Abstract
Air pollution epidemiology studies of ambient fine particulate matter (PM2.5) often use outdoor concentrations as exposure surrogates, which can induce exposure error. The goal of this study was to improve ambient PM2.5 exposure assessments for a repeated measurements study with 22 diabetic individuals in central North Carolina called the Diabetes and Environment Panel Study (DEPS) by applying the Exposure Model for Individuals (EMI), which predicts five tiers of individual-level exposure metrics for ambient PM2.5 using outdoor concentrations, questionnaires, weather, and time-location information. Using EMI, we linked a mechanistic air exchange rate (AER) model to a mass-balance PM2.5 infiltration model to predict residential AER (Tier 1), infiltration factors (Finf_home, Tier 2), indoor concentrations (Cin, Tier 3), personal exposure factors (Fpex, Tier 4), and personal exposures (E, Tier 5) for ambient PM2.5. We applied EMI to predict daily PM2.5 exposure metrics (Tiers 1–5) for 174 participant-days across the 13 months of DEPS. Individual model predictions were compared to a subset of daily measurements of Fpex and E (Tiers 4–5) from the DEPS participants. Model-predicted Fpex and E corresponded well to daily measurements with a median difference of 14% and 23%; respectively. Daily model predictions for all 174 days showed considerable temporal and house-to-house variability of AER, Finf_home, and Cin (Tiers 1–3), and person-to-person variability of Fpex and E (Tiers 4–5). Our study demonstrates the capability of predicting individual-level ambient PM2.5 exposure metrics for an epidemiological study, in support of improving risk estimation.
Keywords: air pollution, exposure modeling, particulate matter, building infiltration modeling
1. Introduction
Epidemiologic studies have reported associations between ambient (i.e., outdoor-generated) fine particulate matter (PM2.5; particulate matter ≤ 2.5 μm in aerodynamic diameter) and various indices of acute cardiopulmonary morbidity and mortality (U.S. EPA, 2009). Due to cost and participant burden of indoor and personal air pollution monitoring, most of these studies used outdoor PM2.5 concentrations as exposure surrogates. However, these exposure surrogates do not account for (1) time spent indoors with ambient PM2.5 levels attenuated from outdoor concentrations, and (2) building-to-building and temporal variability of this attenuation. Differences between exposure surrogates, such as outdoor concentrations, and true exposures contribute to exposure measurement errors. Depending on epidemiological study design, these errors can add bias or uncertainty to health effect estimates, which was highlighted in two National Research Council reports: “Exposure Science in the 21st Century: A Vision and a Strategy” (NRC, 2012) and “Research Priorities for Airborne Particulate Matter” (NRC, 2004), and two National Academies of Sciences reports: “Health Risks of Indoor Exposure to Particulate Matter” (NAS, 2016) and “Using 21st Century Science to Improve Risk-Related Evaluations” (NAS, 2017). To help reduce measurement errors and improve PM2.5 health effect estimation, we developed the Exposure Model for Individuals (EMI), which addresses the recommendations of these reports (Breen et al., 2015). This study describes the application of EMI for ambient PM2.5 in the Diabetes and the Environment Panel Study (DEPS; Schneider et al., 2008).
The goal of DEPS is to examine ambient PM2.5 exposures and cardiovascular and hematologic effects in adults with type 2 diabetes living in central North Carolina (NC). Using PM2.5 measurements from two fixed-site air monitors, significant associations were previously found between daily ambient PM2.5 concentrations and various acute (maximum lag of 4 days) adverse effects: (1) altered endothelial function, (2) increased blood levels of interleukin-6 and tumor necrosis factor-α, (3) changes in indicators of cardiac repolarization, and (4) upregulated expression of receptors on circulating monocytes (Schneider et al., 2008, 2010, 2011). In this study, we applied EMI for a subsequent epidemiological analysis to address the possible limitation of using PM2.5 concentrations from fixed-site monitors as exposure surrogates in DEPS.
The EMI predicts individual-level exposure metrics for actual participants in epidemiological studies using outdoor concentrations, weather (e.g., temperature, wind speed), questionnaires (e.g., building characteristics), and time-location data (Breen et al., 2015). In this study, we predict five tiers of ambient PM2.5 exposure metrics for each health study participant in DEPS (Figure 1). We previously described the development of these five tiers of exposure metrics for ambient PM2.5 (Breen et al., 2015). Briefly, EMI includes three exposure metrics related to PM2.5 infiltration into homes (Tier 1: air exchange rate (AER), Tier 2: infiltration factors, Tier 3: indoor concentrations) and two exposure metrics that account for time spent in different indoor and outdoor locations (Tier 4: personal exposure factors, Tier 5: exposures). The importance of these five tiers of exposure metrics for epidemiological studies was highlighted in the National Academy of Sciences Report “Health Risks of Indoor Exposure to Particulate Matter” (NAS, 2016), and demonstrated in epidemiological studies that applied population-level exposure metrics (Hodas et al., 2013; Sarnat et al., 2013; Jones et al., 2013; Mannshardt et al., 2013).
Figure 1.
Conceptual model of EMI to predict five tiers of individual-level exposure metrics for ambient PM2.5. Tiers 1–3 are related to homes (AER – air exchange rate, Finf_home – infiltration factor, Cin – indoor concentration), and Tiers 4–5 (Fpex – personal exposure factor, E – personal exposure are related to particpants.
Our overall goal is to calibrate and evaluate EMI with extensive exposure data from field studies to reduce model uncertainty, and then apply EMI for epidemiological studies with limited exposure data. Therefore, we previously performed a cross-validation of EMI (Breen et al., 2010, 2015). We used measurement data from the Research Triangle Park Particulate Matter Panel Study (PM Panel Study) to evaluate the five tiers of exposure metrics for ambient PM2.5. The PM Panel Study represents various residential AER and infiltration factors, outdoor and indoor concentrations, and personal exposures for 31 participants for seven consecutive days in each of four consecutive seasons for the same region of central NC as DEPS.
In this paper, we develop ambient PM2.5 exposure metrics for DEPS. We used housing characteristics, time-location diaries, and weather data as inputs for EMI, and measurements of ambient PM2.5 personal exposure factors from DEPS participants for model evaluation. We first describe the DEPS design, and then describe EMI, the method for model evaluation, and the development of daily predictions of five tiers of exposure metrics for each study participant.
2. Materials and methods
2.1. DEPS design
The DEPS was designed to examine the relationship between exposures to ambient PM2.5 and various indices of acute cardiovascular and hematologic effects in a cohort of adults with diabetes. Previous publications describe the study design, clinical measurements, and air pollution measurements from two fixed-site ambient PM2.5 monitors (Schneider et al., 2008, 2010, 2011). Briefly, the study included 22 non-smoking adult participants with type 2 diabetes living in central NC. Each participant visited the U.S. Environmental Protection Agency (EPA) Human Studies Facility (HSF) in Chapel Hill, NC at 9 am (± 1.5 h) on 5 consecutive weekdays between November 2004 and December 2005. Various clinical measurements were collected at baseline and during four follow-up visits to yield a total of 88 participant-days of data. All volunteers signed a written consent form and the study protocol was approved by the University of North Carolina Human Studies Biomedical Institutional Review Board and the EPA.
For fixed-site air pollution monitoring, we used daily concentrations of ambient PM2.5 mass (24-h average, midnight - midnight) from an official network monitoring station located approximately 44 km (27 miles) east of the HSF. We also measured daily concentrations of ambient PM2.5 mass (24-h average, 9 am - 9 am) with a 3000K Versatile Air Pollution Sampler (URG Corp., Chapel Hill, NC, USA) (Williams et al., 2000) located on the HSF rooftop (30 m elevation).
For model evaluation of EMI, we measured daily PM2.5 mass concentrations (24-h average, 9 am - 9 am) with two personal exposure monitors (PEM) operating at 2 L/min (Williams et al., 2003, 2009). One PEM was worn by the participant, and another PEM was located on the HSF rooftop. Personal monitoring was conducted with a nylon vest (Williams et al., 2003, 2009). The vest provided a means to securely and reproducibly position the air inlet in the breathing zone of the participant and allowed safe freedom of movement for normal daily activities. No restrictions were placed on participants as related to their activities. Sulfate concentrations (tracer of ambient PM2.5 mass) for the PEM filters were determined using X-ray fluorescence (Wallace et al., 2005). These sulfate measurements were used for model evaluation, as described below.
Input data for EMI were obtained for weather, home building characteristics, and participant time-activities. Outdoor temperatures and wind speeds (10 m elevation) were obtained from local airport measurements. Daily questionnaires were used to collect occupant behavior related to building operation, including opening windows. Daily time-location diaries were used to determine duration that participants spent in seven microenvironments (indoors: home, work, other locations; outdoors: home, work, other locations; inside vehicles).
2.2. Tiers of exposure metrics
Using EMI, we modeled five tiers of daily exposure metrics for PM2.5 for the 22 study participants and their homes (Figure 1). The five tiers, which have increasing levels of complexity and information needs, include: (Tier 1) residential air exchange rates, (Tier 2) residential infiltration factors, (Tier 3) residential indoor concentrations, (Tier 4) personal exposure factors, (Tier 5) personal exposures. For each participant, 24-h average exposure metrics were modeled on the four consecutive days with clinical measurements, and on the four days before clinical visits to yield a total of 176 participant-days. Two sets of daily 24-h averages were modeled (9 a.m. to 9 a.m., midnight to midnight), and time-matched to the daily measurements from the two fixed-site ambient PM2.5 monitors. We also measured two tiers of exposure metrics (Tiers 4–5) based on 24-h average monitor data, which were time-matched (9 a.m. to 9 a.m.) to the equivalent modeled exposure metric. The modeling and subsequent analysis were implemented using MATLAB software (version R2015a, Mathworks, Natick, MA, USA).
2.3. Modeled exposure metrics for Tiers 1–5
For Tier 1, residential AER were predicted from questionnaires and meteorology using two different models: Lawrence Berkeley Laboratory model (LBL) and the extended LBL model (LBLX) (Breen et al., 2010). These AER models are mechanistic by accounting for the physical driving forces of the airflows (i.e., pressure differences across building envelope from indoor-outdoor temperature differences, called the stack effect, and wind). Both models include leakage airflow through unintentional openings in a building envelope (e.g., cracks around window, doors), whereas the LBLX model also includes natural ventilation through controlled openings in the building envelope (e.g., open windows). We developed separate exposure metrics using these two AER models to evaluate the potential benefit of including natural ventilation for epidemiological studies.
These AER models were previously described and evaluated for homes in the same region of central NC as DEPS (Breen et al., 2010). Briefly, the leakage airflow is defined as
| [1] |
where Aleak is the effective air leakage area, ks is the stack coefficient, kw is the wind coefficient, Tin and Tout are the average indoor and outdoor temperatures, respectively, and U is the average wind speed (see Supplementary Material). The AER is calculated as QLBL divided by the building volume V.
We used the LBLX model to account for natural ventilation airflow on days when participants opened windows (Breen et al., 2010). The days with open windows were determined from daily questionnaires collected on four consecutive days. If a participant reported open windows for 2–4 days, we assumed open windows for the four days before diaries were collected (lag days for the subsequent health outcome analysis).
For the LBLX model, the airflow is defined as:
| [2] |
where QLBL is the leakage airflow as defined above, and Qnat is the natural ventilation airflow through open windows (see Supplementary Material). The AER is calculated as QLBLX divided by V.
For Tier 2, residential PM2.5 infiltration factors were predicted with a steady-state mass balance infiltration model described by
| [3] |
where P is the penetration coefficient (dimensionless), and kr is the indoor removal rate of PM2.5 (h−1) (Breen et al., 2010). The P and kr were estimated by Breen et al. (2010) in the PM Panel Study from homes in the same region of NC as DEPS (P = 0.84, kr = 0.21h−1).
For Tier 3, residential indoor concentrations of ambient PM2.5 ( and ) were predicted from measured outdoor concentrations from the HSF rooftop (Cout_rooftop) and the central-site official network (Cout_central) based on the steady-state equations (Breen et al. 2015; Wallace et al., 2005)
| [4] |
| [5] |
We predicted separate residential indoor concentrations using Cout_rooftop and Cout_central to evaluate the potential benefit of applying outdoor concentrations from the HSF rooftop as compared to the central-site for a future DEPS health analysis.
For Tier 4, personal exposure factors of ambient PM2.5 were predicted as defined by
| [6] |
where f is the fraction of time spent in the seven microenvironments (indoors and outdoors at home, work, other; inside vehicles), which were determined from each participant’s daily time-activity diary collected on four consecutive days. For the four days before diaries were collected (lag days), we set each participant’s time spent in the seven microenvironments to the median across the four days with diaries. The and are the PM2.5 infiltration factors for buildings other than homes and for vehicles, respectively. We set to 0.64 based on the average of three literature-reported PM2.5 infiltration factors for offices, stores, and restaurants (Burke et al., 2001). We set to 0.44 based on the literature-reported PM2.5 infiltration factor for cars (Ott et al., 2008).
For Tier 5, we predicted personal exposures to ambient PM2.5 from Cout_rooftop and Cout_central as defined by (Breen et al., 2015; Wallace et al., 2005)
| [7] |
| [8] |
We predicted separate personal exposures using Cout_rooftop and Cout_central to evaluate the potential benefit of applying outdoor concentrations from the HSF rooftop as compared to the central-site for a future DEPS health analysis.
2.4. Measured exposure metrics for Tiers 4–5
For Tier 4, we determined personal exposure factors of ambient PM2.5 as defined by (Wallace et al., 2005)
| [9] |
where Spersonal and Sout_rooftop are the measured sulfate concentrations derived from the PEM filters worn by the participant and located on the HSF rooftop, respectively. The Fpex is the fraction of ambient PM2.5 that an individual is exposed to. The Fpex accounts for indoor attenuation of ambient PM2.5 and the participant’s daily time within different indoor and outdoor locations. Other studies have used personal/outdoor sulfate ratios to estimate personal exposure factors of ambient PM2.5 based on two assumptions: (1) no indoor sources of sulfate, and (2) physical behavior of sulfate is similar to other ambient PM2.5 components (Ebelt et al., 2005; Sarnat et al., 2002; Koutrakis et al., 1992). For the first assumption, studies show that few indoor sources exist (Koutrakis et al., 1992), and Wallace et al., (2005) verified this assumption for the NC homes in the PM Panel Study. In DEPS, measured Fpex was substantially >1 for one participant-day (1.33), which indicates a possible indoor source of sulfate (Wallace et al., 2005). Therefore, data for that participant-day was removed. For the second assumption, Sarnat et al. (2002) showed daily indoor/outdoor sulfate ratios compared closely to corresponding indoor/outdoor PM2.5 ratios, with no significant bias and a mean relative difference of 14%. For Tier 5, we determined personal exposures of ambient PM2.5 as defined by (Wallace et al., 2005)
| [10] |
2.5. Model evaluation for Tiers 4–5
To evaluate the model predictions for the Tiers 4–5, we calculated the difference (Δ) and relative difference (ε) as:
| [11] |
| [12] |
where TIERi,pred and TIERi,meas are the individual model predictions and measurements, respectively, for Tier i where i = 4 and 5. We also calculated the absolute values |ε| and |Δ|.
3. Results
For the model inputs, summary statistics are provided for the building characteristics of the homes, number of days windows were opened, weather, ambient PM2.5 concentrations, and time-location data (Supplementary Material, Tables S4–S7). Windows were opened on 21% of the study days. The median daily rooftop PM2.5 concentrations varied between 2.2 and 39.9 μg m−3. The mean daily time spent by the participants was 90% indoors, 9% outdoors, and 1% inside vehicles.
3.1. Model evaluation
For the subset of days with measurements of Tier 4 and 5 exposure metrics, summary statistics are provided for measured Fpex and Erooftop (Table 1). We had measurements for 21 participants (1 participant had missing data), which included 76 days for Tier 4 and 75 days (1 day had missing data) for Tier 5. The measured median (minimum-maximum) for Fpex was 0.54 (0.33–1.05) and for Erooftop was 7.1 μg m−3 (2.3–31.9 μg m−3). These measurements are consistent with those previously reported in the PM Panel Study from participants in the same region of NC as DEPS, which measured Fpex from 0.16 to 1.03, and exposures from 1.7 to 35.6 μg m−3. The lack of indoor sources of sulfate can be seen in the measured Fpex not exceeding one, except for one day close to one (1.05). Summary statistics are also provided for the modeled Fpex and Erooftop (Table 1). Using the LBLX model, the modeled and measured Fpex had similar medians (25th-75th percentiles) of 0.59 (0.54–0.63) and 0.54 (0.45–0.66), respectively. The modeled and measured Erooftop also had similar medians (25th-75th percentiles) of 7.4 μg m−3 (4.8–9.3 μg m−3) and 7.1 μg m−3 (5.2–9.2 μg m−3). Using the LBL model, the modeled Fpex and Erooftop had slightly lower medians than using the LBLX model.
Table 1.
Summary statistics for subset of days with measured Tier 4 and 5 exposure metrics, and corresponding model predictions
| Tier of Exposure Metricsa | Measured or Modeled (AER model)b | Number of participants | Measurements (24-h average)c | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample size | Mean | SD | Min | p5 | p25 | p50 | p75 | p95 | Max | |||
| Tier 4: Fpex | Measured | 21 | 76 | 0.55 | 0.16 | 0.33 | 0.35 | 0.46 | 0.54 | 0.66 | 0.85 | 1.05 |
| Modeled (LBL) | 21 | 76 | 0.57 | 0.06 | 0.43 | 0.47 | 0.53 | 0.57 | 0.61 | 0.65 | 0.67 | |
| Modeled (LBLX) | 21 | 76 | 0.58 | 0.06 | 0.43 | 0.48 | 0.53 | 0.59 | 0.63 | 0.67 | 0.69 | |
| Tier 5: Erooftop (μg/m3) | Measured | 21 | 75 | 7.6 | 4.1 | 2.3 | 2.7 | 5.2 | 7.1 | 9.2 | 12.2 | 31.9 |
| Modeled (LBL) | 21 | 75 | 8.2 | 4.6 | 2.5 | 3.2 | 4.8 | 7.3 | 9.3 | 17.7 | 25.8 | |
| Modeled (LBLX) | 21 | 75 | 8.3 | 4.7 | 2.5 | 3.2 | 4.8 | 7.4 | 9.3 | 18.3 | 25.8 | |
Fpex: personal exposure factor (dimensionless), Erooftop: personal PM2.5 exposure
AER model corresponds to LBL or LBLX model
SD corresponds to standard deviation, p5–p95 correspond to percentiles
We compared daily individual modeled and measured Tier 4 and 5 (Figure 2; Supplementary Material, Tables S8–S11, Figures S1–S2). For Tier 4, Fpex had similar quartiles using the LBL and LBLX models with the same median |ε| of 14%, and same median |Δ| of 0.08. Using the LBL and LBLX models, Fpex was generally overestimated with median ε of 6% and 7%, respectively. For Tier 5, Erooftop had similar quartiles using the LBL and LBLX models with the same median |ε| of 23%, and similar median |Δ| of 1.5 μg m−3 and 1.6 μg m−3, respectively. Using the LBL and LBLX models, Erooftop was generally overestimated with median ε of 6% and 10%, respectively. These comparisons are consistent with those previously reported in the PM Panel Study, which showed median |ε| of 20% and 18% for Fpex, and 23% and 20% for exposures using the LBL and LBLX models, respectively.
Figure 2.

Comparison of differences |Δ| and relative differences |ε| between individual model predictions and measurements for Tiers 4 and 5. Results are separated by two AER models (LBL, LBLX) used for mass balance infiltration model. Shown are medians with 25th and 75th percentiles.
3.2. Model predictions for DEPS
To apply EMI for DEPS, we modeled five tiers of daily exposure metrics for all 22 study participants and their homes. Model predictions for the two sets of daily 24-h averages (9 am – 9 am, midnight – midnight) are provided. Since the two sets of averages showed no substantial differences, we will describe the details of only the 9 am – 9 am averages, which are more closely time-matched to the daily health measurements. We modeled a total of 174 participant-days since the ambient PM2.5 monitors had 2 days of missing data.
Summary statistics are provided for the overall distribution of the five tiers of model-predicted exposure metrics across the 174 days (Figures 3–4; Supplementary Material, Tables S12-S13). We first examined the exposure metrics related to the homes (Tiers 1–3). For Tier 1, the median AER (minimum-maximum) was 0.28 h−1 (0.06–0.64 h−1) and 0.30 h−1 (0.06–0.72 h−1) using the LBL and LBLX models, respectively. The median AER are slightly lower than those reported in the PM Panel Study, which showed median AER of 0.37 h−1 and 0.42 h−1 using the LBL and LBLX models, respectively. As compared to the PM Panel Study homes, the DEPS homes were generally newer and larger, and no households were low-income, which yield smaller leakage areas (Equation S2). Since the AER driving forces (i.e., indoor-outdoor temperature, wind speed) were similar in both studies, the lower AER in DEPS was primarily due to the tighter building envelopes.
Figure 3.
Model predictions for Tiers 1–5 using LBL model for home AER model. Results (24-h average, 9 am - 9 am) are sorted by median values from highest to lowest. Shown are medians with 25th and 75th percentiles, and whiskers for minimum and maximum values.
Figure 4.
Model predictions for Tiers 1–5 using LBLX model for home AER model. Results (24-h average, 9 am - 9 am) are sorted by median values from highest to lowest. Shown are medians with 25th and 75th percentiles, and whiskers for minimum and maximum values.
For Tier 2, the median Finf_home (minimum-maximum) was 0.48 (0.18–0.63) and 0.49 (0.18–0.65) using the LBL and LBLX models, respectively. These Finf_home ranges correspond to 37–82% (LBL model) and 35–85% (LBLX model) lower indoor PM2.5 concentrations than outdoors. The median Finf_home are similar to those reported in the PM Panel Study, which showed median Finf_home of 0.53 and 0.56 using the LBL and LBLX models, respectively. For Tier 3, the median Cin,rooftop (minimum-maximum) was 5.8 μg/m3 (1.4–16.7 μg/m3) and 5.9 μg/m3 (1.4–17.8 μg/m3) using the LBL and LBLX models, respectively. As compared to the LBL model, the LBLX model had slightly higher median Cin,rooftop, which is consistent with the slightly higher median AER and Finf_home.
We then examined the overall distributions of the modeled exposure metrics related to personal exposure (Tiers 4–5). For Tier 4, the median Fpex (minimum-maximum) was 0.58 (0.41–0.69) for both the LBL and LBLX models, respectively. This Fpex range corresponds to 31–59% lower PM2.5 exposures than outdoor concentrations. As compared to Finf_home, the median Fpex was slightly higher (i.e., lower attenuation of ambient PM2.5) primarily due to time spent outdoors. The median Fpex are consistent to those reported in the PM Panel Study, which showed median Fpex of 0.58 and 0.60 using the LBL and LBLX models, respectively. For Tier 5, the median Erooftop (minimum-maximum) was 7.0 μg m3 (1.5–23.7 μg/m3) and 7.3 μg m3 (1.5–25.1 μg/m3) using the LBL and LBLX models, respectively. As compared to Cin,rooftop, Erooftop is slightly higher since modeled Fpex tends to be larger than Finf_home, which is consistent with the PM Panel Study.
We compared the daily variability of the modeled exposure metrics for individual homes (Tiers 1–3) and participants (Tier 4–5) (Figures 3–4). For Tier 1, the temporal AER variability (maximum-minimum) within homes was between 0.04 and 0.51 h−1 for both the LBL and LBLX models. This substantial temporal variability, along with the substantial house-to-house variability, was due to variations of wind speed, indoor-outdoor temperature differences, and open windows. The house-to-house variability was also due to building leakage area differences. For the six homes with open windows (3, 4, 6, 9, 11, 18), the AER median and temporal variability were larger using the LBLX model than the LBL model.
For Tier 2, the temporal Finf_home variability within homes was between 0.04 and 0.31 for both the LBL and LBLX models. In plots of the homes ranked by median AER and Finf_home (Figures 3–4), homes with smaller and larger median AER tend to have smaller and larger median Finf_home, respectively. However, the order of a few homes did not match due to the nonlinear relationship between AER and Finf_home (Equation 3).
For Tier 4, the temporal Fpex variability within participants was smaller than Finf_home. This decrease in temporal variability is primarily due to time spent in microenvironments other than indoors at home, which have constant infiltration factors. In plots of the participants ranked by median Fpex and homes ranked by median Finf_home, the order of most subjects and their corresponding homes did not closely match due to variations in time spent in microenvironments with different infiltration factors.
For Tier 5, the temporal Erooftop variability within subjects was larger than Cin_rooftop for both LBL and LBLX models. When a range of outdoor concentrations are multiplied by an attenuation factor (Equations 4, 7) that can vary between 0 and 1, the resulting range of concentrations will vary between 0 and 100% of the outdoor concentration range, respectively. Therefore, the temporal variability of Erooftop > Cin_rooftop is primarily due to typical values of Fpex > Finf_home.
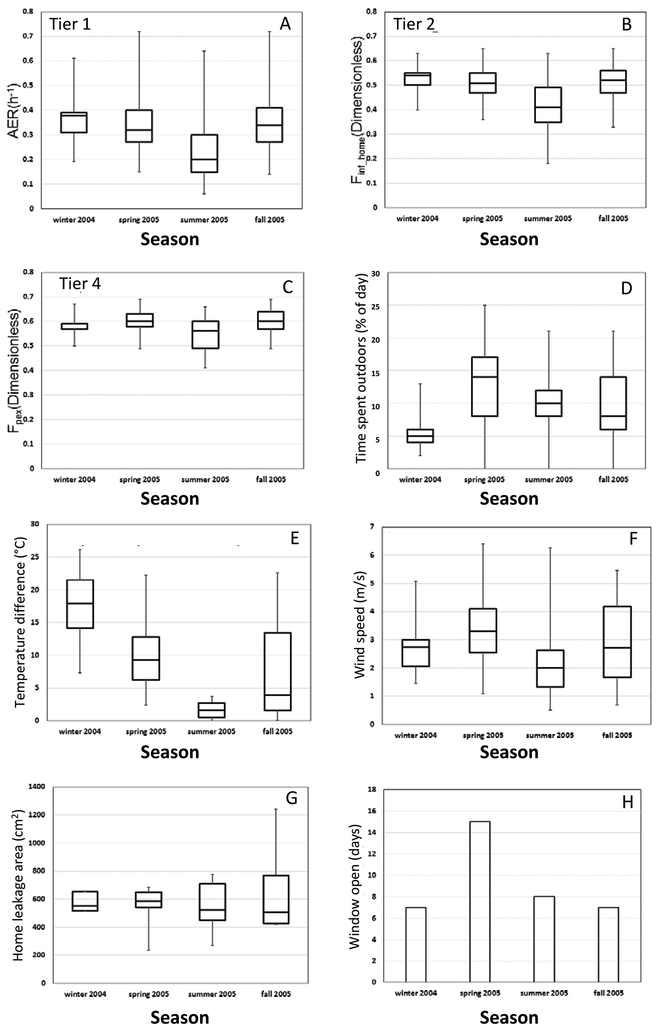
We compared the seasonal variability of the modeled exposure metrics for individual homes (Tiers 1–2) and participants (Tier 4) (Figure 5). For Tier 1, the median AER was lowest and highest in the summer and winter, respectively, for both the LBL and LBLX models. This corresponds with the median indoor-outdoor temperature difference, which was lowest and highest in the summer and winter, respectively. The median wind speeds and home leakage areas had no substantial variability between seasons. The median AER for each season was slightly higher using the LBLX model as compared to the LBL model, which is due to windows opened in each season.
Figure 5.
Model predictions using LBLX model for home AER model across four seasons for Tiers 1, 2, 4 (A-C), time spent outdoors (D), indoor-outdoor temperature difference (E), wind speed (F), home leakage area (G), and window opening (H). Results (24-h average, 9 am - 9 am) shown are medians, 25th and 75th percentiles, and whiskers for minimum and maximum values.
For Tier 2, the median Finf_home was lowest in the summer and highest in the winter, which is consistent with the median AER. For Tier 4, the median Fpex were similar for each season with only a small variation between 0.56 and 0.60. The seasonal increase between the median Fpex and Finf_home was lowest in winter and highest in summer. This is primarily due to the median fraction of time spent outdoors, which was lowest in winter and second highest in summer.
4. Discussion
Our goal was to predict daily ambient PM2.5 exposure metrics for each DEPS participant in support of improving health effect estimation for future epidemiological analysis. Using EMI, we performed an individual-level exposure assessment in DEPS that accounts for daily variations in ambient PM2.5 exposures based on a mechanistic house-specific AER model linked to a mass-balance PM2.5 infiltration model, infiltration factors for nonresidential buildings and vehicles, and comprehensive time-location data from each participant. We demonstrated the ability to calibrate and evaluate EMI using the PM Panel Study with extensive exposure data (Breen et al., 2015), and then apply EMI for DEPS with limited exposure data. The impact of applying EMI for an epidemiological study to improve health effect estimation will depend not only on the accuracy of the exposure assessment, but also other factors such as the health study design and the true exposure distributions (Szpiro et al., 2011, 2013). In support of optimizing the health study design, we predicted multiple tiers of exposure metrics with different levels of complexity and uncertainty, which will be used in the epidemiological analysis to help determine the benefit of more sophisticated exposure metrics.
The EMI can be applied for epidemiological studies with different size and duration. Besides DEPS, we are applying EMI for two other panel studies in central North Carolina with <30 participants across two years, a larger panel study in Detroit, Michigan with >140 children across three years (Breen et al., 2014a), and a long-term study across 10 years with >2300 participants with cardiovascular disease (Mirowsky et al., 2017). For large cohorts, daily house-specific AER and Finf_home can be predicted from public property assessments and weather (Breen et al., 2014a), and time spent in different microenvironments can be determined from questionnaires (Kaufman et al., 2016) or global positioning system (GPS) data from smart phones (Breen et al., 2014b). To determine the home-specific building characteristics needed for the AER model, a list of participant home addresses can be automatically matched to those in city or county property assessment databases.
An important and unique aspect of our exposure assessment for DEPS is the comprehensive evaluation of the exposure model predictions. We evaluated all five tiers of modeled exposure metrics with detailed measurements from an exposure substudy (PM Panel Study) in the same geographic location and housing stock as DEPS (Breen et al. 2015), and evaluated Fpex and Erooftop with measurements from the actual DEPS participants. The uncertainty determined for the modeled exposure metrics can be used in our future epidemiological analysis to assess, and possibly reduce, any uncertainty in the health effect estimates (Spiegelman et al., 2010, Szpiro et al., 2013).
We can compare our individual-level microenvironment exposure model to those used in other panel studies. Two previous epidemiological studies predicted individual-level ambient PM2.5 exposures based on time spent in two microenvironments (indoors and outdoors) and used regression-based infiltration models for the homes (Kaufman et al., 2016; Koenig et al., 2005). Unlike the mechanistic AER models used by our mass balance infiltration model, these regression models are empirical and do not account for the driving forces of AER (stack and wind effects). Wu et al. predicted individual-level ambient PM2.5 exposures for a children’s health study based on time-spent indoors at home, school, and in-vehicle (Wu et al., 2005). For the homes, a mass balance infiltration model was linked to an empirical AER model, which did not consider the stack and wind effects. The in-vehicle concentration was set to a constant value based on measurements from other studies. The time spent in each microenvironment was based on demographics and measured population-level distributions from other studies. In our study, we predicted daily exposures based on a mass-balance infiltration model linked to a mechanistic house-specific AER model, infiltration factors for nonresidential buildings and vehicles, and daily participant-specific time spent in seven microenvironments.
A benefit of using EMI for epidemiology studies, such as DEPS, is that modeled exposure metrics are needed for ambient PM2.5 panel studies to account for the building-to-building and temporal variability of AER and indoor attenuation of ambient PM2.5. Since people spend most of their time indoors, the variability of indoor attenuation can be a substantial source of variability in exposures between individuals, including studies across regions with small spatial variations in outdoor PM2.5 concentrations. The significant variability of indoor attenuation was highlighted in the National Academy of Sciences Report “Health Risks of Indoor Exposure to Particulate Matter” (NAS, 2016). The report states that indoor attenuation of ambient PM2.5 for homes in United States and Canada can range from less than 10% to almost 100%, depending on the home characteristics, season, and location. Furthermore, when outdoor PM2.5 concentration is used as an exposure surrogate in panel studies, the estimated health effect parameter can be biased towards the null since it is the product of the toxicity (i.e., true health effect) and indoor attenuation of ambient PM2.5 (Sheppard et al., 2012). Accounting for indoor attenuation (i.e., Finf_home, Fpex) in epidemiological studies should yield a more accurate health effect estimate (Koenig et al., 2005). For respiratory and cardiac outcomes, a cohort study found ambient PM2.5 exposures had significant associations with larger effect estimates than outdoor PM2.5 concentrations, which were not significant (Ebelt et al., 2005).
One limitation of this study is that EMI requires detailed time-location data to determine time spent outdoors and within different indoor microenvironments. In DEPS, we used daily questionnaires, which can have uncertainty and substantial participant burden. To improve this data collection process for other studies, particularly large long-term health studies, we previously developed a model, called MicroTrac, that uses GPS data to automatically determine time spent outdoors and within different indoor locations (in-vehicle, home, work, school, other) (Breen et al. 2014b).
Another limitation of this study is the exposure metrics do not include non-ambient PM2.5. Wilson et al. showed the importance of separating ambient and non-ambient pollutant exposures (Wilson et al., 2000). These include: (1) the U.S. Environmental Protection Agency regulates only ambient pollutants, and (2) PM2.5 from ambient and non-ambient sources has different physical and chemical properties and temporal patterns, which can induce different health effects. A previous study found stronger associations between ambient PM2.5 and health effects than non-ambient PM2.5 (Ebelt et al., 2005). When we apply EMI for health studies, we plan to separately examine factors associated with non-ambient PM2.5 sources (e.g., environmental tobacco smoke) as categorical variables in the health models, which can remove potential uncertainties that can be introduced by including indoor sources in the exposure predictions.
Another potential limitation of this study is that our exposure model predicts ambient PM2.5 exposures based on two fixed-site PM2.5 monitors. In the PM Panel Study, we found no substantial difference between daily ambient PM2.5 exposures predicted from a fixed-site PM2.5 monitor and those predicted from PM2.5 monitors outside each participant’s home (Breen et al., 2015). This is consistent with data from various U.S. cities that show PM2.5 concentrations are spatially homogeneous within cities, and that local sources have only limited influence (U.S. EPA, 2009). Unlike PM2.5 components that can substantial local spatial variations from nearby sources (e.g., black carbon near major roads with high diesel traffic; U.S. EPA, 2009), this study examined total PM2.5 mass for participants living within 45 km (28 miles) of the PM2.5 monitors, and there were no major local sources. These results strengthen our confidence that using a fixed-site monitor is sufficient to estimate PM2.5 exposures in the same region of central NC.
5. Conclusions
This study demonstrates the ability of applying EMI to predict five tiers of individual-level exposure metrics for the homes and participants in an epidemiological study. To improve exposure assessments in DEPS, EMI accounts for (1) daily house-specific infiltration of ambient PM2.5 and (2) daily participant-specific time spent outdoors, in-vehicles, and indoors at home and other buildings. This capability can help provide more accurate exposure estimates for epidemiological studies, such as DEPS, in support of improving health risk estimation.
Supplementary Material
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
The authors thank John Langstaff, Anne Weaver, and Kathie Dionisio for their reviews and helpful suggestions. Although the manuscript was reviewed by the U.S. EPA and approved for publication, it may not necessarily reflect official Agency policy. Mention of trade names or commercial products does not constitute endorsement or recommendation for use.
Footnotes
Appendix A. Supplementary material
There is supplementary material for this article.
References
- Breen MS, Breen M, Williams RW, Schultz BD. 2010. Predicting residential air exchange rates from questionnaires and meteorology: model evaluation in central North Carolina. Environ Sci Technol 44:9349–9356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breen MS, Burke JM, Batterman SA, Vette AF, Godwin C, Croghan CW, et al. 2014a. Modeling spatial and temporal variability of residential air exchange rates for the Near-Road Exposures and Effects of Urban Air Pollutants Study (NEXUS). Int J Environ Res Public Health 11:11481–11504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breen MS, Long TC, Schultz BD, Crooks J, Breen M, Langstaff JE, et al. 2014b. GPS-based microenvironment tracker (MicroTrac) model to estimate time-location of individuals for air pollution exposure assessments: model evaluation in central North Carolina. J Exp Sci Environ Epidemiol 24:412–420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breen MS, Long TC, Schultz BD, Williams RW, Richmond-Bryant J, Breen M, et al. 2015. Air pollution exposure model for individuals (EMI) in health studies: evaluation for ambient PM in central North Carolina. Environ Sci Technol 49:14184–14194. [DOI] [PubMed] [Google Scholar]
- Burke JM, Zufall MJ, Ozkaynak H. 2001. A population exposure model for particulate matter: case study results for PM2.5 in Philadelphia, PA. J Expo Anal Environ Epidemiol 11:470–489. [DOI] [PubMed] [Google Scholar]
- Ebelt ST, Wilson WE, Brauer M. 2005. Exposure to ambient and nonambient components of particulate matter: a comparison of health effects. Epidemiology 16:396–405. [DOI] [PubMed] [Google Scholar]
- Kaufman JD, Adar SD, Barr RG, Budoff M, Burke GL, Curl CL, et al. 2016. Association between air pollution and coronary artery calcification within six metropolitan areas in the USA (the Multi-Ethnic Study of Atherosclerosis and Air Pollution): a longitudinal cohort study. Lancet 388:696–704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koenig JQ, Mar TF, Allen RW, Jansen K, Lumley T, Sullivan JH, et al. 2005. Pulmonary effects of indoor- and outdoor-generated particles in children with asthma. Environ Health Perspect 113:499–503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koutrakis P, Briggs LK. 1992. Source apportionment of indoor aerosols in Suffolk and Onondaga Counties, New York. Environ Sci Technol 26:521–527. [Google Scholar]
- Mirowsky JE, Devlin RB, Diaz-Sanchez D, Cascio W, Grabich SC, Haynes C, et al. 2017. A novel approach for measuring residential socioeconomic factors associated with cardiovascular and metabolic health. J Expo Sci Environ Epidemiol 27:281–289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- National Research Council (NRC). 2004. Research priorities for airborne particulate matter: I. Immediate priorities and a long-range research portfolio. Washington, D.C.: The National Academies Press; https://doi.org/10.17226/6131. [PubMed] [Google Scholar]
- National Research Council (NRC). 2012. Exposure science in the 21st century: a vision and a strategy. Washington, D.C.: The National Academies Press; https://doi.org/10.17226/13507. [PubMed] [Google Scholar]
- National Academies of Sciences, Engineering, and Medicine (NAS). 2016. Health risks of indoor exposure to particulate matter: workshop summary. Washington, D.C.: The National Academies Press; https://doi.org/10.17226/23531. [PubMed] [Google Scholar]
- National Academies of Sciences, Engineering, and Medicine (NAS). 2017. Using 21st century science to improve risk-related evaluations. Washington, D.C.: The National Academies Press; https://doi.org/10.17226/24635. [PubMed] [Google Scholar]
- Ott W, Klepeis N, Switzer P. 2008. Air change rates of motor vehicles and in-vehicle pollutant concentrations from secondhand smoke. J Expo Sci Environ Epidemiol 18:312–325. [DOI] [PubMed] [Google Scholar]
- Sarnat JA, Long CM, Koutrakis P, Coull BA, Schwartz J, Suh HH. 2002. Using sulfur as a tracer of outdoor fine particulate matter. Environ Sci Technol 36:5305–5314. [DOI] [PubMed] [Google Scholar]
- Sarnat JA, Sarnat SE, Flanders WD, Chang HH, Mulholland J, Baxter L, et al. 2013. Spatiotemporally resolved air exchange rate as a modifier of acute air pollution-related morbidity in Atlanta. J Expo Sci Environ Epidemiol 23:606–615. [DOI] [PubMed] [Google Scholar]
- Schneider A, Neas L, Herbst MC, Case M, Williams RW, Cascio W, et al. 2008. Endothelial dysfunction: associations with exposure to ambient fine particles in diabetic individuals. Environ Health Perspect 116:1666–1674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider A, Neas LM, Graff DW, Herbst MC, Cascio WE, Schmitt MT, et al. 2010. Association of cardiac and vascular changes with ambient PM in diabetic individuals. Part Fibre Toxicol 7:1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider A, Alexis NE, Diaz-Sanchez D, Neas LM, Harder S, Herbst MC, et al. 2011. Ambient PM exposure up-regulates the expression of costimulatory receptors on circulating monocytes in diabetic individuals. Environ Health Perspect 119:778–783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheppard L, Burnett RT, Szpiro AA, Kim SY, Jerrett M, Pope CA III, et al. 2012. Confounding and exposure measurement error in air pollution epidemiology. Air Qual Atmos Health 5:203–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiegelman D 2010. Approaches to uncertainty in exposure assessment in environmental epidemiology. Annu Rev Public Health 31:149–163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szpiro AA, Paciorek CJ, Sheppard L. 2011. Does more accurate exposure prediction necessarily improve health effect estimates? Epidemiology 22:680–685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szpiro AA, Paciorek CJ. 2013. Measurement error in two-stage analyses, with application to air pollution epidemiology. Environmetrics 24:501–517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- U.S. Environmental Protection Agency (U.S. EPA) 2009. Integrated science assessment (ISA) for particulate matter. EPA/600/R-08/139F. Washington, DC: U.S. Environmental Protection Agency. [PubMed] [Google Scholar]
- Wallace L, Williams R. 2005. Use of personal-indoor-outdoor sulfur concentrations to estimate the infiltration factor and outdoor exposure factor for individual homes and persons. Environ Sci Technol 39:1707–1714. [DOI] [PubMed] [Google Scholar]
- Williams R, Suggs J, Rodes C, Lawless P, Zweidinger R, Kwok R, et al. 2000. Comparison of PM2.5 and PM10 monitors. J Expo Anal Environ Epidemiol 10:497–505. [DOI] [PubMed] [Google Scholar]
- Williams R, Suggs J, Rea A, Leovic K, Vette A, Croghan C, et al. 2003. The Research Triangle Park particulate matter panel study: PM mass concentration relationships. Atmos Environ 37:5349–5363. [Google Scholar]
- Wilson W, Mage D, Grant L. 2000. Estimating separately personal exposure to ambient and nonambient particulate matter for epidemiology and risk assessment: why and how. J Air Waste Manag Assoc 50:1167–1183. [DOI] [PubMed] [Google Scholar]
- Williams R, Rea A, Vette A, Croghan C, Whitaker D, Stevens C, et al. 2009. The design and field implementation of the Detroit Exposure and Aerosol Research Study. J Expo Sci Environ Epidemiol 19:643–659. [DOI] [PubMed] [Google Scholar]
- Wu J, Lurmann F, Winer A, Lu R, Turco R, Funk T. 2005. Development of an individual exposure model for application to the Southern California children’s health study. Atmos Environ 39:259–273. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.