Abstract
BACKGROUND
Sickle cell pain crisis continues to challenge patients and health professionals in places like Saudi Arabia, where the disease is common, and use of narcotic analgesics is strictly controlled. We sought to find the most effective and appropriate pain control regime for adult sickle cell pain crisis in Saudi Arabian patients.
PATIENTS AND METHODS
Adult sickle cell disease patients in crisis, treated initially in the emergency room of a private health center, usually undergo further treatment with different pain control regimes in an observation ward. We compared the adequacy of pain conrol during the three recent years (2000–2002) with those of the preceding three (1995–1997).
RESULTS
Treatment with regular opiates supplemented with oral analgesics during the second three-year period produced better results than “on demand” regimes. The former regimen enabled about 83% of patients from the second three-year period to be discharged home within two days compared with 71% during the first three-year period (P<0.05). A minority of patients needed more time for pain resolution. Patient response to oral analgesics was variable and females appeared to fare better than males.
DISCUSSION
Greater empathy and individualized treatment are required for sickle cell pain crisis patients because of their variable clinical presentation, response to medications, and the regularity of pain in their lives. The observed gender differences in pain response require further study.
Sickle cell disease (SCD) comprises several entities that are characterized by chronic haemolytic anaemia and acute episodic severe pain in different and sometimes multiple parts of the body, especially in the extremities, chest, back and abdomen. This “painful crisis” is the hallmark clinical feature in most patients.1,2 Its pathogenesis is complex, but is believed to be caused by small vessel vasocclusion triggered by the sickling and polymerisation on deoxygenation of haemoglobin S-rich red cells.
Episodes occur throughout life and frequently require treatment. The agony experienced by patients may be caused by present pain, recollection of past experiences or a fear of future pain because of its inevitability. Thus pain is always near these patients and managing it is challenging because pain is subjective and difficult for sufferers to describe accurately. This is compounded by poor knowledge and understanding of SCD pain among healthcare professionals,1,3 who frequently fear that relieving it completely, especially with narcotics, may induce addiction in patients.1,4–6
Saudi Arabia follows a very strict controlled drugs policy; offenders of drugs considered as “recreational” in some countries are punished severely and drug traffickers are liable to judicial execution. Consequently, physicians practicing in the country, not wanting to be associated with drug-related crime, are often reluctant to use narcotic drugs for a recurring condition like SCD pain crisis, leaving many patients under-treated for pain.
SCD is common in eastern Saudi Arabia.8, 9 We manage over 500 crisis events annually among our adult health care population of about 40,000. We previously described our initial impression with pain management in these patients.10 Our approach has been modified over the years, in an effort to help these patients. In this article, we describe our experience with different acute pain management modalities in adult SCD.
Patients and Methods
The Stabilization Unit (SU) is an observation ward attached to the Emergency Room (ER) of the Saudi Aramco Al-Hasa Health Center (SA-AHHC), where patients can be treated for 36 to 47 hours.10 SCD pain crisis comprises 20% to 50% of SU cases and is the most common diagnostic entity among patients admitted. The SU had 3 beds up to 1998 and the maximum length of stay (LOS) was 36 hours; bed capacity was increased to 6 and the LOS extended to 47 hours in July 1999. Nursing staff was provided from the ER.
ER physicians initially treated adult SCD patients presenting to SA-AHHC in painful crisis with intravenous rehydration, oxygen, and parenteral analgesics such as diclofenac, ketoralac, or opiates, depending on pain severity. Those with persisting pain were referred to Internal Medicine for further management, which consisted mostly of admission to the SU. Patient data for this study were retrieved from an Access database in which all SU data are stored. All patients were Saudis, drawn from our catchment area. There was no major demographic change in the study population during the study period. We compared patients treated during the three post-renovation years (2000–2002) with those of the preceding three (1995–1997). We excluded patients in the incomplete years (1998 and 1999) to eliminate seasonal influence on SCD pain crisis and to make the two groups comparable.
Pain severity and relief were assessed against patient reports, using either our custom designed color-coded analgesia chart (Figure 1), or the pain faces of Wong-Baker;11 severity was scored numerically from 0–5. Patients were admitted with pain scores of 3–5/5 and discharged when they had maintained a drop in score of ≥2 for at least 4 hours. They were re-evaluated every 6 hours or more often if indicated, and analgesia was adjusted until pain, as reported by patients, was controlled, and confirmed by nurse assessment, using pain score charts. Oxygen and intravenous re-hydration were continued for the first 24 hours, and then reviewed. Pain control was initially attempted with various regimes: intramuscular (IM) diclofenac 75mg every 8 hours; intravenous (IV) ketorolac 30–60 mg every 6 hours, IV pethidine 50–100mg every 4 hours as necessary, IV morphine 5–15mg every 4 hours as necessary (i.e., on demand).
Figure 1.
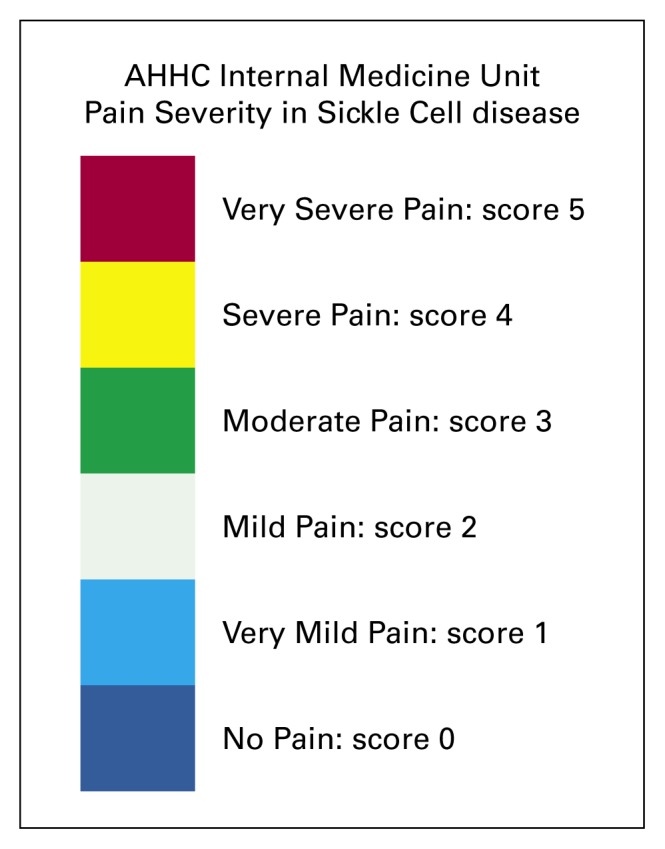
Custom designed color-coded analgesia chart for assessing pain severity and relief.
Our initial pain management review indicated sub-optimal pain control and an inadequate length of stay in the SU.10 Consequently, in 1999, a different strategy of regular round-the-clock opiate for the first 24 hours, supplemented by oral analgesics was applied, and LOS increased to 47 hours. The standard treatment then became IV morphine 5–15 mg regularly every four hours for the first 24 hours, combined with paracetamol 1 g or a non-steroidal anti-inflammatory drug (NSAID) orally. This was reviewed after 24 hours, and adjusted with or without further opiate according to the patients’ pain status. Adjunct intravenous promethazine 12.5–25mg every 8 hours, was given to prevent nausea and vomiting. This approach is similar to the Guidelines for Pain Management in Sickle Cell Disease published by the American Pain Society.12
Our ultimate indicator for adequate pain relief was the proportion of patients discharged home or transferred to hospital. For calculation of means, each patient visit was treated as a different attendance. The null hypothesis was applied to the difference between means and percentages to determine statistical significance by testing the standard errors of difference.
Results
Of the patients admitted to the SU during the two periods (Table 1), there were 305 crisis events in the first group (1995–1997) and 919 in the second (2000–2002), with a near equal sex distribution (Table 2). Patient ages ranged from 14 to 90 years; many attended several times each year in both groups. Patients expressed more satisfaction with the second-period pain management regime. Patient numbers tripled from the first to the second period (P<0.05) even with LOS extended to 47 hours. Twenty-nine patients (13 males and 16 females), whose pain crises usually lasted more than a few days, required 70 hospital transfers. These “repeaters”, who were known to have prolonged crises and should have been admitted directly to hospital, rather than the SU, were excluded from the final analysis. Seventy-one percent of patients in the first group and 83% in the second were discharged home (P<0.05). Seven SCD patients in the second group, and none in the first preferred the SU to the hospital and discharged themselves rather than be transferred to hospital, after a maximum SU stay.
Table 1.
Stabilization unit (SU) and hospital admissions during study periods.
| 1995 | 1996 | 1997 | Total (1995–1997) | 2000 | 2001 | 2002 | Total (2000–2002) | |
|---|---|---|---|---|---|---|---|---|
| SU admissions | 453 | 476 | 497 | 1426 | 806 | 638 | 740 | 2184 |
| SCD | 61 | 113 | 131 | 305 | 275 | 297 | 347 | 919 |
| Male | 32 | 69 | 50 | 151 | 128 | 155 | 168 | 451 |
| Female | 29 | 44 | 81 | 154 | 147 | 142 | 179 | 468 |
| SCD patients as % of SU admissions | 13.5 | 24 | 26 | 21.4 | 34 | 47 | 47 | 42 |
| SCD patients admitted to hospital | 17 | 33 | 39 | 89 | 56 | 75 | 88 | 219 |
| Male | 8 | 22 | 23 | 53 | 25 | 50 | 52 | 127 |
| Female | 9 | 11 | 16 | 36 | 31 | 25 | 32 | 88 |
| SCD as % of SU transfers to hospital | 78 | 36 | 76 | 50.5 | 65 | 74 | ||
| % SCD to hospital | 27.9 | 29.2 | 29.8 | 29.2 | 20 | 25 | 25.4 | 23.8 |
Table 2.
Disposition and characteristics of patients admitted to stabilization unit (SU) and hospital.
| Patient groups | ||
|---|---|---|
| 1995–1997 | 2000–2002 | |
|
| ||
| Total number of patients in SU | 1426 | 2184 |
|
| ||
| SCD repeaters | – | 70 |
|
| ||
| Adjusted numbers | 1426 | 2114* |
|
| ||
| SCD patients (as % of all SU patients) | 305 (21.4) | 849* (40.2) |
|
| ||
| All patients (mean age in years, range) | 26.7 (14–80) | 25.1 (14–90) |
|
| ||
| Male (%) | 152 (49.8) | 422 (49.7) |
|
| ||
| Male (mean age in years, range) | 26.9 (14–80) | 22.2 (14–90) |
|
| ||
| Female (%) | 153 (50.2) | 427 (50.3) |
|
| ||
| Female (mean age in years, range) | 26.4 (14–80) | 27.9 (14–82) |
|
| ||
| SCD patients admitted to hospital (%) | 89 (29.2) | 145 (17.1) |
|
| ||
| Male (%) | 53 (59.6) | 98 (67.6) |
|
| ||
| Female (%) | 36 (40.4) | 47 (32.4) |
|
| ||
| Mean stay in SU (hours) | 26.7 | 36.02 |
Excludes “repeaters”, who were known to have prolonged crises and should have been admitted directly to hospital, rather than the SU
More males than females required transfer to hospital in both groups of patients. Thus, while patients were almost equally distributed between the sexes in the two groups, males constituted approximately 60% and 68%, respectively, of hospital transfers in both periods, a highly statistically significant difference (P<0.0010). They were also over-represented among those requiring multiple SU admissions of ≥4 per year (Table 3).
Table 3.
Patient distribution by number of admissions to stabilization unit and sex.
| Number of admissions (events) | Number of patients | Males | Females |
|---|---|---|---|
| 1 | 60 | 28 | 32 |
| 2 | 38 | 14 | 24 |
| 3 | 16 | 8 | 8 |
| 4 | 10 | 6 | 4 |
| 5 | 9 | 5 | 4 |
| 6 | 5 | 3 | 2 |
| 7 | 2 | 2 | 0 |
| 8 | 3 | 3 | 0 |
| 9–10 | 1 | 0 | 1 |
| ≥4 | 30 | 19 | 11 |
Regular morphine as given during the second period was more effective than on-demand administration and produced the best and most predictable result of all the modalities. Patients reported no major side effects, although a few declined it occasionally because of drowsiness, sometimes worsened by promethazine. Morphine-related respiratory depression was not observed. There were no cases of gastrointestinal bleeding. Five cases of acute chest syndrome occurred in the first group and 10 in the second; those affected were transferred to the Intensive Care Unit and all survived.
In a survey of 100 patients, the most preferred oral non-narcotic analgesics were ibuprofen and naproxen (Table 4). Pethidine pain relief, which lasted 2–3 hours, was too short-lived to be practically useful, and ketorolac rarely proved adequate for severe pain. Intramuscular diclofenac was too painful for routine use, and was inferior to opiates in severe pain; an intravenous preparation was unavailable. Diclofenac tablets were as efficacious as other NSAIDs, but some patients requested it specifically by name or showed the packaging. Patient response to NSAIDs was variable; an agent described as “very good” by one patient, might be “useless” to another. Mefenamic acid capsules were more effective in some patients than the tablets at equipotent dosages. Opiate analogues like tramadol, and Revacod (paracetamol with codeine), were occasionally useful in patients allergic to NSAIDs.
Table 4.
Patient relative preference for oral non-narcotic analgesics.
| Drug | Preference (%) |
|---|---|
| Ibuprofen | 31 |
| Naproxen | 25 |
| Mefenamic acid (Ponstan) | 17 |
| Diclofenac | 17 |
| Paracetamol | 10 |
| Total | 100 |
Discussion
Although acute pain control is only one aspect of the overall management of SCD, it is nevertheless important since pain is intrusive in the life of these patients,1,2,14 and is usually the presenting problem to medical facilities. Other measures like hydration, blood and exchange blood transfusion, and anti-sickling agents like hydroxyurea all have a role as do psychological counseling and social/family support, but assessment of these modalities of management are outside the scope of the current study.
The percentage of patients discharged home increased from 71% in the first group to 83% in the second, a statistically significant difference (P<0.05). The lower patient discharge rate during the “on demand” analgesia period may have been partly caused by delayed drug administration during busy times in the ER, when SU nurses who were part of the ER nursing pool shifted there to support the service.
Although the effectiveness of properly administered intermittent analgesia has been demonstrated, 13,15 it may be hindered by tradition, prejudice, and staff misconceptions.3–5,16 On-demand analgesia has the further disadvantage of responding to pain post-occurrence, rather than anticipating it to break its cycle. Patient-controlled analgesia leads to more patient satisfaction and a reduction in LOS,13,17 and should probably be used for severe pain when available.
Patient numbers tripled, instead of increasing by about a third as would be expected, statistically, with a doubling of bed capacity, and an LOS extension to 47 hours. This statistically significant difference (P<0.05) was attributed to increased patient satisfaction, since there was no change in the base population demographics. The seven patients in the second group who discharged themselves, rather than go to hospital after a maximum SU stay, could also be seen as additional evidence of this popularity.
These results demonstrate that over 80% of acute SCD pain crisis can be controlled within two days, with good pain management. This has important economic implications for school children, employees and housewives in reduced absences from school, work and home, apart from the cost of hospitalization and medications.
The few patients with prolonged pain illustrate the variability in clinical presentation and course of SCD crisis. When identifiable, such patients should be admitted directly to hospital and not treated in short stay units. In one study, 5% of SCD patients perceived as “difficult” provoked a negative attitude from staff, which was eventually transferred to all SCD patients.1 Our “repeater” patients, who demonstrated a pattern of recurrent hospital transfer, may be equivalent to this 5% minority, although numerically they constituted 7.6% of cases.
The inferior patient response to pethidine encouraged morphine usage as the standard opiate, thus familiarizing all staff with it and probably reducing adverse effects.7 Pethidine produces more addiction than and offers no advantages over morphine and can be toxic in patients with renal compromise.18, 19 Its altered pharmacokinetics in SCD crisis may also have limited its benefits.19,20 Ketorolac, usually initiated by ER physicians because of their experience with its efficacy in trauma patients, proved inadequate for severe pain. Our observation is similar to the finding that its use does not reduce the need for narcotics in SCD pain,21 despite suggestions to the contrary;22 it is also the most expensive analgesic. Intramuscular agents, especially if painful themselves, like diclofenac, should not be used for recurrent conditions like SCD pain crisis since they may cause additional problems.23
The prior analgesic experience of patients helped physicians select appropriate agents. The observed difference in efficacy between mefenamic acid tablets and capsules may be related to the drug bioavailability, which may be better with the capsule granules than with the compressed tablet. This possible explanation requires investigation.
Some patients moaned or cried out during physician rounds to indicate inadequate pain relief, often against a background of perceived lack of sympathy from nurses. This behaviour, which may be a pain-coping strategy,1,4,14 should lead to a review of patient treatment. Allowing for the compounding effects of better hydration, the correspondence of serial changes in laboratory indices like hemoglobin, and lactic dehydrogenase with patient complaints of continuing pain, confirmed ongoing sickling; while their stability in similar settings may rarely indicate drug-seeking behaviour.
The finding of more males than females requiring both multiple SU admissions and hospital transfers was unexpected. Further details of this observation are given elsewhere.24
In conclusion, the variability in clinical presentation and course, patient response to medications, and the observed gender differences in acute SCD crisis, all demonstrate that individualised care is necessary in treating these patients. Everyone should recognize the need for adequate pain medication. Empathy, rather than suspicion, misconceptions or hostility, should be the watchword.
Acknowledgements
We are grateful to our patients and colleagues whose cooperation helped to make this study possible and to V. Moore who provided valuable secretarial assistance. Both authors were employed by the Saudi Aramco Medical Services Organisation, (SAMSO) when the study was conducted and the paper written, and acknowledge the use of SAMSO facilities for both the data and study.
Footnotes
No competing or financial interests involved.
References
- 1.Ballas SK. Treatment of pain in adults with sickle cell disease. Am J Hematol. 1990;34:49–54. doi: 10.1002/ajh.2830340111. [DOI] [PubMed] [Google Scholar]
- 2.Yaster M, Kost-Byerly S, Maxwell LG. The management of pain in sickle cell disease. Pediatr Clin North Am. 2000;47(3):699–710. doi: 10.1016/s0031-3955(05)70233-9. [DOI] [PubMed] [Google Scholar]
- 3.Murray N, May A. Painful crises in sickle cell disease—patients’ perspectives. BMJ. 1988;297:452–4. doi: 10.1136/bmj.297.6646.452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shapiro BS, Benjamin LJ, Payne R, et al. Sickle cell-related pain: perceptions of medical practitioners. J Pain Symptom Manage. 1997;14:168–74. doi: 10.1016/S0885-3924(97)00019-5. [DOI] [PubMed] [Google Scholar]
- 5.Waldrop RD, Mandry C. Health professional perceptions of opiod dependence among patients with pain. Am J Emerg Med. 1995;13:529–31. doi: 10.1016/0735-6757(95)90163-9. [DOI] [PubMed] [Google Scholar]
- 6.Yale SH, Nagib N, Guthrie T. Approach to the vaso-occlusive crisis in adults with sickle cell disease. Am Fam Physician. 2000;61(5):1349–56. 1363–4. [PubMed] [Google Scholar]
- 7.McQuay H, Moore A, Justins D. Treating acute pain in hospital. BMJ. 1997;314(7093):1531–5. doi: 10.1136/bmj.314.7093.1531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.El-Hazmi MAF. Haemoglobinopathies, thalassaemias and enzymopathies in Saudi Arabia. Saudi Med J. 1992;13(6):488–99. doi: 10.1159/000205861. [DOI] [PubMed] [Google Scholar]
- 9.Serjeant GR. Sickle-cell disease. Lancet. 1997;350:725–30. doi: 10.1016/S0140-6736(97)07330-3. [DOI] [PubMed] [Google Scholar]
- 10.Udezue E. A five-year experience of a short stay observation unit in Saudi Arabia. Ann Saudi Med. 2003;23(1–2):72–75. doi: 10.5144/0256-4947.2003.72. [DOI] [PubMed] [Google Scholar]
- 11.Wong DL, Baker CM. Pain in children: comparison of assessment scales. Pediatr Nurs. 1988;14:9–17. [PubMed] [Google Scholar]
- 12.American Pain Society. Guideline for the Management of Acute and Chronic Pain in Sickle Cell Disease. Glenview, IL: 1999. [Google Scholar]
- 13.Gonzalez ER, Bahal N, Hansen LA, et al. Intermittent injection vs patient-controlled analgesia for sickle cell crisis pain: comparison in patients in emergency departments. Arch Intern Med. 1991;151:1373–8. [PubMed] [Google Scholar]
- 14.Black J, Laws S. Living with sickle cell disease: an inquiry into the need for health and social service provision for sickle cell suffers in Newham. London: Sickle Cell Society; 1986. [Google Scholar]
- 15.Gould TH, Crosby DL, Harmer M, et al. Policy for controlling pain after surgery: effect of sequential changes in management. BMJ. 1992;305:1187–93. doi: 10.1136/bmj.305.6863.1187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Maxwell K, Streetly A, Bevan D. Experiences of hospital care and treatment seeking for pain from sickle cell disease: qualitative study. BMJ. 1999;318(7198):1585–90. doi: 10.1136/bmj.318.7198.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ballantyne JC, Carr DB, Chalmers TC, et al. Postoperative patient-controlled analgesia: meta analyses of initial randomized control trials. J Clin Anesth. 1993;5:182–93. doi: 10.1016/0952-8180(93)90013-5. [DOI] [PubMed] [Google Scholar]
- 18.Szeto HH, Inturrisi CE, Houde R, et al. Accumulation of normeperidine, an active metabolite of meperidine, in patients with renal failure or cancer. Ann Intern Med. 1977;86:738–41. doi: 10.7326/0003-4819-86-6-738. [DOI] [PubMed] [Google Scholar]
- 19.Abbuhl S, Jacobson S, Murphy JG, et al. Serum concentrations of meperidine in patients with sickle cell crisis. Ann Emerg Med. 1986;15:433–8. doi: 10.1016/s0196-0644(86)80183-4. [DOI] [PubMed] [Google Scholar]
- 20.Williams WD, Chung H. Protein binding diminishes the efficacy of meperidine. Blood. 1985;66(Suppl 1):67a–70a. [Google Scholar]
- 21.Wright SW, Norris RL, Mitchell TR. Ketorolac for sickle cell vaso-occlusive crisis pain in the emergency department: lack of a narcotic-sparing effect. Ann Emerg Med. 1992;8:925–8. doi: 10.1016/s0196-0644(05)82929-4. [DOI] [PubMed] [Google Scholar]
- 22.Gillis JC, Brogden RN. Ketorolac. A reappraisal of its pharmacodynamic and pharmacokinetic properties and therapeutic use in pain management. Drugs. 1997;53(1):139–88. doi: 10.2165/00003495-199753010-00012. [DOI] [PubMed] [Google Scholar]
- 23.Dorwart BB, Gabuzda TG. Symmetric myositis and fascitis: a complication of sickle cell anemia during vasocclusion. J Rheumatol. 1985;12:590–5. [PubMed] [Google Scholar]
- 24.Udezue EO, Girshab AM. Differences between males and females in adult sickle cell pain crisis in eastern Saudi Arabia. Ann Saudi Med. 2004;24(3):180–183. doi: 10.5144/0256-4947.2004.179. [DOI] [PMC free article] [PubMed] [Google Scholar]


