Abstract
Study Objective:
Obese parturients both greatly benefit from neuraxial techniques, and may represent a technical challenge to obstetric anesthesiologists. Several studies address the topic of obesity and neuraxial analgesia in general, but few offer well described definitions or rates of “difficulty” and “failure” of labor epidural analgesia. Providing those definitions, we hypothesized that increasing body mass index (BMI) is associated with negative outcomes in both categories and increased time needed for epidural placement.
Design:
Single center retrospective chart review.
Setting:
Labor and Delivery Unit of an inner city academic teaching hospital.
Patients:
2485 parturients, ASA status 2 to 4, receiving labor epidural analgesia for anticipated vaginal delivery.
Interventions:
none.
Measurements:
We reviewed quality assurance and anesthesia records over a 12-month period. “Failure” was defined as either inadequate analgesia or a positive test dose, requiring replacement, and/or when the anesthesia record stated they failed. “Difficulty” was defined as six or more needle redirections or a note indicating difficulty in the anesthesia record.
Main Results:
Overall epidural failure and difficulty rates were 4.3% and 3.0%, respectively. Patients with a BMI of 30 kg/m2 or higher had a higher chance of both failure and difficulty with two and almost three fold increases, respectively. Regression analysis indicated that failure was best predicted by BMI and less provider training while difficulty was best predicted by BMI. Additionally, increased BMI was associated with increased time of discovery of epidural catheter failure.
Conclusions:
Obesity is associated with increasing technical difficulty and failure of neuraxial analgesia for labor. Practitioners should consider allotting extra time for obese parturients in order to manage potential problems.
Keywords: BMI, Cesarean section, dural puncture, labor epidural, neuraxial, morbid obesity, inadequate analgesia, Quality Assurance
INTRODUCTION
Physicians worldwide are faced with increasing numbers of obese patients, including obese parturients.1 The global obesity pandemic brings unique difficulties to anesthesiologists on labor and delivery units.2,3 More than one third of U.S. adults were obese, as defined by the World Health Organization (WHO) as a Body Mass Index (BMI; in kg m−2) ≥ 30) in 2011–2012.4 Prevalence has remained stable between 2009–2010 and 2011–2012.5 More than one third of women are obese, more than half of pregnant women are overweight or obese,6,7 with obesity representing a risk factor for multiple co-morbidities including gestational diabetes, gestational hypertension, preeclampsia and even death.7,8,9 Obese parturients are also at greater risk for both instrumental and Caesarean delivery (CD)1–3,9 which emphasizes the need for successful neuraxial analgesia. Failed regional techniques may lead to general anesthesia which potentially includes difficult airway, aspiration pneumonitis and failed resuscitation from hemodynamic collapse.7,10,11
While earlier studies defined predictive factors or characteristics associated with difficulty or failure of neuraxial anesthesia, few have focused on the obese parturient, or precisely defined “difficulty” and “failure” or provided exact rates.12–16 Yet, obese patients may benefit most from regional anesthetic management. We hypothesized that increasing BMI would be associated with increased neuraxial analgesic failure and difficulty as well as time needed for epidural placement.
METHODS
Our Institutional Review Board approved this study, including a waiver of patient consent for this retrospective analysis. We reviewed quality assurance (QA) data and anesthesia records of all parturients receiving any form of anesthesia care over a 12-month period (January to December 2008) in one of the obstetric teaching hospitals of the Medical College of Wisconsin: Wheaton Franciscan Healthcare St. Joseph Hospital, Milwaukee, WI. There, neuraxial blocks are performed by residents under the supervision of an attending anesthesiologist. Only patients who received epidural analgesia for labor pain and planned vaginal delivery were studied, including those who eventually had to undergo an unplanned CD. Planned CDs were not included in this particular study.
Data collected included age, height, weight, co-morbidities, anesthetic technique, indications of difficulty, indications of failure, epidural placement duration, time between placement and failure decision, and training level of the anesthesiology resident. Specifically, indications of “difficulty” included the number of lumbar interspaces attempted, greater than six epidural needle redirections or a note in the QA form and/or the anesthesia record. Indications of “failure” included the number of clinician boluses of local anesthetic to the epidural catheter beyond the initial bolus dosing, the need for catheter replacement, a statement indicating failure on the QA form or anesthesia record, or a positive intravascular test dose. “Epidural placement duration” was recorded from the time the anesthesiology team entered the parturient’s labor room until the time they exited the room, and included positioning, working time with the actual catheter placement, and administration of drugs. “Training level” was recorded as months in anesthesiology residency. Minutes between placement of labor epidural and determination of failure were recorded to see any difference between obese and non-obese parturients. Accidental dural punctures were recorded as a separate complication and not counted as failure.
STATISTICAL ANALYSIS
All data are expressed as mean ± standard error or as percentages. Multiple regression analysis was used for factor analysis. Unpaired student t-tests, Mann-Whitney U tests and Chi-square tests were used to directly compare two groups for parametric, non-parametric and categorical data, respectively (SigmaStat 3.5, Systat Software Inc, San Jose, CA). Correlation between BMI and determination of catheter failure was performed using IBM SPSS Statistics. Statistical significance (*) was assumed when P < 0.05 (two-tailed). Binning was used for regression analysis and display of difficulty, failure, and duration of placement with BMI.
RESULTS
During the 12-month study period, anesthesia providers cared for 3229 out of 4103 patients who delivered at our institution. Of these, 2485 patients received epidural analgesia for labor pain and planned vaginal delivery. We excluded 113 patients due to lack of information in their anesthesia records. Patient characteristics are displayed in Table 1. Obesity as defined by BMI ≥ 30 was not associated with age, but with comorbidities such as hypertension including pregnancy-induced hypertension (PIH), asthma, obstructive sleep apnea, gastroesophageal reflux disease, gestational and non-gestational diabetes mellitus, preterm labor and fetal distress. Higher BMI was also associated with an increased rate of CDs (Table 2). Our emergent CD rate overall was 0.24%, and 0.09% in non-obese vs. 0.36% in obese, but because of the low numbers this was not significant with Chi-Square (p =0.35).
Table 1.
Patient characteristics and prevalence of co-morbidities in obese, non-obese and all patients
| All patients (n = 2485) |
BMI < 30 (n = 1095) |
BMI ≥ 30 (n = 1390) |
p value | |
|---|---|---|---|---|
| Age (years) | 26.2±0.1 | 26.2±0.2 | 26.2±0.2 | 0.862 |
| Height (inches) | 64.6±0.1 | 64.8±0.1 | 64.4±0.1 | 0.001 |
| Weight (kg) | 85.9±0.4 | 71.3±0.3 | 97.4±0.5 | < 0.001 |
| HTN (%) | 2.4 | 0.6 | 3.8 | < 0.001 |
| PIH (%) | 9.8 | 6.7 | 12.3 | < 0.001 |
| OSA (%) | 0.3 | 0.0 | 0.5 | 0.049 |
| Asthma (%) | 12.6 | 11.0 | 13.8 | 0.038 |
| GERD (%) | 16.5 | 14.6 | 18.0 | 0.028 |
| Non-gestational DM (%) | 0.9 | 0.2 | 1.4 | 0.002 |
| Gestational DM (%) | 3.4 | 2.4 | 4.3 | 0.015 |
Data are mean ± standard error or percentage. * P<0.05
Abbreviations: BMI = body mass index; HTN = hypertension; PIH = pregnancy induced hypertension; OSA = obstructive sleep apnea; GERD = gastroesophageal reflux disease; DM = diabetes mellitus.
Table 2.
Rate of prior and subsequent Cesarean Delivery and their indications, and characteristics of epidural placements in obese, non-obese and all patients
| All patients (n = 2485) |
BMI < 30 (n = 1095) |
BMI ≥ 30 (n = 1390) |
p value | |
|---|---|---|---|---|
| Overall CD (%) | 11.4 | 6.8 | 15.1 | < 0.001 |
| FTP (%) | 7.0 | 3.7 | 9.5 | < 0.001 |
| 1.0 | 0.5 | 1.4 | 0.026 | |
| Fetal distress (%) | 3.5 | 2.1 | 4.5 | 0.001 |
| Prior CD (%) | 0.6 | 0.3 | 0.8 | 0.150 |
| Placement time (min) | 29.3±0.2 | 27.9±0.3 | 30.4±0.3 | < 0.001 |
| Difficulty (%) | 3.0 | 1.6 | 4.1 | < 0.001 |
| Failure (%) | 4.3 | 2.7 | 5.6 | < 0.001 |
| Clinician bolus (%) | 38.3 | 35.1 | 41.5 | < 0.001 |
| > 1 Interspace (%) | 8.1 | 6.4 | 9.7 | 0.003 |
| Dural Puncture (%) | 0.4 | 0.4 | 0.4 | 0.952 |
Data are mean ± standard error or percentage. * P<0.05.
Abbreviations: BMI = body mass index; CD = Cesarean delivery; FTP = failure to progress; FTD = failure to descend.
Overall epidural failure and difficulty rates were 4.3% and 3.0%, respectively. Obese patients had a higher chance of both difficulty in epidural placement and failure of epidural analgesia, and an increased time needed for placement. Rate of difficulty and failure increased 2.5 and 2.1 fold, respectively, in obese vs. non-obese patients. Increased BMI was associated with the need for one or more clinician boluses (*41.5 vs. 35.1%). When clinician boluses were necessary, their number was higher with increased BMI (*1.63±0.05 vs. 1.43±0.04). *9.7 vs. 6.4% of the obese vs. non-obese patients needed to have attempts at more than one lumbar interspace to place the epidural catheter. There was no significant difference in the number of levels when more than one level was necessary (Table 2). As a result, obese parturients required more clinician boluses and lumbar interspace level attempts than non-obese; the same was true when placement failed. For accidental dural punctures and difficult placements, however, only the number of levels was significantly different, not the number of clinician boluses needed.
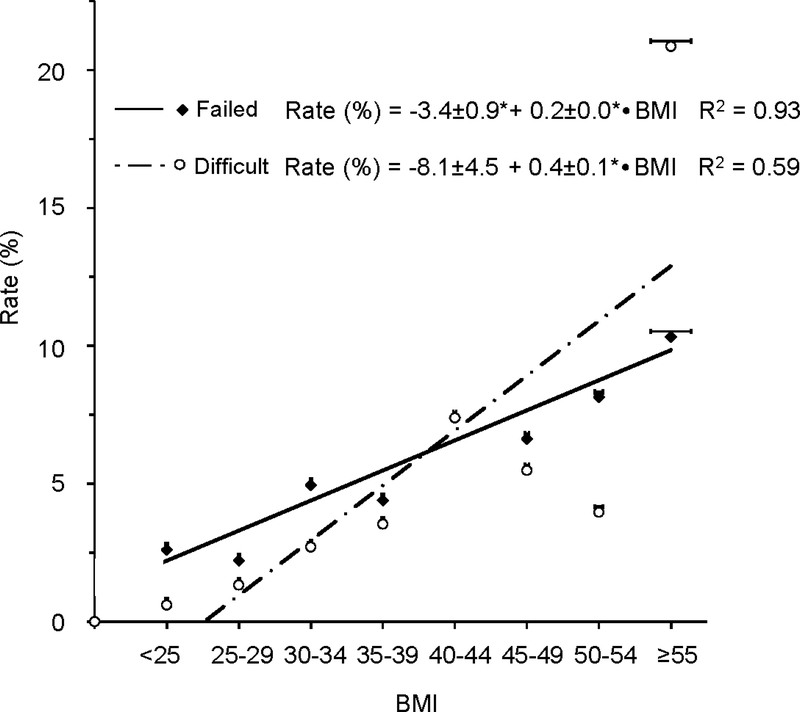
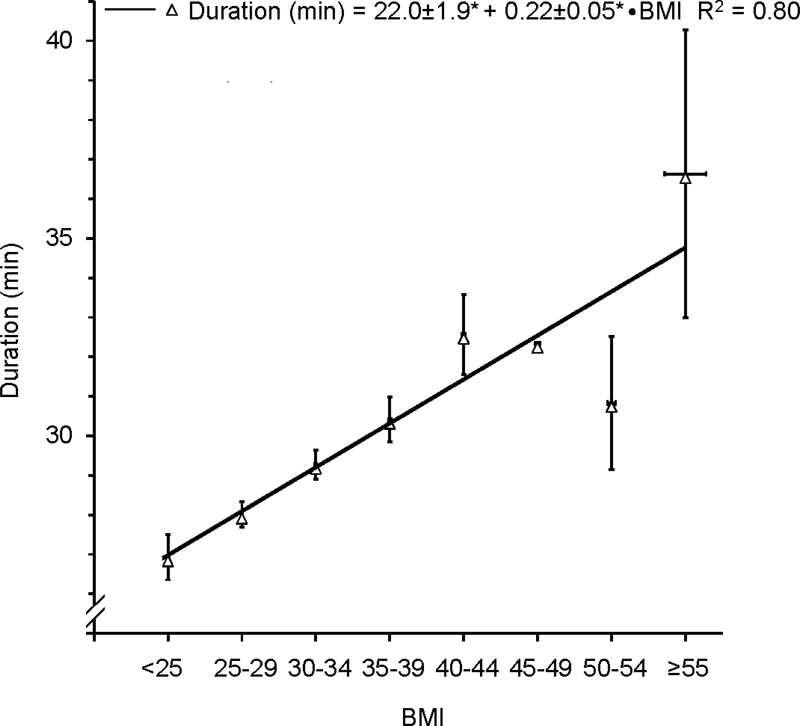
Although epidural placement time was only 2.5 min or 9% longer in average obese (*30.4±0.3 min) vs. non-obese patients (27.9±0.3 min) as defined by BMI, every one point increase in BMI prolonged placement by *13±3 sec (Fig 1) and added *0.4±0.1% and *0.2±0.0% to the overall rate of difficulty and failure of epidural placement, respectively (Fig 2). Moreover, we observed a greater variation (heteroscedasticity) in all parameters as BMI increased, indicating decreased predictability of placement time, difficulty and failure rates at higher BMI.
Figure 1:
The rate (in %) of difficulty (white circles, dotted line) and failure (black diamonds, continuous line) of epidural catheter placement as a function of body mass index (BMI, in kg/m2). Regression analysis of binned data in BMI intervals of 5 revealed a significant linear relationship and correlations of BMI vs. difficulty and failure, respectively. All values are given ± standard error. For easier visibility, standard errors are displayed as capped horizontal bars slightly above the mean symbols. * P < 0.05 (two-tailed).
Figure 2:
Duration of epidural placement (in min; white triangles, continuous line) is plotted against body mass index (BMI, in kg/m2). Regression analysis of binned data in BMI intervals of 5 revealed a significant linear relation and correlation of BMI vs. duration. Values are given ± standard errors. For easier visibility BMI standard errors are displayed as capped horizontal bars slightly above the mean symbols. * P < 0.05 (two-tailed).
Placement duration, difficulty, and failure were additionally interrelated. Placement duration increased by 20 min or 70% when the placement was difficult (*48.7±2.3 min) vs. not difficult (28.7±0.2 min). Conversely, when placement was difficult (Table 3), patients had a higher BMI (*36.5±0.9 vs. 31.8±0.1) and providers had a trend towards less experience (20.8±1.1 vs. 22.9±0.2 months anesthesia training, p = 0.08). Epidural failure was also associated with increased BMI (*34.1±0.7 vs. 31.8±0.1), less provider experience (*19.9±0.9 vs. 22.9±0.3 months anesthesia training) and increased time needed for placement (*36.9±1.8 vs. 28.9±0.2 min) as well as with documented difficulty (Table 3).
Table 3.
BMI and characteristics of epidural placements by difficulty and failure
| All patients | No difficulty | Difficulty | p value | |
|---|---|---|---|---|
| BMI | 31.9±0.1 | 31.8±0.1 | 36.5±0.9 | < 0.001 |
| Placement time (min) | 29.3±0.2 | 28.7±0.2 | 48.7±2.3 | < 0.001 |
| Training (months) | 22.8±0.2 | 22.9±0.2 | 20.8±1.1 | 0.081 |
| Failure (%) | 4.3 | 4.0 | 13.3 | < 0.001 |
| All patients | No failure | Failure | p value | |
| BMI | 31.9±0.1 | 31.8±0.1 | 34.1±0.7 | 0.002 |
| Placement time (min) | 29.3±0.2 | 28.9±0.2 | 36.9±1.8 | < 0.001 |
| Training (months) | 22.8±0.2 | 22.9±0.3 | 19.9±0.9 | 0.002 |
| Difficulty (%) | 3.0 | 2.7 | 9.4 | < 0.001 |
Data are mean ± standard error or percentage. * P<0.05. Abbreviation: BMI = body mass index
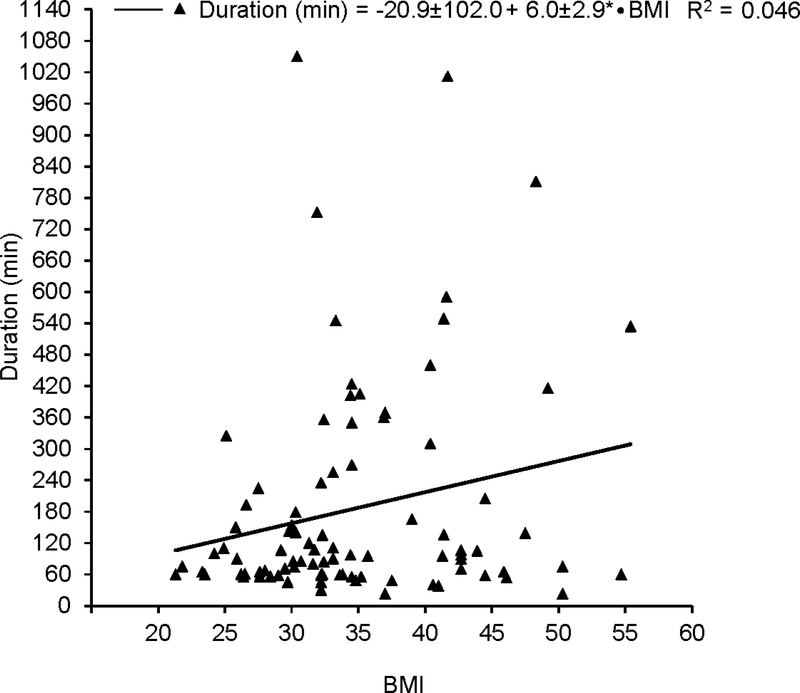
Time between epidural placement and determination of failure was 185±22 min in those that failed; these patients had an average BMI of 34.6±0.8. BMI correlated positively with time until replacement: for every one-unit increase in BMI, the time between placement and determination of failure increased by 6±3 min (Fig 3).
Figure 3:
Time between epidural placement and the decision that the catheter has failed. Every unit of BMI increases the average time to decision regarding failure by almost 6 min while decreasing its predictability as indicated by increasing heteroscedasticity and a low correlation coefficient. Values are given ± standard error; * P < 0.05 (two-tailed).
The need to undergo an unplanned CD was associated with a higher epidural failure rate (*8.8% vs 3.7% for vaginal delivery), but not with more difficulty to place the epidural catheter (3.5% vs 3.0%).
Accidental dural punctures (0.4%) were not associated with increased BMI; they were best predicted by less provider experience (*13.5±0.9 vs. 22.8±0.2 months anesthesia training).
DISCUSSSION
The major findings of this study are that increased BMI correlates with both increasing technical difficulty and failure of labor epidural analgesia as well as longer placement time and delayed recognition of failure. In our study, obesity was assessed by current BMI at delivery because it was the measure available to anesthesiologists for all parturients. Though BMI is an accepted measure of obesity, it does not correlate with adipose tissue distribution in different individuals.2 This distribution can affect palpability of spinous processes, which has been shown to affect ease of placement.12 Our database did not include spine palpability.
It is known that obesity in pregnancy is associated with increased co-morbidities.6,7,14,15 Our results confirm this notion by showing that increasing BMI was age-independent but correlated with co-morbidities such as hypertension including pregnancy-induced hypertension, asthma, obstructive sleep apnea, gastroesophageal reflux disease, gestational and non-gestational diabetes mellitus. As determined by other authors, higher BMI was also associated with an increased CD rate.7,17 Of note, our emergent CD rate overall was 0.24% which did not allow for a statistically significant distinction between non-obese (0.09%) and obese patients (0.36%). Parturients having to undergo an unplanned CD had a higher catheter failure rate, but not a higher rate of difficulty in catheter placement. All of these potential problems emphasize the need to ensure well-functioning neuraxial analgesia in this population to avoid possible difficult airway manipulation and aspiration which may result in aspiration pneumonitis during general anesthesia if needed.
Practitioners assume that in obese parturients, neuraxial techniques will be more difficult to perform, more likely to fail, or both.2,12 However, only a few studies focused on difficulty in initial placement of neuraxial analgesia or specific definitions of difficulty.12,18,21,22,23 We defined difficulty as more than six epidural needle redirections in total in addition to a note mentions difficulty in the chart. Another commonly used measure is the number of attempts. “Attempt” may be described in many different ways and subjective interpretation may cause confusion. Our number of attempts is defined as the number of lumbar interspaces we worked on. The number of interspaces was increased in difficult epidural placements in obese patients. In contrast, the increased number of clinician boluses in obese patients was more a possible indicator of dysfunctional labor. Lee et al.19 also identified an increased number of breakthrough pain episodes needing treatment as a predictor for failure of augmentation of labor epidural for a CD.
Our data indicated overall labor epidural failure and difficulty rates of 4.3% and 3.0%, respectively, which are lower than those reported in the literature. Our study specifically focused on labor epidural failures for planned vaginal deliveries only. We defined labor epidural failure as inadequate analgesia, a positive intravascular test dose, so that the catheter had to be replaced, or when the anesthesia record stated failure. Several previous studies investigated the characteristics and predictive factors for failure of neuraxial anesthesia,13,18 focusing on the failure of augmentation of labor epidural analgesia for CD.19,20,24 Unfortunately, there is no standardized definition of epidural failure in the literature, which may explain the wide range of reported epidural failure rates.25 Another reason for discrepancies may be including different types of failures such as epidural analgesia failure during labor or epidural anesthesia failure during CD. Previous published reports cite epidural failure rates ranging from 8% to 23%.25 Hood et al. described epidural failure as documentation in the anesthetic record as failure or when the epidural catheter had to be replaced. Eappen et al.26 considered an epidural catheter as a failed if replacement of the catheter was needed or a unilateral block was present. Dresner et al. used a midwife satisfaction scoring system, and epidural resite rates to judge epidural failure. They determined that, as BMI increases, epidural catheters are more likely to fail, however they used different definitions of failure.
We chose to use months in training for determining experience of our residents instead of years of residency to be more accurate. Our data showed in difficulty there was a trend towards less experience, but only epidural failures were significantly correlated with less provider experience (Table 3). When planning the anesthetic it is crucial to have a more experienced team for obese parturients.
Our findings indicate that obese parturients are at risk for increased difficulty, failure, and epidural placement time. These three factors were also shown to interact with each other, such that increases in difficulty and failure led to greatly increased neuraxial procedure time. Tonidandel et al.27 showed greater neuraxial procedure times in morbidly obese parturients in their study which supports our findings.
The provision of safe anesthetic care is dependent on timely epidural catheter placement, proper block evaluation, and the ability to rapidly determine a catheter’s efficacy. To examine factors considered by our providers when determining catheter failure, we measured the time between catheter placement and the decision that the catheter had failed. For every one unit increase in BMI, time between placement of the epidural catheter and the decision that the catheter had failed increased by 6 min. Possible explanations for this correlation include actual late catheter failure, possibly caused by catheter dislodgement from the epidural space, difficulty in determining failure, or practitioner reluctance to abandon catheters in obese patients due to fear of facing difficulty and/or failure during replacement. This finding emphasizes the importance of frequent checks of epidural catheters in obese parturients and replacing them in time to prevent further problems.
The present study has natural limitations. As a retrospective paper chart review, it relies on the accurate reporting of individual practitioners on the QA form and/or the anesthesia record. This may result in omissions or bias during data recording. For example, the anesthesia times in our records were measured from entering the room to leaving the room; as a consequence, since more granular data were not available, our reported anesthesia time periods include, e.g., positioning, draping, placement, test dose, and taping of the catheter, all of which contributed to longer reported times. We were not able to distinguish between catheters that failed before the decision for CD, and those that failed when trying to bolus the catheter for CD; therefore, a cause-effect relation between catheter failure and unplanned CD is not possible to determine. These limitations, however, were true for all patients regardless of BMI and are, therefore, not expected to have skewed our data or conclusions. In addition, our meticulous review of both the QA form and the individual anesthesia records may mitigate reporting bias by considering two complementary sources of data. Lastly, we have increased the study’s statistical power by including a high number of patients.
In summary, obesity in parturients correlates significantly with increasing difficulty and time needed for labor epidural placement, epidural failure and time to recognize failure. These are important considerations necessitating an open discussion and a multidisciplinary approach with experienced personnel when caring for obese parturients.
HIGHLIGHTS:
We examined the correlation between BMI and labor epidural failure and difficulty.
Obese parturients had higher rates of both failure and difficulty of labor epidural analgesia.
Obese parturients required more lumbar interspace level attempts for successful placement, and more clinician boluses to achieve adequate analgesia.
Having a less-experienced trainee was correlated with epidural failure, but not with placement difficulty.
Practitioners should consider allotting extra time for obese parturients’ care, in order to manage potential problems.
ACKNOWLEDGEMENT
The authors would like to thank Sandy Abendroth (Department of Anesthesiology, Medical College of Wisconsin) and Wolfgang Gaggl, PhD (Department of Radiology, University of Wisconsin) for their assistance with this study.
Funding: Institutional only. Unrelated research funding to MLR was received from the Department of Veterans Affairs (IK2 BX001278) and the NIH (R01 HL123227).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Soens MA, Birnbach DJ, Ranasinghe JS, van Zundert A. Obstetric anesthesia for the obese and morbidly obese patient: an ounce of prevention is worth more than a pound of treatment. Acta Anaesthesiol Scand 2008;52(1):6–19. [DOI] [PubMed] [Google Scholar]
- 2.Saravanakumar K, Rao SG, Cooper GM. Obesity and obstetric anaesthesia. Anaesthesia 2006;61(1):36–48. [DOI] [PubMed] [Google Scholar]
- 3.Roofthooft E Anesthesia for the morbidly obese parturient. Curr Opin Anaesthesiol 2009;22(3):341–6. [DOI] [PubMed] [Google Scholar]
- 4.Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of childhood and adult obesity in the United States, 2011–2012. JAMA 2014;311(8):806–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity among adults: United States, 2011–2012. NCHS Data Brief 2013;(131)(131):1–8. [PubMed] [Google Scholar]
- 6.Flegal KM, Carroll MD, Kit BK, Ogden CL. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999–2010. JAMA 2012;307(5):491–7. [DOI] [PubMed] [Google Scholar]
- 7.Badve MH, Golfeiz C, Vallejo MC. Anesthetic considerations for the morbid obese parturient. Int Anesthesiol Clin 2014;52(3):132–47. [DOI] [PubMed] [Google Scholar]
- 8.Cedergren MI. Maternal morbid obesity and the risk of adverse pregnancy outcome. Obstet Gynecol 2004;103(2):219–24. [DOI] [PubMed] [Google Scholar]
- 9.McClure JH, Cooper GM, Clutton-Brock TH, Centre for Maternal and Child Enquiries. Saving mothers’ lives: reviewing maternal deaths to make motherhood safer: 2006–8: a review. Br J Anaesth 2011;107(2):127–32. [DOI] [PubMed] [Google Scholar]
- 10.Hawkins JL, Chang J, Palmer SK, Gibbs CP, Callaghan WM. Anesthesia-related maternal mortality in the United States: 1979–2002. Obstet Gynecol 2011;117(1):69–74. [DOI] [PubMed] [Google Scholar]
- 11.Saravanakumar K, Rao SG, Cooper GM. The challenges of obesity and obstetric anaesthesia. Curr Opin Obstet Gynecol 2006;18(6):631–5. [DOI] [PubMed] [Google Scholar]
- 12.Ellinas EH, Eastwood DC, Patel SN, Maitra-D’Cruze AM, Ebert TJ. The effect of obesity on neuraxial technique difficulty in pregnant patients: a prospective, observational study. Anesth Analg 2009;109(4):1225–31. [DOI] [PubMed] [Google Scholar]
- 13.Pan PH, Bogard TD, Owen MD. Incidence and characteristics of failures in obstetric neuraxial analgesia and anesthesia: a retrospective analysis of 19,259 deliveries. Int J Obstet Anesth 2004;13(4):227–33. [DOI] [PubMed] [Google Scholar]
- 14.Hood DD, Dewan DM. Anesthetic and obstetric outcome in morbidly obese parturients. Anesthesiology 1993;79(6):1210–8. [DOI] [PubMed] [Google Scholar]
- 15.Dresner M, Brocklesby J, Bamber J. Audit of the influence of body mass index on the performance of epidural analgesia in labour and the subsequent mode of delivery. BJOG 2006;113(10):1178–81. [DOI] [PubMed] [Google Scholar]
- 16.Bamgbade OA, Khalaf WM, Ajai O, Sharma R, Chidambaram V, Madhavan G. Obstetric anaesthesia outcome in obese and non-obese parturients undergoing caesarean delivery: an observational study. Int J Obstet Anesth 2009;18(3):221–5. [DOI] [PubMed] [Google Scholar]
- 17.Ray A, Hildreth A, Esen UI. Morbid obesity and intra-partum care. J Obstet Gynaecol 2008;28(3):301–4. [DOI] [PubMed] [Google Scholar]
- 18.Agaram R, Douglas MJ, McTaggart RA, Gunka V. Inadequate pain relief with labor epidurals: a multivariate analysis of associated factors. Int J Obstet Anesth 2009;18(1):10–4. [DOI] [PubMed] [Google Scholar]
- 19.Lee S, Lew E, Lim Y, Sia AT. Failure of augmentation of labor epidural analgesia for intrapartum cesarean delivery: a retrospective review. Anesth Analg 2009;108(1):252–4. [DOI] [PubMed] [Google Scholar]
- 20.Halpern SH, Soliman A, Yee J, Angle P, Ioscovich A. Conversion of epidural labour analgesia to anaesthesia for Caesarean section: a prospective study of the incidence and determinants of failure. Br J Anaesth 2009;102(2):240–3. [DOI] [PubMed] [Google Scholar]
- 21.Kim JH, Song SY, Kim BJ. Predicting the difficulty in performing a neuraxial blockade. Korean J Anesthesiol 2011;61(5):377–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sprung J, Bourke DL, Grass J, Hammel J, Mascha E, Thomas P, Tubin I. Predicting the difficult neuraxial block: a prospective study. Anesth Analg 1999;89(2):384–9. [DOI] [PubMed] [Google Scholar]
- 23.Guglielminotti J, Mentre F, Bedairia E, Montravers P, Longrois D. Development and evaluation of a score to predict difficult epidural placement during labor. Reg Anesth Pain Med 2013;38(3):233–8. [DOI] [PubMed] [Google Scholar]
- 24.Orbach-Zinger S, Friedman L, Avramovich A, Ilgiaeva N, Orvieto R, Sulkes J, Eidelman LA. Risk factors for failure to extend labor epidural analgesia to epidural anesthesia for Cesarean section. Acta Anaesthesiol Scand 2006;50(7):793–7. [DOI] [PubMed] [Google Scholar]
- 25.Thangamuthu A, Russell IF, Purva M. Epidural failure rate using a standardised definition. Int J Obstet Anesth 2013;22(4):310–5. [DOI] [PubMed] [Google Scholar]
- 26.Eappen S, Blinn A, Segal S. Incidence of epidural catheter replacement in parturients: a retrospective chart review. Int J Obstet Anesth 1998;7(4):220–5. [DOI] [PubMed] [Google Scholar]
- 27.Tonidandel A, Booth J, D’Angelo R, Harris L, Tonidandel S. Anesthetic and obstetric outcomes in morbidly obese parturients: a 20-year follow-up retrospective cohort study. Int J Obstet Anesth 2014;23(4):357–64 [DOI] [PubMed] [Google Scholar]