Abstract
Domesticated Atlantic salmon grow much faster than wild salmon when reared together in fish tanks under farming conditions (size ratios typically 1:2–3). In contrast, domesticated salmon only display marginally higher growth than wild salmon when reared together in rivers (size ratios typically 1:1–1.2). This begs the question why? Is this a difference in the plastic response driven by divergent energy budgets between the two environments, or is it a result of selection, whereby domesticated salmon that display the greatest growth-potential are those at greatest risk of mortality in the wild? We reared domesticated, hybrid and wild salmon in a river until they smoltified at age 2 or 4, and thereafter in fish tanks for a further 2 years. In the river, there was no difference in the mean size between the groups. In contrast, after being transferred from the river to fish tanks, the domesticated salmon significantly outgrew the wild salmon (maximum size ratio of ~1:1.8). This demonstrates that selection alone cannot be responsible for the lack of growth differences observed between domesticated and wild salmon in rivers. Nevertheless, the final size ratios observed after rearing in tanks were lower than expected in that environment, thus suggesting that plasticity, as for selection, cannot be the sole mechanism. We therefore conclude that a combination of energy-budget plasticity, and selection via growth-potential mortality, cause the differences in growth reaction norms between domesticated and wild salmon across these contrasting environments. Our results imply that if phenotypic changes are not observed in wild populations following introgression of domesticated conspecifics, it does not mean that functional genetic changes have not occurred in the admixed population. Clearly, under the right environmental conditions, the underlying genetic changes will manifest themselves in the phenotype.
Introduction
The domestication of Atlantic salmon (Salmo salar L.) was initiated in the early 1970’s when wild fish were collected from a selection of Norwegian rivers and thereafter used as broodstock in breeding programs1,2. For each generation that passed, fish were exposed to directional selection for an increasing number of economically important traits, such as rapid growth, delayed maturation, desirable flesh characteristics and increased disease resistance2,3. Genetically improved strains of Atlantic salmon for commercial aquaculture have subsequently been established in most of the major salmon producing countries4–7, and this species is regarded as one of the most domesticated fish reared for food8.
Genetic differences in the expression of a wide range of phenotypic traits now exist between domesticated and wild Atlantic salmon9. However, the trait displaying the largest and most consistent difference between domesticated and wild salmon is growth, typically measured as size at age, under controlled farming conditions10–13. This stands to reason given that growth displays a high heritability in salmon3,14, and has represented the most important target of selection. The most recent studies typically reveal between 2 to 3-fold differences in size at age between domesticated and wild fish when reared together in fish tanks under farming conditions11–13,15, although up to 5-fold differences have been documented15. Multiple quantitative trait loci associated with growth have been reported in Atlantic salmon16–18, perhaps not surprisingly, as this trait is regarded to be under polygenic control19. Variation in this trait is primarily, although not exclusively20, explained by an additive genetic model. This is further evidenced by the fact that F1 hybrids typically display intermediate growth rates between their fast growing domesticated and slow growing wild parents13,15,21,22.
As for other species of fish, growth is highly plastic in salmon11,15,22,23. Significantly, there are differences in reaction norms between domesticated and wild salmon, and in the natural environment, growth of domesticated salmon, measured as size at age, is only marginally higher than for wild salmon17,24–27. This stands in stark contrast to the very large differences observed in growth between domesticated and wild salmon when reared together in fish tanks10–13,15, and begs the question, why? Is this driven by a difference in energy budgets between the two environments? For example, as rivers display a limited production capacity28–30, could food be limiting in the natural environment, thus not providing domesticated salmon with enough energy to utilize their high genetic growth-potential and outgrow wild salmon? Also, are domesticated salmon less effective at catching prey in the wild, and do they expend more energy in their search for them? Or alternatively, could individual domesticated salmon displaying the highest growth-potential be more susceptible to predation in the wild than individual domesticated fish displaying lower growth-potential? Such a selection mechanism could result in more similar growth, recorded as size at age, among surviving domesticated salmon and wild salmon in the natural environment. Although some of the selection during freshwater is density-dependent28,29, earlier studies have suggested that deviating growth-potential mortality may influence the growth relationship between domesticated and wild salmon15, and that growth-potential may be negatively correlated with survival in the wild31–33. Survival from egg to smolt is typically ~1–3% for Atlantic salmon in the wild24,34–36, and the offspring of domesticated salmon display higher mortality rates than the offspring of wild salmon in the natural environment24,27,34,37. Thus, natural selection has significant potential to select against sub-optimal phenotypes, such as domesticated fish displaying a high genetic growth capacity. In turn, this could result in more similar size distributions between fish of domestic and wild pedigree in the natural environment.
Using two complimentary approaches, the present study was designed to investigate why domesticated and wild salmon display deviating growth distributions across the contrasting natural and farming environments. In the first approach, we studied growth of domesticated, F1 hybrid and wild Atlantic salmon that lived the first 2–4 years of their lives in a natural river system, that were thereafter transferred to fish tanks and reared on high-energy pellets for a further 2 years. Our reasoning was such: if high growth-potential and/or size-selective mortality (hereon collectively referred to as “growth-potential” mortality) represents the primary mechanism preventing size differences developing between the groups in the wild, then the growth reaction norms of survivors from the wild should be similar when transferred to a high energy commercial diet in fish tanks. If however, this primarily represents a genetic difference in the plastic response to the deviating energy-budgets between the environments, then the domesticated salmon would be expected to out-grow the wild salmon when transferred to fish tanks and provided a high energy diet.
In the second approach, and to compliment the main experiment, we implemented a weight-dependent mortality function to simulate selection via growth-potential mortality on a data set documenting growth of wild, domesticated and hybrid salmon smolts in fish tanks. We did this to investigate whether it was possible to create overlapping weight distributions of these genetic groups, similar to the weigh distributions of these groups observed in a river24, by a size-dependent mortality function alone. The mortality rates required to create overlapping weight distributions between domesticated and wild salmon were thereafter compared to observed mortality rates of these groups in the wild.
Results
Numbers of fish sampled in the river and reared thereafter in tanks
In the period 14–22 May 2012, 360 seaward-migrating salmon smolts that had survived in the river Guddal since being planted out as eyed eggs in 2008 and 2010, were sampled on the downstream fish trap. These survivors were thereafter transferred to fish tanks at the Matre fish farm for continued rearing. DNA parentage testing revealed that the majority of the 360 fish gathered from the trap were 4 year-old smolts from the 2008 cohort, and of wild pedigree (268/10, 14/9 and 56/3 wild, hybrid and domesticated individuals, from the 2008/2010 cohorts respectively). Nevertheless, all three genetic groups, and both smolt age groups, were represented in the total material collected. These data are attached (Supplementary File 1).
Growth of fish sampled in the river and reared thereafter in tanks
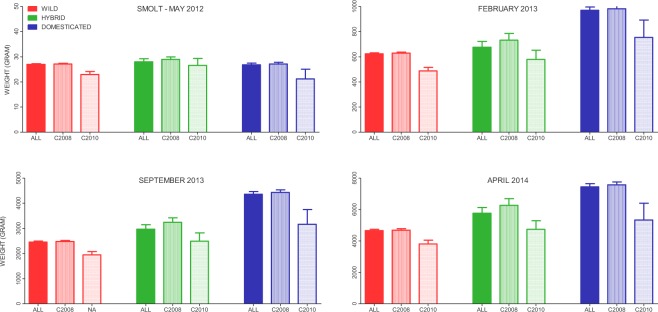
No significant difference in smolt size was observed among salmon of domesticated, hybrid and wild pedigree when measured on the downstream fish migration trap in the river at timepoint 1 (Fig. 1, Table 1). This gave an overall size ratio of ~1:1 at the smolt stage in the wild (Fig. 2). However, after 10, 17 and 24 months of rearing in tanks (timepoints 2, 3 and 4 respectively), significant differences in fish size were detected among salmon of domesticated, hybrid and wild pedigree (Fig. 1, Table 1) (Strainxtime interaction: F6,957 = 65.6, P < 0.001, Strain: F2,21 = 19.7, P < 0.001, Time: F3,957 = 29 332, P < 0.001). The largest difference between these groups was detected after 17 months of tank rearing, resulting in a maximum size ratio of 1:1.8 between wild and domesticated salmon (Fig. 2).
Figure 1.
Mean size (g) of wild, hybrid and domesticated salmon as measured at timepoint 1 (May 2012, sampled in the river, age 2–4 years), and timepoints 2–4 (February 2013-April 2014, sampled in fish tanks, age 3–6 years). Error bars show the standard error.
Table 1.
Statistical results for growth (size) and condition factor of domesticated, hybrid and wild salmon upon sampling at timepoint 1 (aged 2–4 years, sampled in river) and timepoints 2–4 (aged 3–6 years, sampled in fish tanks).
| Domesticated | Hybrid | Wild | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Timepoint | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 | 1 | 2 | 3 | 4 |
| Weight (g) | 27.0 | 979.9 | 4356.5 | 7450.0 | 28.1 | 676.4 | 2968.2 | 5660.0 | 27.2 | 623.6 | 2459.2 | 4659.7 |
| K | 0.83 | 1.21 | 1.40 | 1.29 | 0.86 | 1.11 | 1.27 | 1.27 | 0.85 | 1.14 | 1.27 | 1.23 |
| Domesticated | — | — | — | — | 0.41 | <0.001 | <0.001 | 0.38 | 0.21 | <0.001 | <0.001 | <0.001 |
| Hybrid | 0.23 | 0.001 | 0.002 | 0.03 | — | — | — | — | 1.00 | 0.16 | 0.93 | 0.10 |
| Wild | 0.77 | <0.001 | <0.001 | <0.001 | 0.13 | 0.09 | 0.003 | 0.001 | — | — | — | — |
Upper and lower triangle shows the P-value of the pairwise comparisons of the condition factor (K) and the body weight (italic), respectively, of domesticated, hybrid and wild salmon at the different time points. Significant results are marked in bold.
Figure 2.
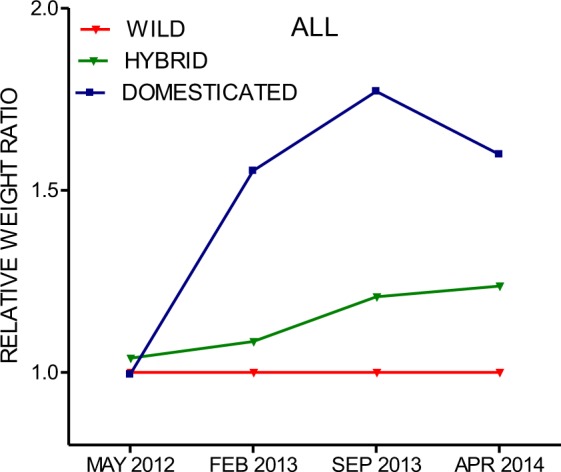
Size ratio among wild, hybrid and domesticated salmon sampled at timepoint 1 (May 2012, sampled in the river, age 2–4 years), and timepoints 3–4 (February 2013–April 2014, sampled in fish tanks, age 3–6 years).
A significant difference in mean size was observed between fish from the two cohorts, with older smolts being larger (cohort/smolt age: F1,26 = 8.3, P = 0.008) (Fig. 1). This difference in weight between cohorts were consistent for all three genetic groups (Strainxcohort interaction: F2,26 = 0.48, P = 0.6), and across all time points (CohortxTime interaction: F3,26 = 0.48, P = 0.6). Thus, the fish that smoltified at an older age, were consistently larger throughout the whole study, within their respective genetic groups.
Mortality and condition factor
After 2 months of being reared in fish tanks on a combined diet of insect larvae (bloodworms, Chironomidae) and pellets, daily observations indicated that all fish were weaned onto pellets. This observation was supported by the low mortality observed during this and subsequent periods. During this transition period, and up until the next sample was taken (timepoint 2, ten months after transfer fish tanks), overall mortality was 8.33%. This corresponded to 8.63% (N = 24), 4.35% (N = 1) and 8.47% (N = 5) of the wild, hybrid and domesticated individuals respectively.
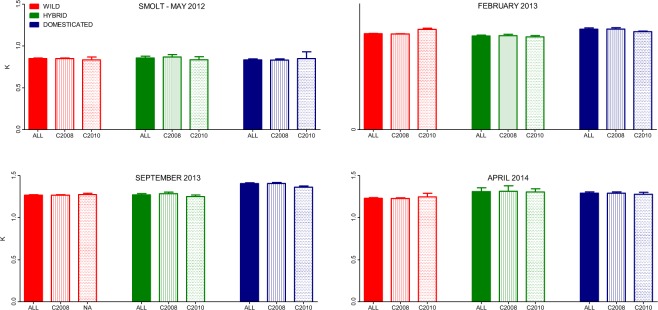
No significant difference in condition factor were observed among salmon of wild, hybrid and domesticated pedigree sampled on the fish trap at timepoint 1 (Fig. 3, Table 1) and all fish displayed a significant increase in condition factor after a period of rearing in the fish tanks (Fig. 3, Table 1). While hybrid and wild salmon displayed similar condition factors throughout the tank rearing stage of the experiment, domesticated salmon displayed an increasing K (Strainxtime interaction: F6,962 = 13.4, P < 0.001, Strain: F2,20 = 12.3, P = 0.003, Time: F3,963 = 775.6, P < 0.001, Table 1).
Figure 3.
Mean condition factor of wild, hybrid and domesticated salmon sampled at timepoint 1 (May 2012, sampled in the river, age 2–4 years), and timepoints 2–4 (February 2013-April 2014, sampled in fish tanks, age 3–6 years). Error bars show the standard error.
No significant difference in condition factor was observed between fish from the two cohorts at any time point, in any of the genetic groups (Strainxcohort interaction: F2,43 = 0.3, P = 0.7, TimexCohort interaction: F3,965 = 1.02, P = 0.4, Cohort: F1,41 = 0.02, P = 0.9).
Modelling size selective mortality
We modelled size-selective mortality, as a proxy for growth-potential mortality, to investigate whether it was possible to create overlapping size distributions using selection alone. This was achieved using a simulated data set consisting of 107 wild, hybrid and domesticated individuals created using the mean and the standard deviation of the genetic groups’ weight in a previously published data set documenting growth of salmon from multiple strains in fish tanks38 (Supplementary File 2).
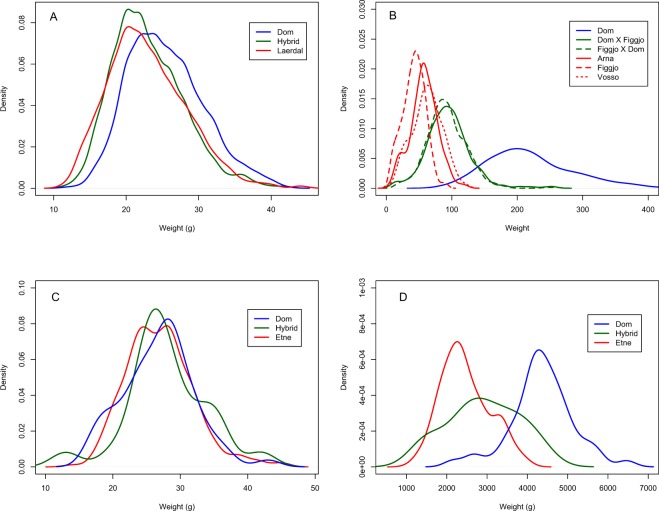
In a previous study where growth of wild, hybrid and domesticated salmon was investigated in the river Guddal24, observed smolt weights were highly overlapping between the genetic groups (22.9 ± 5.3, 23.0 ± 4.9 and 25.4. ± 5.1 g respectively, Fig. 4A). In contrast, divergent weight distributions were observed in the data set documenting growth of multiple wild, hybrid and domesticated salmon smolts reared in fish tanks at the Matre fish farm (54.9 ± 22.5, 93.6 ± 32.5 and 217.6 ± 65.1 g respectively, Fig. 4B)38. This data set was used for the growth-potential mortality simulation here. We set the mean observed weight of the wild fish in the tanks (54.9 g) as the optimum for survival, based upon the assumption that wild fish growth represents the optimum resulting from natural selection. Thereafter, the probability of mortality in all three genetic groups was modelled as a function of each individual fish’s weight-deviation from the optimum. The results of this simulation demonstrated that in order to create overlapping weight distributions between salmon of wild, hybrid and domesticated origin reared under farming conditions, the differential mortality rates of the three genetic groups needed to be far greater than observed in the wild (Table 2 vs.24,27,34,37). Thus, this simulation, which was used to supplement the main growth study, suggests that growth-potential or size-selective mortality alone cannot explain why domesticated salmon do not outgrow wild salmon in the natural environment.
Figure 4.
Weight distributions of domesticated, hybrid and wild salmon from data sets collected in the wild, and from fish tanks: (A) Smolts sampled in the river Guddal from a previously published study of survival and growth24, (B) Smolts sampled from multiple strains in fish tanks that had been reared in that environment from hatching onwards38 and that were used for the modelling simulations in this work, (C) Smolts sampled in the river Guddal in 2012 that form the experimental work in the present study (N = 360), (D) Smolts transferred from the river Guddal to the fish farm and after ~17 months of rearing in fish tanks (N = 321). Dom = domesticated strain, Dom × Figgjo and Figgjo × Dom = hybrid variants, while Lærdal, Figgjo, Arna, Vosso, and Etne = wild populations.
Table 2.
Modelled mortality rates and the resulting weight distributions of domesticated, hybrid and wild salmon.
| x | Weight | Resulting mortality | Weight (gram ± SD) | ||||
|---|---|---|---|---|---|---|---|
| optimum | Domesticated | Hybrid | Wild | Domesticated | Hybrid | Wild | |
| % | % | % | Mean | Mean | Mean | ||
| 0.01 | mean(Wild) | 85.1 | 80.1 | 80 | 198 ± 58 | 93 ± 32 | 55 ± 22 |
| 0.025 | mean(Wild) | 96.0 | 82.0 | 80.2 | 144 ± 51 | 88 ± 29 | 54 ± 22 |
| 0.05 | mean(Wild) | 98.9 | 88.1 | 81.3 | 96 ± 36 | 76 ± 23 | 54 ± 19 |
| 0.075 | mean(Wild) | 99.5 | 91.7 | 83.8 | 76 ± 25 | 68 ± 19 | 54 ± 16 |
| 0.1 | mean(Wild) | 99.8 | 93.7 | 86.2 | 67 ± 19 | 64 ± 16 | 54 ± 14 |
The initial data set used for this modelling was smolt weights of domesticated, hybrid and wild salmon from tank-reared salmon (Fig. 4B). The average weight of the wild salmon was set as the optimal for survival. x represents the parameter changing the strength of weight-dependent mortality function, implemented as a proxy to simulate growth-potential mortality.
Discussion
This is the first study to investigate growth of domesticated, hybrid and wild salmon in the natural environment, and thereafter in fish tanks. After 2–4 years in the river, no difference in size was observed among these genetically distinct groups (Figs 1, 2, 4C). However, once these fish were transferred from the river to tanks, and fed a high-energy pellet diet, the genetic growth potential of the domesticated salmon was suddenly unleashed, resulting in highly significant size differences (Figs 1, 2, 4D). This demonstrates that selection alone cannot be responsible for the lack of growth differences observed between domesticated and wild salmon in rivers. Nevertheless, the largest size ratios observed here after the fish had been transferred to and reared in fish tanks (1:1.8), were lower than typically reported between domesticated and wild fish reared in that environment (typically 1:2–1:3, albeit with exceptions)10–12,15,39. Furthermore, the differences were much lower than observed in a study conducted in fish tanks (1:2.9)11 that used part of the same genetic material as in the present study. We therefore conclude that genetic differences between domesticated and wild salmon, in their plastic response to the divergent energy budgets between the river and fish tanks, cannot be the sole cause of the observed large differences in their norms of reaction across these contrasting environments. It therefore follows that a combination of selection and plasticity are responsible for the large differences in growth reaction norms between domesticated and wild salmon across the natural and farming environments.
The offspring of domesticated salmon display lower survival in the natural environment when compared to the offspring of wild salmon24,27,34,37. Studies have also indicated that the offspring of domesticated salmon are more naïve to predators than the offspring of wild salmon40,41, and that there is a trade-off between fast growing domesticated fish and survival32,33. Nevertheless, while growth-potential selection against the fastest growing domesticated fish could prevent size differences developing between domesticated and wild salmon in the natural environment, our results demonstrate that this cannot be the primary force at work. Clearly, the majority of the domesticated fish surviving for 2–4 years in the river still displayed a significantly higher growth potential than the wild fish, illustrated by the fact that they suddenly out-grew the wild salmon when transferred into fish tanks and onto a pellet diet (Fig. 4C vs. D). If selection via growth-potential mortality was the primary driver, we would not have expected to observe this. Other factors, potentially in combination with a degree of growth-potential selection, must therefore be responsible for the observed differences in growth reaction norms.
After modelling size-selective mortality on a data set documenting hatchery growth of smolts of domesticated, hybrid and wild origin, as a proxy for growth-potential selection, we were still not able to create overlapping weight distributions that are observed in the wild (Fig. 4A,C) at mortality rates similar to those observed for domesticated and wild salmon in rivers24,27,34,37. To create even close to overlapping weight distributions (Table 2), the morality rate of wild salmon had to be modelled far lower than what is observed for egg to smolt survival in the wild24,34–36, while the morality rate of domesticated salmon had to be modelled far higher than has been reported in the wild24,34. While the modelling exercise is arguably simplistic, it nevertheless supports the results from the main experimental part of this study.
It could be reasoned that the domesticated fish were able to outgrow the wild fish after being transferred from the river to fish tanks, due to genetic differences in their abilities to tackle this transition. While it is not possible to unequivocally exclude this possibility, we find it unlikely that it played a major role in our results for the following reasons. After being transferred to the fish tanks, fish of all genetic groups displayed very low mortality, substantial gains in growth (Figs 1, 2), and an increase in condition factor (Fig. 3), all of which demonstrate that fish of all genetic backgrounds managed the transition well. In addition, earlier studies have investigated whether domestication has led to adaptations to specific factors associated with farming conditions, such as; handling stress11, high rearing densities and social interactions15,42, feeding regimes22, and high-energy and pellet-based diets39. However, none of the above factors appear to inflate the relative growth differences between domesticated and wild salmon reared in fish tanks in farms.
Domestication of Atlantic salmon has resulted in a faster growing fish that displays increased consumption, metabolism and potentially feed conversion efficiency when presented with an excess and high energy diet in a protected environment such as a farm, as opposed to adaptations to the specific physical and social conditions of the farm per se9. Significantly, when growth has been investigated under farming and semi-natural conditions, both with restricted access to feed, differences between domesticated and wild salmon were less than under standard farming conditions with excess feed15. Thus, in the light of the results from the present study, it would seem highly likely that part of the difference in growth between domesticated and wild salmon in the natural habitat vs the fish farm is a difference in genetic based plasticity, mediated through the lack of available energy, or the accessibility of that energy, for the domesticated salmon to out-grow wild fish in the wild. This situation may also be exacerbated by domesticated salmon using more energy in the natural environment, as has been reported in growth-enhanced brown trout (Salmo trutta L.)43.
Over the past three to four decades, tens of millions of domesticated salmon have escaped into the wild9,44, and introgression of domesticated salmon has been documented in Norwegian45–48, Irish49,50, Scottish51, and Canadian populations52,53. All available evidence suggests that introgression of domesticated escapees represents a threat to the genetic integrity, abundance and long-term evolutionary viability of native populations9. Despite this, direct evidence of phenotypic, life-history or demographic changes in native populations where introgression of domesticated escapees has been documented, is sparse9. A recent multiple-river study demonstrated that fish hatched in the wild, but displaying domestication-admixture, had slightly higher average size at maturity than pure wild fish from the same river54. However, a modelling study indicated that at low to modest domestication introgression levels, clear differences in mean freshwater growth are unlikely to emerge in wild populations, in part due to selection against the offspring of domesticated salmon, and in part due to plasticity in growth55. In this context, results of the present study provide a salient reminder that even if clear phenotypic changes are not observed following introgression of domesticated escapees, it does not demonstrate that functional genetic changes have not occurred in the wild population. Changes in trait expression may occur in domestication-admixed populations over time, or under changing environmental conditions. To this end, our data clearly show that under the right conditions, the underlying genetic changes may manifest themselves in the phenotype – thus removing the sheep´s clothing, and unveiling the wolf.
Materials and Methods
Genetic material
The study is based upon domesticated, F1 hybrid and wild salmon. To produce the domesticated fish, we used the commercial Mowi strain. This had been in domestication for ≥10 generations at the time of stripping, and has previously been demonstrated to display several-fold higher growth rates, measured as size at age, than wild salmon under farming conditions10,11,23. To produce the wild fish, we used adult salmon from the river Etne. To ensure that these fish had been born in the wild, scales of broodfish were checked56. F1 hybrids between these two strains were produced by fertilizing eggs from a domesticated female with sperm from a wild male. In the autumn of 2007 and 2009, a total of 17 (7 wild, 2 hybrid and 8 domesticated) and 29 (10 wild, 9 hybrid and 10 domesticated) families were produced.
Production of fish in the river Guddal
In the spring of 2008 and 2010, 69 800 (31 800 wild, 9 400 hybrid and 28 600 domesticated) and 106 000 eyed (34 800 wild, 32 000 hybrid and 39 200 domesticated) eggs were planted into the river Guddal in Hardanger, western Norway. This material forms part of an ongoing long-term study of fitness in the natural environment, following up from earlier work24,57. Consequently, details about group and family performance in the river are not presented as the fish used here only represent a small sub-set included in the main study.
The river Guddal is partly-fed by the Folgefonna glacier, so it is a summer-cold river. Consequently, salmon grow relatively slowly in this river, and the typical age for smolts is 3–4 years, although smolt ages of 1–5 years have been reported24. Survival from egg to smolt in this river is typically 1–3%24, and a native brown trout (Salmo trutta L.) population, that predates on emerging salmon juveniles exists57. This river is otherwise typical for a small Norwegian river located on the western part of Norway. After being planted out as eyed eggs, fish representing the above groups were left alone until they smoltified and exited the river.
In the spring of each year, smolts migrating out of the river Guddal are captured on a permanent trapping facility. In the period 14–22 May 2012, 360 fish, representing a random sub-set of the salmon smolts migrating out of the river that season were measured (weight, length, fin clip taken), and thereafter transferred to the Matre research facility (timepoint 1 in the results). The transferred sub-population consisted of a total of 278, 23 and 59 wild, hybrid and domesticated individuals, respectively, whereof 10, 3 and 9 individuals were 2-year-old smolts of the 2010 cohort, while the remaining where 4-year-old smolts of the 2008 cohort. In the sub-population, the 2008 and 2010 cohorts were represented by a total of 16 out of 17 (6 wild, 2 hybrid and 8 domesticated) and 10 out of 29 (4 wild, 4 hybrid and 2 domesticated) families initially planted in the river.
Continued production of the fish in tanks at the Matre research facility
Upon arrival at Matre in late May 2012, fish from all genetic groups were randomly divided into two standard 1.5 m2 tanks, then weaned onto a commercial pellet diet by mixing insect larvae (bloodworm, Chironomidae) in the tanks together with pellets. After approximately 2 months, the fish had been weaned onto the commercial diet and supplementary feeding with bloodworm was ceased. During this transition period, and up until the next sample was taken (timepoint 2) ten months after transfer to Matre, overall mortality was only 8.33%. Fish were fed continuously in excess and held on a standard light regime for Bergen. The fish were reared in tanks for nearly two years. They were sampled in February 2013 (timepoint 2 in the results, N = 330). In this sample, fish were measured, fin clip taken and individually PIT tagged to provide subsequent identification. The fish were thereafter sampled in September 2013 (timepoint 3 in the results, N = 321) and April 2014 (timepoint 4 in the results, N = 310).
DNA identification of fish
The fish included in this study were identified to their type (domesticated, F1 hybrid and wild) using DNA parentage testing. DNA analysis included amplification of six highly polymorphic microsatellite loci on an ABI sequencing machine located in the molecular genetics laboratory at the Institute of Marine Research, Bergen. DNA parentage testing, combined with an exclusion based assignment58, has been previously used in this laboratory to identify >20 000 fish to their families and groups of origin originating from both this river system24,57 and the Matre fish farm11,15,23.
DNA analysis was not only used to identify fish to their groups of origin, it was also used to connect individual fish and their biological data between sampling timepoint 1 and 2. Individual fish were identified to themselves based upon identical match in their multi-locus DNA profile. In the few cases where individuals shared an identical genetic profile to another fish, they were genotyped for a further five microsatellites to provide unambiguous individual identification. For full details regarding this procedure the reader is referred to previous work59,60.
Statistics
All analyses were carried out using the R statistical software, version 3.4.361. Linear mixed effect (LME) models were used to compare the growth trajectories, and the corresponding condition factor, of fish of wild, domesticated and hybrid genetic background. Two models were fitted using the lmer function in the lme4 package62. The full models tested for the effect of strain (S), i.e., wild/hybrid/domesticated, and cohort/smoltage (C), i.e., 2008 and 2010, and time (T), i.e., timepoints 1, 2, 3 and 4, as well as their interactions, upon log-weight (LogW) and condition factor (K). All fixed effects were coded as factors, with three, two and four levels, respectively. Due to repeated measurements of individuals in this study, their identity was included as a random effects (ID). Family (Fam) was also include as random effects, to control for variation between families. Due to the hierarchal structure of the data set, the random effects were nested. The full models:
where α is the intercept and ε is a random error. Model selection was performed backwards by the use of the step function in the lmerTest package61. Insignificant random effects were eliminated before the removal of insignificant fixed effects. Interaction terms were removed before the variables themselves:
Each model’s fit was confirmed by examining the histogram of the model residuals and by plotting the model residuals against the fitted values. The potential autocorrelation of the time series in the models were investigated using the acf function.
Simulation of growth-potential mortality
As a supplement to the main experiment, modelling was conducted to investigate whether selection alone, via growth-potential mortality, can create strongly overlapping size distributions of wild, hybrid and domesticated salmon that are typically observed in the wild. In order to achieve this, we implemented a size-selective mortality function, as a proxy for growth-potential mortality, on a data set documenting growth of three wild strains, a domesticated strain and reciprocal wild-domesticated hybrids in fish tanks under farming conditions where there has not been any selection38 (Supplementary File 2). For details about the production, rearing and sampling of these fish at Matre, see38.
Mortality was implemented in the above-mentioned data set as a function of the fish’s recorded smolt weight, using the observed mean weight of the wild salmon in the data set (54.9 g, Fig. 4B) as the optimum for survival (Wopt). This was based upon the assumption that the mean weight of the wild fish represents the “optimum” growth-potential that has evolved in the wild. Thereafter, a morality function that gives the probability of mortality (Pm) as a function of the fish weight was implemented. The mortality function was then implemented on a simulated data set consisting of 107 wild, hybrid and domesticated individuals created using the mean and the standard deviation of the genetic groups’ weight in the hatchery data set (54.9 ± 22.5, 93.6 ± 32.5 and 217.6 ± 65.1 g respectively, Fig. 4B). The absolute difference (d) between each individual (Wi) and the optimum weight (Wopt) was calculated as:
while the normalized weight difference (dn) was calculated as:
The probability of mortality (Pm) was then derived from a sigmoid function of dn:
where x is a parameter that changes the steepness of the curve, illustrating the strength of size selective mortality. Pm was at minimum when Wi = Wopt, and the minimum mortality probability was set to 80% (i.e, Pm smaller than 0.8 was set to 0.8 a posteriori).
Ethical statement
The experimental protocol (permit number 5296 (main experiment) and 5186 (data used in the modelling simulations)) was approved by the Norwegian Animal Research Authority (NARA). All welfare and use of experimental animals was performed in strict accordance with the Norwegian Animal Welfare Act. This included full anesthesia of fish using metacain (Finquel® Vet, ScanVacc, Årnes, Norway), during all four samples referred to. In addition, all personnel involved in this experiment had undergone training approved by the Norwegian Food Safety Authority, which is mandatory for all personnel running experiments involving animals included in the Animal Welfare Act.
Electronic supplementary material
Acknowledgements
This study was jointly-financed by the Norwegian research council grants INTERACT 200510, and QUANTESCAPE 216105, and the Norwegian Ministry of Trade and Fisheries. We would like to acknowledge the assistance of Ivar Helge Matre and Lise Dyrhovden for rearing fish and sampling, and Anne-Grete Sørvik for DNA analysis. We also acknowledge the helpful comments from referees.
Author Contributions
K.A.G., M.F.S., F.B. and Ø.S. conceived and designed the study. K.A.G. and Ø.S. produced the genetic material and Ø.S. was responsible for the fish in the river. K.A.G. and M.F.S. were responsible for the fish in Matre. M.F.S. conducted all data analysis, F.B. conducted all mortality-based simulations, and K.A.G. wrote the first draft of the manuscript. All authors contributed to data interpretation and writing the final manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Kevin A. Glover and Monica F. Solberg contributed equally.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-32467-2.
References
- 1.Gjedrem T, Gjoen HM, Gjerde B. Genetic-origin of Norwegian farmed Atlantic salmon. Aquaculture. 1991;98:41–50. doi: 10.1016/0044-8486(91)90369-I. [DOI] [Google Scholar]
- 2.Gjedrem T. The first family-based breeding program in aquaculture. Reviews in Aquaculture. 2010;2:2–15. doi: 10.1111/j.1753-5131.2010.01011.x. [DOI] [Google Scholar]
- 3.Gjedrem T. Genetic improvement of cold-water fish species. Aquaculture Research. 2000;31:25–33. doi: 10.1046/j.1365-2109.2000.00389.x. [DOI] [Google Scholar]
- 4.Friars GW, Bailey JK, Oflynn FM. Applications of selection for multiple traits in cage-reared Atlantic salmon (Salmo salar) Aquaculture. 1995;137:213–217. doi: 10.1016/0044-8486(95)01101-3. [DOI] [Google Scholar]
- 5.Powell J, White I, Guy D, Brotherstone S. Genetic parameters of production traits in Atlantic salmon (Salmo salar) Aquaculture. 2008;274:225–231. doi: 10.1016/j.aquaculture.2007.11.036. [DOI] [Google Scholar]
- 6.Taylor RS, Wynne JW, Kube PD, Elliott NG. Genetic variation of resistance to amoebic gill disease in Atlantic salmon (Salmo salar) assessed in a challenge system. Aquaculture. 2007;272:S94–S99. doi: 10.1016/j.aquaculture.2007.08.007. [DOI] [Google Scholar]
- 7.Yanez JM, et al. Genetic co-variation between resistance against both Caligus rogercresseyi and Piscirickettsia salmonis, and body weight in Atlantic salmon (Salmo salar) Aquaculture. 2014;433:295–298. doi: 10.1016/j.aquaculture.2014.06.026. [DOI] [Google Scholar]
- 8.Teletchea F, Fontaine P. Levels of domestication in fish: implications for the sustainable future of aquaculture. Fish. Fish. 2014;15:181–195. doi: 10.1111/faf.12006. [DOI] [Google Scholar]
- 9.Glover KA, et al. Half a century of genetic interaction between farmed and wild Atlan tic salmon: status of knowledge and unanswered questions. Fish. Fish. 2017;18:890–927. doi: 10.1111/faf.12214. [DOI] [Google Scholar]
- 10.Glover KA, et al. A comparison of farmed, wild and hybrid Atlantic salmon (Salmo salar L.) reared under farming conditions. Aquaculture. 2009;286:203–210. doi: 10.1016/j.aquaculture.2008.09.023. [DOI] [Google Scholar]
- 11.Solberg MF, Glover KA, Nilsen F, Skaala Ø. Does domestication cause changes in growth reaction norms? A study of farmed, wild and hybrid Atlantic salmon families exposed to environmental stress. Plos One. 2013;8(1):e54469. doi: 10.1371/journal.pone.0054469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Harvey A, Glover KA, Taylor MI, Creer S, Carvalho GR. A common garden design reveals population-specific variability in potential impacts of hybridization between populations of farmed and wild Atlantic salmon. Salmo salar L. Evolutionary Applications. 2016;9:435–449. doi: 10.1111/eva.12346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Harvey AC, Fjelldal PG, Solberg MF, Hansen T, Glover KA. Ploidy elicits a whole-genome dosage effect: growth of triploid Atlantic salmon is linked to the genetic origin of the second maternal chromosome set. BMC Genet. 2017;18:12. doi: 10.1186/s12863-017-0502-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tsai HY, et al. The genetic architecture of growth and fillet traits in farmed Atlantic salmon (Salmo salar) BMC Genet. 2015;16:51. doi: 10.1186/s12863-015-0215-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Solberg MF, Zhang ZW, Nilsen F, Glover KA. Growth reaction norms of domesticated, wild and hybrid Atlantic salmon families in response to differing social and physical environments. Bmc Evolutionary Biology. 2013;13:234. doi: 10.1186/1471-2148-13-234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Baranski M, Moen T, Vage DI. Mapping of quantitative trait loci for flesh colour and growth traits in Atlantic salmon (Salmo salar) Genetics Selection Evolution. 2010;42:17. doi: 10.1186/1297-9686-42-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Besnier F, et al. Identification of quantitative genetic components of fitness variation in farmed, hybrid and native salmon in the wild. Heredity. 2015;115:47–55. doi: 10.1038/hdy.2015.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Vasemagi A, Kahar S, Ozerov MY. Genes that affect Atlantic salmon growth in hatchery do not have the same effect in the wild. Funct. Ecol. 2016;30:1687–1695. doi: 10.1111/1365-2435.12635. [DOI] [Google Scholar]
- 19.Tsai HY, et al. Genome wide association and genomic prediction for growth traits in juvenile farmed Atlantic salmon using a high density SNP array. Bmc Genomics. 2015;16:969. doi: 10.1186/s12864-015-2117-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Debes PV, Fraser DJ, Yates M, Hutchings JA. The between-population genetic architecture of growth, maturation, and plasticity in Atlantic salmon. Genetics. 2014;196:1277–1291. doi: 10.1534/genetics.114.161729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Solberg MF, Fjelldal PG, Nilsen F, Glover KA. Hatching time and alevin growth prior to the onset of exogenous feeding in farmed, wild and hybrid Norwegian Atlantic salmon. Plos One. 2014;9(12):e113697. doi: 10.1371/journal.pone.0113697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Harvey A, et al. Plasticity in response to feed availability - does feeding regime influence the relative growth performance of domesticated, wild and hybrid Atlantic salmon Salmo salar parr? Journal of Fish Biology. 2016;89:1754–1768. doi: 10.1111/jfb.13076. [DOI] [PubMed] [Google Scholar]
- 23.Solberg MF, Dyrhovden L, Matre IH, Glover KA. Thermal plasticity in farmed, wild and hybrid Atlantic salmon during early development: has domestication caused divergence in low temperature tolerance? Bmc Evolutionary Biology. 2016;16:38. doi: 10.1186/s12862-016-0607-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Skaala Ø, et al. Performance of farmed, hybrid, and wild Atlantic salmon (Salmo salar) families in a natural river environment. Canadian Journal of Fisheries and Aquatic Sciences. 2012;69:1994–2006. doi: 10.1139/f2012-118. [DOI] [Google Scholar]
- 25.Reed TE, et al. Quantifying heritable variation in fitness-related traits of wild, farmed and hybrid Atlantic salmon families in a wild river environment. Heredity. 2015;115:173–184. doi: 10.1038/hdy.2015.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jonsson B, Jonsson N. Maternal inheritance influences homing and growth of hybrid offspring between wild and farmed Atlantic salmon. Aquaculture Environment Interactions. 2017;9:231–238. doi: 10.3354/aei00232. [DOI] [Google Scholar]
- 27.Fleming IA, et al. Lifetime success and interactions of farm salmon invading a native population. Proc. R. Soc. Lond. Ser. B-Biol. Sci. 2000;267:1517–1523. doi: 10.1098/rspb.2000.1173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jonsson N, Jonsson B, Hansen LP. The relative role of density-dependent and density-independent survival in the life cycle of Atlantic salmon Salmo salar. Journal of Animal Ecology. 1998;67:751–762. doi: 10.1046/j.1365-2656.1998.00237.x. [DOI] [Google Scholar]
- 29.Bacon PJ, et al. Can Conservation Stocking Enhance Juvenile Emigrant Production in Wild Atlantic Salmon? Transactions of the American Fisheries Society. 2015;144:642–654. doi: 10.1080/00028487.2015.1017655. [DOI] [Google Scholar]
- 30.Jonsson N, Jonsson B, Hansen LP. Long-term study of the ecology of wild Atlantic salmon smolts in a small Norwegian river. Journal of Fish Biology. 1998;52:638–650. doi: 10.1111/j.1095-8649.1998.tb02023.x. [DOI] [Google Scholar]
- 31.Tymchuk WE, Sundstrom LF, Devlin RH. Growth and survival trade-offs and outbreeding depression in rainbow trout (Oncorhynchus mykiss) Evolution. 2007;61:1225–1237. doi: 10.1111/j.1558-5646.2007.00102.x. [DOI] [PubMed] [Google Scholar]
- 32.Biro PA, Abrahams MV, Post JR, Parkinson EA. Predators select against high growth rates and risk-taking behaviour in domestic trout populations. Proceedings of the Royal Society B-Biological Sciences. 2004;271:2233–2237. doi: 10.1098/rspb.2004.2861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Biro PA, Abrahams MV, Post JR, Parkinson EA. Behavioural trade-offs between growth and mortality explain evolution of submaximal growth rates. Journal of Animal Ecology. 2006;75:1165–1171. doi: 10.1111/j.1365-2656.2006.01137.x. [DOI] [PubMed] [Google Scholar]
- 34.McGinnity P, et al. Genetic impact of escaped farmed Atlantic salmon (Salmo salar L.) on native populations: use of DNA profiling to assess freshwater performance of wild, farmed, and hybrid progeny in a natural river environment. Ices Journal of Marine Science. 1997;54:998–1008. [Google Scholar]
- 35.Buck RJG, Hay DW. The relation between stock size and progeny of Atlantic salmon, Salmo salar L., in a Scottish stream. Journal of Fish Biology. 1984;24:1–11. doi: 10.1111/j.1095-8649.1984.tb04771.x. [DOI] [Google Scholar]
- 36.Chadwick EMP. Stock-recruitment relationship for Atlantic salmon (Salmo salar) in Newfoundland rivers. Canadian Journal of Fisheries and Aquatic Sciences. 1982;39:1496–1501. doi: 10.1139/f82-201. [DOI] [Google Scholar]
- 37.McGinnity P, et al. Fitness reduction and potential extinction of wild populations of Atlantic salmon. Salmo salar, as a result of interactions with escaped farm salmon. Proc. R. Soc. Lond. Ser. B-Biol. Sci. 2003;270:2443–2450. doi: 10.1098/rspb.2003.2520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jørgensen, K. et al. Don’t judge a salmon by its spots: environmental variation is the primary determinant of spot patterns in Salmo salar. Bmc Evolutionary Biology In press (2018). [DOI] [PMC free article] [PubMed]
- 39.Harvey AC, et al. Plasticity in growth of farmed and wild Atlantic salmon: is the increased growth rate of farmed salmon caused by evolutionary adaptations to the commercial diet? Bmc Evolutionary Biology. 2016;16:13. doi: 10.1186/s12862-016-0841-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Houde ALS, Fraser DJ, Hutchings JA. Reduced anti-predator responses in multi-generational hybrids of farmed and wild Atlantic salmon (Salmo salar L.) Conservation Genetics. 2010;11:785–794. doi: 10.1007/s10592-009-9892-2. [DOI] [Google Scholar]
- 41.Fleming IA, Einum S. Experimental tests of genetic divergence of farmed from wild Atlantic salmon due to domestication. Ices Journal of Marine Science. 1997;54:1051–1063. [Google Scholar]
- 42.Harvey A, et al. Does density influence relative growth performance of farmed, wild, and F1 hybrid Atlantic salmon in semi-natural and hatchery common garden conditions? Royal Society Open Science. 2016;3:160152. doi: 10.1098/rsos.160152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sundt-Hansen L, et al. Growth enhanced brown trout show increased movement activity in the wild. Funct. Ecol. 2009;23:551–558. doi: 10.1111/j.1365-2435.2008.01532.x. [DOI] [Google Scholar]
- 44.Skilbrei OT, Heino M, Svåsand T. Using simulated escape events to assess the annual numbers and destinies of escaped farmed Atlantic salmon of different life stages, from farms sites in Norway. Ices Journal of Marine Science. 2015;72:670–685. doi: 10.1093/icesjms/fsu133. [DOI] [Google Scholar]
- 45.Glover KA, et al. Atlantic salmon populations invaded by farmed escapees: quantifying genetic introgression with a Bayesian approach and SNPs. BMC Genet. 2013;14:4. doi: 10.1186/1471-2156-14-74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Karlsson S, Diserud OH, Fiske P, Hindar K. Widespread genetic introgression of escaped farmed Atlantic salmon in wild salmon populations. Ices Journal of Marine Science. 2016;73:2488–2498. doi: 10.1093/icesjms/fsw121. [DOI] [Google Scholar]
- 47.Skaala O, Wennevik V, Glover KA. Evidence of temporal genetic change in wild Atlantic salmon, Salmo salar L., populations affected by farm escapees. Ices Journal of Marine Science. 2006;63:1224–1233. doi: 10.1016/j.icesjms.2006.04.005. [DOI] [Google Scholar]
- 48.Glover KA, et al. Three decades of farmed escapees in the wild: A spatio-temporal analysis of population genetic structure throughout Norway. Plos One. 2012;7(8):e43129. doi: 10.1371/journal.pone.0043129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Clifford SL, McGinnity P, Ferguson A. Genetic changes in Atlantic salmon (Salmo salar) populations of northwest Irish rivers resulting from escapes of adult farm salmon. Canadian Journal of Fisheries and Aquatic Sciences. 1998;55:358–363. doi: 10.1139/f97-229. [DOI] [Google Scholar]
- 50.Crozier WW. Evidence of genetic interaction between escaped farmed salmon and wild Atlantic salmon (Salmo salar L) in a Northern Irish river. Aquaculture. 1993;113:19–29. doi: 10.1016/0044-8486(93)90337-X. [DOI] [Google Scholar]
- 51.Verspoor E, Knox D, Marshall S. Assessment of interbreeding and introgression of farm genes into a small Scottish Atlantic salmon Salmo salar stock: ad hoc samples–ad hoc results? Journal of Fish Biology. 2016;89:2680–2696. doi: 10.1111/jfb.13173. [DOI] [PubMed] [Google Scholar]
- 52.Bourret V, O’Reilly PT, Carr JW, Berg PR, Bernatchez L. Temporal change in genetic integrity suggests loss of local adaptation in a wild Atlantic salmon (Salmo salar) population following introgression by farmed escapees. Heredity. 2011;106:500–510. doi: 10.1038/hdy.2010.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Keyser, F. et al. Predicting the impacts of escaped farmed Atlantic salmon on wild salmon populations. Canadian Journal of Fisheries and Aquatic Sciences 1–7, 10.1139/cjfas-2017-0386 (2018).
- 54.Bolstad GH, et al. Gene flow from domesticated escapees alters the life history of wild Atlantic salmon. Nature Ecology & Evolution. 2017;1:0124. doi: 10.1038/s41559-017-0124. [DOI] [PubMed] [Google Scholar]
- 55.Castellani M, et al. Modelling fitness changes in wild Atlantic salmon populations faced by spawning intrusion of domesticated escapees. Evolutionary Applications, Early online. 2018 doi: 10.1111/eva.12615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lund RA, Hansen LP. Identification of wild and reared Atlantic salmon, Salmo salaar L., using scale characters. Aquaculture and Fisheries Management. 1991;22:499–508. [Google Scholar]
- 57.Skaala O, Glover KA, Barlaup BT, Borgstrom R. Microsatellite DNA used for parentage identification of partly digested Atlantic salmon (Salmo salar) juveniles through non-destructive diet sampling in salmonids. Marine Biology Research. 2014;10:323–328. doi: 10.1080/17451000.2013.810757. [DOI] [Google Scholar]
- 58.Taggart JB. FAP: an exclusion-based parental assignment program with enhanced predictive functions. Mol. Ecol. Notes. 2007;7:412–415. doi: 10.1111/j.1471-8286.2006.01616.x. [DOI] [Google Scholar]
- 59.Solberg MF, Zhang Z, Glover KA. Are farmed salmon more prone to risk than wild salmon? Susceptibility of juvenile farm, hybrid and wild Atlantic salmon Salmo salar L. to an artificial predator. Applied Animal Behaviour Science. 2015;162:67–80. doi: 10.1016/j.applanim.2014.11.012. [DOI] [Google Scholar]
- 60.Harvey AC, Tang YK, Wennevik V, Skaala O, Glover KA. Timing is everything: Fishing-season placement may represent the most important angling-induced evolutionary pressure on Atlantic salmon populations. Ecol. Evol. 2017;7:7490–7502. doi: 10.1002/ece3.3304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.R Development Core Team. R: A Language and Environment for Statistical Computing. Vienna, Asutria, [http://www.R-project.org/] (2014).
- 62.Bates D, Machler M, Bolker BM, Walker SC. Fitting Linear Mixed-Effects Models Using lme4. Journal of Statistical Software. 2015;67:1–48. doi: 10.18637/jss.v067.i01. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.