risk of second primary breast cancer in the WECARE study. Int J Radiat Oncol. 2008;72:1021–30. doi: 10.1016/j.ijrobp.2008.02.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Aznar MC, Duane FK, Darby SC, Wang Z, Taylor CW. Exposure of the lungs in breast cancer radiotherapy: a systematic review of lung doses published 2010–2015. Radiother Oncol. 2018;126:148–54. doi: 10.1016/j.radonc.2017.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bhatnagar AK, Heron DE, Deutsch M, Brandner E, Wu A, Kalnicki S. Does breast size affect the scatter dose to the ipsilateral lung, heart, or contralateral breast in primary breast irradiation using intensity-modulated radiation therapy (IMRT)? Am J Clin Oncol. 2006;29:80–4. doi: 10.1097/01.coc.0000198743.80991.15. [DOI] [PubMed] [Google Scholar]
- 54.Jin G-H, Chen L-X, Deng X-W, Liu X-W, Huang Y, Huang X-B.. A comparative dosimetric study for treating left-sided breast cancer for small breast size using five different radiotherapy techniques: conventional tangential field, filed-in-filed, tangential-IMRT, multi-beam IMRT and VMAT. Radiat Oncol. 2013;8:89. doi: 10.1186/1748-717X-8-89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Osa E-OO, DeWyngaert K, Roses D, Speyer J, Guth A, Axelrod D. et al. Prone Breast Intensity Modulated Radiation Therapy: 5-Year Results. Int J Radiat Oncol. 2014;89:899–906. doi: 10.1016/j.ijrobp.2014.03.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ramella S, Trodella L, Ippolito E, Fiore M, Cellini F, Stimato G. et al. Whole-breast irradiation: a subgroup analysis of criteria to stratify for prone position treatment. Med Dosim. 2012;37:186–91. doi: 10.1016/j.med-dos.2011.06.010. [DOI] [PubMed] [Google Scholar]
- 57.Lymberis SC, deWyngaert JK, Parhar P, Chhabra AM, Fenton-Kerimian M, Chang J. et al. Prospective assessment of optimal individual position (prone versus supine) for breast radiotherapy: volumetric and dosimetric correlations in 100 patients. Int J Radiat Oncol. 2012;84:902–9. doi: 10.1016/j.ijrobp.2012.01.040. [DOI] [PubMed] [Google Scholar]
- 58.Fan L-L, Luo Y-K, Xu J-H, He L, Wang J, Du X. A dosimetry study precisely outlining the heart substructure of left breast cancer patients using intensity-modulated radiation therapy. J Appl Clin Med Phys. 2014;15:4624. doi: 10.1120/jacmp.v15i5.4624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Würschmidt F, Stoltenberg S, Kretschmer M, Petersen C. Incidental dose to coronary arteries is higher in prone than in supine whole breast irradiation. Strahlenther Onkol. 2014;190:563–8. doi: 10.1007/s00066-014-0606-4. [DOI] [PubMed] [Google Scholar]
- 60.Kirova YM, Hijal T, Campana F, Fournier-Bidoz N, Stilhart A, Dendale R. et al. Whole breast radiotherapy in the lateral decubitus position: a dosimetric and clinical solution to decrease the doses to the organs at risk (OAR) Radiother Oncol. 2014;110:477–81. doi: 10.1016/j.radonc.2013.10.038. [DOI] [PubMed] [Google Scholar]
- 61.Bartlett FR, Colgan RM, Donovan EM, Carr K, Landeg S, Clements N. et al. Voluntary breath-hold technique for reducing heart dose in left breast radiotherapy. J Vis Exp. 2014:89. doi: 10.3791/51578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Mulliez T, Veldeman L, Vercauteren T, De Gersem W, Speleers B, Van Greveling A. et al. Reproducibility of deep inspiration breath hold for prone left-sided whole breast irradiation. Radiat Oncol. 2015;10:9. doi: 10.1186/s13014-014-0313-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bartlett FR, Colgan RM, Donovan EM, McNair HA, Carr K, Evans PM. et al. The UK HeartSpare Study (tage IB): randomised comparison of a voluntary breath-hold technique and prone radiotherapy after breast conserving surgery. Radiother Oncol. 2015;114:66–72. doi: 10.1016/j.radonc.2014.11.018. [DOI] [PubMed] [Google Scholar]
- 64.Merino Lara TR, Fleury E, Mashouf S, Helou J, McCann C, Ruschin M. et al. Measurement of mean cardiac dose for various breast irradiation techniques and corresponding risk of major cardiovascular event. Front Oncol. 2014;4:284. doi: 10.3389/fonc.2014.00284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Moran JM, Ben-David MA, Marsh RB, Balter JM, Griffith KA, Hayman JA. et al. Accelerated partial breast irradiation: what is dosimetric effect of advanced technology approaches? Int J Radiat Oncol. 2009;75:294–301. doi: 10.1016/j.ijrobp.2009.03.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lettmaier S, Kreppner S, Lotter M, Walser M, Ott OJ, Fietkau R. et al. Radiation exposure of the heart, lung and skin by radiation therapy for breast cancer: a dosimetric comparison between partial breast irradiation using multicatheter brachytherapy and whole breast teletherapy. Radiother Oncol. 2011;100:189–94. doi: 10.1016/j.radonc.2010.07.011. [DOI] [PubMed] [Google Scholar]
- 67.Mészáros N, Major T, Stelczer G, Zaka Z, Mózsa E, Pukancsik D. et al. Implementation of image-guided intensity-modulated accelerated partial breast irradiation: three-year results of a phase II clinical study. Strahlenther Onkol. 2017;193:70–9. doi: 10.1007/s00066-016-1074-9. [DOI] [PubMed] [Google Scholar]
- 68.Piroth MD, Petz D, Pinkawa M, Holy R, Eble MJ. Usefulness of a thermoplastic breast bra for breast cancer radiotherapy. Strahlenther Onkol. 2016;192:609–16. doi: 10.1007/s00066-016-0981-0. [DOI] [PubMed] [Google Scholar]
- 69.Balaji K, Subramanian B, Yadav P, Anu Radha C, Ramasubramanian V. Radiation therapy for breast cancer: literature review. Med Dosim. 2016;41:253–7. doi: 10.1016/j.meddos.2016.06.005. [DOI] [PubMed] [Google Scholar]
- 70.Arsene-Henry A, Fourquet A, Kirova YM. Evolution of radiation techniques in the treatment of breast cancer (BC) patients: from 3D conformal radiotherapy (3D CRT) to intensity-modulated RT (IMRT) using helical tomotherapy (HT) Radiother Oncol. 2017;124:333–4. doi: 10.1016/j.radonc.2017.07.002. [DOI] [PubMed] [Google Scholar]
- 71.Cozzi L, Lohr F, Fogliata A, Franceschini D, De Rose F, Filippi AR. et al. Critical appraisal of the role of volumetric modulated arc therapy in the radiation therapy management of breast cancer. Radiat Oncol. 2017;12:1–12. doi: 10.1186/s13014-017-0935-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Borca VC, Franco P, Catuzzo P, Migliaccio F, Zenone F, Aimonetto S. et al. Does TomoDirect 3DCRT represent a suitable option for post-operative whole breast irradiation? A hypothesis-generating pilot study. Radiat Oncol. 2012;7:211. doi: 10.1186/1748-717X-7-211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Virén T, Heikkilä J, Myllyoja K, Koskela K, Lahtinen T, Seppälä J.. Tangential volumetric modulated arc therapy technique for left-sided breast cancer radiotherapy. Radiat Oncol. 2015;10:79. doi: 10.1186/s13014-015-0392-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhao H, He M, Cheng G, Han D, Wu N, Shi D. et al. A comparative dosimetric study of left sided breast cancer after breast-conserving surgery treated with VMAT and IMRT. Radiat Oncol. 2015;10:231. doi: 10.1186/s13014-015-0531-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Tan W, Wang X, Qiu D, Liu D, Jia S, Zeng F. et al. Dosimetric comparison of intensity-modulated radiotherapy plans, with or without anterior myocardial territory and left ventricle as organs at risk, in early-stage left-sided breast cancer patients. Int J Radiat Oncol Biol Phys. 2011;81:1544–51. doi: 10.1016/j.ijrobp.2010.09.028. [DOI] [PubMed] [Google Scholar]
- 76.Zhang L, Mei X, Chen X, Hu W, Hu S, Zhang Y. et al. Estimating cardiac substructures exposure from diverse radiotherapy techniques in treating left-sided breast cancer. Medicine (Baltimore) 2015;94:e847. doi: 10.1097/MD.0000000000000847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Haciislamoglu E, Colak F, Canyilmaz E, Dirican B, Gurdalli S, Yilmaz AH. et al. Dosimetric comparison of left-sided whole-breast irradiation with 3DCRT, forward-planned, IMRT, inverse-planned, IMRT helical tomotherapy, and volumetric arc therapy. Phys Medica. 2015;31:360–7. doi: 10.1016/j.ejmp.2015.02.005. [DOI] [PubMed] [Google Scholar]
- 78.Baycan D, Karacetin D, Balkanay AY, Barut Y. Field-in-field IMRT versus 3D-CRT of the breast. Cardiac vessels, ipsilateral lung, and contralateral breast absorbed doses in patients with left-sided lumpectomy: a dosimetric comparison. Jpn J Radiol. 2012;30:819–23. doi: 10.1007/s11604-012-0126-z. [DOI] [PubMed] [Google Scholar]
- 79.Lohr F, El-Haddad M, Dobler B, Grau R, Wertz H-J, Kraus-Tiefenbacher U. et al. Potential effect of robust and simple IMRT approach for left-sided breast cancer on cardiac mortality. Int J Radiat Oncol Biol Phys. 2009;74:73–80. doi: 10.1016/j.ijrobp.2008.07.018. [DOI] [PubMed] [Google Scholar]
- 80.Rudat V, Nour A, Ghaida SA, Alaradi A. Impact of hypofractionation and tangential beam IMRT on the acute skin reaction in adjuvant breast cancer radiotherapy. Radiat Oncol. 2016;11:100. doi: 10.1186/s13014-016-0674-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ciammella P, Podgornii A, Galeandro M, Micera R, Ramundo D, Palmieri T. et al. Toxicity and cosmetic outcome of hypofractionated whole-breast radiotherapy: predictive clinical and dosimetric factors. Radiat Oncol. 2014;9:97. doi: 10.1186/1748-717X-9-97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lee E, Takita C, Wright JL, Reis IM, Zhao W, Nelson OL. et al. Characterization of risk factors for adjuvant radiotherapy-associated pain in a tri-racial/ ethnic breast cancer population. Pain. 2016;157:1122–31. doi: 10.1097/j.pain.0000000000000489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Mak KS, Chen Y-H, Catalano PJ, Punglia RS, Wong JS, Truong L. et al. Dosimetric inhomogeneity predicts for long-term breast pain after breast-conserving therapy. Int J Radiat Oncol Biol Phys. 2014;93:1087–95. doi: 10.1016/j.ijrobp.2014.05.021. [DOI] [PubMed] [Google Scholar]
- 84.Stimato G, Ippolito E, Silipigni S, Venanzio C Di, Rinaldi CG, Gaudino D. et al. A new three-dimensional conformal radiotherapy (3DCRT) technique for large breast and/or high body mass index patients: evaluation of a novel fields assessment aimed to reduce extra–target-tissue irradiation. Br J Radiol. 2016;89:20160039. doi: 10.1259/bjr.20160039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Mulliez T, Speleers B, Madani I, De Gersem W, Veldeman L, De Neve W. Whole breast radiotherapy in prone and supine position: is there a place for multi-beam IMRT? Radiat Oncol. 2013;8:151. doi: 10.1186/1748-717X-8-151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Goldsmith C, Haviland J, Tsang Y, Sydenham M, Yarnold J. Large breast size as a risk factor for late adverse effects of breast radiotherapy: is residual dose inhomogeneity, despite 3D treatment planning and delivery, the main explanation? Radiother Oncol. 2011;100:236–40. doi: 10.1016/j.radonc.2010.12.012. [DOI] [PubMed] [Google Scholar]
- 87.Juneja P, Bonora M, Haviland JS, Harris E, Evans P, Somaiah N. Does breast composition influence late adverse effects in breast radiotherapy? Breast. 2016;26:25–30. doi: 10.1016/j.breast.2015.12.004. [DOI] [PubMed] [Google Scholar]
- 88.Kirby AM, Evans PM, Donovan EM, Convery HM, Haviland JS, Yarnold JR. Prone versus supine positioning for whole and partial-breast radiotherapy: a comparison of non-target tissue dosimetry. Radiother Oncol. 2010;96:178–84. doi: 10.1016/j.radonc.2010.05.014. [DOI] [PubMed] [Google Scholar]
- 89.Aznar MC, Korreman S-S, Pedersen AN, Persson GF, Josipovic M, Specht L. Evaluation of dose to cardiac structures during breast irradiation. Br J Radiol. 2011;84:743–6. doi: 10.1259/bjr/12497075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.van den Bogaard VAB, Ta BDP, van der Schaaf A, Bouma AB, Middag AMH, Bantema-Joppe EJ. et al. Validation and modification of a prediction model for acute cardiac events in patients with breast cancer treated with radiotherapy based on three-dimensional dose distributions to cardiac substructures. J Clin Oncol. 2017;35:1171–8. doi: 10.1200/JCO.2016.69.8480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Haciislamoglu E, Colak F, Canyilmaz E, Zengin AY, Yilmaz AH, Yoney A. et al. The choice of multi-beam IMRT for whole breast radiotherapy in early-stage right breast cancer. Springerplus. 2016;5:688. doi: 10.1186/s40064-016-2314-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Coles CE, Griffin CL, Kirby AM, Titley J, Agrawal RK, Alhasso A. et al. Partial-breast radiotherapy after breast conservation surgery for patients with early breast cancer (UK IMPORT LOW trial): 5-year results from a multicentre, randomised, controlled, phase 3, non-inferiority trial. Lancet. 2017;390:1048–60. doi: 10.1016/S0140-6736(17)31145-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Strnad V, Ott OJ, Hildebrandt G, Kauer-Dorner D, Knauerhase H, Major T. et al. 5-year results of accelerated partial breast irradiation using sole interstitial multicatheter brachytherapy versus whole-breast irradiation with boost after breast-conserving surgery for low-risk invasive and in-situ carcinoma of the female breast: a randomised, phase 3, non-inferiority trial. Lancet. 2016;387:229–38. doi: 10.1016/S0140-6736(15)00471-7. [DOI] [PubMed] [Google Scholar]
- 94.Ott OJ, Strnad V, Hildebrandt G, Kauer-Dorner D, Knauerhase H, Major T. et al. GEC-ESTRO multicenter phase 3-trial: Accelerated partial breast irradiation with interstitial multicatheter brachytherapy versus external beam whole breast irradiation: Early toxicity and patient compliance. Radiother Oncol. 2016;120:119–23. doi: 10.1016/j.radonc.2016.06.0. [DOI] [PubMed] [Google Scholar]


