Abstract
A common feature of solid tumors, including glioblastoma multiforme (GBM), is mitochondrial dysfunction. However, it is reported that the current standard of anti-GBM therapies may potentiate mitochondrial damage and, in effect, support the aggressive character of cancer. As mitochondria are implicated in the modulation of cellular drug sensitivity and chemoresistance mechanisms, activation-stressed mitochondria in GBM cells may represent a new target for anti-GBM therapy that is nontoxic for normal cells.
Methods:
As mitochondria are possible targets for antidepressant drugs used as adjuvant therapy in patients with GBM, we examined their influence on mitochondrial volume and activity, reactive oxygen species level, extracellular lactate concentration, and p65 NF-κB gene expression in GBM cells.
Results:
Our investigation showed, for the first time, that tricyclic antidepressants, imipramine and amitriptyline, partially reverse GBM abnormalities.
Conclusion:
In the light of reported studies, the mitochondrial disturbance observed in glioma cells is a dynamic process that can be reversed or silenced. Moreover, imipramine and amitriptyline are attractive cellular metabolic modulators and can potentially be used to restoring a proper function of mitochondria in GBM cells.
Keywords: mitochondrial disturbances in GBM cells, antidepressant drugs, adjuvants in cancer treatment, metabolic therapy
Introduction
Although glioblastoma multiforme (GBM) is one of the best molecularly characterized human tumors, this knowledge does not significantly promote a prolonged life span in patients with GBM. Therefore, it is necessary to search for new targets for anti-GBM therapies.
A promising link in a chain cancer transformation is the mitochondria. Recent studies focus on mitochondrial involvement in GBM malignancy as well as chemoresistance mechanisms. However, this complex cellular machinery does not always work perfectly. Low respiring “dormant” and “stressed out” mitochondria are observed in cancer cells that differ structurally and functionally from mitochondria in healthy cells.1,2 Therefore, proponents of the metabolic theory suggest the use of metabolic activators during cancer therapy to restore proper mitochondrial function as executioners of cell death and gatekeepers of chemotherapy response.3,4 This idea, however, is not widely accepted and requires further studies.
Antineoplastic drugs used in systemic chemotherapy accumulate in the mitochondria of all fast-proliferating cells and generate mitochondrial reactive oxygen species, which cause mitochondrial dysfunction, DNA damage, and genome instability. On account of the nonselective drug action, a blockade of mitochondrial ribosome synthesis and arrest of RNA transfer necessary for the regenerative processes of cellular organelle is induced in noncancerous cells, leading to their malfunction.5,6 Clinically, cytotoxicity induced by chemotherapy is manifested in multiorgan damage, epilepsy, fatigue, neuropathic pain, chemo brain syndrome, cachexia, and depression, which, when left untreated, complicate the cancer treatment process, deteriorate the quality of the patients’ life, and reduce their survival rate.7,8 In patients with glioma, cytostatics induce chemoresistance mechanisms in glioma cells and generate irreversible changes in the mitochondria of normal cells that are responsible for many cytotoxic/neurological and psychological side effects.9 Therefore, implementation of supportive therapy during a basic antiglioma treatment is always required.
Due to the fact that advanced cancers, especially GBM, often coexist with depression, antidepressant drugs are widely used in these patients as an adjuvant treatment.10 Experimental in vivo and in vitro studies, however, have shown the equivocal effects of antidepressants on cancer prognosis, metastasis, chemotherapy response, cultured tumor cells, cell cycle, apoptosis, and intracellular signaling.11,12 Since antidepressants have been used at various concentrations and in different models in various studies, their results generate an informative dissonance that does not allow the drawing of uniform conclusions about the real effect of antidepressants on the carcinogenic processes of GBM.
GBM is a heterogeneous cell composition residing in an unstable microenvironment in which oxygen concentrations change dynamically, fluctuating from 0% to 5%.13 Besides genetic background, this specific microenvironment can determine its malignancy and is responsible for interindividual differences observed in clinical studies. Therefore, studies conducted in only 1 oxygen model, especially in standard laboratory conditions (20% oxygen), do not reflect the complex character of GBM nor do they create real microenvironments that can be used to progress our knowledge concerning intratumor processes.14 Moreover, such studies do not include palisade cells that migrate from lower to higher oxygenated regions in the tumor that are responsible for GBM micrometastasis.
Our studies were conducted in 2.5% oxygen (physiologic conditions for GBM) and for comparison in a standard laboratory conditions to investigate, for the first time, the involvement of mitochondrial activity in the mechanisms of antidepressant drugs of different classes, namely15:
Imipramine—a tricyclic antidepressant (TCA) used as a drug of second choice in the treatment of major depressive disorders, neuropathic pain, and frequently applied as the referent antidepressant in experimental studies.
Amitriptyline—a TCA used in the treatment of major depressive disorders, anxiety disorders, migraine prevention, neuropathic pain, and posttherapeutic neuralgia.
Fluoxetine—a selective serotonin reuptake inhibitor (SSRI) applied in the treatment of major depressive disorders, obsessive-compulsive disorders, bulimia nervosa, and panic disorders.
Mirtazapine—an atypical antidepressant applied as an anxiolytic, hypnotic, and antiemetic drug as well as an appetite stimulant.
Escitalopram—an SSRI, recommended for depressive and anxiety disorders, such as general anxiety disorders, social anxiety and panic disorders, and obsessive-compulsive disorders.
Agomelatine—a melatonergic antidepressant that restores balance in circadian rhythm disorders.
Materials and Methods
Cell Culture
The human glioblastoma multiforme cell line, T98G (Sigma-Aldrich, St. Louis, Missouri), was utilized for this study. Cell culture media, gentamicin, and fetal bovine serum were purchased from Gibco-BRL (Waltham, Massachusetts). Antidepressant drugs (imipramine, amitriptyline, fluoxetine, escitalopram, agomelatine, and mirtazapine) were obtained from Sigma-Aldrich (Saint Louis, Missouri). Plastics for cell cultures (monolayer and spheres) were purchased from Nunc (Waltham, Massachusetts), Falcon (Lexington, Tennessee), and Eppendorf (Hamburg, Germany).
Study Design and Experimental Oxygen Models
All oxygen models applied in our study and cell density evaluation were described previously.16,17 After achieving 90% confluence, cultures were trypsinized and passaged. On the second day following trypsinization, the medium was replenished with a fresh portion containing antidepressant drugs at a 10 µmol/L concentration (imipramine, amitriptyline, fluoxetine, mirtazapine, escitalopram, or agomelatine). Cultures were placed in an incubator for 24 hours in one of the following oxygen conditions: an environment containing 2.5% or an environment containing 20% oxygen.
All our experiments presented in this article were conducted in 2 types of CO2 incubators: (1) CO2 incubator NuAir (Fernbrook Lane, Centerville, Massachusetts) for experiments conducted in a standard laboratory conditions (5% CO2, 20% oxygen, and 97% humidity) and (2) New Brunswick Galaxy 48R for experiments conducted in 2.5%; this incubator has a regulation for oxygen level (0.1% to 19%) and for 2.5% of oxygen it is 5% CO2 and 94% N2 (gas pressure is 0.35 bar/5 psi).
Microscopic Analyses
Glioma cells were cultured on coverslips, and after reaching confluence, they were incubated with 250 nmol/L MitoTracker Red (ThermoFisher Scientific [Thermofisher, Waltham, Massachusetts]) (1 mM of initial concentration stock solution) for 20 minutes, followed by washing in phosphate-buffered saline (PBS) and fixation. This red fluorescent dye stains mitochondria in living cells, as cellular accumulation depends upon membrane potential. The morphology of GBM (T98G cell line) exposed to antidepressant drugs in 2 oxygen models (2.5% and 20% oxygen) and mitochondrial localization were observed using a fluorescent microscope (FV10i, Olympus) and the Imaris program (8.2.1). For the purposes of visualization, we merged pictures with the fluorescent and contrast-phase filters. No convolution was carried out in the pictures.
Mitochondrial volume
Photos presented in the article were made in the fluorescence and contrast-phase channel using Confocal Fluoview 10 microscope Nikon (Minato,Tokyo, Japan). The microscope software allows taking pictures in the Z axis, scanning the entire thickness of the selected field (it gave several photos of a single area). On this basis, we obtained reconstruction of 3D cell culture space, and we were able to know the actual thickness of the studied area. Using Imaris (Bitplane 8.2.1) software, we analyzed images by removing noise and applying frames to calculate the volume of mitochondria.
Determination of ROS Generation
Quantitative measurements of intracellular reactive oxygen species (ROS) were conducted on T98G monolayer cultured cells exposed to antidepressant drugs in 2 oxygen conditions: 2.5% and 20% oxygen. The oxidative stress kit (Muse, Millipore, Germany), which determines the count and percentage of ROS negative (−) and ROS positive (+) cells, was used. The main component of this assay, dihydroethidium, was applied to detect ROS in cellular populations.
Measurement of Extracellular Lactate Levels
Lactate is a prognostic factor in various tumors, and its formation is related to tumor aggressiveness, invasiveness, and poor clinical outcomes.18 Lactate release into the culture medium was determined by fluorimetric assay (Cayman Chemicals, Ann Arbor, Michigan) according to the manufacturer’s protocol. Samples derived from different monolayer cultures of T98G cells tested in 4 different oxygen models and exposed to antidepressant drugs were collected and prepared according to the manufacturer’s manual. Fluorescence was measured using Fluoroscan Ascent FL (Labsystems, Helsinki, Finland) at the excitation wavelength of 530 to 540 nm and the emission wavelength of 585 to 595 nm.
Detection of p65 NF-κB Gene Expression
Nuclear factor κB plays a pivotal role in tumorigenesis and tumor progression, and its activation has been detected in glioma cells. In cancer, inhibition of NF-κB overactivation is an attractive target for future treatments due to its functional influence on more than 500 genes important for cellular processes. Experimental studies in recent years indicate that overexpression of RelA/p65 subunit of NF-κB contributes to tumor survival, which is a hallmark of inflammation and confers a negative prognosis.19
RNA Extraction, Reverse Transcription, and Real-Time Quantitative Polymerase Chain Reaction
T98G cells were cultured in a monolayer in 60-mm Petri dishes in different oxygen conditions as described earlier. After incubation, the medium was removed, and the culture plates were washed with cold PBS. Next, the cells were scraped, vortexed, and collected in sterile Eppendorf tubes using TRI Reagent (Molecular Research Centre, Cincinnati, Ohio). All ribonucleic acids were extracted from cell cultures using TRI Reagent according to the 1-step extraction method described by Chomczyński et al.20 RNA precipitates were finally dissolved in 100 µL of nuclease-free water and were quantified spectrophotometrically at wavelengths of 260 nm and 280 nm. Next, 1 µg of total RNA was reverse transcribed into complementary DNA using the GoScript Reverse Transcription System (Promega GmbH, Mannheim, Germany) in a total volume of 20 μL per reaction. The thermal profile included 22°C for 5 minutes, 42°C for 15 minutes, and 85°C for 5 minutes. Finally, the reverse transcription reaction mixture was diluted 1:4 with RNAse-free water.
Quantitative analysis of human p65 NF-κB gene expression in T98G cells was carried out by a real-time quantitative polymerase chain reaction (qPCR) assay based on SYBR Green I chemistry (Life Technologies, Warsaw, Poland). An amount of 2 μL of reverse transcription reaction mixture (ie, an equivalent of 50 ng of total RNA used for the reverse transcription) was used as a template for the reaction. Real-time PCR was performed using GoTaq qPCR Master Mix (Promega GmbH, Mannheim, Germany). The reaction mixture contained 10 μL of ×2 qPCR Master Mix in a total volume of 20 μL and 200 nmol/L of each forward and reverse primer. Primer sequences were derived from a real-time PCR primer database (https://primerdepot.nci.nih.gov): p65 NF-κB_F: TTGCTGGTCCCACATAGTTG, NF-κB_R: ATGTATGTGAAGGCCCATCC giving a product of 105 base pairs in length. Primer sequences for a reference human β-actin (ACTB) gene were derived from PrimerDepot database (ACTB_F: TCATGAAGTGTGACGTGGACATC, ACTB_R: CAGGAGGAGCAATGATCTTGATCT, product length: 109 bp). The relative quantification experiment type was chosen for the interpretation of qPCR analysis. Quantitative PCR was performed on a Roche LightCycler 480 Real-Time PCR system (Roche Diagnostics Polska, Warsaw, Poland). The reaction’s thermal profile was as follows: 95°C for 3 minutes, 40 cycles of 94°C for 30 seconds, and 58°C for 1 minute. Thereafter, a melting-curve analysis was conducted. The specificity of each reaction was confirmed by both melting-curve analysis and agarose gel electrophoresis (Agagel Mini, Biometra, Germany). The amount of p65 NF-κB in a sample, normalized to an endogenous reference and relative to a calibrator, was calculated by the 2−ΔΔCt method.
Statistical Analyses
Statistical analysis was performed using a 1-way analysis of variance followed by the post hoc Tukey test. Differences were considered statistically significant when P < .05. The results are presented as the standard error of the mean. Statistical analysis was performed using GraphPad Prism 7.01 software system (GraphPad Software Inc, San Diego, California).
Results
The Influence of Antidepressant Drugs on Cell Morphology, Mitochondrial Volume, and Localization in T98G Cells Maintained in 2.5% and 20% Oxygen (Standard Laboratory Conditions)
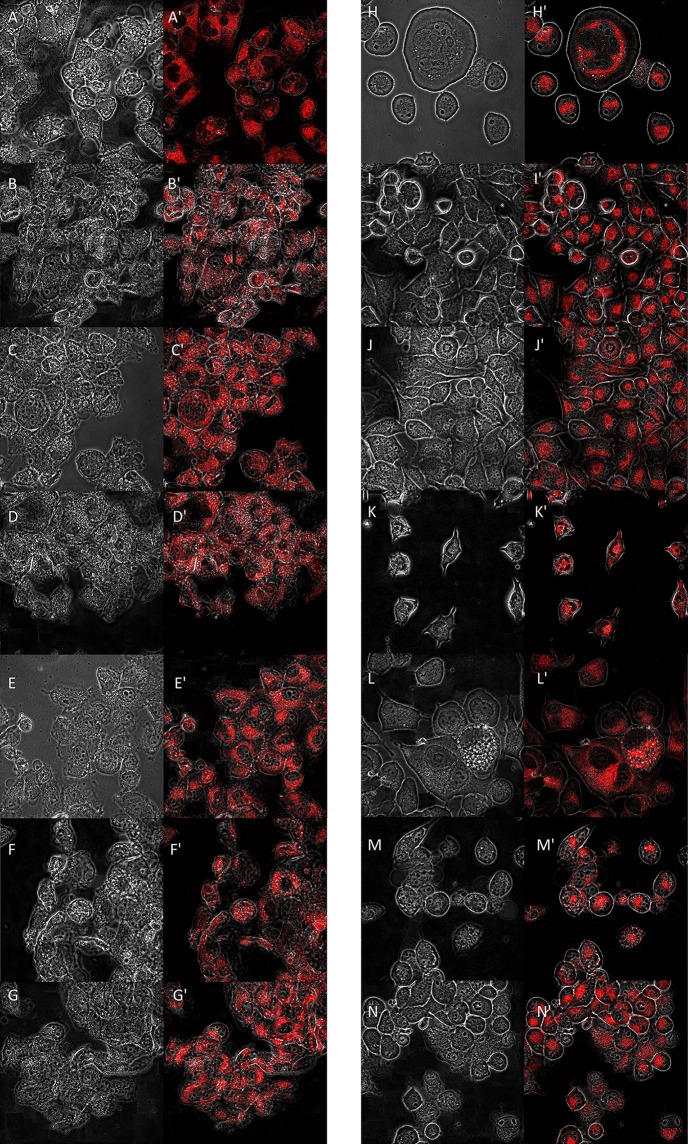
In both oxygen models, GBM cells were characterized with Regard to shape, size (morphological polymorphism), mitochondrial dynamics, and localization. In the 2.5% oxygen model (physiologic conditions for GBM), we observed very strong mitotracker signals in the untreated cells (control group), potentially caused by the loss of mitochondrial inner membrane potential (a fusion phenomenon) that is responsible for respiratory chain defects. In a control group of untreated cells, it was difficult to distinguish between single mitochondria because they formed a large network (Figure 1A and A’). The Imaris program analysis showed that untreated cells presented the typical biochemical form of neoplastic cells. In these cells, lipid droplets were more visible, suggesting higher secretion of inflammatory mediators. In the control cells, the volume of the mitochondria was 0.7 µm3. The antidepressants decreased this mitochondrial parameter, and the most significant effect was induced by imipramine (by approximately 30%), escitalopram (by 27%), and amitriptyline (by 23%). Following exposure to antidepressants, fewer lipid droplets and a weaker mitochondrial signal were detected in GBM cells compared to control cells (Figure 1B-G’). In this oxygen model, we observed anterior localization of the mitochondria.
Figure 1.
Effect of antidepressant drugs (imipramine (B), amitriptyline (C), fluoxetine (D), mirtazapine (E), escitalopram (F), agomelatine (G) (10 µmol/L)) on mitochondrial localization/morphology of glioma T98G cells cultured in 2.5% oxygen conditions; A, control glioma cells not exposed to antidepressant drugs. Cells were observed using the fluorescent microscope (FV10i, Olympus). Analyses were conducted using the Imaris program (8.2.1) after the incubation of cells with MitoTracker (ThermoFisher Scientific) (denoted by ’). The left panel presents pictures in a contrast-phase microscope. Magnification ×40. Effect of antidepressant drugs (imipramine (I), amitriptyline (J), Fluoxetine (K), mirtazapine (L), escitalopram (M), agomelatine (N) (10 µmol/L)) on mitochondrial localization/morphology of glioma T98G cells cultured in 20% oxygen conditions (standard laboratory conditions); H, control glioma cells not exposed to antidepressant drugs. Cells were observed using the fluorescent microscope (FV10i, Olympus). Analyses were conducted using the Imaris program (8.2.1) after the incubation of cells with mitotracker (ThermoFisher Scientific) (denoted by ’). The left panel presents pictures in a contrast-phase microscope. Magnification ×40.
In standard laboratory conditions (20% oxygen), the mitochondrial volume in all experimental groups was significantly smaller, particularly in the control group, where it was 0.44 µm3 (Figure 2), and mitochondria were localized around nucleus. The effect of antidepressant drugs on this parameter, however, was contrary to that in cells maintained in 2.5% oxygen. In cells exposed to antidepressants, we observed an increased mitochondrial volume of up to 22% (mirtazapine) versus the control cells. Lipid droplet aggregation and mitochondrial fusion were not clearly visible (Figure 1A-N).
Figure 2.
Effect of antidepressants (10 µmol/L) on mitochondrial volume of glioma T98G cells cultured in 2.5% oxygen and standard laboratory conditions (20% oxygen). Data were analyzed by Imaris program (8.2.1). *P < .05 compared with the control.
The Influence of Antidepressant Drugs on ROS Generation in T98G Cells Maintained in 2.5% and 20% Oxygen Conditions
All studied antidepressants shifted the ratio ROS (+) to ROS (−) toward ROS (−) in GBM cultures maintained in an atmosphere containing 2.5% oxygen, frequently detected in the intratumor space in vivo. Amitriptyline induced the strongest effect. In the culture exposed to amitriptyline, the ratio of ROS (+) to ROS (−) was 20% to 80% (in the control group, this was 60% to 40%). In standard laboratory conditions (20% oxygen), the ratio of ROS (+) to ROS (−) was approximately 45% to 55% in the cultures exposed to antidepressants. In this model, fluoxetine, mirtazapine, escitalopram, and agomelatine increased ROS (+) levels in comparison to the control cultures (Figure 3A, B).
Figure 3.
(A and B) Effect of antidepressants (10 µmol/L) on generation of reactive oxygen species (ROS, ROS negative (−) and ROS positive (+) cells) in glioma T98G cells cultured in 2.5% oxygen and standard laboratory conditions (20% oxygen). Data are presented as means ± standard error of the mean of triplicate samples (P = 3). *P < .05 compared to untreated cells.
l-Lactate Release by T98G Cells in Response to Antidepressants in Several Oxygen Conditions
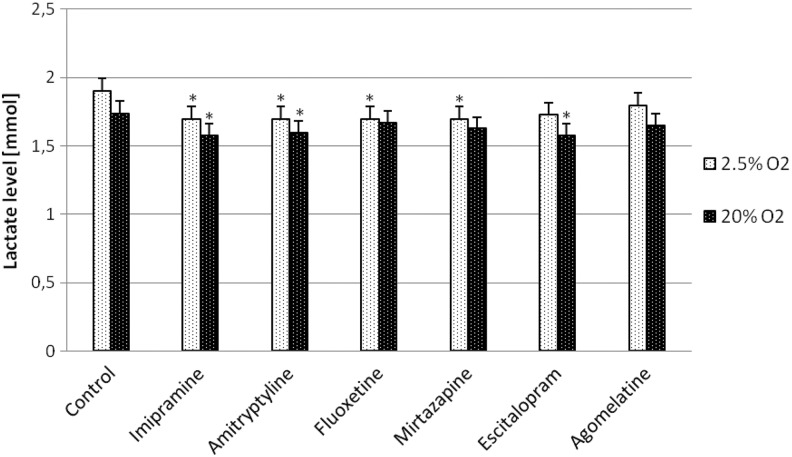
The highest concentration of l-lactate (1.75 and 1.94 mmol/L) was detected in the media harvested from untreated T98G cultures in a both oxygen conditions. Compared to the control groups, imipramine, amitriptyline, fluoxetine, mirtazapine, and escitalopram inhibited lactate release into the culture medium; hovewer, in the hypoxia model, the effect of antidepressants was slightly stronger than in a standard laboratory conditions (2.5% oxygen model: approximately, 18% vs control; 20% oxygen approximately 14% vs control; Figure 4).
Figure 4.
Effect of antidepressants (10 µmol/L) on extracellular level of lactate released from glioma T98G cells cultured in 2.5% oxygen and standard laboratory conditions (20% oxygen). Data points are means ± standard error of the mean of triplicate samples (n = 3); statistical analysis was performed using Student’s t test by comparing antidepressants group versus control (untreated cells). *P < .05 compared with the control (basal) lactate releasing.
p65 NF-κB Expression in T98G Cells Following Their Exposure to Antidepressants Under Several Oxygen Conditions
Following exposure of T98G cells to antidepressant drugs, the inhibition of p65 NF-κB gene expression was observed in all oxygen models, albeit with varying intensity. In the 2.5% oxygen model, the strongest effects were induced by imipramine (41%), amitriptyline (33%), and fluoxetine (14%). These drugs also reduced this parameter in standard laboratory conditions by 31%, 19%, and 7%, respectively. Mirtazapine, escitalopram, and agomelatine did not have a significant impact on p65 NF-κB gene expression in any oxygen condition (Figure 5).
Figure 5.
Effect of antidepressants (10 µmol/L) on p65 nuclear factor κB (NF-κB) gene expression in glioma T98G cells cultured in 2.5% oxygen and standard laboratory conditions (20% oxygen). Data are represented as means ± standard error of the mean of triplicate samples (P = 3). *P < .05 compared with control (100%).
Discussion
Several recent studies have indicated that antidepressant drugs, utilized for years in GBM patients to minimize the side effects of chemotherapy, depression, or neurologic deficits, may contribute to additional anti-GBM effects through their influence on mitochondrial functions.21 However, there is insufficient data describing the role of mitochondria in GBM malignancy and mitochondrial involvement in the mechanism of action of drugs that support anticancer therapy. As mitochondrial disturbances occur in parallel with changes in their morphology, and altered brain energy metabolism has been associated with depression in GBM patients, we undertook a novel investigation of the influence of 6 antidepressants (imipramine, amitriptyline, fluoxetine, escitalopram, mirtazapine, and agomelatine) on select aspects of mitochondrial machinery (volume, morphology, ROS (+) ROS (−) production, lactate release, and p65 NF-κB gene expression).
The present study was performed under 2 different oxygen conditions, as oxygen states critically effect cancer experiments, by employing a 2.5% physiologic oxygen model, which mimicked the tumor microenvironment in vivo as well as standard laboratory conditions (20% oxygen) for comparison. However, the latter condition cannot be defined as “normoxia” because in vivo oxygen level is 10% to 13% (in the brain for normal, noncancerous tissues).22 Thus, in effect, both oxygen models created stress conditions.
Hypoxia in untreated GBM cells resulted in mitochondrial swelling and increased mitochondria volumes compared to standard laboratory conditions. In comparison to untreated cells, GBM cells exposed to antidepressants (particularly imipramine and amitriptyline) in a 2.5% physiologic oxygen model were characterized by smaller cristolysis and mitochondrial volume/fusion as well as centralization of the mitochondria. In contrast, these effects were not observed in cells cultured (under standard laboratory conditions) in the 20% oxygen model. The differing effects of antidepressant drugs on mitochondrial volume and ROS production due to atmospheric oxygen concentration has a strong influence on mitochondrial activity and morphology.
Various studies over the last year have highlighted the impact of hypoxia on mitochondria and mitochondrial metabolism, including changes in ROS production and signaling.23 Under physiological conditions (resting phase), the mitochondrial matrix is expanded with a narrow average intermembrane distance between the inner and outer mitochondrial membranes.24 During ischemia, mitochondrial K+ uptake decreases, resulting in an imbalance between K+ influx and efflux that causes contraction of the mitochondrial matrix. In consequence, a broader intermembrane space is formed, increasing distances between enzymes necessary for cellular function.25
Although changes in mitochondrial volume have been associated with alterations in their biological functions, as well as being a sign of mitochondrial injury or response to stress, mechanisms of mitochondrial volume regulation are still not well understood. Our results suggest that antidepressants, especially imipramine and amitriptyline (as in the case of neuroinflammatory processes in depression, stroke, or mood disorders), may have the ability to restore energy balance and regulate mitochondrial function. Our research also confirms reports by other authors26 of a role for mitochondria in effects of antidepressant drug actions and sheds new light on the influence of these drugs in glioma “mitochondriopathy.” Tricyclic antidepressants can likely modulate mitochondrial function by opening different subtypes of K(+) channels (voltage-gated, K(ATP), and Ca(2+)-gated), blocking sodium channels, inhibiting respiratory complex, and enhancing ATP synthase activity or ROS activity.27
In malignant tumors, including GBM, oxidative stress and ROS act as intracellular messengers and are implicated in the impairment of cellular repair mechanisms, DNA disturbances, genomic instability, and an ineffective execution of apoptosis. When ROS levels exceed the antioxidant defense system, oxidative stress occurs, inducing harmful processes and effects such as disturbed cellular redox, DNA damage, lipid peroxidation, protein oxidation, and enzyme inactivation, ultimately leading to tumorigenesis.28 Therefore, the manipulation of ROS levels may be a target of interest for antiglioma treatment. In our study, we observed that all antidepressants in question, particularly imipramine, amitriptyline, fluoxetine, and mirtazapine, shifted the ratio of ROS (+)–ROS (−) toward ROS (− in cells maintained in a 2.5% physiology oxygen model compared to control cells. Under standard laboratory conditions, however, this effect was weaker and only observed following exposure of glioma cells to imipramine or amitriptyline. The other antidepressants induced an opposite effect, namely, an increase in ROS (+) level compared to controls. This phenomenon may be a result of an adaptation to output ROS levels and a blockade of proapoptotic pathways in cancer cells. For this reason, in many cancer models, strong oxidative stress is required for activation of apoptosis.29 In our experiments, imipramine and amitriptyline shifted the balance between ROS (+) and ROS (−) toward ROS (−), which may be indicative of their protective effects toward normal cells. In a previous study,28 we showed that these drugs had the potential to increase the viability of normal human astrocytes, which could protect the cells from the cytotoxic effects of anti-GBM drugs. Moreover, results from other studies have shown that long-term use of tricyclic antidepressants may have a protective effect against glioma.29 Therefore, we propose that pro-oxidative treatment is not the only effective method available for inducing cancer cell death. Typically, in cancer cells, ROS levels are high but in chemoresistant tumors, low ROS levels have been detected.30 The reported antioxidative effects of antidepressants may be related to their immunomodulatory and anti-inflammatory properties through the modulation of p65 NF-κB gene expression.
Due to strong activation of NF-κB factor in various tumors, and the fact that it is related to cancer development/invasion/progression and inflammatory processes, suppression of NF-κB activity could be an anticancer therapeutic strategy or an important element of adjuvant treatment.31,32 Among the drugs studied, only imipramine and amitriptyline induced strong inhibition of p65 NF-κB gene expression. It is possible that this antidepressant effect may be helpful in maintaining a balance between anti- and proinflammatory mediators. The latter is secreted in excess by microglia cells and may disturb proper cellular immune response, leading to micrometastasis and, finally, tumor progression.33,34 The suppressive effect of imipramine and amitriptyline on p65 NF-κB expression supports their use for the relief of chronic/neuropathic pain or depressive disorders35 in patients with GBM, as chronic inflammation and endocrine mechanisms partly underlie depression and pain pathomechanisms.36
Lactate, an active oncometabolite in cancer cells, is, apart NF-κB, a connecting link between inflammation, immunity, and cancerogenesis. At enhanced concentrations, lactate, as an inflammatory agent, promotes the local invasion of the tumor as a result of tumor niche acidification, inhibition of T-cell motility, and stimulation of proinflammatory cytokine release.37,38 Our study is the first to observe that antidepressants, especially imipramine and amitriptyline, inhibited lactate release from GBM cells to the culture medium under hypoxia conditions. This observation suggests a hitherto unreported mechanism of action of tricyclic antidepressant drugs, likely due to their inhibitory influence on glycolytic pathways and glucose metabolism, whereby suppression is the potential metabolic target of treatment. However, this interesting phenomenon requires further study.
Among the antidepressants studied in this investigation, the tricyclic drugs imipramine and amitriptyline exerted a stronger effect on the analyzed mitochondrial processes than the others. Until now, however, the influence of imipramine and amitriptyline on mitochondrial activity has been largely unknown. Based on previous experimental studies, one might assume that their effect would be concentration dependent. At lower concentrations (up to 10 µmol/L), they have a protective effect on respiratory chain complexes but at higher concentrations (above 20 µmol/L), they inhibit mitochondrial activity.39 Although the precise antidepressant concentrations in the human brain during treatment is unknown, the results of experimental pharmacokinetic studies indicate the brain concentration of imipramine and amitriptyline is about 20 to 30 times higher than their plasma levels. Since the therapeutic levels of plasma tricyclic antidepressants used in the treatment of depression are estimated to range between 0.3 and 1 µmol/L,40,41 and as coanalgesic amitriptyline, for the treatment of neuropathic pain, is prescribed at lower doses than for the treatment of depression, our study used imipramine and amitriptyline at a concentration of 10 µmol/L to mimic in vivo conditions. Tricyclic drugs in this concentration range have the potential to act on selected parameters of mitochondrial activity.
Our observations corroborate results from other studies investigating the underlying mechanism of action of antidepressant drugs as an element of polypharmacotherapy for GBM. Considering mitochondria have largely unexplored therapeutic potential and are an attractive target in metabolic therapy, the modulatory effect of antidepressants on restoring proper mitochondrial function should be further investigated. Epidemiological studies are required to prove the clinical significance of antidepressants in the treatment of patients with glioma. A meta-analysis of clinical data regarding survival, quality of life, and relapse in patients with GBM treated with cytostatic drugs and antidepressants in comparison to the effects of monotherapy with cytostatics alone is warranted.
Acknowledgment
We gratefully thank to Nikon Company (Drs Wojcech Brutkowski and Maciej Stafiński) for lending a microscope and scientific support.
Author’s Note: This article does not contain any studies with human participants or animals performed by any of the authors. No human participants were used in this study. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant from the School of Medicine, Medical University of Silesia, Katowice, Poland (KNW-2-023/N/5/K).
References
- 1. Czarnecka AM, Czarnecki JS, Kukwa W, Capello F, Sicińska A, Kukwa A. Molecular oncology focus—is carcinogenesis a mitochondriopathy? J Biomed Sci. 2010;17:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Masui K, Cavenee WK, Mischel PS. Cancer metabolism as a central driving force of glioma pathogenesis. Brain Tumor Pathol. 2016;33(3):161–168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Cairns RA, Harris IS, Mak T. Regulation of cancer cell metabolism. Nat Rev Cancer. 2011;11(2):85–95. [DOI] [PubMed] [Google Scholar]
- 4. Deighton R, Bihan TL, Martin SF, et al. Interactions among mitochondrial proteins altered in glioblastoma. J neurooncol. 2014;118(2):247–256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Sarosiek KA, Chonghaile TN, Letai A. Mitochondria: gatekeepers of response to chemotherapy. Trends Cell Biol. 2013;23(12):612–619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Bhat TA, Kumar S, Chaudhary AK, Yadav N, Chandra D. Restoration of mitochondria function as a target for cancer therapy. Drug Discov Today. 2015;20(5):635–643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Conklin K, Nicolson GL. Molecular replacement in cancer therapy: reversing cancer metabolic and mitochondrial dysfunction, fatigue and the adverse effects of cancer therapy. Curr Cancer Ther Rev. 2008;4:1–11. [Google Scholar]
- 8. Hess JA, Khasawneth MK. Cancer metabolism and oxidative stress: insights into carcinogenesis and chemotherapy via the non-dihydrofolate reductase effects of methotrexate. BBA Clin. 2015;3:152–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Bae SH, Park MJ, Lee MM, et al. Toxicity profile of temozolomide in the treatment of 300 malignant glioma patients in Korea. J Korean Med Sci. 2014;29(7):980–984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Rooney AG, Brown PD, Reijneveld JC, Grant R. Depression in glioma: a primer for clinicians and researchers. J Neurol Neurosurg Psychiatry. 2014;85(2):230–235. [DOI] [PubMed] [Google Scholar]
- 11. Hroudova J, Fiskar Z. In vitro inhibition of mitochondrial respiratory rate by antidepressants. Toxicol Lett. 2012;213(3):345–352. [DOI] [PubMed] [Google Scholar]
- 12. Jeon SH, Kim SH, Kim YS, Lim Y, Lee YH, Shin SY. The tricyclic antidepressant imipramine induces autophagic cell death in U-87MG glioma cells. Biochem Biophys Res Commun. 2011;413(2):311–317. [DOI] [PubMed] [Google Scholar]
- 13. Sullivan M, Galea P, Latif S. What is the appropriate oxygen tension for in vitro culture? Mol Hum Reprod. 2006;12(11):653. [DOI] [PubMed] [Google Scholar]
- 14. Vaupel P. Tumor microenvironment physiology and its implications for radiation oncology. Semin Radiat Oncol. 2004;14(3):198–206. [DOI] [PubMed] [Google Scholar]
- 15. Brunton LL, Lazo JS, Parker KL. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 11th ed New York, NY: McGraw-Hill Medical Publishing Division; 2006. [Google Scholar]
- 16. Bielecka AM, Obuchowicz E. Chronic physiological hypoxia and high glucose concentration promote resistance of T98G glioblastoma cell line to temozolomide. Drug Des. 2014;3:117. [Google Scholar]
- 17. Bielecka AM, Obuchowicz E. Antidepressant drugs can modify cytotoxic action of temozolomide. Eur J Cancer Care. 2017;26(5):1–28. [DOI] [PubMed] [Google Scholar]
- 18. Bauman F, Leukel P, Doefelt A, et al. Lactate promotes glioma migration by TGF-beta2-dependent regulation of matrix metalloproteinase-2. Neuro Oncol. 2009;11(4):368–380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. McFarland BC, Hong SW, Rajbhandari R, et al. Nf-κB-induced IL-6 ensures STAT3 activation and tumor aggressiveness in glioblastoma. PloS One. 2013;8(11):e78728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Ribaudo R, Gilman M, Kingston RE, Chomczyński P, Sacchi N. Preparation of RNA from tissues and cells. Curr Protoc Neurosci. 2001; Appendix 1:Appendix 1I. [DOI] [PubMed] [Google Scholar]
- 21. Frick LR, Rapanelli M. Antidepressants: influence on cancer and immunity? Life Sci. 2013;92(10):525–532. [DOI] [PubMed] [Google Scholar]
- 22. Isa AY, Ward TH, West CM, Slevin NJ, Homer JJ. Hypoxia in head and neck cancer. Br J Radiol. 2006;79(946):791–798. [DOI] [PubMed] [Google Scholar]
- 23. Westermann B. Molecular machinery mitochondria fusion and fission. J Biol Chem. 2008;283(20):13501–13505. [DOI] [PubMed] [Google Scholar]
- 24. Morillo G. Electron microscopy morphology of the mitochondrial network in human cancer. Int J Biochem Cell Biol. 2009;41(10):2062–2068. [DOI] [PubMed] [Google Scholar]
- 25. Rinaldi M, Caffo M, Minutoli L, et al. ROS and brain gliomas: an overview of potential and innovative therapeutic strategies. Int J Mol Sci. 2016;17(6):pii E984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Pilkington GJ, Parker K, Murray SA. Approaches to mitochondrially mediated cancer therapy. Semin Cancer Biol. 2018;18(3):226–235. [DOI] [PubMed] [Google Scholar]
- 27. Bourin M, Frank C, Haskoet M. The role of sodium channels in the mechanism of action of antidepressants and mood stabilizers. Curr Drug Targets. 2009;10(11);1052–1060. [DOI] [PubMed] [Google Scholar]
- 28. Bielecka-Wajdman AM, Lesiak M, Ludyga T, Sieroń A, Obuchowicz E. Reversing glioma malignancy: a new look at the role of antidepressant drugs as adjuvant therapy for glioblastoma multiforme. Cancer Chemother Pharmacol. 2017;79(6):1249–1256. [DOI] [PubMed] [Google Scholar]
- 29. Pottegard A, Rodriguea LA, Rasmussen L, Damkier P, Fris S, Gaist D. Use of tricyclic antidepressants and risk of glioma: a nationwide case-control study. Br J Cancer. 2016;114(11):1265–1268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Singer R, Judkins J, Salomonis N, et al. Reactive oxygen species-mediated therapeutic response and resistance in glioblastoma. Cell Death Dis. 2015;6:e1601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Troib A, Azab AN. Effects of psychotropic drugs on nuclear factor kappa B. Eur Rev Med Pharmacol Sci. 2015;19(7):1198–1208. [PubMed] [Google Scholar]
- 32. Frick LR, Williams K, Pittenger C. Microglia dysregulation in psychiatric disease. Clin Dev Immun. 2013;2016:8606057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Schiepers OJ, Wischers M, Maes M. Cytokines and major depression. Prog Neuropsychopharmacol Biol Psychiatry. 2005;29(2):201–217. [DOI] [PubMed] [Google Scholar]
- 34. Jaracz J, Gattner K, Moczko J, Hauser J. Comparison of the effects of escitalopram and nortryptiline on painful symptoms in patients with major depression. Gen Hosp Psychiatry. 2015;37(1):36–30. [DOI] [PubMed] [Google Scholar]
- 35. Hwang J, Zheng LT, Ock J, et al. Inhibition of glial inflammatory activation and neurotoxicity by tricyclic antidepressants. Neuropharmacology. 2008;55(5):826–834. [DOI] [PubMed] [Google Scholar]
- 36. Guyon A, Massa F, Rovere C, Nahon JL. How cytokines can influence the brain: a role for chemokines? J Neuroimmunol. 2008;198(1-2):46–55. [DOI] [PubMed] [Google Scholar]
- 37. Haas R, Smith J, Ros VR, et al. Lactate regulates metabolic and pro-inflammatory circuits in control of T cell migration and effector functions. PloS Biol. 2015;13(7):e1002202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Hayashi K, Michiue H, Yamada H, et al. Fluvoxamine, an anti-depressant, inhibits glioblastoma invasion by disrupting actin polymerization. Sci Rep. 2015;6:23372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Steingart AB. Do antidepressants cause, promote, or inhibit cancers? J Clin Epidemiol. 1995;48(11):1407–1412. [DOI] [PubMed] [Google Scholar]
- 40. Glotzbach RK, Preskorn SH. Brain concentrations of tricyclic antidepressants: single-dose kinetics and relationship to plasma concentrations in chronically dosed rats. Psychopharmacology (Berl). 1982;78(1):125–127. [DOI] [PubMed] [Google Scholar]
- 41. Budziszewska B, Jaworska-Feil L, Tetich M, et al. Regulation of the human corticotropin-releasing-hormone gene promotor activity by antidepressant drugs in neuro 2A and At-20 cells. Neuropsychopharmacol. 2004;29(4):785–794. [DOI] [PubMed] [Google Scholar]